1. Introduction
Mass spectrometry technology is employed to separate and detect charged molecular ions according to their mass-to-charge ratio. Given its high sensitivity, selectivity, and throughput capabilities, it assumes a pivotal role in various fields, including proteomics, metabolomics, food safety, biomedical analysis, and chemical imaging. Ionization is one of the core technologies in mass spectrometry. In 2004, Cooks proposed Desorption Electrospray Ionization (DESI), which made it possible to perform direct mass spectrometric analysis on untreated samples [
1]. In 2005, Durst introduced Direct Analysis in Real Time (DART) [
2]. Subsequently, the concept of Ambient Ionization Mass Spectrometry (AIMS) was proposed and has since become a research focus [
3,
4,
5,
6,
7,
8].
Recent advances in 3D printing have significantly impacted chemical analysis due to its rapid design–prototype cycles, cost-effectiveness, and customizable open-source features [
9,
10,
11,
12]. This technology shows particular promise for mass spectrometry enhancement, where commercially available ambient ionization sources remain limited, creating substantial replication challenges for researchers. Crucially, ambient ionization’s relatively lenient material requirements compared to conventional techniques enable effective integration with 3D-printed components. Elviri et al. demonstrated that 3D-printed PLA sample holders for DESI could double the signal intensity compared to conventional PTFE fixtures [
13]. Chen et al. developed textile-based electrofluidic devices coupled with DESI-MS for protein preconcentration [
14]. Zemaitis et al. implemented a fully 3D-printed DESI source for rat brain tissue imaging [
15]. These cases demonstrate how 3D printing’s rapid prototyping, customization, and cost-efficiency can transform specialized, hard-to-commercialize MS technologies. Our work extends this approach by developing a 3D-printed chamber, specifically addressing dual-spray interference challenges while retaining these manufacturing advantages.
The implementation of dual-channel and multi-channel spray in AIMS presents two significant advantages: streamlining sample pretreatment procedures to facilitate high-throughput detection and enhancing analytical performance, such as improving detection sensitivity. Dual-channel and multi-channel spray ionization technologies have extensive application requirements in precise mass analysis [
16,
17], the detection of complex matrix samples [
18,
19], sensitivity improvement [
20,
21], and high-throughput analysis of macromolecular compounds [
22,
23,
24,
25,
26]. Huang et al. developed a high-throughput induced nano-spray array ionization technology [
27]. Li et al. proposed a dual-channel induced nano-spray device for internal calibration [
16]. Chambers et al. designed a glass-based microfluidic device featuring two independent electrospray nozzles to sequentially ionize solutions for mass analysis [
17]. Yu et al. introduced a multi-channel ionization technology based on microfluidic chips for extracting and ionizing complex matrices without the need for sample pretreatment [
20]. Luo’s research group proposed a multi-channel rotating electrospray ionization technique in which desorbed analytes were simultaneously ionized by multiple spray solvents, enabling mass spectrometric detection of various compounds in complex matrices [
21].
In studies employing dual or multiple sprays concurrently for high-throughput analysis, researchers have noted significant electric field interference among independently charged spray tips. To enable simultaneous and independent charged spray functionality in dual- or multi-channel systems, numerous studies have resorted to compromise-based solutions or have implemented ingenious structural designs to alleviate this problem. The mutual interference between charged sprays is a crucial challenge that must be urgently resolved in the development of dual- and multi-channel spray ionization technologies. It is also one of the key issues addressed in this paper.
