Inhibitors of Lipoxygenase and Cyclooxygenase-2 Attenuate Trimethyltin-Induced Neurotoxicity through Regulating Oxidative Stress and Pro-Inflammatory Cytokines in Human Neuroblastoma SH-SY5Y Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents, Chemicals, and Antibodies
2.2. Cell Culture and TMT Treatment
2.3. Photomicrography of Cultured Cells
2.4. Lactate Dehydrogenase (LDH) Activity Measurement
2.5. MTT Assay
2.6. Western Blot Analysis
2.7. Enzyme-Linked Immunoassay (ELISA)
2.8. Protein Assay
2.9. TEM Observations
2.10. Statistical Analyses
3. Results
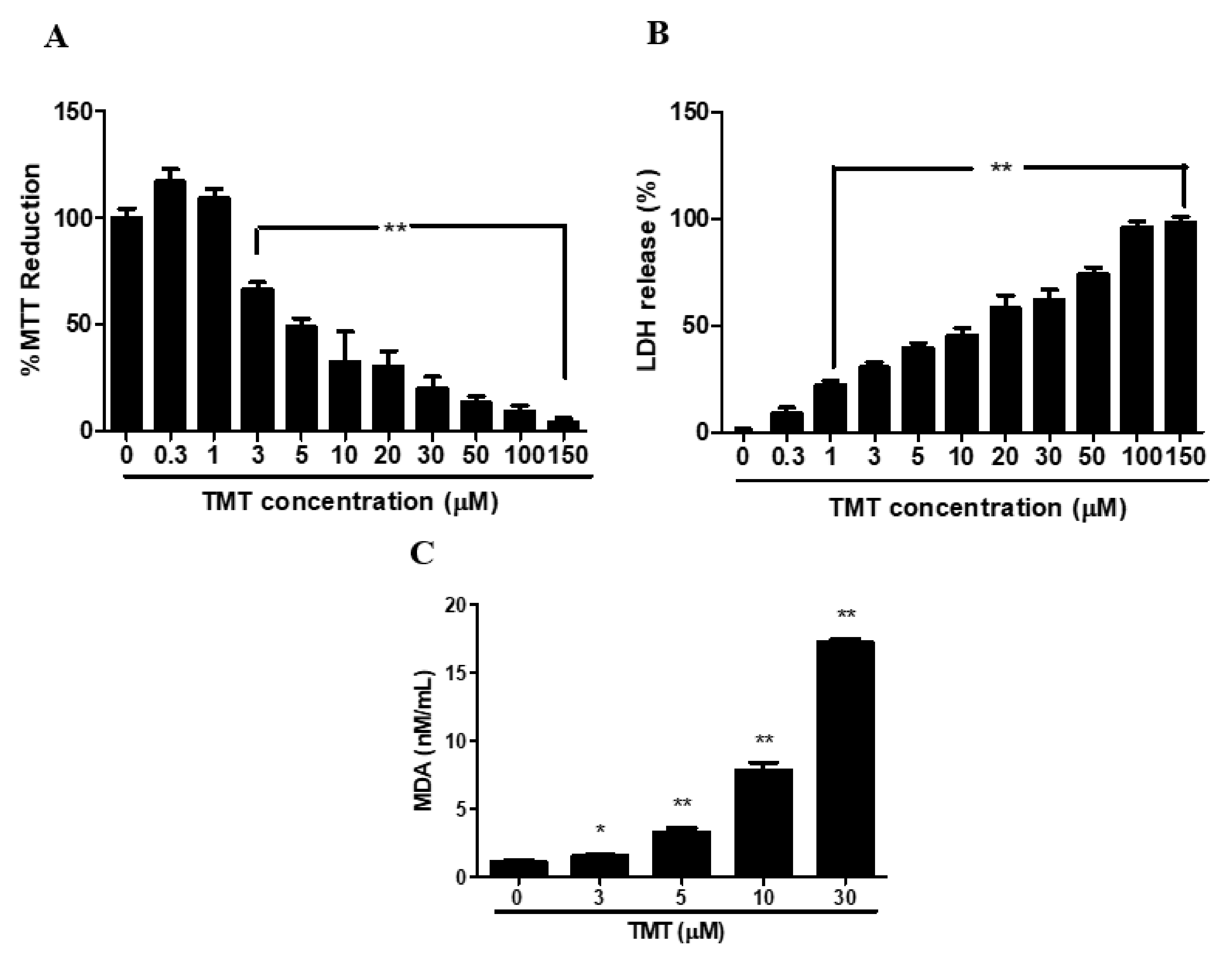
3.1. Dose-Toxicity of Trimethyltin (TMT)
3.2. Photomicrographs of Representative Cell Morphologies
3.3. Melatonin, N-acetylcysteine (NAC), α-tocopherol, and Allopurinol Protected against TMT-Induced Neurotoxicity in SH-SY5Y Cells
3.4. Deferoxamine Reversed Altered Expression of Cleaved Caspase-3, Bax, Bcl-2, and Bcl-xL Induced by TMT
3.5. NAC Improved TMT-Exposed SH-SY5Y Cells Observed via TEM
3.6. Esculetin, Meloxicam, Celecoxib, and Phenidone Attenuated TMT-Induced Neurotoxicity in SH-SY5Y Cells
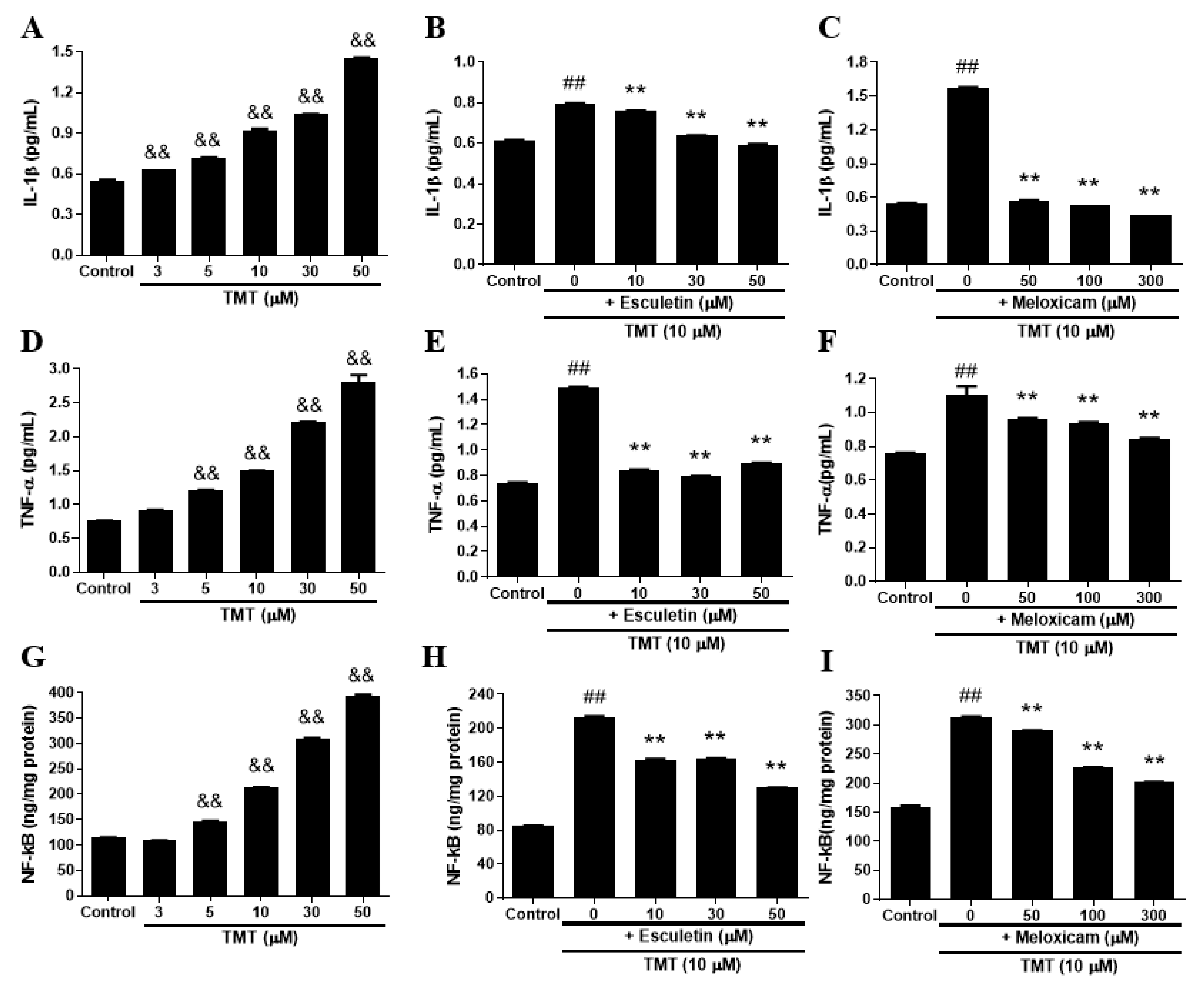
3.7. Esculetin and Meloxicam Inhibited the Elevation of Pro-inflammatory Cytokines IL-1β, TNF-α, and NF-kB Induced by TMT in SH-SY5Y Cells
3.8. Esculetin and Meloxicam Ameliorated Catalase (CAT) and Glutathione Peroxidase-1 (GPx-1) Activity Reduction in SH-SY5Y Cells
3.9. Dextrorphan and CNQX Attenuated TMT-Induced Neurotoxicity in SH-SY5Y Cells
3.10. Calcium and Iron Chelators and A Voltage-gated L-type Calcium Channel (VGCC) Blocker Attenuated TMT Toxicity in SH-SY5Y Cells
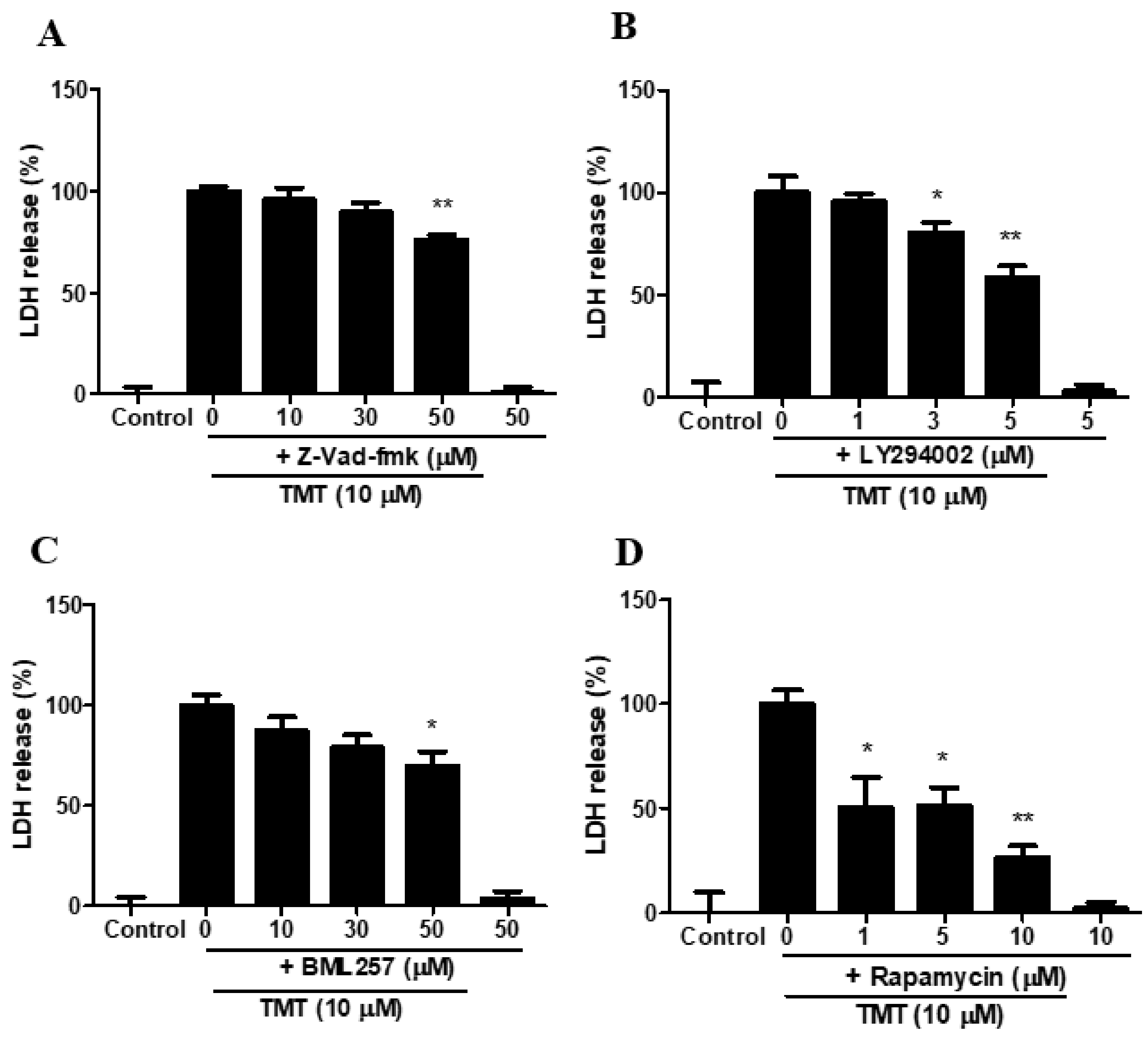
