Estimation of Suspended Matter, Organic Carbon, and Chlorophyll-a Concentrations from Particle Size and Refractive Index Distributions
Abstract
1. Introduction
2. Materials and Methods
2.1. Theory
2.1.1. Particle Mass Modelling from Apparent Density of Hydrated Matter
2.1.2. Particulate Organic Carbon and Chlorophyll-a Cell Volume Scaling
2.1.3. Refractive Index-Based Estimation of Particulate Organic Carbon and Chlorophyll-a
2.2. Methods
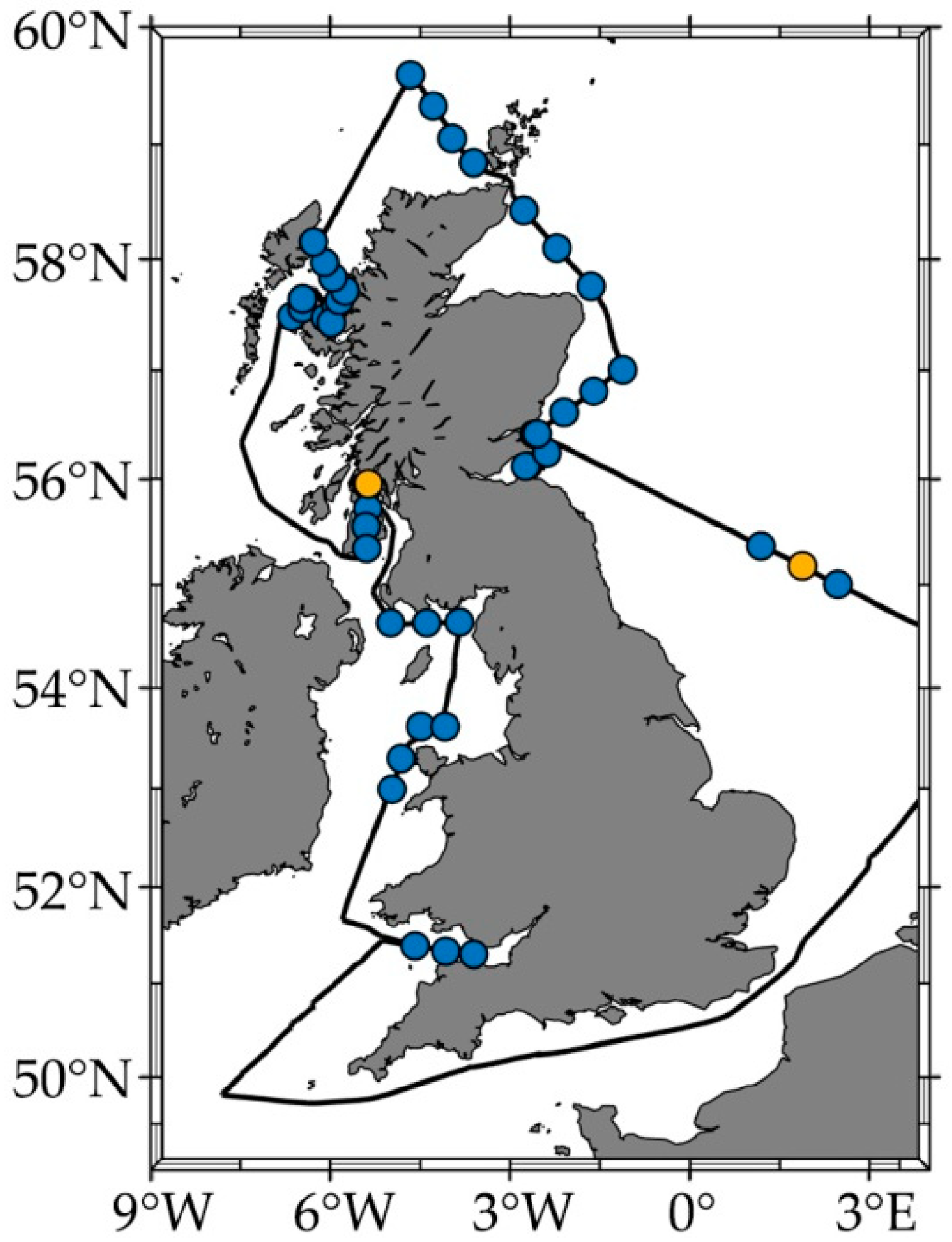
2.2.1. UK Coastal Waters (UKCW) Dataset
2.2.2. Depth Profiling
2.2.3. Flow Cytometry Measurement Protocol
2.2.4. PSD Extrapolations
2.2.5. Suspended Particulate Matter and Inorganic/Organic Suspended Matter
2.2.6. Particulate Organic Carbon
2.2.7. Chlorophyll-a
3. Results
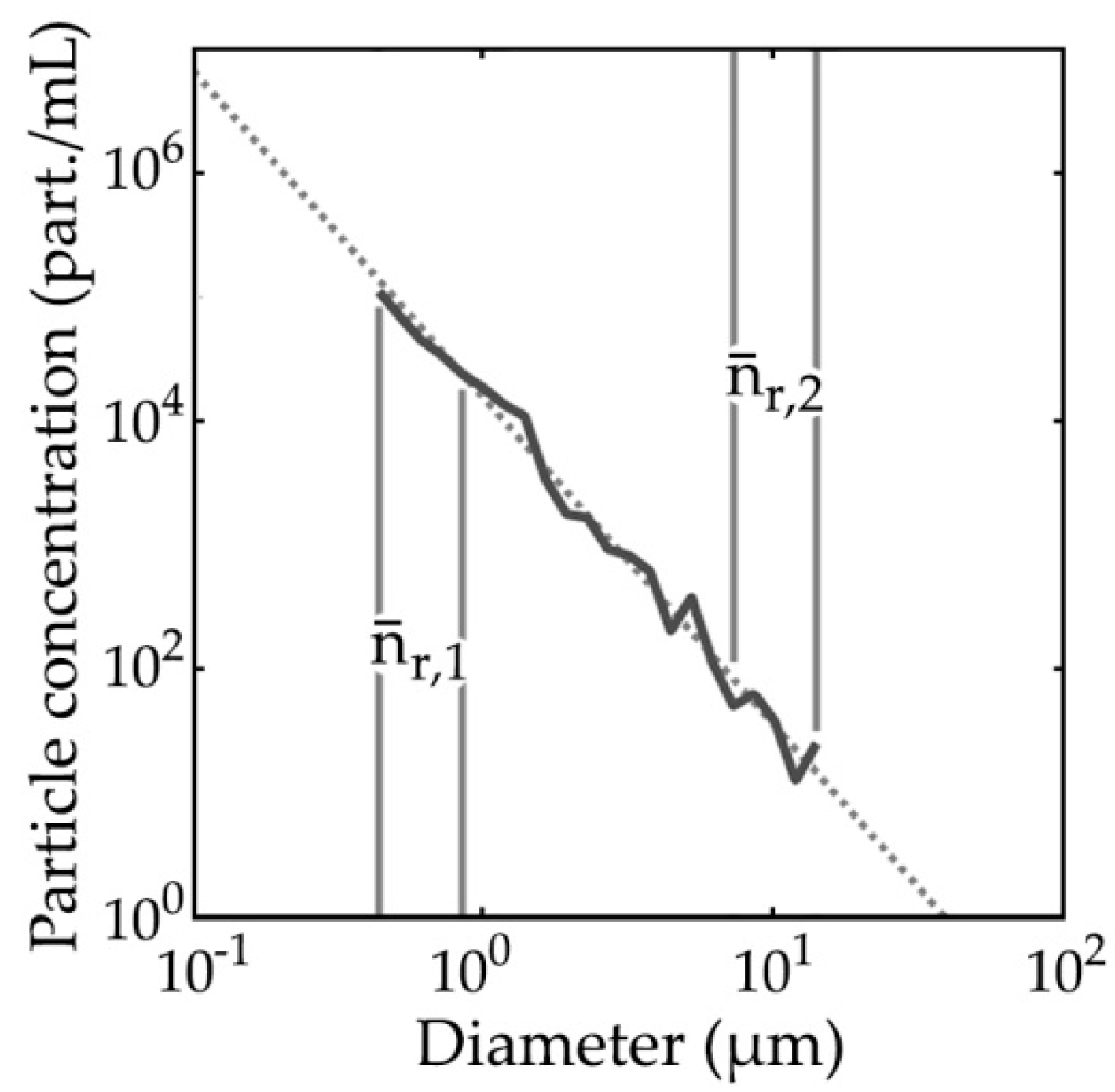
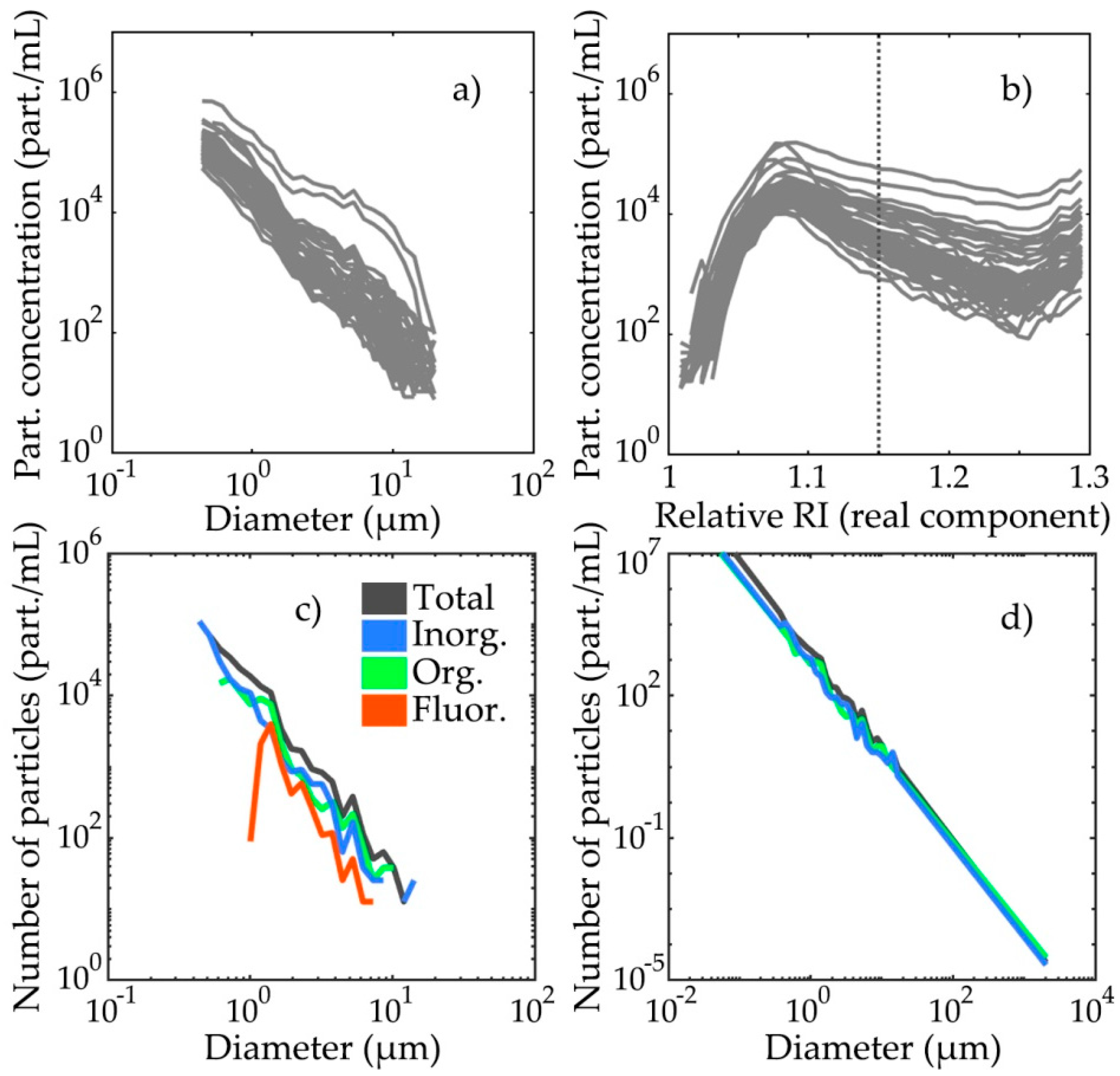
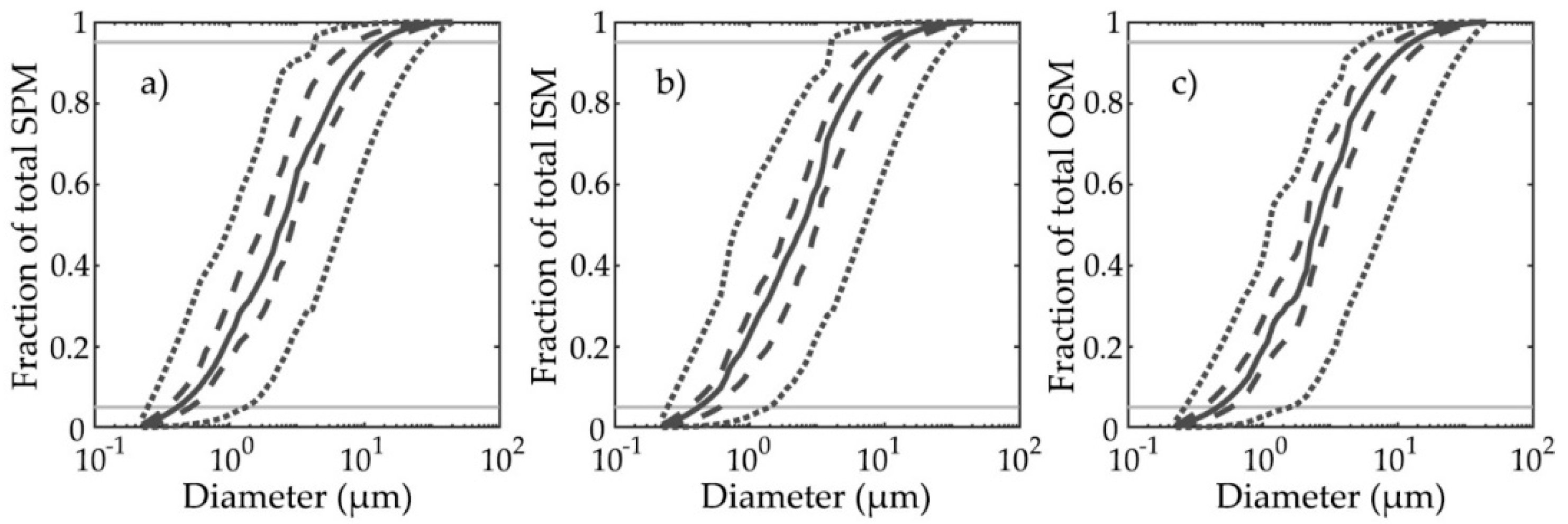
3.1. PSDs and PRIDs
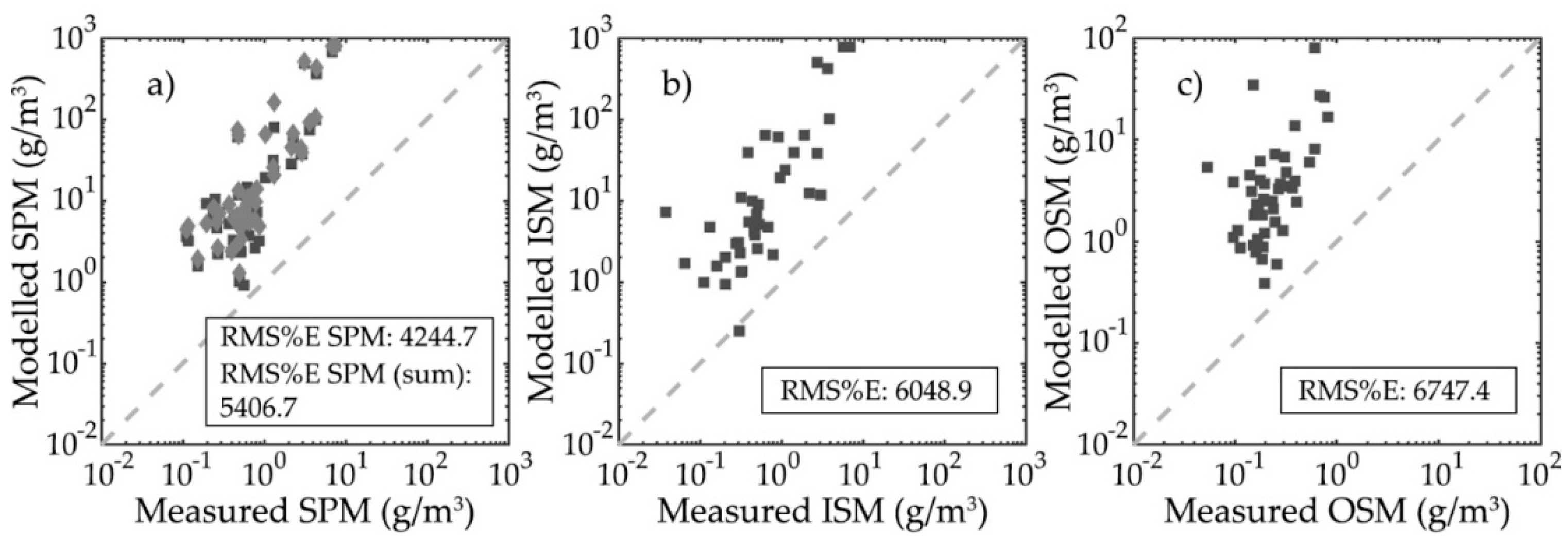
3.2. Particle Mass Modelling
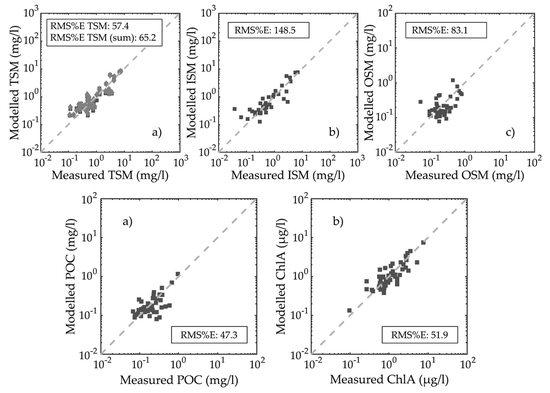
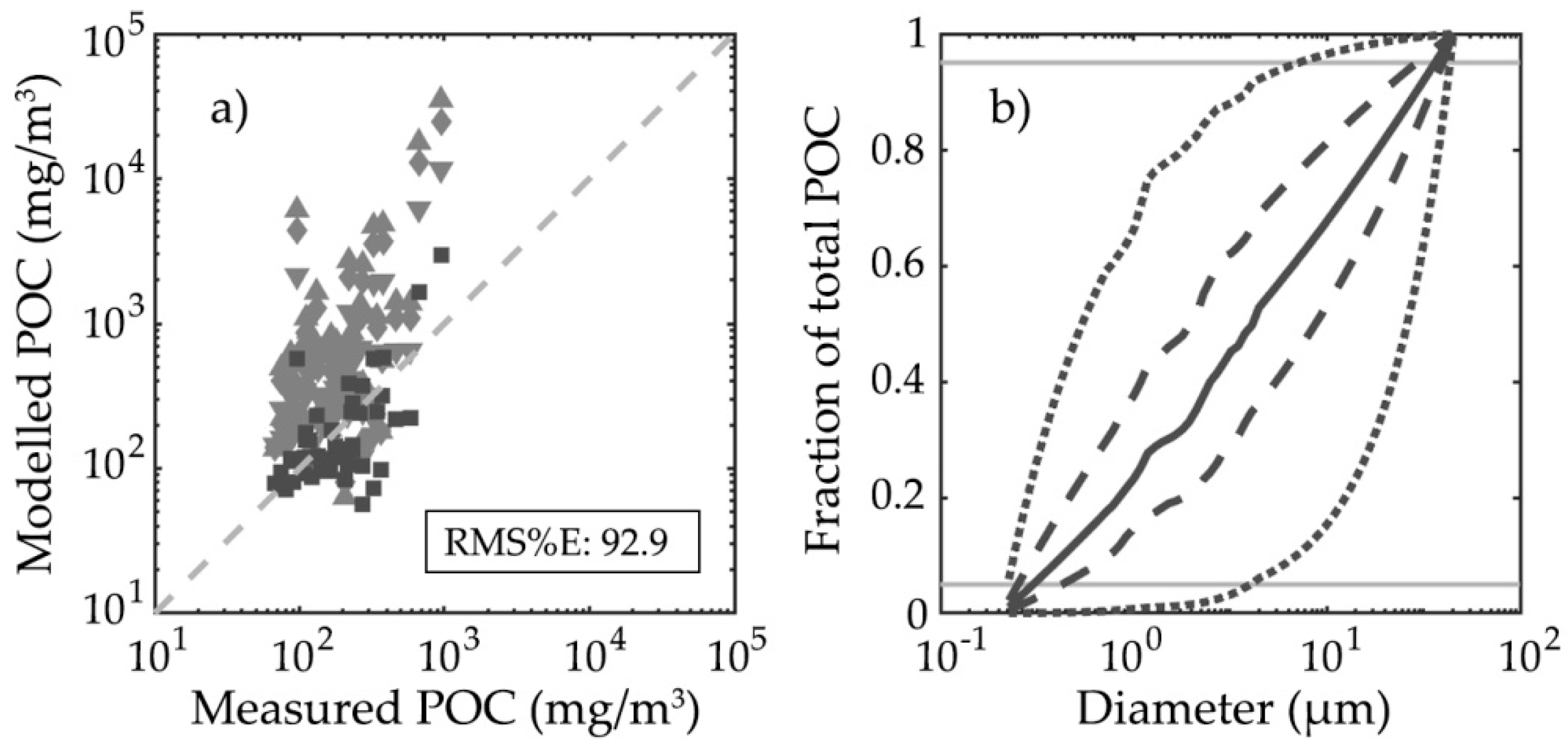
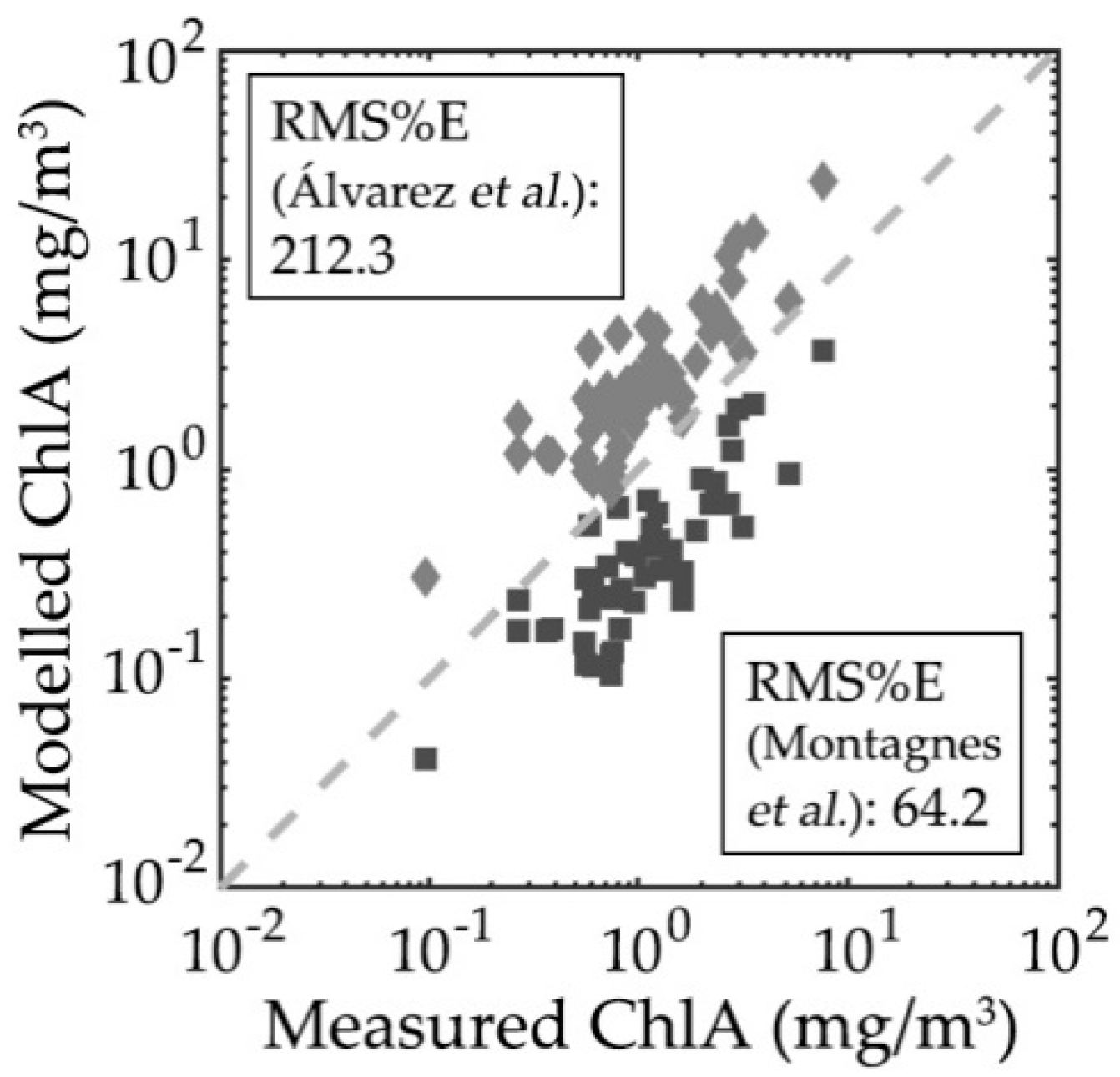
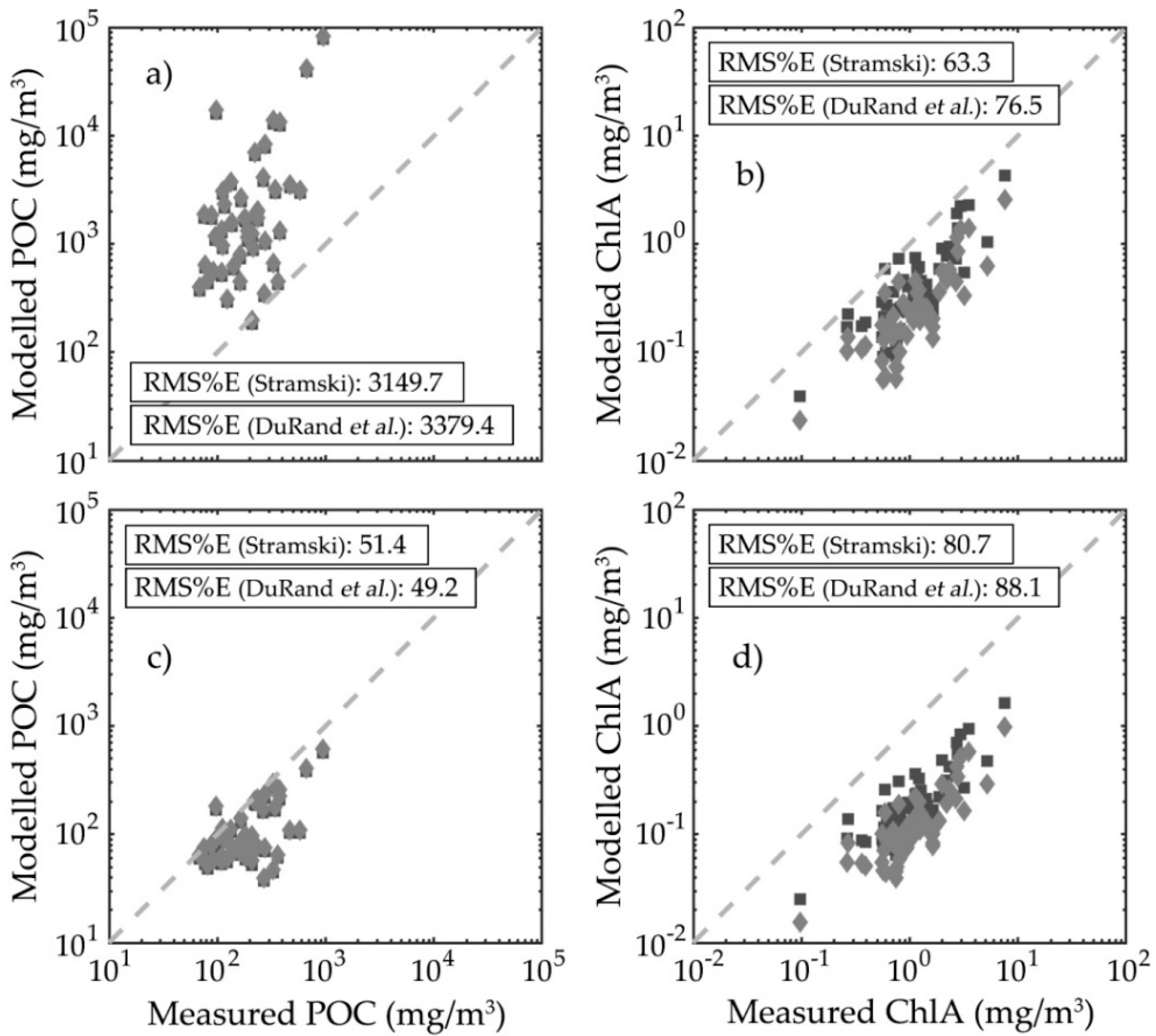
3.3. Particulate Organic Carbon and Chlorophyll-a Concentration Modelling
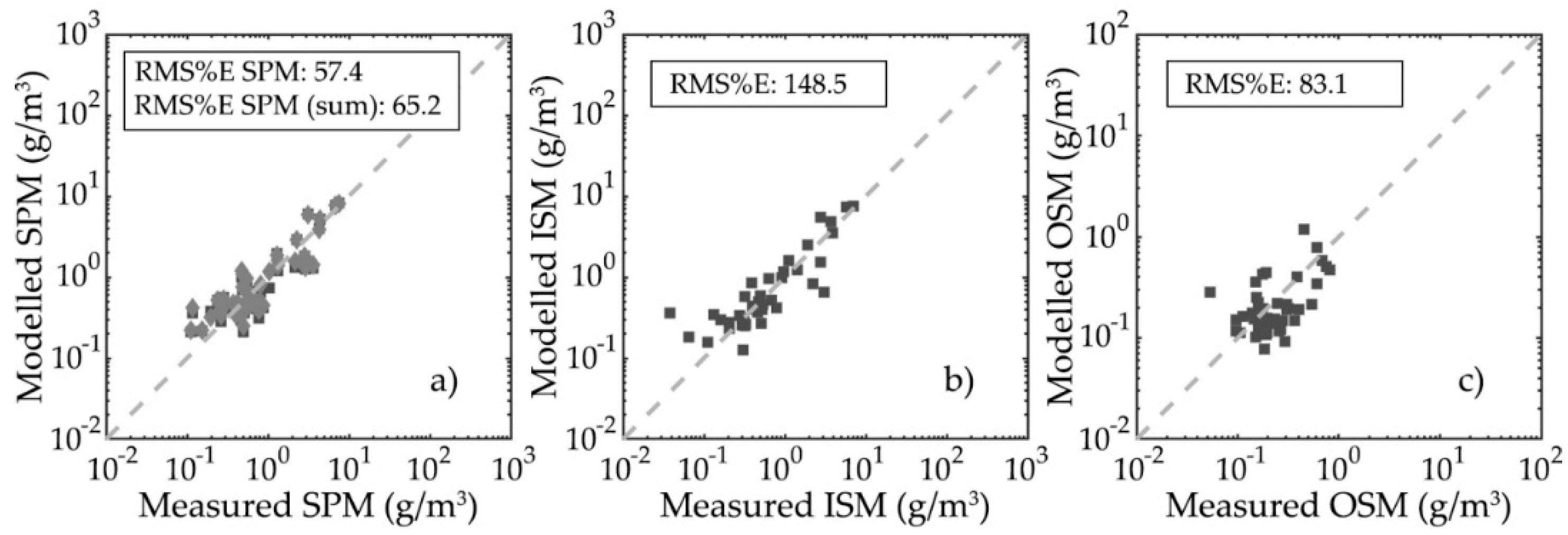
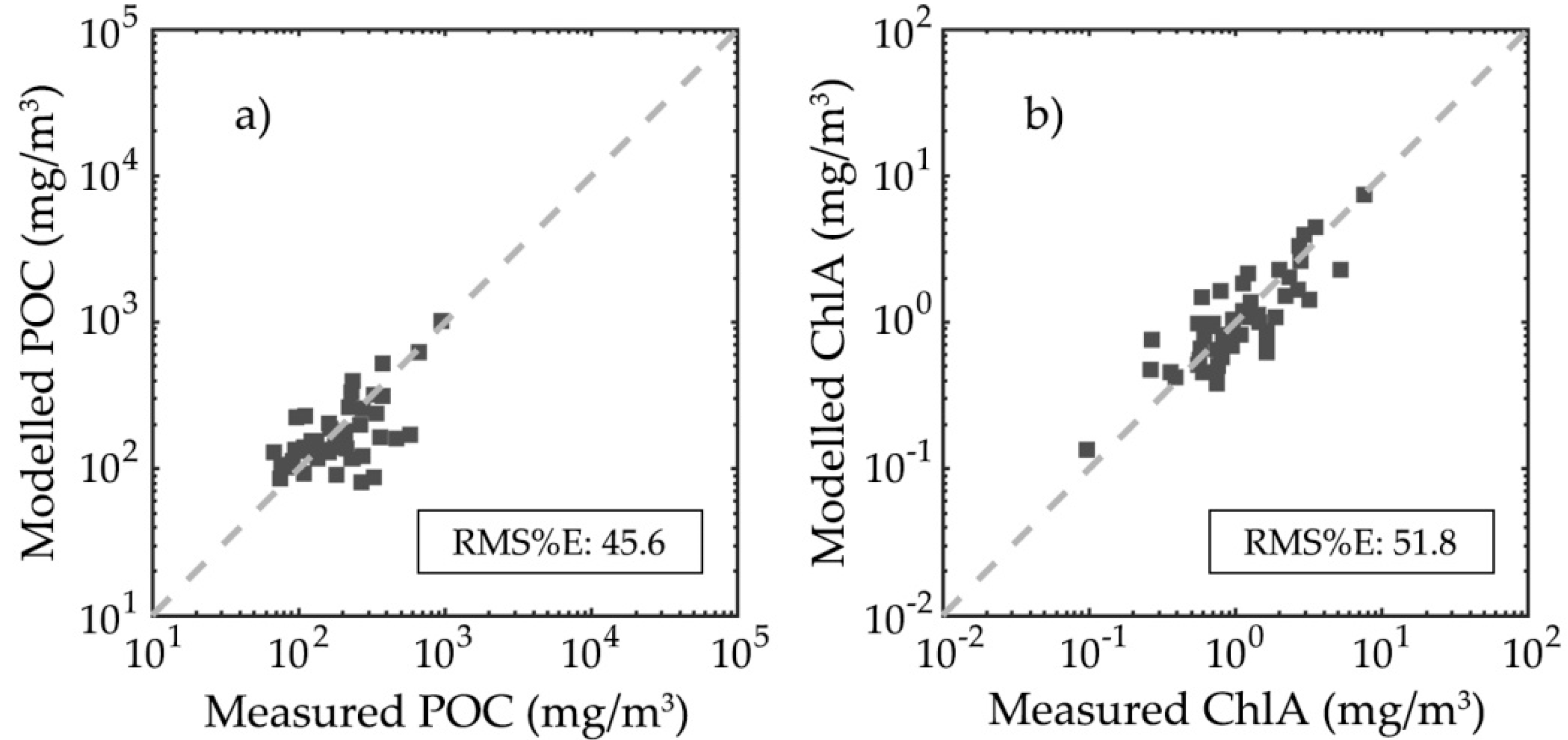
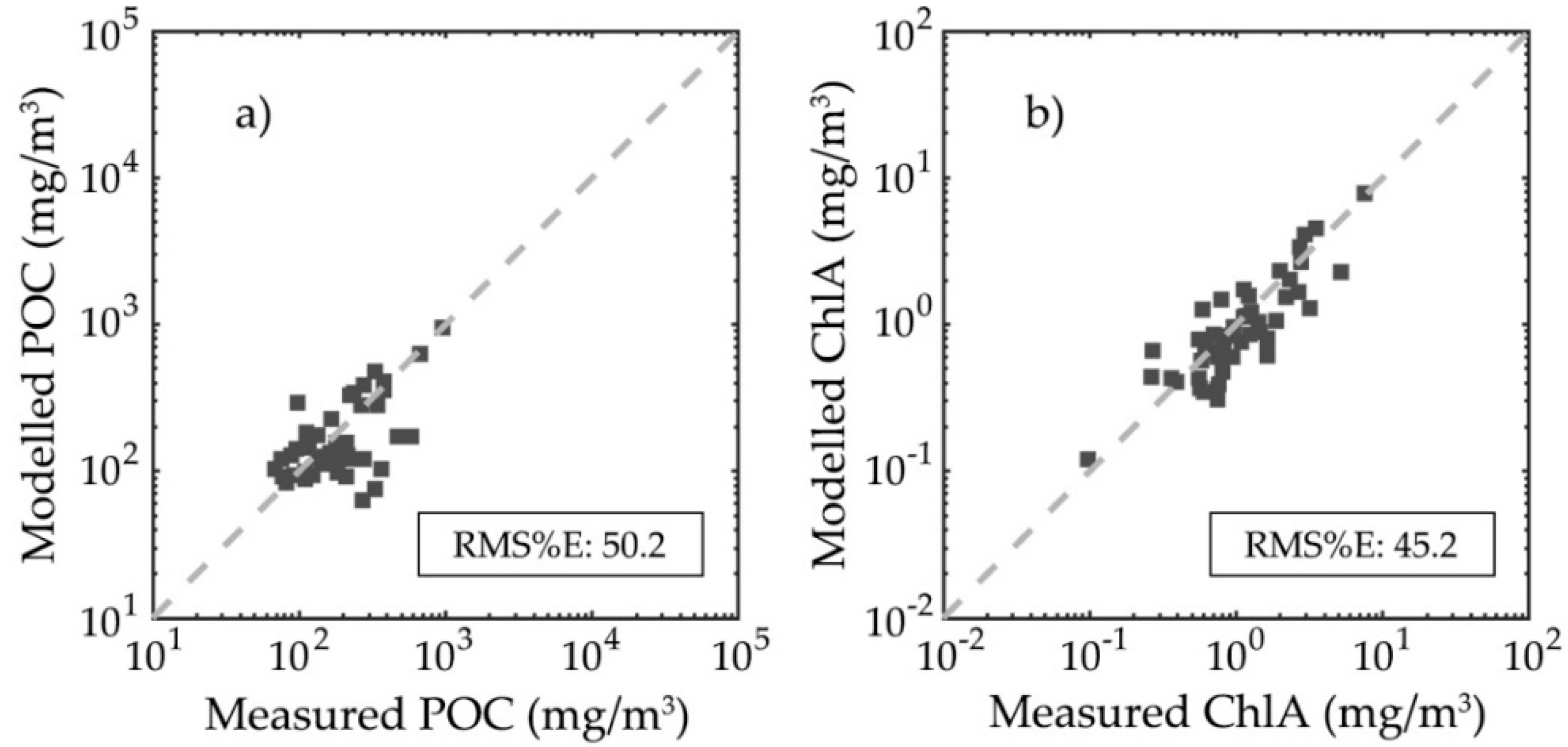
3.4. Particulate Organic Carbon and Chlorophyll-a Concentration Modelling Optimization
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Agagliate, J.; Röttgers, R.; Twardowski, M.S.; McKee, D. Evaluation of a flow cytometry method to determine size and real refractive index distributions in natural marine particle populations. Appl. Opt. 2018, 57, 1705–1716. [Google Scholar] [CrossRef] [PubMed]
