Recent Advancement in the Surface-Enhanced Raman Spectroscopy-Based Biosensors for Infectious Disease Diagnosis
Abstract
1. Introduction
2. Bacterial Infections
2.1. Nucleic Acid Biomarkers
2.2. Cell Membrane Biomarkers
2.3. Metabolites as Biomarkers
3. Mosquito-Borne Infections
3.1. Nucleic Acid Biomarker Detection
3.2. Proteins
3.3. Metabolites as Biomarkers
4. SERS-Based Diagnostic Devices and Assay Platforms
5. Conclusions and Outlook
Author Contributions
Funding
Conflicts of Interest
References
- Qasim, M.; Lim, D.-J.; Park, H.; Na, D. Nanotechnology for Diagnosis and Treatment of Infectious Diseases. J. Nanosci. Nanotechnol. 2014, 14, 7374–7387. [Google Scholar] [CrossRef]
- Peeling, R.W.; Holmes, K.K.; Mabey, D.; Ronald, A. Rapid tests for sexually transmitted infections (STIs): the way forward. Sex. Transm. Infect. 2006, 82, v1–v6. [Google Scholar] [CrossRef] [PubMed]
- Mabey, D.; Peeling, R.W.; Ustianowski, A.; Perkins, M.D. Diagnostics for the developing world. Nat. Rev. Microbiol. 2004, 2, 231–240. [Google Scholar] [CrossRef]
- Manohar, C.; Andrea, T.; Anisha, G.; Gobind, D.; Zaccaria, R.P.; Roman, K.; Eliana, R.; Marco, L.; Carlo, L.; De, A.F.; et al. 3D Nanostar Dimers with a Sub-10-nm Gap for Single-/Few-Molecule Surface-Enhanced Raman Scattering. Adv. Mater. 2014, 26, 2353–2358. [Google Scholar]
- Gracie, K.; Correa, E.; Mabbott, S.; Dougan, J.A.; Graham, D.; Goodacre, R.; Faulds, K. Simultaneous detection and quantification of three bacterial meningitis pathogens by SERS. Chem. Sci. 2014, 5, 1030–1040. [Google Scholar] [CrossRef]
- Domke, K.F. Surface Enhanced Raman Spectroscopy. Analytical, Biophysical and Life Science Applications. Edited by Sebastian Schlücker. Angew. Chem. Int. Ed. 2011, 50, 8226. [Google Scholar] [CrossRef]
- Wang, Y.; Rauf, S.; Grewal, Y.S.; Spadafora, L.J.; Shiddiky, M.J.; Cangelosi, G.A.; Schlucker, S.; Trau, M. Duplex microfluidic SERS detection of pathogen antigens with nanoyeast single-chain variable fragments. Anal. Chem. 2014, 86, 9930–9938. [Google Scholar] [CrossRef] [PubMed]
- Tran, V.; Walkenfort, B.; Konig, M.; Salehi, M.; Schlucker, S. Rapid, Quantitative, and Ultrasensitive Point-of-Care Testing: A Portable SERS Reader for Lateral Flow Assays in Clinical Chemistry. Angew. Chem. Int. Ed. Engl. 2019, 58, 442–446. [Google Scholar] [CrossRef]
- Graham, D.; Mallinder, B.J.; Smith, W.E. Detection and identification of labeled DNA by surface enhanced resonance Raman scattering. Biopolymers 2000, 57, 85–91. [Google Scholar] [CrossRef]
- Harper, M.M.; Dougan, J.A.; Shand, N.C.; Graham, D.; Faulds, K. Detection of SERS active labelled DNA based on surface affinity to silver nanoparticles. Analyst 2012, 137, 2063–2068. [Google Scholar] [CrossRef]
- McQueenie, R.; Stevenson, R.; Benson, R.; MacRitchie, N.; McInnes, I.; Maffia, P.; Faulds, K.; Graham, D.; Brewer, J.; Garside, P. Detection of inflammation in vivo by surface-enhanced Raman scattering provides higher sensitivity than conventional fluorescence imaging. Anal. Chem. 2012, 84, 5968–5975. [Google Scholar] [CrossRef]
- Alvarez-Puebla, R.A.; Liz-Marzan, L.M. SERS-Based Diagnosis and Biodetection. Small 2010, 6, 604–610. [Google Scholar] [CrossRef]
- Bodelon, G.; Montes-Garcia, V.; Lopez-Puente, V.; Hill, E.H.; Hamon, C.; Sanz-Ortiz, M.; Rodal-Cedeira, S.; Costas, C.; Celiksoy, S.; Perez-Juste, I.; et al. Detection and imaging of quorum sensing in Pseudomonas aeruginosa biofilm communities by surface-enhanced resonance Raman scattering. Nat. Mater. 2016, 15, 1203–1211. [Google Scholar] [CrossRef]
- Moskovits, M. Surface-enhanced Raman spectroscopy: A brief retrospective. J. Raman Spectrosc. 2005, 36, 485–496. [Google Scholar] [CrossRef]
- Hao, E.; Schatz, G.; Hupp, J. Synthesis and Optical Properties of Anisotropic Metal Nanoparticles. J. Fluoresc. 2004, 14, 331–341. [Google Scholar] [CrossRef]
- Kelly, K.L.; Coronado, E.; Zhao, L.L.; Schatz, G.C. The optical properties of metal nanoparticles: The influence of size, shape, and dielectric environment. J. Phys. Chem. B 2003, 107, 668. [Google Scholar] [CrossRef]
- De Silva Indrasekara, A.S.; Johnson, S.F.; Odion, R.A.; Vo-Dinh, T. Manipulation of the Geometry and Modulation of the Optical Response of Surfactant-Free Gold Nanostars: A Systematic Bottom-Up Synthesis. ACS Omega 2018, 3, 2202–2210. [Google Scholar] [CrossRef]
- Indrasekara, A.S.D.; Thomas, R.; Fabris, L. Plasmonic properties of regiospecific core–satellite assemblies of gold nanostars and nanospheres. Phys. Chem. Chem. Phys. 2015, 17, 21133–21142. [Google Scholar] [CrossRef]
