1. Introduction
According to statistics from the American Association of Oral and Maxillofacial Surgeons, approximately 70% of adults between the ages of 35 and 44 can lose at least one permanent tooth due to trauma, periodontal, or endodontic complications [
1]. Approximately 5 million dental implants are being placed every year in the United States of America as per the American Dental Association, with a elevated rate of success (<90%), with low risk and/or complication [
2]. Currently, dental implants are used as an alternative in rehabilitative treatments with a good degree of predictability. Several clinical studies have shown good results in treatments in long-term follow-up patients of unitary, partial, or totally edentulous areas [
3,
4,
5]. Despite this, several companies and research centers have invested in the improvement of implants mainly seeking to reduce the time and/or improve the healing of the bone tissue around the implanted surface. However, many events involved in the osseointegration process have not yet been completely elucidated.
Several investigations to improve or accelerate the process of osseointegration have been studied, as well worked on to elaborate new treatments for the surface of the implants (micro topography) with different physical and chemical characteristics [
5,
6,
7,
8,
9]. These modifications have shown good results, mainly in pre-clinical studies, as reported in the literature [
10,
11,
12,
13].
The surgical technique used to elaborate the osteotomy and the macrogeometry of the implant is also a factor considered of great importance in the process of osseointegration. Several models with different macrogeometries and surface treatment have been proposed and are commercialized [
14,
15,
16], with each design following its specific recommendations as to the type of bone where it should be used and the specific surgical technique for its installation [
17]. Conventionally, osteotomy is performed with the last drill having a smaller diameter in relation to the implant diameter, so that it is inserted with a high degree of torque. Obviously, the more sub-dimensioned the bed receiving the implant is, the greater the insertion torque. However, it is speculated that high levels of torque can cause a high compression in the bone tissue, which can lead to extensive bone remodeling over time [
18]. Several other studies have shown that depending on the insertion torque of the implant and whether it is beyond the physiological tolerance limit, it may present microfractures or osteonecrosis by compression [
19,
20,
21].
Recently, studies have proposed that approaching the diameter of the drilling (during the osteotomy) with the diameter of the implant that will be inserted into the bone, can facilitate and improve osseointegration [
22,
23]. This fact was demonstrated by Jimbo and collaborators in a study using a dog animal model, where, in the implants placed with high torque, the samples presented a certain amount of necrotic bone inside the implant threads, whereas in the samples where a larger drilling was used, the samples presented a substantial formation of new bone [
23]. In this case, the free space created inside the implant threads, resulting from the drill-implant diameter ratio, is called the healing chambers (
Figure 1).
Within the consistence of these concepts of the “no bone compression” during the implant installation in the bone tissue, a new implant design with decompression chambers in the threads to improve and accelerate the osseointegration process, was analyzed and compared with a conventional implant macrogeometry, both implants models with and without surface treatment. Histological and biomechanical analyses were performed using the rabbit tibia experimental model. The hypothesis was that the chambers created in the threads can promote decompression of the bone during introduction in the osteotomy and, then, a positive effect on the osseointegration.
2. Materials and Methods
Implants and groups formation: Eighty conical implants were prepared in pure titanium grade IV (Implacil De Bortoli Ltd.a, São Paulo, Brazil) which were 9 mm in length and 4 mm in diameter. The macrogeometry of the implants used presented the traditional design and threads configuration (
Figure 2a) and, the new macrogeometry implant with the presence of decompression chambers in the threads design (
Figure 2b).
Based on the initial proposed hypothesis, both implant macrogeometries were prepared with and without (machined only) surface treatment. The surface treatment used was performed by blasting with microparticles (~100 µm) of titanium oxide and followed by application of maleic acid, showing a roughness with Ra = 0.56 ± 0.10 µm [
8].
Figure 3 shows the scanning electronic microscopy (SEM) of the two surfaces used for the comparison.
Then, the implants were divided into four groups in accordance with the macrogeometry and the surface condition (with or without treatment), as the following: Group 1 (G1), traditional conical implants with surface treatment; group 2 (G2), traditional conical implants without surface treatment (machined surface); group 3 (G3), new conical implant design with surface treatment; group 4 (G4), new conical implant design without surface treatment. All implants were subjected to washing, decontamination, sterilization, and packaging in accordance with the requirements for commercialization of these materials.