- Agagliate, J.; Lefering, I.; McKee, D. Forward modelling of inherent optical properties from flow cytometry estimates of particle size and refractive index. Appl. Opt. 2018, 57, 1777–1788. [Google Scholar] [CrossRef] [PubMed]
- Ackleson, S.G.; Spinrad, R.W. Size and refractive index of individual marine particulates: A flow cytometric approach. Appl. Opt. 1988, 27, 1270–1277. [Google Scholar] [CrossRef] [PubMed]
- Green, R.E.; Sosik, H.M.; Olson, R.J.; DuRand, M.D. Flow cytometric determination of size and complex refractive index for marine particles: Comparison with independent and bulk estimates. Appl. Opt. 2003, 42, 526–541. [Google Scholar] [CrossRef] [PubMed]
- Green, R.E.; Sosik, H.M.; Olson, R.J. Contributions of phytoplankton and other particles to inherent optical properties in New England continental shelf waters. Limnol. Oceanogr. 2003, 48, 2377–2391. [Google Scholar] [CrossRef]
- Zhang, X.; Stavn, R.H.; Falster, A.U.; Gray, D.; Gould, R.W., Jr. New insight into particulate mineral and organic matter in coastal ocean waters through optical inversion. Estuar. Coast. Shelf Sci. 2014, 149, 1–12. [Google Scholar] [CrossRef]
- Morel, A.; Ahn, Y.-H. Optical efficiency factors of free-living marine bacteria: Influence of bacterioplankton upon the optical properties and particulate organic carbon in oceanic waters. J. Mar. Res. 1990, 48, 145–175. [Google Scholar] [CrossRef]
- Babin, M.; Morel, A.; Fournier-Sicre, V.; Fell, F.; Stramski, D. Light scattering properties of marine particles in coastal and open ocean waters as related to the particle mass concentration. Limnol. Oceanogr. 2003, 48, 843–859. [Google Scholar] [CrossRef]