3.11. Inhibitors of Pan-Caspase, PI3K, Akt, and mTOR Attenuated TMT-Induced Toxicity in SH-SY5Y Cells
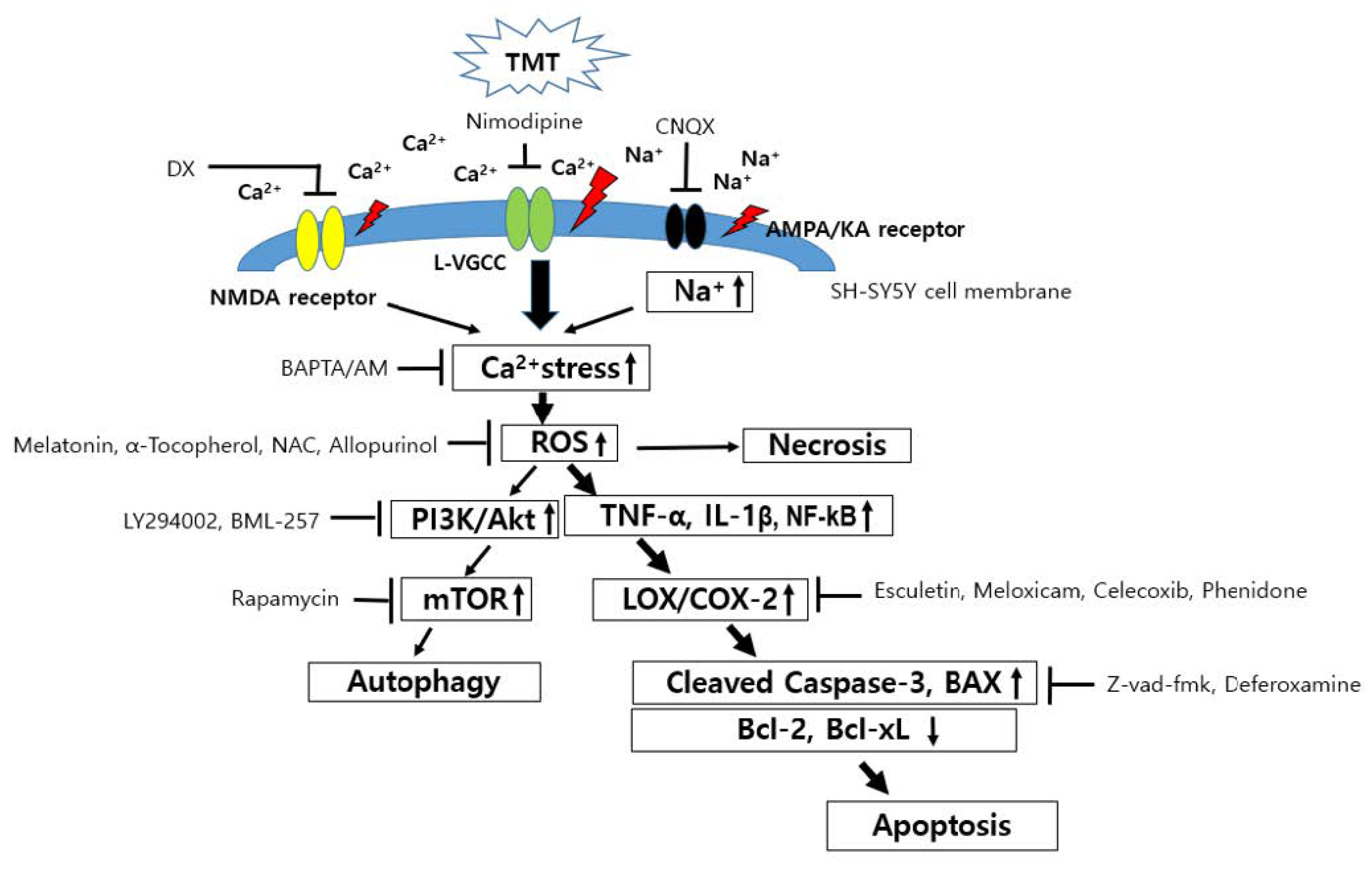
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Fent, K. Ecotoxicology of organotin compounds. Crit. Rev. Toxicol. 1996, 26, 1–117. [Google Scholar] [CrossRef]
- Liu, M.; Pi, H.; Xi, Y.; Wang, L.; Tian, L.; Chen, M.; Xie, J.; Deng, P.; Zhang, T.; Zhou, C.; et al. KIF5A-dependent axonal transport deficiency disrupts autophagic flux in trimethyltin chloride-induced neurotoxicity. Autophagy 2021, 17, 903–924. [Google Scholar] [CrossRef]
- Appel, K.E. Organotin compounds: Toxicokinetic aspects. Drug Metab. Rev. 2004, 36, 763–786. [Google Scholar] [CrossRef]
- Mignini, F.; Nasuti, C.; Artico, M.; Giovannetti, F.; Fabrizi, C.; Fumagalli, L.; Lannetti, G.; Pompili, E. Effects of trimethyltin on hippocampal dopaminergic markers and cognitive behavior. Int. J. Immunopathol. Pharmacol. 2012, 25, 1107–1119. [Google Scholar] [CrossRef]
- Ali, S.F.; Slikker, W., Jr.; Newport, G.D.; Goad, P.T. Cholinergic and dopaminergic alterations in the mouse central nervous system following acute trimethyltin exposure. Acta. Pharmacol. Toxicol. 1986, 59, 179–188. [Google Scholar] [CrossRef] [PubMed]
- Dehaven, D.L.; Krigman, M.R.; Mailman, R.B. Temporal changes in dopaminergic and serotonergic function caused by administration of trimethyltin to adult rat. Neurobehav. Toxicol. Teratol. 1986, 8, 475–479. [Google Scholar]
- Blesa, J.; Trigo-Damas, I.; Quiroga-Varela, A.; Jackson-Lewis, V.R. Oxidative stress and Parkinson’s disease. Front. Neuroanat. 2015, 9, 91. [Google Scholar] [CrossRef]
