The Tolerability and Efficacy of 4 mA Transcranial Direct Current Stimulation on Leg Muscle Fatigability
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Experimental Protocol
2.3. Isokinetic Strength Test
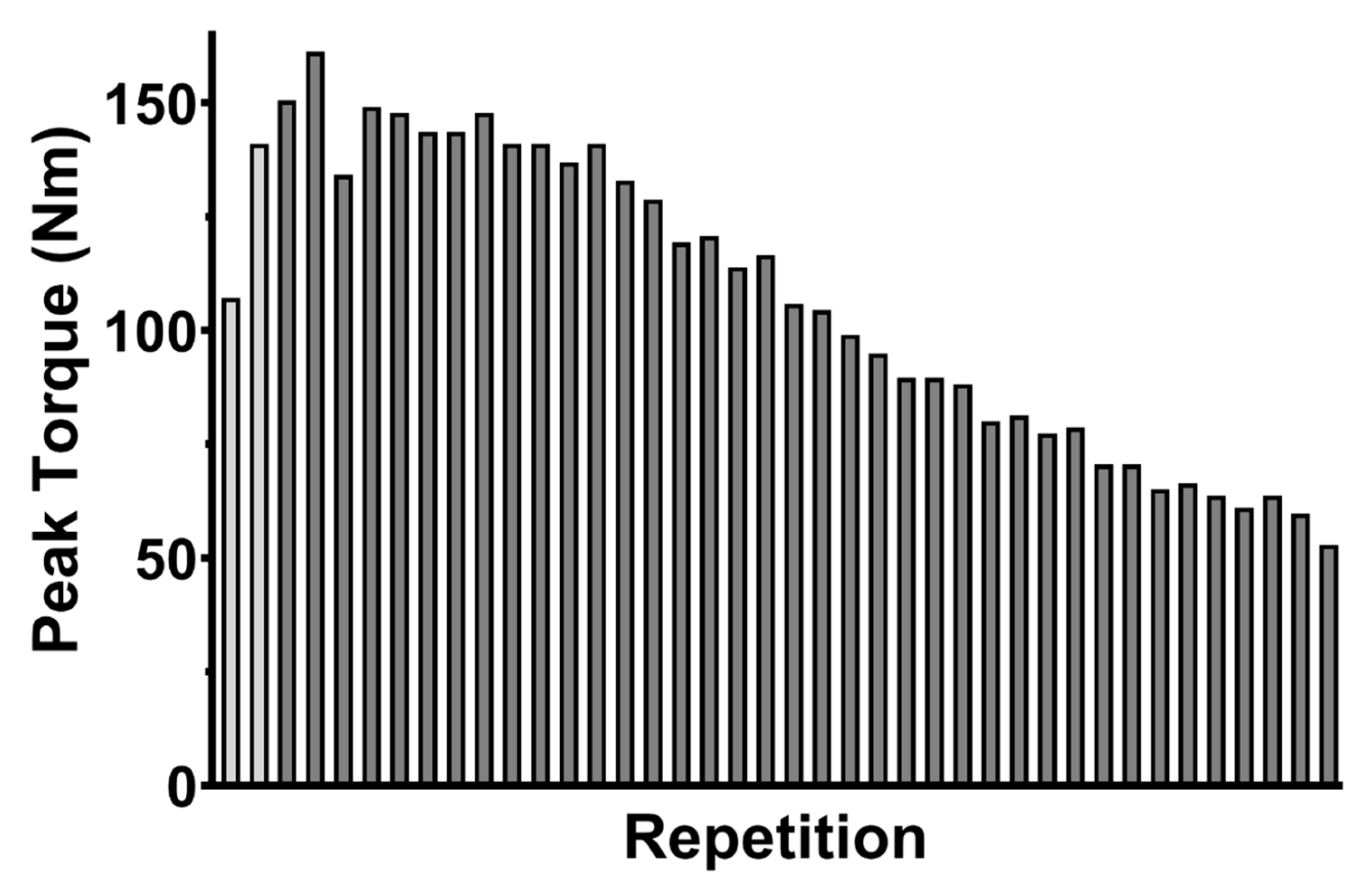
2.4. Isokinetic Fatigue Test
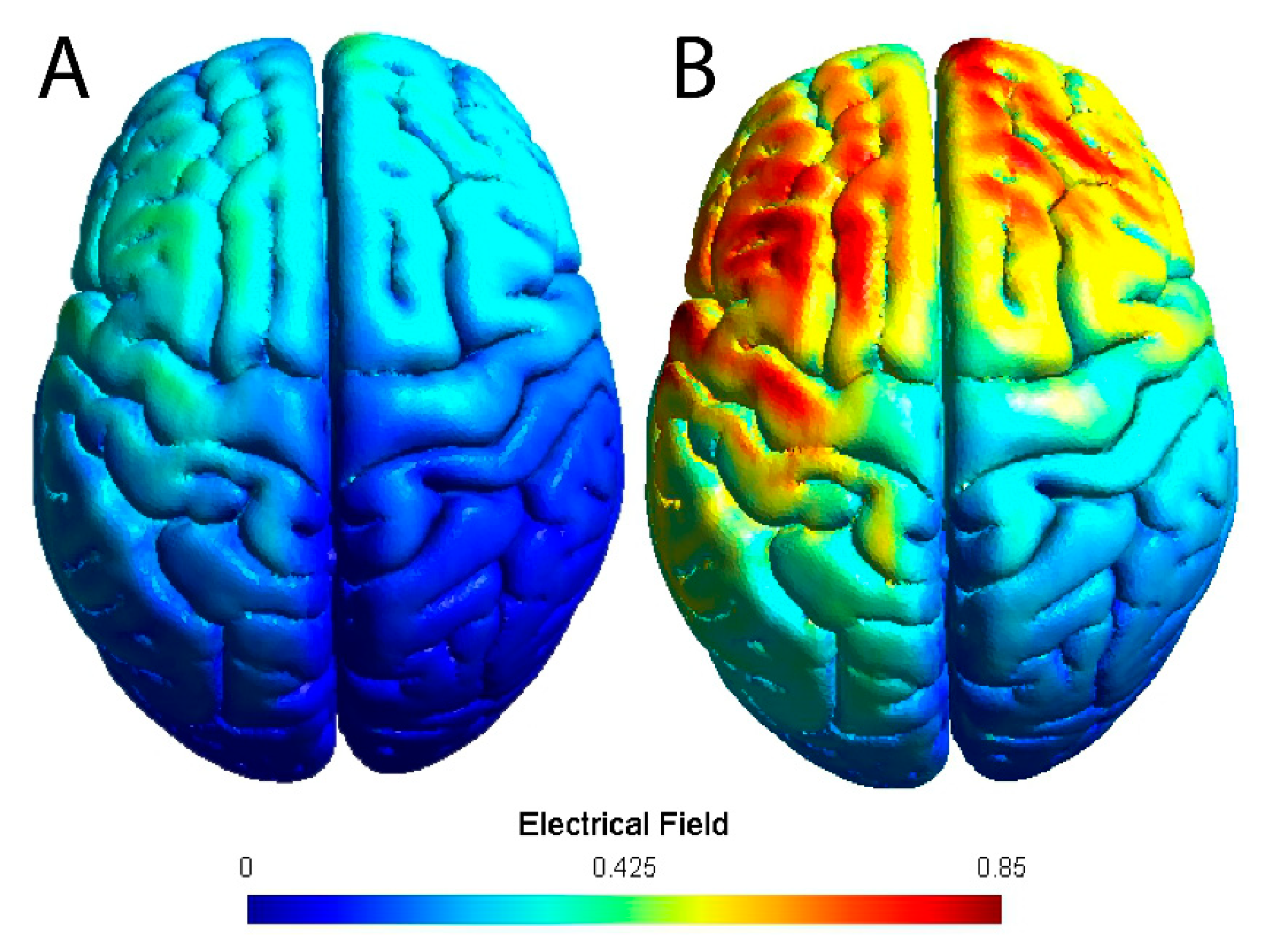
2.5. tDCS Sessions
2.6. Data Analysis
2.7. Statistical Analysis
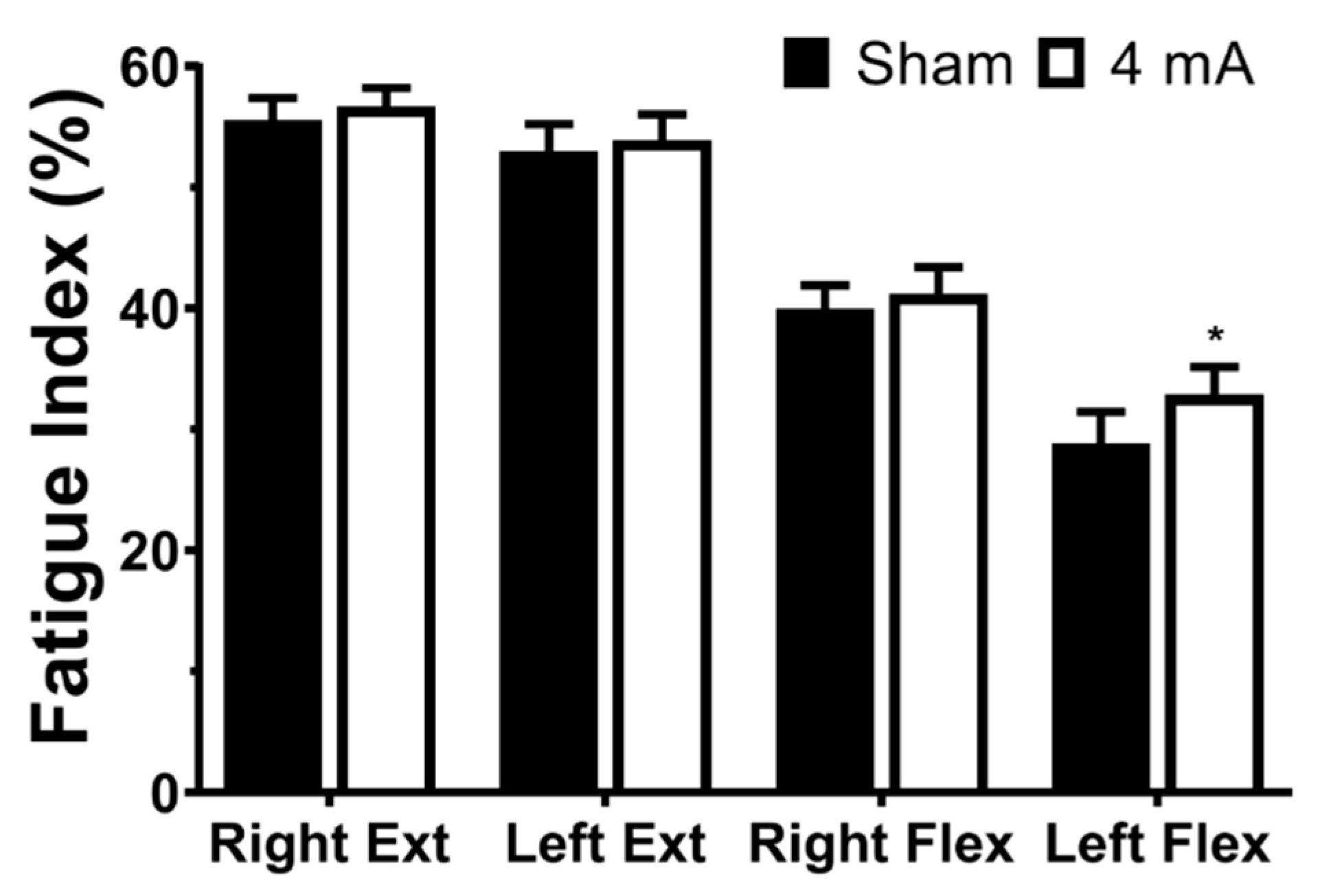
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Poreisz, C.; Boros, K.; Antal, A.; Paulus, W. Safety aspects of transcranial direct current stimulation concerning healthy subjects and patients. Brain Res. Bull. 2007, 72, 208–214. [Google Scholar] [CrossRef] [PubMed]
- Schlaug, G.; Renga, V. Transcranial direct current stimulation: A noninvasive tool to facilitate stroke recovery. Expert Rev. Med. Devices 2008, 5, 759–768. [Google Scholar] [CrossRef] [PubMed]
- Nitsche, M.A.; Liebetanz, D.; Lang, N.; Antal, A.; Tergau, F.; Paulus, W. Safety criteria for transcranial direct current stimulation (tDCS) in humans. Clin. Neurophysiol. 2003, 114, 2220–2222, author reply 2222–2223 (2003). [Google Scholar] [CrossRef]
- Nitsche, M.A.; Liebetanz, D.; Antal, A.; Lang, N.; Tergau, F.; Paulus, W. Modulation of cortical excitability by weak direct current stimulation—Technical, safety and functional aspects. Suppl. Clin. Neurophysiol. 2003, 56, 255–276. [Google Scholar] [PubMed]
- Nitsche, M.A.; Paulus, W. Excitability changes induced in the human motor cortex by weak transcranial direct current stimulation. J. Physiol. 2000, 527, 633–639. [Google Scholar] [CrossRef] [PubMed]
- Dissanayaka, T.; Zoghi, M.; Farrell, M.; Egan, G.F.; Jaberzadeh, S. Does transcranial electrical stimulation enhance corticospinal excitability of the motor cortex in healthy individuals? A systematic review and meta-analysis. Eur. J. Neurosci. 2017, 46, 1968–1990. [Google Scholar] [CrossRef]
- Berryhill, M.E.; Martin, D. Cognitive effects of transcranial direct current stimulation in healthy and clinical populations: An overview. J. ECT 2018, 34, e25–e35. [Google Scholar] [CrossRef]
- Steinberg, F.; Pixa, N.H.; Fregni, F. A review of acute aerobic exercise and transcranial direct current stimulation effects on cognitive functions and their potential synergies. Front. Hum. Neurosci. 2018, 12, 534. [Google Scholar] [CrossRef]
