A Review on Biomaterials for 3D Conductive Scaffolds for Stimulating and Monitoring Cellular Activities
Abstract
1. Introduction
2. Materials and Methods
- the study was a scientific article written in English;
- the study was focused on the use of conductive biomaterials for cell monitoring;
- the study reported information about the choice of the used materials, their properties and the manufacturing process.
3. Results
3.1. Conductive Polymers
- Polypyrrole (PPy)—PPy is conductive conjugated polymer with excellent mechanical, electrical and stimulus-responsive characteristics, which makes it one of the most conductive biomaterials, appropriate to be used in biomedicine [33]. Polypyrrole possesses great chemical stability in water and air [34,35], and high electrical conductivity under biological conditions [36,37,38,39]. It can be synthesized with various different porosities and can be adjusted to make it more feasible for biomedical applications by incorporating with bioactive molecules [40,41,42]. Moreover, its stimulus responsive nature allows to control its properties by applying electrical potential [39,43]. As a biomaterial, PPy also supports cell growth and adhesion of a number of different types of cell, which makes it a suitable for scaffold fabrication in tissue engineering [44,45,46,47].
- Polyaniline (PANI)—The second most studied conductive polymer is polyaniline (PANI) which is commonly known as aniline black. It is classified into three types depending on its oxidation level, such as the fully oxidized form is pernigraniline base, half-oxidized form is emeraldine base and fully reduced form is leucoemeraldine base. Among these, most conductive and stable is PANI emaraldine [48,49]. PANI offers many advantages over other conducting polymers, such as low-cost, easy to synthesize, and able to electrically switch between its resistive and conductive states [50,51,52,53,54]. Studies have shown that PANI and its derivative support cell growth [55] and hence can be used for scaffold fabrication.
- Poly (3,4-ethylenedioxythiophene) (PEDOT)—Another conductive polymer is PEDOT which is a polythiophene (PTh) derivative [48,56]. PEDOT possesses good electrical, environmental and chemical stability [56]. It also have better thermal stability and conductivity than PPy [49,56]. Luo et al. (2008) [57] carried out an in vivo study and examined the biocompatibility of PEDOT by seeding NIH3T3 fibroblasts cells on PEDOT based films. The results showed good inflammatory response at the implantation site with low intrinsic cytotoxicity, hence making them suitable for biological and biosensing applications e.g., in neural electrodes [49,56,58], heart muscle patches, and nerve grafts [56].
3.2. Conductive Fillers
3.2.1. Carbon-Based Solutions
- Carbon Black—Carbon black fillers are preferred over metal fillers as they do not undergo oxidation, whereas, metal fillers get oxidized and create an insulation layer on particles surface [75]. Other advantages of hybrid composites made from carbon black fillers include: flexibility, light weight process capabilities, absorption of mechanical shock, and low production costs [76]. In [77], carbon black nanoparticles have been used to develop an electric responsive scaffold for cardiomyocytes-related experiments, showing good adhesion and viability.
- Carbon Nanotubes—Carbon nanotubes (CNTs) possess extremely high strength together with low density and high electrical conductivity. CNTs are widely used in scaffold-related applications, as they support cell adhesion and their dimensions are comparable with extracellular matrix (ECM) molecules [78]. In [79,80], CNTs are used as carbon filler showing that these fillers has remarkably increased the conductivity and mechanical strength of designed scaffolds. A 3D-structure with tunable porosity for cardiac tissue engineering was developed, showing reinforced properties in terms of mechanical and electrical properties thanks to DWCNTs [81]. Also nerve tissue engineering applications, regarding both central nervous system (CNS) and peripheral nervous system (PNS), were studied in presence of CNTs showing cells proliferation and viability [78,82].
- Carbon Fibers and Nanofibers—Carbon nanofibers (CNFs) could mechanically strengthen scaffolds and have the availability of higher amount of sites on the outer wall respect CNTs, thus facilitating electron transfer for electroactive analytes such as proteins and enzymes [83]. In [83], CNFs were successfully used in a chitosan matrix to fabricate an electrically conductive scaffold with a highly porous and interconnected structure for cardiac tissue engineering exhibiting high electrical properties and an elastic modulus similar to that of rat myocardium. CNFs were also incorporated in a biocompatible scaffold made up of cotton, which was carbonized, oxidized, and coated with polydopamine (PDA) to study nerve cells proliferation and differentiation, obtaining a final material similar to native cellular physiological environment facilitating cell adhesion, proliferation, differentiation, and other biofunctions [84].
- Graphene—Graphene is a single layer of carbon atoms in a hexagonal lattice, capable of high mechanical properties combined with high electrical conductivity and biocompatibility. Graphene showed an important versatility since it was employed as filler in different base materials like chitosan/gelatin matrices, hydrogels and Poly(trimethylene carbonate) (PTMC). Biological properties like better adhesion, spreading and proliferation of cells on the conductive graphene make it a suitable material for scaffold based applications in tissue engineering [85,86,87,88,89,90,91].
3.2.2. Metal-Based Solutions
- Gold—An interesting metal which has attracted attention in fields ranging from drug delivery, cancer therapies and regenerative medicine is gold, specifically gold nanoparticles (AuNPs). Gold is noble metal characterized by fundamental properties for tissue engineering like: biocompatibility, electrical, optical and catalytic properties, ease of functionalization, higher mechanical properties. It has been proven that AuNPs are capable to enhance intercellular electrical communications, augment cardiomyocyte function and cardiac differentiation of MSC. They have been used to produce a unique extracellular milieu that couples tunable conductivity and elasticity for cardiac tissue engineering (CTE) [92,93,94]. AuNPs have been employed together with different polymers to obtain conductive scaffolds evidencing an outgrowth process, where neurites originate from the soma, extend and branch, developing into a complex dendritic tree [95].
- Platinum—Platinum Nanoparticles (PtNPs) have been indicated as promising biocompatible metal, which, when incorporated within scaffold, produce a positive influence both on cell growth and conductivity of the overall scaffold; thus, ensuring the possibility of an electric-based monitoring of cellular activities. Additionally, they protect cells from oxidation-induced inflammation which inhibits pulmonary inflammation and induced bone loss by decreasing osteoclastogenesis. PtNPs also assisted cell growth and could promote cell proliferation [96].
- Silver—Silver (Ag) is a well-known material already used in coatings for biomedical devices and in wound care products thanks to its antibacterial properties. Since it is a metal, it shows electrical conductivity and charge storage capacity [97,98]. It must be underlined that this metal is under debate regarding the aspect of biocompatibility: Ag antibacterial properties are associated to the release of Ag+ ions and their release rate can be affected by silver nanoparticles (AgNPs) surface to volume ratio and capping agents, thus leading in toxicity problems [98].
- Zinc—Complex tissue regeneration of peripheral nervous system (PNS) and, particularly central nervous system (CNS) (no spontaneous regeneration due to inhibitory cues connected to astroglial cells) suggest the usage of nanoparticles due to their ability to reduce activity of undesirable cells. In order to enhance neural tissue regeneration in electrical fields through conductive materials, like piezoelectric materials, zinc is a promising candidate in this field. This metal presents important properties like biodegradability (lower degradation rate than iron and magnesium (0.018–0.145 mm/year)), biocompatibility, suturability, non-toxic behavior and rapid healing capacity [99,100].