- Pham, H.T.N.; Phan, S.V.; Tran, H.N.; Phi, X.T.; Le, X.T.; Nguyen, K.M.; Fujiwara, H.; Yoneyama, M.; Ogita, K.; Yamaguchi, T.; et al. Bacopa monnieri (L.) ameliorates cognitive deficits caused in trimethyltin-induced neurotoxicity model mice. Biol. Pharm. Bull. 2019, 42, 1384–1393. [Google Scholar] [CrossRef] [PubMed]
- Fechter, L.D.; Liu, Y. Elevation of intracellular calcium levels in spiral ganglion cells by trimethyltin. Hear. Res. 1995, 91, 101–109. [Google Scholar] [CrossRef]
- Florea, A.M.; Splettstoesser, F.; Dopp, E.; Rettenmeier, A.W.; Büsselberg, D. Modulation of intracellular calcium homeostasis by trimethyltin chloride in human tumor cells: Neuroblastoma SY5Y and cervix adenocarcinoma HeLa S3. Toxicology 2005, 216, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Fan, Y.; Kim, D.; Zhong, T.; Yi, P.; Fan, C.; Wang, A.; Yang, X.; Lee, S.; Ren, X.; et al. Neuroprotective effect of catechins derivatives isolated from Anhua dark tea on NMDA-induced excitotoxicity in SH-SY5Y cells. Fitoterpia 2019, 137, 104240. [Google Scholar] [CrossRef]
- Sun, Z.W.; Zhang, L.; Zhu, S.J.; Chen, W.C.; Mei, B. Excitotoxicity effects of glutamate on human neuroblastoma SH-SY5Y cells via oxidative damage. Neurosci. Bull. 2010, 26, 8–16. [Google Scholar] [CrossRef]
- Than, A.; Tan, Y.; Ong, W.Y.; Farooqui, A.A.; Chen, P. Kainate receptors mediate regulated exocytosis of secretory phospholipase A2 in SH-SY5Y neuroblastoma cells. Neurosignals 2012, 20, 72–85. [Google Scholar] [CrossRef]
- Bruccoleri, A.; Pennypacker, K.R.; Harry, G.J. Effect of dexamethasone on elevated cytokine mRNA levels in chemical-induced hippocampal injury. J. Neurosci. Res. 1999, 57, 916–926. [Google Scholar] [CrossRef]
