Breastfeeding Duration Is Associated with Regional, but Not Global, Differences in White Matter Tracts
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants and Protocol
2.2. Infant Feeding Methods
2.3. Magnetic Resonance Imaging Scan Procedures
2.4. Anatomical and Diffusion Weighted Scan Acquisition
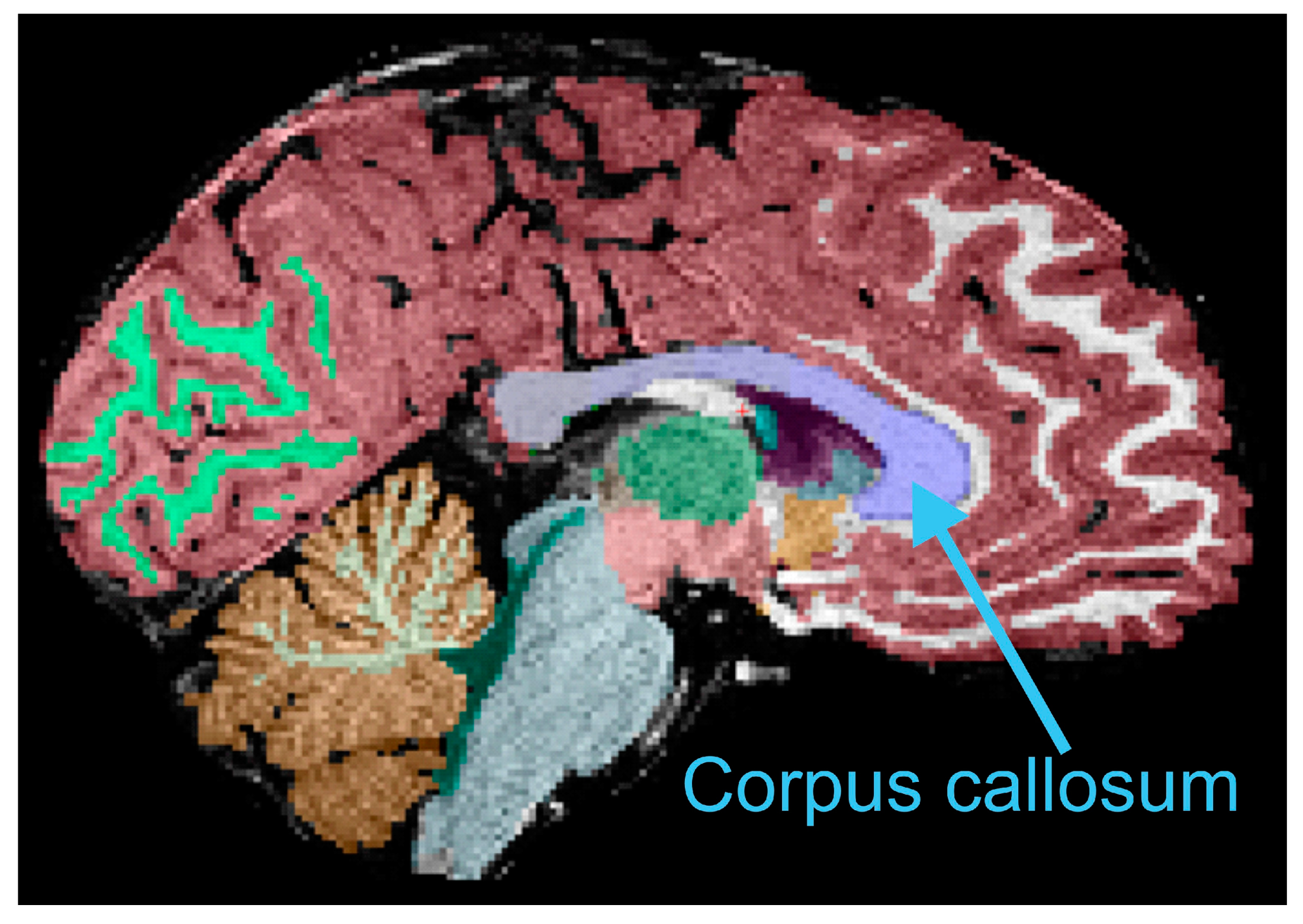
2.5. Anatomical Imaging Processing and Corpus Callosum Measurements
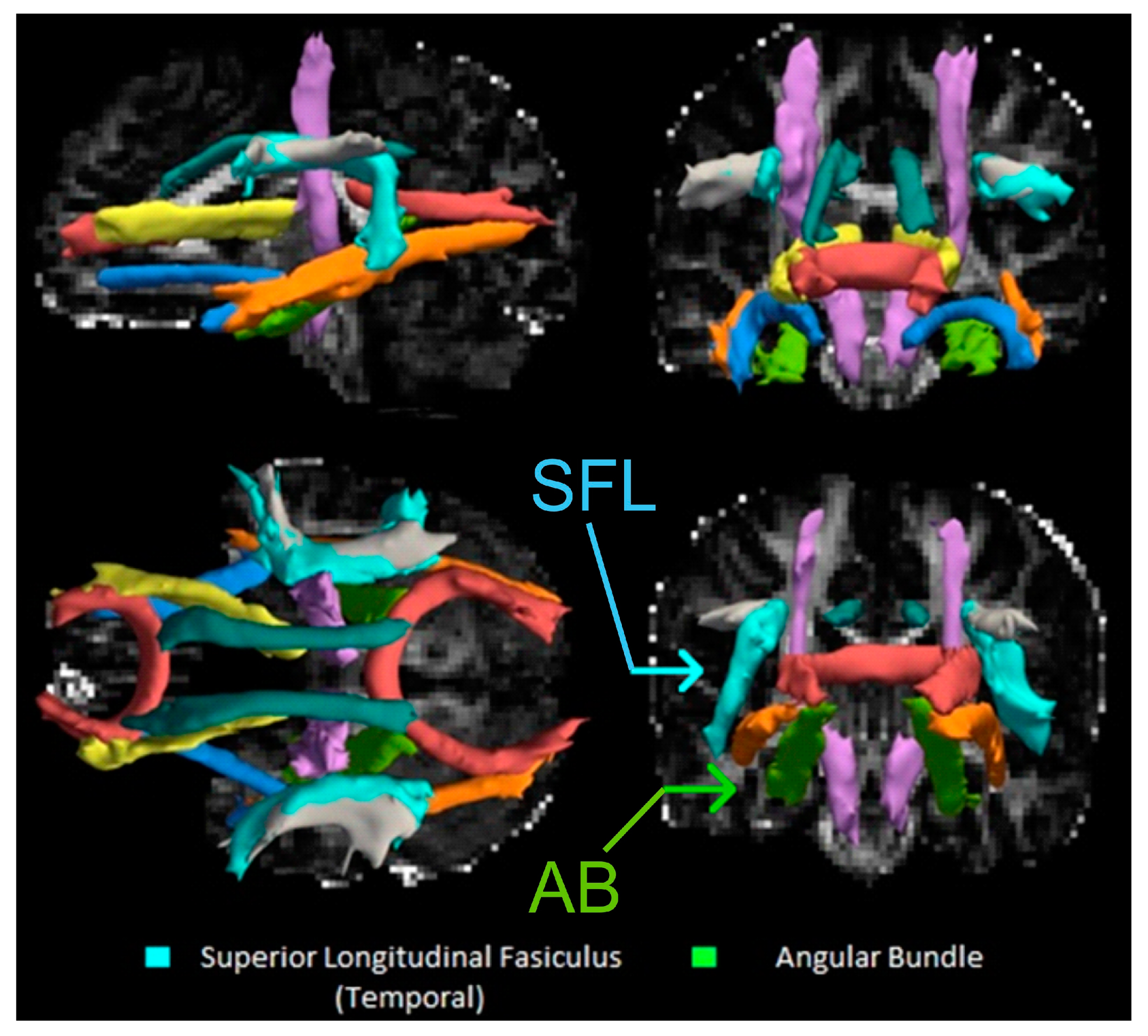
2.6. DTI Processing
2.7. Statistical Analyses
3. Results
3.1. Breastfeeding Duration and CC Volume
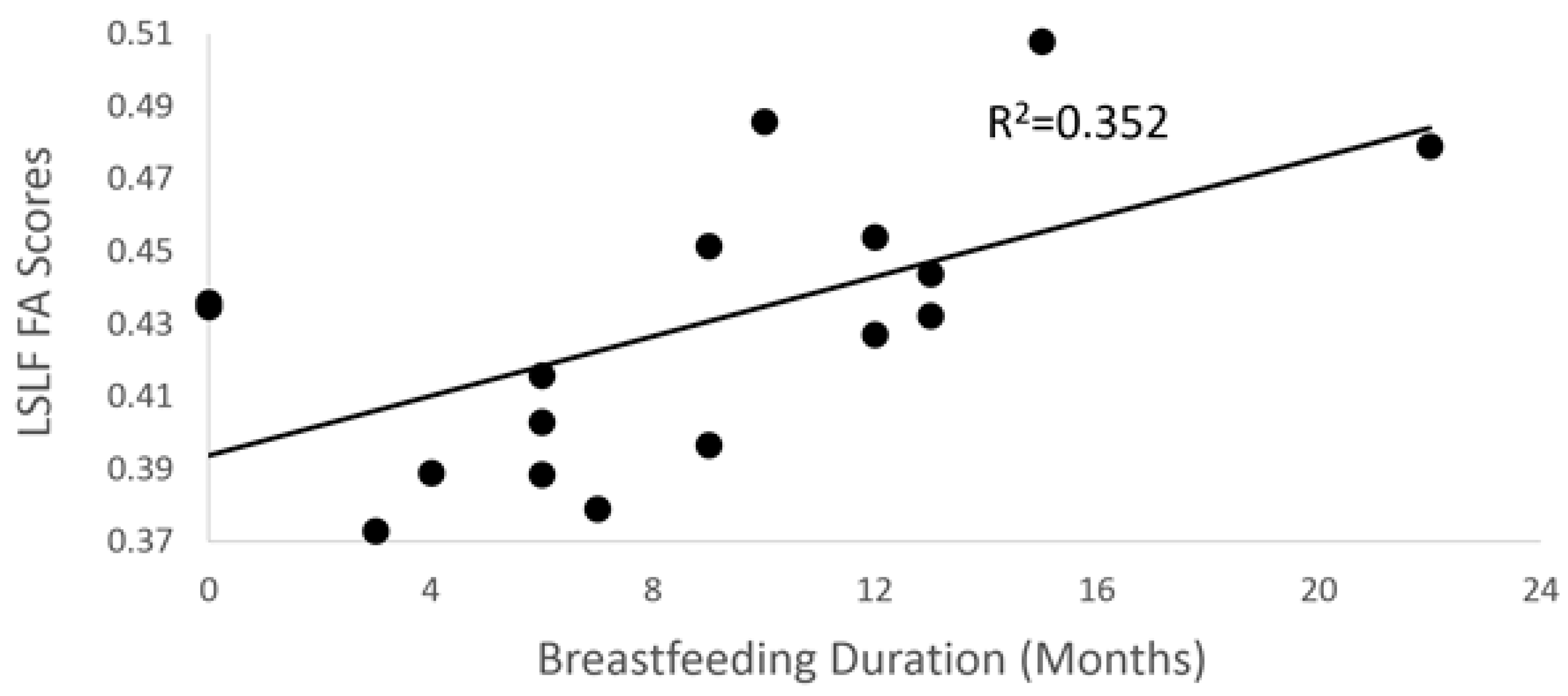
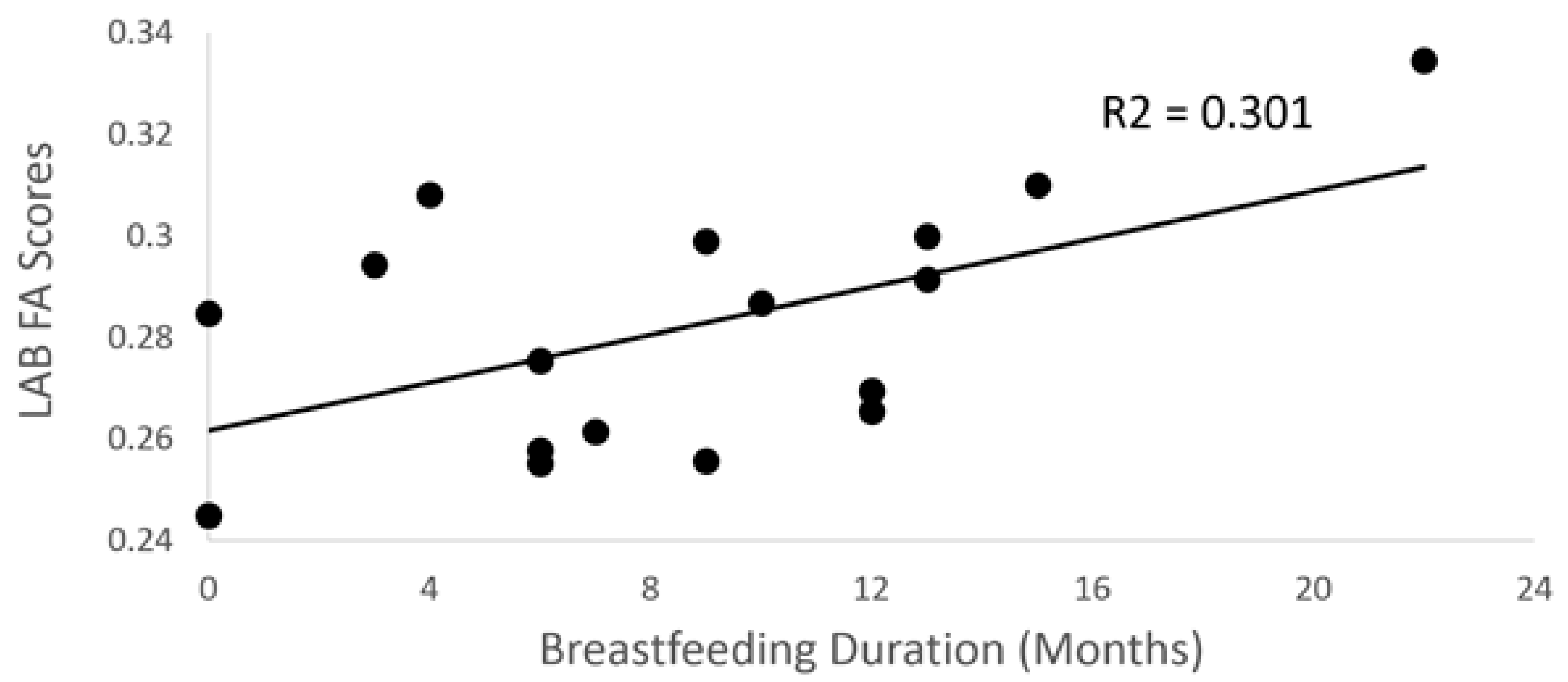
3.2. Breastfeeding Duration and DTI
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mortensen, E.L.; Michaelsen, K.F.; Sanders, S.A.; Reinisch, J.M. The Association Between Duration of Breastfeeding and Adult Intelligence. JAMA J. Am. Med. Assoc. 2002, 287, 2365–2372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kafouri, S.; Kramer, M.; Leonard, G.; Perron, M.; Pike, B.; Richer, L.; Toro, R.; Veillette, S.; Pausova, Z.; Paus, T. Breastfeeding and Brain Structure in Adolescence. Int. J. Epidemiol. 2013, 42, 150–159. [Google Scholar] [CrossRef] [PubMed]
- Isaacs, E.B.; Fischl, B.R.; Quinn, B.T.; Chong, W.K.; Gadian, D.G. Impact of Breast Milk on IQ, Brain Size and White Matter Development. Pediatr. Res. 2010, 67, 357–362. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luby, J.L.; Belden, A.C.; Whalen, D.; Harms, M.P.; Barch, D.M. Breastfeeding and Childhood IQ: The Mediating Role of Gray Matter Volume. J. Am. Acad. Child Adolesc. Psychiatry 2016, 55, 367–375. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oddy, W.H.; Li, J.; Whitehouse, A.J.O.; Zubrick, S.R.; Malacova, E. Breastfeeding Duration and Academic Achievement at 10 Years. Pediatrics 2011, 127, e137–e145. [Google Scholar] [CrossRef] [Green Version]
