Anomalous Increase in Specific Heat of Binary Molten Salt-Based Graphite Nanofluids for Thermal Energy Storage
Abstract
1. Introduction
2. Nanofluid Synthesis and Specific Heat Measurements
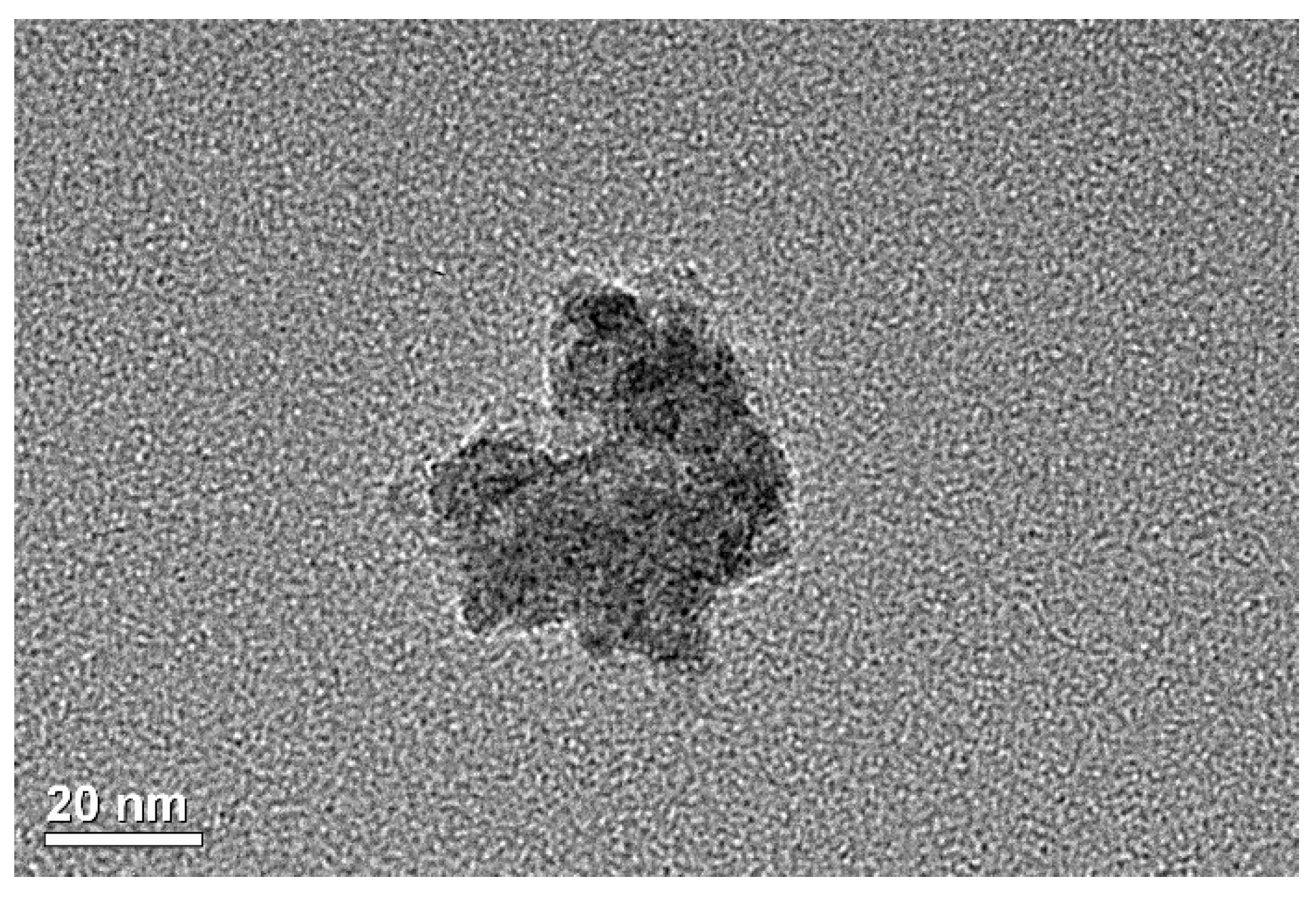
2.1. Materials
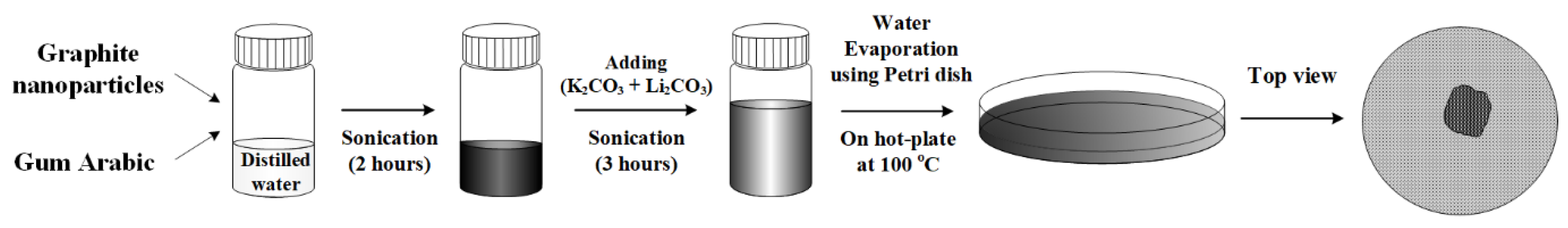
2.2. Synthesis of Nanofluids
2.3. Specific Heat Measurements
2.4. Uncertainty Analysis
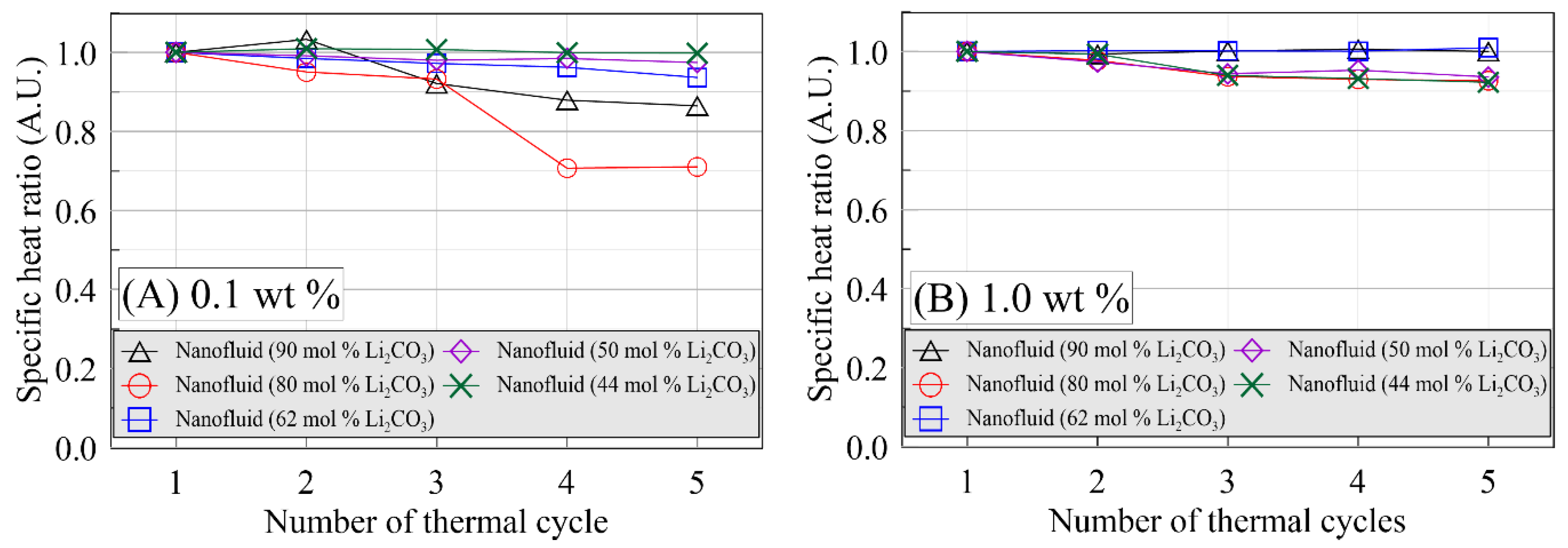
