The Contribution of Functional Magnetic Resonance Imaging to the Understanding of the Effects of Acute Physical Exercise on Cognition
Abstract
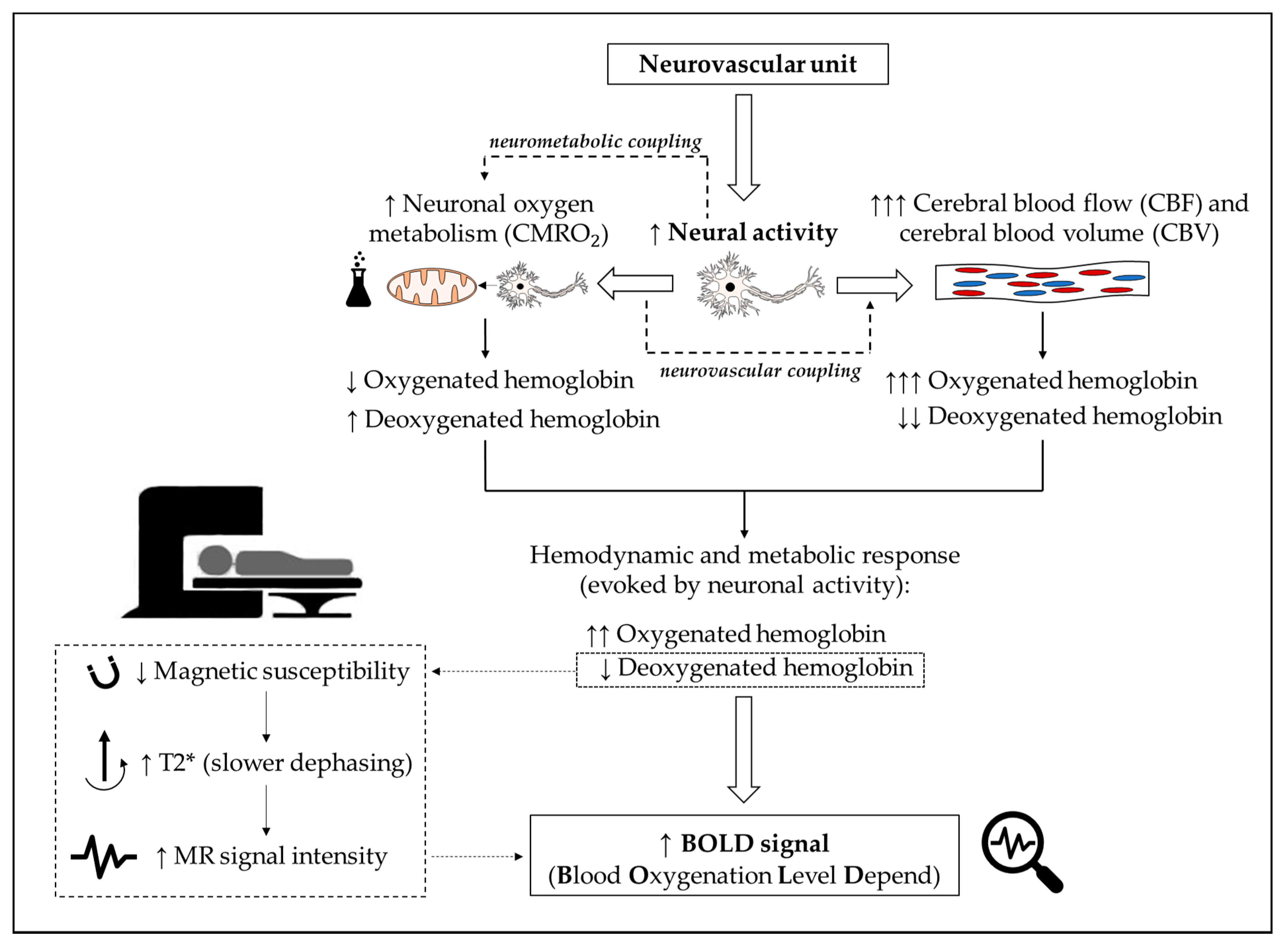
1. Introduction
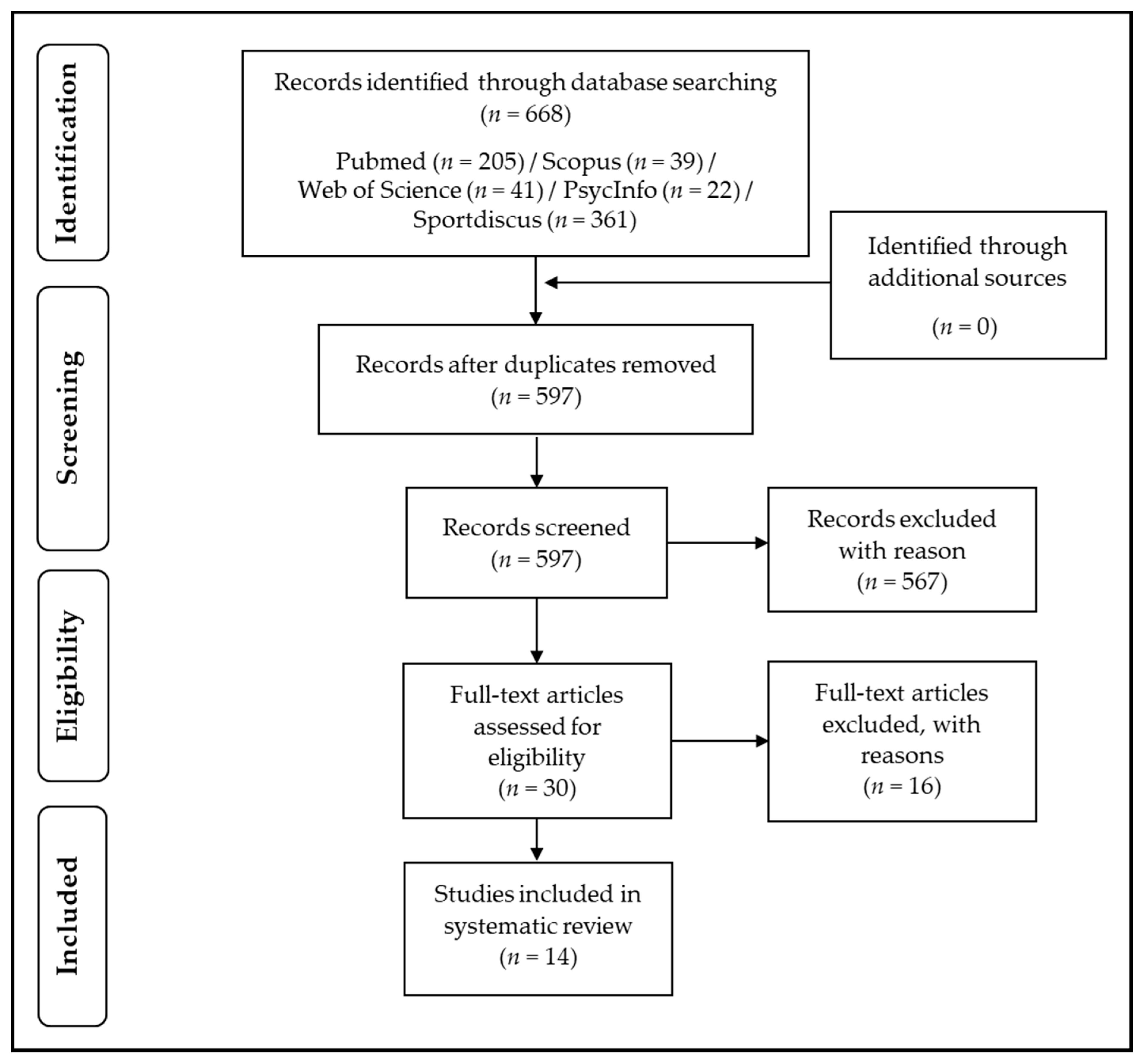
2. Methods
|
| AND |
|
| AND |
|
2.1. Inclusion and Exclusion Criteria
2.2. Data Extraction
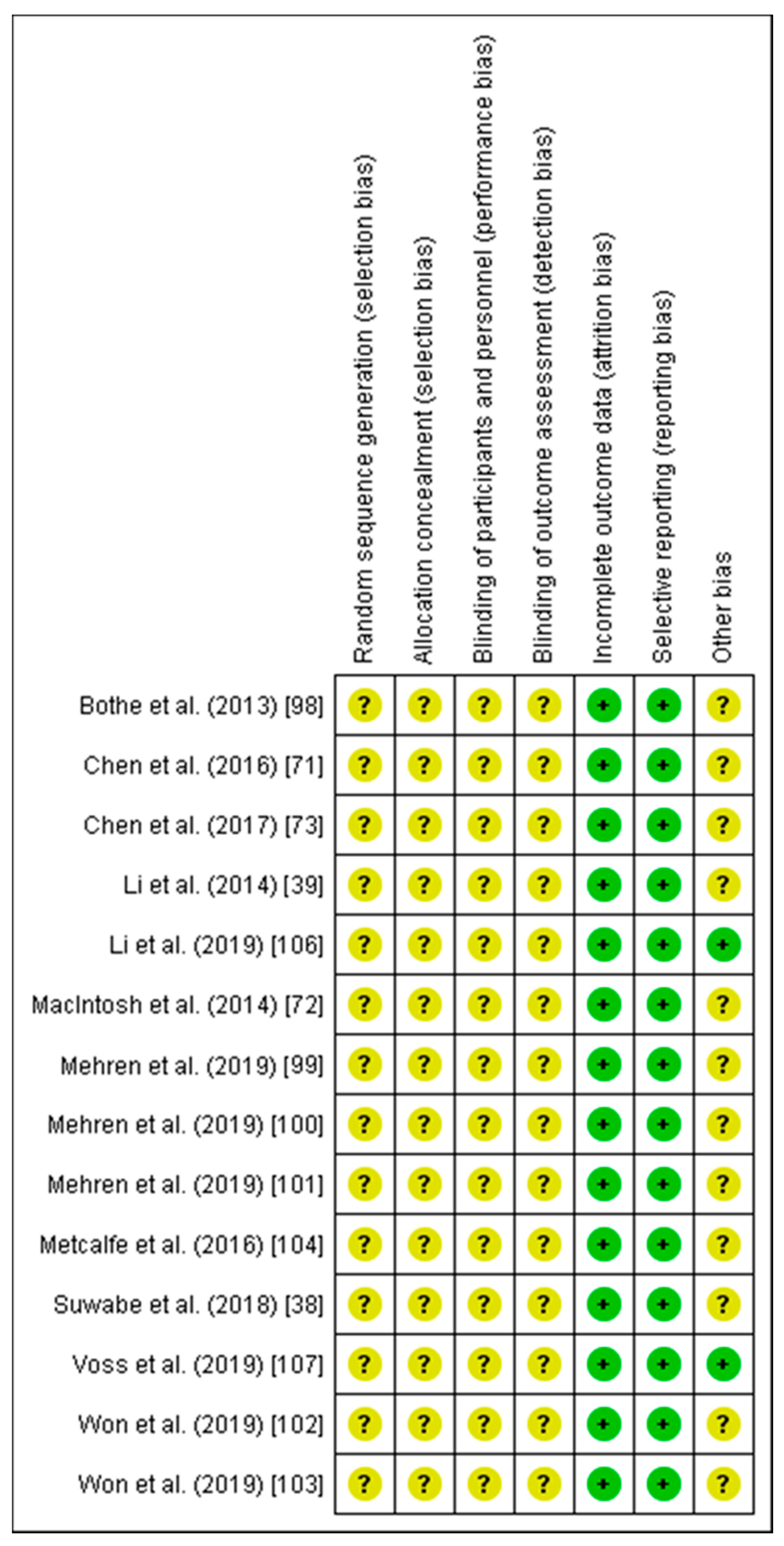
2.3. Risk of Bias Assessment
3. Results
3.1. Risk of Bias
3.2. Study Characteristics
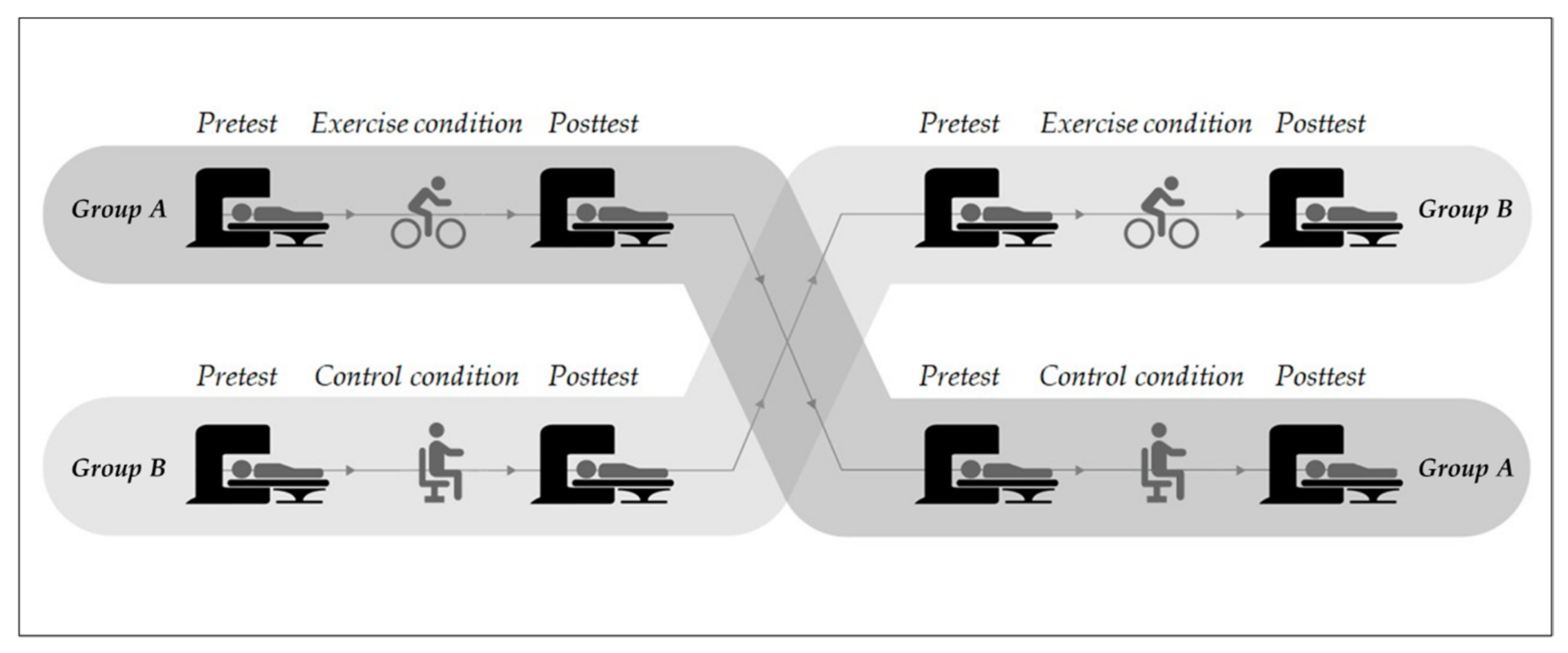
3.3. Exercise Characteristics
3.4. fMRI Characteristics and Data Processing
3.5. Findings
4. Discussion
4.1. Risk of Bias
4.2. Study Characteristics
4.3. Exercise Characteristics
4.4. Data Processing
4.5. Findings
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hill, J.O.; Peters, J.C. Environmental contributions to the obesity epidemic. Science 1998, 280, 1371–1374. [Google Scholar] [CrossRef]
- Bell, A.C.; Ge, K.; Popkin, B.M. The road to obesity or the path to prevention: Motorized transportation and obesity in China. Obes. Res. 2002, 10, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Fotheringham, M.J.; Wonnacott, R.L.; Owen, N. Computer use and physical inactivity in young adults: Public health perils and potentials of new information technologies. Ann. Behav. Med. 2000, 22, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.W. Trends in Job Demands among Older Workers, 1992–2002. Mon. Labor Rev. 2004, 127, 48–56. [Google Scholar]
- Owen, N.; Sparling, P.B.; Healy, G.N.; Dunstan, D.W.; Matthews, C.E. Sedentary behavior: Emerging evidence for a new health risk. Mayo Clin. Proc. 2010, 85, 1138–1141. [Google Scholar] [CrossRef]
- Church, T.S.; Thomas, D.M.; Tudor-Locke, C.; Katzmarzyk, P.T.; Earnest, C.P.; Rodarte, R.Q.; Martin, C.K.; Blair, S.N.; Bouchard, C. Trends over 5 decades in U.S. occupation-related physical activity and their associations with obesity. PLoS ONE 2011, 6, e19657. [Google Scholar] [CrossRef]
- Copeland, J.L.; Clarke, J.; Dogra, S. Objectively measured and self-reported sedentary time in older Canadians. Prev. Med. Rep. 2015, 2, 90–95. [Google Scholar] [CrossRef]
- Blair, S.N. Physical inactivity: The biggest public health problem of the 21st century. Br. J. Sports Med. 2009, 43, 1–2. [Google Scholar]
- Falck, R.S.; Davis, J.C.; Liu-Ambrose, T. What is the association between sedentary behaviour and cognitive function? A systematic review. Br. J. Sports Med. 2016, 51, 800–811. [Google Scholar] [CrossRef]
- Ku, P.-W.; Liu, Y.-T.; Lo, M.-K.; Chen, L.-J.; Stubbs, B. Higher levels of objectively measured sedentary behavior is associated with worse cognitive ability: Two-year follow-up study in community-dwelling older adults. Exp. Gerontol. 2017, 99, 110–114. [Google Scholar] [CrossRef]
- Tan, Z.S.; Spartano, N.L.; Beiser, A.S.; DeCarli, C.; Auerbach, S.H.; Vasan, R.S.; Seshadri, S. Physical Activity, Brain Volume, and Dementia Risk: The Framingham Study. J. Gerontol. A Biol. Sci. Med. Sci. 2017, 72, 789–795. [Google Scholar] [CrossRef]
- Aichberger, M.C.; Busch, M.A.; Reischies, F.M.; Ströhle, A.; Heinz, A.; Rapp, M.A. Effect of Physical Inactivity on Cognitive Performance after 2.5 Years of Follow-Up. GeroPsych 2010, 23, 7–15. [Google Scholar] [CrossRef]
- Laurin, D.; Verreault, R.; Lindsay, J.; MacPherson, K.; Rockwood, K. Physical Activity and Risk of Cognitive Impairment and Dementia in Elderly Persons. Arch. Neurol. 2001, 58, 498–504. [Google Scholar] [CrossRef] [PubMed]
- Bowen, M.E. A prospective examination of the relationship between physical activity and dementia risk in later life. Am. J. Health Promot. 2012, 26, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Grande, G.; Vanacore, N.; Maggiore, L.; Cucumo, V.; Ghiretti, R.; Galimberti, D.; Scarpini, E.; Mariani, C.; Clerici, F. Physical activity reduces the risk of dementia in mild cognitive impairment subjects: A cohort study. J. Alzheimers Dis. 2014, 39, 833–839. [Google Scholar] [CrossRef] [PubMed]
- Paillard-Borg, S.; Fratiglioni, L.; Winblad, B.; Wang, H.-X. Leisure activities in late life in relation to dementia risk: Principal component analysis. Dement. Geriatr. Cogn. Disord. 2009, 28, 136–144. [Google Scholar] [CrossRef]
- Rovio, S.; Kåreholt, I.; Helkala, E.-L.; Viitanen, M.; Winblad, B.; Tuomilehto, J.; Soininen, H.; Nissinen, A.; Kivipelto, M. Leisure-time physical activity at midlife and the risk of dementia and Alzheimer’s disease. Lancet Neurol. 2005, 4, 705–711. [Google Scholar] [CrossRef]
- Paillard, T. Preventive effects of regular physical exercise against cognitive decline and the risk of dementia with age advancement. Sports Med. Open 2015, 1, 4. [Google Scholar] [CrossRef]
- Buchman, A.S.; Boyle, P.A.; Yu, L.; Shah, R.C.; Wilson, R.S.; Bennett, D.A. Total daily physical activity and the risk of AD and cognitive decline in older adults. Neurology 2012, 78, 1323–1329. [Google Scholar] [CrossRef]
- Bruijn, R.F.A.G.; Schrijvers, E.M.C.; Groot, K.A.; Witteman, J.C.M.; Hofman, A.; Franco, O.H.; Koudstaal, P.J.; Ikram, M.A. The association between physical activity and dementia in an elderly population: The Rotterdam Study. Eur. J. Epidemiol. 2013, 28, 277–283. [Google Scholar] [CrossRef]
- Hamer, M.; Chida, Y. Physical activity and risk of neurodegenerative disease: A systematic review of prospective evidence. Psychol. Med. 2009, 39, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Ravaglia, G.; Forti, P.; Lucicesare, A.; Pisacane, N.; Rietti, E.; Bianchin, M.; Dalmonte, E. Physical activity and dementia risk in the elderly: Findings from a prospective Italian study. Neurology 2008, 70, 1786–1794. [Google Scholar] [CrossRef] [PubMed]
- Taylor, W.C. Transforming work breaks to promote health. Am. J. Prev. Med. 2005, 29, 461–465. [Google Scholar] [CrossRef] [PubMed]
- Bergouignan, A.; Legget, K.T.; Jong, N.; Kealey, E.; Nikolovski, J.; Groppel, J.L.; Jordan, C.; O’Day, R.; Hill, J.O.; Bessesen, D.H. Effect of frequent interruptions of prolonged sitting on self-perceived levels of energy, mood, food cravings and cognitive function. Int. J. Behav. Nutr. Phys. Act. 2016, 13, 113. [Google Scholar] [CrossRef]
- Panahi, S.; Tremblay, A. Sedentariness and Health: Is Sedentary Behavior More Than Just Physical Inactivity? Front. Public Health 2018, 6, 258. [Google Scholar] [CrossRef]
- Herold, F.; Hamacher, D.; Schega, L.; Müller, N.G. Thinking While Moving or Moving While Thinking-Concepts of Motor-Cognitive Training for Cognitive Performance Enhancement. Front. Aging Neurosci. 2018, 10, 228. [Google Scholar] [CrossRef]
- Budde, H.; Schwarz, R.; Velasques, B.; Ribeiro, P.; Holzweg, M.; Machado, S.; Brazaitis, M.; Staack, F.; Wegner, M. The need for differentiating between exercise, physical activity, and training. Autoimmun. Rev. 2016, 15, 110–111. [Google Scholar] [CrossRef]
- Caspersen, C.J.; Powell, K.E.; Christenson, G.M. Physical activity, exercise, and physical fitness: Definitions and distinctions for health-related research. Public Health Rep. 1985, 100, 126–131. [Google Scholar]
- Gronwald, T.; Budde, H. Commentary: Physical Exercise as Personalized Medicine for Dementia Prevention? Front. Physiol. 2019, 10, 726. [Google Scholar] [CrossRef]
- Chang, Y.K.; Labban, J.D.; Gapin, J.I.; Etnier, J.L. The effects of acute exercise on cognitive performance: A meta-analysis. Brain Res. 2012, 1453, 87–101. [Google Scholar] [CrossRef]
- Basso, J.C.; Suzuki, W.A. The Effects of Acute Exercise on Mood, Cognition, Neurophysiology, and Neurochemical Pathways: A Review. Brain Plast. 2017, 2, 127–152. [Google Scholar] [CrossRef] [PubMed]
- McSween, M.-P.; Coombes, J.S.; MacKay, C.P.; Rodriguez, A.D.; Erickson, K.I.; Copland, D.A.; McMahon, K.L. The Immediate Effects of Acute Aerobic Exercise on Cognition in Healthy Older Adults: A Systematic Review. Sports Med. 2018, 49, 67–82. [Google Scholar] [CrossRef] [PubMed]
- Pontifex, M.B.; McGowan, A.L.; Chandler, M.C.; Gwizdala, K.L.; Parks, A.C.; Fenn, K.; Kamijo, K. A primer on investigating the after effects of acute bouts of physical activity on cognition. Psychol. Sport Exerc. 2019, 40, 1–22. [Google Scholar] [CrossRef]
- Herold, F.; Wiegel, P.; Scholkmann, F.; Müller, N.G. Applications of Functional Near-Infrared Spectroscopy (fNIRS) Neuroimaging in Exercise⁻Cognition Science: A Systematic, Methodology-Focused Review. J. Clin. Med. 2018, 7, 466. [Google Scholar] [CrossRef]
- Törpel, A.; Herold, F.; Hamacher, D.; Müller, N.G.; Schega, L. Strengthening the Brain—Is Resistance Training with Blood Flow Restriction an Effective Strategy for Cognitive Improvement? J. Clin. Med. 2018, 7, 377. [Google Scholar] [CrossRef]