- Kramer, M.S.; Aboud, F.A.; Mironova, E.; Vanilovich, I.; Platt, R.W.; Matush, L.; Igumnov, S.; Fombonne, E.; Bogdanovich, N.; Ducruet, T.; et al. Breastfeeding and Child Cognitive Development. Arch. Gen. Psychiatry 2008, 65, 578. [Google Scholar] [CrossRef]
- McCrory, C.; Murray, A. The Effect of Breastfeeding on Neuro-Development in Infancy. Matern. Child Health J. 2013, 17, 1680–1688. [Google Scholar] [CrossRef]
- Deoni, S.C.L.; Dean, D.C.; Piryatinsky, I.; O’Muircheartaigh, J.; Waskiewicz, N.; Lehman, K.; Han, M.; Dirks, H. Breastfeeding and Early White Matter Development: A Cross-Sectional Study. Neuroimage 2013, 82, 77–86. [Google Scholar] [CrossRef] [Green Version]
- Smith, R.; Lucas, R. Evaluation of Nursing Knowledge of Early Initiation of Breastfeeding in Preterm Infants in a Hospital Setting. J. Neonatal Nurs. 2016, 22, 138–143. [Google Scholar] [CrossRef]
- Liu, Z.; Neuringer, M.; Erdman, J.W.; Kuchan, M.J.; Renner, L.; Johnson, E.E.; Wang, X.; Kroenke, C.D. The Effects of Breastfeeding versus Formula-Feeding on Cerebral Cortex Maturation in Infant Rhesus Macaques. Neuroimage 2019, 184, 372–385. [Google Scholar] [CrossRef]
- Deoni, S.; Dean, D.I.; Joelson, S.; O’Regan, J.; Schneider, N. Early Nutrition Influences Developmental Myelination and Cognition in Infants and Young Children. Neuroimage 2018, 178, 649–659. [Google Scholar] [CrossRef]
- McNamara, R.K.; Carlson, S.E. Role of Omega-3 Fatty Acids in Brain Development and Function: Potential Implications for the Pathogenesis and Prevention of Psychopathology. Prostaglandins Leukot. Essent. Fat. Acids 2006, 75, 329–349. [Google Scholar] [CrossRef] [PubMed]
