Comparative Phenolic Profiles of Monovarietal Wines from Different Croatian Regions
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Wine Samples
2.3. HPLC Analyses
2.4. DPPH Radical Scavenging Activity
2.5. NO Radical Scavenging Activity
2.6. Lipid Peroxidation Inhibition Assay
2.7. Statistical Analysis
3. Results and Discussion
3.1. Contents of Phenolic Compounds in Croatian Wines
3.1.1. Phenolic Acids in Selected Red and White Wines
3.1.2. Flavan-3-ols, Anthocyanins, and Stilbenes in Selected Red and White Wines
3.1.3. Flavonoids in the Selected Red and White Wines
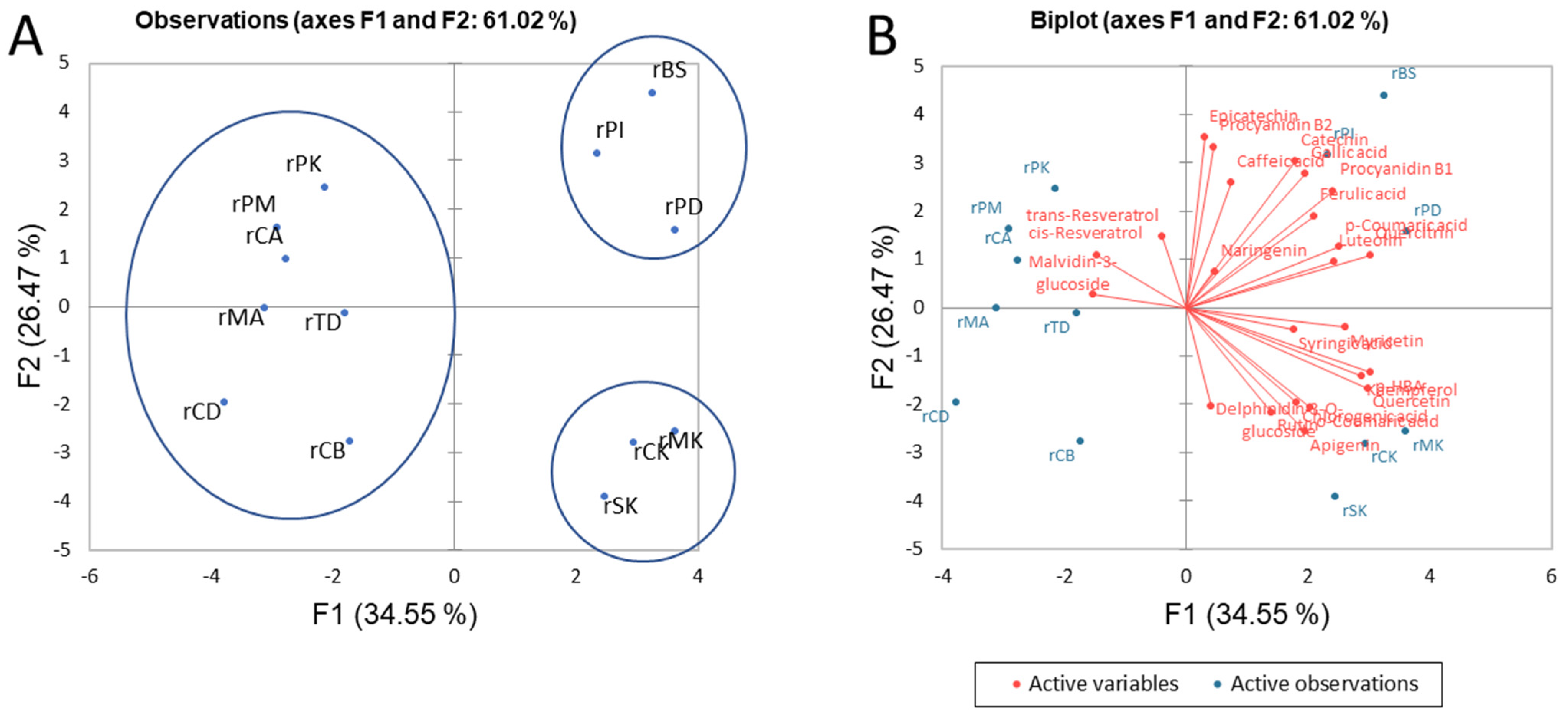
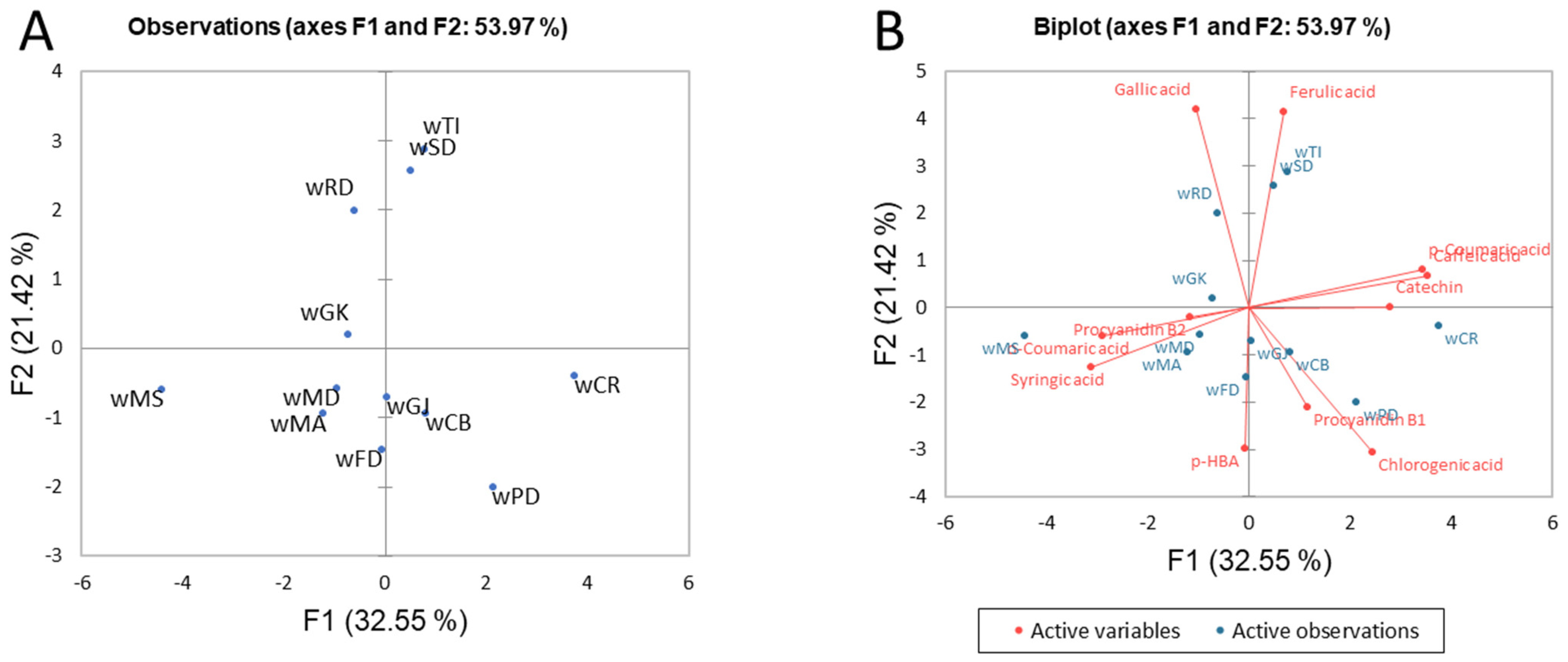
3.1.4. Differentiation of the Selected Monovarietal Red and White Wines
3.2. Antioxidant Activity of Major Phenolic Components in Commercial Croatian Wines
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Merkytė, V.; Longo, E.; Windisch, G.; Boselli, E. Phenolic compounds as markers of wine quality and authenticity. Foods 2020, 9, 1785. [Google Scholar] [CrossRef] [PubMed]
- Garrido, J.; Borges, F. Wine and grape polyphenols—A chemical perspective. Food Res. Int. 2013, 54, 1844–1858. [Google Scholar] [CrossRef]