- Hillman, C.H.; Kamijo, K.; Pontifex, M.B. The Relation of ERP Indices of Exercise to Brain Health and Cognition. In Functional Neuroimaging in Exercise and Sport Sciences; Boecker, H., Hillman, C.H., Scheef, L., Strüder, H.K., Eds.; Springer: New York, NY, USA, 2012; pp. 419–446. ISBN 978-1-4614-3292-0. [Google Scholar]
- Chang, Y.-K. Acute Exercise and Event-Related Potential. In Exercise-Cognition Interaction; Elsevier: Amsterdam, The Netherlands, 2016; pp. 105–130. ISBN 9780128007785. [Google Scholar]
- Suwabe, K.; Byun, K.; Hyodo, K.; Reagh, Z.M.; Roberts, J.M.; Matsushita, A.; Saotome, K.; Ochi, G.; Fukuie, T.; Suzuki, K.; et al. Rapid stimulation of human dentate gyrus function with acute mild exercise. Proc. Natl. Acad. Sci. USA 2018, 115, 10487–10492. [Google Scholar] [CrossRef]
- Li, L.; Men, W.W.; Chang, Y.K.; Fan, M.X.; Ji, L.; Wei, G.X. Acute aerobic exercise increases cortical activity during working memory: A functional MRI study in female college students. PLoS ONE 2014, 9, e99222. [Google Scholar] [CrossRef]
- Herold, F.; Wiegel, P.; Scholkmann, F.; Thiers, A.; Hamacher, D.; Schega, L. Functional near-infrared spectroscopy in movement science: A systematic review on cortical activity in postural and walking tasks. Neurophoton 2017, 4, 41403. [Google Scholar] [CrossRef]
- Pinti, P.; Tachtsidis, I.; Hamilton, A.; Hirsch, J.; Aichelburg, C.; Gilbert, S.; Burgess, P.W. The present and future use of functional near-infrared spectroscopy (fNIRS) for cognitive neuroscience. Ann. N. Y. Acad. Sci. 2018. [Google Scholar] [CrossRef]
- Scarapicchia, V.; Brown, C.; Mayo, C.; Gawryluk, J.R. Functional Magnetic Resonance Imaging and Functional Near-Infrared Spectroscopy: Insights from Combined Recording Studies. Front. Hum. Neurosci. 2017, 11, 419. [Google Scholar] [CrossRef]
- Soltanlou, M.; Sitnikova, M.A.; Nuerk, H.-C.; Dresler, T. Applications of Functional Near-Infrared Spectroscopy (fNIRS) in Studying Cognitive Development: The Case of Mathematics and Language. Front. Psychol. 2018, 9, 277. [Google Scholar] [CrossRef] [PubMed]
- Glover, G.H. Overview of functional magnetic resonance imaging. Neurosurg. Clin. N. Am. 2011, 22, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Voss, J.L.; Bridge, D.J.; Cohen, N.J.; Walker, J.A. A Closer Look at the Hippocampus and Memory. Trends Cogn. Sci. 2017, 21, 577–588. [Google Scholar] [CrossRef] [PubMed]
- Knierim, J.J. The hippocampus. Curr. Biol. 2015, 25, R1116-21. [Google Scholar] [CrossRef] [PubMed]
- Bird, C.M.; Burgess, N. The hippocampus and memory: Insights from spatial processing. Nat. Rev. Neurosci. 2008, 9, 182–194. [Google Scholar] [CrossRef] [PubMed]
- Maguire, E.A.; Frackowiak, R.S.J.; Frith, C.D. Recalling Routes around London: Activation of the Right Hippocampus in Taxi Drivers. J. Neurosci. 1997, 17, 7103–7110. [Google Scholar] [CrossRef]
- Squire, L.R.; Ojemann, J.G.; Miezin, F.M.; Petersen, S.E.; Videen, T.O.; Raichle, M.E. Activation of the hippocampus in normal humans: A functional anatomical study of memory. Proc. Natl. Acad. Sci. USA 1992, 89, 1837–1841. [Google Scholar] [CrossRef]
- O’Brien, J.L.; O’Keefe, K.M.; LaViolette, P.S.; DeLuca, A.N.; Blacker, D.; Dickerson, B.C.; Sperling, R.A. Longitudinal fMRI in elderly reveals loss of hippocampal activation with clinical decline. Neurology 2010, 74, 1969–1976. [Google Scholar] [CrossRef]
- Johnson, S.C.; Schmitz, T.W.; Moritz, C.H.; Meyerand, M.E.; Rowley, H.A.; Alexander, A.L.; Hansen, K.W.; Gleason, C.E.; Carlsson, C.M.; Ries, M.L.; et al. Activation of brain regions vulnerable to Alzheimer’s disease: The effect of mild cognitive impairment. Neurobiol. Aging 2006, 27, 1604–1612. [Google Scholar] [CrossRef]
- Small, S.A.; Perera, G.M.; DeLapaz, R.; Mayeux, R.; Stern, Y. Differential regional dysfunction of the hippocampal formation among elderly with memory decline and Alzheimer’s disease. Ann. Neurol. 1999, 45, 466–472. [Google Scholar] [CrossRef]
- Ogawa, S.; Lee, T.M.; Kay, A.R.; Tank, D.W. Brain magnetic resonance imaging with contrast dependent on blood oxygenation. Proc. Natl. Acad. Sci. USA 1990, 87, 9868–9872. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, S.; Lee, T.M.; Nayak, A.S.; Glynn, P. Oxygenation-sensitive contrast in magnetic resonance image of rodent brain at high magnetic fields. Magn. Reson. Med. 1990, 14, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, S.; Tank, D.W.; Menon, R.; Ellermann, J.M.; Kim, S.G.; Merkle, H.; Ugurbil, K. Intrinsic signal changes accompanying sensory stimulation: Functional brain mapping with magnetic resonance imaging. Proc. Natl. Acad. Sci. USA 1992, 89, 5951–5955. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, S. Finding the BOLD effect in brain images. NeuroImage 2012, 62, 608–609. [Google Scholar] [CrossRef]
- Gore, J.C. Principles and practice of functional MRI of the human brain. J. Clin. Investig. 2003, 112, 4–9. [Google Scholar] [CrossRef]
- Norris, D.G. Principles of magnetic resonance assessment of brain function. J. Magn. Reson. Imaging 2006, 23, 794–807. [Google Scholar] [CrossRef]
- Kim, S.-G.; Ogawa, S. Biophysical and physiological origins of blood oxygenation level-dependent fMRI signals. J. Cereb. Blood Flow Metab. 2012, 32, 1188–1206. [Google Scholar] [CrossRef]
- Logothetis, N.K. What we can do and what we cannot do with fMRI. Nature 2008, 453, 869–878. [Google Scholar] [CrossRef]
- Logothetis, N.K.; Pfeuffer, J. On the nature of the BOLD fMRI contrast mechanism. Magn. Reson. Imaging 2004, 22, 1517–1531. [Google Scholar] [CrossRef]
- Soares, J.M.; Magalhães, R.; Moreira, P.S.; Sousa, A.; Ganz, E.; Sampaio, A.; Alves, V.; Marques, P.; Sousa, N. A Hitchhiker’s Guide to Functional Magnetic Resonance Imaging. Front. Neurosci. 2016, 10, 515. [Google Scholar] [CrossRef]
- Francis, S.; Panchuelo, R.S. Physiological measurements using ultra-high field fMRI: A review. Physiol. Meas. 2014, 35, R167-85. [Google Scholar] [CrossRef] [PubMed]
- Heeger, D.J.; Ress, D. What does fMRI tell us about neuronal activity? Nat. Rev. Neurosci. 2002, 3, 142–151. [Google Scholar] [CrossRef]
- Cinciute, S. Translating the hemodynamic response: Why focused interdisciplinary integration should matter for the future of functional neuroimaging. PeerJ 2019, 7, e6621. [Google Scholar] [CrossRef] [PubMed]
- Huneau, C.; Benali, H.; Chabriat, H. Investigating Human Neurovascular Coupling Using Functional Neuroimaging: A Critical Review of Dynamic Models. Front. Neurosci. 2015, 9, e1002435. [Google Scholar] [CrossRef] [PubMed]
- Leybaert, L. Neurobarrier coupling in the brain: A partner of neurovascular and neurometabolic coupling? J. Cereb. Blood Flow Metab. 2005, 25, 2–16. [Google Scholar] [CrossRef] [PubMed]
- Liao, L.-D.; Tsytsarev, V.; Delgado-Martínez, I.; Li, M.-L.; Erzurumlu, R.; Vipin, A.; Orellana, J.; Lin, Y.-R.; Lai, H.-Y.; Chen, Y.-Y.; et al. Neurovascular coupling: In vivo optical techniques for functional brain imaging. BioMed. Eng. OnLine 2013, 12, 38. [Google Scholar] [CrossRef]
- Nippert, A.R.; Biesecker, K.R.; Newman, E.A. Mechanisms Mediating Functional Hyperemia in the Brain. Neuroscientist 2018, 24, 73–83. [Google Scholar] [CrossRef]
- Minzenberg, M.J. Pharmacological MRI approaches to understanding mechanisms of drug action. Curr. Top. Behav. Neurosci. 2012, 11, 365–388. [Google Scholar] [CrossRef]
- Chen, A.-G.; Zhu, L.-N.; Yan, J.; Yin, H.-C. Neural Basis of Working Memory Enhancement after Acute Aerobic Exercise: FMRI Study of Preadolescent Children. Front. Psychol. 2016, 7, 1804. [Google Scholar] [CrossRef]
- MacIntosh, B.J.; Crane, D.E.; Sage, M.D.; Rajab, A.S.; Donahue, M.J.; McIlroy, W.E.; Middleton, L.E. Impact of a single bout of aerobic exercise on regional brain perfusion and activation responses in healthy young adults. PLoS ONE 2014, 9, e85163. [Google Scholar] [CrossRef]
- Chen, A.-G.; Zhu, L.-N.; Xiong, X.; Li, Y. Acute aerobic exercise alters executive control network in preadolescent children. J. Sport Psychol. 2017, 26, 132–137. [Google Scholar]
- Amaro, E.; Barker, G.J. Study design in fMRI: Basic principles. Brain Cogn. 2006, 60, 220–232. [Google Scholar] [CrossRef] [PubMed]
- Poldrack, R.A.; Fletcher, P.C.; Henson, R.N.; Worsley, K.J.; Brett, M.; Nichols, T.E. Guidelines for reporting an fMRI study. Neuroimage 2008, 40, 409–414. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. J. Clin. Epidemiol. 2009, 62, 1006–1012. [Google Scholar] [CrossRef]
- Harris, J.D.; Quatman, C.E.; Manring, M.M.; Siston, R.A.; Flanigan, D.C. How to write a systematic review. Am. J. Sports Med. 2014, 42, 2761–2768. [Google Scholar] [CrossRef]