- Aas, E. Refractive index of phytoplankton derived from its metabolite composition. J. Plankton Res. 1996, 18, 2223–2249. [Google Scholar] [CrossRef]
- Verity, P.G.; Robertson, C.Y.; Tronzo, C.R.; Andrews, M.G.; Nelson, J.R.; Sieracki, M.E. Relationships between cell volume and the carbon and nitrogen content of marine photosynthetic nanoplankton. Limnol. Oceanogr. 1992, 37, 1434–1446. [Google Scholar] [CrossRef]
- Montagnes, D.J.S.; Berges, J.A.; Harrison, P.J.; Taylor, F.J.R. Estimating carbon, nitrogen, protein, and chlorophyll a from volume in marine phytoplankton. Limnol. Oceanogr. 1994, 39, 1044–1060. [Google Scholar] [CrossRef]
- Menden-Deuer, S.; Lessard, E.J. Carbon to volume relationships for dinoflagellates, diatoms, and other protist plankton. Limnol. Oceanogr. 2000, 45, 569–579. [Google Scholar] [CrossRef]
- Álvarez, E.; Nogueira, E.; López-Urrutia, Á. In Vivo Single-Cell Fluorescence and Size Scaling of Phytoplankton Chlorophyll Content. Appl. Environ. Microbiol. 2017, 83, e03317-16. [Google Scholar] [CrossRef] [PubMed]
- Stramski, D.; Morel, A. Optical properties of photosynthetic picoplankton in different physiological states as affected by growth irradiance. Deep Sea Res. Part A Oceanogr. Res. Pap. 1990, 37, 245–266. [Google Scholar] [CrossRef]
- Stramski, D.; Reynolds, R.A. Diel variations in the optical properties of a marine diatom. Limnol. Oceanogr. 1993, 38, 1347–1364. [Google Scholar] [CrossRef]