Animal procedures: Twenty New Zealand white rabbits, weighing 4 ± 0.5 kg, were used for the present experimental study. The animals received the standards care and management applied in the previous studies performed and described by our research group [
10,
11]. The international guidelines of animal studies were applied. The study was approved by the Animal Experimentation Committee (Number 02-17UnRV), University of Rio Verde (Rio Verde, Brazil). A total of eighty implants (
n = 20 per group) were installed in both tibias (
n = 2 per tibia). The implants distribution was made by the randomization program (
www.randomization.com). Initially, the animals were anesthetized using a combination of 0.35 mg/kg of ketamine (Ketamina Agener
®; Agener União Ltd.a., São Paulo, Brazil) and 0.5 mg/kg of xylazine (Rompum
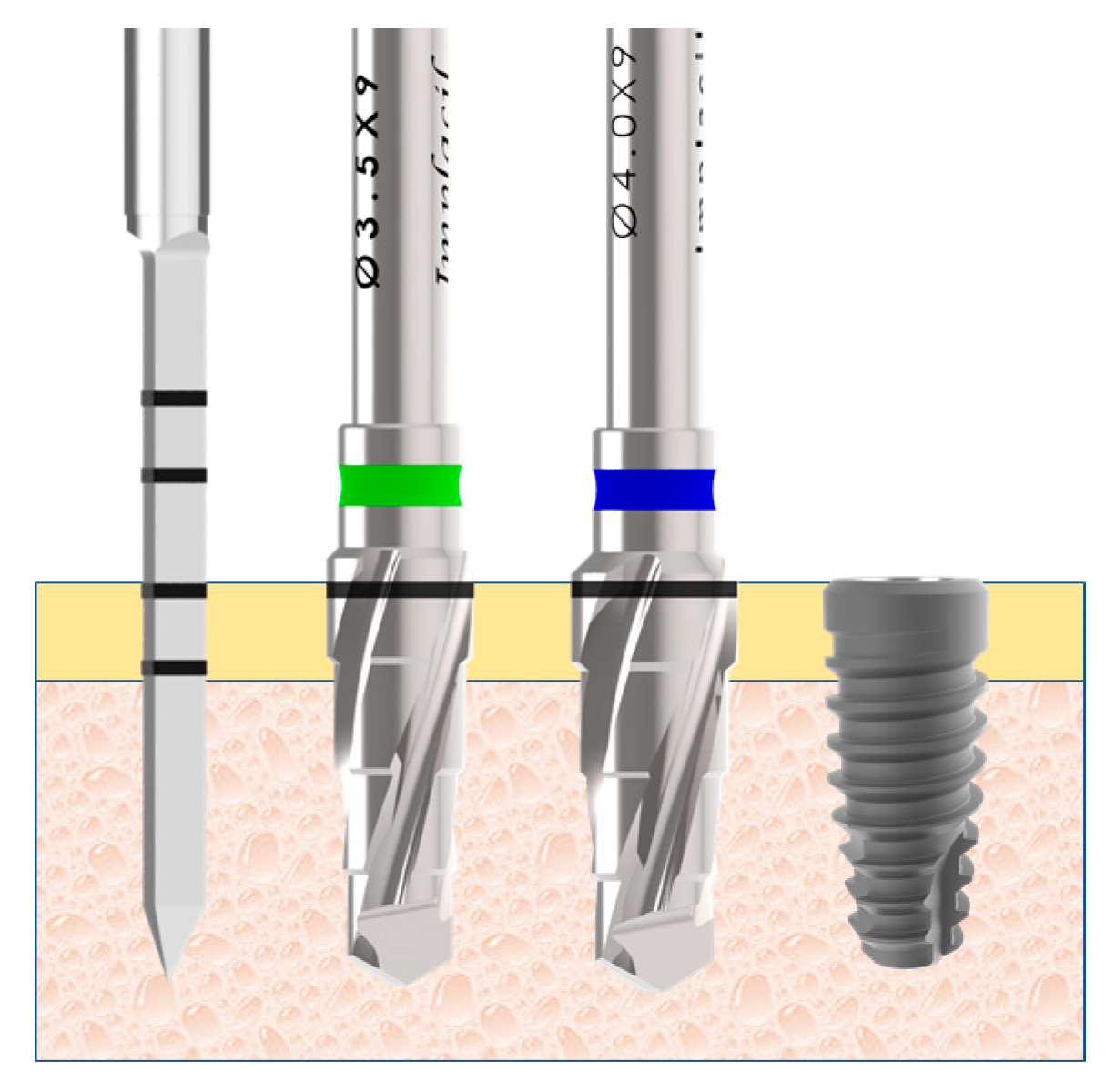
® Bayer S.A., São Paulo, Brazil), with intramuscular application. Both tibias were scraped of hairs and cleansed with antiseptic solutions before the surgical procedures to avoid a contamination. Then, an incision was performed initiating ~10 mm from the knee at distal direction with a length of ~30 mm. The bone tissue was exposed and the osteotomy to install the implant was performed using a predeterminate drilling sequence propria of the implant system (
Figure 4), under intense irrigation with saline solution.