- Bostrom, N.; Sandberg, A. Cognitive enhancement: Methods, ethics, regulatory challenges. Sci. Eng. Ethics 2009, 15, 311–341. [Google Scholar] [CrossRef]
- Borson, S. Cognition, aging, and disabilities: Conceptual issues. Phys. Med. Rehabil. Clin. N. Am. 2010, 21, 375–382. [Google Scholar] [CrossRef]
- Gothe, N.; Pontifex, M.B.; Hillman, C.; McAuley, E. The acute effects of yoga on executive function. J. Phys. Act. Health 2013, 10, 488–495. [Google Scholar] [CrossRef]
- Chang, Y.-K.; Tsai, C.-L.; Hung, T.-M.; So, E.C.; Chen, F.-T.; Etnier, J.L. Effects of Acute Exercise on Executive Function: A Study with a Tower of London Task. J. Sport Exerc. Psychol. 2011, 33, 847–865. [Google Scholar] [CrossRef]
- Won, J.; Alfini, A.J.; Weiss, L.R.; Nyhuis, C.C.; Spira, A.P.; Callow, D.D.; Carson Smith, J. Caudate Volume Mediates the Interaction between Total Sleep Time and Executive Function after Acute Exercise in Healthy Older Adults. Brain Plast. 2019, 5, 69–82. [Google Scholar] [CrossRef]
- Faulkner, J.; Lambrick, D.; Kaufmann, S.; Stoner, L. Effects of Upright and Recumbent Cycling on Executive Function and Prefrontal Cortex Oxygenation in Young, Healthy, Men. J. Phys. Act. Health 2016, 13, 882–887. [Google Scholar] [CrossRef] [PubMed]
- Faulkner, J.; Stoner, L.; Grigg, R.; Fryer, S.; Stone, K.; Lambrick, D. Acute effects of exercise posture on executive function in transient ischemic attack patients. Psychophysiology 2017, 54, 1239–1248. [Google Scholar] [CrossRef] [PubMed]
- Lambrick, D.; Stoner, L.; Grigg, R.; Faulkner, J. Effects of continuous and intermittent exercise on executive function in children aged 8–10 years. Psychophysiology 2016, 53, 1335–1342. [Google Scholar] [CrossRef] [PubMed]
- Pontifex, M.B.; Gwizdala, K.L.; WENG, T.B.; Zhu, D.C.; Voss, M.W. Cerebral blood flow is not modulated following acute aerobic exercise in preadolescent children. Int. J. Psychophysiol. 2018, 134, 44–51. [Google Scholar] [CrossRef]
- Rajab, A.S.; Crane, D.E.; Middleton, L.E.; Robertson, A.D.; Hampson, M.; MacIntosh, B.J. A single session of exercise increases connectivity in sensorimotor-related brain networks: A resting-state fMRI study in young healthy adults. Front. Hum. Neurosci. 2014, 8, 625. [Google Scholar] [CrossRef]
- Scheef, L.; Jankowski, J.; Daamen, M.; Weyer, G.; Klingenberg, M.; Renner, J.; Mueckter, S.; Schürmann, B.; Musshoff, F.; Wagner, M.; et al. An fMRI study on the acute effects of exercise on pain processing in trained athletes. Pain 2012, 153, 1702–1714. [Google Scholar] [CrossRef]
- Schmitt, A.; Martin, J.A.; Rojas, S.; Vafa, R.; Scheef, L.; Strüder, H.K.; Boecker, H. Effects of low- and high-intensity exercise on emotional face processing: An fMRI face-matching study. Soc. Cogn. Affect. Neurosci. 2019, 14, 657–665. [Google Scholar] [CrossRef]
- Weng, T.B.; Pierce, G.L.; Darling, W.G.; Falk, D.; Magnotta, V.A.; Voss, M.W. The Acute Effects of Aerobic Exercise on the Functional Connectivity of Human Brain Networks. Brain Plast. 2017, 2, 171–190. [Google Scholar] [CrossRef]
- van Janse Rensburg, K.; Taylor, A.; Hodgson, T.; Benattayallah, A. Acute exercise modulates cigarette cravings and brain activation in response to smoking-related images: An fMRI study. Psychopharmacology 2009, 203, 589–598. [Google Scholar] [CrossRef]
- Masterson, T.D.; Kirwan, C.B.; Davidson, L.E.; Larson, M.J.; Keller, K.L.; Fearnbach, S.N.; Evans, A.; LeCheminant, J.D. Brain reactivity to visual food stimuli after moderate-intensity exercise in children. Brain Imaging Behav. 2018, 12, 1032–1041. [Google Scholar] [CrossRef]
- Chen, Y.-C.; Chen, C.; Martínez, R.M.; Etnier, J.L.; Cheng, Y. Habitual physical activity mediates the acute exercise-induced modulation of anxiety-related amygdala functional connectivity. Sci. Rep. 2019, 9, 19787. [Google Scholar] [CrossRef]
- Schmitt, A.; Upadhyay, N.; Martin, J.A.; Rojas, S.; Strüder, H.K.; Boecker, H. Modulation of Distinct Intrinsic Resting State Brain Networks by Acute Exercise Bouts of Differing Intensity. Brain Plast. 2019, 5, 39–55. [Google Scholar] [CrossRef] [PubMed]
- Himmelmeier, R.M.; Nouchi, R.; Saito, T.; Burin, D.; Wiltfang, J.; Kawashima, R. Study Protocol: Does an Acute Intervention of High-Intensity Physical Exercise Followed by a Brain Training Video Game Have Immediate Effects on Brain Activity of Older People During Stroop Task in fMRI?-A Randomized Controlled Trial With Crossover Design. Front. Aging Neurosci. 2019, 11, 260. [Google Scholar] [CrossRef]
- Tzourio-Mazoyer, N.; Landeau, B.; Papathanassiou, D.; Crivello, F.; Etard, O.; Delcroix, N.; Mazoyer, B.; Joliot, M. Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. NeuroImage 2002, 15, 273–289. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions. A Cochrane Pocketbook; John Wiley & Sons, Ltd.: Chichester, UK, 2008; ISBN 978-0470699515. [Google Scholar]
- The Cochrane Collaboration. Review Manager (RevMan 5.3) [Computer program]. Available online: https://community.cochrane.org/help/tools-and-software/revman-5 (accessed on 1 April 2019).
- Bothe, N.; Zschucke, E.; Dimeo, F.; Heinz, A.; Wüstenberg, T.; Ströhle, A. Acute exercise influences reward processing in highly trained and untrained men. Med. Sci. Sports Exerc. 2013, 45, 583–591. [Google Scholar] [CrossRef] [PubMed]
- Mehren, A.; Özyurt, J.; Thiel, C.M.; Brandes, M.; Lam, A.P.; Philipsen, A. Effects of Acute Aerobic Exercise on Response Inhibition in Adult Patients with ADHD. Sci. Rep. 2019, 9, 65. [Google Scholar] [CrossRef]
- Mehren, A.; Diaz Luque, C.; Brandes, M.; Lam, A.P.; Thiel, C.M.; Philipsen, A.; Özyurt, J. Intensity-Dependent Effects of Acute Exercise on Executive Function. Neural Plast. 2019, 2019, 8608317. [Google Scholar] [CrossRef]
- Mehren, A.; Özyurt, J.; Lam, A.P.; Brandes, M.; Müller, H.H.O.; Thiel, C.M.; Philipsen, A. Acute Effects of Aerobic Exercise on Executive Function and Attention in Adult Patients With ADHD. Front. Psychiatry 2019, 10, 132. [Google Scholar] [CrossRef]
- Won, J.; Alfini, A.J.; Weiss, L.R.; Callow, D.D.; Smith, J.C. Brain activation during executive control after acute exercise in older adults. Int. J. Psychophysiol. 2019, 146, 240–248. [Google Scholar] [CrossRef]
- Won, J.; Alfini, A.J.; Weiss, L.R.; Michelson, C.S.; Callow, D.D.; Ranadive, S.M.; Gentili, R.J.; Smith, J.C. Semantic Memory Activation After Acute Exercise in Healthy Older Adults. J. Int. Neuropsychol. Soc. 2019, 25, 557–568. [Google Scholar] [CrossRef]
- Metcalfe, A.W.S.; MacIntosh, B.J.; Scavone, A.; Ou, X.; Korczak, D.; Goldstein, B.I. Effects of acute aerobic exercise on neural correlates of attention and inhibition in adolescents with bipolar disorder. Transl. Psychiatry 2016, 6, e814. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Zhang, S.; Cui, J.; Chen, L.-Z.; Wang, X.; Fan, M.; Wei, G.-X. Fitness-Dependent Effect of Acute Aerobic Exercise on Executive Function. Front. Physiol. 2019, 10, 731. [Google Scholar] [CrossRef] [PubMed]
- Voss, M.W.; Weng, T.B.; Narayana-Kumanan, K.; Cole, R.C.; Wharff, C.; Reist, L.; DuBose, L.; Sigurdsson, G.; Mills, J.A.; Long, J.D.; et al. Acute Exercise Effects Predict Training Change in Cognition and Connectivity. Med. Sci. Sports Exerc. 2019. [Google Scholar] [CrossRef] [PubMed]
- Borg, G.A. Psychophysical bases of perceived exertion. Med. Sci. Sports Exerc. 1982, 14, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Day, J.R.; Rossiter, H.B.; Coats, E.M.; Skasick, A.; Whipp, B.J. The maximally attainable VO2 during exercise in humans: The peak vs. maximum issue. J. Appl. Physiol. 2003, 95, 1901–1907. [Google Scholar] [CrossRef]
- Poole, D.C.; Jones, A.M. Measurement of the maximum oxygen uptake o2max: o2peak is no longer acceptable. J. Appl. Physiol. 2017, 122, 997–1002. [Google Scholar] [CrossRef]
- Midgley, A.W.; McNaughton, L.R.; Polman, R.; Marchant, D. Criteria for determination of maximal oxygen uptake: A brief critique and recommendations for future research. Sports Med. 2007, 37, 1019–1028. [Google Scholar] [CrossRef]
- Schulz, K.F.; Altman, D.G.; Moher, D.; Group, C. CONSORT 2010 Statement: Updated guidelines for reporting parallel group randomised trials. BMC Med. 2010, 8, 18. [Google Scholar] [CrossRef]
- Dwan, K.; Li, T.; Altman, D.G.; Elbourne, D. CONSORT 2010 statement: Extension to randomised crossover trials. BMJ 2019, 366, l4378. [Google Scholar] [CrossRef]
- Lindheimer, J.B.; Szabo, A.; Raglin, J.S.; Beedie, C. Advancing the understanding of placebo effects in psychological outcomes of exercise: Lessons learned and future directions. Eur. J. Sport Sci. 2019, 1–28. [Google Scholar] [CrossRef]