- Stramski, D.; Shalapyonok, A.; Reynolds, R.A. Optical characterization of the oceanic unicellular cyanobacterium Synechococcus grown under a day-night cycle in natural irradiance. J. Geophys. Res. 1995, 100, 13295–13307. [Google Scholar] [CrossRef]
- Reynolds, R.A.; Stramski, D.; Kiefer, D.A. The effect of nitrogen limitation on the absorption and scattering properties of the marine diatom Thalassiosira pseudonana. Limnol. Oceanogr. 1997, 42, 881–892. [Google Scholar] [CrossRef]
- Durand, M.D.; Olson, R.J. Diel patterns in optical properties of the chlorophyte Nannochloris sp.: Relating individual-cell to bulk measurements. Limnol. Oceanogr. 1998, 43, 1107–1118. [Google Scholar] [CrossRef]
- Stramski, D. Refractive index of planktonic cells as a measure of cellular carbon and chlorophyll a content. Deep Sea Res. Part I Oceanogr. Res. Pap. 1999, 46, 335–351. [Google Scholar] [CrossRef]
- Durand, M.D.; Green, R.E.; Sosik, H.M.; Olson, R.J. Diel Variations in Optical Properties of Micromonas Pusilla (Prasinophyceae). J. Phycol. 2002, 38, 1132–1142. [Google Scholar] [CrossRef]
- Stramski, D.; Morel, A.; Bricaud, A. Modeling the light attenuation and scattering by spherical phytoplanktonic cells: A retrieval of the bulk refractive index. Appl. Opt. 1988, 27, 3954–3956. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, R.A.; Stramski, D.; Wright, V.M.; Woźniak, S.B. Measurements and characterization of particle size distributions in coastal waters. J. Geophys. Res. 2010, 115. [Google Scholar] [CrossRef]
- Davies, E.J.; McKee, D.; Bowers, D.; Graham, G.W.; Nimmo-Smith, W.A.M. Optically significant particle sizes in seawater. Appl. Opt. 2014, 53, 1067. [Google Scholar] [CrossRef] [PubMed]
