Exercise Intensity May Not Moderate the Acute Effects of Functional Circuit Training on Cognitive Function: A Randomized Crossover Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Standards and Study Design
2.2. Participants
2.3. Intervention
2.4. Outcomes
2.5. Data Processing and Statistics
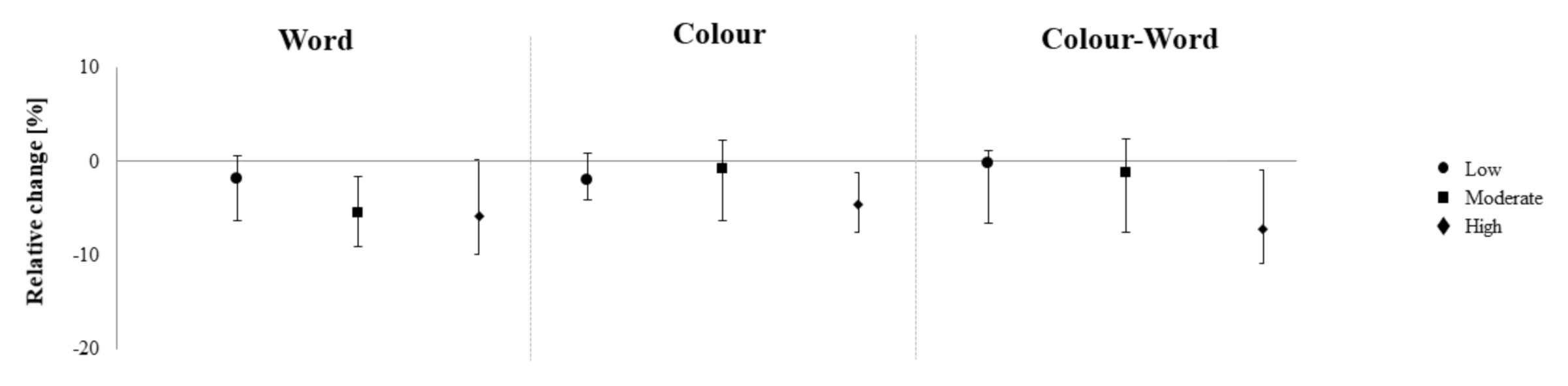
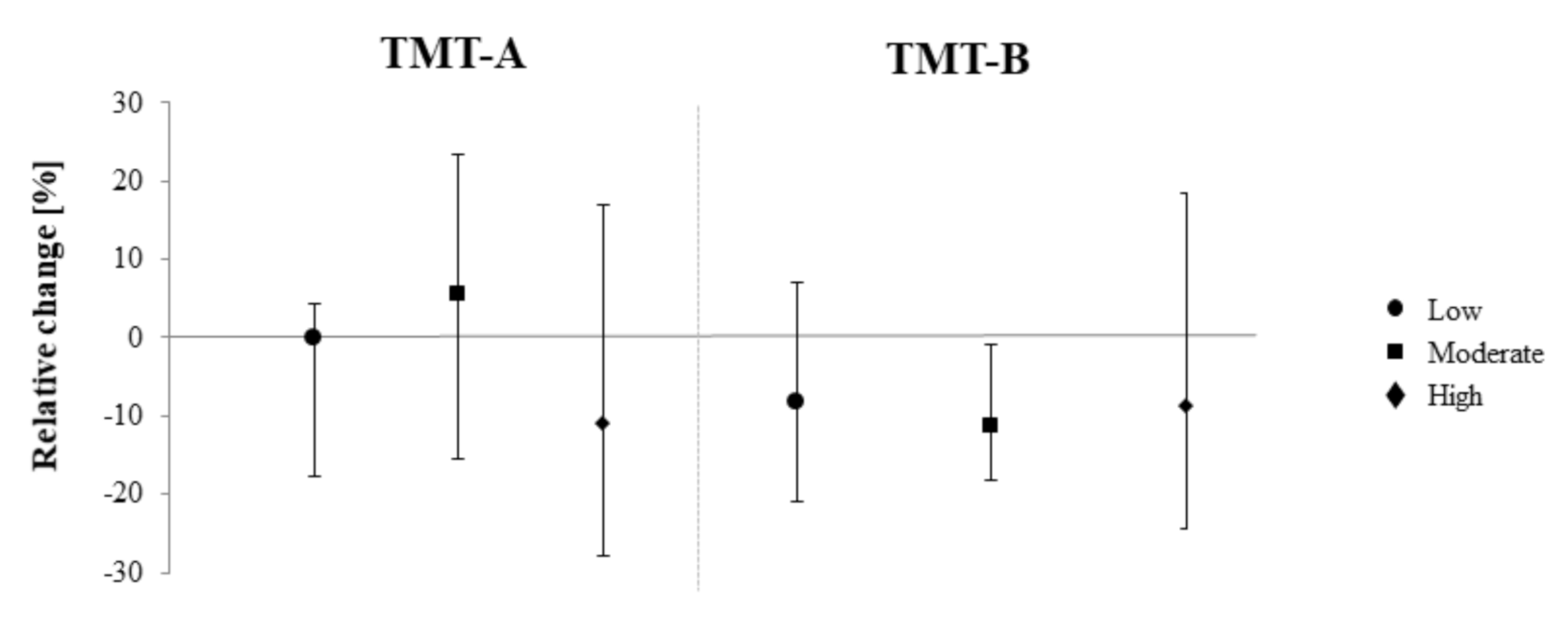
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Data Availability Statement
References
- Basso, J.C.; Suzuki, W.A. The effects of acute exercise on mood, cognition, neurophysiology, and neurochemical pathways: A review. Brain Plast. 2017, 2, 127–152. [Google Scholar] [CrossRef]
- Chang, Y.K.; Labban, J.D.; Gapin, J.I.; Etnier, J.L. The effects of acute exercise on cognitive performance: A meta-analysis. Brain Res. 2012, 1453, 87–101. [Google Scholar] [CrossRef]
- Wilke, J.; Giesche, F.; Klier, K.; Vogt, L.; Herrmann, E.; Banzer, W. Acute effects of resistance exercise on cognitive function in healthy adults: A systematic review with multilevel meta-analysis. Sports Med. 2019, 49, 905–916. [Google Scholar] [CrossRef]
- Brisswalter, J.; Collardeau, M.; René, A. Effects of acute physical exercise characteristics on cognitive performance. Sports Med. 2002, 32, 555–566. [Google Scholar] [CrossRef] [PubMed]
- Northey, J.M.; Cherbuin, N.; Pumpa, K.L.; Smee, D.J.; Rattray, B. Exercise interventions for cognitive function in adults older than 50: A systematic review with meta-analysis. Br. J. Sports Med. 2018, 52, 154–160. [Google Scholar] [CrossRef]
- Loprinzi, P.D.; Blough, J.; Crawford, L.; Ryu, S.; Zou, L.; Li, H. The Temporal Effects of Acute Exercise on Episodic Memory Function: Systematic Review with Meta-Analysis. Brain Sci. 2019, 9, 87. [Google Scholar] [CrossRef]
- Etnier, J.L.; Salazar, W.; Landers, D.M.; Petruzzello, S.J.; Han, M.; Nowell, P. The influence of physical fitness and exercise upon cognitive functioning: A meta-analysis. J Sport Exerc. Psychol. 1997, 19, 249–277. [Google Scholar] [CrossRef]
- Landrigan, J.; Bell, T.; Crowe, M.; Clay, O.J.; Mirman, D. Lifting cognition: A meta-analysis of effects of resistance exercise on cognition. Psychol. Res. 2020, 84, 1167–1183. [Google Scholar] [CrossRef]
- Guadagni, V.; Drogos, L.L.; Tyndall, A.V.; Davenport, M.H.; Anderson, T.J.; Eskes, G.A.; Lognman, R.; Hill, M.D.; Hogan, D.B.; Poulin, M.J. Aerobic exercise improves cognition andcerebrovascular regulation in older adults. Neurology 2020, 94, e2245–e2257. [Google Scholar] [CrossRef]
- Van Praag, H. Neurogenesis and exercise: Past and future directions. Neuromol. Med. 2008, 10, 128–140. [Google Scholar] [CrossRef]