- Lindheimer, J.B.; Szabo, A.; Raglin, J.S.; Beedie, C.; Carmichael, K.E.; O’Connor, P.J. Reconceptualizing the measurement of expectations to better understand placebo and nocebo effects in psychological responses to exercise. Eur. J. Sport Sci. 2019, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Beedie, C.; Benedetti, F.; Barbiani, D.; Camerone, E.; Cohen, E.; Coleman, D.; Davis, A.; Elsworth-Edelsten, C.; Flowers, E.; Foad, A.; et al. Consensus statement on placebo effects in sports and exercise: The need for conceptual clarity, methodological rigour, and the elucidation of neurobiological mechanisms. Eur. J. Sport Sci. 2018, 18, 1383–1389. [Google Scholar] [CrossRef] [PubMed]
- Boot, W.R.; Simons, D.J.; Stothart, C.; Stutts, C. The Pervasive Problem With Placebos in Psychology: Why Active Control Groups Are Not Sufficient to Rule Out Placebo Effects. Perspect. Psychol. Sci. 2013, 8, 445–454. [Google Scholar] [CrossRef] [PubMed]
- Shawn Green, C.; Bavelier, D.; Kramer, A.F.; Vinogradov, S.; Ansorge, U.; Ball, K.K.; Bingel, U.; Chein, J.M.; Colzato, L.S.; Edwards, J.D.; et al. Improving Methodological Standards in Behavioral Interventions for Cognitive Enhancement. J. Cogn. Enhanc. 2019, 33, 316. [Google Scholar] [CrossRef]
- Lange, A.K.; Vanwanseele, B.; Foroughi, N.; Baker, M.K.; Shnier, R.; Smith, R.M.; Singh, M.A.F. Resistive Exercise for Arthritic Cartilage Health (REACH): A randomized double-blind, sham-exercise controlled trial. BMC Geriatr. 2009, 9, 1. [Google Scholar] [CrossRef]
- Vonk, M.; Wikkerink, S.; Regan, K.; Middleton, L.E. Similar changes in executive function after moderate resistance training and loadless movement. PLoS ONE 2019, 14, e0212122. [Google Scholar] [CrossRef]
- McCaskey, M.A.; Wirth, B.; Schuster-Amft, C.; Bruin, E.D. Postural sensorimotor training versus sham exercise in physiotherapy of patients with chronic non-specific low back pain: An exploratory randomised controlled trial. PLoS ONE 2018, 13, e0193358. [Google Scholar] [CrossRef]
- Alhola, P.; Polo-Kantola, P. Sleep deprivation: Impact on cognitive performance. Neuropsychiatr. Dis. Treat. 2007, 3, 553–567. [Google Scholar]
- Lo, J.C.; Groeger, J.A.; Santhi, N.; Arbon, E.L.; Lazar, A.S.; Hasan, S.; Schantz, M.; Archer, S.N.; Dijk, D.-J. Effects of partial and acute total sleep deprivation on performance across cognitive domains, individuals and circadian phase. PLoS ONE 2012, 7, e45987. [Google Scholar] [CrossRef]
- Nilsson, J.P.; Söderström, M.; Karlsson, A.U.; Lekander, M.; Akerstedt, T.; Lindroth, N.E.; Axelsson, J. Less effective executive functioning after one night’s sleep deprivation. J. Sleep Res. 2005, 14, 1–6. [Google Scholar] [CrossRef]
- Randazzo, A.C.; Muehlbach, M.J.; Schweitzer, P.K.; Walsh, J.K. Cognitive function following acute sleep restriction in children ages 10–14. Sleep 1998, 21, 861–868. [Google Scholar] [PubMed]
- Chee, M.W.L.; Chuah, L.Y.M. Functional neuroimaging insights into how sleep and sleep deprivation affect memory and cognition. Curr. Opin. Neurol. 2008, 21, 417–423. [Google Scholar] [CrossRef] [PubMed]
- Drummond, S.P.; Gillin, J.C.; Brown, G.G. Increased cerebral response during a divided attention task following sleep deprivation. J. Sleep Res. 2001, 10, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Killgore, W.D.S.; Weber, M. Sleep Deprivation and Cognitive Performance. In Sleep Deprivation and Disease; Bianchi, M.T., Ed.; Springer: New York, NY, USA, 2014; pp. 209–229. ISBN 978-1-4614-9086-9. [Google Scholar]
- Petersen, S.E.; Dubis, J.W. The mixed block/event-related design. NeuroImage 2012, 62, 1177–1184. [Google Scholar] [CrossRef] [PubMed]
- Meltzer, J.A.; Negishi, M.; Constable, R.T. Biphasic hemodynamic responses influence deactivation and may mask activation in block-design fMRI paradigms. Hum. Brain Mapp. 2008, 29, 385–399. [Google Scholar] [CrossRef] [PubMed]
- Dale, A.M.; Buckner, R.L. Selective averaging of rapidly presented individual trials using fMRI. Hum. Brain Mapp. 1997, 5, 329–340. [Google Scholar] [CrossRef]
- Miezin, F.M.; Maccotta, L.; Ollinger, J.M.; Petersen, S.E.; Buckner, R.L. Characterizing the hemodynamic response: Effects of presentation rate, sampling procedure, and the possibility of ordering brain activity based on relative timing. Neuroimage 2000, 11, 735–759. [Google Scholar] [CrossRef]
- Donaldson, D.I. Parsing brain activity with fMRI and mixed designs: What kind of a state is neuroimaging in? Trends Neurosci. 2004, 27, 442–444. [Google Scholar] [CrossRef]
- Behroozi, M.; Reza Daliri, M.; Boyaci, H. Statistical Analysis Methods for the fMRI Data. Basic Clin. Neurosci. 2011, 2, 67–74. [Google Scholar]
- Otten, L.J.; Henson, R.N.A.; Rugg, M.D. State-related and item-related neural correlates of successful memory encoding. Nat. Neurosci. 2002, 5, 1339–1344. [Google Scholar] [CrossRef]
- Wasfy, M.M.; Baggish, A.L. Exercise Dose in Clinical Practice. Circulation 2016, 133, 2297–2313. [Google Scholar] [CrossRef] [PubMed]
- Gronwald, T.; Bem Alves, A.C.; Murillo-Rodríguez, E.; Latini, A.; Schuette, J.; Budde, H. Standardization of exercise intensity and consideration of a dose-response is essential. Commentary on “Exercise-linked FNDC5/irisin rescues synaptic plasticity and memory defects in Alzheimer’s models”, by Lourenco et al., published 2019 in Nature Medicine. J. Sport Health Sci. 2019, 8, 353–354. [Google Scholar] [CrossRef] [PubMed]
- Herold, F.; Müller, P.; Gronwald, T.; Müller, N.G. Dose-response matters–A perspective on the exercise prescription in exercise-cognition research. Front. Psychol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Impellizzeri, F.M.; Marcora, S.M.; Coutts, A.J. Internal and External Training Load: 15 Years On. Int. J. Sports Physiol. Perform. 2019, 14, 270–273. [Google Scholar] [CrossRef] [PubMed]
- Vanrenterghem, J.; Nedergaard, N.J.; Robinson, M.A.; Drust, B. Training Load Monitoring in Team Sports: A Novel Framework Separating Physiological and Biomechanical Load-Adaptation Pathways. Sports Med. 2017, 47, 2135–2142. [Google Scholar] [CrossRef] [PubMed]
- Wallace, L.K.; Slattery, K.M.; Coutts, A.J. The ecological validity and application of the session-RPE method for quantifying training loads in swimming. J. Strength Cond. Res. 2009, 23, 33–38. [Google Scholar] [CrossRef]
- Halson, S.L. Monitoring training load to understand fatigue in athletes. Sports Med. 2014, 44 (Suppl. 2), S139–S147. [Google Scholar] [CrossRef]
- Bourdon, P.C.; Cardinale, M.; Murray, A.; Gastin, P.; Kellmann, M.; Varley, M.C.; Gabbett, T.J.; Coutts, A.J.; Burgess, D.J.; Gregson, W.; et al. Monitoring Athlete Training Loads: Consensus Statement. Int. J. Sports Physiol. Perform. 2017, 12, S2-161–S2-170. [Google Scholar] [CrossRef]
- Burgess, D.J. The Research Doesn’t Always Apply: Practical Solutions to Evidence-Based Training-Load Monitoring in Elite Team Sports. Int. J. Sports Physiol. Perform. 2017, 12, S2136–S2141. [Google Scholar] [CrossRef]
- McLaren, S.J.; Macpherson, T.W.; Coutts, A.J.; Hurst, C.; Spears, I.R.; Weston, M. The Relationships between Internal and External Measures of Training Load and Intensity in Team Sports: A Meta-Analysis. Sports Med. 2018, 48, 641–658. [Google Scholar] [CrossRef]
- Hillman, C.H.; Erickson, K.I.; Kramer, A.F. Be smart, exercise your heart: Exercise effects on brain and cognition. Nat. Rev. Neurosci. 2008, 9, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Soga, K.; Masaki, H.; Gerber, M.; Ludyga, S. Acute and Long-term Effects of Resistance Training on Executive Function. J. Cogn. Enhanc. 2018, 56, 729. [Google Scholar] [CrossRef]
- Wilke, J.; Giesche, F.; Klier, K.; Vogt, L.; Herrmann, E.; Banzer, W. Acute Effects of Resistance Exercise on Cognitive Function in Healthy Adults: A Systematic Review with Multilevel Meta-Analysis. Sports Med. 2019, 49, 905–916. [Google Scholar] [CrossRef] [PubMed]
- Budde, H.; Voelcker-Rehage, C.; Pietrabyk-Kendziorra, S.; Ribeiro, P.; Tidow, G. Acute coordinative exercise improves attentional performance in adolescents. Neurosci. Lett. 2008, 441, 219–223. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, S.; Grove, P.M. Comparison of the effects of running and badminton on executive function: A within-subjects design. PLoS ONE 2019, 14, e0216842. [Google Scholar] [CrossRef] [PubMed]
- Zach, S.; Shalom, E. The Influence of Acute Physical Activity on Working Memory. Percept. Mot. Ski. 2016, 122, 365–374. [Google Scholar] [CrossRef] [PubMed]