In recent years, 3D-printed chambers have been used in self-aspiration capillary electrospray ionization [
28] (SACESI) and self-aspiration corona discharge ionization ion sources [
29] (SACDI), resulting in a very stable ionization effect; however, the multi-channel electrospray system used for extraction and desorption lacks such standardized interface designs. Thus, in this paper, we propose a lateral extraction enhanced desorption electrospray ionization source (LEE-DESI). As illustrated in
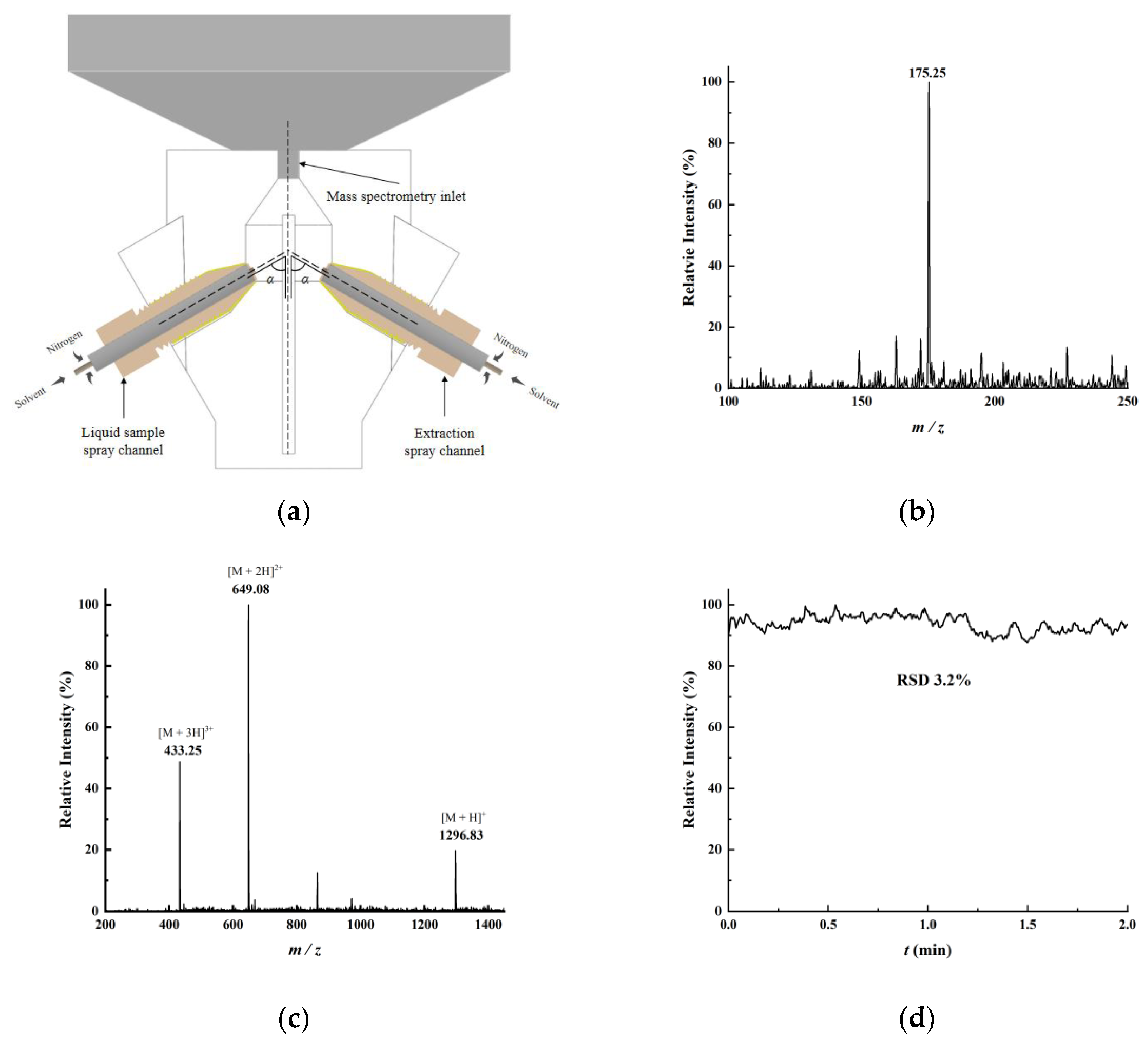
Figure 1, a glass slide physically separated the two spray channels to prevent direct interaction. The sample was deposited on one side of the slide, where the desorption spray desorbed charged droplets from the sample surface. Due to the inherently low ion collection efficiency of DESI and the aerosolizing effect of high-pressure nitrogen, some neutral sample droplets were generated. These neutral droplets were then transported to the region near the mass spectrometer inlet, where the extraction spray from the opposite side generated charged droplets. Collisions between the charged extraction droplets and neutral desorption droplets facilitate energy and charge transfer, enabling the extraction and ionization of neutral species.
