Phase Change Material Selection for Thermal Processes Working under Partial Load Operating Conditions in the Temperature Range between 120 and 200 °C
Abstract
:1. Introduction
2. Materials Description
3. Methodology
3.1. Health Hazard
3.2. Thermal Stability
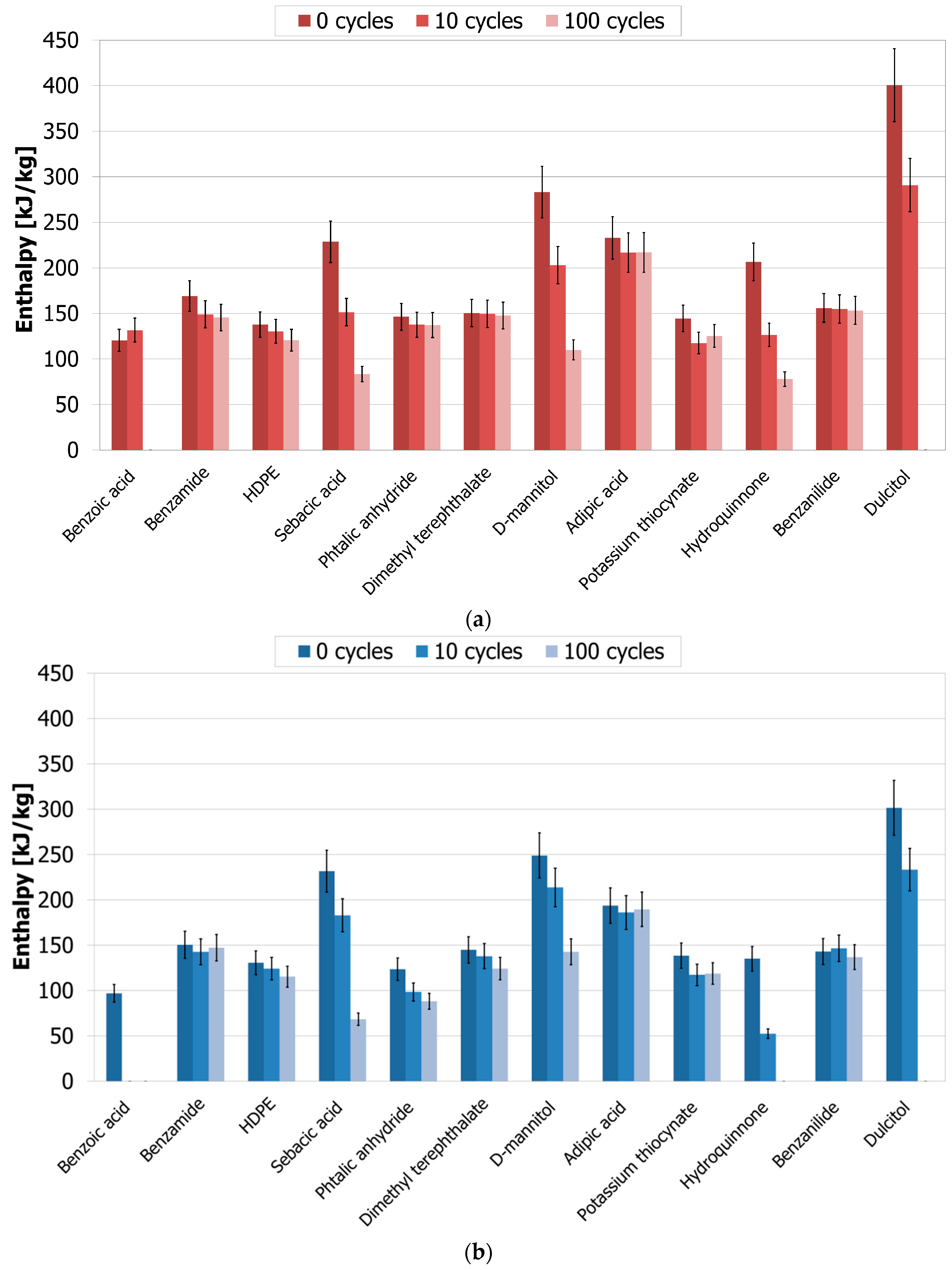
3.3. Cycling Stability
- Measurement 1: 0 cycle;
- Measurement 2: 10th cycle;
- Measurement 3: 100th cycle.
