A Phase 1/2 Study of Flavocoxid, an Oral NF-κB Inhibitor, in Duchenne Muscular Dystrophy
Abstract
1. Introduction
2. Patients and Methods
2.1. Inclusion Criteria
2.2. Exclusion Criteria
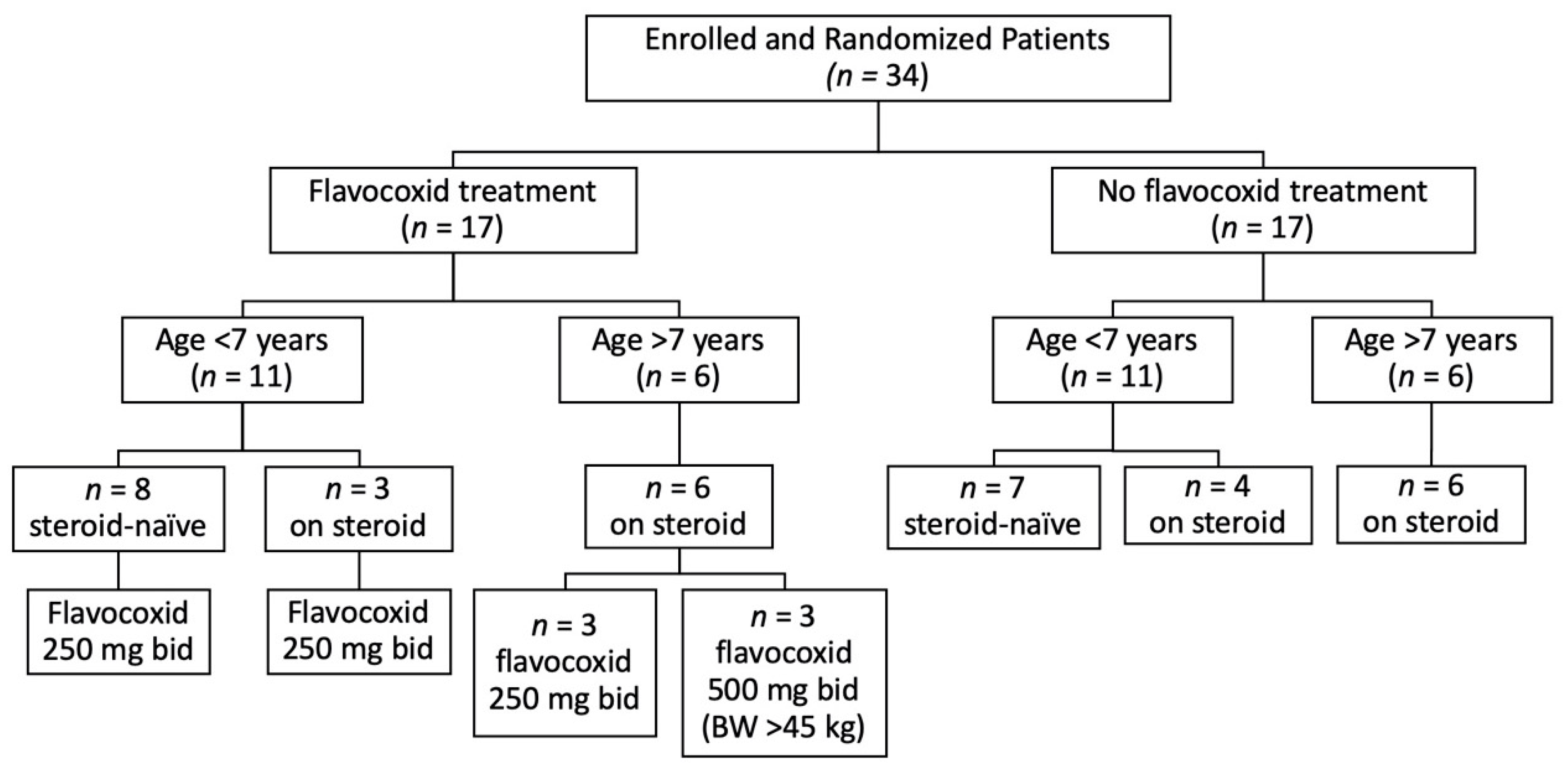
2.3. Randomization
