Transient Liquid Phase Bonding of Copper Using Sn Coated Cu MWCNT Composite Powders for Power Electronics
Abstract
:1. Introduction
2. Materials and Methods
2.1. Electroless Plating of MWCNT
2.1.1. Electroless Plating of Copper on MWCNT
2.1.2. Electroless Plating of Sn on Cu-MWCNT Composite Powders
2.2. Transient Liquid Phase Joining
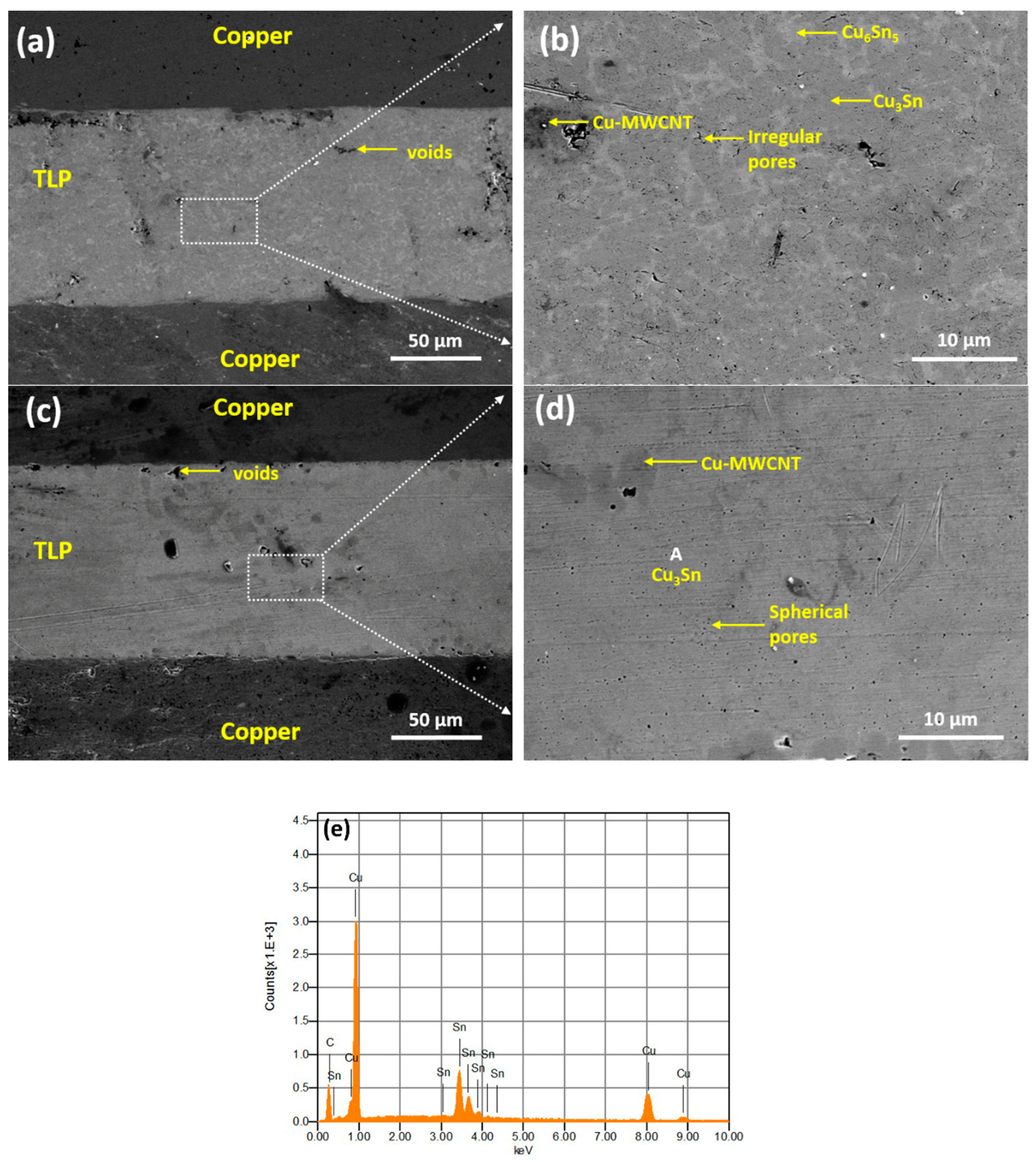
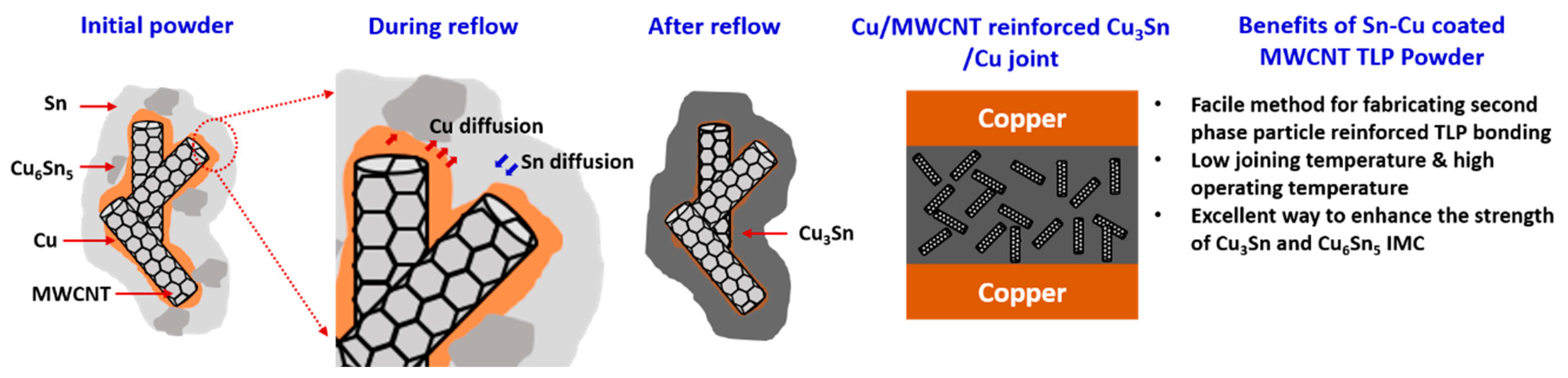
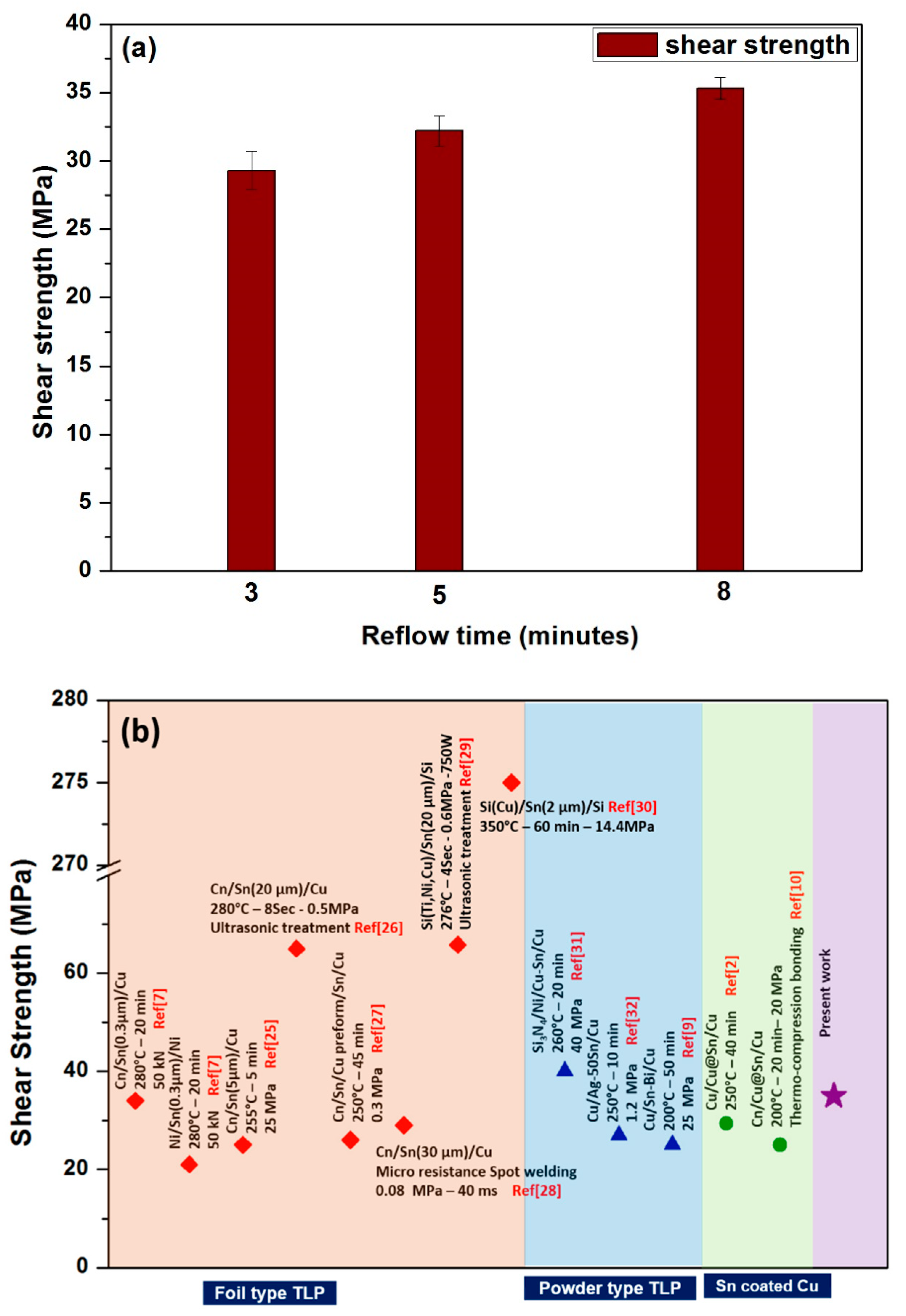
3. Results and Discussions
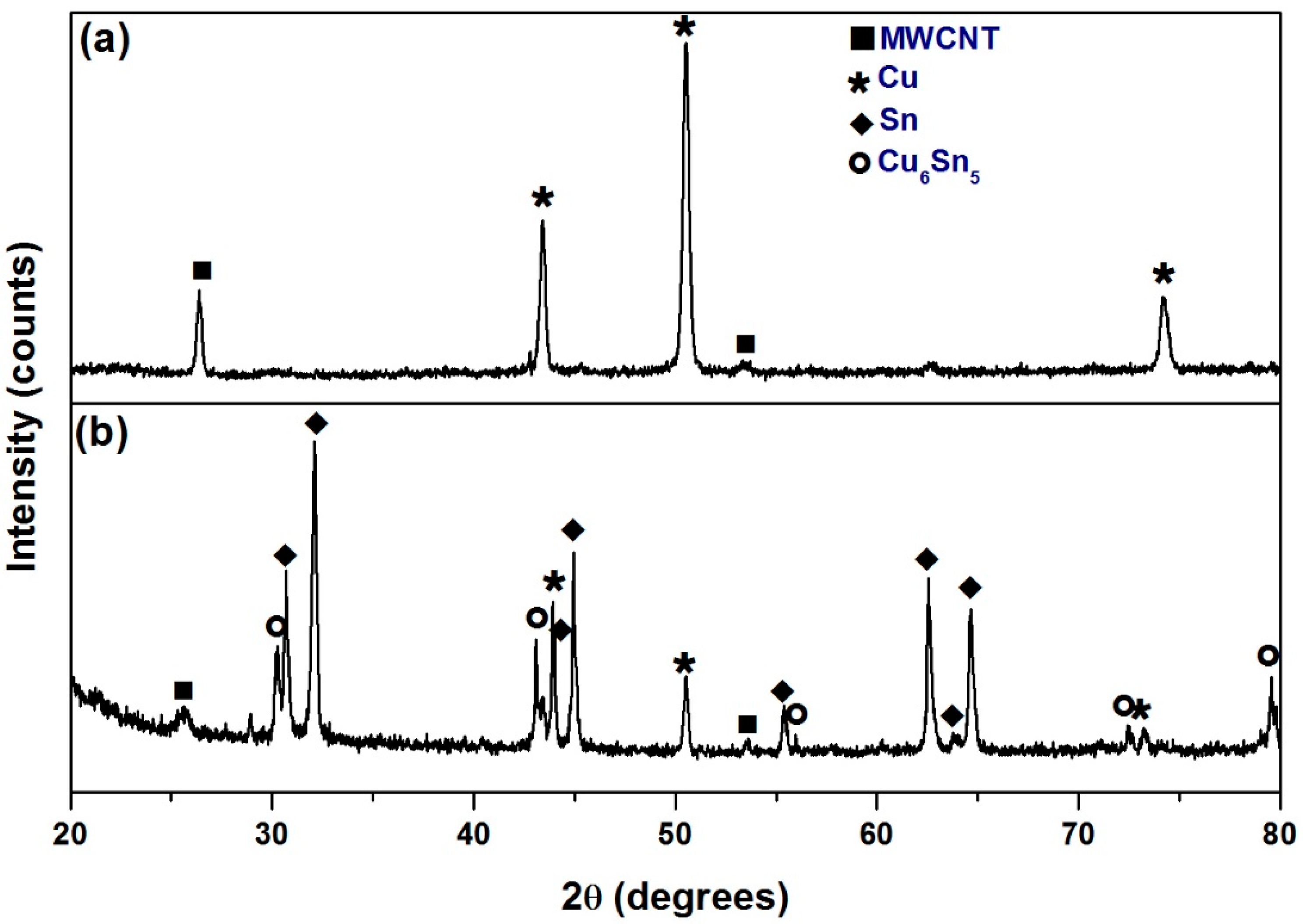
Characterization of Sn-Cu Coated MWCNT
4. Conclusions
Author Contributions
Acknowledgements
Conflicts of Interest
References
- Liu, X.; Nishikawa, H. Microstructure of Transient Liquid Phase Sintering Joint by Sn-Coated Cu Particles for High Temperature Packaging. In Proceedings of the IMAPS 2015 Orlando—48th Annual International Symposium on Microelectronics, Orlando, FL, USA, 26–29 October 2015; pp. 000449–000452. [Google Scholar]
- Hu, T.; Chen, H.; Li, M. Die attach materials with high remelting temperatures created by bonding Cu@Sn micro particles at lower temperatures. Mater. Des. 2016, 108, 383–390. [Google Scholar] [CrossRef]
- Fan, J.; Tan, C.S. Low temperature wafer-level metal thermos-compression bonding technology for 3D integration. In Metallurgy: Advances in Materials and Processes; IntechOpen Ltd.: London, UK, 2012; pp. 71–94. [Google Scholar]
- Chidambaram, V.; Hattel, J.; Hald, J. High-temperature lead free solder alternatives. Microelectron. Eng. 2011, 88, 981–989. [Google Scholar] [CrossRef]