- Fabris, L.; Indrasekara, A.S. Gold Nanostar Substrates for SERS Sensing in the Femtomolar Regime. US Patent No US10024800B2, 2015. [Google Scholar]
- Perets, E.A.; Indrasekara, A.S.D.S.; Kurmis, A.; Atlasevich, N.; Fabris, L.; Arslanoglu, J. Carboxy-terminated immuno-SERS tags overcome non-specific aggregation for the robust detection and localization of organic media in artworks. Analyst 2015, 140, 5971–5980. [Google Scholar] [CrossRef]
- Indrasekara, A.S.D.S.; Paladini, B.J.; Naczynski, D.J.; Starovoytov, V.; Moghe, P.V.; Fabris, L. Dimeric Gold Nanoparticle Assemblies as Tags for SERS-Based Cancer Detection. Adv. Healthc. Mater. 2013, 2, 1370–1376. [Google Scholar] [CrossRef]
- Personick, M.L.; Langille, M.R.; Zhang, J.; Mirkin, C.A. Shape Control of Gold Nanoparticles by Silver Underpotential Deposition. Nano Lett. 2011, 11, 3394–3398. [Google Scholar] [CrossRef]
- Xue, C.; Millstone, J.E.; Li, S.; Mirkin, C.A. Plasmon-Driven Synthesis of Triangular Core?Shell Nanoprisms from Gold Seeds. Angew. Chem. Int. Ed. 2007, 46, 8436–8439. [Google Scholar] [CrossRef]
- Rosi, N.L.; Mirkin, C.A. Nanostructures in biodiagnostics. Chem. Rev. 2005, 105, 1547–1562. [Google Scholar] [CrossRef]
- Murphy, C.J.; Sau, T.K.; Gole, A.M.; Orendorff, C.J.; Gao, J.; Gou, L.; Hunyadi, S.E.; Li, T. Anisotropic Metal Nanoparticles: Synthesis, Assembly, and Optical Applications. J. Phys. Chem. B 2005, 109, 13857–13870. [Google Scholar] [CrossRef]
- Kleinman, S.L.; Sharma, B.; Blaber, M.G.; Henry, A.-I.; Valley, N.; Freeman, R.G.; Natan, M.J.; Schatz, G.C.; Van Duyne, R.P. Structure Enhancement Factor Relationships in Single Gold Nanoantennas by Surface-Enhanced Raman Excitation Spectroscopy. J. Am. Chem. Soc. 2013, 135, 301–308. [Google Scholar] [CrossRef]
- Sharma, B.; Fernanda Cardinal, M.; Kleinman, S.L.; Greeneltch, N.G.; Frontiera, R.R.; Blaber, M.G.; Schatz, G.C.; Van Duyne, R.P. High-performance SERS substrates: Advances and challenges. MRS Bull. 2013, 38, 615–624. [Google Scholar] [CrossRef]
- Greeneltch, N.G.; Blaber, M.G.; Henry, A.-I.; Schatz, G.C.; Van Duyne, R.P. Immobilized Nanorod Assemblies: Fabrication and Understanding of Large Area Surface-Enhanced Raman Spectroscopy Substrates. Anal. Chem. 2013, 85, 2297–2303. [Google Scholar] [CrossRef]
- Stiles, P.L.; Dieringer, J.A.; Shah, N.C.; Van Duyne, R.P. Surface-Enhanced Raman Spectroscopy. Annu. Rev. Anal. Chem. 2008, 1, 601–626. [Google Scholar] [CrossRef]
- Kneipp, K.; Dasari, R.R.; Wang, Y. Near-Infrared Surface-Enhanced Raman-Scattering (Nir Sers) on Colloidal Silver and Gold. Appl. Spectrosc. 1994, 48, 951–955. [Google Scholar] [CrossRef]
- Michota, A.; Bukowska, J. Surface-enhanced Raman scattering (SERS) of 4-mercaptobenzoic acid on silver and gold substrates. J. Raman Spectrosc. 2003, 34, 21–25. [Google Scholar] [CrossRef]
- Alvarez-Puebla, R.; Dos, S., Jr.; Aroca, R.F. Surface-enhanced Raman scattering for ultrasensitive chemical analysis of 1 and 2-naphthalenethiols. Analyst 2004, 129, 1251–1256. [Google Scholar] [CrossRef]
- Fabris, L.; Dante, M.; Nguyen, T.-Q.; Tok, J.B.H.; Bazan, G.C. SERS Aptatags: New Responsive Metallic Nanostructures for Heterogeneous Protein Detection by Surface Enhanced Raman Spectroscopy. Adv. Funct. Mater. 2008, 18, 2518–2525. [Google Scholar] [CrossRef]
- Indrasekara, A.S.D.S.; Meyers, S.; Shubeita, S.; Feldman, L.C.; Gustafsson, T.; Fabris, L. Gold nanostar substrates for SERS-based chemical sensing in the femtomolar regime. Nanoscale 2014, 6, 8891–8899. [Google Scholar] [CrossRef]
- Millstone, J.E.; Park, S.; Shuford, K.L.; Qin, L.; Schatz, G.C.; Mirkin, C.A. Observation of a Quadrupole Plasmon Mode for a Colloidal Solution of Gold Nanoprisms. J. Am. Chem. Soc. 2005, 127, 5312–5313. [Google Scholar] [CrossRef]
- Fales, A.M.; Yuan, H.; Vo-Dinh, T. Silica-Coated Gold Nanostars for Combined Surface-Enhanced Raman Scattering (SERS) Detection and Singlet-Oxygen Generation: A Potential Nanoplatform for Theranostics. Langmuir 2011, 27, 12186–12190. [Google Scholar] [CrossRef]
- Hsiangkuo, Y.; Christopher, G.K.; Hanjun, H.; Christy, M.W.; Gerald A Grant and, T.V. Gold nanostars: surfactant-free synthesis, 3D modelling, and two-photon photoluminescence imaging. Nanotechnology 2012, 23, 075102. [Google Scholar]
- Hao, F.; Nehl, C.L.; Hafner, J.H.; Nordlander, P. Plasmon Resonances of a Gold Nanostar. Nano Lett. 2007, 7, 729–732. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Puebla, R.; Liz-MarzaÌn, L.M.; de Abajo, F.J.G. Light Concentration at the Nanometer Scale. J. Phys. Chem. Lett. 2010, 1, 2428–2434. [Google Scholar] [CrossRef]