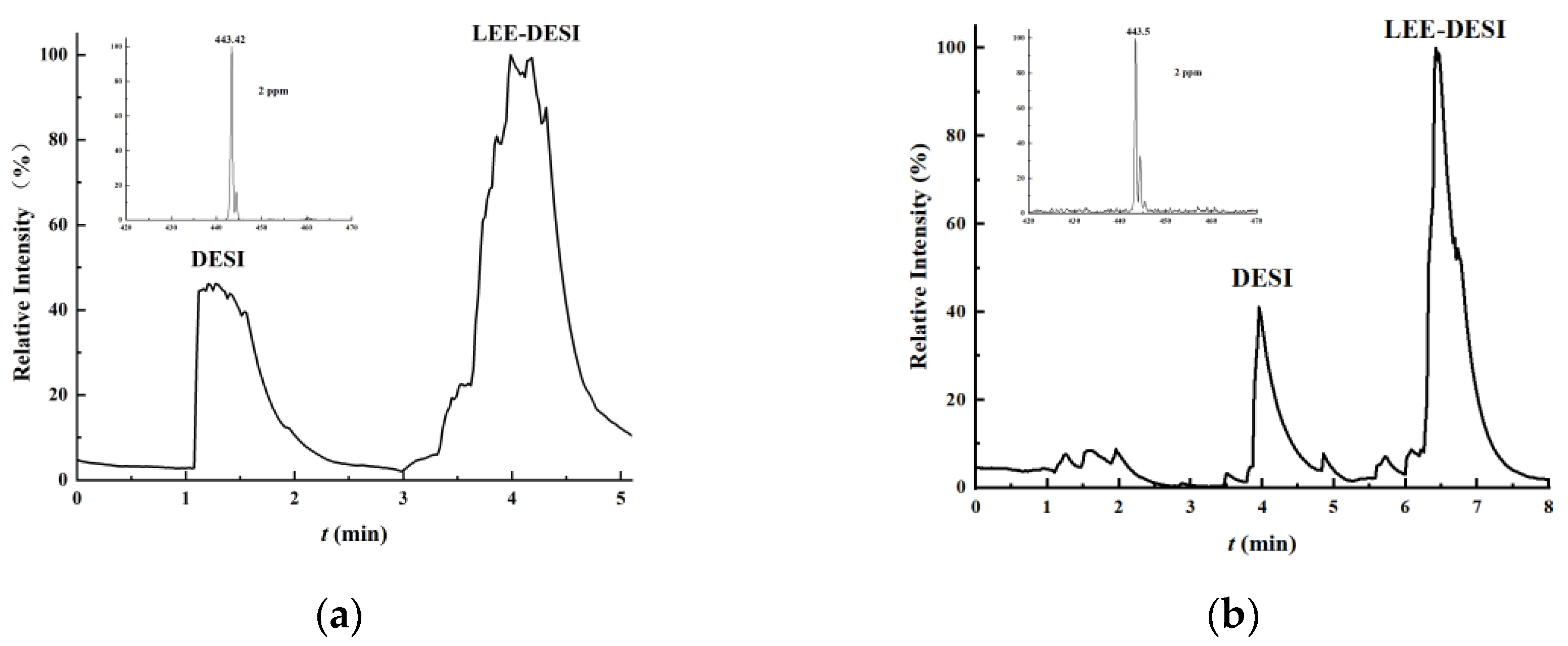
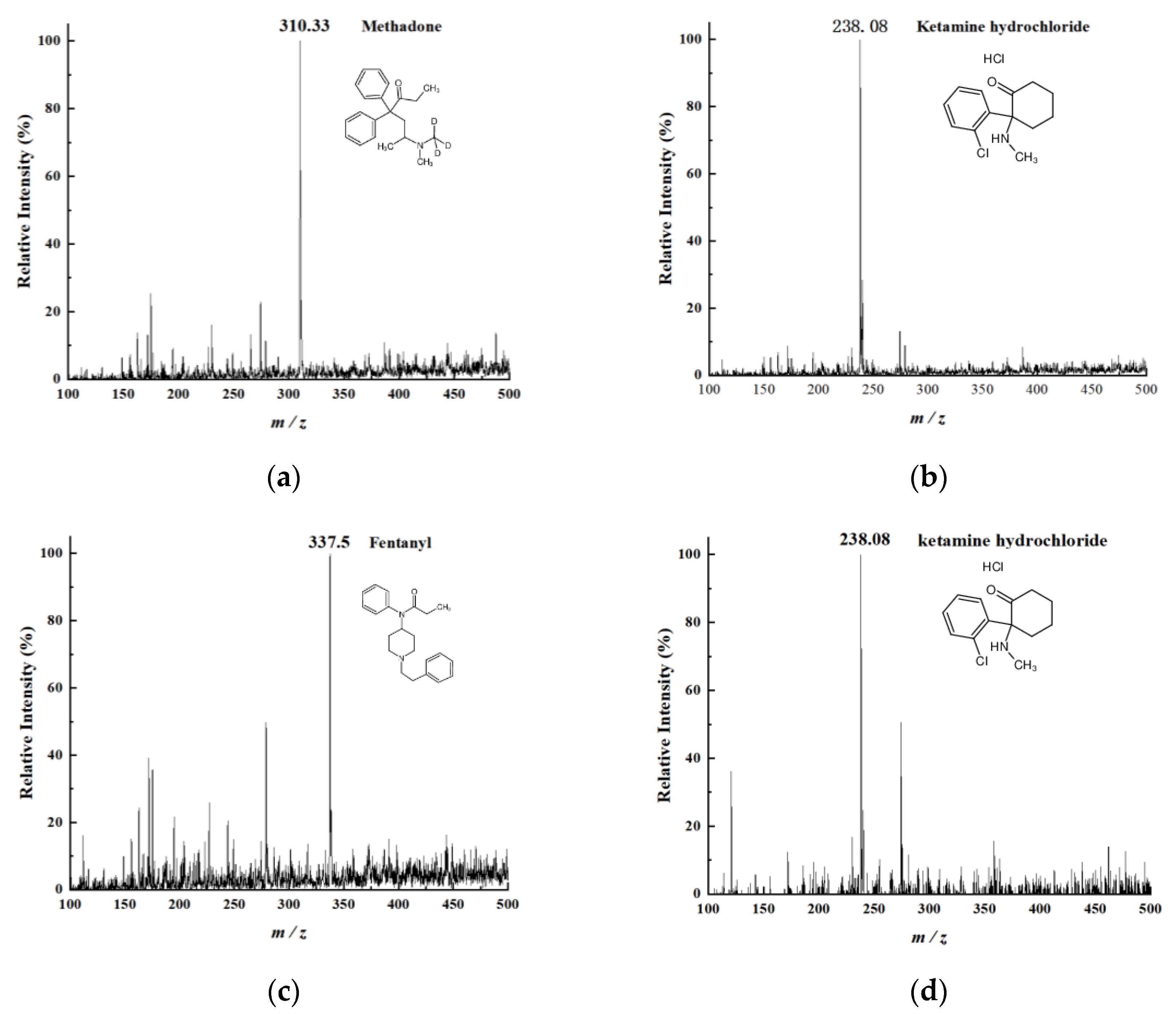
By incorporating dual-channel sprays into DESI—one for desorption and the other for extraction—the ionization efficiency during the desorption process is enhanced, thus boosting the desorption signal of samples. The ion source makes use of 3D printing technology to fix the optimized spatial parameters of the DESI setup and prevent electric field interference between the dual-channel charged sprays. Compared with traditional DESI, it demonstrates advantages such as high sensitivity, low detection limits, good reproducibility, and user-friendly operation. These advantages were validated using a range of representative compounds: Rhodamine B as a model dye for sensitivity testing, L-arginine and Angiotensin I as representatives of small molecules and peptides, respectively, and methadone, ketamine, and fentanyl as examples of controlled substances to demonstrate applicability in forensic and biomedical analysis.
2. Materials and Methods
2.1. Materials and Instruments
Methanol and formic acid were purchased from ANPEL (Shanghai, China) and Aladdin (Shanghai, China), respectively. We used Rhodamine B from Amper (Shanghai, China) and L-arginine and Angiotensin I (Ang I) from Aladdin (Shanghai, China) for verifying the devices. Methadone, ketamine hydrochloride, and fentanyl were provided by the Guangdong Nantian Institute of Forensic Science (Shenzhen, China). Artificial urine purchased from GEMIC (Shanghai, China) was used to dilute the ketamine hydrochloride. All of the above chemicals were of analytical reagent grade.