- O’Brien, A.T.; Bertolucci, F.; Torrealba-Acosta, G.; Huerta, R.; Fregni, F.; Thibaut, A. Non-invasive brain stimulation for fine motor improvement after stroke: A meta-analysis. Eur. J. Neurol. 2018, 25, 1017–1026. [Google Scholar] [CrossRef]
- Sanchez-Kuhn, A.; Perez-Fernandez, C.; Canovas, R.; Flores, P.; Sanchez-Santed, F. Transcranial direct current stimulation as a motor neurorehabilitation tool: An empirical review. Biomed. Eng. Online 2017, 16, 76. [Google Scholar] [CrossRef]
- Simpson, M.W.; Mak, M. The effect of transcranial direct current stimulation on upper limb motor performance in parkinson’s disease: A systematic review. J. Neurol. 2019. [Online ahead of print]. [Google Scholar] [CrossRef] [PubMed]
- Bikson, M.; Grossman, P.; Thomas, C.; Zannou, A.L.; Jiang, J.; Adnan, T.; Mourdoukoutas, A.P.; Kronberg, G.; Truong, D.; Boggio, P.; et al. Safety of transcranial direct current stimulation: Evidence based update 2016. Brain Stimul. 2016, 9, 641–661. [Google Scholar] [CrossRef] [PubMed]
- Lippold, O.C.; Redfearn, J.W. Mental changes resulting from the passage of small direct currents through the human brain. Br. J. Psychiatry 1964, 110, 768–772. [Google Scholar] [CrossRef] [PubMed]
- Nitsche, M.A.; Bikson, M. Extending the parameter range for tDCS: Safety and tolerability of 4 mA stimulation. Brain Stimul. 2017, 10, 541–542. [Google Scholar] [CrossRef] [PubMed]
- Fertonani, A.; Ferrari, C.; Miniussi, C. What do you feel if I apply transcranial electric stimulation? Safety, sensations and secondary induced effects. Clin. Neurophysiol. 2015, 126, 2181–2188. [Google Scholar] [CrossRef] [PubMed]
- Chhatbar, P.Y.; Chen, R.; Deardorff, R.; Dellenbach, B.; Kautz, S.A.; George, M.S.; Feng, W. Safety and tolerability of transcranial direct current stimulation to stroke patients—A phase I current escalation study. Brain Stimul. 2017, 10, 553–559. [Google Scholar] [CrossRef]
- Chhatbar, P.Y.; Kautz, S.A.; Takacs, I.; Rowland, N.C.; Revuelta, G.J.; George, M.S.; Bikson, M.; Feng, W. Evidence of transcranial direct current stimulation-generated electric fields at subthalamic level in human brain in vivo. Brain Stimul. 2018, 11, 727–733. [Google Scholar] [CrossRef]
- Trapp, N.T.; Xiong, W.; Gott, B.M.; Espejo, G.D.; Bikson, M.; Conway, C.R. Proceedings #51: 4 mA adaptive transcranial direct current stimulation for treatment-resistant depression: Early demonstration of feasibility with a 20-session course. Brain Stimul. Basic Transl. Clin. Res. Neuromodul. 2019, 12, e124–e125. [Google Scholar]
