Smartphone-Based Point-of-Care Cholesterol Blood Test Performance Evaluation Compared with a Clinical Diagnostic Laboratory Method
Abstract
Featured Application
Abstract
1. Introduction
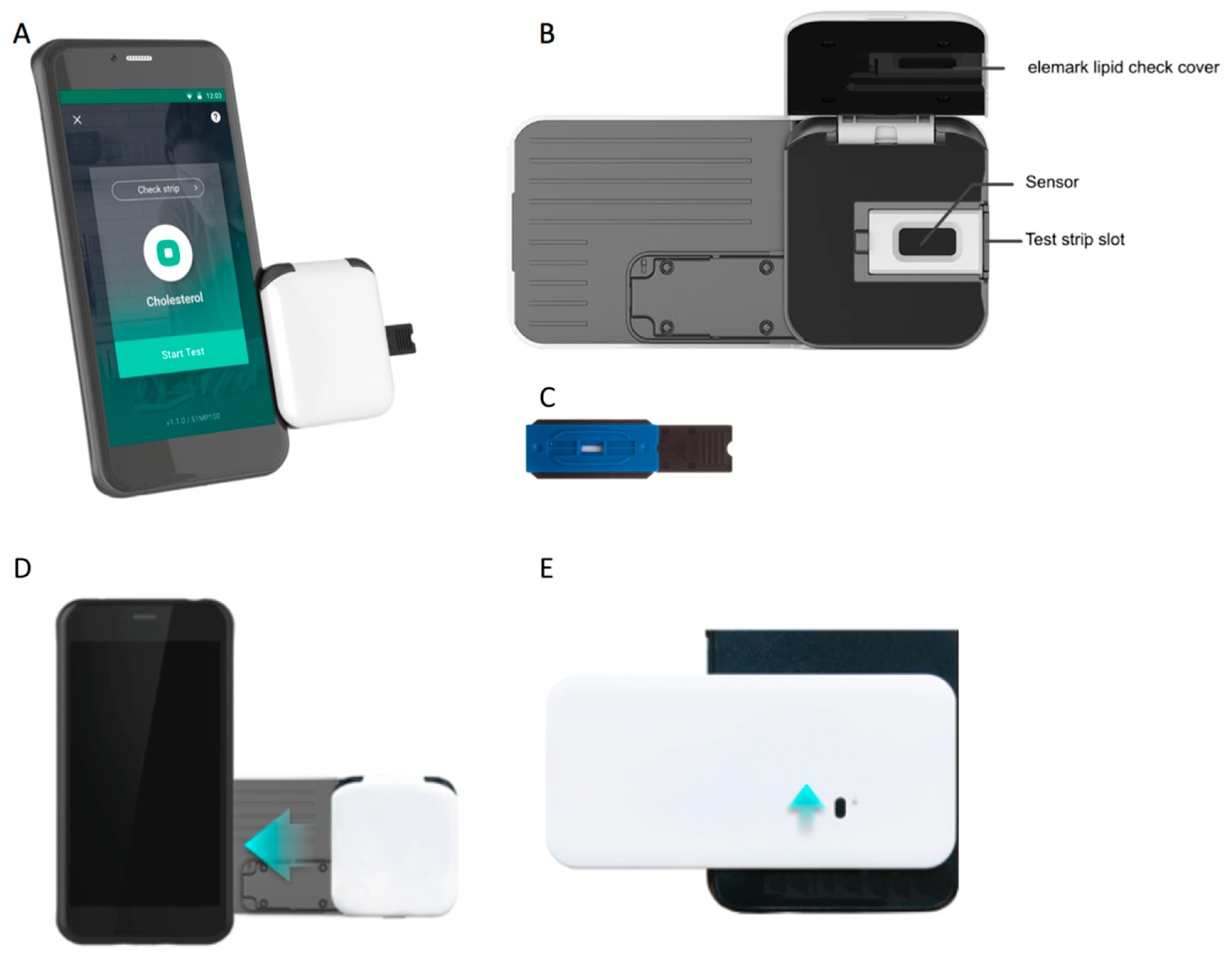
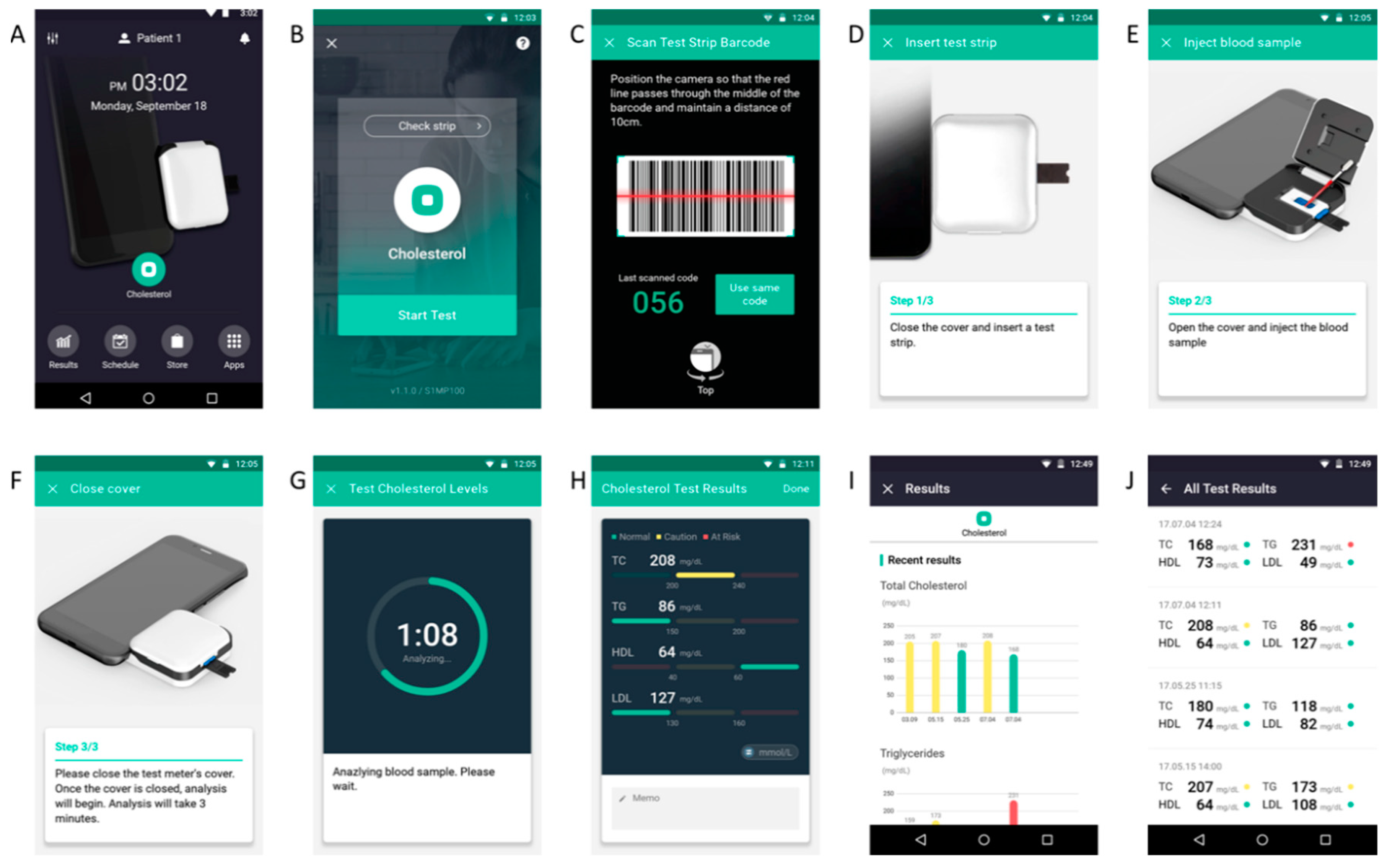
2. Materials and Methods
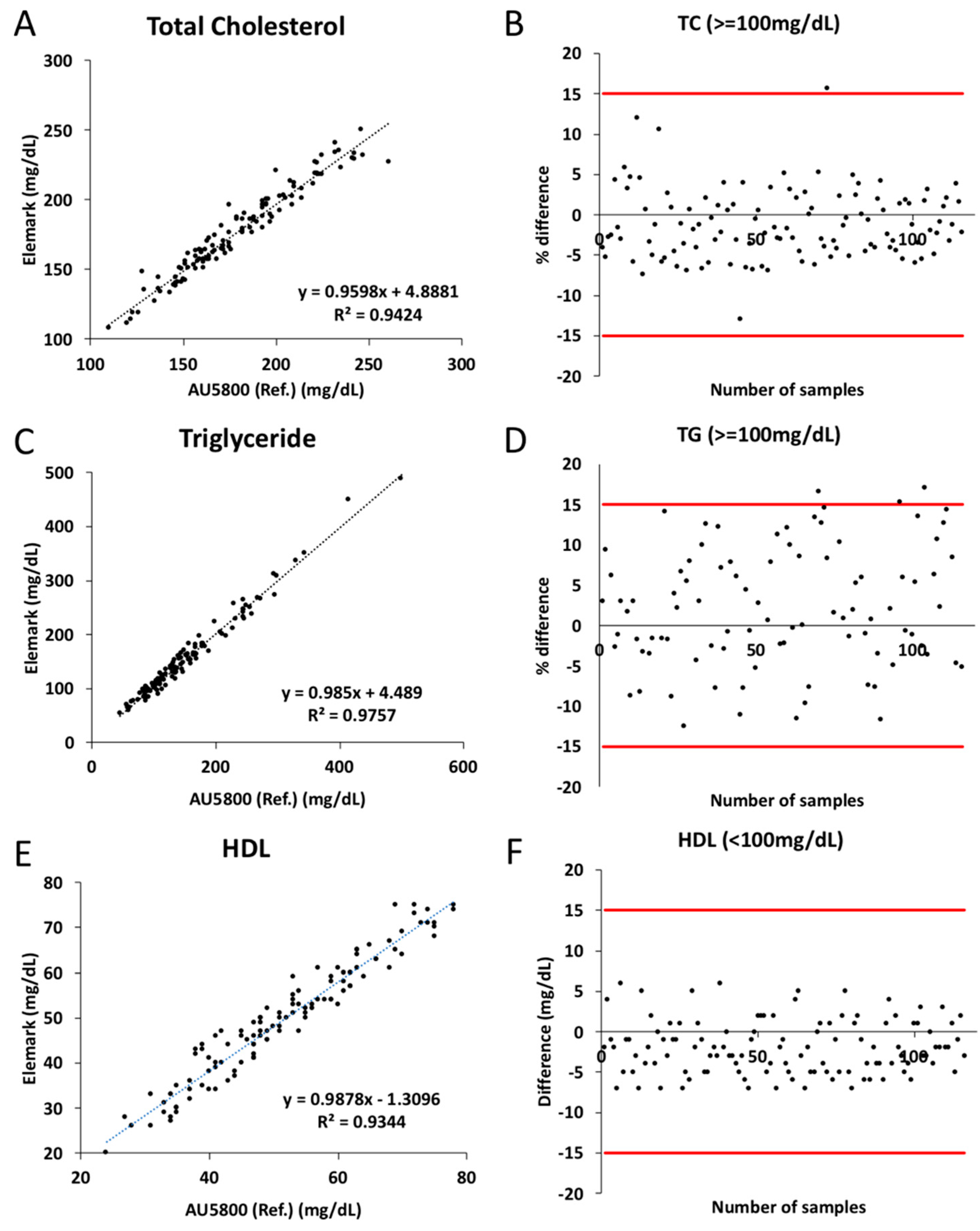
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- New, M.I.; Roberts, T.N.; Bierman, E.L.; Reader, G.G. The Significance of Blood Lipid Alterations in Diabetes Mellitus. Diabetes 1963, 12, 208–212. [Google Scholar] [CrossRef] [PubMed]
- Frantz, I.D.; Dawson, E.A.; Ashman, P.L.; Gatewood, L.C.; Bartsch, G.E.; Kuba, K.; Brewer, E.R. Test of effect of lipid lowering by diet on cardiovascular risk. The Minnesota Coronary Survey. Arterioscler. Thromb. Vasc. Biol. 1989, 9, 129–135. [Google Scholar]
- Ravelli, A.; van der Meulen, J.H.P.; Osmond, C.; Barker, D.; Bleker, O. Infant feeding and adult glucose tolerance, lipid profile, blood pressure, and obesity. Arch. Dis. Child. 2000, 82, 248–252. [Google Scholar] [CrossRef] [PubMed]
- Pai, N.P.; Vadnais, C.; Denkinger, C.; Engel, N.; Pai, M. Point-of-Care Testing for Infectious Diseases: Diversity, Complexity, and Barriers in Low- And Middle-Income Countries. PLoS Med. 2012, 9, e1001306. [Google Scholar] [CrossRef] [PubMed]