The implant introduction in the bone site was made with manual technique, ending with a torque of ~20 N. Distances of 10 mm between the implants and from the knee articulation were maintained. Finally, a simple point suture was performed using an Ethicon nylon 4-0 (Johnson & Johnson Medical, New Brunswick, NJ, USA). After the surgeries, all medication was administered intramuscularly as follows: A single dose of 0.1 ml/kg of Benzetacil (Bayer, São Paulo, Brazil); three doses (one per day) of 3 mg/kg of ketoprofen (Ketoflex, Mundo Animal, São Paulo, Brazil). Euthanization was performed using an overdose of anesthesia two times after the implantations, at 15 and 30 days. All tibias with the implants (
Figure 5) were removed and immediately immersed in a 4% formaldehyde solution.
Implant stability measurement: The stability of all implants was measured using the Osstell (Osstell AB, Gothenburg, Sweden) device. A smartpeg sensor was installed in each implant and a controlled torque of 10 Ncm was applied, as recommended by a recent previous study [
24]. Measurements were performed in two directions (
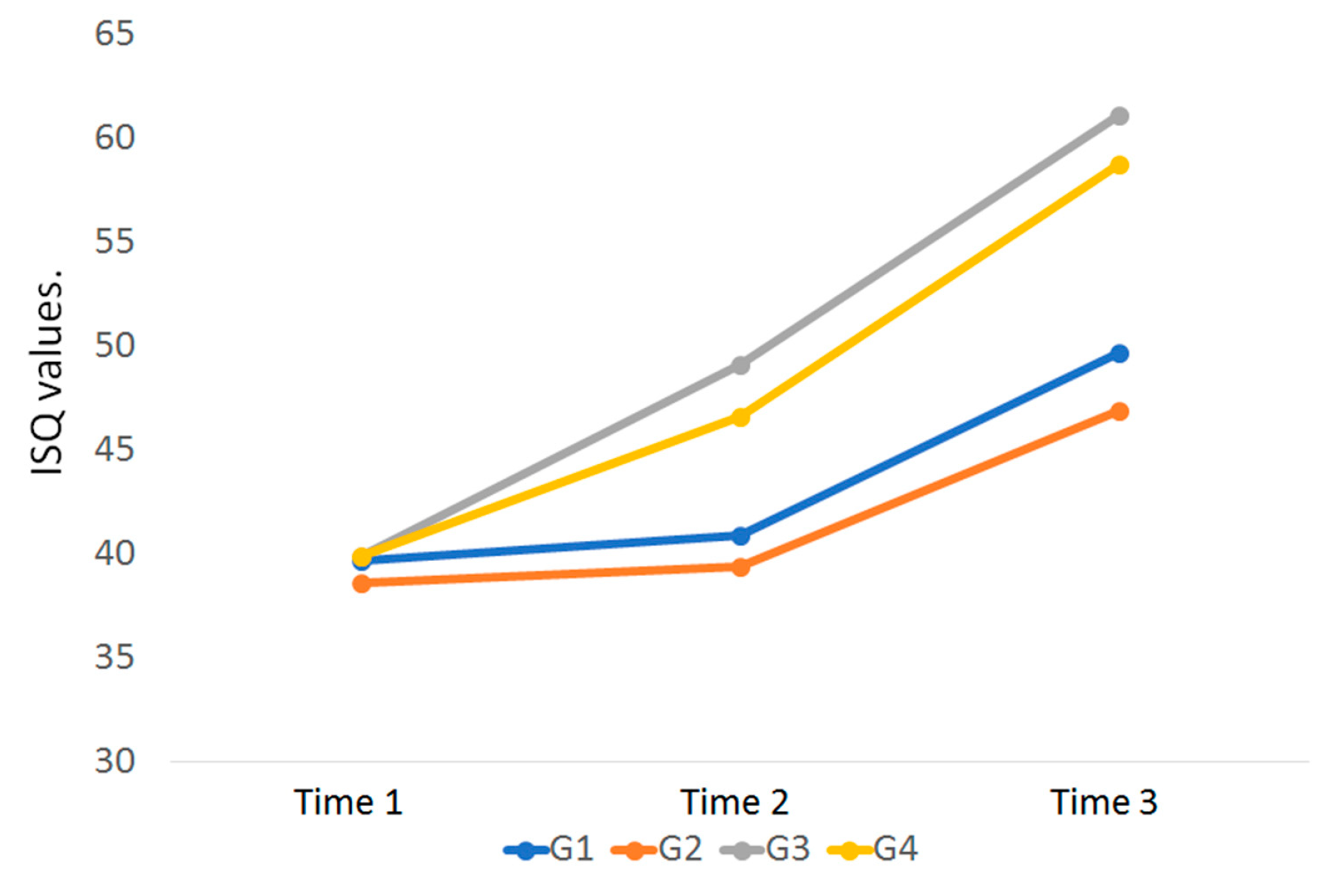
Figure 6): Proximo-distal and antero-posterior; and, a mean was made for each implant sample. Analysis was conducted three times: Immediately after the implant installation (Time 1), in the first animal lot euthanized after 15 days (Time 2) and, in the second animal lot euthanized after 30 days (Time 3).
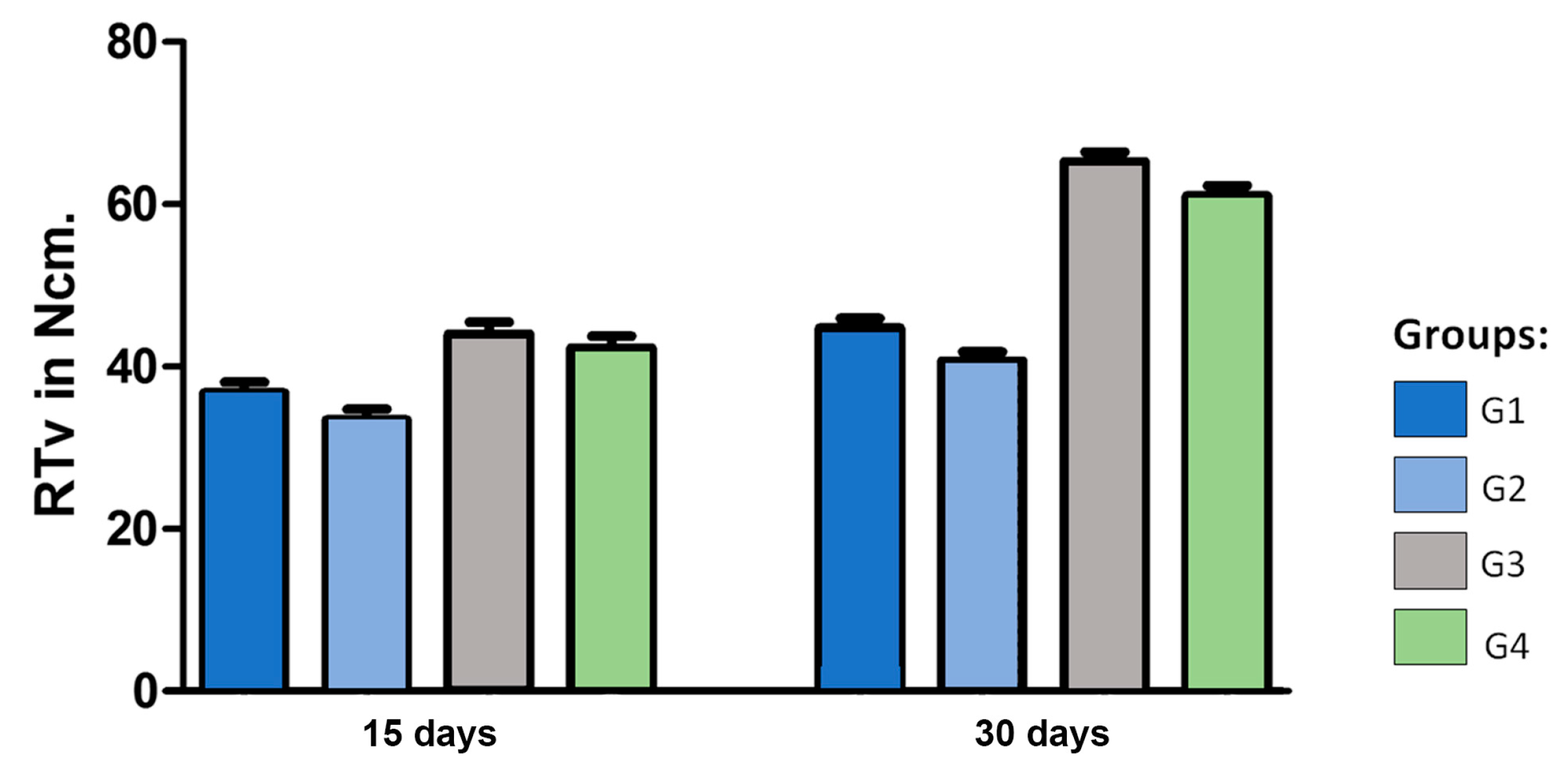