3. Results and Discussion
4. Conclusions
- (a)
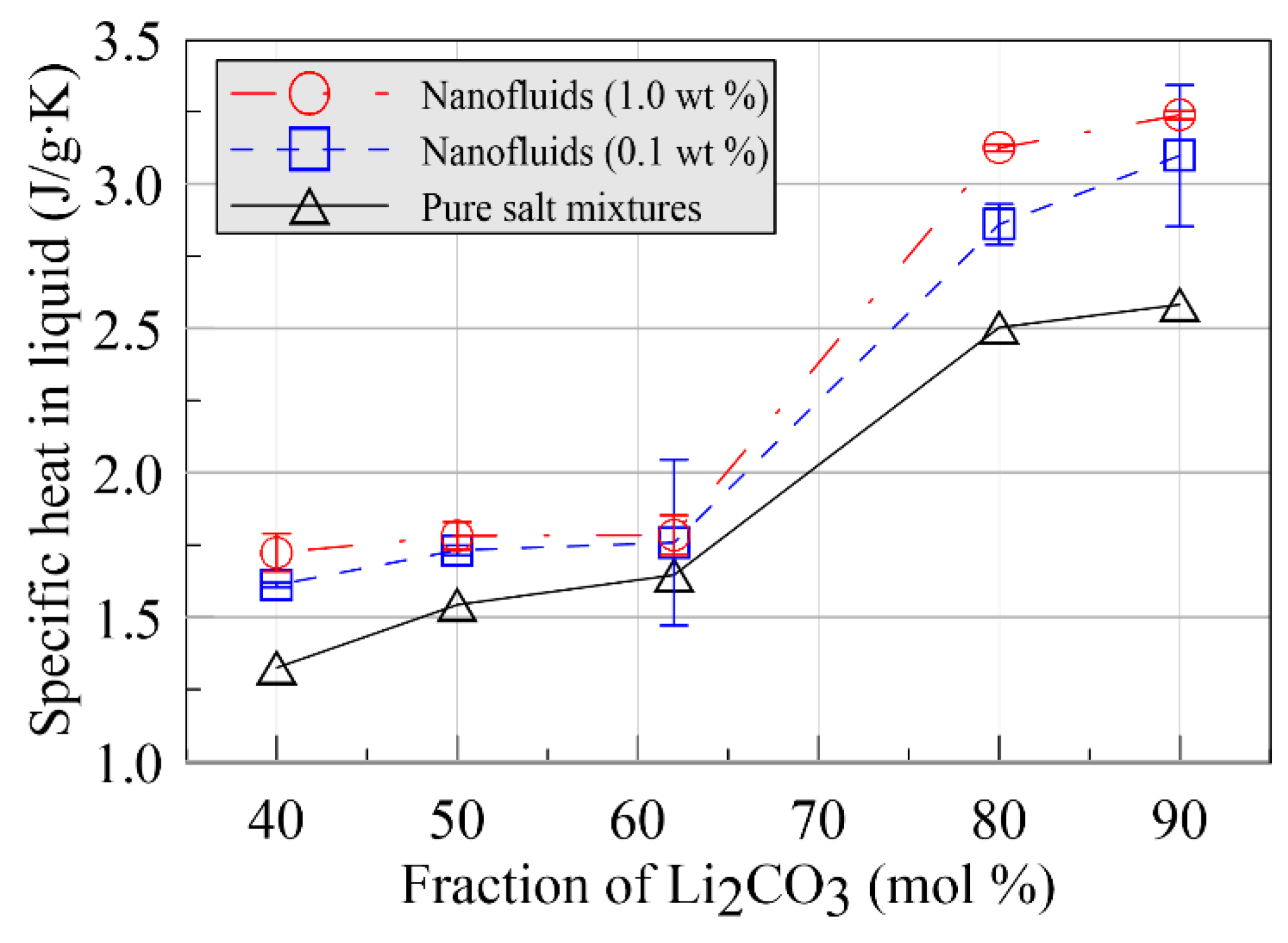
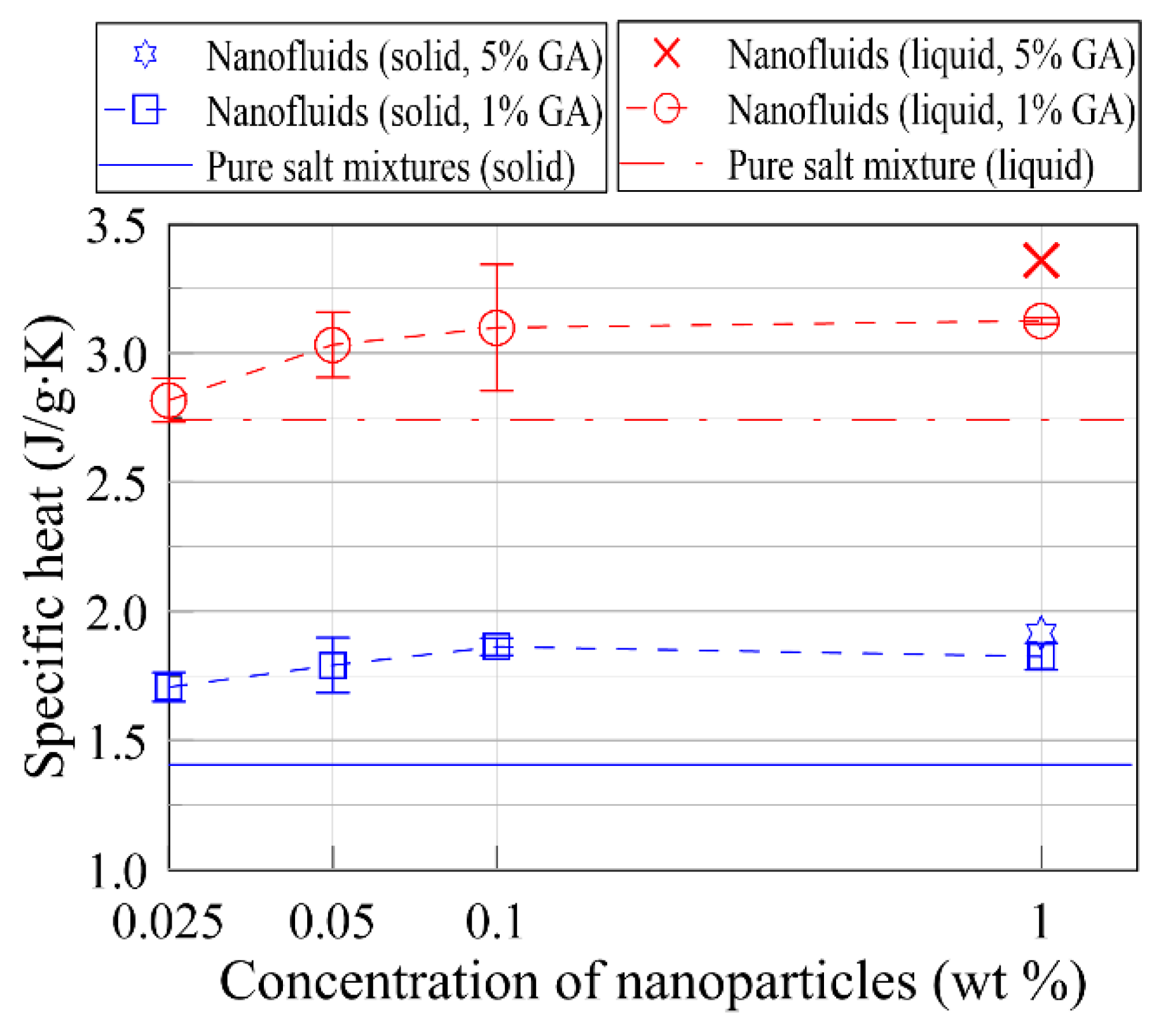
- The specific heat was anomalously enhanced by up to >30% in the liquid phase and 36% in the solid phase via the dispersion of graphite nanoparticles. The enhancements were strongly dependent on the chemical composition of the base fluid: the increments, as well as the enhancements in the specific heat, increased when the chemical composition of the base fluid was far from the eutectic composition.
- (b)
- The specific heat increased with the concentration of the nanoparticles. A noticeable increase was observed at the nanoparticle concentration of 0.025 wt % for the base fluid including 75 mol % lithium carbonate and 25 mol % potassium carbonate. A critical concentration of nanoparticles was observed. For nanoparticle concentrations exceeding 0.1 wt %, the increment was negligible.
- (c)
- The specific heat enhancements increased as the amount of the surfactant (GA) increased from 1 to 5 wt %. Larger enhancements were measured for the nanofluids with 5 wt % GA, and the actual specific heat value was almost double that of the eutectic.