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Edmondson, R.; Broglie, J.J.; Adcock, A.F.; Yang, L. Three-Dimensional Cell Culture Systems and Their Applications in Drug Discovery and Cell-Based Biosensors. Assay Drug Dev. Technol. 2014, 12, 207–218. [Google Scholar] [CrossRef] [PubMed]
- Kujala, V.J.; Jimenez, Z.C.; Väisänen, J.; Tanskanen, J.M.A.; Kerkelä, E.; Hyttinen, J.; Aalto-Setälä, K. Averaging in vitro cardiac field potential recordings obtained with microelectrode arrays. Comput. Methods Programs Biomed. 2011, 104, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Y.; Liao, R.; Zhang, X. Real-time monitoring primary cardiomyocyte adhesion based on electrochemical impedance spectroscopy and electrical cell-substrate impedance sensing. Anal. Chem. 2008, 80, 990–996. [Google Scholar] [CrossRef] [PubMed]
- Rangarajan, S.; Madden, L.; Bursac, N. Use of flow, electrical, and mechanical stimulation to promote engineering of striated muscles. Ann. Biomed. Eng. 2014, 42, 1391–1405. [Google Scholar] [CrossRef] [PubMed]
- Gu, W.; Zhao, Y. Cellular electrical impedance spectroscopy: An emerging technology of microscale biosensors. Expert Rev. Med. Devices 2010, 7, 767–779. [Google Scholar] [CrossRef] [PubMed]
- Thomas, C.A.; Springer, P.A.; Loeb, G.E.; Berwald-Netter, Y.; Okun, L.M. A miniature microelectrode array to monitor the bioelectric activity of cultured cells. Exp. Cell Res. 1972, 74, 61–66. [Google Scholar] [CrossRef]
- Giaever, I.; Keese, C.R. Use of electric fields to monitor the dynamical aspect of cell behavior in tissue culture. IEEE Trans. Biomed. Eng. 1986, 33, 242–247. [Google Scholar] [CrossRef] [PubMed]
- Gross, G.W.; Rieske, E.; Kreutzberg, G.W.; Meyer, A. A new fixed-array multi-microelectrode system designed for long-term monitoring of extracellular single unit neuronal activity in vitro. Neurosci. Lett. 1977, 6, 101–105. [Google Scholar] [CrossRef]
- Reppel, M.; Pillekamp, F.; Lu, Z.J.; Halbach, M.; Brockmeier, K.; Fleischmann, B.K.; Hescheler, J. Microelectrode arrays: A new tool to measure embryonic heart activity. J. Electrocardiol. 2004, 37, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Fendyur, A.; Spira, M.E. Toward on-chip, in-cell recordings from cultured cardiomyocytes by arrays of gold mushroom-shaped microelectrodes. Front. Neuroeng. 2012, 5, 21. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Xie, X.; Duan, Y.; Wang, L.; Cheng, Z.; Cheng, J. A review of impedance measurements of whole cells. Biosens. Bioelectron. 2016, 77, 824–836. [Google Scholar] [CrossRef] [PubMed]
- Poulton, E.-J. Impedance Measurement in Induced Pluripotent Stem Cell-Derived Cardiomyocytes. In Drug Safety Evaluation; Gautier, J.-C., Ed.; Springer: New York, NY, USA, 2017; pp. 201–209. [Google Scholar]
- Szulcek, R.; Bogaard, H.J.; Amerongen, G.P.V.N. Electric Cell-substrate Impedance Sensing for the Quantification of Endothelial Proliferation, Barrier Function, and Motility. J. Vis. Exp. 2014, 51300. [Google Scholar] [CrossRef] [PubMed]
- Ramasamy, S.; Bennet, D.; Kim, S. Drug and bioactive molecule screening based on a bioelectrical impedance cell culture platform. Int. J. Nanomed. 2014, 9, 5789–5809. [Google Scholar] [CrossRef]
- Sun, T.; Tsuda, S.; Zauner, K.-P.; Morgan, H. On-chip electrical impedance tomography for imaging biological cells. Biosens. Bioelectron. 2010, 25, 1109–1115. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Zhou, W.; Yang, Y.; Jia, J.; Bagnaninchi, P. Exploring the Potential of Electrical Impedance Tomography for Tissue Engineering Applications. Materials 2018, 11, 930. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Zhang, Q.; Gao, S.; Song, Q.; Huang, R.; Wang, L.; Liu, L.; Dai, J.; Tang, M.; Cheng, G. Three-dimensional graphene foam as a biocompatible and conductive scaffold for neural stem cells. Sci. Rep. 2013, 3, 1604. [Google Scholar] [CrossRef] [PubMed]
- Sain, M.; Ummartyotin, S.; Juntaro, J.; Wu, C.; Manuspiya, H. Deposition of PEDOT: PSS nanoparticles as a conductive microlayer anode in OLEDs device by desktop inkjet printer. J. Nanomater. 2011, 2011, 606714. [Google Scholar] [CrossRef]
- Khan, M.A.; Lopomo, N.F.; Serpelloni, M.; Sardini, E.; Sartore, L. Characterization of Sensorized Porous 3D Gelatin/Chitosan Scaffolds via Bio-impedance Spectroscopy. In Sensors, Lecture Notes in Electrical Engineering; Springer: Cham, Switzerland, 2018; Volume 539. [Google Scholar]
- Pissis, P.; Kyritsis, A. Electrical conductivity studies in hydrogels. Solid State Ion. 1997, 97, 105–113. [Google Scholar] [CrossRef]
- Mawad, D.; Lauto, A.; Wallace, G.G. Conductive Polymer Hydrogels. In Polymeric Hydrogels as Smart Biomaterials; Kalia, S., Ed.; Springer International Publishing: Cham, Switzerland, 2016; pp. 19–44. [Google Scholar]
- Balint, R.; Cassidy, N.J.; Cartmell, S.H. Conductive polymers: Towards a smart biomaterial for tissue engineering. Acta Biomater. 2014, 10, 2341–2353. [Google Scholar] [CrossRef] [PubMed]
- Choi, W.J.; Jung, J.; Lee, S.; Chung, Y.J.; Yang, C.-S.; Lee, Y.K.; Lee, Y.-S.; Park, J.K.; Ko, H.W.; Lee, J.-O. Effects of substrate conductivity on cell morphogenesis and proliferation using tailored, atomic layer deposition-grown ZnO thin films. Sci. Rep. 2015, 5, 9974. [Google Scholar] [CrossRef] [PubMed]