- Ceccariglia, S.; Alvino, A.; Del Fà, A.; Parolini, O.; Michetti, F.; Gangitano, C. Autophagy is activated in vivo during trimethyltin-induced apoptotic neurodegeneration: A study in the rat hippocampus. Int. J. Mol. Sci. 2019, 21, 175. [Google Scholar] [CrossRef] [PubMed]
- Fabrizi, C.; Somma, F.; Pompili, E.; Biagioni, F.; Lenzi, P.; Fornai, F.; Fumagalli, L. Role of autophagy inhibitors and inducers in modulating the toxicity of trimethyltin in neuronal cell cultures. J. Neural. Transm. 2012, 119, 1295–1305. [Google Scholar] [CrossRef]
- Gunasekar, P.; Li, L.; Prabhakaran, K.; Eybl, V.; Borowitz, J.L.; Isom, G.E. Mechanisms of the apoptotic and necrotic actions of trimethyltin in cerebellar granule cells. Toxicol. Sci. 2001, 64, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Shin, E.J.; Nam, Y.; Tu, T.T.; Lim, Y.K.; Wie, M.B.; Kim, D.J.; Jeong, J.H.; Kim, H.C. Protein kinase Cδ mediates trimethyltin-induced neurotoxicity in mice in vivo via inhibition of glutathione defense mechanism. Arch. Toxicol. 2015, 90, 937–953. [Google Scholar] [CrossRef] [PubMed]
- Qing, Y.; Liang, Y.; Du, Q.; Fan, P.; Xu, H.; Xu, Y.; Shi, N. Apoptosis induced by trimethyltin chloride in human neuroblastoma cells SY5Y is regulated by a balance and cross-talk between NF-kB and MAPKs signaling pathways. Arch. Toxicol. 2013, 87, 1273–1285. [Google Scholar] [CrossRef]
- Xi, Y.; Liu, M.; Xu, S.; Hong, H.; Chen, M.; Tian, L.; Xie, J.; Deng, P.; Zhou, C.; Zhang, L.; et al. Inhibition of SERPINA3N-dependent neuroinflammation is essential for melatonin to ameliorate trimethyltin chloride-induced neurotoxicity. J. Pineal Res. 2019, 67, e12596. [Google Scholar] [CrossRef]