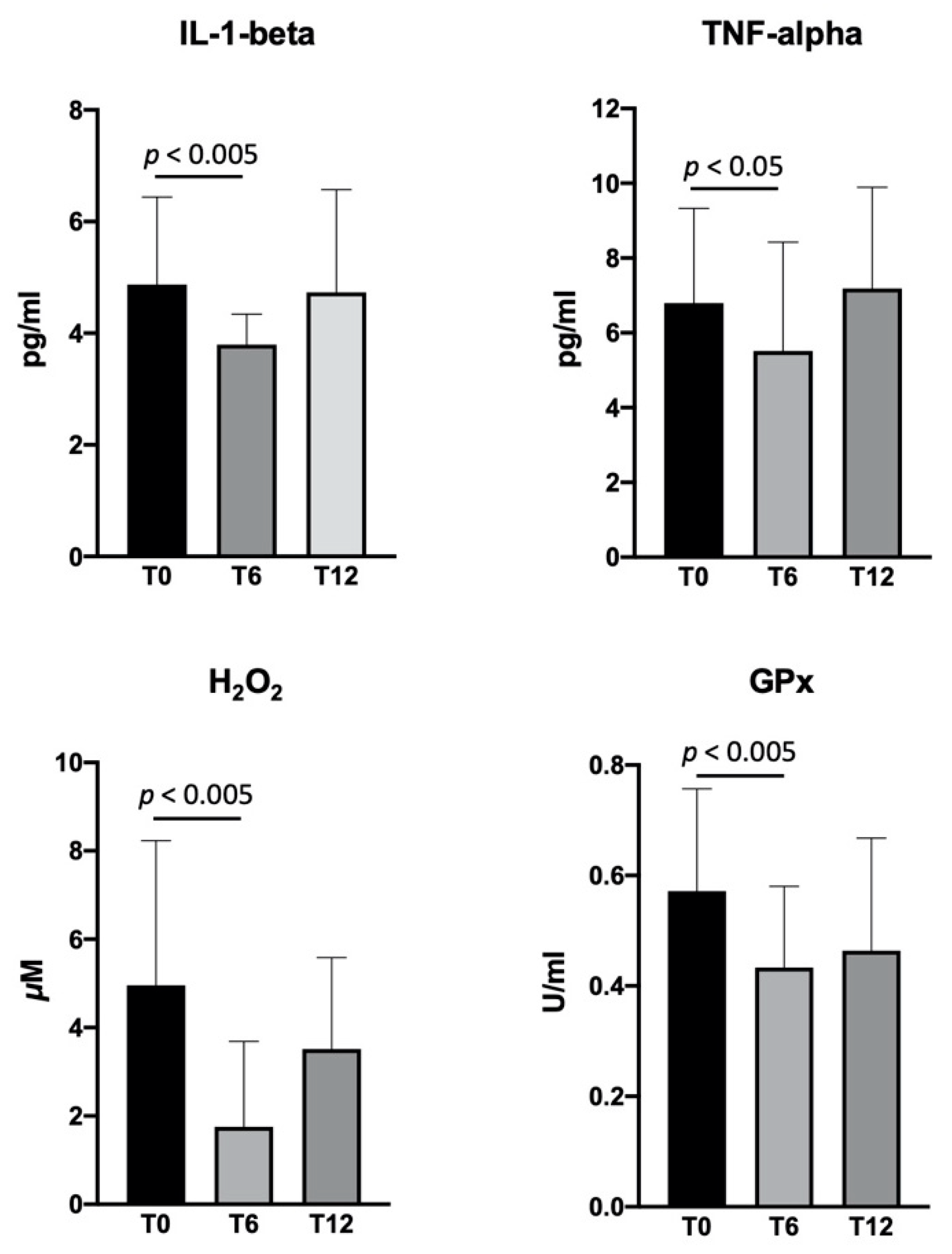
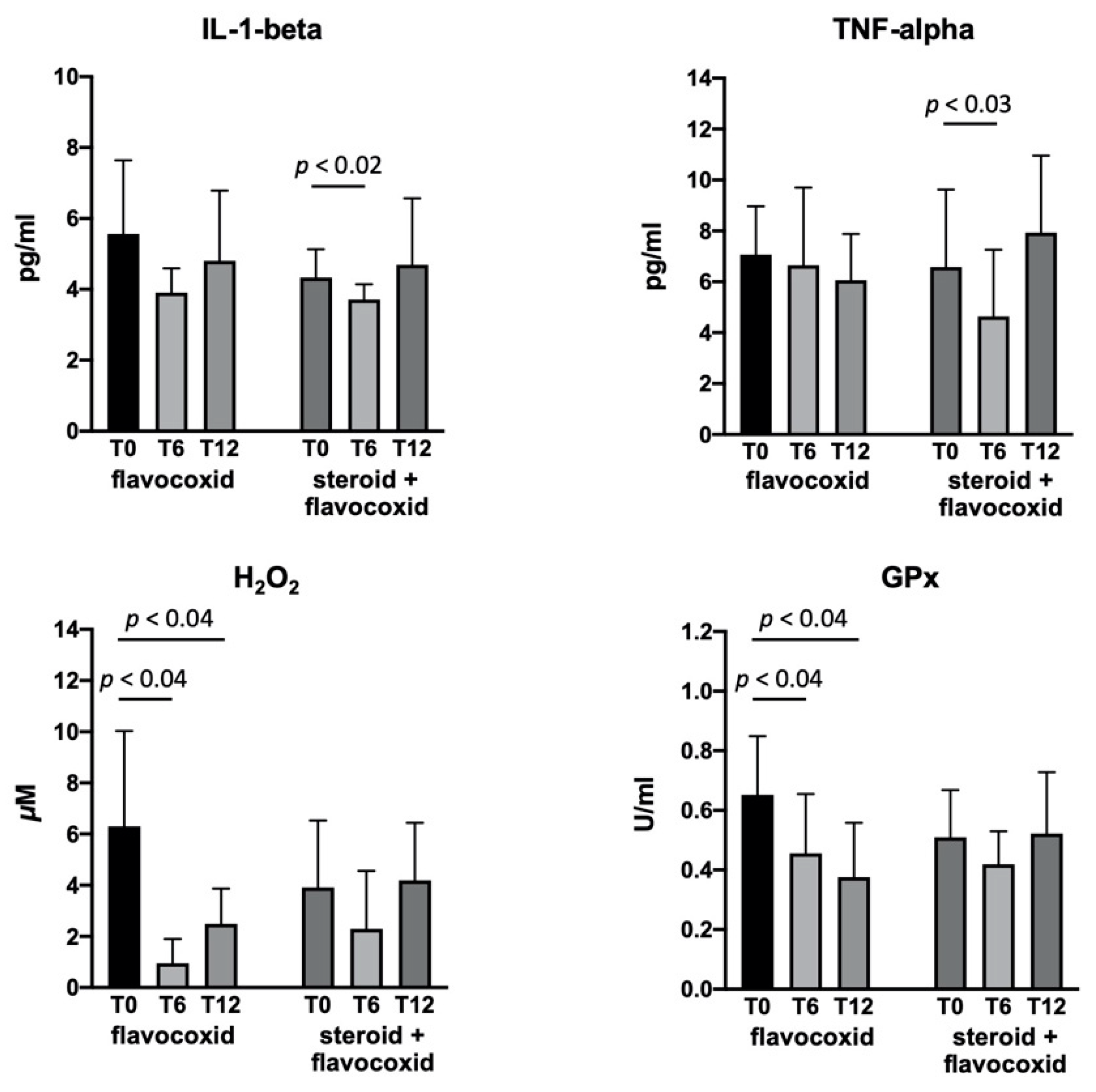
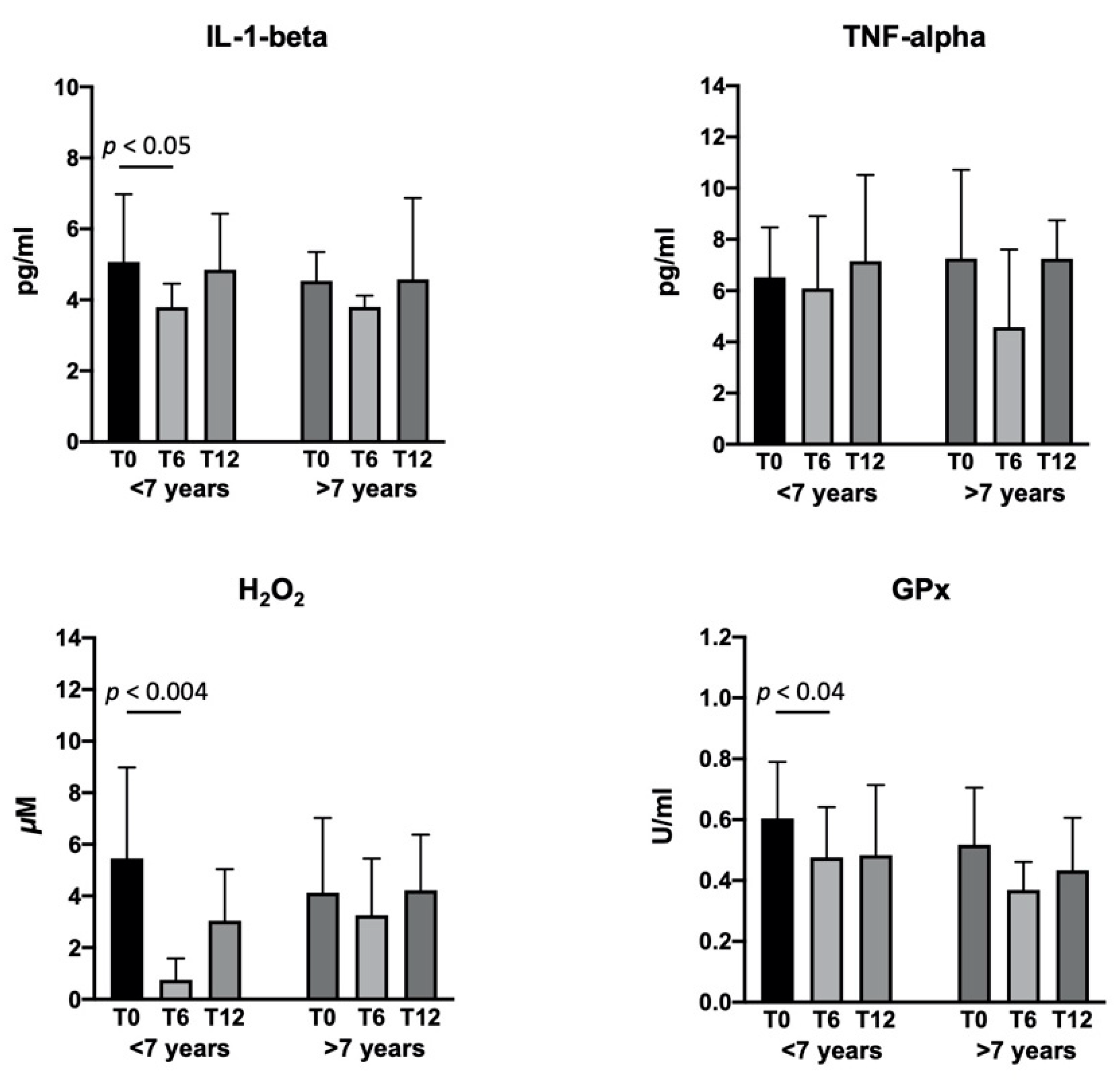
2.4. Biological Activity
2.5. Endpoints
- North Star Ambulatory Assessment (NSAA), which is a 17-item scale, ranging from standing (item 1) to running (item 17) and including several items assessing abilities that are necessary to remain functionally ambulant, with a total score ranging from 0 if all the activities are failed to 34 if all the activities are achieved [30].