- Ravikumar, K.; Kar, G.P.; Bose, S.; Basu, B. Synergistic effect of polymorphism, substrate conductivity and electric field stimulation towards enhancing muscle cell growth in vitro. RSC Adv. 2016, 6, 10837–10845. [Google Scholar] [CrossRef]
- Chronakis, I.S.; Grapenson, S.; Jakob, A. Conductive polypyrrole nanofibers via electrospinning: Electrical and morphological properties. Polymer 2006, 47, 1597–1603. [Google Scholar] [CrossRef]
- Skotheim, T.A.; Reynolds, J.R. Handbook of Conducting Polymers, 3rd ed.; CRC Press: Gainesville, FL, USA, 2007. [Google Scholar]
- Zhang, Q.; Yan, Y.; Li, S.; Feng, T. The synthesis and characterization of a novel biodegradable and electroactive polyphosphazene for nerve regeneration. Mater. Sci. Eng. C 2010, 30, 160–166. [Google Scholar] [CrossRef]
- Bettinger, C.J.; Bruggeman, J.P.; Misra, A.; Borenstein, J.T.; Langer, R. Biocompatibility of biodegradable semiconducting melanin films for nerve tissue engineering. Biomaterials 2009, 30, 3050–3057. [Google Scholar] [CrossRef] [PubMed]
- Guimard, N.K.; Gomez, N.; Schmidt, C.E. Conducting polymers in biomedical engineering. Prog. Polym. Sci. 2007, 32, 876–921. [Google Scholar] [CrossRef]
- MacDiarmid, A.G. “Synthetic Metals”: A Novel Role for Organic Polymers (Nobel Lecture). Angew. Chem. Int. Ed. 2001, 40, 2581–2590. [Google Scholar] [CrossRef]
- Heeger, A.J. Semiconducting and Metallic Polymers: The Fourth Generation of Polymeric Materials (Nobel Lecture). Angew. Chem. Int. Ed. 2001, 40, 2591–2611. [Google Scholar] [CrossRef]
- Ravichandran, R.; Sundarrajan, S.; Venugopal, J.R.; Mukherjee, S.; Ramakrishna, S. Applications of conducting polymers and their issues in biomedical engineering. J. R. Soc. Interface 2010, 7, S559–S579. [Google Scholar] [CrossRef] [PubMed]
- Garner, B.; Georgevich, A.; Hodgson, A.J.; Liu, L.; Wallace, G.G. Polypyrrole–heparin composites as stimulus-responsive substrates for endothelial cell growth. J. Biomed. Mater. Res. 1999, 44, 121–129. [Google Scholar] [CrossRef]
- Bousalem, S.; Mangeney, C.; Chehimi, M.M.; Basinska, T.; Miksa, B.; Slomkowski, S. Synthesis, characterization and potential biomedical applications of Nsuccinimidyl ester functionalized, polypyrrole-coated polystyrene latex particles. Colloid Polym. Sci. 2004, 282, 1301–1307. [Google Scholar] [CrossRef]
- Cetiner, S.; Kalaoglu, F.; Karakas, H.; Sarac, A.S. Electrospun nanofibers of polypyrrole–poly(acrylonitrile co-vinyl acetate). Text. Res. J. 2010, 80, 1784–1792. [Google Scholar] [CrossRef]
- Kim, D.H.; Richardson-Burns, S.M.; Hendricks, J.L.; Sequera, C.; Martin, D.C. Effect of immobilized nerve growth factor on conductive polymers: Electrical properties and cellular response. Adv. Funct. Mater. 2007, 17, 79–86. [Google Scholar] [CrossRef]
- Li, Y.; Neoh, K.G.; Kang, E.T. Plasma protein adsorption and thrombus formation on surface functionalized polypyrrole with and without electrical stimulation. J. Colloid Interfaces Sci. 2004, 275, 488–495. [Google Scholar] [CrossRef] [PubMed]
- Akkouch, A.; Shi, G.; Zhang, Z.; Rouabhia, M. Bioactivating electrically conducting polypyrrole with fibronectin and bovine serum albumin. J. Biomed. Mater. Res. A 2010, 92, 221–231. [Google Scholar] [CrossRef] [PubMed]
- Ferraz, N.; Strømme, M.; Fellstrom, B.; Pradhan, S.; Nyholm, L.; Mihranyan, A. In vitro and in vivo toxicity of rinsed and aged nanocellulose–Polypyrrole composites. J. Biomed. Mater. Res. A 2012, 100, 2128–2138. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Hetke, J.E.; Wiler, J.A.; Anderson, D.J.; Martin, D.C. Electrochemical deposition and characterization of conducting polymer polypyrole/PSS on multichannel neural probes. Sens. Actuator A Phys. 2001, 93, 8–18. [Google Scholar] [CrossRef]
- Gomez, N.; Schmidt, C.E. Nerve growth factor-immobilized polypyrrole: Bioactive electrically conducting polymer for enhanced neurite extension. J. Biomed. Mater. Res. A 2007, 81, 135–149. [Google Scholar] [CrossRef] [PubMed]
- Song, H.K.; Toste, B.; Ahmann, K.; Hoffman-Kim, D.; Palmore, G.T.R. Micropatterns of positive guidance cues anchored to polypyrrole doped with polyglutamic acid: A new platform for characterizie neurite extension in complex environments. Biomaterials 2006, 27, 473–484. [Google Scholar] [CrossRef] [PubMed]
- Garner, B.; Hodgson, A.J.; Wallace, G.G.; Underwood, P.A. Human endothelial cell attachment to and growth on polypyrrole–heparin is fibronectin dependent. J. Mater. Sci. Mater. Med. 1999, 10, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Gomez, N.; Lee, J.Y.; Nickels, J.D.; Schmidt, C.E. Micropatterned polypyrrole: A combination of electrical and topographical characteristics for the stimulation of cells. Adv. Funct. Mater. 2007, 17, 1645–1653. [Google Scholar] [CrossRef] [PubMed]
- Castano, H.; O’Rear, E.A.; McFetridge, P.S. Polypyrrole thin films formed by admicellar polymerization support the osteogenic differentiation of mesenchymal stem cells. Macromol. Biosci. 2004, 4, 785–794. [Google Scholar] [CrossRef] [PubMed]
- Evans, G.R. Peripheral nerve injury: A review and approach to tissue engineered constructs. Anat. Rec. 2001, 263, 396–404. [Google Scholar] [CrossRef] [PubMed]