- Bader, H. The hyperbolic distribution of particle sizes. J. Geophys. Res. 1970, 75, 2822–2830. [Google Scholar] [CrossRef]
- Sheldon, R.W.; Prakash, A.; Sutcliffe, W.H., Jr. The size distribution of particles in the ocean. Limnol. Oceanogr. 1972, 17, 327–340. [Google Scholar] [CrossRef]
- Junge, C.E. Air Chemistry and Radioactivity; Academic Press: New York, NY, USA, 1963; ISBN-13 9780123921505. [Google Scholar]
- Stramski, D.; Kiefer, D.A. Light scattering by microorganisms in the open ocean. Prog. Oceanogr. 1991, 28, 343–383. [Google Scholar] [CrossRef]
- Ulloa, O.; Sathyendranath, S.; Platt, T. Effect of the particle-size distribution on the backscattering ratio in seawater. Appl. Opt. 1994, 33, 7070. [Google Scholar] [CrossRef] [PubMed]
- Röttgers, R.; Heymann, K.; Krasemann, H. Suspended matter concentrations in coastal waters: Methodological improvements to quantify individual measurement uncertainty. Estuar. Coast. Shelf Sci. 2014, 151, 148–155. [Google Scholar] [CrossRef]
- Zapata, M.; Rodríguez, F.; Garrido, J.L. Separation of chlorophylls and carotenoids from marine phytoplankton: A new HPLC method using a reversed phase C8 column and pyridine-containing mobile phases. Mar. Ecol. Prog. Ser. 2000, 195, 29–45. [Google Scholar] [CrossRef]
- Risović, D. Two-component model of sea particle size distribution. Deep Sea Res. Part I Oceanogr. Res. Pap. 1993, 40, 1459–1473. [Google Scholar] [CrossRef]
- Twardowski, M.S.; Boss, E.; Macdonald, J.B.; Pegau, W.S.; Barnard, A.H.; Zaneveld, J.R.V. A model for estimating bulk refractive index from the optical backscattering ratio and the implications for understanding particle composition in case I and case II waters. J. Geophys. Res. 2001, 106, 14129–14142. [Google Scholar] [CrossRef]