- Brenna, J.T.; Varamini, B.; Jensen, R.G.; Diersen-schade, D.A.; Boettcher, J.A.; Arterburn, L.M. Docosahexaenoic and Arachidonic Acid Concentrations in Human Breast Milk Worldwide. Am. J. Clin. Nutr. 2007, 85, 1457–1464. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stewart, A.L.; Rifkin, L.; Amess, P.N.; Kirkbride, V.; Townsend, J.P.; Miller, D.H.; Lewis, S.W.; Kingsley, D.P.E.; Moseley, I.F.; Foster, O.; et al. Brain Structure and Neurocognitive and Behavioral Function in Adolescents Who Were Born Very Preterm. Lancet 1999, 353, 1653–1657. [Google Scholar] [CrossRef]
- Northam, G.B.; Liégeois, F.; Chong, W.K.; Wyatt, J.S.; Baldeweg, T. Total Brain White Matter is a Major Determinant of IQ in Adolescents Born Preterm. Ann. Neurol. 2011, 69, 702–711. [Google Scholar] [CrossRef]
- Peterson, B.S.; Vohr, B.; Staib, L.H.; Cannistraci, C.J.; Dolberg, A.; Schneider, K.C.; Katz, K.H.; Westerveld, M.; Sparrow, S.; Anderson, A.W.; et al. Regional Brain Volume Abnormalities and Long-Term Cognitive Outcome in Preterm Infants. JAMA 2000, 284, 1939–1947. [Google Scholar] [CrossRef]
- Stadler, D.D.; Musser, E.D.; Holton, K.F.; Shannon, J.; Nigg, J.T. Recalled Initiation and Duration of Maternal Breastfeeding among Children with and without ADHD in a Well Characterized Case-Control Sample. J. Abnorm. Child Psychol. 2016, 44, 347–355. [Google Scholar] [CrossRef] [Green Version]
- Montgomery-Downs, H.E.; O’Brien, L.M.; Holbrook, C.R.; Gozal, D. Snoring and Sleep-Disordered Breathing in Young Children: Subjective and Objective Correlates. Sleep 2004, 27, 87–94. [Google Scholar] [CrossRef] [Green Version]
- Fischl, B.; Van Der Kouwe, A.; Destrieux, C.; Halgren, E.; Ségonne, F.; Salat, D.H.; Busa, E.; Seidman, L.J.; Goldstein, J.; Kennedy, D.; et al. Automatically Parcellating the Human Cerebral Cortex. Cereb. Cortex 2004, 14, 11–22. [Google Scholar] [CrossRef] [Green Version]
