Detecting the Potential for Consciousness in Unresponsive Patients Using the Perturbational Complexity Index
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Experimental Protocol
2.3. Data Analysis
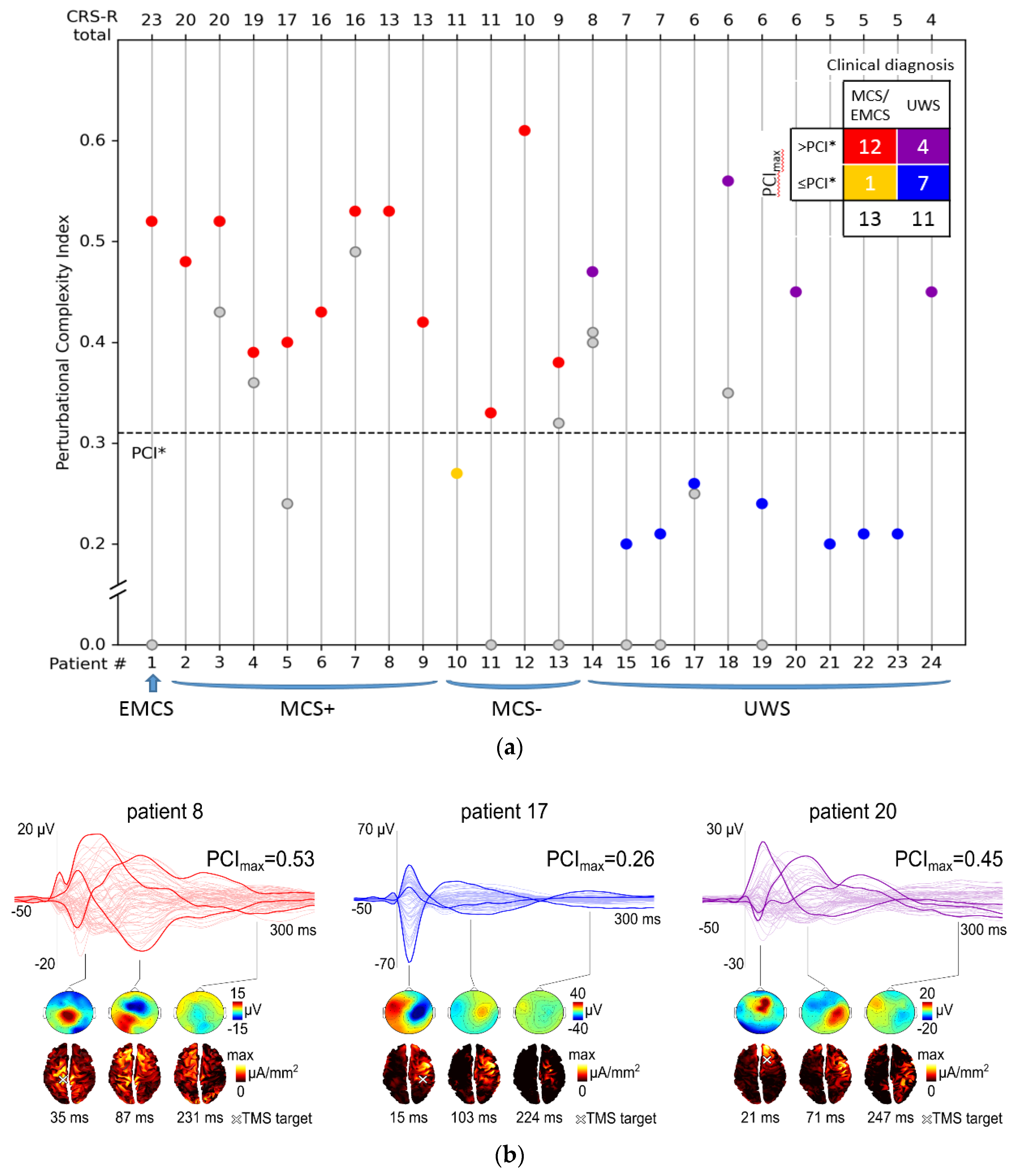
3. Results
3.1. Clinical Assessment
3.2. Neurophysiological Assessment
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Behavioral Assessment in Patients with Disorders of Consciousness: Gold Standard or Fool’s Gold? Available online: https://www.sciencedirect.com/science/article/pii/S007961230917704X (accessed on 26 November 2020).