- (d)
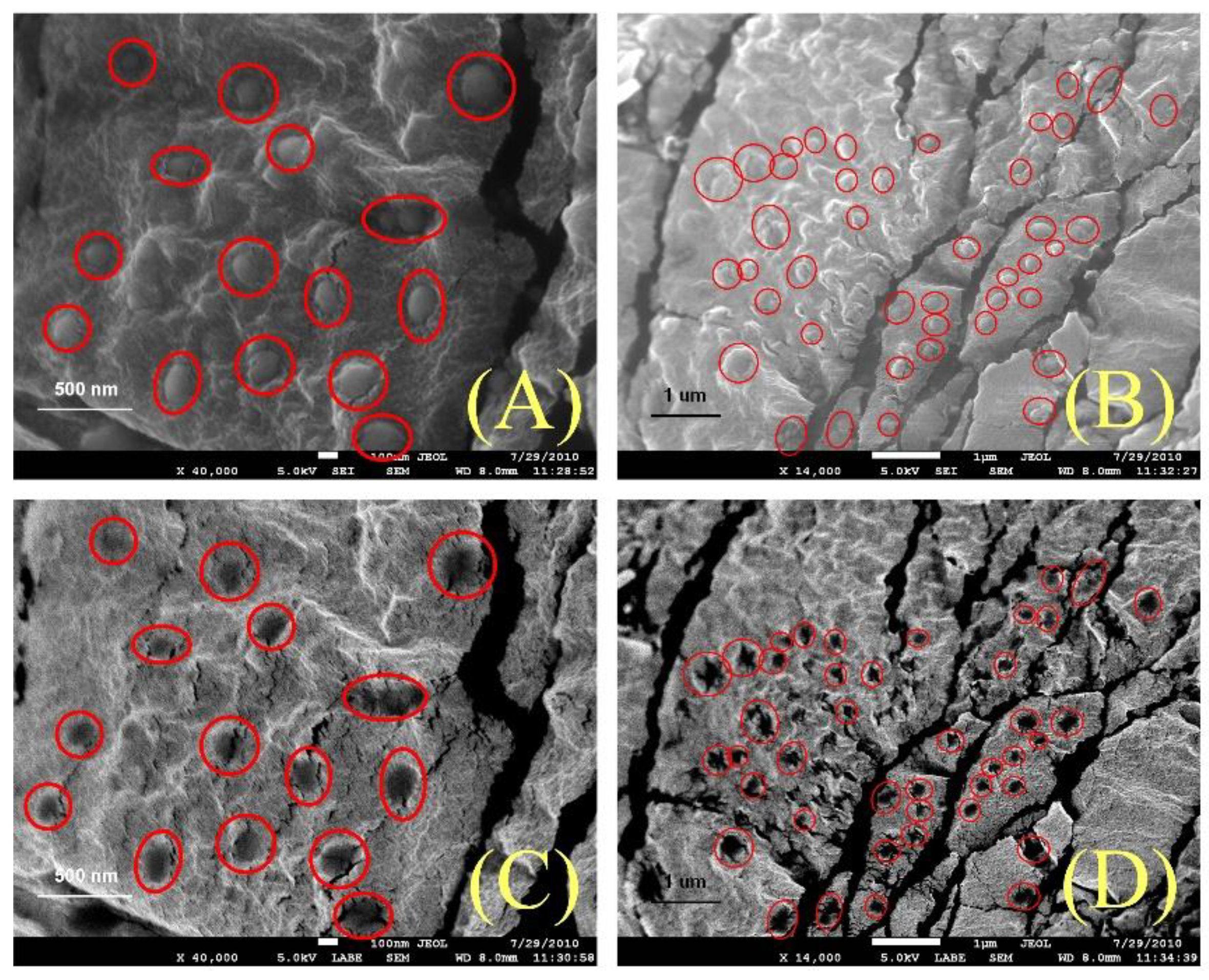
- The homogeneous dispersion of the graphite nanoparticles was visually confirmed via SEM (secondary electron images and backscatter images) for the nanofluid after a DSC test. In the SEM images, the graphite nanoparticles were spherical, having smooth curvature (not disks), so it was presumed that special structures, nanoparticle-induced compressed liquid layer, were formed in the nanofluids. The SEM images conclusively proved that the dispersion homogeneity (quality) of the nanoparticles is due to the specific heat enhancements of the nanofluids.