A single-sided frosted glass slide (Yancheng Wanyang Instrument Co., Ltd., Yancheng, China) was used to apply the sample to the surface. Quartz capillaries (100 μm i.d., 360 μm o.d., Zhengzhou Innatek Co., Ltd., Zhengzhou, China) were used for the transportation of solutions. The nitrogen (Shenzhen Huateng Special Gas Co., Ltd., Shenzhen, China) was supplied via a gas cylinder. The solution was injected using a syringe pump (KD Scientific, Holliston, MA, USA) and a microsyringe from an LCQ Fleet mass spectrometer (Thermo Fisher Scientific, Waltham, MA, USA). Three-dimensional printing (Shenzhen SANDI Technology Co., Ltd., Shenzhen, China) was applied to manufacture the LEE-DESI chamber. This design was executed using SLA 3D printing, a cost-effective manufacturing method where the material expense for a single chamber is around 150 RMB. The LEE-DESI source was tested and characterized using the LCQ Fleet mass spectrometer (Thermo Fisher Scientific, Waltham, MA, USA) and the Kirin 3 miniaturized mass spectrometer (Shenzhen Zhiqin Instrument Co., Ltd., Shenzhen, China).
Data acquisition and processing were performed using the following software: Xcalibur Qual Browser (version 2.2; Thermo Fisher Scientific, Waltham, MA, USA) for viewing, and processing raw data; and Origin (version 2022; OriginLab Corporation, Northampton, MA, USA) for statistical analysis and figure plotting.
2.2. Device Structure
Prior to finalizing the spatial parameters through 3D printing, we established two experimental platforms: (1) a laboratory DESI platform for systematic optimization of the basic spray geometry and (2) a laboratory LEE-DESI platform for investigating the feasibility of dual independent charged sprays and verifying whether the significant electric field interference between charged spray tips reported in the literature actually exists.