- Oncescu, V.; Mancuso, M.; Erickson, D. Cholesterol testing on a smartphone. Lab Chip 2014, 14, 759–763. [Google Scholar] [CrossRef] [PubMed]
- Kroll, M.H.; Biswas, B.; Budd, J.R.; Durham, P.; Gorman, R.T.; Gwise, T.E.; Halim, A.; Hatjimihail, A.T.; Hilden, J.; Kyunghee, S. Assessment of the diagnostic accuracy of laboratory tests using receiver operating characteristic curves; approved guideline. Clin. Lab. Stand. Inst. Doc. 2011, 31, 1–45. [Google Scholar]
- International Organization for Standardization. In Vitro Diagnostic Test Systems: Requirements for Blood-glucose Monitoring Systems for Self-testing in Managing Diabetes Mellitus; ISO: Geneva, Switzerland, 2003. [Google Scholar]
- 45 CFR 46. Available online: https://www.hhs.gov/ohrp/regulations-and-policy/regulations/45-cfr-46/index.html (accessed on 26 March 2018).
- Hoaglin, D.C.; Iglewicz, B.; Tukey, J.W. Performance of some resistant rules for outlier labeling. J. Am. Stat. Assoc. 1986, 81, 991–999. [Google Scholar] [CrossRef]
- Hoaglin, D.C.; John, W. Tukey and data analysis. Stat. Sci. 2003, 18, 311–318. [Google Scholar]
- Shephard, M.D.; Mazzachi, B.C.; Shephard, A.K. Comparative performance of two point-of-care analysers for lipid testing. Clin. Lab. 2007, 53, 561–566. [Google Scholar] [PubMed]