- Ateh, D.D.; Navsaria, H.A.; Vadgama, P. Review: Polypyrrole-based conducting polymers and interactions with biological tissues. J. R. Soc. Interface 2006, 3, 741–752. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi-Mobarakeh, L.; Prabhakaran, M.P.; Morshed, M.; Nasr-Esfahani, M.H.; Baharvand, H.; Kiani, S.; Al-Deyab, S.S.; Ramakrishna, S. Application of conductive polymers, scaffolds and electrical stimulation for nerve tissue engineering. J. Tissue Eng. Regen. Med. 2011, 5, e17–e35. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.D.; Cui, X.T.; Hines, A.; Greenberg, R.J. Conducting polymers in neural stimulation applications. In Implantable Neural Prostheses; Zhou, D.D., Greenbaum, E., Eds.; Springer: Berlin, Germany, 2010; Volume 2, pp. 217–252. [Google Scholar]
- Cullen, D.K.; Patel, A.R.; Doorish, J.F.; Smith, D.H.; Pfister, B.J. Developing a tissueengineered neural-electrical relay using encapsulated neuronal constructs on conducting polymer fibers. J. Neural Eng. 2008, 5, 374–384. [Google Scholar] [CrossRef] [PubMed]
- Borriello, A.; Guarino, V.; Schiavo, L.; Alvarez-Perez, M.A.; Ambrosio, L. Optimizing PANi doped electroactive substrates as patches for the regeneration of cardiac muscle. J. Mater. Sci. Mater. Med. 2011, 22, 1053–1062. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Li, M.; Mylonakis, A.; Han, J.; MacDiarmid, A.G.; Chen, X.; Lelkes, P.I.; Wei, Y. Electroactive oligoaniline-containing self-assembled monolayers for tissue engineering applications. Biomacromolecules 2007, 8, 3025–3034. [Google Scholar] [CrossRef] [PubMed]
- Prabhakaran, M.P.; Ghasemi-Mobarakeh, L.; Jin, G.; Ramakrishna, S. Electrospun conducting polymer nanofibers and electrical stimulation of nerve stem cells. J. Biosci. Bioeng. 2011, 112, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Yu, Q.Z.; Shi, M.M.; Deng, M.; Wang, M.; Chen, H.Z. Morphology and conductivity of polyaniline sub-micron fibers prepared by electrospinning. Mater. Sci. Eng. B Solid 2008, 150, 70–76. [Google Scholar] [CrossRef]
- Mattioli-Belmonte, M.; Giavaresi, G.; Biagini, G.; Virgili, L.; Giacomini, M.; Fini, M.; Giantomassi, F.; Natali, D.; Torricelli, P.; Giardino, R. Tailoring biomaterial compatibility: In vivo tissue response versus in vitro cell behavior. Int. J. Artif. Organs 2003, 26, 1077–1085. [Google Scholar] [CrossRef] [PubMed]
- Peramo, A.; Urbanchek, M.G.; Spanninga, S.A.; Povlich, L.K.; Cederna, P.; Martin, D.C. In situ polymerization of a conductive polymer in acellular muscle tissue constructs. Tissue Eng. Part A 2008, 14, 423–432. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.C.; Mohamed Ali, E.; Tansil, N.C.; Yu, H.H.; Gao, S.; Kantchev, E.A.; Ying, J.Y. Poly(3,4-ethylenedioxythiophene) (PEDOT) nanobiointerfaces: Thin, ultrasmooth, and functionalized PEDOT films with in vitro and in vivo biocompatibility. Langmuir 2008, 24, 8071–8077. [Google Scholar] [CrossRef] [PubMed]
- Asplund, M.; Thaning, E.; Lundberg, J.; Sandberg-Nordqvist, A.C.; Kostyszyn, B.; Inganäs, O.; von Holst, H. Toxicity evaluation of PEDOT/biomolecular composites intended for neural communication electrodes. Biomed. Mater. 2009, 4, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Karimullah, A.; Cumming, R.S.; Riehle, M.; Gadegaard, N. Development of a Conducting Polymer Cell Impedance Sensor. Sens. Actuators B 2013, 176, 667–674. [Google Scholar] [CrossRef]
- Del Agua, I.; Mantione, D.; Marina, S.; Pitsalidis, C.; Ferro, M.; Sanchez-Sanchez, A.; Owens, R.M.; Malliaras, G.G.; Mecerreyes, D. Conducting Polymer Scaffolds based on PEDOT and Xanthan Gum for live-cell Monitoring. ACS Omega 2018, 3, 7424–7431. [Google Scholar] [CrossRef] [PubMed]
- Atanasov, V.; Knorr, N.; Duran, R.S.; Ingebrandt, S.; Offenhausser, A.; Knoll, W.; Köper, I. Membrane on a Chip: A Functional Tethered Lipid Bilayer Membrane on Silicon Oxide Surfaces. Biophys. J. 2005, 89, 1780–1788. [Google Scholar] [CrossRef] [PubMed]
- Dai, L. Conducting polymers. In Intelligent Macromolecules for Smart Devices: From Materials Synthesis to Device Applications; Dai, L., Ed.; Springer: London, UK, 2004; pp. 41–80. [Google Scholar]
- Zhou, J.F.; Wang, Y.G.; Cheng, L.; Wu, Z.; Sun, X.D.; Peng, J. Preparation of polypyrrole-embedded electrospun poly(lactic acid) nanofibrous scaffolds for nerve tissue engineering. Neural Regen. Res. 2016, 11, 1644–1652. [Google Scholar] [PubMed]
- Liu, X.; Chen, J.; Gilmore, K.J.; Higgins, M.J.; Liu, Y.; Wallace, G.G. Guidance of neurite outgrowth on aligned electrospun polypyrrole/poly-(styrene-β-isobutylene-β-styrene) fiber platforms. J. Biomed. Mater. Res. A 2010, 94, 1004–1011. [Google Scholar] [CrossRef] [PubMed]
- Pelto, J.; Björninen, M.; Pälli, A.; Talvitie, E.; Hyttinen, J.; Mannerström, B.; Suuronen Seppanen, R.; Kellomäki, M.; Miettinen, S.; Haimi, S. Novel polypyrrole-coated polylactide scaffolds enhance adipose stem cell proliferation and early osteogenic differentiation. Tissue Eng. Part A 2013, 19, 882–892. [Google Scholar] [CrossRef] [PubMed]
- Yu, Q.Z.; Xu, S.L.; Zhang, K.H.; Shan, Y.M. Multi-porous electroactive poly(L-lactic acid)/polypyrrole composite micro/nano fibrous scaffolds promote neurite outgrowth in PC12 cells. Neural Regen. Res. 2013, 8, 31–38. [Google Scholar] [PubMed]
