Transcranial Direct Current Stimulation to Facilitate Lower Limb Recovery Following Stroke: Current Evidence and Future Directions
Abstract
1. Introduction
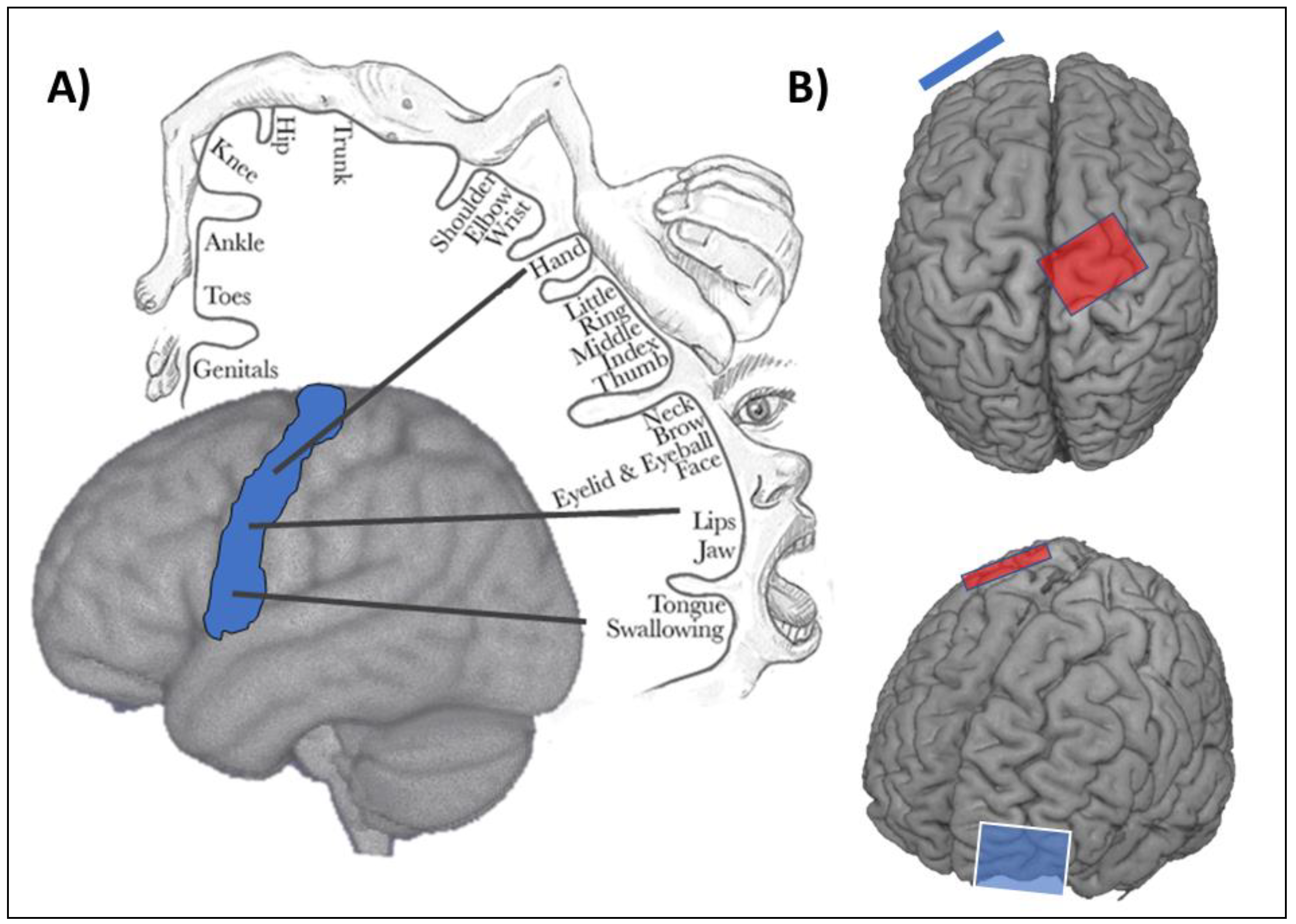
2. Is it Possible to Modify Lower Limb Motor Networks with Transcranial Direct Current Stimulation?
3. Transcranial Direct Current Stimulation to Improve Lower Limb Motor Performance in Healthy Adults and People with Stroke
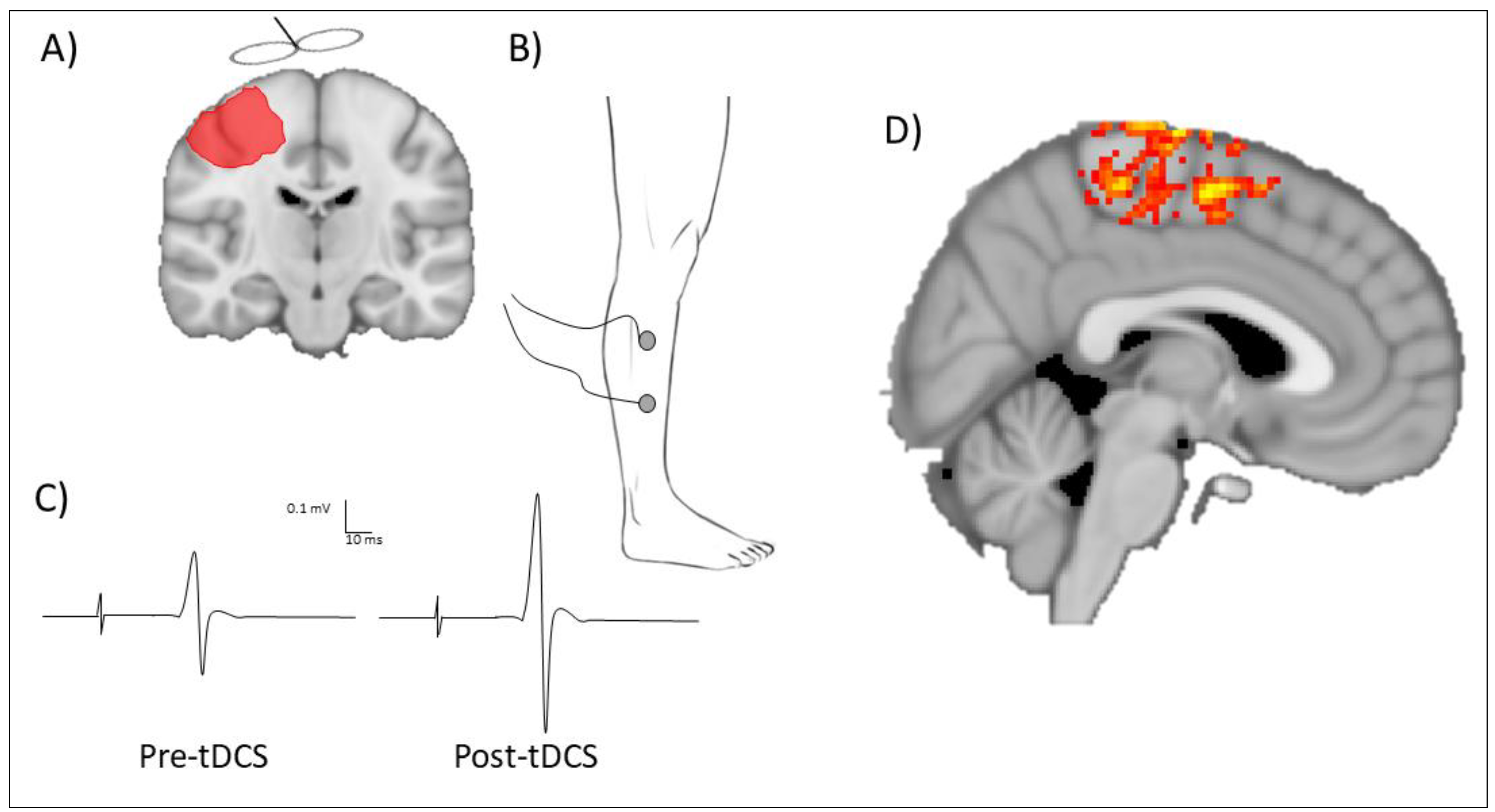
4. Principles of tDCS Application in Stroke
5. Quantifying Response to tDCS Application
6. Biomarkers and Determinants of Response to tDCS
7. Limitations
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Johnson, W.; Onuma, O.; Owolabi, M.; Sachdev, S. Stroke: A global response is needed. Bull. World Health Organ. 2016, 94, 634. [Google Scholar] [CrossRef] [PubMed]
- Murphy, T.H.; Corbett, D. Plasticity during stroke recovery: From synapse to behaviour. Nat. Rev. Neurosci. 2009, 10, 861–872. [Google Scholar] [CrossRef] [PubMed]
- Carmichael, S.T. Cellular and molecular mechanisms of neural repair after stroke: Making waves. Ann. Neurol. 2006, 59, 735–742. [Google Scholar] [CrossRef] [PubMed]
- Overman, J.J.; Carmichael, S.T. Plasticity in the injured brain: More than molecules matter. Neuroscientist 2014, 20, 15–28. [Google Scholar] [CrossRef]
- Biernaskie, J.; Chernenko, G.; Corbett, D. Efficacy of rehabilitative experience declines with time after focal ischemic brain injury. J. Neurosci. 2004, 24, 1245–1254. [Google Scholar] [CrossRef]
- McDonnell, M.N.; Koblar, S.; Ward, N.S.; Rothwell, J.C.; Hordacre, B.; Ridding, M.C. An investigation of cortical neuroplasticity following stroke in adults: Is there evidence for a critical window for rehabilitation? BMC Neurol. 2015, 15, 109. [Google Scholar] [CrossRef]
- Ward, N.S.; Brown, M.M.; Thompson, A.J.; Frackowiak, R.S. Neural correlates of motor recovery after stroke: A longitudinal fmri study. Brain 2003, 126, 2476–2496. [Google Scholar] [CrossRef]
- Fan, Y.-T.; Lin, K.-C.; Liu, H.-L.; Chen, Y.-L.; Wu, C.-Y. Changes in structural integrity are correlated with motor and functional recovery after post-stroke rehabilitation. Restor. Neurol. Neuros. 2015, 33, 835–844. [Google Scholar] [CrossRef]
- Hordacre, B.; Moezzi, B.; Ridding, M.C. Neuroplasticity and network connectivity of the motor cortex following stroke: A transcranial direct current stimulation study. Hum. Brain Mapp. 2018, 39, 3326–3339. [Google Scholar] [CrossRef]
- Hordacre, B.; Ghosh, R.; Goldsworthy, M.R.; Ridding, M.C. Transcranial magnetic stimulation-eeg biomarkers of poststroke upper-limb motor function. J. Stroke Cerebrovasc. Dis. 2019, 28, 104452. [Google Scholar] [CrossRef]