- Shin, E.J.; Suh, S.K.; Lim, Y.K.; Jhoo, W.K.; Hjelle, O.P.; Otterson, O.P.; Shin, C.Y.; Ko, K.H.; Kim, W.K.; Kim, D.S.; et al. Ascorbate attenuates trimethyltin-induced oxidative burden and neuronal degeneration in the rat hippocampus by maintaining glutathione homeostasis. Neuroscience 2005, 133, 715–727. [Google Scholar] [CrossRef] [PubMed]
- Bouldin, T.W.; Goines, N.D.; Bagnell, C.R.; Krigman, M.R. Pathogenesis of trimethyltin neuronal toxicity. Ultrastructural and cytochemical observations. Am. J. Pathol. 1981, 104, 237–249. [Google Scholar]
- Chang, L.W. Hippocampal lesions induced by trimethyltin in the neonatal rat brain. Neurotoxicology 1984, 5, 205–215. [Google Scholar] [PubMed]
- Shirakawa, T.; Nakano, K.; Hachiya, N.S.; Kato, N.; Kaneko, K. Temporospatial patterns of COX-2 expression and pyramidal cell degeneration in the rat hippocampus after trimethyltin administration. Neurosci. Res. 2007, 59, 117–123. [Google Scholar] [CrossRef]
- Shirakawa, T.; Nakano, K.; Hachiya, N.S.; Kato, N.; Kaneko, K. The involvement of P2X1 receptor in pyramidal cell degeneration in the rat hippocampus after trimethyltin administration. Neurosci. Res. 2011, 71, 396–404. [Google Scholar] [CrossRef] [PubMed]
- Houng, N.Q.; Nakamura, Y.; Kuramoto, N.; Yoneyama, M.; Nagashima, R.; Shiba, T.; Yamaguchi, T.; Hasebe, S.; Ogita, K. Indomethacin ameliorates trimethyltin-induced neuronal damage in vivo by attenuating oxidative stress in the dentate gyrus of mice. Biol. Pharm. Bull. 2011, 34, 1856–1863. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fiedorowicz, A.; Figiel, I.; Zaremba, M.; Dzwonek, K.; Schliebs, R.; Oderfeld-Nowak, B. Trimethyltin-evoked apoptosis of murine hippocampal granule neurons is accompanied by the expression of interleukin-1beta and interleukin-1 receptor antagonist in cells of ameboid phenotype, the majority of which are NG-2-positive. Brain Res. Bull. 2008, 77, 19–26. [Google Scholar] [CrossRef]