- Sudwilai, T.; Ng, J.J.; Boonkrai, C.; Israsena, N.; Chuangchote, S.; Supaphol, P. Polypyrrole-coated electrospun poly(lactic acid) fibrous scaffold: Effects of coating on electrical conductivity and neural cell growth. J. Biomater. Sci. Polym. Ed. 2014, 25, 1240–1252. [Google Scholar] [CrossRef] [PubMed]
- Ufere, S.K.J.; Sultana, N. Fabrication and characterization of pcl/ha/ppy composite scaffold using freeze-drying technique. J. Teknol. 2016, 78, 89–94. [Google Scholar]
- Sharma, Y.; Tiwari, A.; Hattori, S.; Terada, D.; Sharma, A.K.; Ramalingam, M.; Kobayashi, H. Fabrication of conducting electrospun nanofibers scaffold for three-dimensional cells culture. Int. J. Biol. Macromol. 2012, 51, 627–631. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.I.; Jun, I.D.; Choi, M.J.; Nho, Y.C.; Lee, Y.M.; Shin, H. Development of Electroactive and Elastic Nanofibers That Contain Polyaniline and Poly (L-Lactide-Co-e-Caprolactone) for the Control of Cell Adhesion. Macromol. Biosci. 2008, 8, 627–637. [Google Scholar] [CrossRef] [PubMed]
- Iandolo, D.; Ravichandran, A.; Liu, X.; Wen, F.; Chan, J.K.Y.; Berggren, M.; Teoh, S.H.; Simon, D.T. Development and characterization of organic electronic scaffolds for bone tissue engineering. Adv. Healthc. Mater. 2016, 5, 1505–1512. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudinezhad, M.H.; Karkhaneh, A.; Jadidi, K. Effect of PEDOT:PSS in tissue engineering composite scaffold on improvement and maintenance of endothelial cell function. J. Biosci. 2018, 43, 307–319. [Google Scholar] [CrossRef] [PubMed]
- Jin, G.; Li, K. The electrically conductive scaffold as the skeleton of stem cell niche in regenerative medicine. Mater. Sci. Eng. C 2014, 45, 671–681. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.C. Carbon black filled conducting polymers and polymer blends. Adv. Polym. Technol. 2002, 21, 299–313. [Google Scholar] [CrossRef]
- Zois, H.; Apekis, L.; Mamunya, Y.P. Dielectric properties and morphology of polymer composites filled with dispersed iron. J. Appl. Polym. Sci. 2003, 88, 3013–3020. [Google Scholar] [CrossRef]
- Tanasa, F.; Zanoaga, M.; Mamunya, Y. Conductive Thermoplastic Polymer Nanocomposites with Ultralow Percolation Threshold; Scientific Research & Education in the Air Force-AFASES: Brasov, Slovak Republic, 2014; Volume 2. [Google Scholar]
- Kim, H.; Zhu, B.; Chen, H.; Adetiba, O.; Agrawal, A.; Ajayan, P.; Jacot, J.G.; Verduzco, R. Preparation of Monodomain Liquid Crystal Elastomers and Liquid Crystal Elastomer Nanocomposites. J. Vis. Exp. 2016, 53688. [Google Scholar] [CrossRef] [PubMed]
- Whulanza, Y.; Ucciferri, N.; Domenici, C.; Vozzi, G.; Ahluwalia, A. Biosensors and Bioelectronics Sensing scaffolds to monitor cellular activity using impedance measurements. Biosens. Bioelectron. 2011, 26, 3303–3308. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.F.; Shen, L.; Zhang, W.D.; Tong, Y.J. Preparation and Mechanical Properties of Chitosan/Carbon Nanotubes Composites. Biomacromolecules 2005, 6, 3067–3072. [Google Scholar] [CrossRef] [PubMed]
- Whulanza, Y.; Battini, E.; Vannozzi, L.; Vomero, M.; Ahluwalia, A.; Vozzi, G. Electrical and mechanical characterisation of single wall carbon nanotubes based composites for Tissue engineering applications. J. Nanosci. Nanotechnol. 2013, 13, 188–197. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Kataoka, M.; Jung, Y.C.; Ko, Y.I.; Fujisawa, K.; Hayashi, T.; Kim, Y.A.; Endo, M. Mechanically Tough, Electrically Conductive Polyethylene Oxide Nanofiber Web Incorporating DNA-Wrapped Double-Walled Carbon Nanotubes. ACS Appl. Mater. Interfaces 2013, 5, 4150–4154. [Google Scholar] [CrossRef] [PubMed]
- Behan, B.L.; DeWitt, D.G.; Bogdanowicz, D.R.; Koppes, A.N.; Bale, S.S.; Thompson, D.M. Single-walled carbon nanotubes alter Schwann cell behavior differentially within 2D and 3D environments. J. Biomed. Mater. Res. Part A 2010, 96, 46–57. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.M.; Eng, G.; Caridade, G.; Mano, F.; Reis, R.L. Electrically Conductive Chitosan/Carbon Scaffolds for Cardiac Tissue Engineering. Biomacromolecules 2014, 15, 635–643. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Wu, Y.; Ranjan, V.D.; Zhang, Y. Three-dimensional electrical conductive scaffold from biomaterial-based carbon micro fiber sponge with bioinspired coating for cell proliferation and differentiation. Carbon 2018, 134, 174–182. [Google Scholar] [CrossRef]
- Baniasadi, H.; SA, A.R.; Mashayekhan, S.; Ghaderinezhad, F. Preparation of conductive polyaniline/graphene nanocomposites via in situ emulsion polymerization and product characterization. Synth. Met. 2014, 196, 199–205. [Google Scholar] [CrossRef]
- Javed, K.; Krumme, A.; Viirsalu, M.; Krasnou, I.; Plamus, T.; Vassiljeva, V.; Tarasova, E.; Savest, N.; Mere, A.; Mikli, V.; et al. A method for producing conductive graphene biopolymer nano fibrous fabrics by exploitation of an ionic liquid dispersant in electrospinning. Carbon 2018, 140, 148–156. [Google Scholar] [CrossRef]
- Sayyar, S.; Bjorninen, M.; Haimi, S.; Miettinen, S.; Gilmore, K.; Grijpma, D.; Wallace, G. UV Cross-Linkable Graphene/Poly(trimethylene Carbonate) Composites for 3D Printing of Electrically Conductive Scaffolds. ACS Appl. Mater. Interfaces 2016, 8, 31916–31925. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Zhang, X.; Yu, X.; Wang, S.; Qiu, J.; Tang, W.; Li, L.; Liu, H.; Wang, Z.L. Self-Powered Electrical Stimulation for Enhancing Neural Differentiation of Mesenchymal Stem Cells on Graphene–Poly(3,4-ethylenedioxythiophene) Hybrid Microfibers. ACS Nano 2016, 10, 5086–5095. [Google Scholar] [CrossRef] [PubMed]
