Silver Nanowires: Synthesis, Antibacterial Activity and Biomedical Applications
Abstract
1. Introduction
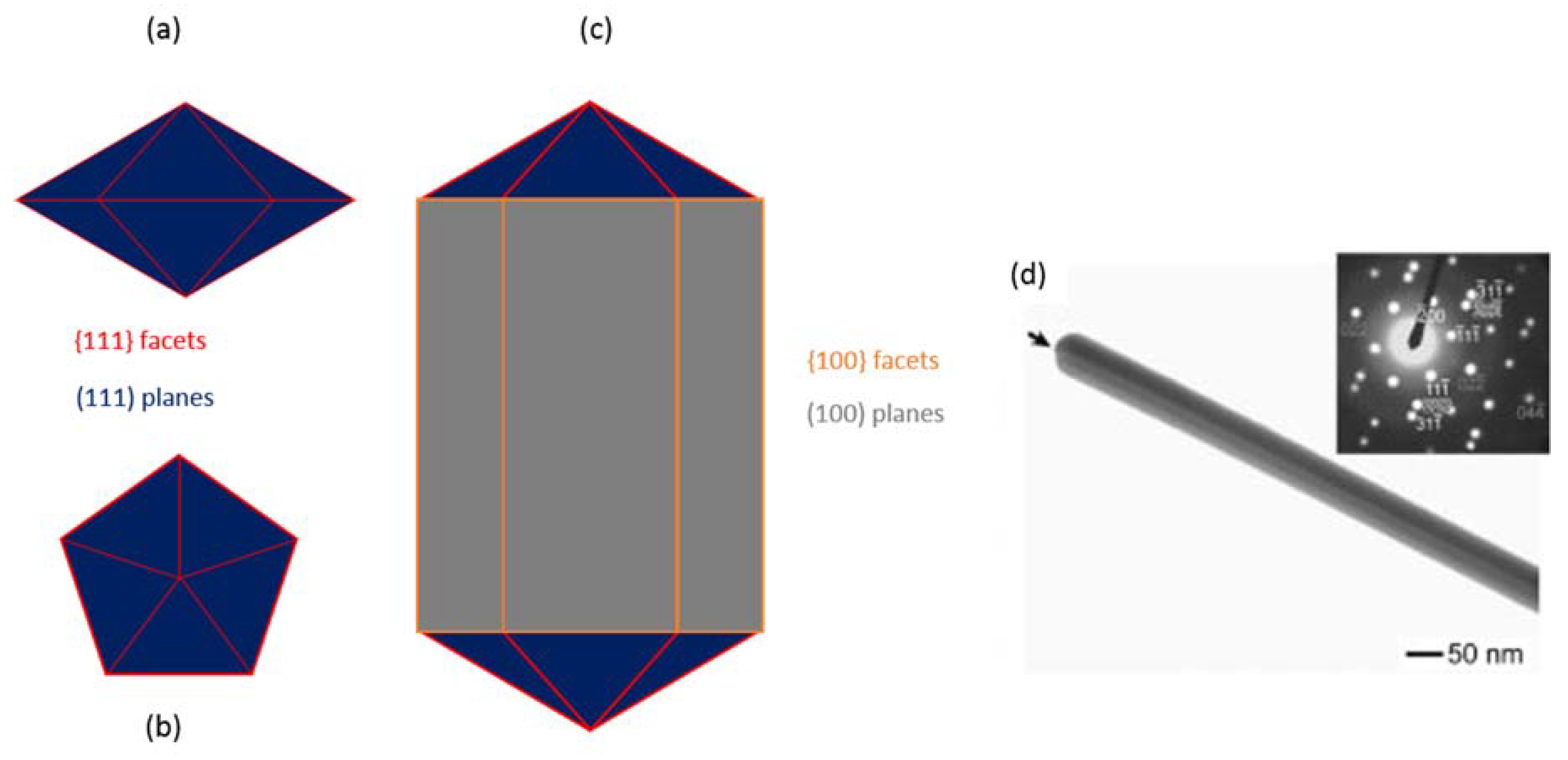
2. Synthesis of AgNWs
2.1. Hard-Template Synthesis
2.2. Soft-Template Synthesis
3. Mechanism of Antibacterial Activity
3.1. Silver Nanoparticles Mechanism of Action
3.2. Silver Nanowires Mechanism of Action
4. Toxicity of Silver Nanowires
5. Emerging Biomedical Applications of Silver Nanowires
5.1. Textiles and Fibrous Membranes
5.2. Surface Coating of Medical Devices
5.3. Drug Delivery
6. Conclusions and Future Prospects
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Kumar, S.S.D.; Houreld, N.N.; Kroukamp, E.M.; Abrahamse, H. Cellular imaging and bactericidal mechanism of green-synthesized silver nanoparticles against human pathogenic bacteria. J. Photochem. Photobiol. B 2018, 178, 259–269. [Google Scholar] [CrossRef] [PubMed]
- Korshed, P.; Li, L.; Liu, Z.; Mironov, A.; Wang, T. Antibacterial mechanisms of a novel type picosecond laser-generated silver-titanium nanoparticles and their toxicity to human cells. Int. J. Nanomed. 2018, 13, 89–101. [Google Scholar] [CrossRef] [PubMed]
- Atiyeh, B.S.; Costagliola, M.; Hayek, S.N.; Dibo, S.A. Effect of silver on burn wound infection control and healing: Review of the literature. Burns 2018, 33, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Phuong Phong, N.T.; Ke Thanh, N.V.; Phuong, P.H. Fabrication of antibacterial water filter by coating silver nanoparticles on flexible polyurethane foams. J. Phys. Conf. Ser. 2009, 187. [Google Scholar] [CrossRef]
- Laxminarayan, R.; Duse, A.; Wattal, C.; Zaidi, A.K.M.; Wertheim, H.F.L.; Sumpradit, N.; Vlieghe, E.; Hara, G.L.; Gould, I.M.; Goossens, H.; et al. Antibiotic resistance-the need for global solutions. Lancet Infect. Dis. 2013, 13, 1057–1098. [Google Scholar] [CrossRef]
- Jenks, J. Antibiotic resistance needs global solutions. Lancet Infect. Dis. 2018, 14, 550. [Google Scholar] [CrossRef]
- Singh, M.; Movia, D.; Mahfoud Omar, K.; Volkov, Y.; Prina-Mello, A. Silver nanowires as prospective carriers for drug delivery in cancer treatment: An in vitro biocompatibility study on lung adenocarcinoma cells and fibroblasts. Eur. J. Nanomed. 2013, 5, 195. [Google Scholar] [CrossRef]
- Huang, Z.; Jiang, X.; Guo, D.; Gu, N. Controllable synthesis and biomedical applications of silver nanomaterials. J. Nanosci. Nanotechnol. 2011, 11, 9395–9408. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Yang, P.; Sun, Y.; Wu, Y.; Mayers, B.; Gates, B.; Yin, Y.; Kim, F.; Yan, H. One-Dimensional Nanostructures: Synthesis, Characterization, and Applications. Adv. Mater. 2003, 15, 353–389. [Google Scholar] [CrossRef]