- The timed items are included in NSAA, i.e., the time to rise from the floor (Gowers’ time) and the 10-m walk test (10MWT) [30].
- The 6MWT, performed according to the established guidelines [31]. The evaluator instructed the patient to walk at his own pace while covering as much distance as possible during the allotted time. During the test, the patient was allowed to rest or stop and then continue as soon as he could resume walking. At the end of the six minutes, the distance covered was recorded. The 6MWT was performed along a long, flat, straight, corridor with turnaround points at an interval of 25 m.
- Forced vital capacity (FVC), measured using a standard spirometer, taking the best of three attempts [32].
2.6. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Muntoni, F.; Torelli, S.; Ferlini, A. Dystrophin and mutations: One gene, several proteins, multiple phenotypes. Lancet Neurol. 2003, 2, 731–740. [Google Scholar] [CrossRef]
- Naidoo, M.; Anthony, K. Dystrophin Dp71 and the Neuropathophysiology of Duchenne Muscular Dystrophy. Mol. Neurobiol. 2020, 57, 1748–1767. [Google Scholar] [CrossRef] [PubMed]
- Dalkilic, I.; Kunkel, L.M. Muscular dystrophies: Genes to pathogenesis. Curr. Opin. Genet. Dev. 2003, 13, 231–238. [Google Scholar] [CrossRef]
- Messina, S.; Vita, G.L. Clinical management of Duchenne muscular dystrophy: The state of the art. Neurol. Sci. 2018, 39, 1837–1845. [Google Scholar] [CrossRef] [PubMed]
- Mah, J.K. An Overview of Recent Therapeutics Advances for Duchenne Muscular Dystrophy. Methods Mol. Biol. 2018, 1687, 3–17. [Google Scholar]
- Syed, Y.Y. Eteplirsen: First Global Approval. Drugs 2016, 76, 1699–1704. [Google Scholar] [CrossRef]
- Heo, Y.A. Golodirsen: First Approval. Drugs 2020, 80, 329–333. [Google Scholar] [CrossRef]
- Dhillon, S. Viltolarsen: First Approval. Drugs 2020, 80, 1027–1031. [Google Scholar] [CrossRef]
- Messina, S.; Bitto, A.; Aguennouz, M.; Vita, G.L.; Polito, F.; Irrera, N.; Altavilla, D.; Marini, H.; Migliorato, A.; Squadrito, F.; et al. The soy isoflavone genistein blunts nuclear factor kappa-B, MAPKs and TNF-α activation and ameliorates muscle function and morphology in mdx mice. Neuromuscul. Disord. 2011, 21, 579–589. [Google Scholar] [CrossRef]
- Ruegg, U.T. Pharmacological prospects in the treatment of Duchenne muscular dystrophy. Curr. Opin. Neurol. 2013, 26, 577–584. [Google Scholar] [CrossRef]
- Messina, S.; Bitto, A.; Vita, G.L.; Aguennouz, M.; Irrera, N.; Licata, N.; Sframeli, M.; Bruschetta, D.; Minutoli, L.; Altavilla, D.; et al. Modulation of neuronal nitric oxide synthase and apoptosis by the isoflavone genistein in Mdx mice. BioFactors 2015, 41, 324–329. [Google Scholar] [CrossRef] [PubMed]
- Miyatake, S.; Shimizu-Motohashi, Y.; Takeda, S.; Aoki, Y. Anti-inflammatory drugs for Duchenne muscular dystrophy: Focus on skeletal muscle-releasing factors. Drug Des. Dev. Ther. 2016, 10, 2745–2758. [Google Scholar]