4. Results
4.1. Health Hazard
4.2. Thermal Stability
4.3. Cycling Stability
4.3.1. Thermophysical Characterization (Phase Change Temperature and Enthalpy)
4.3.2. Chemical Characterization
5. General Discussion
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zalba, B.; Marı́n, J.M.; Cabeza, L.F.; Mehling, H. Review on thermal energy storage with phase change: Materials, heat transfer analysis and applications. Appl. Therm. Eng. 2003, 23, 251–283. [Google Scholar] [CrossRef]
- Sharma, A.; Tyagi, V.V.; Chen, C.R.; Buddhi, D. Review on thermal energy storage with phase change materials and applications. Renew. Sustain. Energy Rev. 2009, 13, 318–345. [Google Scholar] [CrossRef]
- Hasnain, S.M. Review on sustainable thermal energy storage technologies; Part I: Heat storage materials and techniques. Energy Convers. Manag. 1998, 39, 1127–1138. [Google Scholar] [CrossRef]
- Gasia, J.; Miró, L.; Cabeza, L.F. Materials and system requirements of high temperature thermal energy storage systems: A review; Part 1: General requirements. Renew. Sustain. Energy Rev. 2017, 75, 1320–1601. [Google Scholar] [CrossRef]
- Bayón, R.; Coco, S.; Barcenilla, M.; Espinet, P.; Imbuluzqueta, G. Feasibility of Storing Latent Heat with Liquid Crystals: Proof of Concept at Lab Scale. Appl. Sci. 2016, 6, 120. [Google Scholar] [CrossRef]
- Del Barrio, E.P.; Godin, A.; Duquesne, M.; Daranlot, J.; Jolly, J.; Alshaer, W.; Kouadio, T.; Sommier, A. Characterization of different sugar alcohols as phase change materials for thermal energy storage applications. Sol. Energ. Mater. Sol. Cells 2017, 159, 560–569. [Google Scholar] [CrossRef]
- Bauer, D.T.; Kröner, U.; Tamme, R. Thermal analysis of phase change materials in the temperature range 120–150 °C. Thermochim. Acta 2011, 513, 49–59. [Google Scholar]
- Miró, L.; Barreneche, C.; Ferrer, G.; Solé, A.; Martorell, I.; Cabeza, L.F. Health hazard, cycling and thermal stability as key parameters when selecting a suitable phase change material (PCM). Thermochim. Acta 2016, 627–629, 39–47. [Google Scholar] [CrossRef]
- Buschle, J.; Steinmann, W.D.; Tamme, R. Latent heat storage for process heat applications. In Proceedings of the 10th International Conference on Thermal Energy Storage ECOSTOCK, Atlantic City, NJ, USA, 31 May–2 June 2006. [Google Scholar]
- Ge, H.; Li, H.; Mei, S.; Liu, J. Low melting point liquid metal as a new class of phase change material: An emerging frontier in energy area. Renew. Sustain. Energy Rev. 2013, 21, 331–346. [Google Scholar] [CrossRef]
- Solé, A.; Neumann, H.; Niedermaier, S.; Martorell, I.; Schossig, P.; Cabeza, L.F. Stability of sugar alcohols as PCM for thermal energy storage. Sol. Energ. Mater. Sol. Cells 2014, 126, 125–134. [Google Scholar]
- Oró, E.; Gil, A.; Miró, L.; Peiró, G.; Álvarez, S.; Cabeza, L.F. Thermal energy storage implementation using phase change materials for solar cooling and refrigeration applications. Energy Procedia 2012, 30, 947–956. [Google Scholar]
- Waschull, J.; Müller, R.; Römer, S. Investigation of phase change materials for elevated temperatures. In Proceedings of the 11th International Conference on Thermal Energy Storage EFFSTOCK, Stockholm, Sweden, 14–17 June 2009. [Google Scholar]
- Martin, J.M.; Solé, A.; Barreneche, C.; Cabeza, L.F. Dataset on thermal and cycling stability tests of phase change materials within 120–200 °C temperature range. Data Brief 2017. submitted for publication. [Google Scholar]
- Available online: https://www.sciencelab.com (accessed on 16 January 2017).
- Available online: http://pubchem.ncbi.nlm.nih.gov/compound (accessed on 16 January 2017).