Removal torque measurement: Five samples of each group and each time (15 and 30 days) were used to measure the removal torque value (RTv). The analysis was performed in a computed torquimeter machine (Torque BioPDI, São Paulo, Brazil). The blocks (bone and implant) were fixed in the apparatus and the maximum value of removal torque was measured and tabulated.
Figure 7 shows the machine during the assay realization.
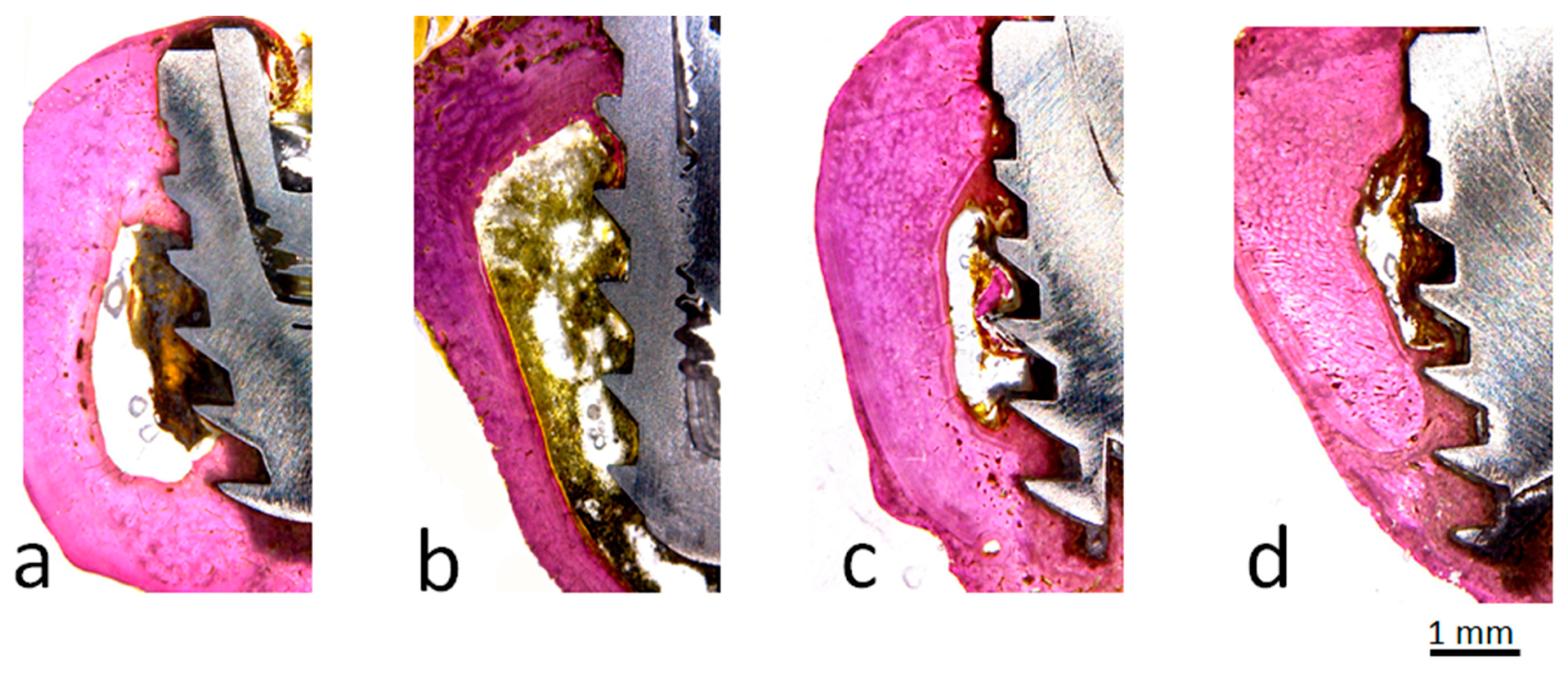
Histomorphometric and histological analysis: Three days after fixation in formaldehyde solution, the samples were washed in running tap water per 12 hours and then gradually dehydrated in a progressive series of ethanol solution (60% to 100%). After the dehydration, the blocks (bone with the implant) were embedded in historesin (Technovit 7200 VLC, Kultzer & Co, Wehrhein, Germany), polymerized, and cut in the central region of the implants using a metallographic cutter machine (Isomet 1000; Buehler, Germany). Then, the samples were polished using a sequence of abrasive paper (180 to 1200 mesh) in a polishing machine (Polipan-U, Panambra Zwick, São Paulo, Brazil). The samples were stained using the picrosirus hematoxylin staining technique. Images using an optical microscopy (Nykon E200, Tokyo, Japan) were obtained around all samples and, the percentage of bone-to-implant contact (BIC%) and bone area fraction occupancy (BAFO%) inside of the threads were measured using the ImageJ program (National Institute of Health, Bethesda, MD, USA). For the BIC% calculation, the