- Vita, G.L.; Polito, F.; Oteri, R.; Arrigo, R.; Ciranni, A.M.; Musumeci, O.; Messina, S.; Rodolico, C.; Di Giorgio, R.M.; Vita, G.; et al. Hippo signaling pathway is altered in Duchenne muscular dystrophy. PLoS ONE 2018, 13, e0205514. [Google Scholar] [CrossRef] [PubMed]
- Monici, M.C.; Aguennouz, M.; Mazzeo, A.; Messina, C.; Vita, G. Activation of nuclear factor-kappaB in inflammatory myopathies and Duchenne muscular dystrophy. Neurology 2003, 60, 993–997. [Google Scholar] [CrossRef] [PubMed]
- Messina, S.; Altavilla, D.; Aguennouz, M.; Seminara, P.; Minutoli, L.; Monici, M.C.; Bitto, A.; Mazzeo, A.; Marini, H.; Squadrito, F.; et al. Lipid peroxidation inhibition blunts nuclear factor-kappaB activation, reduces skeletal muscle degeneration, and enhances muscle function in mdx mice. Am. J. Pathol. 2006, 168, 918–926. [Google Scholar] [CrossRef]
- Messina, S.; Bitto, A.; Aguennouz, M.; Minutoli, L.; Monici, M.C.; Altavilla, D.; Squadrito, F.; Vita, G. Nuclear factor kappa-B blockade reduces skeletal muscle degeneration and enhances muscle function in Mdx mice. Exp. Neurol. 2006, 198, 234–241. [Google Scholar] [CrossRef]
- Mourkioti, F.; Kratsios, P.; Luedde, T.; Song, Y.H.; Delafontaine, P.; Adami, R.; Parente, V.; Bottinelli, R.; Pasparakis, M.; Rosenthal, N. Targeted ablation of IKK2 improves skeletal muscle strength, maintains mass, and promotes regeneration. J. Clin. Investig. 2006, 116, 2945–2954. [Google Scholar] [CrossRef]
- Acharyya, S.; Villalta, S.A.; Bakkar, N.; Bupha-Intr, T.; Janssen, P.M.; Carathers, M.; Li, Z.W.; Beg, A.A.; Ghosh, S.; Sahenk, Z.; et al. Interplay of IKK/NF-kappaB signaling in macrophages and myofibers promotes muscle degeneration in Duchenne muscular dystrophy. J. Clin. Investig. 2007, 117, 889–901. [Google Scholar] [CrossRef]
- Hnia, K.; Gayraud, J.; Hugon, G.; Ramonatxo, M.; De La Porte, S.; Matecki, S.; Mornet, D. L-arginine decreases inflammation and modulates the nuclear factor-kappaB/matrix metalloproteinase cascade in mdx muscle fibers. Am. J. Pathol. 2008, 172, 1509–1519. [Google Scholar] [CrossRef]
- Pan, Y.; Chen, C.; Shen, Y.; Zhu, C.H.; Wang, G.; Wang, X.C.; Chen, H.Q.; Zhu, M.S. Curcumin alleviates dystrophic muscle pathology in mdx mice. Mol. Cells 2008, 25, 531–537. [Google Scholar]
- Peterson, J.M.; Wang, D.J.; Shettigar, V.; Roof, S.R.; Canan, B.D.; Bakkar, N.; Shintaku, J.; Gu, J.M.; Little, S.C.; Ratnam, N.M.; et al. NF-κB inhibition rescues cardiac function by remodeling calcium genes in a Duchenne muscular dystrophy model. Nat. Commun. 2018, 9, 3431. [Google Scholar] [CrossRef] [PubMed]
- Levy, R.M.; Khokhlov, A.; Kopenkin, S.; Bart, B.; Ermolova, T.; Kantemirova, R.; Mazurov, V.; Bell, M.; Caldron, P.; Pillai, L.; et al. Efficacy and safety of flavocoxid, a novel therapeutic, compared with naproxen: A randomized multicenter controlled trial in subjects with osteoarthritis of the knee. Adv. Ther. 2010, 27, 731–742. [Google Scholar] [CrossRef]
- Morgan, S.L.; Baggott, J.E.; Moreland, L.; Desmond, R.; Kendrach, A.C. The safety of flavocoxid, a medical food, in the dietary management of knee osteoarthritis. J. Med. Food 2009, 12, 1143–1148. [Google Scholar] [CrossRef] [PubMed]