- Braun, G.B.; Lee, S.J.; Laurence, T.; Fera, N.; Fabris, L.; Bazan, G.C.; Moskovits, M.; Reich, N.O. Generalized Approach to SERS-Active Nanomaterials via Controlled Nanoparticle Linking, Polymer Encapsulation, and Small-Molecule Infusion. J. Phys. Chem. C 2009, 113, 13622–13629. [Google Scholar] [CrossRef]
- Talley, C.E.; Jackson, J.B.; Oubre, C.; Grady, N.K.; Hollars, C.W.; Lane, S.M.; Huser, T.R.; Nordlander, P.; Halas, N.J. Surface-Enhanced Raman Scattering from Individual Au Nanoparticles and Nanoparticle Dimer Substrates. Nano Lett. 2005, 5, 1569–1574. [Google Scholar] [CrossRef]
- Schutz, M.; Schlucker, S. Molecularly linked 3D plasmonic nanoparticle core/satellite assemblies: SERS nanotags with single-particle Raman sensitivity. Phys. Chem. Chem. Phys. 2015, 17, 24356. [Google Scholar] [CrossRef]
- Romo-Herrera, J.; Alvarez-Puebla, R.; Liz-Marzan, L. Controlled assembly of plasmonic colloidal nanoparticle clusters. Nanoscale 2011, 3, 1304–1315. [Google Scholar] [CrossRef]
- Chen, G.; Wang, Y.; Yang, M.; Xu, J.; Goh, S.J.; Pan, M.; Chen, H. Measuring Ensemble-Averaged Surface-Enhanced Raman Scattering in the Hotspots of Colloidal Nanoparticle Dimers and Trimers. J. Am. Chem. Soc. 2010, 132, 3644–3645. [Google Scholar] [CrossRef]
- Malinsky, M.D.; Kelly, K.L.; Schatz, G.C.; Van Duyne, R.P. Chain Length Dependence and Sensing Capabilities of the Localized Surface Plasmon Resonance of Silver Nanoparticles Chemically Modified with Alkanethiol Self-Assembled Monolayers. J. Am. Chem. Soc. 2001, 123, 1471–1482. [Google Scholar] [CrossRef]
- Wustholz, K.L.; Henry, A.-I.; McMahon, J.M.; Freeman, R.G.; Valley, N.; Piotti, M.E.; Natan, M.J.; Schatz, G.C.; Duyne, R.P.V. Structure-Activity Relationships in Gold Nanoparticle Dimers and Trimers for Surface-Enhanced Raman Spectroscopy. J. Am. Chem. Soc. 2010, 132, 10903–10910. [Google Scholar] [CrossRef]
- Rahme, K.; Chen, L.; Hobbs, R.G.; Morris, M.A.; O'Driscoll, C.; Holmes, J.D. PEGylated gold nanoparticles: Polymer quantification as a function of PEG lengths and nanoparticle dimensions. RSC Adv. 2013, 3, 6085–6094. [Google Scholar] [CrossRef]
- Chen, S.; Yang, M.; Hong, S.; Lu, C. Nonionic fluorosurfactant as an ideal candidate for one-step modification of gold nanorods. Nanoscale 2014. [Google Scholar] [CrossRef]
- Austin, L.A.; Mackey, M.A.; Dreaden, E.C.; El-Sayed, M.A. The optical, photothermal, and facile surface chemical properties of gold and silver nanoparticles in biodiagnostics, therapy, and drug delivery. Arch. Toxicol. 2014, 88, 1391–1417. [Google Scholar] [CrossRef]
- Park, S.Y.; Lee, J.-S.; Georganopoulou, D.; Mirkin, C.A.; Schatz, G.C. Structures of DNA-Linked Nanoparticle Aggregates. J. Phys. Chem. B 2006, 110, 12673–12681. [Google Scholar] [CrossRef]
- Brennan, J.L.; Hatzakis, N.S.; Tshikhudo, T.R.; Razumas, V.; Patkar, S.; Vind, J.; Svendsen, A.; Nolte, R.J.M.; Rowan, A.E.; Brust, M. Bionanoconjugation via Click Chemistry: The Creation of Functional Hybrids of Lipases and Gold Nanoparticles. Bioconj. Chem. 2006, 17, 1373–1375. [Google Scholar] [CrossRef]
- Krpetić, Ž.; Nativo, P.; Porta, F.; Brust, M. A Multidentate Peptide for Stabilization and Facile Bioconjugation of Gold Nanoparticles. Bioconj. Chem. 2009, 20, 619–624. [Google Scholar] [CrossRef]
- Kanaras, A.G.; Kamounah, F.S.; Schaumburg, K.; Kiely, C.J.; Brust, M. Thioalkylated tetraethylene glycol: A new ligand for water soluble monolayer protected gold clusters. Chem. Commun. 2002, 2294–2295. [Google Scholar] [CrossRef]
- Barhoumi, A.; Halas, N.J. Label-Free Detection of DNA Hybridization Using Surface Enhanced Raman Spectroscopy. J. Am. Chem. Soc. 2010, 132, 12792–12793. [Google Scholar] [CrossRef]
- Dondapati, S.K.; Sau, T.K.; Hrelescu, C.; Klar, T.A.; Stefani, F.D.; Feldmann, J. Label-free Biosensing Based on Single Gold Nanostars as Plasmonic Transducers. ACS Nano 2010, 4, 6318–6322. [Google Scholar] [CrossRef]
- Papadopoulou, E.; Bell, S.E.J. Label-Free Detection of Single-Base Mismatches in DNA by Surface-Enhanced Raman Spectroscopy. Angew. Chem. Int. Ed. 2011, 50, 9058–9061. [Google Scholar] [CrossRef]
- Ngo, H.T.; Wang, H.-N.; Fales, A.M.; Vo-Dinh, T. Label-Free DNA Biosensor Based on SERS Molecular Sentinel on Nanowave Chip. Anal. Chem. 2013, 85, 6378–6383. [Google Scholar] [CrossRef]