- Boroojeni, F.R.R.; Mashayekhan, S.; Abbaszadeh, H.; Ansarizadeh, M. Conductive Nanofiber Scaffold for Bone Tissue Engineering. In Proceedings of the 2017 24th National and 2nd International Iranian Conference on Biomedical Engineering (ICBME), Tehran, Iran, 30 November–1 December 2017; pp. 1–5. [Google Scholar]
- Baniasadi, H.; SA, A.R.; Mashayekhan, S. International Journal of Biological Macromolecules Fabrication and characterization of conductive chitosan/gelatin-based scaffolds for nerve tissue engineering. Int. J. Biol. Macromol. 2015, 74, 360–366. [Google Scholar] [CrossRef] [PubMed]
- Sartore, L.; Dey, K.; Agnelli, S.; Bignotti, F.; Lopomo, N.; Khan, M.A.; Barbera, V.; Galimberti, M. Novel nanobiocomposite hydrogels based on gelatin/chitosan and functionalized grapheme. In Proceedings of the 9th International Conference on Times of Polymers and Composites, Ischia, Italy, 17–21 June 2018. [Google Scholar]
- McKeon-Fischer, K.D.; Freeman, J.W. Characterization of electrospun poly(L-lactide) and gold nanoparticle composite scaffolds for skeletal muscle tissue engineering. J. Tissue Eng. Regen. Med. 2010, 5, 560–568. [Google Scholar] [CrossRef] [PubMed]
- Ravichandran, R.; Sridhar, R.; Venugopal, J.R.; Sundarrajan, S.; Mukherjee, S.; Ramakrishna, S. Gold Nanoparticle Loaded Hybrid Nanofibers for Cardiogenic Differentiation of Stem Cells for Infarcted Myocardium Regeneration. Macromol. Biosci. 2014, 14, 515–525. [Google Scholar] [CrossRef] [PubMed]
- Baei, P.; Jalili-firoozinezhad, S.; Rajabi-zeleti, S.; Tafazzoli-shadpour, M. Electrically conductive gold nanoparticle-chitosan thermosensitive hydrogels for cardiac tissue engineering. Mater. Sci. Eng. C 2016, 63, 131–141. [Google Scholar] [CrossRef] [PubMed]
- Baranes, K.; Shevach, M.; Shefi, O.; Dvir, T. Gold Nanoparticle-Decorated Scaffolds Promote Neuronal Differentiation and Maturation. Nano Lett. 2016, 16, 2916–2920. [Google Scholar] [CrossRef] [PubMed]
- Eid, K.; Eldesouky, A.; Fahmy, A.; Shahat, A.; Abdelaal, R. Calcium Phosphate Scaffold Loaded with Platinum Nanoparticles for Bone Allograft. Am. J. Biomed. Sci. 1937, 5, 242–249. [Google Scholar] [CrossRef]
- Nguyen, H.L.; Jo, Y.; Cha, M.; Cha, Y.; Yoon, D.; Sanandiya, N.; Prajatelistia, E.; Oh, D.; Hwang, D. Mussel-Inspired Anisotropic Nanocellulose and Silver Nanoparticle Composite with Improved Mechanical Properties, Electrical Conductivity and Antibacterial Activity. Polymers 2016, 8, 102. [Google Scholar] [CrossRef]
- Wickham, A.; Vagin, M.; Khalaf, H.; Bertazzo, S.; Hodder, P.; Dånmark, S.; Bengtsson, T.; Altimiras, J.; Aili, D. Electroactive biomimetic collagen-silver nanowire composite scaffolds. Nanoscale 2016, 8, 14146–14155. [Google Scholar] [CrossRef] [PubMed]
- Webster, T.J. Decreased astroglial cell adhesion and proliferation on zinc oxide nanoparticle polyurethane composites. Int. J. Nanomed. 2008, 3, 523–531. [Google Scholar] [CrossRef]
- Sezer, U.A.; Ozturk, K.; Aru, B. Zero valent zinc nanoparticles promote neuroglial cell proliferation: A biodegradable and conductive filler candidate for nerve regeneration. J. Mater. Sci. Mater. Med. 2017, 28, 19. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Holzwarth, J.M.; Yan, Y.; Xu, P.; Zheng, H.; Yin, Y.; Li, S.; Ma, P.X. Conductive PPY/PDLLA conduit for peripheral nerve regeneration. Biomaterials 2014, 35, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Bashur, C.A.; Milroy, C.A.; Forciniti, L.; Goldstein, A.S.; Schmidt, C.E. Nerve growth factor-immobilized electrically conducting fibrous scaffolds for potential use in neural engineering applications. IEEE Trans. Nanobiosci. 2012, 11, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Bashur, C.A.; Goldstein, A.S.; Schmidt, C.E. Polypyrrole-coated electrospun PLGA nanofibers for neural tissue applications. Biomaterials 2009, 30, 4325–4335. [Google Scholar] [CrossRef] [PubMed]
- Runge, M.B.; Dadsetan, M.; Baltrusaitis, J.; Knight, A.M.; Ruesink, T.; Lazcano, E.A.; Lu, L.; Windebank, A.J.; Yaszemski, M.J. The development of electrically conductive polycaprolactone fumarate–polypyrrole composite materials for nerve regeneration. Biomaterials 2010, 31, 5916–5926. [Google Scholar] [CrossRef] [PubMed]
- Abidian, M.R.; Daneshvar, E.D.; Egeland, B.M.; Kipke, D.R.; Cederna, P.S.; Urbanchek, M.G. Hybrid conducting polymer–hydrogel conduits for axonal growth and neural tissue engineering. Adv. Healthc. Mater. 2012, 1, 762–767. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Guan, S.; Wang, J.; Liu, H.; Liu, T.; Ma, X.; Cui, Z. Fabrication and characterization of conductive poly (3, 4-ethylenedioxythiophene) doped with hyaluronic acid/poly (L-lactic acid) composite film for biomedical application. J. Biosci. Bioeng. 2017, 123, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Bidez, P.R.; Li, S.; MacDiarmid, A.G.; Venancio, E.C.; Wei, Y.; Lelkes, P.I. Polyaniline, an electroactive polymer, supports adhesion and proliferation of cardiac myoblasts. J. Biomater. Sci. Polym. Ed. 2006, 17, 199–212. [Google Scholar] [CrossRef] [PubMed]