- Jia, Q. Generating and screening a natural product library for cyclooxygenase and lipoxygenase dual inhibitors. In Studies in Natural Products Chemistry; Rahman, A.U., Ed.; Elsevier Science: New York, NY, USA, 2003; Volume 29, pp. 643–718. [Google Scholar]
- Burnett, B.P.; Jia, Q.; Zhao, Y.; Levy, R.M. A medicinal extract of Scutellaria baicalensis and Acacia catechu acts as a dual inhibitor of cyclooxygenase and 5-lipoxygenase to reduce inflammation. J. Med. Food 2007, 10, 442–451. [Google Scholar] [CrossRef] [PubMed]
- Bremner, P.; Heinrich, M. Natural products as targeted modulators of the nuclear factor-kappaB pathway. J. Pharm. Pharmacol. 2002, 54, 453–472. [Google Scholar] [CrossRef]
- Bitto, A.; Squadrito, F.; Irrera, N.; Pizzino, G.; Pallio, G.; Mecchio, A.; Galfo, F.; Altavilla, D. Flavocoxid, a nutraceutical approach to blunt inflammatory conditions. Mediat. Inflamm. 2014, 2014, 790851. [Google Scholar] [CrossRef]
- Messina, S.; Bitto, A.; Aguennouz, M.; Mazzeo, A.; Migliorato, A.; Polito, F.; Irrera, N.; Altavilla, D.; Vita, G.L.; Russo, M.; et al. Flavocoxid counteracts muscle necrosis and improves functional properties in mdx mice: A comparison study with methylprednisolone. Exp. Neurol. 2009, 220, 349–358. [Google Scholar] [CrossRef]
- Schulz, K.F.; Grimes, D.A. Unequal group sizes in randomised trials: Guarding against guessing. Lancet 2002, 359, 966–970. [Google Scholar] [CrossRef]
- Mazzone, E.S.; Pane, M.; Sormani, M.P.; Scalise, R.; Berardinelli, A.; Messina, S.; Torrente, Y.; D’Amico, A.; Doglio, L.; Viggiano, E.; et al. 24 month longitudinal data in ambulant boys with Duchenne muscular dystrophy. PLoS ONE 2013, 8, e52512. [Google Scholar] [CrossRef]
- ATS Committee on Proficiency Standards for Clinical Pulmonary Function Laboratories. ATS statement: Guidelines for the six-minute walk test. Am. J. Respir. Crit. Care Med. 2002, 166, 111–117. [Google Scholar] [CrossRef]
- Mercuri, E.; Bertini, E.; Messina, S.; Solari, A.; D’Amico, A.; Angelozzi, C.; Battini, R.; Berardinelli, A.; Boffi, P.; Bruno, C.; et al. Randomized, double-blind, placebo-controlled trial of phenylbutyrate in spinal muscular atrophy. Neurology 2007, 68, 51–55. [Google Scholar] [CrossRef] [PubMed]
- Puri, B.K.; White, N.; Monro, J.A. The effect of supplementation with Scutellaria baicalensis on hepatic function. Med. Hypotheses 2019, 133, 109402. [Google Scholar] [CrossRef] [PubMed]
- Curtis, J.R.; Owensby, J.K.; Xie, F. Comparative safety of flavocoxid vs prescription NSAIDs among osteoarthritis patients. Osteoarthr. Cartil. 2020, 28, 917–923. [Google Scholar] [CrossRef] [PubMed]
- Oliveira-Marques, V.; Marinho, H.S.; Cyrne, L.; Antunes, F. Role of hydrogen peroxide in NF-kappaB activation: From inducer to modulator. Antioxid. Redox Signal. 2009, 11, 2223–2243. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, A.S.; Puig, M.; Nagaraju, K.; Hoffman, E.P.; Villalta, S.A.; Rao, V.A.; Wakefield, L.M.; Woodcock, J. Immune-mediated pathology in Duchenne muscular dystrophy. Sci. Transl. Med. 2015, 7, 299rv4. [Google Scholar] [CrossRef]