- Bachenheimer, L.; Scherzer, R.; Elliott, P.; Stagon, S.; Gasparov, L.; Huang, H. Degradation Mechanism of Ag Nanorods for Surface Enhanced Raman Spectroscopy. Sci. Rep. 2017, 7, 4–7. [Google Scholar] [CrossRef] [PubMed]
- Liang, H.; Wang, W.; Huang, Y.; Zhang, S.; Wei, H.; Xu, H. Controlled Synthesis of Uniform Silver Nanospheres. J. Phys. Chem. C 2010, 114, 7427–7431. [Google Scholar] [CrossRef]
- Korte, K. Rapid Synthesis of Silver Nanowires. Natl. Nanotechnol. Infrastruct. Netw. 2007, 28–29. [Google Scholar] [CrossRef]
- Hoop, M.; Shen, Y.; Chen, X.Z.; Mushtaq, F.; Iuliano, L.M.; Sakar, M.S.; Petruska, A.; Loessner, M.J.; Nelson, B.J.; Pané, S. Magnetically Driven Silver-Coated Nanocoils for Efficient Bacterial Contact Killing. Adv. Funct. Mater. 2016, 26, 1063–1069. [Google Scholar] [CrossRef]
- Ge, L.; Li, Q.; Wang, M.; Ouyang, J.; Li, X.; Xing, M.M.Q. Nanosilver particles in medical applications: Synthesis, performance, and toxicity. Int. J. Nanomed. 2014, 9, 2399–2407. [Google Scholar] [CrossRef]
- Seil, J.T.; Webster, T.J. Antimicrobial applications of nanotechnology: Methods and literature. Int. J. Nanomed. 2012, 7, 2767–2781. [Google Scholar] [CrossRef]
- Dhapte, V.; Kadam, S.; Moghe, A.; Pokharkar, V. Probing the wound healing potential of biogenic silver nanoparticles. J. Wound Care 2014, 23, 431–432, 434, 436 passim. [Google Scholar] [CrossRef] [PubMed]
- Jung, W.K.; Koo, H.C.; Kim, K.W.; Shin, S.; Kim, S.H.; Park, Y.H. Antibacterial Activity and Mechanism of Action of the Silver Ion in Staphylococcus aureus and Escherichia coli. Appl. Environ. Microbiol. 2008, 74, 2171–2178. [Google Scholar] [CrossRef] [PubMed]
- Kalpana, D.; Lee, Y.S. Synthesis and characterization of bactericidal silver nanoparticles using cultural filtrate of simulated microgravity grown Klebsiella pneumoniae. Enzyme Microb. Technol. 2013, 52, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Ovington, L.G. The truth about silver. Ostomy Wound Manag. 2004, 50, 1S–10S. [Google Scholar]
- Tang, X.; Tsuji, M. Syntheses of Silver Nanowires in Liquid Phase. In Nanowires Science and Technology; Lupu, N., Ed.; InTech: Rijeka, Croatia, 2010; pp. 25–42. ISBN 9789537619343. [Google Scholar]
- Han, Y.-J.; Kim, J.M.; Stucky, G.D. Preparation of Noble Metal Nanowires Using Hexagonal Mesoporous Silica SBA-15. Chem. Mater. 2000, 12, 2068–2069. [Google Scholar] [CrossRef]
- Takai, A.; Doi, Y.; Yamauchi, Y.; Kuroda, K. Soft-Chemical Approach of Noble Metal Nanowires Templated from Mesoporous Silica (SBA-15) through Vapor Infiltration of a Reducing Agent. J. Phys. Chem. C 2010, 114, 7586–7593. [Google Scholar] [CrossRef]
- Kim, K.-J.; Lee, E.-S.; Kwon, Y.-U. Syntheses of micrometer-long Pt and Ag nanowires through SBA-15 templating. J. Nanopart. Res. 2012, 14, 1270. [Google Scholar] [CrossRef]