- Jun, I.; Jeong, S.; Shin, H. The stimulation of myoblast differentiation by electrically conductive sub-micron fibers. Biomaterials 2009, 30, 2038–2047. [Google Scholar] [CrossRef] [PubMed]
- Ku, S.H.; Lee, S.H.; Park, C.B. Synergic effects of nanofiber alignment and electroactivity on myoblast differentiation. Biomaterials 2012, 33, 6098–6104. [Google Scholar] [CrossRef] [PubMed]
- Nishizawa, M.; Nozaki, H.; Kaji, H.; Kitazume, T.; Kobayashi, N.; Ishibashi, T.; Abe, T. Electrodeposition of anchored polypyrrole film on microelectrodes and stimulation of cultured cardiac myocytes. Biomaterials 2007, 28, 1480–1485. [Google Scholar] [CrossRef] [PubMed]
- Kai, D.; Prabhakaran, M.P.; Jin, G.; Ramakrishna, S. Polypyrrole-contained electrospun conductive nanofibrous membranes for cardiac tissue engineering. J. Biomed. Mater. Res. A 2011, 99, 376–385. [Google Scholar] [CrossRef] [PubMed]
- Kharaziha, M.; Shin, S.R.; Nikkhah, M.; Topkaya, S.N.; Masoumi, N.; Annabi, N.; Dokmeci, M.R.; Khademhosseini, A. Tough and flexible CNT–polymeric hybrid scaffolds for engineering cardiac constructs. Biomaterials 2014, 35, 7346–7354. [Google Scholar] [CrossRef] [PubMed]
- Azhar, F.F.; Olad, A.; Salehi, R. Fabrication and characterization of chitosan–gelatin/nanohydroxyapatite–polyaniline composite with potential application in tissue engineering scaffolds. Des. Monomers Polym. 2014, 17, 654–667. [Google Scholar] [CrossRef]
- Chen, J.; Yu, M.; Guo, B.; Ma, P.X.; Yin, Z. Conductive nanofibrous composite scaffolds based on in-situ formed polyaniline nanoparticle and polylactide for bone regeneration. J. Colloid Interface Sci. 2018, 514, 517–527. [Google Scholar] [CrossRef] [PubMed]
- Pan, L.; Pei, X.; He, R.; Wan, Q.; Wang, J. Multiwall carbon nanotubes/polycaprolactone composites for bone tissue engineering application. Colloids Surf. B 2012, 93, 226–234. [Google Scholar] [CrossRef] [PubMed]
- Shokuhfar, T.; Makradi, A.; Titus, E.; Cabral, G.; Ahzi, S.; Sousa, A.C.M.; Belouettar, S.; Gracio, J. Prediction of the mechanical properties of hydroxyapatite/polymethyl methacrylate/carbon nanotubes nanocomposite. J. Nanosci. Nanotechnol. 2008, 8, 4279–4284. [Google Scholar] [CrossRef] [PubMed]
- Kotov, N.A.; Winter, J.O.; Clements, I.P.; Jan, E.; Timko, B.P.; Campidelli, S.; Pathak, S.; Mazzatenta, A.; Lieber, C.M.; Prato, M.; et al. Nanomaterials for neural interfaces. Adv. Mater. 2009, 21, 3970–4004. [Google Scholar] [CrossRef]
- Huang, L.; Hu, J.; Lang, L.; Wang, X.; Zhang, P.; Jing, X.; Wang, X.; Chen, X.; Lelkes, P.I.; MacDiarmid, A.G.; et al. Synthesis and characterization of electroactive and biodegradable ABA block copolymer of polylactide and aniline pentamer. Biomaterials 2007, 28, 1741–1751. [Google Scholar] [CrossRef] [PubMed]
- Rivers, T.J.; Hudson, T.W.; Schmidt, C.E. Synthesis of a novel biodegradable electrically conducting polymer for biomedical applications. Adv. Funct. Mater. 2002, 12, 33–37. [Google Scholar] [CrossRef]
- Shi, G.; Rouabhia, M.; Wang, Z.; Dao, L.H.; Zhang, Z. A novel electrically conductive and biodegradable composite made of Polypyrrole nanoparticles and polylactide. Biomaterials 2004, 25, 2477–2488. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Hu, X.; Lu, L.; Ye, Z.; Zhang, Q.; Luo, Z. Electrical regulation of Schwann cells using conductive polypyrrole/chitosan polymers. J. Biomed. Mater. Res. A 2010, 93, 164–174. [Google Scholar] [CrossRef] [PubMed]
- Mermoud, Y.; Felder, M.; Stucki, J.D.; Guenat, O.T. Microimpedance tomography system to monitor cell activity and membrane movements in a breathing lung-onchip. Sens. Actuators B 2018, 225, 3647–3653. [Google Scholar] [CrossRef]
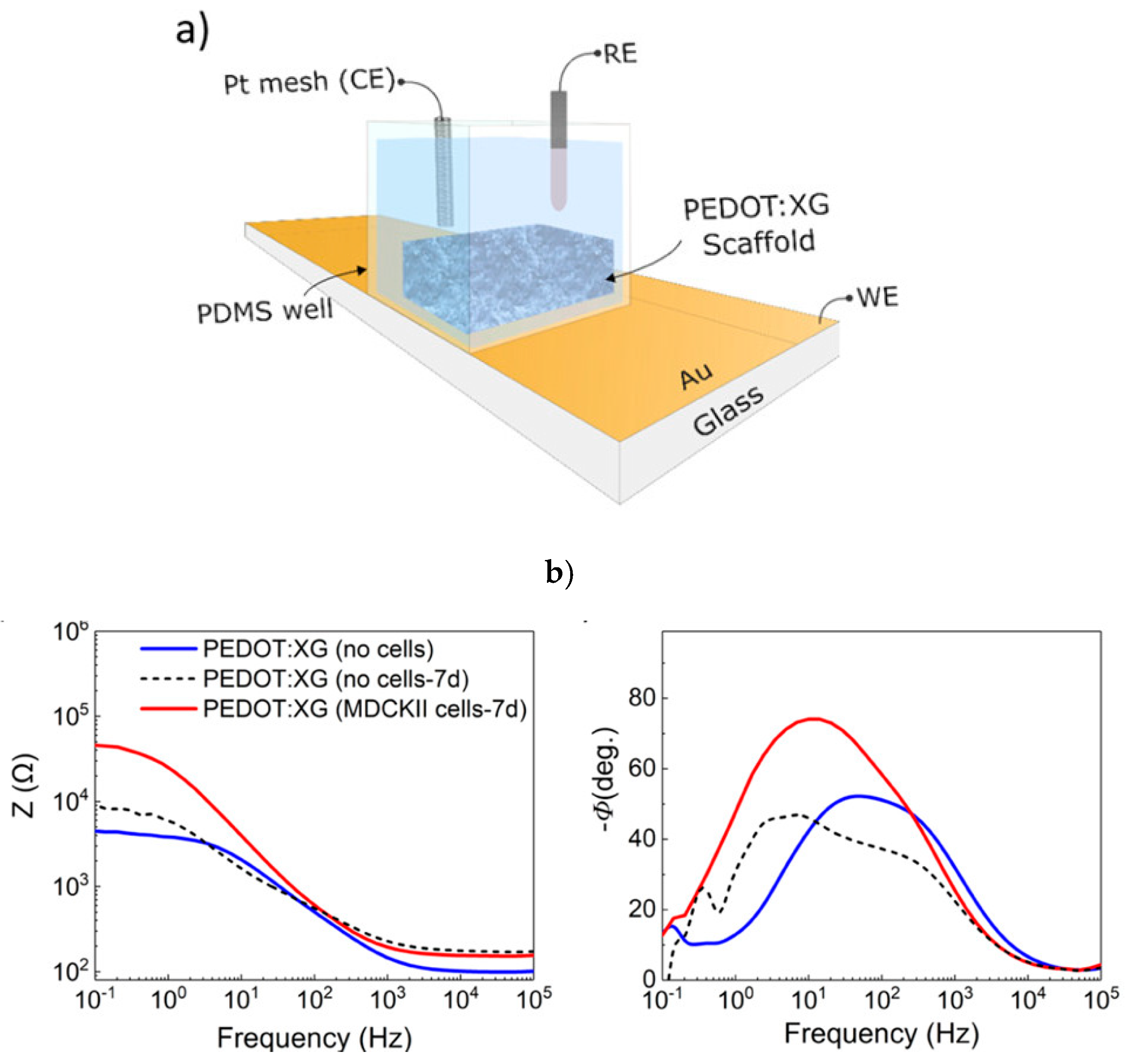
| Conductive Polymers | Manufacturing Techniques | Conductivity (Scm−1) | References |
|---|---|---|---|
| PPy or PPy | Electrospinning, freeze drying, in situ polymerization | 102–7.5 × 103 | [62,63,64,65,66,67,68] |
| PAN or PAN | Electrospinning | 30–200 | [62,69,70] |
| PEDOT or PEDOT Composites | Freeze drying, vapor-phase polymerization, solvent casting | 10–103 | [60,62,71,72] |
| Fillers | Manufacturing Techniques | Conductivity (Scm−1) | References |
|---|---|---|---|
| Carbon black nanofillers | Reversible esterification reaction and two crosslinking steps | / | [77] |
| Carbon fibers and nano fibers | Precipitation, Heat treatment + surface oxidization + PDA coating | 4 × 10−4–128, 2 × 10−2 | [83,84] |
| CNTs | Electrospinning, chemical vapor deposition and oxidative purification; Pressure-Activated Microsyringe (PAM); High pressure carbon monoxide conversion synthesis and suspension mixing. | 1 × 10−9–3.72 × 102 | [78,81,82] |
| Graphene | Electrospinning and hydrazine vapors; co-electrospinning; Electrospinning and 3D ultrasound expansion; Emulsion polymerization by chemical oxidation method; Bioplotter extrusion printing with UV cross-linking | 0.93 × 10−7–2.52 | [85,86,87,88,89,90,91] |
| Fillers | Manufacturing Techniques | Conductivity (Scm−1) | References |
|---|---|---|---|
| Gold nanofillers | Electrospinning, capillary force lithography + electron beam evaporation | 10−4–2 × 105 | [92,93,94,95] |
| Silver nanofillers | Capillary force lithography + electron beam evaporation | 4 | [97,98] |