- Arena, R.; Myers, J.; Kaminsky, L.A. Revisiting age-predicted maximal heart rate: Can it be used as a valid measure of effort? Am. Heart J. 2016, 173, 49–56. [Google Scholar] [CrossRef]
- Correa Mesa, J.F.; Cruz Martinez, L.E.; Correa Morales, J.C.; Rojas Valencia, J.T. Maximum Heart Rate during exercise: Reliability of the 220-age and Tanaka formulas in healthy young people at a moderate altitude. Rev. Fac. Med. 2015, 62, 579–585. [Google Scholar] [CrossRef]
- Esco, M.R.; Chamberlain, N.; Flatt, A.A.; Snarr, R.L.; Bishop, P.A.; Williford, H.N. Cross-Validation of Age-Predicted Maximal Heart Rate Equations Among Female Collegiate Athletes. J. Strength Cond. Res. 2015, 29, 3053–3059. [Google Scholar] [CrossRef]
- Robergs, R.A.; Landwehr, R. The surprising history of the “HRmax = 220-age” equiation. J. Exerc. Physiol. Online 2002, 5, 1–10. [Google Scholar]
- Sarzynski, M.A.; Rankinen, T.; Earnest, C.P.; Leon, A.S.; Rao, D.C.; Skinner, J.S.; Bouchard, C. Measured maximal heart rates compared to commonly used age-based prediction equations in the Heritage Family Study. Am. J. Hum. Biol. 2013, 25, 695–701. [Google Scholar] [CrossRef] [PubMed]
- Silva, V.A.P.; Bottaro, M.; Justino, M.A.; Ribeiro, M.M.; Lima, R.M.; Oliveira, R.J. Maximum Heart Rate in Brazilian Elderly Women: Comparing Measured and Predicted Values. Arq. Bras. Cardiol. 2007, 88, 147–162. [Google Scholar] [CrossRef]
- Strzelczyk, T.A.; Quigg, R.J.; Pfeifer, P.B.; Parker, M.A.; Greenland, P. Accuracy of estimating exercise prescription intensity in patients with left ventricular systolic dysfunction. J. Cardiopulm. Rehabil. 2001, 21, 158–163. [Google Scholar] [CrossRef] [PubMed]
- Gronwald, T.; Velasques, B.; Ribeiro, P.; Machado, S.; Murillo-Rodríguez, E.; Ludyga, S.; Yamamoto, T.; Budde, H. Increasing exercise’s effect on mental health: Exercise intensity does matter. Proc. Natl. Acad. Sci. USA 2018, 115, E11890–E11891. [Google Scholar] [CrossRef]
- Murphy, K.; Birn, R.M.; Bandettini, P.A. Resting-state fMRI confounds and cleanup. Neuroimage 2013, 80, 349–359. [Google Scholar] [CrossRef]
- Liu, T.T. Noise contributions to the fMRI signal: An overview. Neuroimage 2016, 143, 141–151. [Google Scholar] [CrossRef]
- Peeters, R.; Sunaert, S. Clinical BOLD fMRI: Artifacts, Tips and Tricks. In Clinical Functional MRI; Springer: Berlin/Heidelberg, Germany, 2007; Volume 23, pp. 227–249. [Google Scholar] [CrossRef]
- Peeters, R.; Sunaert, S. Clinical BOLD fMRI and DTI: Artifacts, Tips, and Tricks. In Clinical Functional MRI; Stippich, C., Ed.; Springer: Berlin/Heidelberg, Germany, 2015; pp. 313–336. ISBN 978-3-662-45122-9. [Google Scholar]
- Caballero-Gaudes, C.; Reynolds, R.C. Methods for cleaning the BOLD fMRI signal. Neuroimage 2017, 154, 128–149. [Google Scholar] [CrossRef]
- Hutton, C.; Josephs, O.; Stadler, J.; Featherstone, E.; Reid, A.; Speck, O.; Bernarding, J.; Weiskopf, N. The impact of physiological noise correction on fMRI at 7 T. Neuroimage 2011, 57, 101–112. [Google Scholar] [CrossRef]
- Bulte, D.; Wartolowska, K. Monitoring cardiac and respiratory physiology during FMRI. Neuroimage 2017, 154, 81–91. [Google Scholar] [CrossRef]
- Özbay, P.S.; Chang, C.; Picchioni, D.; Mandelkow, H.; Chappel-Farley, M.G.; van Gelderen, P.; Zwart, J.A.; Duyn, J. Sympathetic activity contributes to the fMRI signal. Commun. Biol. 2019, 2, 421. [Google Scholar] [CrossRef]
- Strother, S.C. Evaluating fMRI preprocessing pipelines. IEEE Eng. Med. Biol. Mag. 2006, 25, 27–41. [Google Scholar] [CrossRef] [PubMed]
- Power, J.D.; Schlaggar, B.L.; Petersen, S.E. Recent progress and outstanding issues in motion correction in resting state fMRI. Neuroimage 2015, 105, 536–551. [Google Scholar] [CrossRef] [PubMed]
- Power, J.D.; Mitra, A.; Laumann, T.O.; Snyder, A.Z.; Schlaggar, B.L.; Petersen, S.E. Methods to detect, characterize, and remove motion artifact in resting state fMRI. Neuroimage 2014, 84, 320–341. [Google Scholar] [CrossRef]
- Gorgolewski, K.J.; Storkey, A.J.; Bastin, M.E.; Whittle, I.; Pernet, C. Single subject fMRI test-retest reliability metrics and confounding factors. Neuroimage 2013, 69, 231–243. [Google Scholar] [CrossRef] [PubMed]
- Patriat, R.; Molloy, E.K.; Meier, T.B.; Kirk, G.R.; Nair, V.A.; Meyerand, M.E.; Prabhakaran, V.; Birn, R.M. The effect of resting condition on resting-state fMRI reliability and consistency: A comparison between resting with eyes open, closed, and fixated. Neuroimage 2013, 78, 463–473. [Google Scholar] [CrossRef] [PubMed]
- Pruim, R.H.R.; Mennes, M.; Buitelaar, J.K.; Beckmann, C.F. Evaluation of ICA-AROMA and alternative strategies for motion artifact removal in resting state fMRI. Neuroimage 2015, 112, 278–287. [Google Scholar] [CrossRef] [PubMed]
- Pruim, R.H.R.; Mennes, M.; van Rooij, D.; Llera, A.; Buitelaar, J.K.; Beckmann, C.F. ICA-AROMA: A robust ICA-based strategy for removing motion artifacts from fMRI data. Neuroimage 2015, 112, 267–277. [Google Scholar] [CrossRef]
- Oakes, T.R.; Johnstone, T.; Ores Walsh, K.S.; Greischar, L.L.; Alexander, A.L.; Fox, A.S.; Davidson, R.J. Comparison of fMRI motion correction software tools. Neuroimage 2005, 28, 529–543. [Google Scholar] [CrossRef]
- Morgan, V.L.; Dawant, B.M.; Li, Y.; Pickens, D.R. Comparison of fMRI statistical software packages and strategies for analysis of images containing random and stimulus-correlated motion. Comput. Med. Imaging Graph. 2007, 31, 436–446. [Google Scholar] [CrossRef]
- Edward, V.; Windischberger, C.; Cunnington, R.; Erdler, M.; Lanzenberger, R.; Mayer, D.; Endl, W.; Beisteiner, R. Quantification of fMRI artifact reduction by a novel plaster cast head holder. Hum. Brain Mapp. 2000, 11, 207–213. [Google Scholar] [CrossRef]
- Heim, S.; Amunts, K.; Mohlberg, H.; Wilms, M.; Friederici, A.D. Head motion during overt language production in functional magnetic resonance imaging. Neuroreport 2006, 17, 579–582. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.; Cunningham, J.P.; Glover, G.H. Influence of heart rate on the BOLD signal: The cardiac response function. Neuroimage 2009, 44, 857–869. [Google Scholar] [CrossRef] [PubMed]
- Frank, L.R.; Buxton, R.B.; Wong, E.C. Estimation of respiration-induced noise fluctuations from undersampled multislice fMRI data. Magn. Reson. Med. 2001, 45, 635–644. [Google Scholar] [CrossRef] [PubMed]
- Shmueli, K.; van Gelderen, P.; Zwart, J.A.; Horovitz, S.G.; Fukunaga, M.; Jansma, J.M.; Duyn, J.H. Low-frequency fluctuations in the cardiac rate as a source of variance in the resting-state fMRI BOLD signal. Neuroimage 2007, 38, 306–320. [Google Scholar] [CrossRef]
- Weiss, F.; Zamoscik, V.; Schmidt, S.N.L.; Halli, P.; Kirsch, P.; Gerchen, M.F. Just a very expensive breathing training? Risk of respiratory artefacts in functional connectivity-based real-time fMRI neurofeedback. Neuroimage 2020, 210, 116580. [Google Scholar] [CrossRef] [PubMed]
- Abbott, D.F.; Opdam, H.I.; Briellmann, R.S.; Jackson, G.D. Brief breath holding may confound functional magnetic resonance imaging studies. Hum. Brain Mapp. 2005, 24, 284–290. [Google Scholar] [CrossRef]
- Birn, R.M.; Diamond, J.B.; Smith, M.A.; Bandettini, P.A. Separating respiratory-variation-related fluctuations from neuronal-activity-related fluctuations in fMRI. Neuroimage 2006, 31, 1536–1548. [Google Scholar] [CrossRef]
- Birn, R.M.; Murphy, K.; Handwerker, D.A.; Bandettini, P.A. fMRI in the presence of task-correlated breathing variations. Neuroimage 2009, 47, 1092–1104. [Google Scholar] [CrossRef]
- Gretton, A.; Belitski, A.; Murayama, Y.; Schölkopf, B.; Logothetis, N. The effect of artifacts on dependence measurement in fMRI. Magn. Reson. Imaging 2006, 24, 401–409. [Google Scholar] [CrossRef]
- Kemna, L.J.; Posse, S. Effect of respiratory CO(2) changes on the temporal dynamics of the hemodynamic response in functional MR imaging. Neuroimage 2001, 14, 642–649. [Google Scholar] [CrossRef][Green Version]
- Magon, S.; Basso, G.; Farace, P.; Ricciardi, G.K.; Beltramello, A.; Sbarbati, A. Reproducibility of BOLD signal change induced by breath holding. Neuroimage 2009, 45, 702–712. [Google Scholar] [CrossRef]
- Raj, D.; Anderson, A.W.; Gore, J.C. Respiratory effects in human functional magnetic resonance imaging due to bulk susceptibility changes. Phys. Med. Biol. 2001, 46, 3331–3340. [Google Scholar] [CrossRef] [PubMed]
- Wong, S.W.H.; Xue, G.; Bechara, A. Integrating fMRI with psychophysiological measurements in the study of decision-making. J. Neurosci. Psychol. Econ. 2011, 4, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Kugel, H.; Bremer, C.; Püschel, M.; Fischbach, R.; Lenzen, H.; Tombach, B.; van Aken, H.; Heindel, W. Hazardous situation in the MR bore: Induction in ECG leads causes fire. Eur. Radiol. 2003, 13, 690–694. [Google Scholar] [CrossRef] [PubMed]