- Sun, Y.; Mayers, B.; Herricks, T.; Xia, Y. Polyol Synthesis of Uniform Silver Nanowires: A Plausible Growth Mechanism and the Supporting Evidence. Nano Lett. 2003, 3, 955–960. [Google Scholar] [CrossRef]
- Sun, Y.; Yin, Y.; Mayers, B.T.; Herricks, T.; Xia, Y. Uniform silver nanowires synthesis by reducing AgNO3 with ethylene glycol in the presence of seeds and poly(vinyl pyrrolidone). Chem. Mater. 2002, 14, 4736–4745. [Google Scholar] [CrossRef]
- Sun, Y.; Xia, Y. Large-scale synthesis of uniform silver nanowires through a soft, self-seeding, polyol process. Adv. Mater. 2002, 14, 833–837. [Google Scholar] [CrossRef]
- Korte, K.E.; Skrabalak, S.E.; Xia, Y. Rapid synthesis of silver nanowires through a CuCl- or CuCl2-mediated polyol process. J. Mater. Chem. 2008, 18, 437–441. [Google Scholar] [CrossRef]
- Chen, D.; Qiao, X.; Qiu, X.; Chen, J.; Jiang, R. Convenient synthesis of silver nanowires with adjustable diameters via a solvothermal method. J. Colloid Interface Sci. 2010, 344, 286–291. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.-H.; Chae, Y.-S.; Lee, J.-H.; Kwon, Y.; Kim, Y.-S. Mechanism of metal nanowire formation via the polyol process. Electron. Mater. Lett. 2015, 11, 735–740. [Google Scholar] [CrossRef]
- Bergin, S.M.; Chen, Y.-H.; Rathmell, A.R.; Charbonneau, P.; Li, Z.-Y.; Wiley, B.J. The effect of nanowire length and diameter on the properties of transparent, conducting nanowire films. Nanoscale 2012, 4, 1996. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.H.; Jiang, Z.Y.; Xie, Z.X.; Xu, X.; Huang, R.B.; Zheng, L.S. Growth of silver nanowires from solutions: A cyclic penta-twinned-crystal growth mechanism. J. Phys. Chem. B 2005, 109, 9416–9421. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Tang, Y.; Su, Z.; Zhang, Z.; Fang, C. Preparation of Silver Nanowires via a Rapid, Scalable and Green Pathway. J. Mater. Sci. Technol. 2015, 31, 16–22. [Google Scholar] [CrossRef]
- Visnapuu, M.; Joost, U.; Juganson, K.; Künnis-Beres, K.; Kahru, A.; Kisand, V.; Ivask, A. Dissolution of silver nanowires and nanospheres dictates their toxicity to escherichia coli. Biomed. Res. Int. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Hong, X.; Wen, J.; Xiong, X.; Hu, Y. Shape effect on the antibacterial activity of silver nanoparticles synthesized via a microwave-assisted method. Environ. Sci. Pollut. Res. 2016, 23, 4489–4497. [Google Scholar] [CrossRef] [PubMed]
- Helmlinger, J.; Sengstock, C.; Mayer, C.; Schildhauer, T.A.; Epple, M. Silver nanoparticles with different size and shape: Equal cytotoxicity, but different antibacterial effect. RSC Adv. 2016, 6, 18490–18501. [Google Scholar] [CrossRef]
- Cui, J.; Liu, Y. Preparation of graphene oxide with silver nanowires to enhance antibacterial properties and cell compatibility. RSC Adv. 2015, 5, 85748–85755. [Google Scholar] [CrossRef]