- Xu, L.-J.; Lei, Z.-C.; Li, J.; Zong, C.; Yang, C.J.; Ren, B. Label-Free Surface-Enhanced Raman Spectroscopy Detection of DNA with Single-Base Sensitivity. J. Am. Chem. Soc. 2015, 137, 5149–5154. [Google Scholar] [CrossRef]
- Dina, N.E.; Zhou, H.; Colnita, A.; Leopold, N.; Szoke-Nagy, T.; Coman, C.; Haisch, C. Rapid single-cell detection and identification of pathogens by using surface-enhanced Raman spectroscopy. Analyst 2017, 142, 1782–1789. [Google Scholar] [CrossRef]
- Graham, D.; Mallinder, B.J.; Whitcombe, D.; Watson, N.D.; Smith, W.E. Simple Multiplex Genotyping by Surface-Enhanced Resonance Raman Scattering. Anal. Chem. 2002, 74, 1069–1074. [Google Scholar] [CrossRef]
- Yu, C.; Irudayaraj, J. Quantitative Evaluation of Sensitivity and Selectivity of Multiplex NanoSPR Biosensor Assays. Biophys. J. 2007, 93, 3684–3692. [Google Scholar] [CrossRef]
- Sun, L.; Yu, C.; Irudayaraj, J. Raman multiplexers for alternative gene splicing. Anal. Chem. 2008, 80, 3342–3349. [Google Scholar] [CrossRef]
- von Maltzahn, G.; Centrone, A.; Park, J.-H.; Ramanathan, R.; Sailor, M.J.; Hatton, T.A.; Bhatia, S.N. SERS-Coded Gold Nanorods as a Multifunctional Platform for Densely Multiplexed Near-Infrared Imaging and Photothermal Heating. Adv. Mater. 2009, 21, 3175–3180. [Google Scholar] [CrossRef]
- Zavaleta, C.L.; Smith, B.R.; Walton, I.; Doering, W.; Davis, G.; Shojaei, B.; Natan, M.J.; Gambhir, S.S. Multiplexed imaging of surface enhanced Raman scattering nanotags in living mice using noninvasive Raman spectroscopy. Proc. Natl. Acad. Sci. USA 2009, 106, 13511–13516. [Google Scholar] [CrossRef]
- Fabris, L.; Schierhorn, M.; Moskovits, M.; Bazan, G.C. Aptatag-Based Multiplexed Assay for Protein Detection by Surface-Enhanced Raman Spectroscopy. Small 2010, 6, 1550–1557. [Google Scholar] [CrossRef]
- Kang, T.; Yoo, S.M.; Yoon, I.; Lee, S.Y.; Kim, B. Patterned Multiplex Pathogen DNA Detection by Au Particle-on-Wire SERS Sensor. Nano Lett. 2010, 10, 1189–1193. [Google Scholar] [CrossRef]
- Pallaoro, A.; Braun, G.B.; Moskovits, M. Quantitative ratiometric discrimination between noncancerous and cancerous prostate cells based on neuropilin-1 overexpression. Proc. Natl. Acad. Sci. USA 2011. [Google Scholar] [CrossRef]
- Kim, J.-Y.; Lee, J.-S. Multiplexed DNA Detection with DNA-Functionalized Silver and Silver/Gold Nanoparticle Superstructure Probes. Bull. Korean Chem. Soc. 2012, 33, 221–226. [Google Scholar] [CrossRef]
- Wang, H.-N.; Crawford, B.M.; Fales, A.M.; Bowie, M.L.; Seewaldt, V.L.; Vo-Dinh, T. Multiplexed Detection of MicroRNA Biomarkers Using SERS-Based Inverse Molecular Sentinel (iMS) Nanoprobes. J. Phys. Chem. C 2016, 120, 21047–21055. [Google Scholar] [CrossRef]
- Sánchez-Purrà, M.; Roig-Solvas, B.; Versiani, A.; Rodriguez-Quijada, C.; de Puig, H.; Bosch, I.; Gehrke, L.; Hamad-Schifferli, K. Design of SERS nanotags for multiplexed lateral flow immunoassays. Mol. Syst. Des. Eng. 2017, 2, 401–409. [Google Scholar] [CrossRef]
- Wang, H.-N.; Fales, A.M.; Zaas, A.K.; Woods, C.W.; Burke, T.; Ginsburg, G.S.; Vo-Dinh, T. Surface-enhanced Raman scattering molecular sentinel nanoprobes for viral infection diagnostics. Anal. Chim. Acta 2013, 786, 153–158. [Google Scholar] [CrossRef]
- Strelau, K.K.; Brinker, A.; Schnee, C.; Weber, K.; Moeller, R.; Popp, J. Detection of PCR products amplified from DNA of epizootic pathogens using magnetic nanoparticles and SERS. J. Raman Spectrosc. 2011, 42, 243–250. [Google Scholar] [CrossRef]
- Lu, X.; Samuelson, D.R.; Xu, Y.; Zhang, H.; Wang, S.; Rasco, B.A.; Xu, J.; Konkel, M.E. Detecting and tracking nosocomial methicillin-resistant Staphylococcus aureus using a microfluidic SERS biosensor. Anal. Chem. 2013, 85, 2320–2327. [Google Scholar] [CrossRef]
- Kamińska, A.; Witkowska, E.; Kowalska, A.; Skoczyńska, A.; Ronkiewicz, P.; Szymborski, T.; Waluk, J. Rapid detection and identification of bacterial meningitis pathogens in ex vivo clinical samples by SERS method and principal component analysis. Anal. Methods 2016, 8, 4521–4529. [Google Scholar] [CrossRef]
- De, S.I.; Fabris, L. SERS-based approaches toward genetic profiling. Bioanalysis 2015, 7, 263–278. [Google Scholar]
- Negri, P.; Dluhy, R.A. Detection of genetic markers related to high pathogenicity in influenza by SERS. Analyst 2013, 138, 4877–4884. [Google Scholar] [CrossRef][Green Version]
- Lee, C.; Carney, R.P.; Hazari, S.; Smith, Z.J.; Knudson, A.; Robertson, C.S.; Lam, K.S.; Wachsmann-Hogiu, S. 3D plasmonic nanobowl platform for the study of exosomes in solution. Nanoscale 2015, 7, 9290–9297. [Google Scholar] [CrossRef]