- Khadka, N.; Borges, H.; Paneri, B.; Kaufman, T.; Nassis, E.; Zannou, A.L.; Shin, Y.; Choi, H.; Kim, S.; Lee, K.; et al. Adaptive Current tDCS up to 4 mA. Brain Stimul. 2020, 13, 69–79. [Google Scholar] [CrossRef]
- Nitsche, M.A.; Cohen, L.G.; Wassermann, E.M.; Priori, A.; Lang, N.; Antal, A.; Paulus, W.; Hummel, F.; Boggio, P.S.; Fregni, F.; et al. Transcranial direct current stimulation: State of the art 2008. Brain Stimul. 2008, 1, 206–223. [Google Scholar] [CrossRef]
- Machado, S.; Jansen, P.; Almeida, V.; Veldema, J. Is tDCS an adjunct ergogenic resource for improving muscular strength and endurance performance? A systematic review. Front. Psychol. 2019, 10, 1127. [Google Scholar] [CrossRef] [PubMed]
- Cogiamanian, F.; Marceglia, S.; Ardolino, G.; Barbieri, S.; Priori, A. Improved isometric force endurance after transcranial direct current stimulation over the human motor cortical areas. Eur. J. Neurosci. 2007, 26, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Angius, L.; Mauger, A.R.; Hopker, J.; Pascual-Leone, A.; Santarnecchi, E.; Marcora, S.M. Bilateral extracephalic transcranial direct current stimulation improves endurance performance in healthy individuals. Brain Stimul. 2018, 11, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Angius, L.; Pageaux, B.; Hopker, J.; Marcora, S.M.; Mauger, A.R. Transcranial direct current stimulation improves isometric time to exhaustion of the knee extensors. Neuroscience 2016, 339, 363–375. [Google Scholar] [CrossRef] [PubMed]
- Lefaucheur, J.P.; Chalah, M.A.; Mhalla, A.; Palm, U.; Ayache, S.S.; Mylius, V. The treatment of fatigue by non-invasive brain stimulation. Neurophysiol. Clin. 2017, 47, 173–184. [Google Scholar] [CrossRef] [PubMed]
- Ayache, S.S.; Chalah, M.A. Transcranial direct current stimulation: A glimmer of hope for multiple sclerosis fatigue? J. Clin. Neurosci. 2018, 55, 10–12. [Google Scholar] [CrossRef] [PubMed]
- Chalah, M.A.; Riachi, N.; Ahdab, R.; Creange, A.; Lefaucheur, J.P.; Ayache, S.S. Fatigue in multiple sclerosis: Neural correlates and the role of non-invasive brain stimulation. Front. Cell Neurosci. 2015, 9, 460. [Google Scholar] [CrossRef]
- Proessl, F.; Poston, B.; Rudroff, T. Does a single application of anodal tDCS improve knee extensor fatigability in people with multiple sclerosis? Brain Stimul. 2018, 11, 1388–1390. [Google Scholar] [CrossRef]