- Yan, J.F.; Zou, G.S.; Wu, A.P. Pressure less bonding process using Ag nanoparticles paste for flexible electronics packaging. Scr. Mater. 2012, 66, 582–585. [Google Scholar] [CrossRef]
- Li, Z.; Li, M.; Xiao, Y.; Wang, C. Ultra rapid formation of homogeneous Cu6Sn5 and Cu3Sn intermetallic compound joints at room temperature using ultrasonic waves. Ultrason Sonochem. 2014, 21, 924–929. [Google Scholar] [CrossRef] [PubMed]
- Chu, K.; Sohn, Y.; Moon, C. A comparative study of Cn/Sn/Cu and Ni/Sn/Ni solder joints for low temperature stable transient liquid phase bonding. Scr. Mater. 2015, 109, 113–117. [Google Scholar] [CrossRef]
- Li, J.F.; Agyakwa, J.F.; Johnson, C.M. Kinetics of Ag3Sn growth in Ag–Sn–Ag system during transient liquid phase soldering process. Acta Mater. 2010, 58, 3429–3443. [Google Scholar] [CrossRef]
- Mokhtari, O.; Nishikawa, H. The shear strength of transient liquid phase bonded Sn–Bi solder joint with added Cu particles. Adv. Powder Technol. 2016, 27, 1000–1005. [Google Scholar] [CrossRef]
- Liu, X.; He, S.; Nishikawa, H. Low temperature solid-state bonding using Sn-coated Cu particles for high temperature die attach. J. Alloys Compd. 2017, 695, 2165–2172. [Google Scholar] [CrossRef]
- Aryasomayajula, L.; Wolter, K.J. Carbon nanotube composites for electronic packaging applications: A review. J. Nanotechnol. 2013, 2013, 296517. [Google Scholar] [CrossRef]
- Bal, S.; Samal, S.S. Carbon nanotube reinforced polymer composites—A state of the art. Bull. Mater. Sci. 2007, 30, 379–386. [Google Scholar] [CrossRef]