- Giacino, J.T.; Katz, D.I.; Schiff, N.D.; Whyte, J.; Ashman, E.J.; Ashwal, S.; Barbano, R.; Hammond, F.M.; Laureys, S.; Ling, G.S.F.; et al. Comprehensive systematic review update summary: Disorders of consciousness. Neurology 2018, 91, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Luaute, J.; Maucort-Boulch, D.; Tell, L.; Quelard, F.; Sarraf, T.; Iwaz, J.; Boisson, D.; Fischer, C. Long-term outcomes of chronic minimally conscious and vegetative states. Neurology 2010, 75, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Faugeras, F.; Rohaut, B.; Valente, M.; Sitt, J.; Demeret, S.; Bolgert, F.; Weiss, N.; Grinea, A.; Marois, C.; Quirins, M.; et al. Survival and consciousness recovery are better in the minimally conscious state than in the vegetative state. Brain Inj. 2018, 32, 72–77. [Google Scholar] [CrossRef] [PubMed]
- Thibaut, A.; Schiff, N.; Giacino, J.; Laureys, S.; Gosseries, O. Therapeutic interventions in patients with prolonged disorders of consciousness. Lancet Neurol. 2019, 18, 600–614. [Google Scholar] [CrossRef]
- Thibaut, A.; Bruno, M.-A.; Ledoux, D.; Demertzi, A.; Laureys, S. tDCS in patients with disorders of consciousness: Sham-controlled randomized double-blind study. Neurology 2014, 82, 1112–1118. [Google Scholar] [CrossRef]
- Zhang, Y.; Song, W.; Du, J.; Huo, S.; Shan, G.; Li, R. Transcranial direct current stimulation in patients with prolonged disorders of consciousness: Combined behavioral and event-related potential evidence. Front. Neurol. 2017, 8, 620. [Google Scholar] [CrossRef]
- Martens, G.; Lejeune, N.; O’Brien, A.T.; Fregni, F.; Martial, C.; Wannez, S.; Laureys, S.; Thibaut, A. Randomized controlled trial of home-based 4-week tDCS in chronic minimally conscious state. Brain Stimul. 2018, 11, 982–990. [Google Scholar] [CrossRef]
- Angelakis, E.; Liouta, E.; Andreadis, N.; Korfias, S.; Ktonas, P.; Stranjalis, G.; Sakas, D.E. Transcranial direct current stimulation effects in disorders of consciousness. Arch. Phys. Med. Rehabil. 2014, 95, 283–289. [Google Scholar] [CrossRef]
- Legostaeva, L.; Poydasheva, A.; Iazeva, E.; Sinitsyn, D.; Sergeev, D.; Bakulin, I.; Lagoda, D.; Kremneva, E.; Morozova, S.; Ryabinkina, Y.; et al. Stimulation of the angular gyrus improves the level of consciousness. Brain Sci. 2019, 9, 103. [Google Scholar] [CrossRef]
- Schnakers, C. Update on diagnosis in disorders of consciousness. Expert Rev. Neurother. 2020, 20, 997–1004. [Google Scholar] [CrossRef]
- Cortese, M.; Riganello, F.; Arcuri, F.; Pugliese, M.; Lucca, L.; Dolce, G.; Sannita, W. Coma recovery scale-r: Variability in the disorder of consciousness. BMC Neurol. 2015, 15, 186. [Google Scholar] [CrossRef] [PubMed]
- Giacino, J.T.; Kalmar, K.; Whyte, J. The JFK Coma Recovery Scale-Revised: Measurement characteristics and diagnostic utility. Arch. Phys. Med. Rehabil. 2004, 85, 2020–2029. [Google Scholar] [CrossRef] [PubMed]