- Nateghi, M.R.; Shateri-Khalilabad, M. Silver nanowire-functionalized cotton fabric. Carbohydr. Polym. 2015, 117, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Wu, Y.; Wang, Z.; Zhang, X.; Zhao, Y.; Sun, L. Electrospinning of Ag Nanowires/polyvinyl alcohol hybrid nanofibers for their antibacterial properties. Mater. Sci. Eng. C 2017, 78, 706–714. [Google Scholar] [CrossRef] [PubMed]
- Shahzadi, K.; Wu, L.; Ge, X.; Zhao, F.; Li, H.; Pang, S.; Jiang, Y.; Guan, J.; Mu, X. Preparation and characterization of bio-based hybrid film containing chitosan and silver nanowires. Carbohydr. Polym. 2016, 137, 732–738. [Google Scholar] [CrossRef] [PubMed]
- Lei, J.; Zhou, L.; Tang, Y.; Luo, Y.; Duan, T.; Zhu, W. High-strength konjac glucomannan/silver nanowires composite films with antibacterial properties. Materials 2017, 10, 524. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Teng, C.P. Fabrication of silver nanowires-loaded polydimethylsiloxane film with antimicrobial activities and cell compatibility. Mater. Sci. Eng. C 2017, 70, 1011–1017. [Google Scholar] [CrossRef] [PubMed]
- Polívková, M.; Štrublová, V.; Hubáček, T.; Rimpelová, S.; Švorčík, V.; Siegel, J. Surface characterization and antibacterial response of silver nanowire arrays supported on laser-treated polyethylene naphthalate. Mater. Sci. Eng. C 2017, 72, 512–518. [Google Scholar] [CrossRef] [PubMed]
- Ajayan, P.M.; lijima, S. Capillarity-induced filling of carbon nanotubes. Nature 1993, 361, 333. [Google Scholar] [CrossRef]
- Ugarte, D.; Chatelain, A.; de Heer, W.A. Nanocapillarity and Chemistry in Carbon Nanotubes. Science 1996, 274, 1897–1899. [Google Scholar] [CrossRef] [PubMed]
- Martin, C.R. Nanomaterials—A membrane-based synthetic approach. Science 1994, 266, 1961–1966. [Google Scholar] [CrossRef] [PubMed]
- Kawamura, G.; Muto, H.; Matsuda, A. Hard template synthesis of metal nanowires. Front. Chem. 2014, 2, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Viau, G.; Fiévet-Vincent, F.; Fiévet, F. Nucleation and growth of bimetallic CoNi and FeNi monodisperse particles prepared in polyols. Solid State Ion. 1996, 84, 259–270. [Google Scholar] [CrossRef]
- Xia, Y.; Xiong, Y.; Lim, B.; Skrabalak, S.E. Shape-controlled synthesis of metal nanocrystals: Simple chemistry meets complex physics? Angew. Chem. Int. Ed. 2009, 48, 60–103. [Google Scholar] [CrossRef] [PubMed]
- Rycenga, M.; Cobley, C.M.; Zeng, J.; Li, W.; Moran, C.H.; Zhang, Q.; Qin, D.; Xia, Y. Controlling the synthesis and assembly of silver nanostructures for plasmonic applications. Chem. Rev. 2011, 111, 3669–3712. [Google Scholar] [CrossRef] [PubMed]
- Mao, H.; Feng, J.; Ma, X.; Wu, C.; Zhao, X. One-dimensional silver nanowires synthesized by self-seeding polyol process. J. Nanopart. Res. 2012, 14. [Google Scholar] [CrossRef]