total perimeter around the implant was considered 100% and, then, the areas where the bone is in contact with the implant surface were measured. Whereas, for the BAFO% calculation, the total area of threads was measured for the implant model used, and, then, the percentage of this area of threads occupied by the bone.
Statistical analysis: The ANOVA one-way statistical test was used following Bonferoni’s multiple comparison test to determine individual difference among groups. All analyses were performed using GraphPad Prism version 5.01 for Windows (GraphPad Software, San Diego, CA, USA). When p < 0.05, the differences were considered significant.
4. Discussion
The search for the development of new micro- and macro-geometries of implants which aim to improve and/or accelerate osseointegration has been a constant research topic in implantology worldwide. However, biological factors involved in the process are poorly considered, such as the intensity of the trauma generated during the surgical maneuvers resulting from each event, especially the steps of drilling and implant installation. It is known that the primary stability of the implants is considered fundamental for osseointegration [
25,
26,
27] and, that there is a high probability of failure in implants (~32%) presenting inadequate initial stability [
28]. The achievement of adequate primary stability is directly influenced by bone tissue strength (density), macrogeometry of the implant, and the surgical technique used [
26,
27,
28,
29]. Recent studies have shown that decompression of bone tissue by creating healing chambers with the use of an undersized drilling technique may improve the osseointegration process, however, this technique may compromise the implant’s fixation force (stability) on the bone. Then, a new implant macrogeometry was developed where healing chambers were created in the threads, and the purpose of this study was to compare different variables (ISQ, BIC%, BAFO%, and RTv). For this, we compared the conventional implant macrogeometry with the new implant macrogeometry during the initial phase of osseointegration, at 15 and 30 days after the installation in the bone.
The initial hypothesis, that this implant design does not change the values of initial stability, was proven and, the increase of the torque removal, BIC%, and BAFO% values in the tested samples, regardless of whether the surface is treated, showed that this macrogeometry with healing chambers generates a positive influence on the osseointegration process during the early time tested. Other studies have demonstrated the efficacy of healing chambers, however, most of them reported the possibility that there was a decrease in the primary stability of the implants by the technique (undersized drilling) of these spaces (healing chambers) inside the implants’ threads [
22,
23]. In the measurements obtained in our study there were no statistical differences (
p < 0.05) between the two models of implants tested regarding the primary stability values.