- Swayne, O.B.C.; Rothwell, J.C.; Ward, N.S.; Greenwood, R.J. Stages of motor output reorganization after hemispheric stroke suggested by longitudinal studies of cortical physiology. Cereb. Cortex 2008, 18, 1909–1922. [Google Scholar] [CrossRef] [PubMed]
- Blicher, J.U.; Near, J.; Næss-Schmidt, E.; Stagg, C.J.; Johansen-Berg, H.; Nielsen, J.F.; Østergaard, L.; Ho, Y.-C.L. Gaba levels are decreased after stroke and gaba changes during rehabilitation correlate with motor improvement. Neurorehabil. Neural Repair 2015, 29, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Park, C.-H.; Chang, W.H.; Ohn, S.H.; Kim, S.T.; Bang, O.Y.; Pascual-Leone, A.; Kim, Y.-H. Longitudinal changes of resting-state functional connectivity during motor recovery after stroke. Stroke 2011, 42, 1357–1362. [Google Scholar] [CrossRef] [PubMed]
- Nitsche, M.A.; Paulus, W. Excitability changes induced in the human motor cortex by weak transcranial direct current stimulation. J. Physiol. (Lond) 2000, 527, 633–639. [Google Scholar] [CrossRef] [PubMed]
- Nitsche, M.A.; Paulus, W. Sustained excitability elevations induced by transcranial dc motor cortex stimulation in humans. Neurology 2001, 57, 1899–1901. [Google Scholar] [CrossRef] [PubMed]
- Bindman, L.J.; Lippold, O.C.J.; Redfearn, J.W.T. The action of brief polarizing currents on the cerebral cortex of the rat (1) during current flow and (2) in the production of long-lasting after-effects. J. Physiol. 1964, 172, 369–382. [Google Scholar] [CrossRef] [PubMed]
- Islam, N.; Aftabuddin, M.; Moriwaki, A.; Hattori, Y.; Hori, Y. Increase in the calcium level following anodal polarization in the rat brain. Brain Res. 1995, 684, 206–208. [Google Scholar] [CrossRef]
- Catterall, W.A.; Few, A.P. Calcium channel regulation and presynaptic plasticity. Neuron 2008, 59, 882–901. [Google Scholar] [CrossRef]
- Liebetanz, D.; Nitsche, M.A.; Tergau, F.; Paulus, W. Pharmacological approach to the mechanisms of transcranial dc-stimulation-induced after-effects of human motor cortex excitability. Brain 2002, 125, 2238–2247. [Google Scholar] [CrossRef]
- Boggio, P.S.; Castro, L.O.; Savagim, E.A.; Braite, R.; Cruz, V.C.; Rocha, R.R.; Rigonatti, S.P.; Silva, M.T.A.; Fregni, F. Enhancement of non-dominant hand motor function by anodal transcranial direct current stimulation. Neurosci. Lett. 2006, 404, 232–236. [Google Scholar] [CrossRef]
- Allman, C.; Amadi, U.; Winkler, A.M.; Wilkins, L.; Filippini, N.; Kischka, U.; Stagg, C.J.; Johansen-Berg, H. Ipsilesional anodal tdcs enhances the functional benefits of rehabilitation in patients after stroke. Sci. Transl. Med. 2016, 8, 330re331. [Google Scholar] [CrossRef] [PubMed]