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Masuda, H.; Ebata, A.; Teramae, K. Alternation of thermal conductivity and viscosity of liquid by dispersing ultra-fine particles (Dispersion of γ-Al2O3, SiO2, and TiO2 ultra-fine particles). Netsu Bussei 1993, 4, 227–233. [Google Scholar] [CrossRef]
- Choi, S.U.S.; Eastman, J.A. Enhancing thermal conductivity of fluids with nanoparticles. In Proceedings of the International Mechanical Engineering Congress and Exhibition, San Francisco, CA, USA, 12–17 November 1995. [Google Scholar]
- You, S.M.; Kim, J.H.; Kim, K.H. Effect of nanoparticles on critical heat flux of water in pool boiling heat transfer. Appl. Phys. Lett. 2003, 83, 3374–3376. [Google Scholar] [CrossRef]
- Jo, B.; Jeon, P.S.; Yoo, J.; Kim, H.J. Wide rage parametric study for the pool boiling of nano-fluids with a circular plate heater. J. Vis. 2009, 47, 407–411. [Google Scholar]
- Wang, X.; Mujumdar, A.S. Heat transfer characteristics of nanofluids: A Review. Int. J. Therm. Sci. 2007, 46, 1–19. [Google Scholar] [CrossRef]
- Keblinski, P.; Eastman, J.A.; Cahill, D.G. Nanofluids for thermal transport. Mater. Today 2005, 8, 36–44. [Google Scholar] [CrossRef]
- Kim, S.J.; Bang, I.C.; Buongiorno, J. Effects of nanoparticle deposition on surface wettability influencing boiling heat transfer in nanofluids. Appl. Phys. Lett. 2006, 89, 153107. [Google Scholar] [CrossRef]
- Kim, H.D.; Kim, J.; Kim, M.H. Experimental studies on CHF characteristics of nano-fluids at pool boiling. Int. J. Multiph. Flow 2007, 33, 691–706. [Google Scholar] [CrossRef]
- Ujereh, S.; Fisher, T.; Mudawar, I. Effects of carbon nanotube arrays on nucleate pool boiling. Int. J. Heat Mass Transf. 2007, 50, 4023–4038. [Google Scholar] [CrossRef]
- Sathyamurthi, V.; Ahn, H.S.; Banerjee, D. Subcooled pool boiling experiments on horizontal heaters coated with carbon nanotubes. J. Heat Transf. 2009, 131, 071501. [Google Scholar] [CrossRef]
- Chen, R.; Lu, M.; Srinivasan, V. Nanowires for enhanced boiling heat transfer. Nano Lett. 2009, 9, 548–553. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kim, H.D.; Kim, H. Effects of nano-fluid and surfaces with nano structure on the increase of CHF. Exp. Therm. Fluid Sci. 2010, 34, 487–495. [Google Scholar] [CrossRef]
- Namburu, P.K.; Kulkarni, D.P.; Dandekar, A. Experimental investigation of viscosity and specific heat of silicon dioxide nanofluids. Micro Nano Lett. 2007, 2, 67–71. [Google Scholar] [CrossRef]
- Zhou, S.; Ni, R. Measurement of the specific heat capacity of water-based Al2O3 nanofluid. Appl. Phys. Lett. 2008, 92, 1–3. [Google Scholar] [CrossRef]
- Vajjha, R.S.; Das, D.K. Specific heat measurement of three nanofluids and development of new correlations. J. Heat Transf. 2009, 131, 071601. [Google Scholar] [CrossRef]
- Zhou, L.; Wang, B.; Peng, X. On the specific heat capacity of CuO nanofluid. Adv. Mech. Eng. 2010, 2010, 1–4. [Google Scholar] [CrossRef]
- Nelson, I.C.; Banerjee, D.; Ponnappan, R. Flow loop experiments using polyalphaolefin nanofluids. J. Thermophys. Heat Transf. 2009, 23, 752–761. [Google Scholar] [CrossRef]
- Shin, D.; Banerjee, D. Enhanced specific heat of silica nanofluid. J. Heat Transf. 2011, 133, 024501. [Google Scholar] [CrossRef]
- Shin, D.; Banerjee, D. Enhancement of specific heat capacity of high-temperature silica-nanofluids synthesized in alkali chloride salt eutectics for solar thermal-energy storage applications. Int. J. Heat Mass Transf. 2011, 54, 1064–1070. [Google Scholar] [CrossRef]