- Fischl, B.; Salat, D.H.; Busa, E.; Albert, M.; Dieterich, M.; Haselgrove, C.; Van Der Kouwe, A.; Killiany, R.; Kennedy, D.; Klaveness, S.; et al. Whole Brain Segmentation: Automated Labeling of Neuroanatomical Structures in the Human Brain. Neuron 2002, 33, 341–355. [Google Scholar] [CrossRef] [Green Version]
- Yendiki, A.; Panneck, P.; Srinivasan, P.; Stevens, A.; Zollei, L.; Augustinack, J.; Wang, R.; Salat, D.; Ehrlich, S.; Behrens, T.; et al. Automated Probabilistic Reconstruction of White-Matter Pathways in Health and Disease Using an Atlas of the Underlying Anatomy. Front. Neuroinform. 2011, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yendiki, A.; Koldewyn, K.; Kakunoori, S.; Kanwisher, N.; Fischl, B. Spurious Group Differences Due to Head Motion in a Diffusion MRI Study. Neuroimage 2013, 88, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Colen, C.G.; Ramey, D.M. Is Breast Truly Best? Estimating the Effects of Breastfeeding on Long-Term Child Health and Wellbeing in the United States Using Sibling Comparisons. Soc. Sci. Med. 2014, 109, 55–65. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McFadden, A.; Toole, G. Exploring Women’s Views of Breastfeeding: A Focus Group Study within an Area with High Levels of Socio-Economic Deprivation. Matern. Child Nutr. 2006, 2, 156–168. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, E.V.; Rosenbloom, M.J.; Desmond, J.E.; Pfefferbaum, A. Sex Differences in Corpus Callosum Size: Relationship to Age and Intracranial Size. Neurobiol. Aging 2001, 22, 603–611. [Google Scholar] [CrossRef]
- Jarbo, K.; Verstynen, T.; Schneider, W. In Vivo Quantification of Global Connectivity in the Human Corpus Callosum. Neuroimage 2012, 59, 1988–1996. [Google Scholar] [CrossRef]