- Saiote, C.; Goldschmidt, T.; Timaus, C.; Steenwijk, M.D.; Opitz, A.; Antal, A.; Paulus, W.; Nitsche, M.A. Impact of transcranial direct current stimulation on fatigue in multiple sclerosis. Restor. Neurol. Neurosci. 2014, 32, 423–436. [Google Scholar] [CrossRef]
- Chalah, M.A.; Riachi, N.; Ahdab, R.; Mhalla, A.; Abdellaoui, M.; Creange, A.; Lefaucheur, J.P.; Ayache, S.S. Effects of left DLPFC versus right PPC tDCS on multiple sclerosis fatigue. J. Neurol. Sci. 2017, 372, 131–137. [Google Scholar] [CrossRef]
- Ayache, S.S.; Palm, U.; Chalah, M.A.; Al-Ani, T.; Brignol, A.; Abdellaoui, M.; Dimitri, D.; Sorel, M.; Creange, A.; Lefaucheur, J.P. Prefrontal tDCS decreases pain in patients with multiple sclerosis. Front. Neurosci. 2016, 10, 147. [Google Scholar] [CrossRef]
- Tecchio, F.; Cancelli, A.; Cottone, C.; Zito, G.; Pasqualetti, P.; Ghazaryan, A.; Rossini, P.M.; Filippi, M.M. Multiple sclerosis fatigue relief by bilateral somatosensory cortex neuromodulation. J. Neurol. 2014, 261, 1552–1558. [Google Scholar] [CrossRef] [PubMed]
- Tecchio, F.; Cancelli, A.; Cottone, C.; Ferrucci, R.; Vergari, M.; Zito, G.; Pasqualetti, P.; Filippi, M.M.; Ghazaryan, A.; Lupoi, D.; et al. Brain plasticity effects of neuromodulation against multiple sclerosis fatigue. Front. Neurol. 2015, 6, 141. [Google Scholar] [CrossRef]
- Cancelli, A.; Cottone, C.; Giordani, A.; Migliore, S.; Lupoi, D.; Porcaro, C.; Mirabella, M.; Rossini, P.M.; Filippi, M.M.; Tecchio, F. Personalized, bilateral whole-body somatosensory cortex stimulation to relieve fatigue in multiple sclerosis. Mult. Scler. 2018, 24, 1366–1374. [Google Scholar] [CrossRef] [PubMed]
- Ferrucci, R.; Vergari, M.; Cogiamanian, F.; Bocci, T.; Ciocca, M.; Tomasini, E.; De Riz, M.; Scarpini, E.; Priori, A. Transcranial direct current stimulation (tDCS) for fatigue in multiple sclerosis. Neurorehabilitation 2014, 34, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Jang, H.; Lee, J.Y.; Lee, K.I.; Park, K.M. Are there differences in brain morphology according to handedness? Brain Behav. 2017, 7, e00730. [Google Scholar] [CrossRef] [PubMed]
- Montenegro, R.; Okano, A.; Gurgel, J.; Porto, F.; Cunha, F.; Massaferri, R.; Farinatti, P. Motor cortex tDCS does not improve strength performance in healthy subjects. Motriz Rev. Educ. Física 2015, 21, 185–193. [Google Scholar] [CrossRef]
- Saenz, A.; Avellanet, M.; Hijos, E.; Chaler, J.; Garreta, R.; Pujol, E.; Sandoval, B.; Buen, C.; Farreny, A. Knee isokinetic test-retest: A multicentre knee isokinetic test-retest study of a fatigue protocol. Eur. J. Phys. Rehabil. Med. 2010, 46, 81–88. [Google Scholar]