- Bright, M.G.; Tench, C.R.; Murphy, K. Potential pitfalls when denoising resting state fMRI data using nuisance regression. Neuroimage 2017, 154, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Churchill, N.W.; Strother, S.C. PHYCAA+: An optimized, adaptive procedure for measuring and controlling physiological noise in BOLD fMRI. Neuroimage 2013, 82, 306–325. [Google Scholar] [CrossRef]
- Churchill, N.W.; Yourganov, G.; Spring, R.; Rasmussen, P.M.; Lee, W.; Ween, J.E.; Strother, S.C. PHYCAA: Data-driven measurement and removal of physiological noise in BOLD fMRI. Neuroimage 2012, 59, 1299–1314. [Google Scholar] [CrossRef]
- Kasper, L.; Bollmann, S.; Diaconescu, A.O.; Hutton, C.; Heinzle, J.; Iglesias, S.; Hauser, T.U.; Sebold, M.; Manjaly, Z.-M.; Pruessmann, K.P.; et al. The PhysIO Toolbox for Modeling Physiological Noise in fMRI Data. J. Neurosci. Methods 2017, 276, 56–72. [Google Scholar] [CrossRef]
- Murphy, K.; Fox, M.D. Towards a consensus regarding global signal regression for resting state functional connectivity MRI. Neuroimage 2017, 154, 169–173. [Google Scholar] [CrossRef]
- Power, J.D.; Plitt, M.; Laumann, T.O.; Martin, A. Sources and implications of whole-brain fMRI signals in humans. Neuroimage 2017, 146, 609–625. [Google Scholar] [CrossRef]
- Parkes, L.; Fulcher, B.; Yücel, M.; Fornito, A. An evaluation of the efficacy, reliability, and sensitivity of motion correction strategies for resting-state functional MRI. Neuroimage 2018, 171, 415–436. [Google Scholar] [CrossRef] [PubMed]
- Ludyga, S.; Gerber, M.; Brand, S.; Holsboer-Trachsler, E.; Puhse, U. Acute effects of moderate aerobic exercise on specific aspects of executive function in different age and fitness groups: A meta-analysis. Psychophysiology 2016, 11, 1611–1626. [Google Scholar] [CrossRef] [PubMed]
- Oberste, M.; Javelle, F.; Sharma, S.; Joisten, N.; Walzik, D.; Bloch, W.; Zimmer, P. Effects and Moderators of Acute Aerobic Exercise on Subsequent Interference Control: A Systematic Review and Meta-Analysis. Front. Psychol. 2019, 10, 609. [Google Scholar] [CrossRef] [PubMed]
- Boecker, H.; Henriksen, G.; Sprenger, T.; Miederer, I.; Willoch, F.; Valet, M.; Berthele, A.; Tölle, T.R. Positron emission tomography ligand activation studies in the sports sciences: Measuring neurochemistry in vivo. Methods 2008, 45, 307–318. [Google Scholar] [CrossRef] [PubMed]
- Boecker, H.; Othman, A.; Mueckter, S.; Scheef, L.; Pensel, M.; Daamen, M.; Jankowski, J.; Schild, H.; Tölle, T.; Schreckenberger, M. Advocating neuroimaging studies of transmitter release in human physical exercise challenges studies. Open Access J. Sports Med. 2010, 1, 167–175. [Google Scholar] [CrossRef]
- Boecker, H.; Sprenger, T.; Spilker, M.E.; Henriksen, G.; Koppenhoefer, M.; Wagner, K.J.; Valet, M.; Berthele, A.; Tolle, T.R. The runner’s high: Opioidergic mechanisms in the human brain. Cereb. Cortex 2008, 18, 2523–2531. [Google Scholar] [CrossRef]
- Boecker, H.; Tölle, T.R.; Valet, M.; Sprenger, T. Effects of Aerobic Exercise on Mood and Human Opioidergic Activation Measured by Positron Emission Tomography. In Functional Neuroimaging in Exercise and Sport Sciences; Boecker, H., Hillman, C.H., Scheef, L., Strüder, H.K., Eds.; Springer: New York, NY, USA, 2012; pp. 499–510. ISBN 9781461432920. [Google Scholar]
- van Hall, G.; Stromstad, M.; Rasmussen, P.; Jans, O.; Zaar, M.; Gam, C.; Quistorff, B.; Secher, N.H.; Nielsen, H.B. Blood lactate is an important energy source for the human brain. J. Cereb. Blood Flow Metab. 2009, 29, 1121–1129. [Google Scholar] [CrossRef]
- Quistorff, B.; Secher, N.H.; van Lieshout, J.J. Lactate fuels the human brain during exercise. FASEB J. 2008, 22, 3443–3449. [Google Scholar] [CrossRef]
- Proia, P.; Di Liegro, C.M.; Schiera, G.; Fricano, A.; Di Liegro, I. Lactate as a Metabolite and a Regulator in the Central Nervous System. Int. J. Mol. Sci. 2016, 17, 1450. [Google Scholar] [CrossRef]
- Dennis, A.; Thomas, A.G.; Rawlings, N.B.; Near, J.; Nichols, T.E.; Clare, S.; Johansen-Berg, H.; Stagg, C.J. An Ultra-High Field Magnetic Resonance Spectroscopy Study of Post Exercise Lactate, Glutamate and Glutamine Change in the Human Brain. Front. Physiol. 2015, 6, 351. [Google Scholar] [CrossRef]
- Taher, M.; Leen, W.G.; Wevers, R.A.; Willemsen, M.A. Lactate and its many faces. Eur. J. Paediatr. Neurol. 2016, 20, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Riske, L.; Thomas, R.K.; Baker, G.B.; Dursun, S.M. Lactate in the brain: An update on its relevance to brain energy, neurons, glia and panic disorder. Ther. Adv. Psychopharmacol. 2017, 7, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Brooks, G.A. The Science and Translation of Lactate Shuttle Theory. Cell Metab. 2018, 27, 757–785. [Google Scholar] [CrossRef] [PubMed]
- Sobral-Monteiro-Junior, R.; Maillot, P.; Gatica-Rojas, V.; Ávila, W.R.M.; Paula, A.M.B.; Guimarães, A.L.S.; Santos, S.H.S.; Pupe, C.C.B.; Deslandes, A.C. Is the “lactormone” a key-factor for exercise-related neuroplasticity? A hypothesis based on an alternative lactate neurobiological pathway. Med. Hypotheses 2019, 123, 63–66. [Google Scholar] [CrossRef]
- Kemppainen, J.; Aalto, S.; Fujimoto, T.; Kalliokoski, K.K.; Långsjö, J.; Oikonen, V.; Rinne, J.; Nuutila, P.; Knuuti, J. High intensity exercise decreases global brain glucose uptake in humans. J. Physiol. 2005, 568, 323–332. [Google Scholar] [CrossRef]
- Tsukamoto, H.; Suga, T.; Takenaka, S.; Tanaka, D.; Takeuchi, T.; Hamaoka, T.; Isaka, T.; Ogoh, S.; Hashimoto, T. Repeated high-intensity interval exercise shortens the positive effect on executive function during post-exercise recovery in healthy young males. Physiol. Behav. 2016, 160, 26–34. [Google Scholar] [CrossRef]
- Hashimoto, T.; Tsukamoto, H.; Takenaka, S.; Olesen, N.D.; Petersen, L.G.; Sørensen, H.; Nielsen, H.B.; Secher, N.H.; Ogoh, S. Maintained exercise-enhanced brain executive function related to cerebral lactate metabolism in men. FASEB J. 2017, 32, 1417–1427. [Google Scholar] [CrossRef]
- Li, M.; Newton, A.T.; Anderson, A.W.; Ding, Z.; Gore, J.C. Characterization of the hemodynamic response function in white matter tracts for event-related fMRI. Nat. Commun. 2019, 10, 1140. [Google Scholar] [CrossRef]
- Ding, Z.; Huang, Y.; Bailey, S.K.; Gao, Y.; Cutting, L.E.; Rogers, B.P.; Newton, A.T.; Gore, J.C. Detection of synchronous brain activity in white matter tracts at rest and under functional loading. Proc. Natl. Acad. Sci. USA 2018, 115, 595–600. [Google Scholar] [CrossRef]
- Fraser, L.M.; Stevens, M.T.; Beyea, S.D.; D’Arcy, R.C.N. White versus gray matter: fMRI hemodynamic responses show similar characteristics, but differ in peak amplitude. BMC Neurosci. 2012, 13, 91. [Google Scholar] [CrossRef]
- Gawryluk, J.R.; D’Arcy, R.C.N.; Mazerolle, E.L.; Brewer, K.D.; Beyea, S.D. Functional mapping in the corpus callosum: A 4T fMRI study of white matter. Neuroimage 2011, 54, 10–15. [Google Scholar] [CrossRef]
- Gawryluk, J.R.; Mazerolle, E.L.; D’Arcy, R.C.N. Does functional MRI detect activation in white matter? A review of emerging evidence, issues, and future directions. Front. Neurosci. 2014, 8, 239. [Google Scholar] [CrossRef] [PubMed]
- Mazerolle, E.L.; Beyea, S.D.; Gawryluk, J.R.; Brewer, K.D.; Bowen, C.V.; D’Arcy, R.C.N. Confirming white matter fMRI activation in the corpus callosum: Co-Localization with DTI tractography. Neuroimage 2010, 50, 616–621. [Google Scholar] [CrossRef] [PubMed]
- Tae, W.S.; Yakunina, N.; Kim, T.S.; Kim, S.S.; Nam, E.-C. Activation of auditory white matter tracts as revealed by functional magnetic resonance imaging. Neuroradiology 2014, 56, 597–605. [Google Scholar] [CrossRef] [PubMed]
- Yarkoni, T.; Barch, D.M.; Gray, J.R.; Conturo, T.E.; Braver, T.S. BOLD correlates of trial-by-trial reaction time variability in gray and white matter: A multi-study fMRI analysis. PLoS ONE 2009, 4, e4257. [Google Scholar] [CrossRef]
- Garrett, D.D.; Samanez-Larkin, G.R.; MacDonald, S.W.S.; Lindenberger, U.; McIntosh, A.R.; Grady, C.L. Moment-to-moment brain signal variability: A next frontier in human brain mapping? Neurosci. Biobehav. Rev. 2013, 37, 610–624. [Google Scholar] [CrossRef]
- Grady, C.L.; Garrett, D.D. Brain signal variability is modulated as a function of internal and external demand in younger and older adults. Neuroimage 2017, 169, 510–523. [Google Scholar] [CrossRef]