- Nai, S.M.L.; Wei, J.; Gupta, M. Effect of carbon nanotubes on the shear strength and electrical resistivity of a lead-free solder. J. Electron. Mater. 2008, 37, 515–522. [Google Scholar] [CrossRef]
- Kreupl, F.; Graham, A.P.; Liebau, M.; Duesberg, G.S.; Seidel, R.; Unger, E. Carbon nanotubes for interconnect applications. In Proceedings of the IEEE International Electron Devices Meeting (IEDM ’04), San Francisco, CA, USA, 13–15 December 2004; pp. 683–686. [Google Scholar]
- Hu, T.; Chen, H.T.; Wang, C.Q.; Huang, M.S.; Zhao, F.F. Study of electroless Sn coated Cu microparticles and their application as a high temperature thermal interface material. Surf. Coat. Technol. 2017, 319, 230–240. [Google Scholar] [CrossRef]
- Mookam, N.; Tunthawiroon, P.; Kanlayasiri, K. Effects of Copper content in Sn based solder on the intermetallic phase formation and growth during soldering. IOP Conf. Ser. Mater. Sci. Eng. 2018, 361, 012008. [Google Scholar] [CrossRef]
- Yao, P.; Li, X.; Liang, X.; Yu, B. Investigation of soldering process and interfacial microstructure evolution for the formation of full Cu3Sn joints in electronic packaging. Mater. Sci. Semicond. Process. 2017, 58, 39–50. [Google Scholar] [CrossRef]
- Kumar, S.; Handwerker, C.A.; Dayananda, M.A. Intrinsic and interdiffusion in Cu–Sn system. J. Phase Equilib. Diff. 2011, 32, 309–319. [Google Scholar] [CrossRef]
- Chuang, H.Y.; Yu, J.J.; Kuo, M.S.; Tong, H.M.; Kao, C.R. Elimination of voids in reactions between Ni and Sn: a novel effect of solver. Scr. Mater. 2012, 66, 171–174. [Google Scholar] [CrossRef]