- Kondziella, D.; Bender, A.; Diserens, K.; van Erp, W.; Estraneo, A.; Formisano, R.; Laureys, S.; Naccache, L.; Ozturk, S.; Rohaut, B.; et al. European Academy of Neurology guideline on the diagnosis of coma and other disorders of consciousness. Eur. J. Neurol. 2020, 27, 741–756. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Lin, Y.; Ziemann, U. Managing disorders of consciousness: The role of electroencephalography. J. Neurol. 2020, in press. [Google Scholar] [CrossRef]
- Comanducci, A.; Boly, M.; Claassen, J.; De Lucia, M.; Gibson, R.M.; Juan, E.; Laureys, S.; Naccache, L.; Owen, A.M.; Rosanova, M.; et al. Clinical and advanced neurophysiology in the prognostic and diagnostic evaluation of disorders of consciousness: Review of an IFCN-endorsed expert group. Clin. Neurophysiol. 2020, 131, 2736–2765. [Google Scholar] [CrossRef]
- Casali, A.G.; Gosseries, O.; Rosanova, M.; Boly, M.; Sarasso, S.; Casali, K.R.; Casarotto, S.; Bruno, M.-A.; Laureys, S.; Tononi, G.; et al. A theoretically based index of consciousness independent of sensory processing and behavior. Sci. Transl. Med. 2013, 5, ra105–ra198. [Google Scholar] [CrossRef]
- Casarotto, S.; Comanducci, A.; Rosanova, M.; Sarasso, S.; Fecchio, M.; Napolitani, M.; Pigorini, A.; Casali, A.G.; Trimarchi, P.D.; Boly, M.; et al. Stratification of unresponsive patients by an independently validated index of brain complexity. Ann. Neurol. 2016, 80, 718–729. [Google Scholar] [CrossRef]
- Giacino, J.T.; Katz, D.I.; Schiff, N.D.; Whyte, J.; Ashman, E.J.; Ashwal, S.; Barbano, R.; Hammond, F.M.; Laureys, S.; Ling, G.S.F.; et al. Practice guideline update recommendations summary: Disorders of consciousness. Neurology 2018, 91, 450–460. [Google Scholar] [CrossRef]
- Iazeva, E.G.; Legostaeva, L.A.; Zimin, A.A.; Sergeev, D.V.; Domashenko, M.A.; Samorukov, V.Y.; Yusupova, D.G.; Ryabinkina, J.V.; Suponeva, N.A.; Piradov, M.A.; et al. A Russian validation study of the Coma Recovery Scale-Revised (CRS-R). Brain Inj. 2019, 33, 218–225. [Google Scholar] [CrossRef]
- Thibaut, A.; Bodien, Y.G.; Laureys, S.; Giacino, J.T. Minimally conscious state “plus”: Diagnostic criteria and relation to functional recovery. J. Neurol. 2020, 267, 1245–1254, Erratum in 2020, 267, 1255–1259. [Google Scholar] [CrossRef]
- Gosseries, O.; Sarasso, S.; Casarotto, S.; Boly, M.; Schnakers, C.; Napolitani, M.; Bruno, M.-A.; Ledoux, D.; Tshibanda, J.-F.; Massimini, M.; et al. On the cerebral origin of EEG responses to TMS: Insights from severe cortical lesions. Brain Stimul. 2015, 8, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Forgacs, P.B.; Conte, M.M.; Fridman, E.A.; Voss, H.U.; Victor, J.D.; Schiff, N.D. Preservation of electroencephalographic organization in patients with impaired consciousness and imaging-based evidence of command-following. Ann. Neurol. 2014, 76, 869–879. [Google Scholar] [CrossRef] [PubMed]
- Estraneo, A.; Loreto, V.; Guarino, I.; Boemia, V.; Paone, G.; Moretta, P.; Trojano, L. Standard EEG in diagnostic process of prolonged disorders of consciousness. Clin. Neurophysiol. 2016, 127, 2379–2385. [Google Scholar] [CrossRef] [PubMed]