- Jo, B.; Banerjee, D. Enhanced specific heat capacity of molten salt-based nanomaterials: Effects of nanoparticle dispersion and solvent material. Acta Mater. 2014, 75, 80–91. [Google Scholar] [CrossRef]
- Jo, B.; Banerjee, D. Effect of dispersion homogeneity on specific heat capacity enhancement of molten salt nanomaterials using carbon nanotubes. J. Sol. Energy Eng. 2015, 137, 011011. [Google Scholar] [CrossRef]
- Jo, B.; Banerjee, D. Enhanced specific heat capacity of molten salt-based carbon nanotubes nanomaterials. J. Heat Transf. 2015, 137, 091013. [Google Scholar] [CrossRef]
- Chieruzzi, M.; Cerritelli, G.F.; Miliozzi, A.; Kenny, J.M. Effect of nanoparticles on heat capacity of nanofluids based on molten salts as PCM for thermal energy storage. Nanoscale Res. Lett. 2013, 8, 448. [Google Scholar] [CrossRef] [PubMed]
- Chieruzzi, M.; Miliozzi, A.; Crescenzi, T.; Torre, L.; Kenny, J.M. A new phase change material based on potassium nitrate with silica and alumina nanoparticles for thermal energy storage. Nanoscale Res. Lett. 2015, 10, 273–282. [Google Scholar] [CrossRef] [PubMed]
- Tiznobaik, H.; Shin, D. Enhanced specific heat capacity of high-temperature molten salt-based nanofluids. Int. J. Heat Mass Transf. 2013, 57, 542–548. [Google Scholar] [CrossRef]
- Dudda, B.; Shin, D. Effect of nanoparticle dispersion on specific heat capacity of a binary nitrate salt eutectic for concentrated solar power applications. Int. J. Therm. Sci. 2013, 69, 37–42. [Google Scholar] [CrossRef]
- Ho, M.X.; Pan, C. Optimal concentration of alumina nanoparticles in molten Hitec salt to maximize its specific heat capacity. Int. J. Heat Mass Transf. 2014, 70, 174–184. [Google Scholar] [CrossRef]
- Jo, B.; Banerjee, D. Effect of solvent on specific heat capacity enhancement of binary molten salt-based carbon nanotube nanomaterials for thermal energy storage. Int. J. Therm. Sci. 2015, 98, 219–227. [Google Scholar] [CrossRef]
- Mohebbi, A. Prediction of specific heat and thermal conductivity of nanofluids by a combined equilibrium and non-equilibrium molecular dynamics simulation. J. Mol. Liq. 2012, 175, 51–58. [Google Scholar] [CrossRef]
- Jo, B.; Banerjee, D. Molecular dynamics study on interfacial thermal resistance between organic nanoparticles and alkali molten salt mixtures. Int. J. Multiscale Comput. Eng. 2017, 15, 199–217. [Google Scholar] [CrossRef]
- Jo, B.; Banerjee, D. Thermal properties measurements of binary carbonate salt mixtures for concentrating solar power plants. J. Renew. Sustain. Energy 2015, 7, 033121. [Google Scholar] [CrossRef]
- Jo, B.; Banerjee, D. Viscosity measurements of multi-walled carbon nanotubes-based high temperature nanofluids. Mater. Lett. 2014, 122, 212–215. [Google Scholar] [CrossRef]
- Grosu, Y.; Udayashankar, N.; Bondarchuk, O.; Gonzalez, F.; Faik, A. Unexpected effect of nanoparticles doping on the corrosivity of molten nitrate salt for thermal energy storage. Sol. Energy Mater. Sol. Cells 2018, 178, 91–97. [Google Scholar] [CrossRef]
- Qiao, G.; Lasfargues, M.; Alexiadis, A.; Ding, Y. Simulation and experimental study of the specific heat capacity of molten salt based nanofluids. Appl. Therm. Eng. 2017, 111, 033121. [Google Scholar] [CrossRef]
- Luo, Y.; Du, X.; Awad, A.; Wen, D. Thermal energy storage enhancement of a binary molten salt via in-situ produced nanoparticles. Int. J. Heat Mass Transf. 2017, 104, 658–664. [Google Scholar] [CrossRef]