- Kennedy, G.; Hardman, R.J.; Macpherson, H.; Scholey, A.B.; Pigingas, A. How does exercise reduce the rate of age-associated cognitive decline? A review of potential mechanism. J. Alzheimer Dis. 2016, 55, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Oberste, M.; Javelle, F.; Sharma, S.; Joisten, N.; Walzik, D.; Bloch, W.; Zimmer, P. Effects and Moderators of Acute Aerobic Exercise on Subsequent Interference Control: A Systematic Review and Meta-Analysis. Front. Psychol. 2019. [Google Scholar] [CrossRef]
- McMorris, T.; Hale, B.J. Differential effects of differing intensities of acute exercise on speed and accuracy of cognition: A meta-analytical investigation. Brain Cogn. 2012, 80, 338–351. [Google Scholar] [CrossRef]
- Thompson, W.R. worldwide survey of fitness trends for 2019. Acsm’s Health Fit. J. 2018, 22, 10–17. [Google Scholar] [CrossRef]
- Feito, Y.; Heinrich, K.M.; Butcher, S.J.; Poston, W.S.C. High-intensity functional training (HIFT): Definition and research implications for improved fitness. Sports 2018, 6, 76. [Google Scholar] [CrossRef]
- Wilke, J.; Mohr, L. Chronic effects of high-intensity functional training on motor function: A systematic review with multilevel meta-analysis. Sci. Rep. 2020. under review. [Google Scholar]
- Wilke, J. Functional high-intensity exercise is more effective in acutely increasing working memory than aerobic walking: An exploratory randomized, controlled trial. Sci. Rep. 2020, 10. [Google Scholar] [CrossRef]
- Schulz, K.F.; Altman, D.G.; Moher, D. CONSORT 2010 Statement: Updated guidelines for reporting parallel group randomised trials. BMC Med. 2010, 8, 18. [Google Scholar] [CrossRef] [PubMed]
- Valdez, P.; Ramírez, C.; García, A. Circadian rhythms in cognitive performance: Implications for neuropsychological assessment. Chronophysiol. Ther. 2012, 2, 81–92. [Google Scholar] [CrossRef]
- ACSM’s Guidelines for Exercise Testing and Prescription, 9th ed.; Pescatello, L.S., Ed.; Wolters Kluwer/Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2014; ISBN 978-1-60913-605-5. [Google Scholar]
- Tanaka, H.; Monahan, K.D.; Seals, D.R. Age-predicted maximal heart rate revisited. J. Am. Coll. Cardiol. 2001, 37, 153–156. [Google Scholar] [CrossRef]
- Wöstmann, N.M.; Aichert, D.S.; Costa, A.; Rubia, K.; Möller, H.-J.; Ettinger, U. Reliability and plasticity of response inhibition and interference control. Brain Cogn. 2013, 81, 82–94. [Google Scholar] [CrossRef]
- Wagner, S.; Helmreich, I.; Dahmen, N.; Lieb, K.; Tadic, A. Reliability of three alternate forms of the trail making tests a and B. Arch. Clin. Neuropsychol. 2011, 26, 314–321. [Google Scholar] [CrossRef]