As depicted in
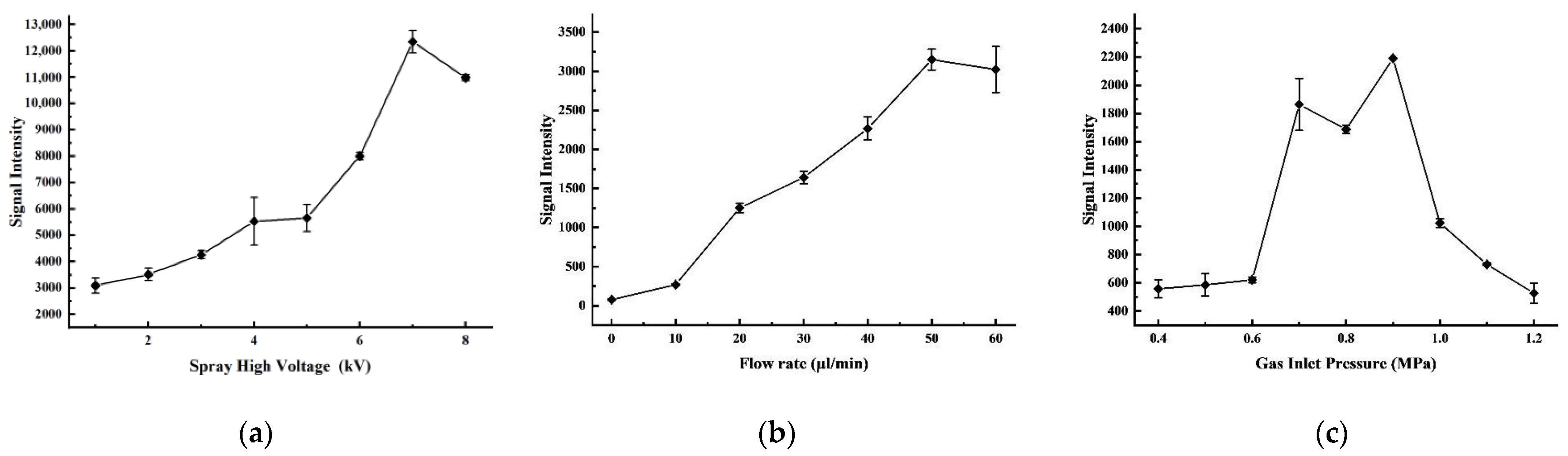
Figure 2a, the key optimized parameters for the DESI platform consist of an angle α of 50°, a distance of 3 mm from the sprayer tip to the glass slide surface, and a distance of 6 mm from the mass spectrometer inlet to the edge of the glass slide. These parameters were derived through iterative testing on the laboratory DESI platform to maximize ionization efficiency and signal stability. The liquid solution of the desorption spray channel is connected to the mass spectrometer inlet system, while its gas interface is connected to a nitrogen cylinder to provide auxiliary gas delivery. The liquid solution utilizes a fused silica capillary with an outer diameter of 360 μm and an inner diameter of 100 μm. A stainless steel tube with an inner diameter of 500 μm coaxially encloses the capillary, leaving a small gap. The capillary spray tip extends 0.5 mm beyond the steel tube to facilitate the alignment of the auxiliary gas flow with the electrospray direction.
The LEE-DESI platform extends the conventional DESI platform by incorporating a precision-controlled secondary spray channel. As shown in
Figure 2b, the right channel serves as the desorption electrospray path while the left channel functions as the extraction spray path. Both channels permit precise adjustment of sprayer angle and displacement, maintaining an inter-spray angle of 50°. The system operates in standard DESI mode when only the right channel is activated, whereas simultaneous activation of both channels enables LEE-DESI functionality. The extraction sprayer (left) maintains an identical configuration to the desorption sprayer (right) and thus requires no additional description. Experimental results revealed strong electric field interference between the two charged spray channels. Under laser illumination, distinct mutual repulsion between the sprays was observed with clearly visible boundaries. Furthermore, the high-speed gas flow from the desorption spray channel caused significant spray dispersion. These combined effects substantially compromised the sample detection signal intensity, demonstrating that the current LEE-DESI platform does not yet meet the requirements for successful dual-spray operation.
While the dual-spray configuration in our prototype LEE-DESI platform (
Figure 2b) demonstrated suboptimal performance, we observed several notable advantages over single-channel systems. The dual-channel arrangement produced a significantly larger total spray volume. More importantly, the high-speed gas flow facilitated the convergence of charged solvent droplets from the extraction channel with both charged and neutral sample droplets from the desorption channel near the mass spectrometer inlet. This interaction enabled effective energy and charge transfer through droplet collisions, while the gas flow simultaneously enhanced desolvation of charged droplets—collectively resulting in improved sample ionization efficiency.