- Khelifa, A.; Hill, P.S. Models for effective density and settling velocity of flocs. J. Hydraul. Res. 2006, 44, 390–401. [Google Scholar] [CrossRef]
- Morel, A.; Bricaud, A. Theoretical results concerning light absorption in a discrete medium, and application to specific absorption by phytoplankton. Deep Sea Res. Part A Oceanogr. Res. Pap. 1981, 28, 1375–1393. [Google Scholar] [CrossRef]
- Marañón, E.; Cermeño, P.; Rodríguez, J.; Zubkov, M.V.; Harris, R.P. Scaling of phytoplankton photosynthesis and cell size in the ocean. Limnol. Oceanogr. 2007, 52, 2190–2198. [Google Scholar] [CrossRef]
- Stramski, D.; Bricaud, A.; Morel, A. Modeling the inherent optical properties of the ocean based on the detailed composition of the planktonic community. Appl. Opt. 2001, 40, 2929–2945. [Google Scholar] [CrossRef] [PubMed]
- Finkel, Z.V.; Irwin, A.J.; Schofield, O. Resource limitation alters the 3/4 size scaling of metabolic rates in phytoplankton. Mar. Ecol. Prog. Ser. 2004, 273, 269–279. [Google Scholar] [CrossRef]
- Mei, Z.P.; Finkel, Z.V.; Irwin, A.J. Light and nutrient availability affect the size-scaling of growth in phytoplankton. J. Theor. Biol. 2009, 259, 582–588. [Google Scholar] [CrossRef] [PubMed]
| Notation | Definition |
|---|---|
| ChlA | Chlorophyll-a concentration, mg m−3 |
| FC | Flow cytometer; flow cytometry |
| IOP | Inherent optical property |
| ISM | Inorganic suspended matter, g m−3 |
| OSM | Organic suspended matter, g m−3 |
| SPM | Suspended particulate matter, g m−3 |
| POC | Particulate organic carbon, mg m−3 |