- Grounds, M.D.; Terrill, J.R.; Al-Mshhdani, B.A.; Duong, M.N.; Radley-Crabb, H.G.; Arthur, P.G. Biomarkers for Duchenne muscular dystrophy: Myonecrosis, inflammation and oxidative stress. Dis. Models Mech. 2020, 13, dmm043638. [Google Scholar] [CrossRef]
- Hammers, D.W.; Sleeper, M.M.; Forbes, S.C.; Coker, C.C.; Jirousek, M.R.; Zimmer, M.; Walter, G.A.; Sweeney, H.L. Disease-modifying effects of orally bioavailable NF-κB inhibitors in dystrophin-deficient muscle. JCI Insight 2016, 1, e90341. [Google Scholar] [CrossRef]
- Capogrosso, R.F.; Cozzoli, A.; Mantuano, P.; Camerino, G.M.; Massari, A.M.; Sblendorio, V.T.; De Bellis, M.; Tamma, R.; Giustino, A.; Nico, B.; et al. Assessment of resveratrol, apocynin and taurine on mechanical-metabolic uncoupling and oxidative stress in a mouse model of Duchenne muscular dystrophy: A comparison with the gold standard, α-methyl prednisolone. Pharmacol. Res. 2016, 106, 101–113. [Google Scholar] [CrossRef]
- Finanger, E.; Vandenborne, K.; Finkel, R.S.; Lee Sweeney, H.; Tennekoon, G.; Yum, S.; Mancini, M.; Bista, P.; Nichols, A.; Liu, H.; et al. Phase 1 Study of Edasalonexent (CAT-1004), an Oral NF-κB Inhibitor, in Pediatric Patients with Duchenne Muscular Dystrophy. J. Neuromuscul. Dis. 2019, 6, 43–54. [Google Scholar] [CrossRef]
- Matthews, E.; Brassington, R.; Kuntzer, T.; Jichi, F.; Manzur, A.Y. Corticosteroids for the treatment of Duchenne muscular dystrophy. Cochrane Database Syst. Rev. 2016, 5, CD003725. [Google Scholar] [CrossRef]
- El-Sheakh, A.R.; Ghoneim, H.A.; Suddek, G.M.; Ammar, E.M. Antioxidant and anti-inflammatory effects of flavocoxid in high-cholesterol-fed rabbits. Naunyn Schmiedeberg’s Arch. Pharmacol. 2015, 388, 1333–1344. [Google Scholar] [CrossRef] [PubMed]
- Merlini, L.; Gennari, M.; Malaspina, E.; Cecconi, I.; Armaroli, A.; Gnudi, S.; Talim, B.; Ferlini, A.; Cicognani, A.; Franzoni, E. Early corticosteroid treatment in 4 Duchenne muscular dystrophy patients: 14-year follow-up. Muscle Nerve 2012, 45, 796–802. [Google Scholar] [CrossRef] [PubMed]
- Shimizu-Motohashi, Y.; Murakami, T.; Kimura, E.; Komaki, H.; Watanabe, N. Exon skipping for Duchenne muscular dystrophy: A systematic review and meta-analysis. Orphanet J. Rare Dis. 2018, 13, 93. [Google Scholar] [CrossRef] [PubMed]
- Guglieri, M.; Bushby, K.; McDermott, M.P.; Hart, K.A.; Tawil, R.; Martens, W.B.; Herr, B.E.; McColl, E.; Wilkinson, J.; Kirschner, J.; et al. Developing standardized corticosteroid treatment for Duchenne muscular dystrophy. Contemp. Clin. Trials 2017, 58, 34–39. [Google Scholar] [CrossRef] [PubMed]
| Flavocoxid-Treated (n = 17) | Untreated (n = 17) | Significance | |
|---|---|---|---|
| Age, years | 6.5 ± 2.2 (4–12) | 6.1 ± 2.4 (4–12) | n.s. |
| Steroid, dosage | prednisone, 0.7 mg/kg/alternate days (n = 2) deflazacort, 0.9 mg/kg/alternate days (n = 5) deflazacort, 0.9 mg/kg/day (n = 2) | prednisone, 0.7 mg/kg/alternate days (n = 3) deflazacort, 0.9 mg/kg/alternate days (n = 4) deflazacort, 0.9 mg/kg/day (n = 3) | |