- Wu, X.; Chen, J.; Li, X.; Zhao, Y.; Zughaier, S.M. Culture-free diagnostics of Pseudomonas aeruginosa infection by silver nanorod array based SERS from clinical sputum samples. Nanomedicine 2014, 10, 1863–1870. [Google Scholar] [CrossRef]
- Hennigan, S.L.; Driskell, J.D.; Ferguson-Noel, N.; Dluhy, R.A.; Zhao, Y.; Tripp, R.A.; Krause, D.C. Detection and differentiation of avian mycoplasmas by surface-enhanced Raman spectroscopy based on a silver nanorod array. Appl. Environ. Microbiol. 2012, 78, 1930–1935. [Google Scholar] [CrossRef]
- Hennigan, S.L.; Driskell, J.D.; Dluhy, R.A.; Zhao, Y.; Tripp, R.A.; Waites, K.B.; Krause, D.C. Detection of Mycoplasma pneumoniae in simulated and true clinical throat swab specimens by nanorod array-surface-enhanced Raman spectroscopy. PLoS ONE 2010, 5, e13633. [Google Scholar] [CrossRef]
- Fan, Z.; Senapati, D.; Khan, S.A.; Singh, A.K.; Hamme, A.; Yust, B.; Sardar, D.; Ray, P.C. Popcorn-shaped magnetic core-plasmonic shell multifunctional nanoparticles for the targeted magnetic separation and enrichment, label-free SERS imaging, and photothermal destruction of multidrug-resistant bacteria. Chemistry 2013, 19, 2839–2847. [Google Scholar] [CrossRef]
- Muhamadali, H.; Subaihi, A.; Mohammadtaheri, M.; Xu, Y.; Ellis, D.I.; Ramanathan, R.; Bansal, V.; Goodacre, R. Rapid, accurate, and comparative differentiation of clinically and industrially relevant microorganisms via multiple vibrational spectroscopic fingerprinting. Analyst 2016, 141, 5127–5136. [Google Scholar] [CrossRef]
- Prucek, R.; Ranc, V.; Kvitek, L.; Panacek, A.; Zboril, R.; Kolar, M. Reproducible discrimination between gram-positive and gram-negative bacteria using surface enhanced Raman spectroscopy with infrared excitation. Analyst 2012, 137, 2866–2870. [Google Scholar] [CrossRef]
- Guven, B.; Basaran-Akgul, N.; Temur, E.; Tamer, U.; Boyaci, I.H. SERS-based sandwich immunoassay using antibody coated magnetic nanoparticles for Escherichia coli enumeration. Analyst 2011, 136, 740–748. [Google Scholar] [CrossRef]
- Mircescu, N.E.; Zhou, H.; Leopold, N.; Chis, V.; Ivleva, N.P.; Niessner, R.; Wieser, A.; Haisch, C. Towards a receptor-free immobilization and SERS detection of urinary tract infections causative pathogens. Anal. Bioanal. Chem. 2014, 406, 3051–3058. [Google Scholar] [CrossRef]
- Fan, Z.; Kanchanapally, R.; Ray, P.C. Hybrid Graphene Oxide Based Ultrasensitive SERS Probe for Label-Free Biosensing. J. Phys. Chem. Lett. 2013, 4, 3813–3818. [Google Scholar] [CrossRef]
- Premasiri, W.R.; Chen, Y.; Williamson, P.M.; Bandarage, D.C.; Pyles, C.; Ziegler, L.D. Rapid urinary tract infection diagnostics by surface-enhanced Raman spectroscopy (SERS): identification and antibiotic susceptibilities. Anal. Bioanal. Chem. 2017, 409, 3043–3054. [Google Scholar] [CrossRef]
- Chen, K.; Han, H.; Luo, Z. Streptococcus suis II immunoassay based on thorny gold nanoparticles and surface enhanced Raman scattering. Analyst 2012, 137, 1259–1264. [Google Scholar] [CrossRef]
- MacLaughlin, C.M.; Mullaithilaga, N.; Yang, G.; Ip, S.Y.; Wang, C.; Walker, G.C. Surface-enhanced Raman scattering dye-labeled Au nanoparticles for triplexed detection of leukemia and lymphoma cells and SERS flow cytometry. Langmuir 2013, 29, 1908–1919. [Google Scholar] [CrossRef]
- Owens, N.A.; Laurentius, L.B.; Porter, M.D.; Li, Q.; Wang, S.; Chatterjee, D. Handheld Raman Spectrometer Instrumentation for Quantitative Tuberculosis Biomarker Detection: A Performance Assessment for Point-of-Need Infectious Disease Diagnostics. Appl. Spectrosc. 2018, 72, 1104–1115. [Google Scholar] [CrossRef]
- Kim, W.; Lee, J.C.; Shin, J.H.; Jin, K.H.; Park, H.K.; Choi, S. Instrument-Free Synthesizable Fabrication of Label-Free Optical Biosensing Paper Strips for the Early Detection of Infectious Keratoconjunctivitides. Anal. Chem. 2016, 88, 5531–5537. [Google Scholar] [CrossRef]
- Zhang, H.; Harpster, M.H.; Park, H.J.; Johnson, P.A. Surface-Enhanced Raman Scattering Detection of DNA Derived from the West Nile Virus Genome Using Magnetic Capture of Raman-Active Gold Nanoparticles. Anal. Chem. 2011, 83, 254–260. [Google Scholar] [CrossRef]
- Zhang, H.; Harpster, M.H.; Wilson, W.C.; Johnson, P.A. Surface-Enhanced Raman Scattering Detection of DNAs Derived from Virus Genomes Using Au-Coated Paramagnetic Nanoparticles. Langmuir 2012, 28, 4030–4037. [Google Scholar] [CrossRef]
- Ngo, H.T.; Wang, H.N.; Fales, A.M.; Nicholson, B.P.; Woods, C.W.; Vo-Dinh, T. DNA bioassay-on-chip using SERS detection for dengue diagnosis. Analyst 2014, 139, 5655–5659. [Google Scholar] [CrossRef]