- Bosco, N.S.; Zok, F.W. Critical interlayer thickness for transient liquid phase bonding in the Cu-Sn system. Acta Mater. 2004, 52, 2965–2972. [Google Scholar] [CrossRef]
- Gao, F.; Qiu, J.M. calculating the diffusivity of Cu and Sn in Cu3Sn Intermetallic by molecular dynamic simulation. Mater Lett. 2012, 73, 92–94. [Google Scholar] [CrossRef]
- Bao, Y.; Wu, A.; Shao, H.; Zhao, Y.; Zou, G. Effect of powders on microstructures and mechanical properties for Sn–Ag transient liquid phase bonding in air. J. Mater. Sci. Mater. Electron. 2018, 29, 10246–10257. [Google Scholar] [CrossRef]
- Lee, B.S.; Hyun, S.K.; Yoon, J.W. Cu-Sn and Ni-Sn transient liquid phase bonding for die attach technology applications in high temperature power electronics packaging. J. Mater. Sci. Mater. Electron. 2017, 28, 7827–7833. [Google Scholar] [CrossRef]
- Chiu, W.L.; Liu, C.M.; Haung, Y.S.; Chen, C. formation of plate like channels in Cu6Sn5 and Cu3Sn Intermetallic compounds during transient liquid reaction of Cu/Sn/Cu structures. Mater. Lett. 2016, 164, 5–8. [Google Scholar] [CrossRef]
- Brincker, M.; Sohl, S.; Eisele, R.; Popok, V.N. Strength and reliability of low temperature transient liquid phase bonded Cu-Sn-Cu interconnects. Microelectron. Reliab. 2017, 76–77, 378–382. [Google Scholar] [CrossRef]
- Zhou, H.Y.; Liu, J.H.; Li, Z.L.; Song, X.G.; Zhao, Y.X.; Niu, H.W.; Tian, H.; Dong, H.J.; Feng, J.C. A Comparative Study on the Microstructure and Mechanical Properties of Cu6Sn5 and Cu3Sn Joints Formed by TLP Soldering With/Without the Assistance of Ultrasonic Waves. Metall. Mater. Trans. A 2018, 49, 2739–2749. [Google Scholar] [CrossRef]