- Figlewski, K.; Blicher, J.U.; Mortensen, J.; Severinsen, K.E.; Nielsen, J.F.; Andersen, H. Transcranial direct current stimulation potentiates improvements in functional ability in patients with chronic stroke receiving constraint-induced movement therapy. Stroke 2017, 48, 229–232. [Google Scholar] [CrossRef] [PubMed]
- Fleming, M.K.; Rothwell, J.C.; Sztriha, L.; Teo, J.T.; Newham, D.J. The effect of transcranial direct current stimulation on motor sequence learning and upper limb function after stroke. Clin. Neurophysiol. 2017, 128, 1389–1398. [Google Scholar] [CrossRef] [PubMed]
- Tedesco Triccas, L.; Burridge, J.H.; Hughes, A.; Verheyden, G.; Desikan, M.; Rothwell, J. A double-blinded randomised controlled trial exploring the effect of anodal transcranial direct current stimulation and uni-lateral robot therapy for the impaired upper limb in sub-acute and chronic stroke. NeuroRehabilitation 2015, 37, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.M.; Cui, H.; Han, S.J.; Black-Schaffer, R.; Volz, M.S.; Lee, Y.T.; Herman, S.; Latif, L.A.; Zafonte, R.; Fregni, F. Combination of transcranial direct current stimulation and methylphenidate in subacute stroke. Neurosci. Lett. 2014, 569, 6–11. [Google Scholar] [CrossRef] [PubMed]
- Fusco, A.; Iosa, M.; Venturiero, V.; De Angelis, D.; Morone, G.; Maglione, L.; Bragoni, M.; Coiro, P.; Pratesi, L.; Paolucci, S. After vs. Priming effects of anodal transcranial direct current stimulation on upper extremity motor recovery in patients with subacute stroke. Restor. Neurol. Neurosci. 2014, 32, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Stagg, C.J.; Bachtiar, V.; O’Shea, J.; Allman, C.; Bosnell, R.A.; Kischka, U.; Matthews, P.M.; Johansen-Berg, H. Cortical activation changes underlying stimulation-induced behavioural gains in chronic stroke. Brain 2012, 135, 276–284. [Google Scholar] [CrossRef]
- Kang, N.; Summers, J.J.; Cauraugh, J.H. Non-invasive brain stimulation improves paretic limb force production: A systematic review and meta-analysis. Brain Stimul. 2016, 9, 662–670. [Google Scholar] [CrossRef]
- Danielsson, A.; Willén, C.; Sunnerhagen, K.S. Is walking endurance associated with activity and participation late after stroke?? Disabil. Rehabil. 2011, 33, 2053–2057. [Google Scholar] [CrossRef] [PubMed]
- Pohl, M.; Werner, C.; Holzgraefe, M.; Kroczek, G.; Wingendorf, I.; Hoölig, G.; Koch, R.; Hesse, S. Repetitive locomotor training and physiotherapy improve walking and basic activities of daily living after stroke: A single-blind, randomized multicentre trial (deutsche gangtrainerstudie, degas). Clin. Rehabil. 2007, 21, 17–27. [Google Scholar] [CrossRef]
- Cumming, T.B.; Collier, J.; Thrift, A.G.; Bernhardt, J. The effect of very early mobilization after stroke on psychological well-being. J. Rehabil. Med. 2008, 40, 609–614. [Google Scholar] [CrossRef]