- Garrett, D.D.; Lindenberger, U.; Hoge, R.D.; Gauthier, C.J. Age differences in brain signal variability are robust to multiple vascular controls. Sci. Rep. 2017, 7, 10149. [Google Scholar] [CrossRef]
- Armbruster-Genç, D.J.N.; Ueltzhöffer, K.; Fiebach, C.J. Brain Signal Variability Differentially Affects Cognitive Flexibility and Cognitive Stability. J. Neurosci. 2016, 36, 3978–3987. [Google Scholar] [CrossRef]
- van Horn, J.D.; Grafton, S.T.; Miller, M.B. Individual Variability in Brain Activity: A Nuisance or an Opportunity? Brain Imaging Behav. 2008, 2, 327–334. [Google Scholar] [CrossRef]
- Dinstein, I.; Heeger, D.J.; Behrmann, M. Neural variability: Friend or foe? Trends Cogn. Sci. 2015, 19, 322–328. [Google Scholar] [CrossRef]
- Garrett, D.D.; Kovacevic, N.; McIntosh, A.R.; Grady, C.L. Blood oxygen level-dependent signal variability is more than just noise. J. Neurosci. 2010, 30, 4914–4921. [Google Scholar] [CrossRef] [PubMed]
- Garrett, D.D.; Nagel, I.E.; Preuschhof, C.; Burzynska, A.Z.; Marchner, J.; Wiegert, S.; Jungehülsing, G.J.; Nyberg, L.; Villringer, A.; Li, S.-C.; et al. Amphetamine modulates brain signal variability and working memory in younger and older adults. Proc. Natl. Acad. Sci. USA 2015, 112, 7593–7598. [Google Scholar] [CrossRef]
- Nomi, J.S.; Bolt, T.S.; Ezie, C.E.C.; Uddin, L.Q.; Heller, A.S. Moment-to-Moment BOLD Signal Variability Reflects Regional Changes in Neural Flexibility across the Lifespan. J. Neurosci. 2017, 37, 5539–5548. [Google Scholar] [CrossRef] [PubMed]
- Protzner, A.B.; Kovacevic, N.; Cohn, M.; McAndrews, M.P. Characterizing functional integrity: Intraindividual brain signal variability predicts memory performance in patients with medial temporal lobe epilepsy. J. Neurosci. 2013, 33, 9855–9865. [Google Scholar] [CrossRef] [PubMed]
- Button, K.S.; Ioannidis, J.P.; Mokrysz, C.; Nosek, B.A.; Flint, J.; Robinson, E.S.J.; Munafò, M.R. Power failure: Why small sample size undermines the reliability of neuroscience. Nat. Rev. Neurosci. 2013, 14, 365–376. [Google Scholar] [CrossRef]
- de Vet, H.C.; Terwee, C.B.; Knol, D.L.; Bouter, L.M. When to use agreement versus reliability measures. J. Clin. Epidemiol. 2006, 59, 1033–1039. [Google Scholar] [CrossRef]
- Atkinson, G.; Nevill, A.M. Statistical methods for assessing measurement error (reliability) in variables relevant to sports medicine. Sports Med. 1998, 26, 217–238. [Google Scholar] [CrossRef]
- Donath, L.; Ludyga, S.; Hammes, D.; Rossmeissl, A.; Andergassen, N.; Zahner, L.; Faude, O. Absolute and relative reliability of acute effects of aerobic exercise on executive function in seniors. BMC Geriatr. 2017, 17, 247. [Google Scholar] [CrossRef]
- Birn, R.M.; Molloy, E.K.; Patriat, R.; Parker, T.; Meier, T.B.; Kirk, G.R.; Nair, V.A.; Meyerand, M.E.; Prabhakaran, V. The effect of scan length on the reliability of resting-state fMRI connectivity estimates. Neuroimage 2013, 83, 550–558. [Google Scholar] [CrossRef]
- Bennett, C.M.; Miller, M.B. fMRI reliability: Influences of task and experimental design. Cogn. Affect. Behav. Neurosci. 2013, 13, 690–702. [Google Scholar] [CrossRef] [PubMed]
- Burke, T.M.; Scheer, F.A.J.L.; Ronda, J.M.; Czeisler, C.A.; Wright, K.P. Sleep inertia, sleep homeostatic and circadian influences on higher-order cognitive functions. J. Sleep Res. 2015, 24, 364–371. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.A.E.; Campbell, K.L.; Amer, T.; Grady, C.L.; Hasher, L. Timing is everything: Age differences in the cognitive control network are modulated by time of day. Psychol. Aging 2014, 29, 648–657. [Google Scholar] [CrossRef] [PubMed]
- Hodyl, N.A.; Schneider, L.; Vallence, A.-M.; Clow, A.; Ridding, M.C.; Pitcher, J.B. The cortisol awakening response is associated with performance of a serial sequence reaction time task. Int. J. Psychophysiol. 2016, 100, 12–18. [Google Scholar] [CrossRef]
- Kojima, S.; Abe, T.; Morishita, S.; Inagaki, Y.; Qin, W.; Hotta, K.; Tsubaki, A. Acute moderate-intensity exercise improves 24-h sleep deprivation-induced cognitive decline and cerebral oxygenation: A near-infrared spectroscopy study. Respir. Physiol. Neurobiol. 2019, 274, 103354. [Google Scholar] [CrossRef]
- Byun, K.; Hyodo, K.; Suwabe, K.; Ochi, G.; Sakairi, Y.; Kato, M.; Dan, I.; Soya, H. Positive effect of acute mild exercise on executive function via arousal-related prefrontal activations: An fNIRS study. Neuroimage 2014, 98, 336–345. [Google Scholar] [CrossRef]
- Kato, K.; Miyata, S.; Ando, M.; Matsuoka, H.; Yasuma, F.; Iwamoto, K.; Kawano, N.; Banno, M.; Ozaki, N.; Noda, A. Influence of sleep duration on cortical oxygenation in elderly individuals. Psychiatry Clin. Neurosci. 2017, 71, 44–51. [Google Scholar] [CrossRef]
- Kato, K.; Iwamoto, K.; Kawano, N.; Noda, Y.; Ozaki, N.; Noda, A. Differential effects of physical activity and sleep duration on cognitive function in young adults. J. Sport Health Sci. 2017, 7, 227–236. [Google Scholar] [CrossRef]
- Nishida, M.; Kikuchi, S.; Miwakeichi, F.; Suda, S. Night duty and decreased brain activity of medical residents: A wearable optical topography study. Med. Educ. Online 2017, 22, 1379345. [Google Scholar] [CrossRef]
- Suda, M.; Sato, T.; Kameyama, M.; Ito, M.; Suto, T.; Yamagishi, Y.; Uehara, T.; Fukuda, M.; Mikuni, M. Decreased cortical reactivity underlies subjective daytime light sleepiness in healthy subjects: A multichannel near-infrared spectroscopy study. Neurosci. Res. 2008, 60, 319–326. [Google Scholar] [CrossRef]
| First Author (year) | Sample Characteristics and Study Design (1.) Health Status (2.) Cardiorespiratory Fitness Level (mean ± SD in ml/kg/min) (3.) Number of Participants (gender) (4.) Age (mean ± SD or range in years) (5.) (i) Body Height (mean ± SD or range in cm)/ (ii) Body Mass (mean ± SD or range in kg)/ (iii) BMI (mean ± SD) | Main Findings (1.) Behavioral Level (2.) Functional Level |
|---|---|---|
| Bothe et al. (2013) [99] | (1.) Young healthy trained and inactive adults (2.) HT (T) 56.0 ± 5.2; HT (P) 55.1 ± 6.2/ PIA (T) 48.4 ± 6.8; PIA (P) 50.2 ± 2.9 (3.) HT (T) N = 9 (9 m); HT (P) N = 10 (10 m)/ PIA (T) N = 11; PIA (P) N = 13 (13 m) (4.) HT (T) 24.0 ± 4.0; HT (P) 26.1 ± 3.4/ PIA (T) 25.1 ± 3.0; PIA (P) 23.9 ± 2.7 (5.) (i) N.A./ (ii) N.A./ (iii) HT (T) 22.8 ± 2.0; HT (P) 23.0 ± 1.9, PIA (T) 22.4 ± 1.5; PIA (P) 21.6 ± 1.5 | Treadmill running vs. placebo (stretching): (1.) - no significant differences (2.) ↓ lt. caudate nucleus in gain anticipation ↓ rt. caudate nucleus in gain feedback |
| Chen et al. (2016) [71] | (1.) Healthy children (2.) N.A. (3.) N = 9 (4 f/ 5 m) (4.) 10.0 ± 0.0 (5.) (i) N.A./ (ii) N.A./ (iii) N.A. | Cycling vs. seated rest: (1.) ↓ mean RT in the 2-back condition (2.) ↑ lt. superior/inferior parietal gyrus, rt. superior parietal gyrus, lt. hippocampus, lt. cerebellum, rt. cerebellum |
| Chen et al. (2017) [73] | (1.) Healthy children (2.) N.A. (3.) N = 9 (4 f/ 5 m) (4.) 10.0 ± 0.0 (5.) (i) N.A./ (ii) N.A./ (iii) N.A. | Cycling vs. seated rest: (1.) ↓ mean RT in Eriksen flanker task (2.) ↑ resting-state functional connectivity in the lt. cerebellum-rt. inferior gyrus is significantly negatively correlated with better performance (e.g., shorter reaction time) in an Eriksen flanker task |
| Li et al. (2014) [39] | (1.) Healthy younger adults (2.) N.A. (3.) N = 15 (15 f) (4.) 19-22 (5.) (i) 161.6 ± 3.1/ (ii) 50.8 ± 3.4/ (iii) 19.5 ± 1.4 | Cycling vs. seated rest: (1.) - no significant differences (2.) ↑ rt. middle frontal gyrus, rt. lingual gyrus, lt. fusiform gyrus/↓ anterior cingulate gyrus, rt. paracentral lobule, and lt. inferior frontal gyrus in the 2-back condition |
| Li et al. (2019) [106] | (1.) Healthy high-fit and low-fit younger adults (2.) HF 26.5 ± 1.9/ LF 19.9 ± 1.0 (3.) HF N = 12 (12 f)/ LF N = 12 (12 f) (4.) HF 25.5 ± 0.7/ LF 25.8 ± 0.6 (5.) (i) HF 164.8 ± 5.9; LF 161.1 ± 4.9/ (ii) HF 55.7 ± 5.1; LF 52.0 ± 5.0/ (iii) HF 20.5 ± 1.1; LF 20.0 ± 1.5 | After cycling vs. prior cycling: (1.) ↑ accuracy in LF in 0-back ↓ accuracy in HF in 1-back (2.) ↓ rt. cerebellum in HF in 0-back and 1-back ↑ rt. cerebellum in LF in 0-back and 1-back ↑ lt. and rt. cerebellum, rt. paracentral lobule, and rt. medial temporal pole in HF and LF in 1-back ↑ lt. anterior cingulate gyrus and in lt. pallidum in HF in 2-back ↓ lt. anterior cingulate cortex in LF in 2-back HF vs. LF: (1.) ↑ accuracy in 1-back and 2-back (before cycling) (2.) ↑ rt. cerebellum in 0-back (before cycling) ↓ rt. cerebellum in 0-back and 1-back (after cycling) ↑ lt. inferior parietal lobule in 0-back and 1-back ↓ lt. anterior cingulate gyrus and in lt. globus pallidus in 2-back (before cycling) ↑ lt. anterior cingulate gyrus and in lt. globus pallidus in 2-back (after cycling) ↑ rt. superior frontal gyrus in 2-back |