- Harry, G.J.; Tyler, K.; d’Hellencourt, C.L.; Tilson, H.A.; Maier, W.E. Morphological alterations and elevations in tumor necrosis factor-α, interleukin (IL)-1α, and IL-6 in mixed glia cultures following exposure to trimethyltin: Modulation by proinflammatory cytokine recombinant proteins and neutralizing antibodies. Toxicol. Appl. Pharmacol. 2002, 180, 205–218. [Google Scholar] [CrossRef]
- Maier, W.E.; Bartenbach, M.J.; Brown, H.W.; Tilson, H.A.; Harry, G.J. Induction of tumor necrosis factor alpha in cultured glial cells by trimethyltin. Neurochem. Int. 1997, 30, 385–392. [Google Scholar] [CrossRef]
- Reali, C.; Scintu, F.; Pillai, R.; Donato, R.; Michetti, F.; Sogos, V. S100b counteracts effects of the neurotoxicant trimethyltin on astrocyte and microglia. J. Neurosci. Res. 2005, 81, 677–686. [Google Scholar] [CrossRef]
- Kaur, S.; Sharma, N.; Nehru, B. Anti-inflammatory effects of Ginko biloba extract against trimethyltin-induced hippocampal neuronal injury. Inflammopharmacology 2018, 26, 87–104. [Google Scholar] [CrossRef] [PubMed]
- Kassed, C.A.; Butler, T.L.; Patton, G.W.; Demesquita, D.D.; Navidomskis, M.T.; Mémet, S.; Israël, A.; Pennypacker, K.R. Injury-induced NF-kappaB activation in the hippocampus: Implications for neuronal survival. FASEB J. 2004, 18, 723–724. [Google Scholar] [CrossRef]
- Elgarhi, R.; Shehata, M.M.; Abdelsameea, A.A.; Salem, A.E. Effects of Diclofenac versus meloxicam in pentylenetetrazol-kindled mice. Neurochem.Res. 2020, 45, 1913–1919. [Google Scholar] [CrossRef] [PubMed]