- Huang, Y.; Cheng, X.; Li, Y.; Yu, G.; Xu, K.; Li, G. Effect of in-situ synthesized nano-MgO on thermal properties of NaNO3-KNO3. Sol. Energy 2018, 160, 208–215. [Google Scholar] [CrossRef]
- Mondragon, R.; Julia, J.E.; Cabedo, L.; Navarrete, N. On the relationship between the specific heat enhancement of salt-based nanofluids and the ionic exchange capacity of nanoparticles. Sci. Rep. 2018, 8, 7532. [Google Scholar] [CrossRef] [PubMed]
- Janz, G.J.; Allen, C.B.; Bansal, N.P.; Murphpy, R.M.; Tomkins, R.P.T. Physical properties data compilations relevant to energy storage-2. In Molten Salts: Data on Single and Multi-Component Salt Systems; US Govt. Print. Off.: Washington, DC, USA, 1979; Volume 61, pp. 1–452. [Google Scholar]
- Hilding, J.; Grulke, E.A.; Zhang, Z.G. Dispersion of carbon nanotubes in liquids. J. Dispers. Sci. Technol. 2003, 24, 1–41. [Google Scholar] [CrossRef]
- Ding, Y.; Alias, H.; Wen, D. Heat transfer of aqueous suspensions of carbon nanotubes (CNT nanofluids). Int. J. Heat Mass Transf. 2006, 49, 240–250. [Google Scholar] [CrossRef]
- ASTM E1269-95. Standard Test Method for Determining Specific Heat Capacity by Differential Scanning Calorimetry; American Society for Testing and Materials: West Conshohocken, PA, USA, 2005. [Google Scholar]
- Daoub, R.M.A.; Elmubarak, A.H.; Misran, M.; Hassan, E.A.; Osman, M.E. Characterization and functional properties of some natural Acacia gums. J. Saudi Soc. Agric. Sci. 2018, 17, 241–249. [Google Scholar] [CrossRef]
- Kline, S.J.; McClintock, F.A. Describing uncertainties in single-sample experiments. Mech. Eng. 1953, 75, 3–8. [Google Scholar]
- Araki, N.; Futamura, M.; Makino, A. Measurements of thermophysical properties of sodium acetate hydrate. Int. J. Thermophys. 1995, 16, 1455–1466. [Google Scholar] [CrossRef]
- Shin, D.; Banerjee, D. Specific heat of nanofluids synthesized by dispersing alumina nanoparticles in alkali salt eutectic. Int. J. Heat Mass Transf. 2014, 74, 210–214. [Google Scholar] [CrossRef]
- Oh, S.H.; Kauffmann, Y.; Scheu, C.; Kapan, W.D.; Ruhle, M. Ordered liquid aluminum at the interface with sapphire. Science 2005, 310, 661–663. [Google Scholar] [CrossRef] [PubMed]
- Buongiorno, J. Convective transport in nanofluids. J. Heat Transf. 2006, 128, 240–250. [Google Scholar] [CrossRef]



| Mole Fraction of Each Salt | Specific Heat in Liquid Phase [J/g·K] | ||
|---|---|---|---|
| Sample Name | Li2CO3 [mol %] | K2CO3 [mol %] | |
| Sample 1 | 90 | 10 | 2.582 |
| Sample 2 | 80 | 20 | 2.503 |
| Sample 3 | 62 | 38 | 1.646 |
| Sample 4 | 50 | 50 | 1.543 |
| Sample 5 | 40 | 60 | 1.325 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, H.J.; Jo, B. Anomalous Increase in Specific Heat of Binary Molten Salt-Based Graphite Nanofluids for Thermal Energy Storage. Appl. Sci. 2018, 8, 1305. https://doi.org/10.3390/app8081305
Kim HJ, Jo B. Anomalous Increase in Specific Heat of Binary Molten Salt-Based Graphite Nanofluids for Thermal Energy Storage. Applied Sciences. 2018; 8(8):1305. https://doi.org/10.3390/app8081305
Chicago/Turabian StyleKim, Hyun Jung, and Byeongnam Jo. 2018. "Anomalous Increase in Specific Heat of Binary Molten Salt-Based Graphite Nanofluids for Thermal Energy Storage" Applied Sciences 8, no. 8: 1305. https://doi.org/10.3390/app8081305
APA StyleKim, H. J., & Jo, B. (2018). Anomalous Increase in Specific Heat of Binary Molten Salt-Based Graphite Nanofluids for Thermal Energy Storage. Applied Sciences, 8(8), 1305. https://doi.org/10.3390/app8081305