- Gao, Y.; Jiang, P.; Song, L.; Liu, L.; Yan, X.; Zhou, Z.; Liu, D.; Wang, J.; Yuan, H.; Zhang, Z.; et al. Growth mechanism of silver nanowires synthesized by polyvinylpyrrolidone-assisted polyol reduction. J. Phys. D Appl. Phys. 2005, 38, 1061–1067. [Google Scholar] [CrossRef]
- Marambio-Jones, C.; Hoek, E.M.V. A review of the antibacterial effects of silver nanomaterials and potential implications for human health and the environment. J. Nanopart. Res. 2010, 12, 1531–1551. [Google Scholar] [CrossRef]
- AshaRani, P.V.; Low Kah Mun, G.; Hande, M.P.; Valiyaveettil, S. Cytotoxicity and Genotoxicity of Silver Nanoparticles in Human Cells. ACS Nano 2009, 3, 279–290. [Google Scholar] [CrossRef] [PubMed]
- Choi, O.; Deng, K.K.; Kim, N.J.; Ross, L.; Surampalli, R.Y.; Hu, Z. The inhibitory effects of silver nanoparticles, silver ions, and silver chloride colloids on microbial growth. Water Res. 2008, 42, 3066–3074. [Google Scholar] [CrossRef] [PubMed]
- Dakal, T.C.; Kumar, A.; Majumdar, R.S.; Yadav, V. Mechanistic Basis of Antimicrobial Actions of Silver Nanoparticles. Front. Microbiol. 2016, 7, 1831. [Google Scholar] [CrossRef] [PubMed]
- Ravindran, A.; Chandran, P.; Khan, S.S. Biofunctionalized silver nanoparticles: Advances and prospects. Colloids Surf. B Biointerfaces 2013, 105, 342–352. [Google Scholar] [CrossRef] [PubMed]
- Schreurs, W.J.A.; Rosenberg, H. Effect of silver ions on transport and retention of phosphate by Escherichia coli. J. Bacteriol. 1982, 152, 7–13. [Google Scholar] [PubMed]
- Ramalingam, B.; Parandhaman, T.; Das, S.K. Antibacterial Effects of Biosynthesized Silver Nanoparticles on Surface Ultrastructure and Nanomechanical Properties of Gram-Negative Bacteria viz. Escherichia coli and Pseudomonas aeruginosa. ACS Appl. Mater. Interfaces 2016, 8, 4963–4976. [Google Scholar] [CrossRef] [PubMed]
- Caro, A.A.; Cederbaum, A.I. Oxidative stress, toxicology, and pharmacology of CYP2E1. Annu. Rev. Pharmacol. Toxicol. 2004, 44, 27–42. [Google Scholar] [CrossRef] [PubMed]
- Apel, K.; Hirt, H. Reactive oxygen species: Metabolism, oxidative stress, and signal transduction. Annu. Rev. Plant Biol. 2004, 55, 373–399. [Google Scholar] [CrossRef] [PubMed]
- Holt, K.B.; Bard, A.J. Interaction of Silver(I) Ions with the Respiratory Chain of Escherichia coli: An Electrochemical and Scanning Electrochemical Microscopy Study of the Antimicrobial Mechanism of Micromolar Ag+. Biochemistry 2005, 44, 13214–13223. [Google Scholar] [CrossRef] [PubMed]
- Park, H.-J.; Kim, J.Y.; Kim, J.; Lee, J.-H.; Hahn, J.-S.; Gu, M.B.; Yoon, J. Silver-ion-mediated reactive oxygen species generation affecting bactericidal activity. Water Res. 2009, 43, 1027–1032. [Google Scholar] [CrossRef] [PubMed]