The intensity of surgical trauma during implant procedures maneuvers may vary during the drilling or insertion of the implant, as was demonstrated in our previous studies where NF-kB, which is a transcription factor involved in controlling the expression of several genes linked to the inflammatory response, was measured [
30]. The excess trauma caused by the inadequate drilling process or the excess compression of the bone tissue during implant installation was reported in some studies [
20,
31,
32]. The bone tissue has its elasticity limitation according to its density, which can dissipate the stress caused by the insertion torque of the implant [
33], which indicates that the bone can withstand a certain amount of compression. Thus, this justifies the possibility of applying a high torque with great initial stability until the implant obtains its biological stability. On the other hand, it was demonstrated that in case the bone tissue is damaged by excessive compression, excessive trauma during osteotomy, the bone may undergo necrosis, causing the implant to lose its stability. In this sense, the new implant design proposed and analyzed in the present study considered the possibility of obtaining an adequate primary stability without generating and/or decreasing the degree of bone compression after its insertion, as shown schematically by the image in
Figure 14. In implants with conventional threads, condensation of bone tissue will always occur, whereas in the implant model with healing cameras, these bone particles take place to lodge thereby decreasing compression.
Moreover, certain implant design features allow peri-implant osteocytes to retain a less aged phenotype, despite highly advanced extracellular matrix maturation. Then, the physicochemical properties of the material can stimulate bone formation and remodeling by regulating the expression of RANKL (receptor activator of nuclear factor–κB ligand), RANK, and OPG (osteoprotegerin) from implant-adherent cells. Modulation of certain osteocyte-related molecular signaling mechanisms (e.g., sclerostin blockade) may enhance the biomechanical anchorage of implants [
34].
Several studies have proposed that the morphological alterations on the implant surface characteristics can improve and accelerate the processes of healing of the bone tissue [
10,
11,
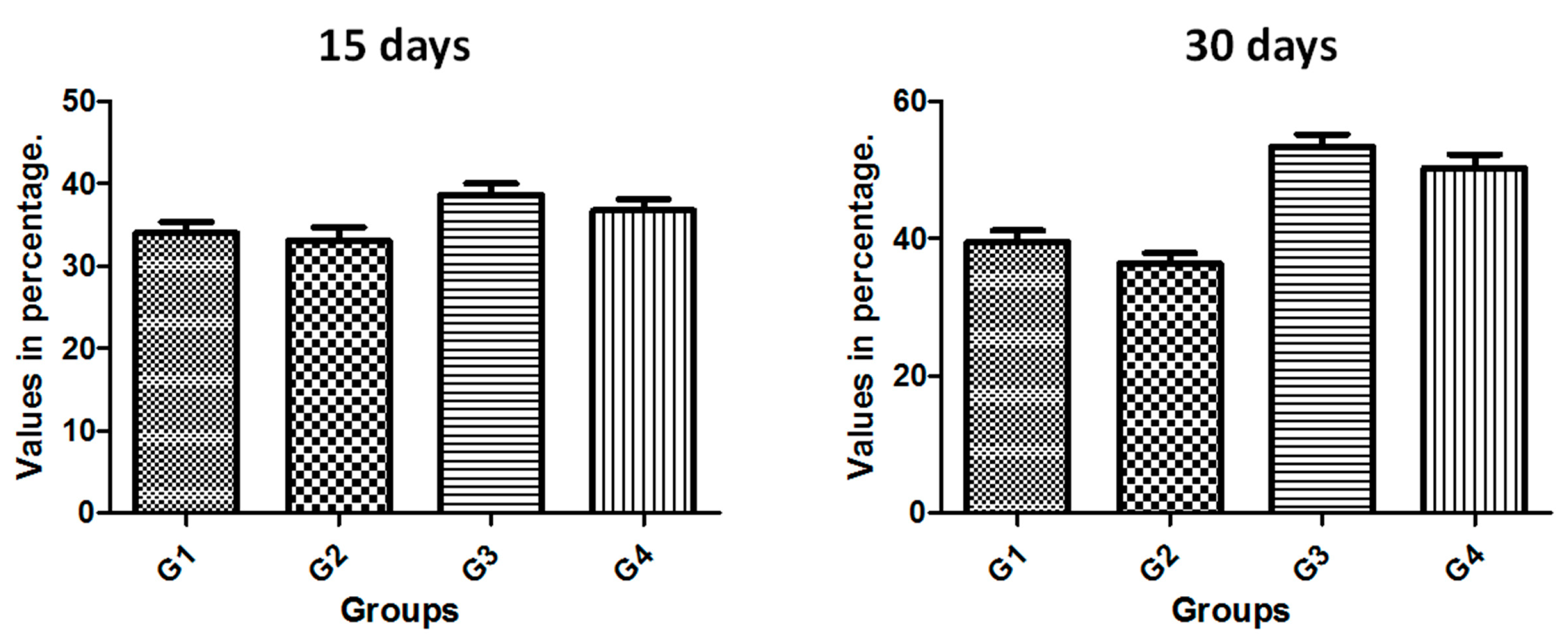
12]. Thus, the present study had the aim to evaluate both implant models (conventional and new implant design) using the same conditions of the surface treatment, with and without treatment, to verify the importance of this factor in the early time period of osseointegration. The results showed that the macrogeometry of the implant had a much higher importance than the surface treatment at the proposed evaluation times (15 and 30 days). Regarding implant stability (ISQ), the G3 and G4 implants had a significant increase in relation to the implants of the G1 and G2 groups, i.e., at 15 days in the general average 19% higher and at 30 days, 25% bigger. In addition, comparing the evolution of ISQ within the same implant model between the 3 measured times, the groups G1 and G2 showed only a time 3 evolution in relation to time 1 (21%), whereas in the G3 and G4 groups the increase was 20% between time 1 and 2, and 25% between time 2 and 3, totaling a 45% increase between time 1 and 3. These data clearly demonstrate the benefit of healing chambers created in the new implant design.
The removal torque measurement is a biomechanical analysis used to analyze the force of the interaction between the implant and bone tissue [
35,