- Yuliani, S.; Mustofa; Partadiredja, G. The neuroprotective effects of an ethanolic turmeric (Curcuma longa L.) extract against trimethyltin-induced oxidative stress in rats. Nutr. Neurosci. 2019, 22, 797–804. [Google Scholar] [CrossRef] [PubMed]
- Kaur, S.; Chhabra, R.; Nehru, B. Ginkgo biloba extract attenuates hippocampal neuronal loss and cognitive dysfunction resulting from trimethyltin in mice. Phytomedicine 2013, 20, 178–186. [Google Scholar] [CrossRef] [PubMed]
- Dawson, R., Jr.; Patterson, T.A.; Eppler, B. Endogenous excitatory amino acid release from brain slices and astrocyte cultures evoked by trimethyltin and other neurotoxic agents. Neurochem. Res. 1995, 20, 847–858. [Google Scholar] [CrossRef] [PubMed]
- Rakshit, J.; Priyam, A.; Gowrishetty, K.K.; Mishra, S.; Bandyopadhyay, J. Iron chelator deferoxamine protects human neuroblastoma cell line SH-SY5Y from 6-hydroxydopamine-induced apoptosis and autophagy dysfunction. J. Trace Elem. Med. Biol. 2020, 57, 126406. [Google Scholar] [CrossRef]
- Heras-Sandoval, D.; Pérez-Rojas, J.M.; Hernández-Damián, J.; Pedraza-Chaverri, J. The role of PI3K/Akt/mTOR pathway in the modulation of autophagy and the clearance of protein aggregates in neurodegeneration. Cell Signal. 2014, 26, 2694–2701. [Google Scholar] [CrossRef] [PubMed]
- Hodges, S.; Lugo, J.N. Therapeutic role of targeting mTOR signaling and neuroinflammation in epilepsy. Epilepsy Res. 2020, 161, 106282. [Google Scholar] [CrossRef]
- Fulda, S. The PI3K/Akt/mTOR pathway as therapeutic target in neuroblastoma. Curr. Cancer Drug Targets. 2009, 9, 729–737. [Google Scholar] [CrossRef] [PubMed]