| PRID | Particle real refractive index distribution |
| PSD | Particle size distribution |
| RMS%E | Root-mean-square percentage error |
| RMSE | Root-mean-square error |
| UKCW | UK coastal waters (dataset) |
| a, aC, achl | Slopes of the refractive index-based models and of the of the Ci and Chli optimized refractive index-based models respectively, kg m−3 |
| b | y-intercepts of the refractive index-based models, kg m−3 |
| F(r) | Fractal dimension, dimensionless |
| h1, h2 | Power law exponents of the optimized POC and ChlA volume scaling functions respectively, dimensionless |
| k | Particle size distribution scaling coefficient, mL−1 |
| k1, k2 | Scaling coefficients of the optimized POC and ChlA volume scaling functions respectively, pg µm−3 |
| mtot | Total particle mass, mg |
| Ci | Intraparticle carbon concentration, kg m−3 |
| Chli | Intraparticle chlorophyll-a concentration, kg m−3 |
| N(D) | Number concentration of particles within particle size bin corresponding to particle diameter D, mL−1 |
| N’(D) | Density function of the particle size distribution, mL−1 µm−1 |
| Nr | Number of particles within particle size bin corresponding to particle radius r, dimensionless |
| ND,nr | Number of particles within particle bin corresponding to particle diameter D and real refractive index nr, dimensionless |
| no | Real refractive index of the dry matter fraction of the particle, dimensionless |
| nr | Real refractive index of the particle, dimensionless |
| ni | Imaginary refractive index of the particle, dimensionless |
| Average real refractive indices at the upper and lower extremes of the particle size distribution respectively, dimensionless | |