- Shao, H.; Wu, A.; Bao, Y.; Zhao, Y.; Zou, G.; Liu, L. Novel transient liquid phase bonding through capillary action for high temperature power devices packaging. Mater. Sci. Eng. A 2018, 724, 231–238. [Google Scholar] [CrossRef]
- Liu, B.; Tian, Y.; Wang, C.; An, R.; Liu, Y. Extremely fast formation of Cu3Sn intermetallic compounds in Cu/Sn/Cu system via a micro-resistance spot welding process. J. Alloys Compd. 2016, 687, 667–673. [Google Scholar] [CrossRef]
- Li, M.; Li, Z.; Xiao, Y.; Wang, C. Rapid formation of Cu/Cu3Sn/Cu joints using ultrasonic bonding processes at ambient temperature. Appl. Phys. Lett. 2013, 102, 094104. [Google Scholar] [CrossRef]
- Rautiainan, A.; Xu, H.; Österlund, E.; Li, J.; Vuorinen, V.; Krockel, M.P. Microstructural characterization and mechanical performance of Wafer-Level SLID bonded Au-Sn and Cu-Sn seal rings for MEMS encapsulation. J. Electron. Mater. 2015, 44, 4533–4548. [Google Scholar] [CrossRef]
- Lang, F.; Yamaguchi, H.; Nakagawa, H.; Sato, H. Thermally Stable Bonding of SiC Devices with Ceramic Substrates: Transient Liquid Phase Sintering Using Cu/Sn Powders. J. Electrochem. Soc. 2013, 160, 315–319. [Google Scholar] [CrossRef]
- Sharif, A.; Gan, C.L.; Chen, Z. Transient liquid phase Ag-based solder technology for high-temperature packaging applications. J. Alloys Compd. 2014, 587, 365–368. [Google Scholar] [CrossRef]
- Xia, Z.; Riester, L.; Curtin, W.A.; Li, H.; Sheldon, B.W.; Liang, J.; Chang, B.; Xu, J.M. Direct observation of toughening mechanisms in carbon nano tube ceramic matrix composites. Acta Mater. 2004, 52, 931–944. [Google Scholar] [CrossRef]