- Sitt, J.D.; King, J.-R.; El Karoui, I.; Rohaut, B.; Faugeras, F.; Gramfort, A.; Cohen, L.; Sigman, M.; Dehaene, S.; Naccache, L. Large scale screening of neural signatures of consciousness in patients in a vegetative or minimally conscious state. Brain 2014, 137, 2258–2270. [Google Scholar] [CrossRef] [PubMed]
- Engemann, D.A.; Raimondo, F.; King, J.-R.; Rohaut, B.; Louppe, G.; Faugeras, F.; Annen, J.; Cassol, H.; Gosseries, O.; Fernandez-Slezak, D.; et al. Robust EEG-based cross-site and cross-protocol classification of states of consciousness. Brain 2018, 141, 3179–3192. [Google Scholar] [CrossRef]
- Sarasso, S.; Boly, M.; Napolitani, M.; Gosseries, O.; Charland-Verville, V.; Casarotto, S.; Rosanova, M.; Casali, A.G.; Brichant, J.-F.; Boveroux, P.; et al. Consciousness and complexity during unresponsiveness induced by propofol, xenon, and ketamine. Curr. Biol. 2015, 25, 3099–3105. [Google Scholar] [CrossRef]
- Rosanova, M.; Fecchio, M.; Casarotto, S.; Sarasso, S.; Casali, A.G.; Pigorini, A.; Comanducci, A.; Seregni, F.; Devalle, G.; Citerio, G.; et al. Sleep-like cortical OFF-periods disrupt causality and complexity in the brain of unresponsive wakefulness syndrome patients. Nat. Commun. 2018, 9, 4427. [Google Scholar] [CrossRef]
- Coyle, D.; Stow, J.; McCreadie, K.; McElligott, J.; Carroll, Á. Sensorimotor modulation assessment and brain-computer interface training in disorders of consciousness. Arch. Phys. Med. Rehabil. 2015, 96, S62–S70. [Google Scholar] [CrossRef]
- Ortner, R.; Allison, B.Z.; Pichler, G.; Heilinger, A.; Sabathiel, N.; Guger, C. Assessment and communication for people with disorders of consciousness. J. Vis. Exp. 2017, e53639. [Google Scholar] [CrossRef]
- Chatelle, C.; Spencer, C.A.; Cash, S.S.; Hochberg, L.R.; Edlow, B.L. Feasibility of an EEG-based brain-computer interface in the intensive care unit. Clin. Neurophysiol. 2018, 129, 1519–1525. [Google Scholar] [CrossRef]

| CRS-R | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Patient Number | Age [Years]/Gender | Time Since Injury [Months] | Etiology | Clinical Diagnosis | Auditory | Visual | Motor | Verbal | Comm | Arousal | TOT | Outcome | Stimulated Sites Number | PCImax Brain Side | PCImax Brain Area | PCImax | EEG Category ‡ |
| 1 | 20/m | 24 | T | EMCS | 4 | 5 | 6 | 3 | 2 | 3 | 23 | conscious | 2 | L | F | 0.52 | Mi |
| 2 | 20/m | 15 | T | MCS+ | 4 | 5 | 5 | 2 | 1 | 3 | 20 | conscious | 1 | L | F | 0.48 | Mi |
| 3 | 29/m | 17 | T | MCS+ | 4 | 4 | 5 | 3 | 1 | 3 | 20 | unknown | 3 | R | F | 0.52 | Mo |
| 4 | 21/f | 12 | T | MCS+ | 4 | 5 | 5 | 2 | 0 | 3 | 19 | conscious | 2 | L | F | 0.39 | n/a |
| 5 | 32/m | 32 | T | MCS+ | 4 | 4 | 5 | 1 | 1 | 2 | 17 | unknown | 2 | R | P | 0.40 | Mo |
| 6 | 31/f | 5 | hV | MCS+ | 4 | 4 | 5 | 1 | 0 | 2 | 16 | unknown | 1 | R | P | 0.43 | Mo |
| 7 | 29/m | 56 | T | MCS+ | 2 | 4 | 5 | 2 | 1 | 2 | 16 | conscious | 2 | L | F | 0.53 | Mi |
| 8 | 55/m | 4 | A | MCS+ | 2 | 2 | 4 | 2 | 1 | 2 | 13 | unknown | 1 | L | P | 0.53 | Se |