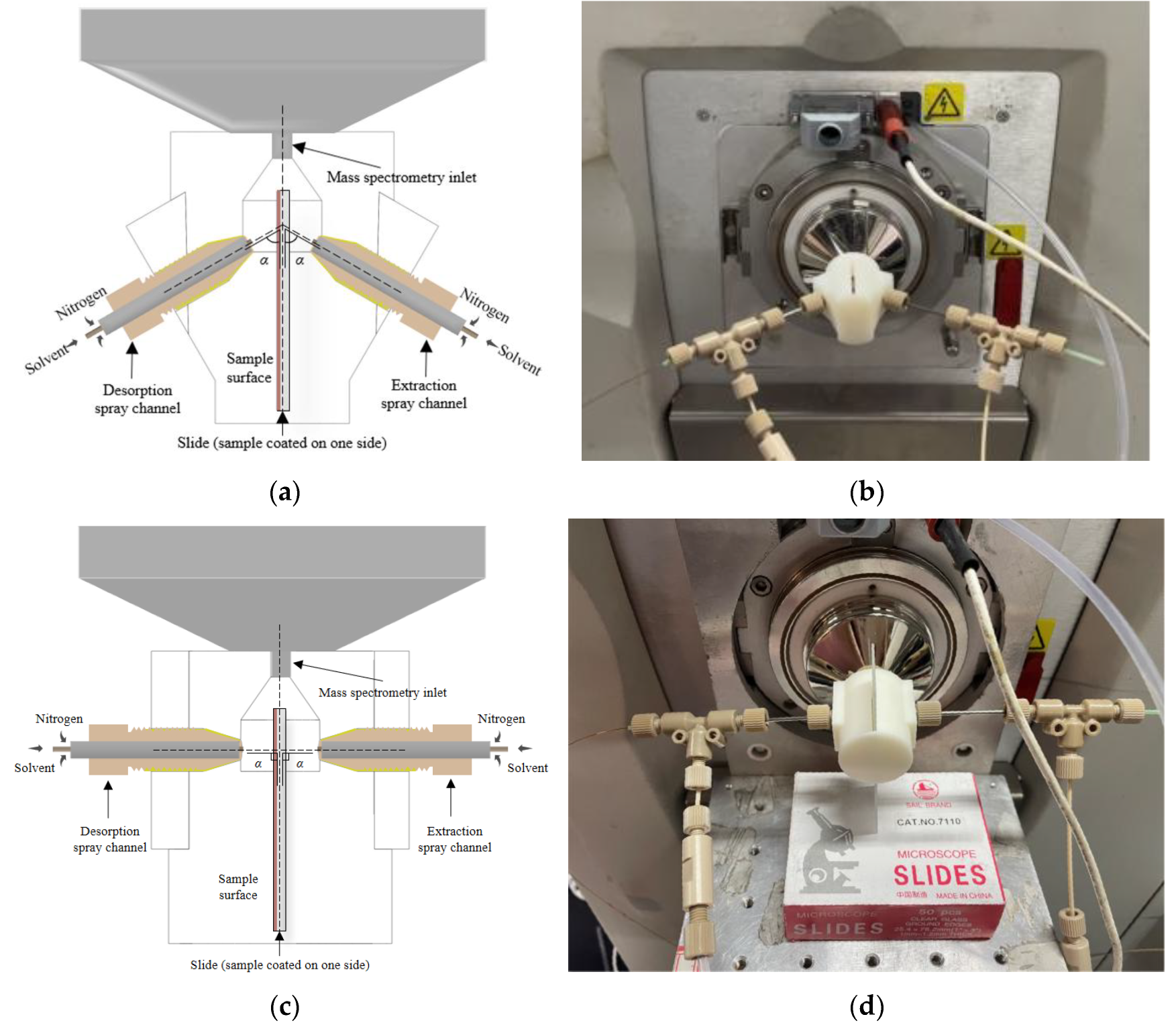
The investigation focused on developing an approach that would maintain the optimized DESI parameters while eliminating interference between dual spray channels. Three-dimensional printing technology was selected for this application due to its rapid prototyping capabilities. The resulting 3D-printed chamber was designed according to mass spectrometer interface specifications, featuring an integrated coupling interface with an orifice diameter of 2.2 mm, and incorporated the optimized DESI spatial parameters (α = 50°, 3 mm sprayer-to-surface distance, 6 mm inlet-to-slide distance).