- Klem, G.H.; Luders, H.O.; Jasper, H.H.; Elger, C. The Ten-Twenty Electrode System of the International Federation—The International Federation of Clinical Neurophysiology. Electroencephalogr. Clin. Neurophysiol. Suppl. 1999, 52, 3–6. [Google Scholar]
- Schambra, H.M.; Abe, M.; Luckenbaugh, D.A.; Reis, J.; Krakauer, J.W.; Cohen, L.G. Probing for hemispheric specialization for motor skill learning: A transcranial direct current stimulation study. J. Neurophysiol. 2011, 106, 652–661. [Google Scholar] [CrossRef]
- Aparicio, L.V.M.; Guarienti, F.; Razza, L.B.; Carvalho, A.F.; Fregni, F.; Brunoni, A.R. A systematic review on the acceptability and tolerability of transcranial direct current stimulation treatment in neuropsychiatry trials. Brain Stimul. 2016, 9, 671–681. [Google Scholar] [CrossRef] [PubMed]
- Mackey, C.S.; Thiele, R.M.; Conchola, E.C.; DeFreitas, J.M. Comparison of fatigue responses and rapid force characteristics between explosive-and traditional-resistance-trained males. Eur. J. Appl. Physiol. 2018, 118, 1539–1546. [Google Scholar] [CrossRef] [PubMed]
- Thorstensson, A.; Karlsson, J. Fatiguability and fibre composition of human skeletal muscle. Acta Physiol. Scand. 1976, 98, 318–322. [Google Scholar] [CrossRef] [PubMed]
- Hameau, S.; Bensmail, D.; Roche, N.; Zory, R. Adaptations of fatigue and fatigability after a short intensive, combined rehabilitation program in patients with multiple sclerosis. J. Rehabil. Med. 2018, 50, 59–66. [Google Scholar] [CrossRef]
- Jamil, A.; Batsikadze, G.; Kuo, H.I.; Labruna, L.; Hasan, A.; Paulus, W.; Nitsche, M.A. Systematic evaluation of the impact of stimulation intensity on neuroplastic after-effects induced by transcranial direct current stimulation. J. Physiol. 2017, 595, 1273–1288. [Google Scholar] [CrossRef]
- Gur, H.; Akova, B.; Punduk, Z.; Kucukoglu, S. Effects of age on the reciprocal peak torque ratios during knee muscle contractions in elite soccer players. Scand. J. Med. Sci. Sports 1999, 9, 81–87. [Google Scholar] [CrossRef]
- Muff, G.; Dufour, S.; Meyer, A.; Severac, F.; Favret, F.; Geny, B.; Lecocq, J.; Isner-Horobeti, M.-E. Comparative assessment of knee extensor and flexor muscle strength measured using a hand-held vs. isokinetic dynamometer. J. Phys. Ther. Sci. 2016, 28, 2445–2451. [Google Scholar] [CrossRef]