- Erickson, D.; O’Dell, D.; Jiang, L.; Oncescu, V.; Gumus, A.; Lee, S.; Mancuso, M.; Mehta, S. Smartphone technology can be transformative to the deployment of lab-on-chip diagnostics. Lab Chip 2014, 14, 3159–3164. [Google Scholar] [CrossRef] [PubMed]
- Roda, A.; Calabretta, M.M.; Calabria, D.; Caliceti, C.; Cevenini, L.; Lopreside, A.; Zangheri, M. Smartphone-Based Biosensors for Bioanalytics: A Critical Review. In Past, Present and Future Challenges of Biosensors and Bioanalytical Tools in Analytical Chemistry: A Tribute to Professor Marco Mascini, Volume 77; Elsevier Science Ltd.: Oxford, UK, 2017; Chapter 8; p. 237. [Google Scholar]
- Haggerty, L.; Tran, D. Cholesterol point-of-care testing for community pharmacies: A review of the current literature. J. Pharm. Pract. 2017, 30, 451–458. [Google Scholar] [CrossRef] [PubMed]
- El-Osta, A.; Woringer, M.; Pizzo, E.; Verhoef, T.; Dickie, C.; Ni, M.Z.; Huddy, J.R.; Soljak, M.; Hanna, G.B.; Majeed, A. Does use of point-of-care testing improve cost-effectiveness of the NHS Health Check programme in the primary care setting? A cost-minimisation analysis. BMJ Open 2017, 7, e015494. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.; Yun, K.; Hwang, J.-H.; Chae, J.-H. Classification of suicide attempts through a Machine learning algorithm Based on Multiple systemic Psychiatric scales. Front. Psychiatry 2017, 8, 192. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.; Yun, K.; Maoz, U.; Kim, T.-S.; Chae, J.-H. Identifying Depression in the National Health and Nutrition Examination Survey Data using a Deep Learning Algorithm. J. Affect. Disord. 2019. [Google Scholar] [CrossRef] [PubMed]
- Kupke, I.R.; Zeugner, S.; Gottschalk, A.; Kather, B. Differences in lipid and lipoprotein concentrations of capillary and venous blood samples. Clin. Chim. Acta 1979, 97, 279–283. [Google Scholar] [CrossRef]
| TC Concentration (mg/dL) | N | Total | % |
| Below 200 mg/dL (Normal) | 86 | 116 | 74.1 |
| 200~239 mg/dL (Borderline High) | 24 | 116 | 20.7 |
| Above 240 mg/dL (High) | 6 | 116 | 5.2 |
| TG concentration (mg/dL) | N | Total | % |
| Below 150 mg/dL (Normal) | 71 | 116 | 61.2 |
| 150~199 mg/dL (Borderline High) | 22 | 116 | 19.0 |
| 200~499 mg/dL (High) | 22 | 116 | 19.0 |
| Above 500 mg/dL (Very high) | 1 | 116 | 0.9 |
| HDL concentration (mg/dL) | N | Total | % |
| Below 40 mg/dL (Low) | 22 | 116 | 19.0 |
| 40~59 mg/dL (Normal) | 60 | 116 | 51.7 |
| Above 60 mg/dL (High) | 34 | 116 | 29.3 |
| <100 mg/dL | ≥100 mg/dL | ||||||
|---|---|---|---|---|---|---|---|
| Within ±5 mg/dL | Within ±10 mg/dL | Within ±15 mg/dL | Within ±5% | Within ±10% | Within ±15% | Within ±5% | |
| TC | N/A (Device range: 100~450 mg/dL) | 87/116 | 112/116 | 115/116 | 116/116 | ||