- Whyte, E.M.; Mulsant, B.H. Post stroke depression: Epidemiology, pathophysiology, and biological treatment. Biol. Psychiatry 2002, 52, 253–264. [Google Scholar] [CrossRef]
- Paolucci, S. Epidemiology and treatment of post-stroke depression. Neuropsychiatr. Dis. Treat. 2008, 4, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Robinson, R.G.; Spalletta, G. Poststroke depression: A review. Can. J. Psychiatry. 2010, 55, 341–349. [Google Scholar] [CrossRef]
- Foerster, Á.S.; Rezaee, Z.; Paulus, W.; Nitsche, M.A.; Dutta, A. Effects of cathode location and the size of anode on anodal transcranial direct current stimulation over the leg motor area in healthy humans. Front. Neurosci. 2018, 12, 443. [Google Scholar] [CrossRef] [PubMed]
- Jeffery, D.T.; Norton, J.A.; Roy, F.D.; Gorassini, M.A. Effects of transcranial direct current stimulation on the excitability of the leg motor cortex. Exp. Brain Res. 2007, 182, 281–287. [Google Scholar] [CrossRef]
- Chang, M.C.; Kim, D.Y.; Park, D.H. Enhancement of cortical excitability and lower limb motor function in patients with stroke by transcranial direct current stimulation. Brain Stimul. 2015, 8, 561–566. [Google Scholar] [CrossRef]
- Rampersad, S.M.; Janssen, A.M.; Lucka, F.; Aydin, U.; Lanfer, B.; Lew, S.; Wolters, C.H.; Stegeman, D.F.; Oostendorp, T.F. Simulating transcranial direct current stimulation with a detailed anisotropic human head model. IEEE Trans. Neural Syst. Rehabil. Eng. 2014, 22, 441–452. [Google Scholar] [CrossRef]
- Sakayori, N.; Kato, S.; Sugawara, M.; Setogawa, S.; Fukushima, H.; Ishikawa, R.; Kida, S.; Kobayashi, K. Motor skills mediated through cerebellothalamic tracts projecting to the central lateral nucleus. Mol. Brain 2019, 12, 13. [Google Scholar] [CrossRef]
- Galea, J.M.; Jayaram, G.; Ajagbe, L.; Celnik, P. Modulation of cerebellar excitability by polarity-specific noninvasive direct current stimulation. J. Neurosci. 2009, 29, 9115–9122. [Google Scholar] [CrossRef]
- Summers, R.L.S.; Chen, M.; Hatch, A.; Kimberley, T.J. Cerebellar transcranial direct current stimulation modulates corticospinal excitability during motor training. Front. Hum. Neurosci. 2018, 12, 118. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, S.; Hanakawa, T.; Honda, M.; Watanabe, K. Enhancement of pinch force in the lower leg by anodal transcranial direct current stimulation. Exp. Brain Res. 2009, 196, 459–465. [Google Scholar] [CrossRef]
- Devanathan, D.; Madhavan, S. Effects of anodal tdcs of the lower limb m1 on ankle reaction time in young adults. Exp. Brain Res. 2016, 234, 377–385. [Google Scholar] [CrossRef]
- Shah, B.; Nguyen, T.T.; Madhavan, S. Polarity independent effects of cerebellar tdcs on short term ankle visuomotor learning. Brain Stimul. 2013, 6, 966–968. [Google Scholar] [CrossRef] [PubMed]