36], where the higher values to implant removal indicate a good interaction between the bone and implant surface [
36], and a signal of good mineralization of the new bone formed. We compared the four groups proposed based on the two time points (15 and 30 days) after implantation, which was highly significant, and it is thus concluded that there is an important effect among the groups (
p < 0.05). Thus, as in the comparison made to ISQ values, when comparing the mean values of groups G1 and G2 with the groups G3 and G4, the latter presented a mean value 23% higher after 15 days and 48% higher after 30 days. When comparing the same groups between the times, the groups G1 and G2 had an increase of 21%, while the G3 and G4 groups showed a 47% increase in the removal torque of the implants. Again, the values indicate an acceleration in the process of osseointegration of the implants with the new design. Furthermore, the values compared statistically between the groups (G1 X G2 and G3 X G4) showed no statistically significant differences when evaluated implants were treated and not treated with the same macrogeometry for the time of 15 days and, showed no significant difference after 30 days.
Other studies have shown that implant design can present different osseointegration levels [
36,
37], depending on its variables presented in the micro- and macrogeometry. Histologically, the values related to the quantity and quality of the bone healing around the implants are evaluated by the percentage of contact of bone to implant (BIC%) and percentage of occupation of the bone area fraction (BAFO%). In the present study, these both analyses showed similar values and evolution on the tested times (15 and 30 days) in all groups. However, the comparison between the groups showed a higher value for the groups with the new implant macrogeometry (G3 and G4 groups) in comparison to the conventional implant macrogeometry (G1 and G2 groups). In the group G3, where the implants presented a new design associated with the surface treatment, the samples showed an important increase in the values of BIC% and BAFO% in the time of 30 days. Still, the healing chamber of group G3 presented a higher amount of BAFO%, indicating that the cellular reaction differed between the implant thread configurations. Other animal studies, where the healing chambers were examined in a longer time period (2 months), showed that healing chambers inserted in the cortical bone did not increase the BIC%, but increased implant biomechanical fixation at early times when compared to the conventional thread design [
38]. This data sought in the literature helps to reinforce the initial hypothesis that the new implant design with the healing chambers elaborated in the implant threads more strongly in the initial stages of osseointegration of the implants. However, new in vivo studies are necessary to prove these findings.
Osteocytes are important indicators of bone tissue quality and are also important structural markers of osseointegration. In addition, osteocytes are exceptionally valuable in characterizing bone tissue response to implanted materials [
34,
39]. Thus, we can cite, one of the limitations of the present study was to evaluate through immunohistochemical assay the amount of osteocytes around the different groups of implants used. This information would be of great importance because in the bone tissue surrounding the implants, osteocytes physically communicate with implant surfaces through the canaliculi and respond to mechanical loading (e.g., bone compression during the implant insertion), leading to changes in osteocyte numbers and morphology [
34].