Figure 3 illustrates the integrated chamber design, where desorption and extraction spray channels are positioned on opposite sides. The chamber features a plug-and-play interface with the mass spectrometer, eliminating time-consuming alignment procedures and significantly enhancing experimental efficiency.
A critical design element involves the strategic placement of a glass slide that physically separates the two spray channels while permitting controlled droplet interactions near the MS inlet. This configuration effectively prevents direct spray interference while preserving the beneficial charge transfer effects identified in preliminary studies. The assembly protocol for the 3D-printed LEE-DESI platform mirrors that of the initial prototype (described in preceding sections), maintaining operational consistency while addressing the limitations of the earlier design.
In addition to the 50° dual-channel spray configuration, a 90° dual-channel spray setup was also tested. This extreme-angle design was implemented to investigate the operational limits of dual-spray functionality, particularly whether the spatial constraints at 90° would lead to spray failure or signal instability. The spatial parameters for the 90° setup included a distance of 3mm from the sprayer tip to the glass slide surface and a distance of 6mm from the mass spectrometer inlet to the edge of the glass slide. The liquid and gas interfaces remained identical to the 50° configuration.
2.3. Sample Preparation
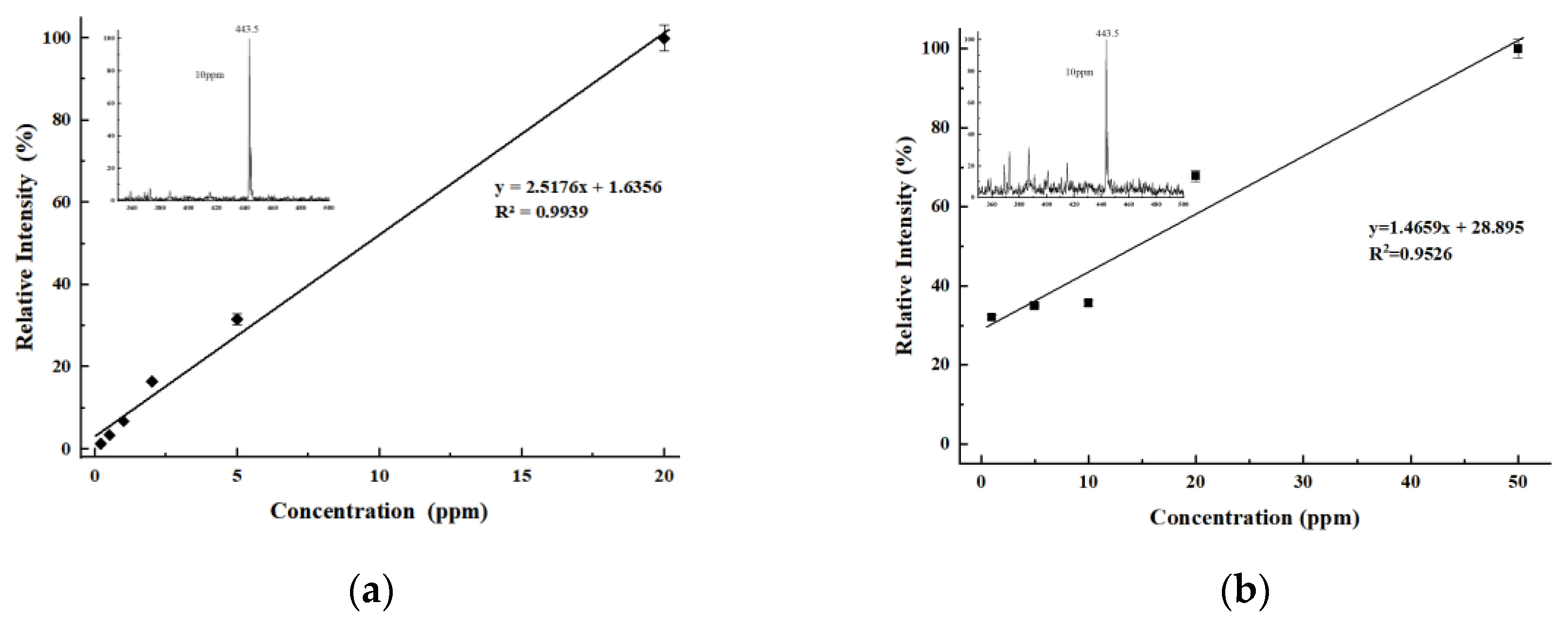
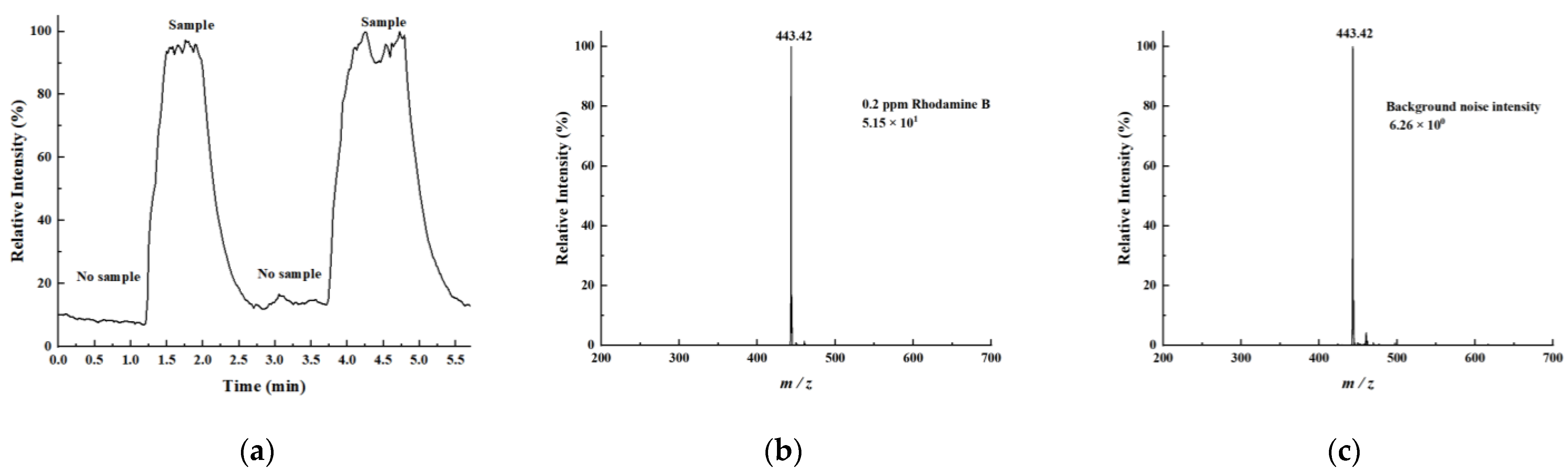
Rhodamine B was dissolved in a methanol–water mixture (1:1, v/v) to prepare a 100 ppm (parts per million) standard solution. This standard solution was subsequently diluted with the same methanol–water mixture (1:1, v/v) to obtain Rhodamine B test solutions at concentrations of 50, 20, 10, 5, 2, 1, 0.5, and 0.2 ppm. Using an identical method, a 10 ppm L-arginine solution and a 100 ppm angiotensin solution were also prepared. Methadone and fentanyl were prepared as 10 ppm standard solutions, while ketamine hydrochloride was provided as a 100 ppm standard. The 100 ppm ketamine hydrochloride solution was subsequently diluted to 10 ppm using artificial urine. The prepared test solutions were applied to single-sided frosted glass slides with cotton swabs and left to dry completely. The slides were then inserted into the LEE-DESI platform for analysis, with the frosted side facing the desorption electrospray channel. A methanol–water mixture (1:1, v/v, containing 0.1% formic acid) was used as the spray solvent.