- Foerster, Á.; Melo, L.; Mello, M.; Castro, R.; Shirahige, L.; Rocha, S.; Monte-Silva, K. Cerebellar transcranial direct current stimulation (ctdcs) impairs balance control in healthy individuals. Cerebellum 2017, 16, 872–875. [Google Scholar] [CrossRef] [PubMed]
- Kumari, N.; Taylor, D.; Signal, N. The effect of cerebellar transcranial direct current stimulation on motor learning: A systematic review of randomized controlled trials. Front. Hum. Neurosci. 2019, 13, 328. [Google Scholar] [CrossRef] [PubMed]
- Maeda, K.; Yamaguchi, T.; Tatemoto, T.; Kondo, K.; Otaka, Y.; Tanaka, S. Transcranial direct current stimulation does not affect lower extremity muscle strength training in healthy individuals: A triple-blind, sham-controlled study. Front. Neurosci. 2017, 11, 179. [Google Scholar] [CrossRef]
- Tanaka, S.; Takeda, K.; Otaka, Y.; Kita, K.; Osu, R.; Honda, M.; Sadato, N.; Hanakawa, T.; Watanabe, K. Single session of transcranial direct current stimulation transiently increases knee extensor force in patients with hemiparetic stroke. Neurorehabil. Neural Repair 2011, 25, 565–569. [Google Scholar] [CrossRef]
- Madhavan, S.; Weber, K.A., 2nd; Stinear, J.W. Non-invasive brain stimulation enhances fine motor control of the hemiparetic ankle: Implications for rehabilitation. Exp. Brain Res. 2011, 209, 9–17. [Google Scholar] [CrossRef]
- Klomjai, W.; Aneksan, B.; Pheungphrarattanatrai, A.; Chantanachai, T.; Choowong, N.; Bunleukhet, S.; Auvichayapat, P.; Nilanon, Y.; Hiengkaew, V. Effect of single-session dual-tdcs before physical therapy on lower-limb performance in sub-acute stroke patients: A randomized sham-controlled crossover study. Ann. Phys. Rehabil. Med. 2018, 61, 286–291. [Google Scholar] [CrossRef]
- Geroin, C.; Picelli, A.; Munari, D.; Waldner, A.; Tomelleri, C.; Smania, N. Combined transcranial direct current stimulation and robot-assisted gait training in patients with chronic stroke: A preliminary comparison. Clin. Rehabil. 2011, 25, 537–548. [Google Scholar] [CrossRef]
- Leon, D.; Cortes, M.; Elder, J.; Kumru, H.; Laxe, S.; Edwards, D.J.; Tormos, J.M.; Bernabeu, M.; Pascual-Leone, A. Tdcs does not enhance the effects of robot-assisted gait training in patients with subacute stroke. Restor. Neurol. Neurosci. 2017, 35, 377–384. [Google Scholar] [CrossRef]
- Hordacre, B.; Ridding, M.C.; Goldsworthy, M.R. Response variability to non-invasive brain stimulation protocols. Clin. Neurophysiol. 2015, 126, 2249–2250. [Google Scholar] [CrossRef]
- Ridding, M.C.; Ziemann, U. Determinants of the induction of cortical plasticity by non-invasive brain stimulation in healthy subjects. J. Physiol. (Lond) 2010, 588, 2291–2304. [Google Scholar] [CrossRef]