- Han, Y.D.; Jing, H.Y.; Nai, S.M.L.; Xu, L.Y.; Tan, C.M.; Wei, J. Interfacial reaction and shear strength of Ni-coated carbon nano tubes reinforced Sn-Ag-Cu solder joints during thermal cycling. Intermetallics 2012, 31, 72–78. [Google Scholar] [CrossRef]
- Lee, H.T.; Chen, M.H.; Jao, H.M.; Liao, L.T. Influence of interfacial intermetallic compound on fracture behavior of solder joints. Mater. Sci. Eng. A 2003, 358, 134–141. [Google Scholar] [CrossRef]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rajendran, S.H.; Do Hyun, J.; Wook Sang, J.; Jae Pil, J. Transient Liquid Phase Bonding of Copper Using Sn Coated Cu MWCNT Composite Powders for Power Electronics. Appl. Sci. 2019, 9, 529. https://doi.org/10.3390/app9030529
Rajendran SH, Do Hyun J, Wook Sang J, Jae Pil J. Transient Liquid Phase Bonding of Copper Using Sn Coated Cu MWCNT Composite Powders for Power Electronics. Applied Sciences. 2019; 9(3):529. https://doi.org/10.3390/app9030529
Chicago/Turabian StyleRajendran, Sri Harini, Jung Do Hyun, Jeon Wook Sang, and Jung Jae Pil. 2019. "Transient Liquid Phase Bonding of Copper Using Sn Coated Cu MWCNT Composite Powders for Power Electronics" Applied Sciences 9, no. 3: 529. https://doi.org/10.3390/app9030529
APA StyleRajendran, S. H., Do Hyun, J., Wook Sang, J., & Jae Pil, J. (2019). Transient Liquid Phase Bonding of Copper Using Sn Coated Cu MWCNT Composite Powders for Power Electronics. Applied Sciences, 9(3), 529. https://doi.org/10.3390/app9030529