- Fabrizi, C.; De Vito, S.; Somma, F.; Pompili, E.; Catizone, A.; Leone, S.; Lenzi, P.; Fornai, F.; Fumagalli, L. Lithium improves survival of PC12 pheochromocytoma cells in high-density cultures and after exposure to toxic compounds. Int. J. Cell Biol. 2014, 2014, 135908. [Google Scholar] [CrossRef] [PubMed]








Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, W.-J.; Yun, J.-H.; Jeong, M.-S.; Kim, K.-N.; Shin, T.; Kim, H.-C.; Wie, M.-B. Inhibitors of Lipoxygenase and Cyclooxygenase-2 Attenuate Trimethyltin-Induced Neurotoxicity through Regulating Oxidative Stress and Pro-Inflammatory Cytokines in Human Neuroblastoma SH-SY5Y Cells. Brain Sci. 2021, 11, 1116. https://doi.org/10.3390/brainsci11091116
Song W-J, Yun J-H, Jeong M-S, Kim K-N, Shin T, Kim H-C, Wie M-B. Inhibitors of Lipoxygenase and Cyclooxygenase-2 Attenuate Trimethyltin-Induced Neurotoxicity through Regulating Oxidative Stress and Pro-Inflammatory Cytokines in Human Neuroblastoma SH-SY5Y Cells. Brain Sciences. 2021; 11(9):1116. https://doi.org/10.3390/brainsci11091116
Chicago/Turabian StyleSong, Woo-Ju, Jang-Hyuk Yun, Myeong-Seon Jeong, Kil-Nam Kim, Taekyun Shin, Hyoung-Chun Kim, and Myung-Bok Wie. 2021. "Inhibitors of Lipoxygenase and Cyclooxygenase-2 Attenuate Trimethyltin-Induced Neurotoxicity through Regulating Oxidative Stress and Pro-Inflammatory Cytokines in Human Neuroblastoma SH-SY5Y Cells" Brain Sciences 11, no. 9: 1116. https://doi.org/10.3390/brainsci11091116
APA StyleSong, W.-J., Yun, J.-H., Jeong, M.-S., Kim, K.-N., Shin, T., Kim, H.-C., & Wie, M.-B. (2021). Inhibitors of Lipoxygenase and Cyclooxygenase-2 Attenuate Trimethyltin-Induced Neurotoxicity through Regulating Oxidative Stress and Pro-Inflammatory Cytokines in Human Neuroblastoma SH-SY5Y Cells. Brain Sciences, 11(9), 1116. https://doi.org/10.3390/brainsci11091116