- Rhoads, D.M.; Umbach, A.L.; Subbaiah, C.C.; Siedow, J.N. Mitochondrial Reactive Oxygen Species. Contribution to Oxidative Stress and Interorganellar Signaling. Plant Physiol. 2006, 141, 357–366. [Google Scholar] [CrossRef] [PubMed]
- Lobo, V.; Patil, A.; Phatak, A.; Chandra, N. Free radicals, antioxidants and functional foods: Impact on human health. Pharmacogn. Rev. 2010, 4, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Imlay, J.A. The molecular mechanisms and physiological consequences of oxidative stress: Lessons from a model bacterium. Nat. Rev. Microbiol. 2013, 11, 443–454. [Google Scholar] [CrossRef] [PubMed]
- Lok, C.-N.; Ho, C.-M.; Chen, R.; He, Q.-Y.; Yu, W.-Y.; Sun, H.; Tam, P.K.-H.; Chiu, J.-F.; Che, C.-M. Proteomic analysis of the mode of antibacterial action of silver nanoparticles. J. Proteome Res. 2006, 5, 916–924. [Google Scholar] [CrossRef] [PubMed]
- Durán, N.; Durán, M.; de Jesus, M.B.; Seabra, A.B.; Fávaro, W.J.; Nakazato, G. Silver nanoparticles: A new view on mechanistic aspects on antimicrobial activity. Nanomed. Nanotechnol. Biol. Med. 2016, 12, 789–799. [Google Scholar] [CrossRef] [PubMed]
- Hwang, E.T.; Lee, J.H.; Chae, Y.J.; Kim, Y.S.; Kim, B.C.; Sang, B.-I.; Gu, M.B. Analysis of the toxic mode of action of silver nanoparticles using stress-specific bioluminescent bacteria. Small 2008, 4, 746–750. [Google Scholar] [CrossRef] [PubMed]
- Carlson, C.; Hussain, S.M.; Schrand, A.M.; Braydich-Stolle, L.K.; Hess, K.L.; Jones, R.L.; Schlager, J.J. Unique cellular interaction of silver nanoparticles: Size dependent generation of reactive oxygen species. J. Phys. Chem. B 2008, 112, 13608–13619. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.F.; Imlay, J.A. Silver(I), mercury(II), cadmium(II), and zinc(II) target exposed enzymic iron-sulfur clusters when they toxify Escherichia coli. Appl. Environ. Microbiol. 2012, 78, 3614–3621. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Theodorou, I.G.; Goode, A.E.; Gow, A.; Schwander, S.; Zhang, J.; Chung, K.F.; Tetley, T.D.; Shaffer, M.S.; Ryan, M.P.; et al. High-resolution analytical electron microscopy reveals cell culture media-induced changes to the chemistry of silver nanowires. Environ. Sci. Technol. 2013, 47, 13813–13821. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.J.; Shin, S. Toxic effects of silver nanoparticles and nanowires on erythrocyte rheology. Food Chem. Toxicol. 2014, 67, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Silva, R.M.; Xu, J.; Saiki, C.; Anderson, D.S.; Franzi, L.M.; Vulpe, C.D.; Gilbert, B.; Van Winkle, L.S.; Pinkerton, K.E. Short versus long silver nanowires: A comparison of in vivo pulmonary effects post instillation. Part. Fibre Toxicol. 2014, 11. [Google Scholar] [CrossRef] [PubMed]