- Sánchez-Cubillo, I.; Periáñez, J.A.; Adrover-Roig, D.; Rodríguez-Sánchez, J.M.; Ríos-Lago, M.; Tirapu, J.; Barceló, F. Construct validity of the trail making test: Role of task-switching, working memory, inhibition/interference control, and visuomotor abilities. J. Int. Neuropsychol. Soc. 2009, 15, 438–450. [Google Scholar] [CrossRef]
- Unsworth, N.; Engle, R.W. On the division of short-term and working memory: An examination of simple and complex span and their relation to higher order abilities. Psychol. Bull. 2007, 133, 1038–1066. [Google Scholar] [CrossRef]
- Youngjohn, J.R.; Larrabee, G.J.; Crook, T.H. Test-retest reliability of computerized, everyday memory measures and traditional memory tests. Clin. Neuropsychol. 1992, 6, 276–286. [Google Scholar] [CrossRef]
- Hausknecht, J.P.; Halpert, J.A.; Di Paolo, N.T.; Gerrard, M.O.M. Retesting in selection: A meta-analysis of coaching and practice effects for tests of cognitive ability. J. Appl. Psychol. 2007, 92, 373–385. [Google Scholar] [CrossRef]
- Chen, M.J.; Fan, X.; Moe, S.T. Criterion-related validity of the Borg ratings of perceived exertion scale in healthy individuals: A meta-analysis. J. Sports Sci. 2002, 20, 873–899. [Google Scholar] [CrossRef]
- Woodruff, R.S. Confidence Intervals for Medians and Other Position Measures. J. Am. Stat. Ass. 1952, 47, 635–646. [Google Scholar] [CrossRef]
- Pesce, C. Shifting the focus from quantitative to qualitative exercise characteristics in exercise and cognition research. J. Sport Exerc. Psychol. 2012, 34, 766–786. [Google Scholar] [CrossRef]
- Wilke, J.; Stricker, V.; Usedly, S. Free-weight resistance exercise is more effective in enhancing inhibitory control than machine-based training: A randomized, controlled trial. Brain Sci. 2020, 10, 702. [Google Scholar] [CrossRef]
- Leff, D.R.; Orihuela-Espina, F.; Elwell, C.E.; Athanasiou, T.; Delpy, D.T.; Darzi, A.W.; Yang, G.-Z. Assessment of the cerebral cortex during motor task behaviours in adults: A systematic review of functional near infrared spectroscopy (fNIRS) studies. Neuroimage 2011, 54, 2922–2936. [Google Scholar] [CrossRef]
- Holper, L.; Biallas, M.; Wolf, M. Task complexity relates to activation of cortical motor areas during uni- and bimanual performance: A functional NIRS study. Neuroimage 2009, 46, 1105–1113. [Google Scholar] [CrossRef]
- Verstynen, T.; Diedrichsen, J.; Albert, N.; Aparicio, P.; Ivry, R.B. Ipsilateral motor cortex activity during unimanual hand movements relates to task complexity. J. Neurophysiol. 2005, 93, 1209–1222. [Google Scholar] [CrossRef]