- Ngo, H.T.; Gandra, N.; Fales, A.M.; Taylor, S.M.; Vo-Dinh, T. Sensitive DNA detection and SNP discrimination using ultrabright SERS nanorattles and magnetic beads for malaria diagnostics. Biosens. Bioelectron. 2016, 81, 8–14. [Google Scholar] [CrossRef]
- Ngo, H.T.; Freedman, E.; Odion, R.A.; Strobbia, P.; De Silva Indrasekara, A.S.; Vohra, P.; Taylor, S.M.; Vo-Dinh, T. Direct Detection of Unamplified Pathogen RNA in Blood Lysate using an Integrated Lab-in-a-Stick Device and Ultrabright SERS Nanorattles. Sci. Rep. 2018, 8, 4075. [Google Scholar] [CrossRef]
- Camacho, S.A.; Sobral-Filho, R.G.; Aoki, P.H.B.; Constantino, C.J.L.; Brolo, A.G. Zika Immunoassay Based on Surface-Enhanced Raman Scattering Nanoprobes. ACS Sens. 2018, 3, 587–594. [Google Scholar] [CrossRef]
- Neng, J.; Harpster, M.H.; Zhang, H.; Mecham, J.O.; Wilson, W.C.; Johnson, P.A. A versatile SERS-based immunoassay for immunoglobulin detection using antigen-coated gold nanoparticles and malachite green-conjugated protein A/G. Biosens. Bioelectron. 2010, 26, 1009–1015. [Google Scholar] [CrossRef]
- Neng, J.; Harpster, M.H.; Wilson, W.C.; Johnson, P.A. Surface-enhanced Raman scattering (SERS) detection of multiple viral antigens using magnetic capture of SERS-active nanoparticles. Biosens. Bioelectron. 2013, 41, 316–321. [Google Scholar] [CrossRef]
- Neng, J.; Li, Y.; Driscoll, A.J.; Wilson, W.C.; Johnson, P.A. Detection of Multiple Pathogens in Serum Using Silica-Encapsulated Nanotags in a Surface-Enhanced Raman Scattering-Based Immunoassay. J. Agric. Food Chem. 2018, 66, 5707–5712. [Google Scholar] [CrossRef]
- Reyes, M.; Piotrowski, M.; Ang, S.K.; Chan, J.; He, S.; Chu, J.J.H.; Kah, J.C.Y. Exploiting the Anti-Aggregation of Gold Nanostars for Rapid Detection of Hand, Foot, and Mouth Disease Causing Enterovirus 71 Using Surface-Enhanced Raman Spectroscopy. Anal. Chem. 2017, 89, 5373–5381. [Google Scholar] [CrossRef]
- Sanchez-Purra, M.; Carre-Camps, M.; de Puig, H.; Bosch, I.; Gehrke, L.; Hamad-Schifferli, K. Surface-Enhanced Raman Spectroscopy-Based Sandwich Immunoassays for Multiplexed Detection of Zika and Dengue Viral Biomarkers. ACS Infect. Dis. 2017, 3, 767–776. [Google Scholar] [CrossRef]
- Paul, A.M.; Fan, Z.; Sinha, S.S.; Shi, Y.; Le, L.; Bai, F.; Ray, P.C. Bio-Conjugated Gold Nanoparticle Based SERS Probe for Ultrasensitive Identification of Mosquito-Borne Viruses Using Raman Fingerprinting. J. Phys. Chem. C Nanomater. Interfaces 2015, 119, 23669–23775. [Google Scholar] [CrossRef]
- Chen, F.; Flaherty, B.R.; Cohen, C.E.; Peterson, D.S.; Zhao, Y. Direct detection of malaria infected red blood cells by surface enhanced Raman spectroscopy. Nanomedicine 2016, 12, 1445–1451. [Google Scholar] [CrossRef]
- Bilal, M.; Saleem, M.; Amanat, S.T.; Shakoor, H.A.; Rashid, R.; Mahmood, A.; Ahmed, M. Optical diagnosis of malaria infection in human plasma using Raman spectroscopy. J. Biomed. Opt. 2015, 20, 017002. [Google Scholar] [CrossRef]
- Bruckner, M.; Becker, K.; Popp, J.; Frosch, T. Fiber array based hyperspectral Raman imaging for chemical selective analysis of malaria-infected red blood cells. Anal. Chim. Acta 2015, 894, 76–84. [Google Scholar] [CrossRef]
- Hobro, A.J.; Konishi, A.; Coban, C.; Smith, N.I. Raman spectroscopic analysis of malaria disease progression via blood and plasma samples. Analyst 2013, 138, 3927–3933. [Google Scholar] [CrossRef]
- Kozicki, M.; Czepiel, J.; Biesiada, G.; Nowak, P.; Garlicki, A.; Weselucha-Birczynska, A. The ring-stage of Plasmodium falciparum observed in RBCs of hospitalized malaria patients. Analyst 2015, 140, 8007–8016. [Google Scholar] [CrossRef]
- Chen, K.; Yuen, C.; Aniweh, Y.; Preiser, P.; Liu, Q. Towards ultrasensitive malaria diagnosis using surface enhanced Raman spectroscopy. Sci. Rep. 2016, 6, 20177. [Google Scholar] [CrossRef]
- Wood, B.R.; Langford, S.J.; Cooke, B.M.; Glenister, F.K.; Lim, J.; McNaughton, D. Raman imaging of hemozoin within the food vacuole ofPlasmodium falciparumtrophozoites. FEBS Lett. 2003, 554, 247–252. [Google Scholar] [CrossRef]
- Wood, B.R.; Langford, S.J.; Cooke, B.M.; Lim, J.; Glenister, F.K.; Duriska, M.; Unthank, J.K.; McNaughton, D. Resonance Raman Spectroscopy Reveals New Insight into the Electronic Structure of β-Hematin and Malaria Pigment. J. Am. Chem. Soc. 2004, 126, 9233–9239. [Google Scholar] [CrossRef]