- Krishnan, C.; Ranganathan, R.; Kantak, S.S.; Dhaher, Y.Y.; Rymer, W.Z. Anodal transcranial direct current stimulation alters elbow flexor muscle recruitment strategies. Brain Stimul. 2014, 7, 443–450. [Google Scholar] [CrossRef]
- Fregni, F.; Boggio, P.S.; Lima, M.C.; Ferreira, M.J.; Wagner, T.; Rigonatti, S.P.; Castro, A.W.; Souza, D.R.; Riberto, M.; Freedman, S.D.; et al. A sham-controlled, phase ii trial of transcranial direct current stimulation for the treatment of central pain in traumatic spinal cord injury. Pain 2006, 122, 197–209. [Google Scholar] [CrossRef]
- Sales, M.M.; De Sousa, C.V.; Browne, R.A.V.; Fontes, E.B.; Olher, R.d.R.V.; Ernesto, C.; Simões, H.G. Transcranial direct current stimulation improves muscle isokinetic performance of young trained individuals. Med. Sport 2016, 69, 163–172. [Google Scholar]
- Mondini, V.; Mangia, A.L.; Cappello, A. Single-Session tDCS over the dominant hemisphere affects contralateral spectral eeg power, but does not enhance neurofeedback-guided event-related desynchronization of the non-dominant hemisphere’s sensorimotor rhythm. PLoS ONE 2018, 13, e0193004. [Google Scholar] [CrossRef] [PubMed]
- Park, C.-H.; Chang, W.H.; Park, J.-Y.; Shin, Y.-I.; Kim, S.T.; Kim, Y.-H. Transcranial direct current stimulation increases resting state interhemispheric connectivity. Neurosci. Lett. 2013, 539, 7–10. [Google Scholar] [CrossRef] [PubMed]
- Waters, S.; Wiestler, T.; Diedrichsen, J. Cooperation not competition: Bihemispheric tDCS and fmri show role for ipsilateral hemisphere in motor learning. J. Neurosci. 2017, 37, 7500–7512. [Google Scholar] [CrossRef] [PubMed]
- Naros, G.; Geyer, M.; Koch, S.; Mayr, L.; Ellinger, T.; Grimm, F.; Gharabaghi, A. Enhanced motor learning with bilateral transcranial direct current stimulation: Impact of polarity or current flow direction? Clin. Neurophysiol. 2016, 127, 2119–2126. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Lee, A.; Kim, H.; Shin, M.; Yun, S.M.; Jung, Y.; Chang, W.H.; Kim, Y.H. Different brain connectivity between responders and nonresponders to dual-mode noninvasive brain stimulation over bilateral primary motor cortices in stroke patients. Neural. Plast. 2019, 2019, 3826495. [Google Scholar] [CrossRef]
- Nejadgholi, I.; Davidson, T.; Blais, C.; Tremblay, F.; Bolic, M. Classification of Responders Versus Non-Responders to Tdcs by Analyzing Voltage between Anode and Cathode During Treatment Session. In Proceedings of the World Congress on Medical Physics and Biomedical Engineering, Toronto, ON, Canada, 7–12 June 2015; pp. 990–993. [Google Scholar]