| 9 | 43/f | 7 | other ¥ | MCS+ | 3 | 3 | 3 | 1 | 1 | 2 | 13 | conscious | 1 | L | P | 0.42 | Mo |
| 10 | 44/m | 9 | T | MCS− | 2 | 2 | 3 | 2 | 0 | 2 | 11 | unknown | 1 | L | F | 0.27 | Mo |
| 11 | 26/m | 5 | T | MCS− | 2 | 3 | 3 | 1 | 0 | 2 | 11 | unknown | 2 | R | P | 0.33 | Mi |
| 12 | 47/m | 9 | A | MCS− | 2 | 3 | 2 | 1 | 0 | 2 | 10 | unknown | 1 | R | P | 0.61 | Se |
| 13 | 48/m | 13 | hV | MCS− | 2 | 2 | 2 | 1 | 0 | 2 | 9 | unknown | 1 | R | F | 0.38 | Mi |
| 14 | 24/f | 22 | T | UWS | 1 | 1 | 2 | 2 | 0 | 2 | 8 | MCS | 3 | L | F | 0.47 | Mi |
| 15 | 25/f | 12 | T | UWS | 1 | 1 | 2 | 1 | 0 | 2 | 7 | unknown | 2 | R | F | 0.20 | Se |
| 16 | 47/m | 3 | A | UWS | 2 | 0 | 2 | 1 | 0 | 2 | 7 | unknown | 2 | R | F | 0.21 | Se |
| 17 | 49/f | 7 | other § | UWS | 1 | 0 | 2 | 1 | 0 | 2 | 6 | unknown | 2 | R | F | 0.26 | Se |
| 18 | 34/f | 9 | A | UWS | 1 | 0 | 2 | 1 | 0 | 2 | 6 | UWS | 2 | L | F | 0.56 | Mo |
| 19 | 47/m | 30 | iV | UWS | 1 | 0 | 2 | 1 | 0 | 2 | 6 | unknown | 2 | R | P | 0.24 | Mo |
| 20 | 19/f | 6 | T | UWS | 1 | 0 | 2 | 1 | 0 | 2 | 6 | UWS | 1 | R | F | 0.45 | Se |
| 21 | 47/f | 26 | hV | UWS | 0 | 0 | 2 | 1 | 0 | 2 | 5 | unknown | 1 | L | P | 0.20 | Se |
| 22 | 22/m | 12 | T | UWS | 1 | 0 | 2 | 1 | 0 | 1 | 5 | unknown | 1 | R | F | 0.21 | Se |
| 23 | 51/m | 10 | A | UWS | 0 | 1 | 1 | 1 | 0 | 2 | 5 | unknown | 1 | L | P | 0.21 | Se |
| 24 | 27/f | 2 | A | UWS | 0 | 0 | 1 | 1 | 0 | 2 | 4 | unknown | 1 | L | P | 0.45 | n/a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sinitsyn, D.O.; Poydasheva, A.G.; Bakulin, I.S.; Legostaeva, L.A.; Iazeva, E.G.; Sergeev, D.V.; Sergeeva, A.N.; Kremneva, E.I.; Morozova, S.N.; Lagoda, D.Y.; et al. Detecting the Potential for Consciousness in Unresponsive Patients Using the Perturbational Complexity Index. Brain Sci. 2020, 10, 917. https://doi.org/10.3390/brainsci10120917
Sinitsyn DO, Poydasheva AG, Bakulin IS, Legostaeva LA, Iazeva EG, Sergeev DV, Sergeeva AN, Kremneva EI, Morozova SN, Lagoda DY, et al. Detecting the Potential for Consciousness in Unresponsive Patients Using the Perturbational Complexity Index. Brain Sciences. 2020; 10(12):917. https://doi.org/10.3390/brainsci10120917
Chicago/Turabian StyleSinitsyn, Dmitry O., Alexandra G. Poydasheva, Ilya S. Bakulin, Liudmila A. Legostaeva, Elizaveta G. Iazeva, Dmitry V. Sergeev, Anastasia N. Sergeeva, Elena I. Kremneva, Sofya N. Morozova, Dmitry Yu. Lagoda, and et al. 2020. "Detecting the Potential for Consciousness in Unresponsive Patients Using the Perturbational Complexity Index" Brain Sciences 10, no. 12: 917. https://doi.org/10.3390/brainsci10120917
APA StyleSinitsyn, D. O., Poydasheva, A. G., Bakulin, I. S., Legostaeva, L. A., Iazeva, E. G., Sergeev, D. V., Sergeeva, A. N., Kremneva, E. I., Morozova, S. N., Lagoda, D. Y., Casarotto, S., Comanducci, A., Ryabinkina, Y. V., Suponeva, N. A., & Piradov, M. A. (2020). Detecting the Potential for Consciousness in Unresponsive Patients Using the Perturbational Complexity Index. Brain Sciences, 10(12), 917. https://doi.org/10.3390/brainsci10120917