- Sweeney, S.; Theodorou, I.G.; Zambianchi, M.; Chen, S.; Gow, A.; Schwander, S.; Zhang, J.; Chung, K.F.; Shaffer, M.S.P.; Ryan, M.P.; et al. Silver nanowire interactions with primary human alveolar type-II epithelial cell secretions: Contrasting bioreactivity with human alveolar type-I and type-II epithelial cells. Nanoscale 2015, 7, 10398–10409. [Google Scholar] [CrossRef] [PubMed]
- Cui, R.; Chae, Y.; An, Y.J. Dimension-dependent toxicity of silver nanomaterials on the cladocerans Daphnia magna and Daphnia galeata. Chemosphere 2017, 185, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Sohn, E.K.; Johari, S.A.; Kim, T.G.; Kim, J.K.; Kim, E.; Lee, J.H.; Chung, Y.S.; Yu, I.J. Aquatic toxicity comparison of silver nanoparticles and silver nanowires. Biomed. Res. Int. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Kwak, J.I.; Park, J.W.; An, Y.J. Effects of silver nanowire length and exposure route on cytotoxicity to earthworms. Environ. Sci. Pollut. Res. 2017, 24, 14516–14524. [Google Scholar] [CrossRef] [PubMed]
- Zaman, H.U.; Islam, J.M.M.; Khan, M.A.; Khan, R.A. Physico-mechanical properties of wound dressing material and its biomedical application. J. Mech. Behav. Biomed. Mater. 2011, 4, 1369–1375. [Google Scholar] [CrossRef] [PubMed]
- De Mori, A.; Peña, M.; Blunn, G.; Tozzi, G.; Roldo, M. 3D Printing and Electrospinning of Composite Hydrogels for Cartilage and Bone Tissue Engineering. Polymers 2018, 10, 285. [Google Scholar] [CrossRef]
- Tozzi, G.; De Mori, A.; Oliveira, A.; Roldo, M. Composite Hydrogels for Bone Regeneration. Materials 2016, 9, 267. [Google Scholar] [CrossRef] [PubMed]


| Hard-Template Synthesis | ||||
| AgNW Diameter | AgNW Length | Microbes Examined | Key Findings | Reference |
| 7 nm | 50 nm—1 µm | Not tested | Microporous silica (SBA-15) template Length controlled via loading percentage, annealing temp/time | [21] |
| 7–8 nm | over 1 µm | Not tested | Microporous silica (SBA-15) template Length controlled by reduction time | [22] |
| 7 nm | up to 4 µm | Not tested | Surface modification of SBA-15 SBA-15 synthesis at low temp, AgNWs must be capped | [23] |
| Soft-Template Synthesis | ||||
| AgNW Diameter | AgNW Length | Microbes Examined | Key Findings | Reference |
| 30–40 nm | 2–50 µm | Not tested | Temperature and seed number important | [24] |
| 30–40 nm | Not stated | Not tested | Pt nanoparticle seeding AgNO3 and PVP added dropwise | [25] |
| 45–60 nm | 2–5 µm | Not tested | Self-seeding with syringe pump; PVP to AgNO3 molar ratio critical; PVP molecular weight important | [26] |
| 100 nm | 10–50 µm | Not tested | Addition of CuCl2 or CuCl important | [27] |
| 170–310 nm | Not stated | Not tested | Solvothermal method with H2S Controllable diameters by H2S concentration | [28] |