- Rathgeber, C.; Miró, L.; Cabeza, L.F.; Hiebler, S. Measurement of enthalpy curves of phase change materials via DSC and T-History: When are both methods needed to estimate the behaviour of the bulk material in applications. Thermochim. Acta 2014, 596, 79–88. [Google Scholar] [CrossRef]
- Sharma, S.D. Latent heat storage materials and systems: A review. Int. J. Green Energy 2005, 2, 1–56. [Google Scholar] [CrossRef]



| Thermal Process | Range of Temperatures |
|---|---|
| Absorption refrigeration | From 80 to 230 °C |
| Adsorption refrigeration | From −60 to 350 °C |
| Transportation exhaust heat recovery | From 55 to 800 °C |
| Solar cooling | From 60 to 250 °C |
| Industrial waste heat recovery | From 30 to 1600 °C |
| No. | Material | Material Type | Thermophysical Properties | Manufacturer Information | |||||
|---|---|---|---|---|---|---|---|---|---|
| Phase Change Temperature (°C) | Phase Change Enthalpy (kJ/kg) | Manufacturer | Purity (%) | ||||||
| Value | Ref. | Value | Ref. | ||||||
| 1 | Benzoic acid | Aromatic hydrocarbon | 121–123 | [13] | 114–147 | [13] | PanReac AppliChem, Barcelona, Spain | >99.5 | |
| 2 | Benzamide | Aromatic hydrocarbon | 124–127 | [13] | 169 | [13] | Alfa Aesar, Ward Hill, MA, USA | >98 | |
| 3 | High density polyethylene (HDPE) | Polymeric hydrocarbon | 130 | [13] | 211–233 | [13] | Alfa Aesar, Ward Hill, MA, USA | n.a. | |
| 4 | Sebacic acid | Dicarboxylic acid | 130–134 | [13] | 228 | [13] | Alfa Aesar, Ward Hill, MA, USA | >98 | |
| 5 | Phtalic anhydride | Dicarboxylic acid anhydride | 131 | [13] | 159 | [13] | Alfa Aesar, Ward Hill, MA, USA | 99 | |
| 6 | Maleic acid | Dicarboxylic acid | 131–140 | [13] | 235 | [13] | PanReac AppliChem, Barcelona, Spain | >99 | |
| 7 | Urea | Organic compound | 133–135 | [13] | 170–258 | [13] | PanReac AppliChem, Barcelona, Spain | >99 | |
| 8 | Dimethyl terephthalate | Aromatic hydrocarbon | 142 | [13] | 170 | [13] | Alfa Aesar, Ward Hill, MA, USA | 99 | |
| 9 | D-mannitol | Sugar alcohol | 150 | [11] | 224–234 | [11] | Sigma-Aldrich, St. Louis, MO, USA | 98 | |
| 10 | Adipic acid | Dicarboxylic acid | 150–152 | [9] | 213–260 | [9] | Sigma-Aldrich, St. Louis, MO, USA | >99.5 | |
| 11 | Salycilic acid | phenolic acid | 157–159 | [2] | 199 | [2] | Sigma-Aldrich, St. Louis, MO, USA | >99 | |
| 12 | Potassium thiocynate | Inorganic salt | 157–177 | [12] | 112–114 | [12] | Sigma-Aldrich, St. Louis, MO, USA | >99 | |
| 13 | Hydroquinnone | Aromatic hydrocarbon | 160–173 | [12] | 179–235 | [12] | Sigma-Aldrich, St. Louis, MO, USA | >99 | |
| 14 | Benzanilide | Amide | 161 | [12] | 129–139 | [12] | Sigma-Aldrich, St. Louis, MO, USA | 98 | |
| 15 | Dulcitol | Sugar alcohol | 167–185 | [9,13] | 246–257 | [9,13] | Sigma-Aldrich, St. Louis, MO, USA | 99 | |
| 16 | 2,2-Bis(hydroxymethyl) propionic acid (DMPA) | Carboxylic acid | 185 | [7] | 289 | [7] | Sigma-Aldrich, St. Louis, MO, USA | 98 | |
| Material | NFPA 704 | GHS Statement |
|---|---|---|
| HDPE | 0 | This material is not hazardous |
| Sebacic acid | 1 | Causes skin irritation |
| Causes serious eye irritation | ||
| May cause respiratory irritation | ||
| Dimethyl terephthalate | 1 | May cause an allergic skin reaction |
| D-mannitol | 1 | Hazardous in case of ingestion. Slightly hazardous in case of skin contact (irritant), of eye contact (irritant) or inhalation |
| Adipic acid | 1 | Causes serious eye irritation |
| Benzanilide | 1 | Harmful if swallowed |
| May cause respiratory irritation | ||
| Causes skin irritation | ||
| Causes serious eye irritation | ||
| Dulcitol | 1 | Hazardous in case of ingestion. Slightly hazardous in case of skin contact (irritant), of eye contact (irritant), of inhalation * |
| Benzoic acid | 2 | Harmful if swallowed |
| May cause respiratory irritation | ||
| Causes skin irritation | ||
| Causes serious eye irritation | ||
| Causes damage to organs through prolonged or repeated exposure | ||
| Causes serious eye damage | ||
| Benzamide | 2 | Harmful if swallowed |
| Suspected of causing genetic defects | ||