- Wolff, J.J.; Gerig, G.; Lewis, J.D.; Soda, T.; Styner, M.A.; Vachet, C.; Botteron, K.N.; Elison, J.T.; Dager, S.R.; Estes, A.M.; et al. Altered Corpus Callosum Morphology Associated with Autism over the First 2 Years of Life. Brain 2015, 138, 2046–2058. [Google Scholar] [CrossRef] [Green Version]
- Travers, B.G.; Tromp, D.P.M.; Adluru, N.; Lange, N.; Destiche, D.; Ennis, C.; Nielsen, J.A.; Froehlich, A.L.; Prigge, M.B.D.; Fletcher, P.T.; et al. Atypical Development of White Matter Microstructure of the Corpus Callosum in Males with Autism: A Longitudinal Investigation. Mol. Autism 2015, 6, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Rilling, J.K.; Glasser, M.F.; Preuss, T.M.; Ma, X.; Zhao, T.; Hu, X.; Behrens, T.E.J. The Evolution of the Arcuate Fasciculus Revealed with Comparative DTI. Nat. Neurosci. 2008, 11, 1–3. [Google Scholar] [CrossRef]
- Bernal, B.; Altman, N. The Connectivity of the Superior Longitudinal Fasciculus: A Tractography DTI Study. Magn. Reson. Imaging 2010, 28, 217–225. [Google Scholar] [CrossRef]
- López-Barroso, D.; Catani, M.; Ripollés, P.; Dell’Acqua, F.; Rodríguez-Fornells, A.; de Diego-Balaguer, R. Word Learning Is Mediated by the Left Arcuate Fasciculus. Proc. Natl. Acad. Sci. USA 2013, 11, 13168–13173. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brown, E.C.; Jeong, J.W.; Muzik, O.; Rothermel, R.; Matsuzaki, N.; Juhász, C.; Sood, S.; Asano, E. Evaluating the Arcuate Fasciculus with Combined Diffusion-Weighted MRI Tractography and Electrocorticography. Hum. Brain Mapp. 2014, 35, 2333–2347. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nagae, L.M.; Zarnow, D.M.; Blaskey, L.; Dell, J.; Khan, S.Y.; Qasmieh, S.; Levy, S.E.; Roberts, T.P.L. Elevated Mean Diffusivity in the Left Hemisphere Superior Longitudinal Fasciculus in Autism Spectrum Disorders Increases with More Profound Language Impairment. Am. J. Neuroradiol. 2012, 33, 1720–1725. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hua, K.; Zhang, J.; Wakana, S.; Jiang, H.; Li, X.; Reich, D.S.; Calabresi, P.A.; Pekar, J.J.; van Zijl, P.C.M.; Mori, S. Tract Probability Maps in Stereotaxic Spaces: Analyses of White Matter Anatomy and Tract-Specific Quantification. Neuroimage 2008, 39, 336–347. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ezzati, A.; Katz, M.J.; Lipton, M.L.; Zimmerman, M.E.; Lipton, R.B. Hippocampal Volume and Cingulum Bundle Fractional Anisotropy Are Independently Associated with Verbal Memory in Older Adults. Brain Imaging Behav. 2016, 10, 652–659. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Phan, T.V.; Smeets, D.; Talcott, J.B.; Vandermosten, M. Processing of Structural Neuroimaging Data in Young Children: Bridging the Gap between Current Practice and State-of-the-Art Methods. Dev. Cogn. Neurosci. 2018, 33, 206–223. [Google Scholar] [CrossRef]
- Mukherjee, P.; Miller, J.; Shimony, J.; Conturo, T.; Lee, B.; Almli, C.; McKinstry, R. Normal Brain Maturation during Childhood: Developmental Trends Characterized with Diffusion-Tensor MR Imaging. Radiology 2001, 221, 349–358. [Google Scholar] [CrossRef]
| Characteristic | Value |
|---|---|
| Age (Years) | 6.4 (1.6) |
| Female (n, %) | 7 (36.8) |
| Annual Income (Dollars; Thousands) | 92.5 (43) * |
| Maternal Education (Years) | 17.4 (1.9) |
| Parental Smoking (n, %) | 3 (15.8) |
| Diagnosed ADD/ADHD (n, %) | 1 (5.3) |
| Exclusive Breastfeeding (n, %) | 7 (36.8) |
| Breastfeeding Duration (Months) | 9.9 (7.1) |
| Exclusive Formula Feeding (n, %) | 2 (10.5) |