| MacIntosh et al. (2014) [72] | (1.) Healthy younger adults (2.) N.A. (3.) N = 16 (10 f/ 6 m) (4.) 26.7 ± 4.1 (20-35) (5.) (i) 171.0 ± 10.5/ (ii) 67.9 ± 13.9/ (iii) N.A. | After vs. prior cycling: (1.) - no significant change (2.) ↓ lt. parietal operculum |
| Mehren et al. (2019) [100] | (1.) Younger adults with and without ADHD (2.) ADHD 36.6 ± 7.5/ HC 42.0 ± 7.3 (3.) ADHD N = 23 (3 f/ 20 m)/ HC N = 23 (4 f/ 20 m) (4.) ADHD 31.4 ± 9.6/HC 29.5 ± 7.0 (5.) (i) N.A./ (ii) N.A./ (iii) ADHD 25.6 ± 4.3; HC 24.1 ± 2.6 | Cycling vs. rest (watching movie): (1.) – no significant differences (2.) ↑ lt. middle occipital gyrus, rt. middle occipital gyrus, rt. supramarginal gyrus, and lt. inferior parietal gyrus during correct inhibitions in Go/No-Go task in ADHD - negative correlation between exercise-related changes in brain activation in lt. insula, lt. precentral gyrus, and rt. postcentral gyrus during correct inhibitions in Go/No-Go task and task performance in the control condition in ADHD ADHD vs. HC (1.) – no significant differences (2.) ↑ lt. superior occipital gyrus, rt. precuneus, and lt. supramarginal gyrus |
| Mehren et al. (2019) [101] | (1.) Younger healthy adults (2.) MIE 39.6 ± 7.1/ HIE 37.0 ± 8.3 (3.) MIE N = 32 (16 f/ 16 m)/ HIE N = 31 (16 f/ 15 m) (4.) MIE 29.3 ± 8.5/ HIE 28.6 ± 7.7 (5.) (i) N.A./ (ii) N.A./ (iii) MIE 23.8 ± 2.3; HIE 24.5 ± 4.8 | Cycling vs. rest (watching movie): (1.) ↓ reaction times in a congruent and incongruent condition (Flanker task) in HIIE ↑ sensitivity index (Go/No-Go task) in MICE (2.) ↑ lt. superior frontal gyrus, rt. precentral gyrus and the triangular part of the lt. inferior frontal gyrus in Go/No-Go task (contrast “hits”) in MICE ↓ lt. anterior cingulate gyrus in a visual task in MICE ↑ lt. lingual gyrus and precuneus in a visual task in HIIE ↑ rt. precentral gyrus in Go/No-Go task (contrast “hits”) in MICE (males vs. females) MICE vs. HIIE: (1.) - no significant differences (2.) ↑ lt. superior frontal gyrus and rt. insula in Go/No-Go task (contrast “hits”) ↓ lt. lingual gyrus, rt. precuneus, and lt. anterior cingulate gyrus in a visual task Associations between brain activation and CRF: - positive association between activation of rt. insula and VO2 peak in MICE (Flanker task contrast “incongruent – congruent”) - negative association between activation of rt. postcentral gyrus VO2 peak in HIIE (Go/No-Go task – contrast “hits”) - positive association between activation of lt. rolandic operculum VO2 peak in MICE (Go/No-Go task – contrast “correct inhibitions - hits”) |
| Mehren et al. (2019) [102] | (1.) Younger adults with and without ADHD (2.) ADHD 37.1 ± 7.2/ HC 41.5 ± 7.3 (3.) ADHD N = 20 (4 f/ 16 m)/ HC N = 20 (5 f/ 15 m) (4.) ADHD 29.9 ± 9.5/ HC 29.0 ± 7.4 (5.) (i) N.A./ (ii) N.A./ (iii) ADHD 25.0 ± 3.8; HC 24.3 ± 2.7 | Cycling vs. rest (watching movie): (1.) ↓ interference score (incongruent-congruent) in HC ↓ mean RT in a congruent and incongruent condition in ADHD ↓ RT variability in a congruent condition in ADHD - significant positive correlation between RT differences (i.e., differences between exercise and control condition) in incongruent trials and VO2 peak in ADHD (2.) ↓ rt. precentral gyrus and rt. middle temporal gyrus in a congruent condition in ADHD ↓ rt. superior frontal gyrus, rt. middle frontal gyrus and paracentral lobule in incongruent condition in ADHD ↓ rt. superior frontal gyrus and rt. middle frontal gyrus in a visual task in ADHD |
| Metcalfe et al. (2016) [105] | (1.) Adolescents with and without BD (2.) N.A. (3.) BD N = 30 (17 f/13 m)/ HC N = 20 (11 f/9 m) (4.) BD 16.8 ± 1.4/ HC 16.1 ± 1.4 (5.) (i) N.A./ (ii) N.A./ (iii) N.A. | BD vs. HC: (1.) – no significant differences (2.) ↓ orbital part of the lt. inferior frontal gyrus, rt. frontal pole extending to temporal pole, rt. and lt. hippocampus and rt. amygdala in Go trials |
| Suwabe et al. (2018) [38] | (1.) Healthy younger adults (2.) 37.9 ± 8.2 (3.) N = 16 (12 f/4 m) (4.) 21.1 ± 2.0 (5.) (i) 164.2 ± 9.1/ (ii) 55.4 ± 7.7/ (iii) 20.5 ± 1.6 | Cycling vs. seated rest: (1.) ↑ better performance in mnemonic discrimination task (in high- and medium-similarity lures) (2.) - significant positive associations between dentate gyrus/CA3 subfield bilaterally and lt. angular gyrus, lt. fusiform gyrus, lt. parahippocampal cortex, and lt. primary visual cortex - significant negative association between dentate gyrus/CA3 subfield bilaterally and lt. temporal pole - significant association between the whole hippocampus (CA1, subiculum and dentate gyrus/CA3) and bilateral parahippocampal cortex - higher correlations between dentate gyrus/CA3 and angular gyrus, fusiform gyrus as well as parahippocampal cortex predicted improvements in cognitive performance |
| Voss et al. (2019) [107] | (1.) Healthy older adults (2.) 20.1 ± 5.0 (3.) N = 34 (20 f/ 14 m) (4.) 67.1 ± 4.3 (60-80) (5.) (i) N.A./ (ii) N.A./ (iii) 29.1 ± 5.3 | After vs. prior cycling: (1.) ↑ accuracy in 1-back and 2-back condition in LIE and MIE and ↑ accuracy in 1-back and 2-back condition in MIE vs. LIE - acute improvements in working memory performance predict long-term improvements in working memory performance (after 12-weeks of training) (2.) - acute changes in the following connections predict long-term changes in the same connections: (i) lt. middle occipital lobule and rt. angular gyrus, (ii) lt. superior frontal gyrus and lt. inferior temporal gyrus, (iii) lt. superior frontal gyrus and rt. inferior parietal gyrus, (iv) rt inferior parietal gyrus and rt. triangular part of the inferior frontal gyrus, (v) rt. inferior parietal gyrus and opercular part of the lt. inferior frontal gyrus, (vi) lt. lingual gyrus and rt. fusiform gyrus, (vii) rt. superior frontal gyrus and rt. middle temporal gyrus, (viii) rt. middle temporal gyrus and rt. inferior parietal gyrus, (ix) lt. and rt. precuneus, (x) lt. superior frontal gyrus and lt. medial superior frontal gyrus, (xi) rt. middle temporal gyrus and opercular part of the lt. inferior frontal gyrus, and (xii) rt. superior frontal gyrus and rt. inferior parietal gyrus - acute and long-term changes in (i) connections between rt. middle temporal gyrus and rt. superior frontal gyrus, and (ii) connections between rt. inferior parietal gyrus and opercular part of the rt. inferior frontal gyrus are associated with performance in the 2-back condition |
| Won et al. (2019) [103] | (1.) Healthy older adults (2.) N.A. (3.) N = 32 (24 f/ 8 m) (4.) 66.2 ± 7.3 (55-85) (5.) (i) 166.6 ± 9.0/ (ii) 71.3 ± 14.2/ (iii) 25.5 ± 4.1 | Cycling vs. seated rest: (1.) ↑ accuracy in a congruent and incongruent condition (2.) ↑ orbital part of the lt. inferior frontal gyrus and lt. inferior parietal gyrus in incongruent and incongruent-congruent condition ↓ rt. cingulate gyrus in incongruent and incongruent-congruent condition |
| Won et al. (2019) [104] | (1.) Healthy older adults (2.) N.A. (3.) N = 26 (20 f/ 6 m) (4.) 65.9 ± 7.2 (55-85) (5.) (i) 166.5 ± 8.5/ (ii) 73.6 ± 14.1/ (iii) 26.1 ± 4.3 | Cycling vs. seated rest: (1.) - no significant differences (2.) ↑ orbital part of lt. inferior frontal gyrus, lt. inferior temporal gyrus, rt. middle temporal gyrus, lt. fusiform gyrus, and rt. and lt. hippocampus |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Herold, F.; Aye, N.; Lehmann, N.; Taubert, M.; Müller, N.G. The Contribution of Functional Magnetic Resonance Imaging to the Understanding of the Effects of Acute Physical Exercise on Cognition. Brain Sci. 2020, 10, 175. https://doi.org/10.3390/brainsci10030175
Herold F, Aye N, Lehmann N, Taubert M, Müller NG. The Contribution of Functional Magnetic Resonance Imaging to the Understanding of the Effects of Acute Physical Exercise on Cognition. Brain Sciences. 2020; 10(3):175. https://doi.org/10.3390/brainsci10030175
Chicago/Turabian StyleHerold, Fabian, Norman Aye, Nico Lehmann, Marco Taubert, and Notger G. Müller. 2020. "The Contribution of Functional Magnetic Resonance Imaging to the Understanding of the Effects of Acute Physical Exercise on Cognition" Brain Sciences 10, no. 3: 175. https://doi.org/10.3390/brainsci10030175
APA StyleHerold, F., Aye, N., Lehmann, N., Taubert, M., & Müller, N. G. (2020). The Contribution of Functional Magnetic Resonance Imaging to the Understanding of the Effects of Acute Physical Exercise on Cognition. Brain Sciences, 10(3), 175. https://doi.org/10.3390/brainsci10030175