- Frosch, T.; Koncarevic, S.; Becker, K.; Popp, J. Morphology-sensitive Raman modes of the malaria pigment hemozoin. Analyst 2009, 134, 1126–1132. [Google Scholar] [CrossRef]
- Yuen, C.; Liu, Q. Magnetic field enriched surface enhanced resonance Raman spectroscopy for early malaria diagnosis. Biomed. Opt. 2012, 17, 017005. [Google Scholar] [CrossRef]
- Garrett, N.L.; Sekine, R.; Dixon, M.W.; Tilley, L.; Bambery, K.R.; Wood, B.R. Bio-sensing with butterfly wings: Naturally occurring nano-structures for SERS-based malaria parasite detection. Phys. Chem. Chem. Phys. 2015, 17, 21164–21168. [Google Scholar] [CrossRef]
- DeChiara, N.S.; Wilson, D.J.; Mace, C.R. An Open Software Platform for the Automated Design of Paper-Based Microfluidic Devices. Sci. Rep. 2017, 7, 16224. [Google Scholar] [CrossRef]
- Fu, E.; Liang, T.; Spicar-Mihalic, P.; Houghtaling, J.; Ramachandran, S.; Yager, P. Two-dimensional paper network format that enables simple multistep assays for use in low-resource settings in the context of malaria antigen detection. Anal. Chem. 2012, 84, 4574–4579. [Google Scholar] [CrossRef]
- Anderson, C.E.; Holstein, C.A.; Strauch, E.M.; Bennett, S.; Chevalier, A.; Nelson, J.; Fu, E.; Baker, D.; Yager, P. Rapid Diagnostic Assay for Intact Influenza Virus Using a High Affinity Hemagglutinin Binding Protein. Anal. Chem. 2017, 89, 6608–6615. [Google Scholar] [CrossRef]
- Zagorovsky, K.; Chan, W.C. A plasmonic DNAzyme strategy for point-of-care genetic detection of infectious pathogens. Angew. Chem. Int. Ed. Engl. 2013, 52, 3168–3171. [Google Scholar] [CrossRef]
- Grant, B.D.; Smith, C.A.; Karvonen, K.; Richards-Kortum, R. Highly Sensitive Two-Dimensional Paper Network Incorporating Biotin-Streptavidin for the Detection of Malaria. Anal. Chem. 2016, 88, 2553–2557. [Google Scholar] [CrossRef]
- Shanmukh, S.; Jones, L.; Zhao, Y.P.; Driskell, J.D.; Tripp, R.A.; Dluhy, R.A. Identification and classification of respiratory syncytial virus (RSV) strains by surface-enhanced Raman spectroscopy and multivariate statistical techniques. Anal. Bioanal. Chem. 2008, 390, 1551–1555. [Google Scholar] [CrossRef]
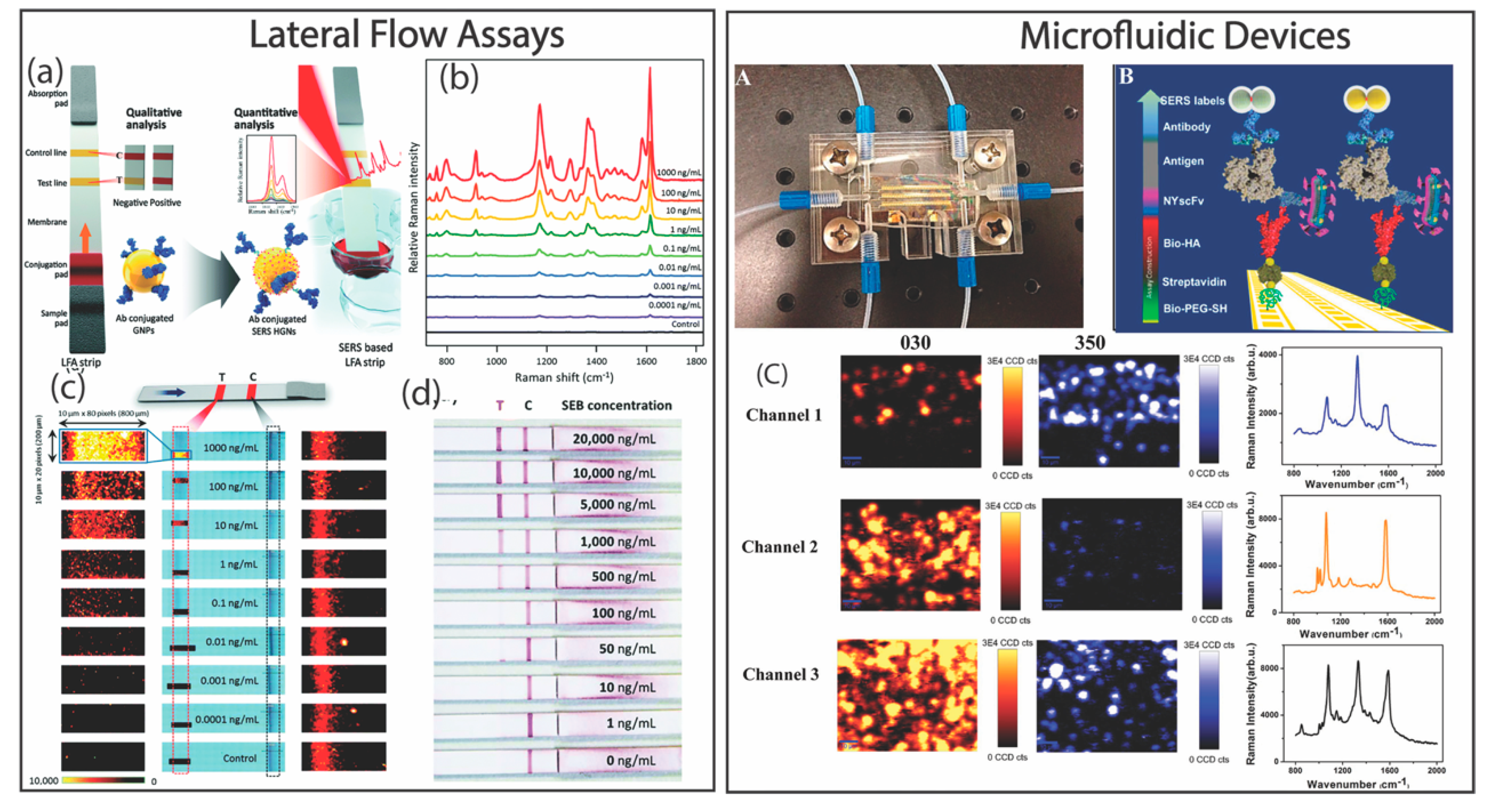
- Fu, X.; Cheng, Z.; Yu, J.; Choo, P.; Chen, L.; Choo, J. A SERS-based lateral flow assay biosensor for highly sensitive detection of HIV-1 DNA. Biosens. Bioelectron. 2016, 78, 530–537. [Google Scholar] [CrossRef]
- Hwang, J.; Lee, S.; Choo, J. Application of a SERS-based lateral flow immunoassay strip for the rapid and sensitive detection of staphylococcal enterotoxin B. Nanoscale 2016, 8, 11418–11425. [Google Scholar] [CrossRef]
- Park, H.J.; Yang, S.C.; Choo, J. Early Diagnosis of Influenza Virus A Using Surface-enhanced Raman Scattering-based Lateral Flow Assay. Bull. Korean Chem. Soc. 2016, 37, 2019–2024. [Google Scholar] [CrossRef]
- Kaminska, A.; Witkowska, E.; Winkler, K.; Dziecielewski, I.; Weyher, J.L.; Waluk, J. Detection of Hepatitis B virus antigen from human blood: SERS immunoassay in a microfluidic system. Biosens. Bioelectron. 2015, 66, 461–467. [Google Scholar] [CrossRef]
- Sanchez-Purra, M.; Roig-Solvas, B.; Rodriguez-Quijada, C.; Leonardo, B.M.; Hamad-Schifferli, K. Reporter Selection for Nanotags in Multiplexed Surface Enhanced Raman Spectroscopy Assays. ACS Omega 2018, 3, 10733–10742. [Google Scholar] [CrossRef]
- De Silva Indrasekara, A.S.; Norton, S.J.; Geitner, N.K.; Crawford, B.M.; Wiesner, M.R.; Vo-Dinh, T. Tailoring the Core–Satellite Nanoassembly Architectures by Tuning Internanoparticle Electrostatic Interactions. Langmuir 2018, 34, 14617–14623. [Google Scholar] [CrossRef]
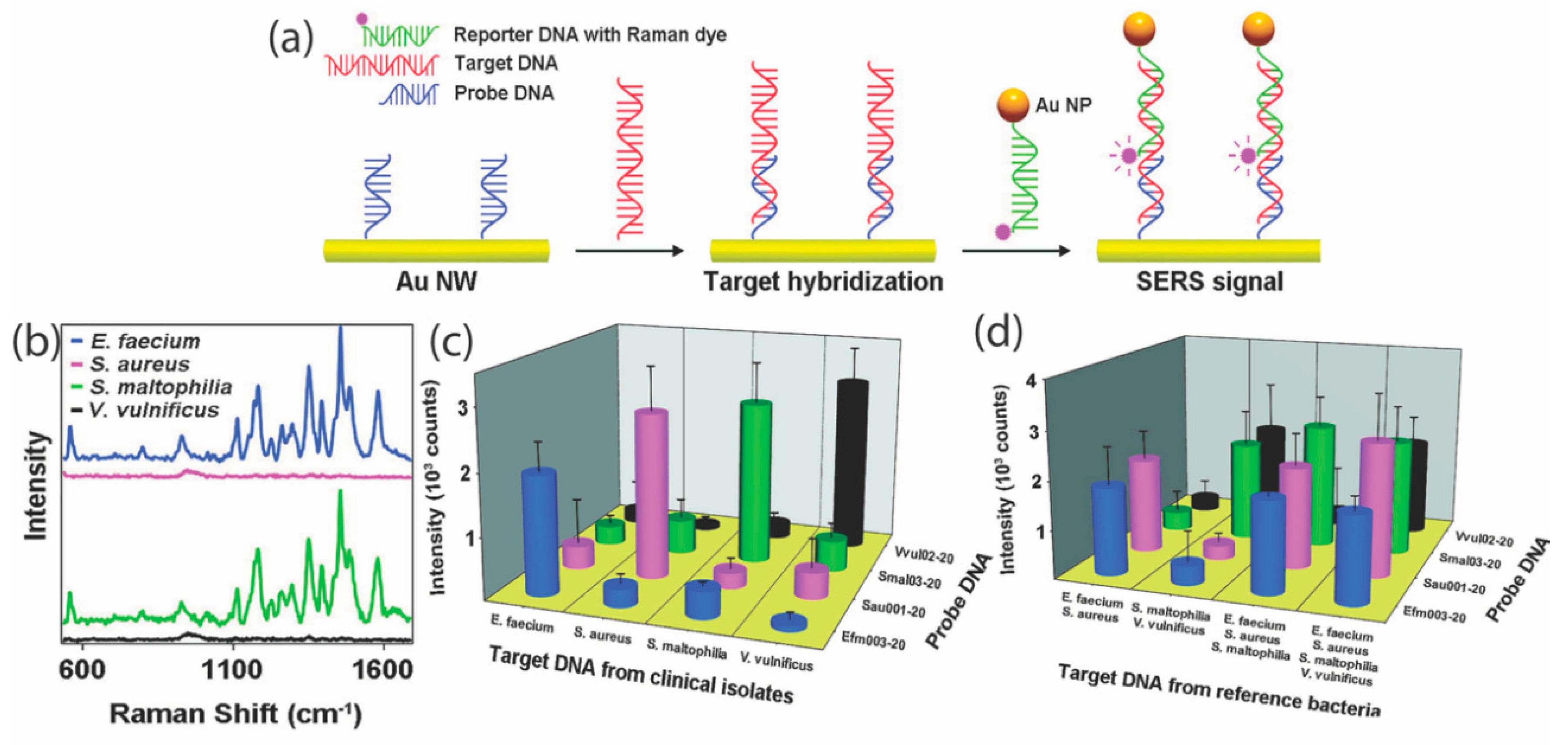
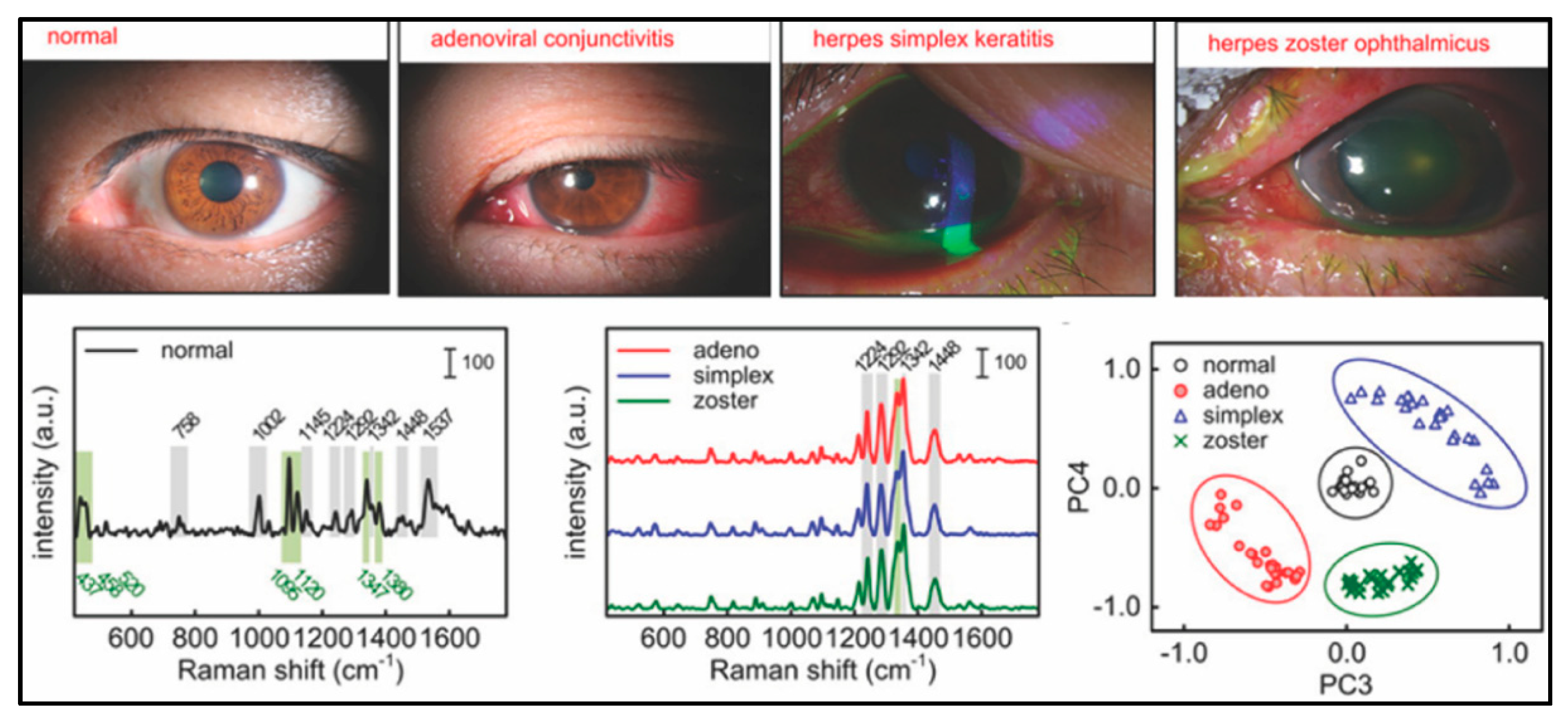
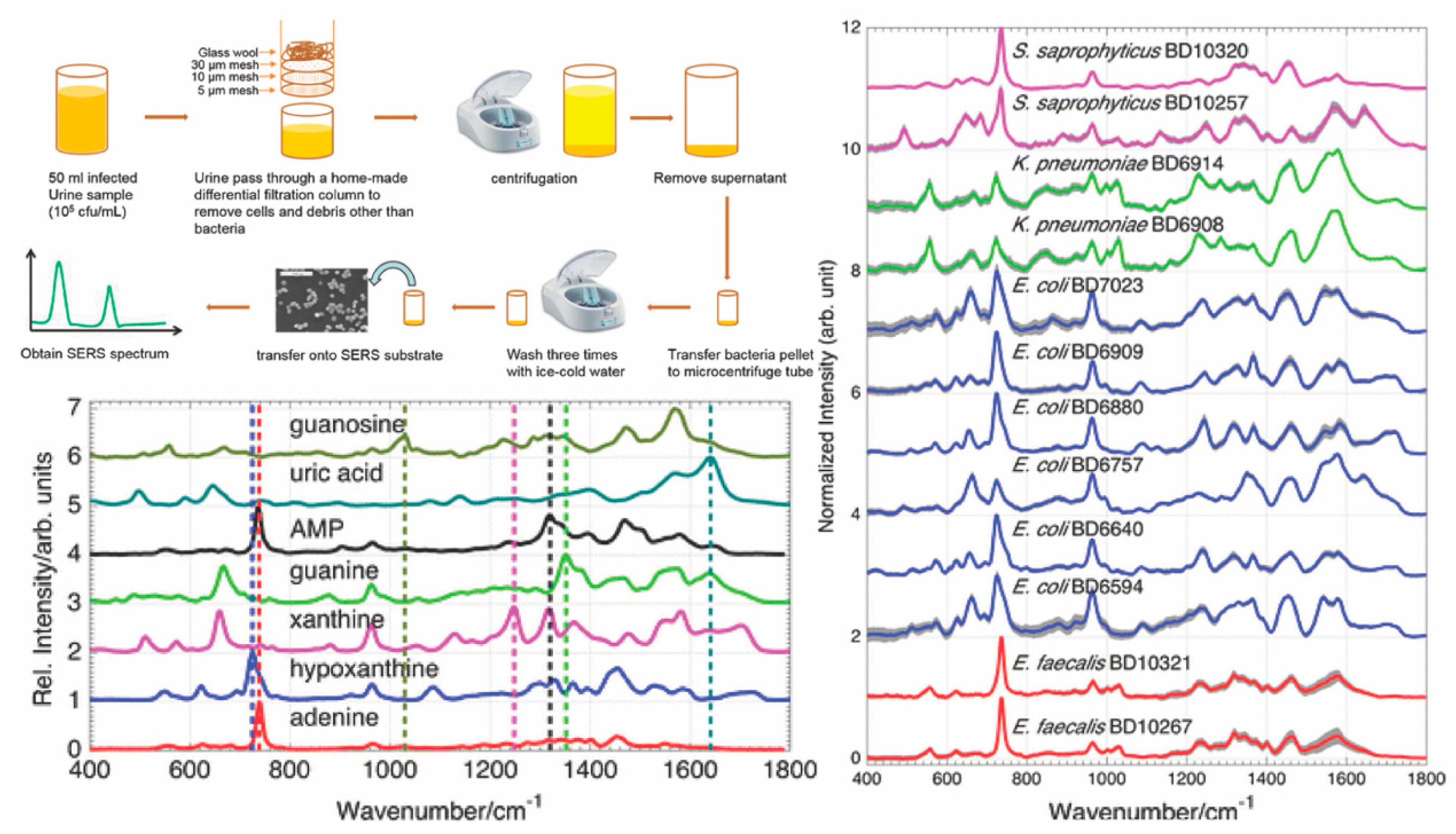
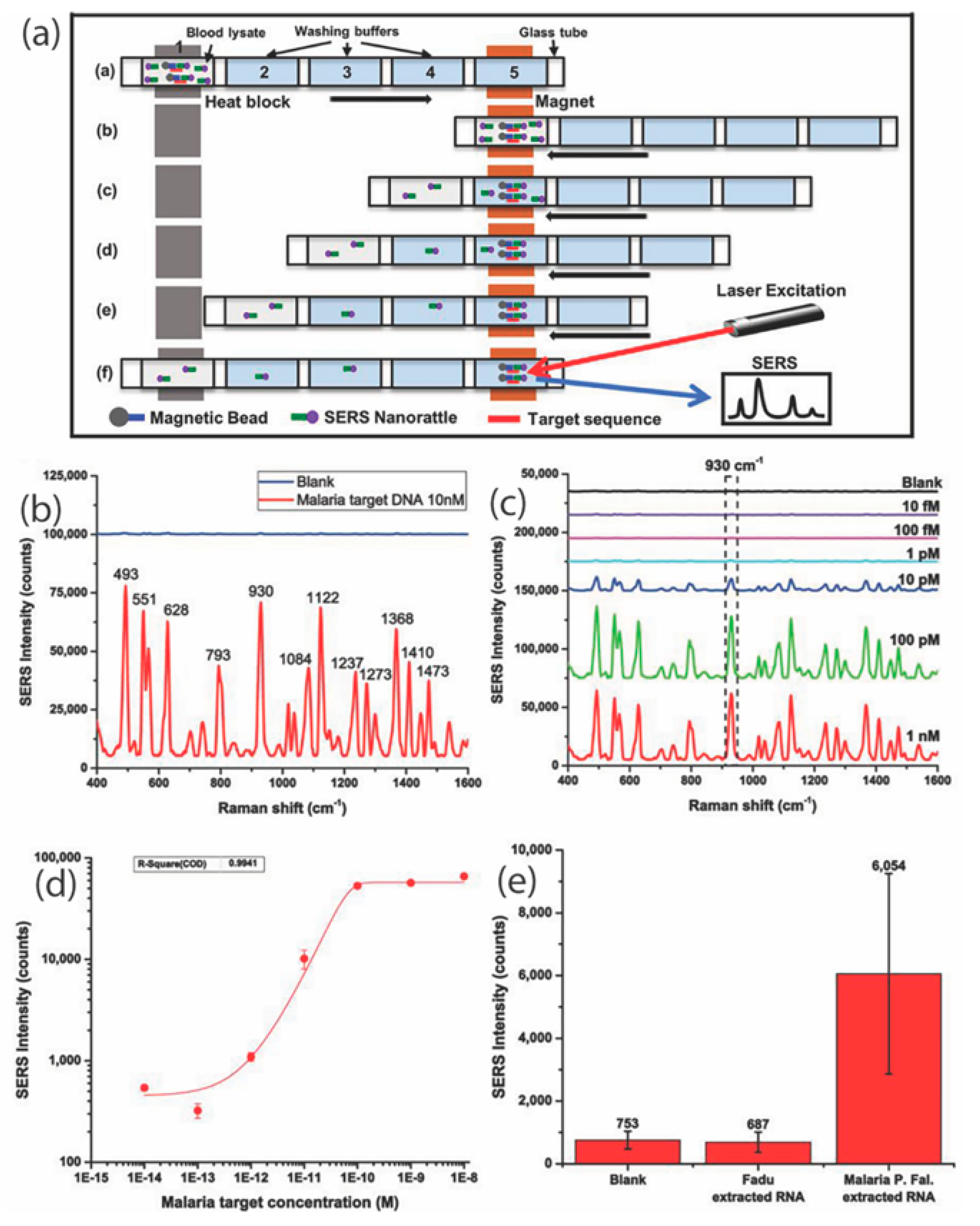
| Disease | Target | Detection Scheme | Transducer | LOD | Ref. |
|---|---|---|---|---|---|
| Bacterial Infections | |||||
| Various bacterial infections: E. faecium, S. aureus, S. Maltophilia and V. vulnificus | DNA extracted from clinical samples of cerebrospinal fluid, stool, and sputum | Indirect SERS | Gold nanowires & nanoparticles | 10 pM | [66] |
| Bacterial meningitis pathogens: N. meningitidis, S. pneumoniae and H. influenzae | Synthetic DNA | Indirect SERS | Silver nanoparticles | pM range | [5] |
| contagious bovine pleuropneumonia (CBPP) | MmmSC_1046 (lipoprotein lppQ), MmmSC_0136 (hypothetical transmembrane protein), and Botau_12S (mitochondrial 12S rRNA) | Indirect SERS | streptavidin-modified magnetic nanoparticles & silver nanoparticles | Not reported | [72] |