- Casassa, L.F.; Gannet, P.A.; Steele, N.B.; Huff, R. Multi-year study of the chemical and sensory effects of microwave-assisted extraction of musts and stems in Cabernet Sauvignon, Merlot and Syrah wines from the central coast of California. Molecules 2022, 27, 1270. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Escobar, R.; Aliaño-González, M.J.; Cantos-Villar, E. Wine polyphenol content and its influence on wine quality and properties: A review. Molecules 2021, 26, 718. [Google Scholar] [CrossRef]
- Weaver, S.R.; Rendeiro, C.; McGettrick, H.M.; Philp, A.; Lucas, S.J.E. Fine wine or sour grapes? A systematic review and meta-analysis of the impact of red wine polyphenols on vascular health. Eur. J. Nutr. 2021, 60, 1–28. [Google Scholar] [CrossRef]
- Radeka, S.; Rossi, S.; Bestulić, E.; Budić-Leto, I.; Kovačević Ganić, K.; Horvat, I.; Orbanić, F.; Zaninović Jurjević, T.; Dvornik, Š. Bioactive compounds and antioxidant activity of red and white wines produced from autochthonous Croatian varieties: Effect of moderate consumption on human health. Foods 2022, 11, 1804. [Google Scholar] [CrossRef]
- Lucarini, C.M.; Durazzo, A.; Lombardi-Bocci, G.; Souto, E.B.; Cecchini, F.; Santini, A. Wine polyphenols and health: Quantitative research literature analysis. Appl. Sci. 2021, 11, 4762. [Google Scholar] [CrossRef]