| Platinum nanofillers | Heat sintering | / | [96] |
| Zinc nanofillers | Electrospinning | 0.01–0.08 | [99,100] |
| Application Areas | Conductive Material | Effect on Cellular Activities | References |
|---|---|---|---|
| Neural Tissue Engineering | PPy: i. PPy/PLA ii. PPy/PLGA iii. PPy/PCLF | An increased c-Fos gene expression level and enhanced nerve regeneration Formation of longer neurites by electrical stimulation of PC12 cells Enhancing the PC12 cell attachment. | [101,102,103,104] |
| PEDOT: i. PEDOT/agarose gel hydrogel ii. PEDOT/HA/PLA | Development of nerve conduits for axonal regeneration. Electrical stimulation of PC12 cells for neural out growth. | [105,106] | |
| Carbon fibers and nanofibers: Cotton/CNFs/Poly-dopamine (PDA) | Electrical stimulation accelerated cell proliferation and differentiation of nerve cells | [84] | |
| CNTs: Hydrogel containing single-walled carbon nanotubes (SWCNTs) | Schwann cell (SC) response to SWCNT is examined in both 2D and 3D microenvironments | [82] | |
| Graphene: i. Conductive polyaniline/graphene (PAG) nanoparticles into a chitosan/gelatin matrix ii. Polyaniline/graphene (PAG) nanocomposites | Electrical and mechanical properties increased depending on the PAG content Improved electrical conductivity, stabilized thermal behavior | [85,90] | |
| Au: polycaprolactone (PCL)-gelatin | Enhances neuronal proliferation with encouraged axonal elongation over forming a complex branching trees | [95] | |
| Ag: Carboxylated Cellulose Nanofibers–Dopamine/Silver Nanoparticles (CCNF-DA/AgNPs) | Improved mechanical properties, antimicrobial activity (Gram-positive and Gram-negative bacteria), and electrical conductivity | [97] | |
| Zn: i. ZnO-NPs polymer composites 50:50, 75:25, 90:10, 98:2, and 100:0 (PU:ZnO wt.%) ii. ZnNPs in poly(εcaprolactone) (PCL) matrix | Decreased astroglial cell adhesion and proliferation Promoted neuroglial cell proliferation | [99,100] | |
| Cardiac Tissue Engineering | PAN: i. PAN substrate ii. PAN/PLCL iii. PAN/CSA iv. PAN/PLGA | Supporting adhesion and proliferation of H9c2 cardiac myoblasts. Increasing cell differentiation of C2C12 myoblasts. In vitro differentiation of hMSC into cardiomyocytes Integration of cardiomyocytes. | [107,108,109] |
| PPy: i. PPy/Pt ii. PPy/PC/gelatin | Stimulating cardiac myocytes. Regeneration of infarct myocardium and cardiac defects. | [110,111] | |
| CNTs: i. CNT/PGS/gelatin ii. CNT/PLA | Supporting for improved alignment of cardiomyocyte In vitro cardiomyogenesis | [94,112] | |
| Carbon black: Liquid crystal elastomers with carbon black nanoparticles | Enhances cell attachment and viability of Cardiomyocytes cells. | [77] | |
| Carbon fibers and nanofibers: Chitosan/carbon nanofibers composite scaffold | Increasing expression of cardiac-specific genes involved in muscle contraction and electrical coupling. | [83] | |
| Au: Chitosan/Au (Thermosensitive conductive hydrogel) | Enhanced cardiomyogenic differentiation and proliferation of MSCs | [94] | |
| Bone Tissue Engineering | PPy: i. PPy/PLA | Stimulating cell growth and proliferation of ADSCs. | [65] |
| PAN: i. PAN/HAP-CS/Gel ii. PAN/PLA | Improves attachment and proliferation of dental pulp stem cells. Enhance bone regeneration | [113,114] | |
| CNTs: i. CNT/PCL ii. CNT/ HAP/PMMA | Enhanced osteogenic signal expression of rat bone-marrow-derived stroma cells. Increased mechanical strength of scaffold | [115,116] | |
| Graphene: i. Poly-caprolactone (PCL), gelatin and polyaniline/graphene(PAG) nanoparticles ii. Polyaniline solution + conductive hydrogel precursor charged with graphene nanoparticles iii. Poly(trimethylene carbonate) (PTMC) with addition of just 3 wt % graphene | Mechanical and electrical properties increased and enhances rat bone marrow-derived mesenchymal stem cells adhesion and proliferation Increased elastic modulus and electrical conductivity. Better supported human osteoblast-like cell adhesion, proliferation, and morphology comparing hydrogel alone Improved tensile strength and electrical conductivity, enhanced mesenchymal stem cell (MSC) attachment and proliferation. | [87,89,91] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, M.A.; Cantù, E.; Tonello, S.; Serpelloni, M.; Lopomo, N.F.; Sardini, E. A Review on Biomaterials for 3D Conductive Scaffolds for Stimulating and Monitoring Cellular Activities. Appl. Sci. 2019, 9, 961. https://doi.org/10.3390/app9050961
Khan MA, Cantù E, Tonello S, Serpelloni M, Lopomo NF, Sardini E. A Review on Biomaterials for 3D Conductive Scaffolds for Stimulating and Monitoring Cellular Activities. Applied Sciences. 2019; 9(5):961. https://doi.org/10.3390/app9050961
Chicago/Turabian StyleKhan, Muhammad Ahmed, Edoardo Cantù, Sarah Tonello, Mauro Serpelloni, Nicola Francesco Lopomo, and Emilio Sardini. 2019. "A Review on Biomaterials for 3D Conductive Scaffolds for Stimulating and Monitoring Cellular Activities" Applied Sciences 9, no. 5: 961. https://doi.org/10.3390/app9050961
APA StyleKhan, M. A., Cantù, E., Tonello, S., Serpelloni, M., Lopomo, N. F., & Sardini, E. (2019). A Review on Biomaterials for 3D Conductive Scaffolds for Stimulating and Monitoring Cellular Activities. Applied Sciences, 9(5), 961. https://doi.org/10.3390/app9050961