| Bacterial meningitis pathogens: N. meningitidis, S. pneumoniae and H. influenzae | Unique Nitrogen base composition of target DNA & Neopterin (metabolites) | Direct SERS | polycarbonate membranes coated with gold & silver | Neopterin: nM range | [74] |
| Tuberculosis | mannose-capped lipoarabinomannan (ManLAM), a stimulant of antigenic biomarker in serum | Indirect SERS | Gold film-coated substrate & gold nanoparticles | handheld Raman system (0.18 ng/mL) & benchtop Ramaninstrument (0.032 ng/mL) | [90] |
| methicillin-resistant S. aureus (MRSA) and methicillin-sensitive S. aureus (MSSA) | mecA gene and cellular proteins | Direct SERS | Silver nanocolloids | 5% to 100% | [73] |
| Urinary tract infection caused by E. coli and P. mirabilis | cell membrane proteins | Direct SERS | Silver nanoparticles | Not reported | [85] |
| Pneumonia caused by M. pneumoniae | Surface proteins of bacteria in clinical throat swabs | Direct SERS | Silver nanorod arrays | 82 CFU/sample | [80] |
| Infections caused by Streptococcus suis II | Muramidase released protein (MRP) antibody in pig serum | Indirect SERS | Thorny gold nanoparticles | 0.1 pg/mL | [88] |
| E. coli | Cellular surface antigens in real water samples | Indirect SERS | Gold nanorods with gold-coated magnetic nanoparticles | 8 CFU/mL | [84] |
| Keratoconjunctivitides | Protein biomarkers in eye fluid | Direct SERS | Gold nanoparticles | 1 pM | [91] |
| Pseudomonas aeruginosa | Pyocyanin metabolite in clinical sputum samples | Indirect SERS | Silver nanorod array | 5 ppb | [78] |
| Mosquito-borne Infectious Diseases | |||||
| West Nile Virus | Viral DNA | Indirect SERS | Paramagnetic nanoparticles and gold nanoparticles | 20–100 nM | [92] |
| Dengue | Viral DNA | Indirect SERS | Gold and silver Nanowaves | 6 attomoles | [94] |
| Malaria | Parasitic DNA | Indirect SERS | nanorattles and magnetic beads | 3 picomolar | [95] |
| Malaria | Ribosomal RNA in infected blood lysates | Indirect SERS | Nanorattles and magnetic beads | 200 fM | [96] |
| Zika | ZIKV Nonstructural protein 1 (NS1) protein | Indirect SERS | Silica-coated gold nanoparticles | 10 ng/mL | [97] |
| West Nile Virus | WNV protein | Indirect SERS | Gold Nanostars | 2 ng/mL | [98] |
| West Nile Virus and Rift Valley Virus | Antigens of WNV and RVF | Indirect SERS | Gold nanoparticles and Paramagnetic nanoparticles | 5 fg/mL | [99] |
| West Nile Virus, Valley Fever Virus and Yersinia pestis | Viral pathogenic antigens | Indirect SERS | Silica-coated gold nanoparticles and Paramagnetic nanoparticles | 10 pg/mL | [100] |
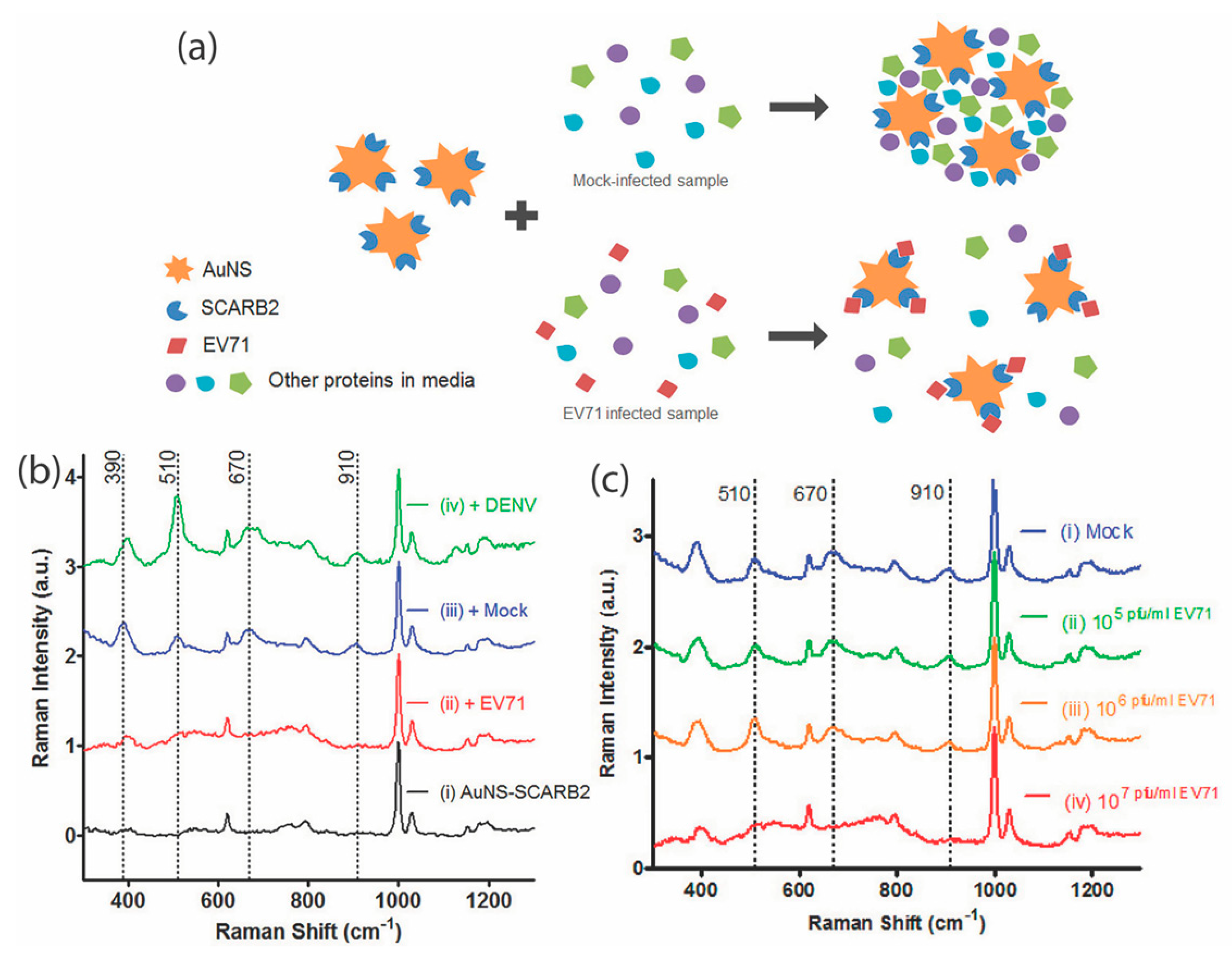
| Hand, Foot and Mouth Disease causing virus | Enterovirus 71 (EV71) | Direct SERS | Gold Nanostars | 106 to 108 pfu/mL | [101] |
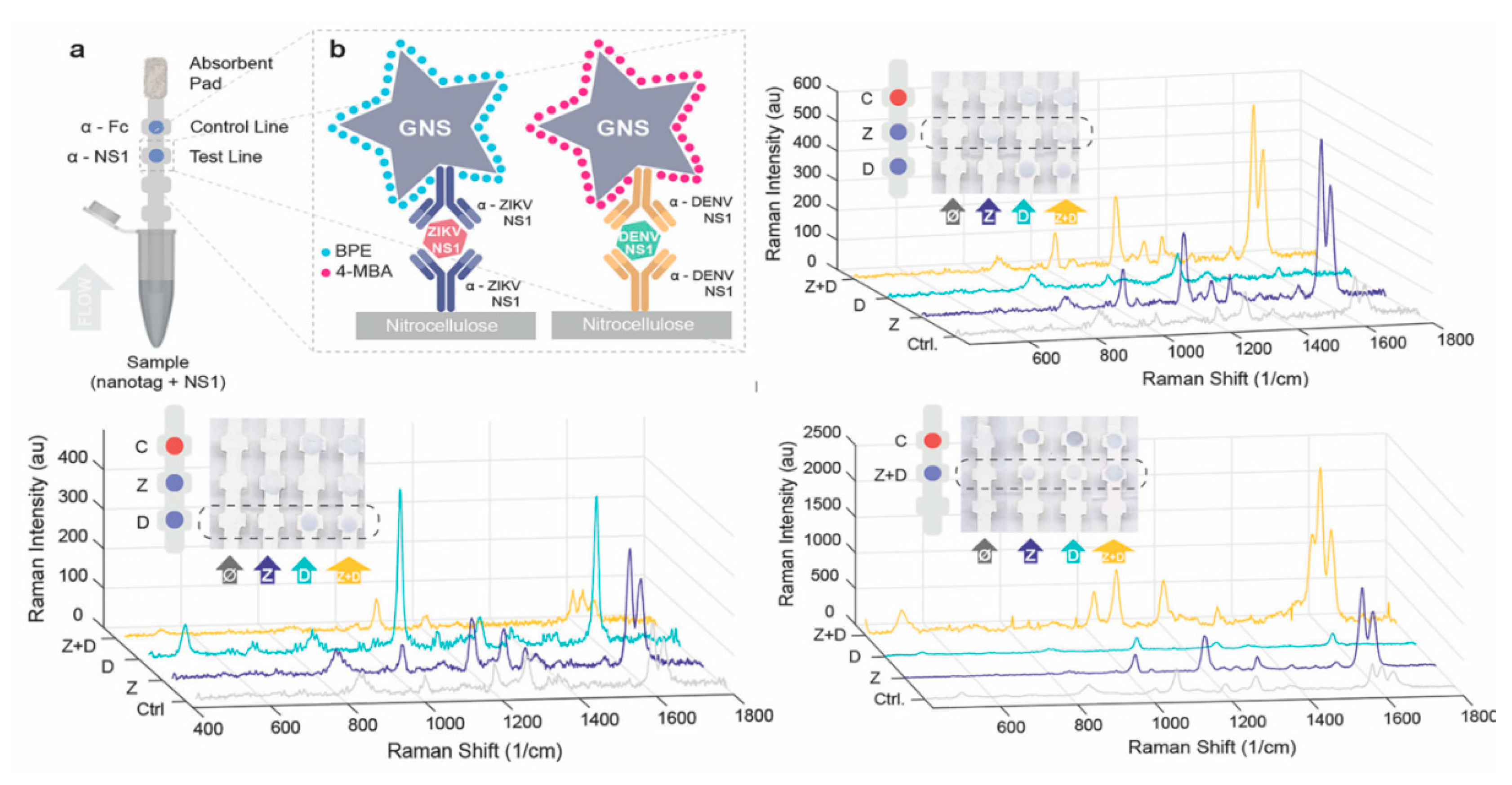
| Zika and Dengue | Nonstructural protein 1 (NS1) | Indirect SERS | Gold Nanostars | 0.72 ng/mL of ZIKV NS1 and 7.67 ng/mL of DENV NS1 | [102] |
| West Nile Virus and Dengue | DENV-2 and WNV proteins in serum | Direct SERS | Gold nanoparticles | 10 pfu/mL | [103] |
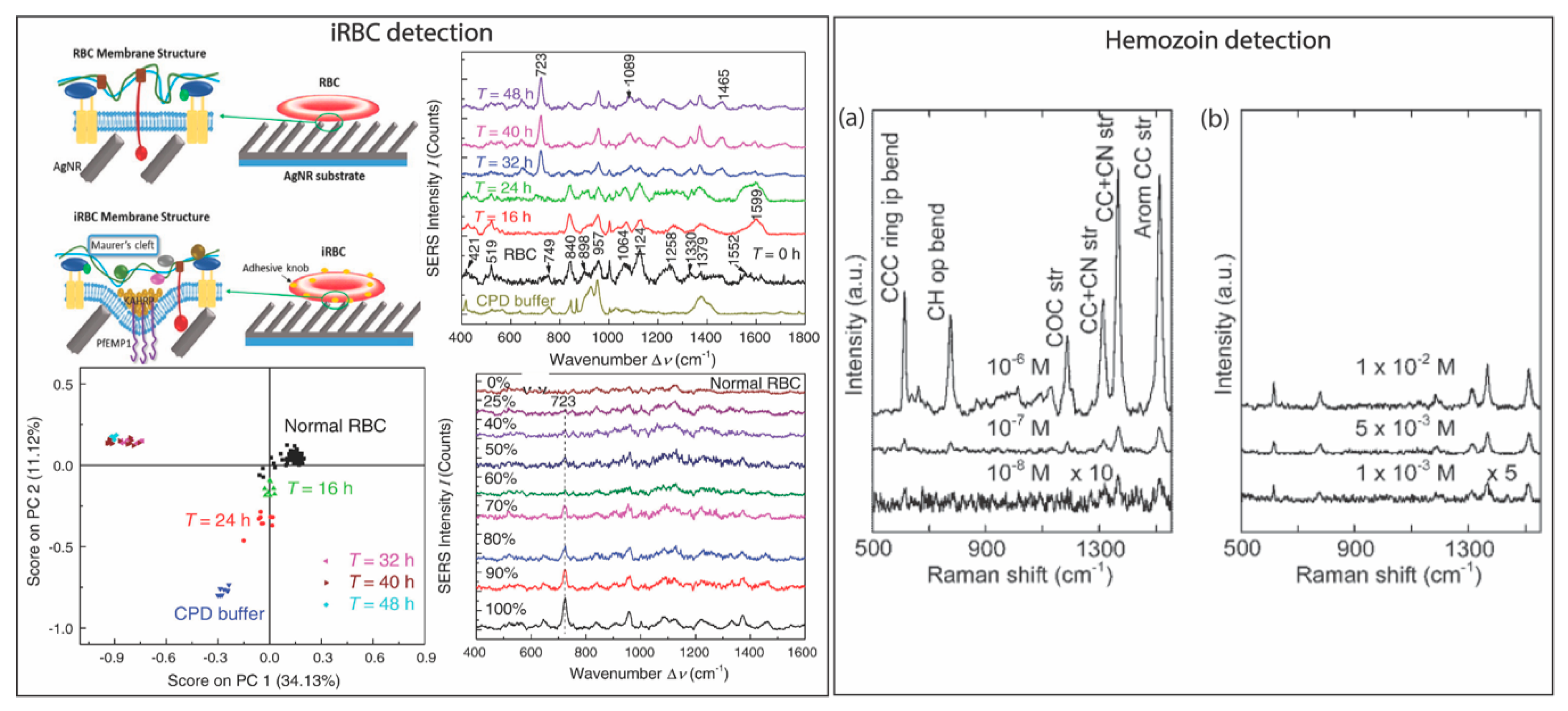
| Malaria | Plasmodium falciparum infected red blood cells (iRBC) | Direct SERS | Silver nanorod | 1.5 × 107/mL | [104] |
| Malaria | Hemozoin in iRBC | direct SERS | Iron oxide core and silver shell nanoparticles | 5 nM | [113] |
| Malaria | Hemozoin in iRBC | direct SERS | Silver nanoparticles | 2.5 parasites/μL | [109] |
| Malaria | Hemozoin in iRBC lysate | direct SERS | Gold-coated butterfly wings | 0.0005% and 0.005% | [114] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hamm, L.; Gee, A.; De Silva Indrasekara, A.S. Recent Advancement in the Surface-Enhanced Raman Spectroscopy-Based Biosensors for Infectious Disease Diagnosis. Appl. Sci. 2019, 9, 1448. https://doi.org/10.3390/app9071448
Hamm L, Gee A, De Silva Indrasekara AS. Recent Advancement in the Surface-Enhanced Raman Spectroscopy-Based Biosensors for Infectious Disease Diagnosis. Applied Sciences. 2019; 9(7):1448. https://doi.org/10.3390/app9071448
Chicago/Turabian StyleHamm, Logan, Amira Gee, and A. Swarnapali De Silva Indrasekara. 2019. "Recent Advancement in the Surface-Enhanced Raman Spectroscopy-Based Biosensors for Infectious Disease Diagnosis" Applied Sciences 9, no. 7: 1448. https://doi.org/10.3390/app9071448
APA StyleHamm, L., Gee, A., & De Silva Indrasekara, A. S. (2019). Recent Advancement in the Surface-Enhanced Raman Spectroscopy-Based Biosensors for Infectious Disease Diagnosis. Applied Sciences, 9(7), 1448. https://doi.org/10.3390/app9071448