- Pavlidou, E.; Mantzorou, M.; Fasoulas, A.; Tryfonos, C.; Petridis, D.; Giaginis, C. Wine: An aspiring agent in promoting longevity and preventing chronic diseases. Diseases 2018, 6, 73. [Google Scholar] [CrossRef]
- Nash, V.; Ranadheera, C.S.; Georgousopoulou, E.N.; Mellor, D.D.; Panagiotakos, D.B.; McKune, A.J.; Kellett, J.; Naumovski, N. The effects of grape and red wine polyphenols on gut microbiota—A systematic review. Food Res. Int. 2018, 113, 277–287. [Google Scholar] [CrossRef]
- Nemzer, B.; Kalita, D.; Yashin, A.Y.; Yashin, Y.I. Chemical composition and polyphenolic compounds of red wines: Their antioxidant activities and effects on human health—A review. Beverages 2022, 8, 1. [Google Scholar] [CrossRef]
- Žurga, P.; Vahčić, N.; Pasković, I.; Banovi, M.; Malenica Staver, M. Croatian wines from native grape varieties have higher distinct phenolic (nutraceutic) profiles than wines from non-native varieties with the same geographic origin. Chem. Biodivers. 2019, 16, e1900218. [Google Scholar] [CrossRef] [PubMed]
- Leder, R.; Petric, I.V.; Jusup, J.; Banović, M. Geographical discrimination of Croatian wines by stable isotope ratios and multielemental composition analysis. Front. Nutr. 2021, 8, 625613. [Google Scholar] [CrossRef] [PubMed]
- Zakon o Vinu (Law on Wine). Available online: https://narodne-novine.nn.hr/clanci/sluzbeni/2019_03_32_641.html (accessed on 13 August 2022).