| 50 nm | 20 µm | Not tested | NaCl instead of CuCl2 | [29] |
| 30–70 nm | 1–25 µm | Not tested | Altering temp and time controls size of generated AgNWs | [30] |
| 30–50 nm 80 nm | Not stated | Not tested | 100° C w/o PVP 200° C w/o PVP | [31] |
| 60–90 nm | 6–12 µm | Not tested | Glycerol substituted for ethylene glycol (green process) | [32] |
| Antibacterial Activity of AgNWs Synthesised by Soft-Template Methods | ||||
| AgNW Diameter | AgNW Length | Microbes Examined | Key Findings | Reference |
| 70–150 nm | 3–8 µm | E. coli MC1061 (pSLlux) | Bioluminscent recombinant E. coli employed No shape-dependent AgNP toxicity | [33] |
| 60 nm | 2–4 µm | E. coli ATCC 25922 | Shape of AgNPs dictates contact Exposed facet type important | [34] |
| 50–100 nm | 1–20 µm | S. aureus DSMZ 1104 | Particle morphology dictates dissolution and inhibition | [35] |
| 50 nm | 10–100 µm | E. coli K-12 S. aureus | AgNWs added to graphene oxide (GO) sheets Enhanced activity when GO and AgNW combined Slower release of Ag+/enhanced ROS | [36] |
| Emerging Applications for AgNWs | ||||
| AgNW Diameter | AgNW Length | Microbes Examined | Key Findings | Reference |
| 40–50 nm | Not stated | E. coli ATCC 1399 S. aureus ATCC 1431 | Drip and dry coating of cotton fabric Total bacterial elimination | [37] |
| 70–100 nm | up to 10 µm | E. coli ATCC 23282 S. aureus ATCC 35696 | Electrospun fibres of AgNW-PVA better than AgNW alone Better against S. aureus than E. coli | [38] |
| 60 nm | 7–12 µm | E. coli B. subtilis | Production of chitosan-AgNW hybrid films Better against B. subtilis than E. coli | [39] |
| 30–40 nm | more than 50 µm | E. coli S. aureus | Plant-based renewable polysaccharide, KGM-AgNW film Better against S. aureus than E. coli | [40] |
| 100 nm | 10–50 µm | E. coli S. aureus | AgNW-loaded PDMS films Better against E. coli than S. aureus | [41] |
| Arrays 20 nm thick | E. coli DBM 3138 S. aureus DBM 3179 | AgNW-PEN arrays | [42] | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jones, R.S.; Draheim, R.R.; Roldo, M. Silver Nanowires: Synthesis, Antibacterial Activity and Biomedical Applications. Appl. Sci. 2018, 8, 673. https://doi.org/10.3390/app8050673
Jones RS, Draheim RR, Roldo M. Silver Nanowires: Synthesis, Antibacterial Activity and Biomedical Applications. Applied Sciences. 2018; 8(5):673. https://doi.org/10.3390/app8050673
Chicago/Turabian StyleJones, Richard S., Roger R. Draheim, and Marta Roldo. 2018. "Silver Nanowires: Synthesis, Antibacterial Activity and Biomedical Applications" Applied Sciences 8, no. 5: 673. https://doi.org/10.3390/app8050673
APA StyleJones, R. S., Draheim, R. R., & Roldo, M. (2018). Silver Nanowires: Synthesis, Antibacterial Activity and Biomedical Applications. Applied Sciences, 8(5), 673. https://doi.org/10.3390/app8050673