- Tremblay, S.; Larochelle-Brunet, F.; Lafleur, L.P.; El Mouderrib, S.; Lepage, J.F.; Theoret, H. Systematic assessment of duration and intensity of anodal transcranial direct current stimulation on primary motor cortex excitability. Eur. J. Neurosci. 2016, 44, 2184–2190. [Google Scholar] [CrossRef]
- Rawji, V.; Ciocca, M.; Zacharia, A.; Soares, D.; Truong, D.; Bikson, M.; Rothwell, J.; Bestmann, S. tDCS changes in motor excitability are specific to orientation of current flow. Brain Stimul. 2018, 11, 289–298. [Google Scholar] [CrossRef]
- Varoli, E.; Pisoni, A.; Mattavelli, G.C.; Vergallito, A.; Gallucci, A.; Mauro, L.D.; Rosanova, M.; Bolognini, N.; Vallar, G.; Romero Lauro, L.J. Tracking the effect of cathodal transcranial direct current stimulation on cortical excitability and connectivity by means of TMS-EEG. Front. Neurosci. 2018, 12, 319. [Google Scholar] [CrossRef]
- Liu, A.; Vöröslakos, M.; Kronberg, G.; Henin, S.; Krause, M.R.; Huang, Y.; Opitz, A.; Mehta, A.; Pack, C.C.; Krekelberg, B.; et al. Immediate neurophysiological effects of transcranial electrical stimulation. Nat. Commun. 2018, 9, 5092. [Google Scholar] [CrossRef]
- Thoumie, P.; Lamotte, D.; Cantalloube, S.; Faucher, M.; Amarenco, G. Motor determinants of gait in 100 ambulatory patients with multiple sclerosis. Mult. Scler. 2005, 11, 485–491. [Google Scholar] [CrossRef]
- Broekmans, T.; Gijbels, D.; Eijnde, B.O.; Alders, G.; Lamers, I.; Roelants, M.; Feys, P. The relationship between upper leg muscle strength and walking capacity in persons with multiple sclerosis. Mult. Scler. 2013, 19, 112–119. [Google Scholar] [CrossRef]
- Larson, R.D.; McCully, K.K.; Larson, D.J.; Pryor, W.M.; White, L.J. Bilateral differences in lower-limb performance in individuals with multiple sclerosis. J. Rehabil. Res. Dev. 2013, 50, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Kollock, R.; Van Lunen, B.L.; Ringleb, S.I.; Onate, J.A. Measures of functional performance and their association with hip and thigh strength. J. Athl. Train. 2015, 50, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Batsikadze, G.; Moliadze, V.; Paulus, W.; Kuo, M.F.; Nitsche, M.A. Partially non-linear stimulation intensity-dependent effects of direct current stimulation on motor cortex excitability in humans. J. Physiol. 2013, 591, 1987–2000. [Google Scholar] [CrossRef] [PubMed]