- Hordacre, B.; Moezzi, B.; Goldsworthy, M.R.; Rogasch, N.C.; Graetz, L.J.; Ridding, M.C. Resting state functional connectivity measures correlate with the response to anodal transcranial direct current stimulation. Eur. J. Neurosci. 2017, 45, 837–845. [Google Scholar] [CrossRef]
- McCambridge, A.B.; Stinear, J.W.; Byblow, W.D. ‘I-wave’ recruitment determines response to tdcs in the upper limb, but only so far. Brain Stimul. 2015, 8, 1124–1129. [Google Scholar] [CrossRef]
- Patel, R.; Madhavan, S. Comparison of transcranial direct current stimulation electrode montages for the lower limb motor cortex. Brain Sci. 2019, 9, 189. [Google Scholar] [CrossRef]
- Kuo, H.-I.; Bikson, M.; Datta, A.; Minhas, P.; Paulus, W.; Kuo, M.-F.; Nitsche, M.A. Comparing cortical plasticity induced by conventional and high-definition 4× 1 ring tdcs: A neurophysiological study. Brain Stimul. 2013, 6, 644–648. [Google Scholar] [CrossRef]
- Goldsworthy, M.R.; Hordacre, B. Dose-dependency of transcranial direct current stimulation: Implications for neuroplasticity induction in health and disease. J. Physiol. 2017, 595, 3265–3266. [Google Scholar] [CrossRef]
- Jamil, A.; Batsikadze, G.; Kuo, H.I.; Labruna, L.; Hasan, A.; Paulus, W.; Nitsche, M.A. Systematic evaluation of the impact of stimulation intensity on neuroplastic after-effects induced by transcranial direct current stimulation. J. Physiol. 2017, 595, 1273–1288. [Google Scholar] [CrossRef]
- Takeuchi, N.; Tada, T.; Toshima, M.; Ikoma, K. Correlation of motor function with transcallosal and intracortical inhibition after stroke. J. Rehabil. Med. 2010, 42, 962–966. [Google Scholar] [CrossRef]
- Liepert, J.; Hamzei, F.; Weiller, C. Motor cortex disinhibition of the unaffected hemisphere after acute stroke. Muscle Nerve 2000, 23, 1761–1763. [Google Scholar] [CrossRef]
- Takechi, U.; Matsunaga, K.; Nakanishi, R.; Yamanaga, H.; Murayama, N.; Mafune, K.; Tsuji, S. Longitudinal changes of motor cortical excitability and transcallosal inhibition after subcortical stroke. Clin. Neurophysiol. 2014, 125, 2055–2069. [Google Scholar] [CrossRef]
- Butefisch, C.M.; Wessling, M.; Netz, J.; Seitz, R.J.; Homberg, V. Relationship between interhemispheric inhibition and motor cortex excitability in subacute stroke patients. Neurorehabil. Neural Repair 2008, 22, 4–21. [Google Scholar] [CrossRef]
- Murase, N.; Duque, J.; Mazzocchio, R.; Cohen, L.G. Influence of interhemispheric interactions on motor function in chronic stroke. Ann. Neurol. 2004, 55, 400–409. [Google Scholar] [CrossRef]
- Shimizu, T.; Hosaki, A.; Hino, T.; Sato, M.; Komori, T.; Hirai, S.; Rossini, P.M. Motor cortical disinhibition in the unaffected hemisphere after unilateral cortical stroke. Brain 2002, 125, 1896–1907. [Google Scholar] [CrossRef]