| Steroid duration, months | 26.4 ± 12.5 (8–46) | 25 ± 19.3 (7–60) | n.s. |
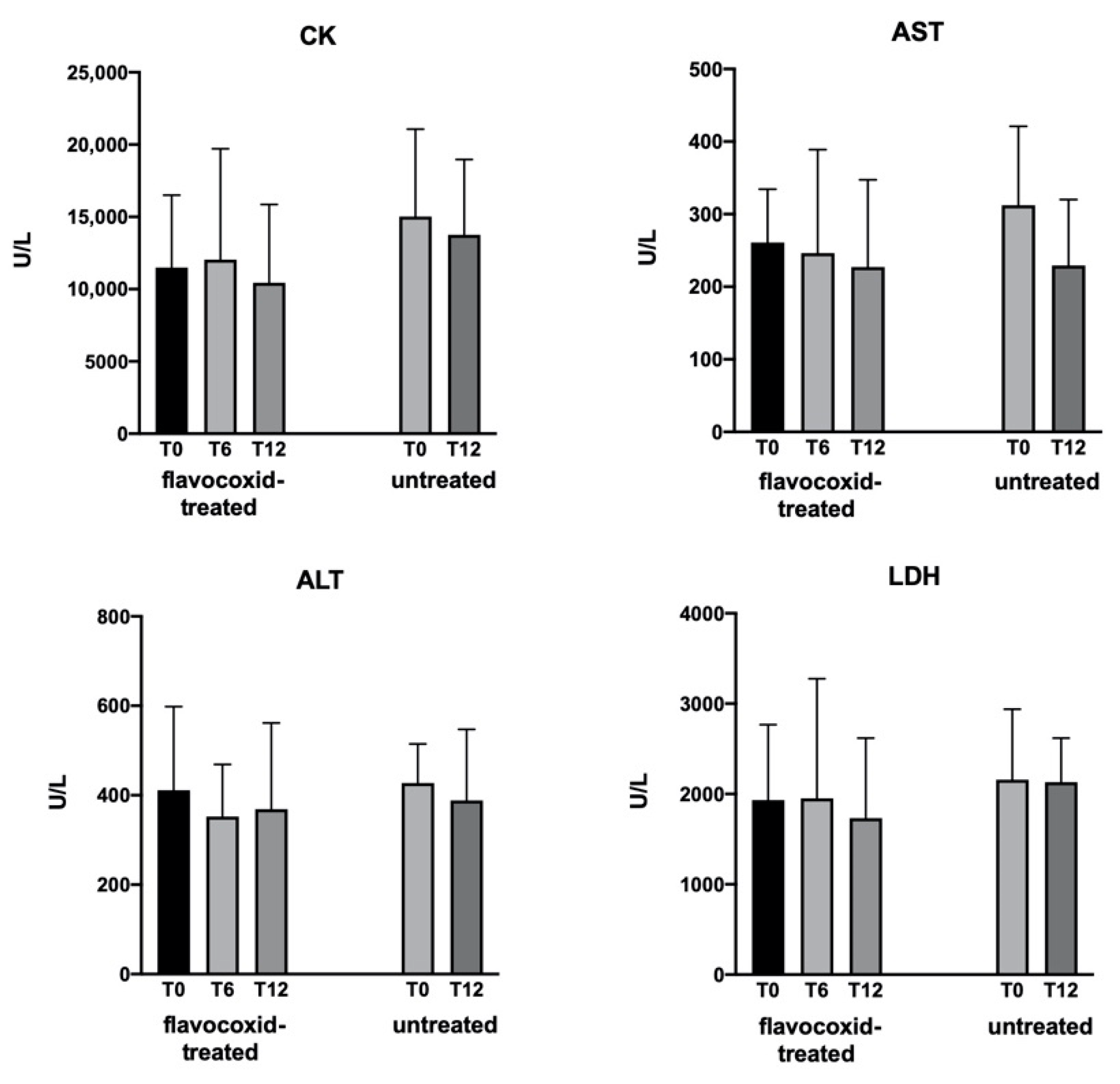
| CK, U/L | 11,490 ± 5010 (3122–20,011) | 15,028 ± 6046 (3494–21,378) | n.s. |
| AST, U/L | 261 ± 73 (120–365) | 312 ± 109 (193–585) | n.s. |
| ALT, U/L | 411 ± 187 (178–831) | 428 ± 87 (295–575) | n.s. |
| LDH, U/L | 1932 ± 836 (810–3380) | 2158 ± 780 (1132–3075) | n.s. |
| NSAA | 28.8 ± 6.25 (16–34) | 27.2 ± 5.7 (16–34) | n.s. |
| Gowers’ time, s | 8.59 ± 13.8 (3.1–60) | 8.47 ± 13.5 (2.1–60) | n.s. |
| 10MWT, s | 6.1 ± 2.4 (3.8–13.1) | 5.5 ± 1.68 (3–10) | n.s. |
| 6MWT, m | 387 ± 96.7 (175–521) | 358 ± 55.6 (225–475) | n.s. |
| FVC, L | 1.18 ± 0.56 (0.47–2.79) | 1.11 ± 0.54 (0.63–2.78) | n.s. |
| Treated | Untreated | Treated < 7 Years | Untreated < 7 Years | Treated > 7 Years | Untreated > 7 Years | ||||
|---|---|---|---|---|---|---|---|---|---|
| Age, years | 6.5 ± 0.54 | 6.1 ± 0.57 | n.s. | 5.1 ± 0.2 | 4.6 ± 0.2 | n.s. | 9.1 ± 0.7 | 8.7 ± 0.8 | n.s. |
| NSAA | −3.2 ± 1.4 | −1.8 ± 1.1 | n.s. | −1.7 ± 1.7 | −0.8 ± 1.9 | n.s. | −6.0 ± 2.4 | −3.1 ± 0.7 | n.s. |
| Gowers’ time, s | 4.5 ± 2.4 | 4.9 ± 2.7 | n.s. | 3.0 ± 1.5 | 6.9 ± 4.4 | n.s. | 7.2 ± 6.6 | 2.1 ± 1.6 | n.s. |
| 10MWT, s | 0.6 ± 0.5 | 1.3 ± 0.7 | n.s. | 0.5 ± 0.5 | 0.5 ± 0.8 | n.s. | 0.8 ± 1 | 2.3 ± 1.3 | n.s. |
| 6MWT, m | −9.7 ± 14 | −2.6 ± 20 | n.s. | −2.1 ± 20 | 17 ± 28 | n.s. | −24 ± 14 | −25 ± 27 | n.s. |
| FVC, L | 0.07 ± 0.07 | 0.04 ± 0.06 | n.s. | 0.09 ± 0.06 | 0.05 ± 0.09 | n.s. | 0.03 ± 0.15 | 0.03 ± 0.06 | n.s. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vita, G.L.; Sframeli, M.; Licata, N.; Bitto, A.; Romeo, S.; Frisone, F.; Ciranni, A.; Pallio, G.; Mannino, F.; Aguennouz, M.; et al. A Phase 1/2 Study of Flavocoxid, an Oral NF-κB Inhibitor, in Duchenne Muscular Dystrophy. Brain Sci. 2021, 11, 115. https://doi.org/10.3390/brainsci11010115
Vita GL, Sframeli M, Licata N, Bitto A, Romeo S, Frisone F, Ciranni A, Pallio G, Mannino F, Aguennouz M, et al. A Phase 1/2 Study of Flavocoxid, an Oral NF-κB Inhibitor, in Duchenne Muscular Dystrophy. Brain Sciences. 2021; 11(1):115. https://doi.org/10.3390/brainsci11010115
Chicago/Turabian StyleVita, Gian Luca, Maria Sframeli, Norma Licata, Alessandra Bitto, Sara Romeo, Francesca Frisone, Annamaria Ciranni, Giovanni Pallio, Federica Mannino, M’Hammed Aguennouz, and et al. 2021. "A Phase 1/2 Study of Flavocoxid, an Oral NF-κB Inhibitor, in Duchenne Muscular Dystrophy" Brain Sciences 11, no. 1: 115. https://doi.org/10.3390/brainsci11010115
APA StyleVita, G. L., Sframeli, M., Licata, N., Bitto, A., Romeo, S., Frisone, F., Ciranni, A., Pallio, G., Mannino, F., Aguennouz, M., Rodolico, C., Squadrito, F., Toscano, A., Messina, S., & Vita, G. (2021). A Phase 1/2 Study of Flavocoxid, an Oral NF-κB Inhibitor, in Duchenne Muscular Dystrophy. Brain Sciences, 11(1), 115. https://doi.org/10.3390/brainsci11010115