- Jäncke, L.; Himmelbach, M.; Shah, N.J.; Zilles, K. The effect of switching between sequential and repetitive movements on cortical activation. Neuroimage 2000, 12, 528–537. [Google Scholar] [CrossRef] [PubMed]
| Parameter | Value |
|---|---|
| Weight (kg) | 70 ± 11 |
| Height (cm) | 174 ± 10 |
| BMI | 23 ± 2 |
| Resting heart rate (bpm) | 67 ± 9 |
| Maximal heart rate (bpm) | 189 ± 3 |
| Perceived exertion during exercise (RPE scale) | L: 9 ± 2, M: 12 ± 2, H: 16 ± 2 |
| Light | Moderate | High | p Value | |
|---|---|---|---|---|
| Stroop Word (t) | 25.1 (21.1/31.5) | 25.8 (19.3/30.8) | 25.7 (19.5/33.5) | 0.72 |
| Stroop Color (t) | 36.3 (25.5/46.1) | 34.6 (24.5/44.3) | 35.6 (26.2/52.3) | 0.13 |
| Stroop Interference (t) | 55.1 (36.9/60.2) | 52.0 (39.1/65.4) | 54.5 (36.7/70.7) | 0.25 |
| Trail Making Test A (t) | 22.1 (15.0/44.5) | 21.6 (13.0/44.5) | 22.9 (13.9/60.6) | 0.10 |
| Trail Making Test B (t) | 24.5 (11.1/44.8) | 21.8 (12.6/47.2) | 21.5 (12.5/48.2) | 0.42 |
| Digit Span Score (pts) | 11.5 (5/19) | 11.5 (5/20) | 11.0 (5/22) | 0.95 |
| Light | Moderate | High | |
|---|---|---|---|
| Stroop Word (t) | −0.51 (−1.57 to 0.18) | −1.22 (−2.66 to 0.05) | −1.35 (−3.9 to 4.39) |
| Stroop Color (t) | −1.17 (−2.87 to 0.16) | −0.28 (−2.0 to 0.78) | −1.65 (−2.81 to −0.22) |
| Stroop Interference (t) | −0.15 (−3.78 to 0.65) | −0.71 (−4.21 to 1.49) | −3.72 (−6.31 to −0.38) |
| Trail Making Test A (t) | 0.11 (−4.05 to 6.09) | 2.64 (−3.05 to 6.48) | −1.46 (−8.43 to 4.36) |
| Trail Making Test B (t) | −2.71 (−5.78 to 1.87) | −2.56 (−6.75 to −0.93) | −1.80 (−8.44 to 3.13) |
| Digit Span Score (pts) | 0 (−2 to 1.75) | 0 (−4 to 5) | −0.5 (−1 to 1.75) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wilke, J.; Royé, C. Exercise Intensity May Not Moderate the Acute Effects of Functional Circuit Training on Cognitive Function: A Randomized Crossover Trial. Brain Sci. 2020, 10, 738. https://doi.org/10.3390/brainsci10100738
Wilke J, Royé C. Exercise Intensity May Not Moderate the Acute Effects of Functional Circuit Training on Cognitive Function: A Randomized Crossover Trial. Brain Sciences. 2020; 10(10):738. https://doi.org/10.3390/brainsci10100738
Chicago/Turabian StyleWilke, Jan, and Caroline Royé. 2020. "Exercise Intensity May Not Moderate the Acute Effects of Functional Circuit Training on Cognitive Function: A Randomized Crossover Trial" Brain Sciences 10, no. 10: 738. https://doi.org/10.3390/brainsci10100738
APA StyleWilke, J., & Royé, C. (2020). Exercise Intensity May Not Moderate the Acute Effects of Functional Circuit Training on Cognitive Function: A Randomized Crossover Trial. Brain Sciences, 10(10), 738. https://doi.org/10.3390/brainsci10100738