| r, D | Particle radius and particle diameter, µm |
| ro | Primary particle radius, µm |
| VD | Particle volume, µm3 |
| Vo | Volume of the dry matter fraction of the particle, µm3 |
| y(r) | Volume scaling function |
| B | Fractal dimension exponent, dimensionless |
| γ | Power law slope, dimensionless |
| P | Particle density, g/m3 |
| ρnr | Density of a particle with real refractive index nr, g/m3 |
| ρo | Density of the dry matter fraction of the particle, g/m3 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Agagliate, J.; Röttgers, R.; Heymann, K.; McKee, D. Estimation of Suspended Matter, Organic Carbon, and Chlorophyll-a Concentrations from Particle Size and Refractive Index Distributions. Appl. Sci. 2018, 8, 2676. https://doi.org/10.3390/app8122676
Agagliate J, Röttgers R, Heymann K, McKee D. Estimation of Suspended Matter, Organic Carbon, and Chlorophyll-a Concentrations from Particle Size and Refractive Index Distributions. Applied Sciences. 2018; 8(12):2676. https://doi.org/10.3390/app8122676
Chicago/Turabian StyleAgagliate, Jacopo, Rüdiger Röttgers, Kerstin Heymann, and David McKee. 2018. "Estimation of Suspended Matter, Organic Carbon, and Chlorophyll-a Concentrations from Particle Size and Refractive Index Distributions" Applied Sciences 8, no. 12: 2676. https://doi.org/10.3390/app8122676
APA StyleAgagliate, J., Röttgers, R., Heymann, K., & McKee, D. (2018). Estimation of Suspended Matter, Organic Carbon, and Chlorophyll-a Concentrations from Particle Size and Refractive Index Distributions. Applied Sciences, 8(12), 2676. https://doi.org/10.3390/app8122676