| Urea | 2 | May cause respiratory irritation |
| Causes skin irritation | ||
| Suspected of causing cancer | ||
| May cause damage to organs | ||
| Causes serious eye irritation | ||
| Salycilic acid | 2 | Toxic if swallowed |
| Causes serious eye irritation | ||
| Hydroquinnone | 2 | Harmful if swallowed |
| Harmful in contact with skin | ||
| Suspected of causing genetic defects | ||
| Suspected of causing cancer | ||
| Causes serious eye damage | ||
| May cause an allergic skin reaction | ||
| Phtalic anhydride | 3 | Harmful if swallowed |
| May cause respiratory irritation | ||
| Causes skin irritation | ||
| May cause an allergic skin reaction | ||
| Causes serious eye damage | ||
| May cause allergy or asthma symptoms or breathing difficulties if inhaled | ||
| Maleic acid | 3 | Harmful if swallowed |
| Harmful in contact with skin | ||
| May cause respiratory irritation | ||
| Causes skin irritation | ||
| Causes serious eye irritation | ||
| May cause an allergic skin reaction | ||
| Potassium thiocynate | 3 | Harmful if swallowed |
| Harmful in contact with skin | ||
| Harmful if inhaled | ||
| Causes serious eye irritation | ||
| DMPA | ** | Causes serious eye irritation |
| May cause respiratory irritation |
| Material | Maximum Thermal-Stable Temperature (°C) | Final Degradation Temperature (°C) |
|---|---|---|
| Potassium thiocynate | 540 | >600 |
| HDPE | 309 | 540 |
| Dulcitol | 293 | 481 |
| D-mannitol | 259 | 424 |
| Adipic acid | 203 | 379 |
| Benzanilide | 196 | 315 |
| DMPA | 190 | 431 |
| Hydroquinone | 157 | 240 |
| Urea | 148 | 500 |
| Maleic acid | 141 | 212 |
| Benzamide | 138 | 225 |
| Salycilic acid | 133 | 203 |
| Phtalic anhydride | 129 | 210 |
| Dimethyl terephthalate | 128 | 265 |
| Benzoic acid | 121 | 195 |
| Sebacic acid | 118 | 201 |
| Material | Health Hazard | Cycling Stability (after 100 Cycles) | Thermal Stability | Suitable Material for Partial Load Applications | |
|---|---|---|---|---|---|
| Phase Change Enthalpy Loss (%) | Chemical Degradation a | Maximum Thermal-Stable Temperature | |||
| Benzoic acid | 2 | 100 | ++ | 121 | No |
| Benzamide | 2 | 14 | ++ | 138 | No |
| High density polyethylene (HDPE) | 0 | 12 | + | 309 | Yes |
| Sebacic acid | 1 | 63 | ++ | 118 | No |
| Phtalic anhydride | 3 | 6 | + | 129 | No |
| Maleic acid | 3 | ** | ++ | 141 | No |
| Urea | 2 | ** | ++ | 148 | No |
| Dimethyl terephthalate | 1 | 2 | + | 128 | No |
| D-mannitol | 1 | 61 | ++ | 259 | No |
| Adipic acid | 1 | 7 | - | 203 | Yes |
| Salycilic acid | 2 | ** | ++ | 133 | No |
| Potassium thiocynate | 3 | 13 | + | 540 | No |
| Hydroquinnone | 2 | 62 | ++ | 157 | No |
| Benzanilide | 1 | 2 *** | - | 196 | No |
| Dulcitol | 1 | 100 | ++ | 293 | No |
| 2,2-Bis(hydroxymethyl) propionic acid (DMPA) | * | ** | ++ | 190 | No |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gasia, J.; Martin, M.; Solé, A.; Barreneche, C.; Cabeza, L.F. Phase Change Material Selection for Thermal Processes Working under Partial Load Operating Conditions in the Temperature Range between 120 and 200 °C. Appl. Sci. 2017, 7, 722. https://doi.org/10.3390/app7070722
Gasia J, Martin M, Solé A, Barreneche C, Cabeza LF. Phase Change Material Selection for Thermal Processes Working under Partial Load Operating Conditions in the Temperature Range between 120 and 200 °C. Applied Sciences. 2017; 7(7):722. https://doi.org/10.3390/app7070722
Chicago/Turabian StyleGasia, Jaume, Marc Martin, Aran Solé, Camila Barreneche, and Luisa F. Cabeza. 2017. "Phase Change Material Selection for Thermal Processes Working under Partial Load Operating Conditions in the Temperature Range between 120 and 200 °C" Applied Sciences 7, no. 7: 722. https://doi.org/10.3390/app7070722
APA StyleGasia, J., Martin, M., Solé, A., Barreneche, C., & Cabeza, L. F. (2017). Phase Change Material Selection for Thermal Processes Working under Partial Load Operating Conditions in the Temperature Range between 120 and 200 °C. Applied Sciences, 7(7), 722. https://doi.org/10.3390/app7070722