| 75.00% | 96.60% | 99.10% | 100% | ||||
| TG | 8/24 | 17/24 | 23/24 | 42/92 | 71/92 | 89/92 | 92/92 |
| 33.30% | 70.80% | 95.80% | 45.70% | 77.20% | 96.70% | 100% | |
| HDL | 98/116 | 116/116 | 116/116 | N/A (device range: 25~95 mg/dL) | |||
| 84.50% | 100% | 100% | |||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yun, K.; Choi, J.; Song, I.-U.; Chung, Y.-A. Smartphone-Based Point-of-Care Cholesterol Blood Test Performance Evaluation Compared with a Clinical Diagnostic Laboratory Method. Appl. Sci. 2019, 9, 3334. https://doi.org/10.3390/app9163334
Yun K, Choi J, Song I-U, Chung Y-A. Smartphone-Based Point-of-Care Cholesterol Blood Test Performance Evaluation Compared with a Clinical Diagnostic Laboratory Method. Applied Sciences. 2019; 9(16):3334. https://doi.org/10.3390/app9163334
Chicago/Turabian StyleYun, Kyongsik, Jaekyu Choi, In-Uk Song, and Yong-An Chung. 2019. "Smartphone-Based Point-of-Care Cholesterol Blood Test Performance Evaluation Compared with a Clinical Diagnostic Laboratory Method" Applied Sciences 9, no. 16: 3334. https://doi.org/10.3390/app9163334
APA StyleYun, K., Choi, J., Song, I.-U., & Chung, Y.-A. (2019). Smartphone-Based Point-of-Care Cholesterol Blood Test Performance Evaluation Compared with a Clinical Diagnostic Laboratory Method. Applied Sciences, 9(16), 3334. https://doi.org/10.3390/app9163334