- Khedr, E.M.; Shawky, O.A.; El-Hammady, D.H.; Rothwell, J.C.; Darwish, E.S.; Mostafa, O.M.; Tohamy, A.M. Effect of anodal versus cathodal transcranial direct current stimulation on stroke rehabilitation: A pilot randomized controlled trial. Neurorehabil. Neural Repair 2013, 27, 592–601. [Google Scholar] [CrossRef]
- Fusco, A.; De Angelis, D.; Morone, G.; Maglione, L.; Paolucci, T.; Bragoni, M.; Venturiero, V. The abc of tdcs: Effects of anodal, bilateral and cathodal montages of transcranial direct current stimulation in patients with stroke—A pilot study. Stroke Res. Treat. 2013, 2013, 6. [Google Scholar] [CrossRef]
- Di Pino, G.; Pellegrino, G.; Assenza, G.; Capone, F.; Ferreri, F.; Formica, D.; Ranieri, F.; Tombini, M.; Ziemann, U.; Rothwell, J.C.; et al. Modulation of brain plasticity in stroke: A novel model for neurorehabilitation. Nat. Rev. Neurol. 2014, 10, 597–608. [Google Scholar] [CrossRef]
- Rehme, A.K.; Fink, G.R.; Von Cramon, D.Y.; Grefkes, C. The role of the contralesional motor cortex for motor recovery in the early days after stroke assessed with longitudinal fmri. Cereb. Cortex 2011, 21, 756–768. [Google Scholar] [CrossRef]
- Bradnam, L.V.; Stinear, C.M.; Barber, P.A.; Byblow, W.D. Contralesional hemisphere control of the proximal paretic upper limb following stroke. Cereb. Cortex 2012, 22, 2662–2671. [Google Scholar] [CrossRef]
- Capaday, C.; Lavoie, B.A.; Barbeau, H.; Schneider, C.; Bonnard, M. Studies on the corticospinal control of human walking. I. Responses to focal transcranial magnetic stimulation of the motor cortex. J. Neurophysiol. 1999, 81, 129–139. [Google Scholar] [CrossRef]
- Petersen, N.; Christensen, L.O.D.; Nielsen, J. The effect of transcranial magnetic stimulation on the soleus h reflex during human walking. J. Physiol. (Lond) 1998, 513, 599–610. [Google Scholar] [CrossRef]
- Kwakkel, G.; Lannin, N.A.; Borschmann, K.; English, C.; Ali, M.; Churilov, L.; Saposnik, G.; Winstein, C.; van Wegen, E.E.; Wolf, S.L.; et al. Standardized measurement of sensorimotor recovery in stroke trials: Consensus-based core recommendations from the stroke recovery and rehabilitation roundtable. Int. J. Stroke 2017, 12, 451–461. [Google Scholar] [CrossRef]
- Hordacre, B.; Bradnam, L.V.; Barr, C.; Patritti, B.L.; Crotty, M. Ipsilateral corticomotor excitability is associated with increased gait variability in unilateral transtibial amputees. Eur. J. Neurosci. 2014, 40, 2454–2462. [Google Scholar] [CrossRef][Green Version]
- Hordacre, B.; Bradnam, L.; Barr, C.; Patritti, B.; Crotty, M. Intracortical inhibition is modulated by phase of prosthetic rehabilitation in transtibial amputees. Front. Hum. Neurosci. 2015, 9, 276. [Google Scholar] [CrossRef]