4. Conclusions
The LEE-DESI successfully addressed the electric field interference issues in dual-channel spray systems via innovative spatial separation and 3D-printed structural optimization. By integrating desorption and extraction sprays within an optimized geometry, the system attains enhanced ionization efficiency and sensitivity while maintaining operational simplicity. The fixed spatial parameters provided by 3D printing ensure reproducible performance across different experimental setups. Performance evaluations with standard compounds confirmed the system’s reliability for quantitative analysis, showing consistent signal response across multiple measurements. The dual-spray configuration demonstrates clear advantages for direct sample analysis, particularly in maintaining signal stability while reducing preparation requirements. The design’s flexibility allows for straightforward adaptation to different mass spectrometer interfaces, as shown through successful coupling with both laboratory and portable instruments. These features make the LEE-DESI a practical solution for the direct analysis of untreated samples within complex matrices.
Building on this foundation, two promising future directions are immediately apparent. First, the fixed geometry of the source makes it ideally suited for adaptation to imaging mass spectrometry by integrating a motorized stage into raster samples, which could revolutionize the detection of spatial chemical distributions in biological tissues. Second, while this study demonstrates efficacy in standardized scenarios, a critical next step is the rigorous validation of the system with truly complex matrices—such as clinical biofluids and tissue homogenates—to fully unlock its potential in proteomic and metabolomic workflows.
Future applications may therefore extend to multi-channel systems for high-throughput analysis, as well as to the challenging realms of chemical imaging and in situ bioanalysis, thereby further promoting ambient ionization technologies in real-time analytical workflows.