| Characteristic | Total | Exclusively Breastfed | Any Formula |
|---|---|---|---|
| n | 16 | 7 | 9 |
| Total CC Volume | 7353 (790) | 7287 (893) | 7405 (752) |
| Anterior CC Volume | 2092 (263) | 2043 (352) | 2130 (181) |
| Anterior-Central CC Volume | 942 (173) | 966 (92) | 924 (221) |
| Central CC Volume | 948 (130) | 955 (117) | 943 (147) |
| Posterior-Central CC Volume | 1200 (244) | 1114 (198) | 1266 (266) |
| Posterior CC Volume | 2171 (344) | 2208 (258) | 2143 (412) |
| eTIV | 1456 (109) | 1486 (102) | 1433 (114) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bauer, C.E.; Lewis, J.W.; Brefczynski-Lewis, J.; Frum, C.; Schade, M.M.; Haut, M.W.; Montgomery-Downs, H.E. Breastfeeding Duration Is Associated with Regional, but Not Global, Differences in White Matter Tracts. Brain Sci. 2020, 10, 19. https://doi.org/10.3390/brainsci10010019
Bauer CE, Lewis JW, Brefczynski-Lewis J, Frum C, Schade MM, Haut MW, Montgomery-Downs HE. Breastfeeding Duration Is Associated with Regional, but Not Global, Differences in White Matter Tracts. Brain Sciences. 2020; 10(1):19. https://doi.org/10.3390/brainsci10010019
Chicago/Turabian StyleBauer, Christopher E., James W. Lewis, Julie Brefczynski-Lewis, Chris Frum, Margeaux M. Schade, Marc W. Haut, and Hawley E. Montgomery-Downs. 2020. "Breastfeeding Duration Is Associated with Regional, but Not Global, Differences in White Matter Tracts" Brain Sciences 10, no. 1: 19. https://doi.org/10.3390/brainsci10010019
APA StyleBauer, C. E., Lewis, J. W., Brefczynski-Lewis, J., Frum, C., Schade, M. M., Haut, M. W., & Montgomery-Downs, H. E. (2020). Breastfeeding Duration Is Associated with Regional, but Not Global, Differences in White Matter Tracts. Brain Sciences, 10(1), 19. https://doi.org/10.3390/brainsci10010019