- Deng, Z.-D.; Lisanby, S.H.; Peterchev, A.V. Coil design considerations for deep transcranial magnetic stimulation. Clin. Neurophysiol. 2014, 125, 1202–1212. [Google Scholar] [CrossRef]
- Kim, C.R.; Kim, D.-Y.; Kim, L.S.; Chun, M.H.; Kim, S.J.; Park, C.H. Modulation of cortical activity after anodal transcranial direct current stimulation of the lower limb motor cortex: A functional mri study. Brain Stimul. 2012, 5, 462–467. [Google Scholar] [CrossRef]
- Ameli, M.; Grefkes, C.; Kemper, F.; Riegg, F.P.; Rehme, A.K.; Karbe, H.; Fink, G.R.; Nowak, D.A. Differential effects of high-frequency repetitive transcranial magnetic stimulation over ipsilesional primary motor cortex in cortical and subcortical middle cerebral artery stroke. Ann. Neurol. 2009, 66, 298–309. [Google Scholar] [CrossRef]
- Emara, T.; El Nahas, N.; Elkader, H.A.; Ashour, S.; El Etrebi, A. Mri can predict the response to therapeutic repetitive transcranial magnetic stimulation (rtms) in stroke patients. J. Vasc. Interv. Neurol. 2009, 2, 163–168. [Google Scholar]
- Khaleel, S.H.; Bayoumy, I.M.; El-Nabil, L.M.; Moustafa, R.R. Differential hemodynamic response to repetitive transcranial magnetic stimulation in acute stroke patients with cortical versus subcortical infarcts. Eur. Neurol. 2010, 63, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Boyd, L.A.; Hayward, K.S.; Ward, N.S.; Stinear, C.M.; Rosso, C.; Fisher, R.J.; Carter, A.R.; Leff, A.P.; Copland, D.A.; Carey, L.M.; et al. Biomarkers of stroke recovery: Consensus-based core recommendations from the stroke recovery and rehabilitation roundtable. Int. J. Stroke 2017, 12, 480–493. [Google Scholar] [CrossRef] [PubMed]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gowan, S.; Hordacre, B. Transcranial Direct Current Stimulation to Facilitate Lower Limb Recovery Following Stroke: Current Evidence and Future Directions. Brain Sci. 2020, 10, 310. https://doi.org/10.3390/brainsci10050310
Gowan S, Hordacre B. Transcranial Direct Current Stimulation to Facilitate Lower Limb Recovery Following Stroke: Current Evidence and Future Directions. Brain Sciences. 2020; 10(5):310. https://doi.org/10.3390/brainsci10050310
Chicago/Turabian StyleGowan, Samuel, and Brenton Hordacre. 2020. "Transcranial Direct Current Stimulation to Facilitate Lower Limb Recovery Following Stroke: Current Evidence and Future Directions" Brain Sciences 10, no. 5: 310. https://doi.org/10.3390/brainsci10050310
APA StyleGowan, S., & Hordacre, B. (2020). Transcranial Direct Current Stimulation to Facilitate Lower Limb Recovery Following Stroke: Current Evidence and Future Directions. Brain Sciences, 10(5), 310. https://doi.org/10.3390/brainsci10050310