- Esmaeilpour, Z.; Marangolo, P.; Hampstead, B.M.; Bestmann, S.; Galletta, E.; Knotkova, H.; Bikson, M. Incomplete evidence that increasing current intensity of tDCS boosts outcomes. Brain Stimul. 2018, 11, 310–321. [Google Scholar] [CrossRef] [PubMed]
- Monte-Silva, K.; Kuo, M.F.; Hessenthaler, S.; Fresnoza, S.; Liebetanz, D.; Paulus, W.; Nitsche, M.A. Induction of late LTP-like plasticity in the human motor cortex by repeated non-invasive brain stimulation. Brain Stimul. 2013, 6, 424–432. [Google Scholar] [CrossRef] [PubMed]
- Brunoni, A.R.; Amadera, J.; Berbel, B.; Volz, M.S.; Rizzerio, B.G.; Fregni, F. A systematic review on reporting and assessment of adverse effects associated with transcranial direct current stimulation. Int. J. Neuropsychopharmacol. 2011, 14, 1133–1145. [Google Scholar] [CrossRef]
| Sensation 1 | Sham | 4 mA |
|---|---|---|
| Blinding 2 Accuracy | 66.7% | 45.2% |
| Tingling | 1.5 ± 1.0 (n = 13) | 2.8 ± 1.6 (n = 11) |
| Burning | 3.1 ± 1.5 (n = 10) | 4.6 ± 1.7 (n = 18) |
| Itching | 2.3 ± 1.3 (n = 8) | 3.5 ± 2.0 (n = 12) |
| Prickling | 2.8 ± 1.1 (n = 5) | 3.0 ± 1.4 (n = 2) |
| Poking | 3.5 ± 3.5 (n = 2) | 3.0 ± 1.4 (n = 2) |
| Stinging | 2.3 ± 1.5 (n = 3) | |
| Watery Eyes | 1.0 ± 0.0 (n = 1) | 2.0 ± 0.0 (n = 1) |
| Needle | 3.5 ± 2.1 (n = 4) | |
| Headache | 7.0 ± 1.4 (n = 2) | |
| Pinching | 2.0 ± 0.0 (n = 1) | |
| Tickling | 2.0 ± 0.0 (n = 1) | |
| Pressure | 7.0 ± 0.0 (n = 1) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Workman, C.D.; Kamholz, J.; Rudroff, T. The Tolerability and Efficacy of 4 mA Transcranial Direct Current Stimulation on Leg Muscle Fatigability. Brain Sci. 2020, 10, 12. https://doi.org/10.3390/brainsci10010012
Workman CD, Kamholz J, Rudroff T. The Tolerability and Efficacy of 4 mA Transcranial Direct Current Stimulation on Leg Muscle Fatigability. Brain Sciences. 2020; 10(1):12. https://doi.org/10.3390/brainsci10010012
Chicago/Turabian StyleWorkman, Craig D., John Kamholz, and Thorsten Rudroff. 2020. "The Tolerability and Efficacy of 4 mA Transcranial Direct Current Stimulation on Leg Muscle Fatigability" Brain Sciences 10, no. 1: 12. https://doi.org/10.3390/brainsci10010012
APA StyleWorkman, C. D., Kamholz, J., & Rudroff, T. (2020). The Tolerability and Efficacy of 4 mA Transcranial Direct Current Stimulation on Leg Muscle Fatigability. Brain Sciences, 10(1), 12. https://doi.org/10.3390/brainsci10010012