- Rastija, V.; Srečnik, G.; Medić-Šarić, M. Polyphenolic composition of Croatian wines with different geographical origins. Food Chem. 2009, 115, 54–60. [Google Scholar] [CrossRef]
- Šeruga, M.; Novak, L.; Jakobek, L. Determination of polyphenols content and antioxidant activity of some red wines by differential pulse voltammetry, HPLC and spectrophotometric methods. Food Chem. 2011, 124, 1208–1216. [Google Scholar] [CrossRef]
- Lukić, I.; Radeka, S.; Budić-Leto, I.; Bubola, M.; Vrhovsek, U. Targeted UPLC-QqQ-MS/MS profiling of phenolic compounds for differentiation of monovarietal wines and corroboration of particular varietal typicity concepts. Food Chem. 2019, 300, 125251. [Google Scholar] [CrossRef]
- Jagatić Korenika, A.-M.; Tomaz, I.; Preiner, D.; Plichta, V.; Jeromel, A. Impact of commercial yeasts on phenolic profile of Plavac Mali wines from Croatia. Fermentation 2021, 7, 92. [Google Scholar] [CrossRef]
- Mucalo, A.; Zdunić, G.; Maletić, E. Prolonged ripening on the vine affects the polyphenolic profile of grapes and wine of ’Plavac Mali’ (Vitis vinifera L.). Acta Hortic. 2019, 1248, 417–424. [Google Scholar] [CrossRef]
- Budić-Leto, I.; Zdunić, G.; Gajdoš Kljusurić, J.; Pezo, I.; Alpeza, I.; Lovrić, T. Effects of polyphenolic composition on sensory perception of Croatian red wine Babić. J. Food Agric. Environ. 2008, 6, 138–142. [Google Scholar]
- Budić-Leto, I.; Lovrić, T.; Gajdoš-Kljusurić, J.; Pezo, I.; Vrhovšek, U. Anthocyanin composition of the red wine Babić affected by maceration treatment. Eur. Food Res. Technol. 2006, 222, 397–402. [Google Scholar] [CrossRef]
- Bubola, M.; Rusjan, D.; Lukić, I. Crop level vs. leaf removal: Effects on Istrian Malvasia wine aroma and phenolic acids composition. Food Chem. 2020, 312, 126046. [Google Scholar] [CrossRef]
- Osrečak, M.; Karoglan, M.; Kozina, B.; Preiner, D. Influence of leaf removal and reflective mulch on phenolic composition of white wines. J. Int. Sci. Vigne Vin 2015, 49, 183–193. [Google Scholar] [CrossRef]
- Maletić, E.; Kontić, K.; Preiner, D.; Jeromel, A.; Patz, C.-D.; Dietrich, H. Anthocyanin profile and antioxidative capacity of some autochthonous Croatian red wines. J. Food Agric. Environ. 2009, 7, 48–51. [Google Scholar] [CrossRef]
- Katalinić, V.; Milos, M.; Modun, D.; Musić, I.; Boban, M. Antioxidant effectiveness of selected wines in comparison with (+)-catechin. Food Chem. 2006, 86, 593–600. [Google Scholar] [CrossRef]
- Vinković Vrček, I.; Bojić, M.; Žuntar, I.; Mendaš, G.; Medić-Šarić, M. Phenol content, antioxidant activity and metal composition of Croatian wines deriving from organically and conventionally grown grapes. Food Chem. 2011, 124, 354–361. [Google Scholar] [CrossRef]
- Visioli, F.; Panaite, S.-A.; Tomé-Carneiro, J. Wine’s phenolic compounds and health: A Pythagorean view. Molecules 2020, 25, 4105. [Google Scholar] [CrossRef]
- Nour, V.; Trandafir, I.; Muntean, C. Ultraviolet irradiation of trans-resveratrol and HPLC determination of trans-resveratrol and cis-resveratrol in Romanian red wines. J. Chromatogr. Sci. 2012, 50, 920–927. [Google Scholar] [CrossRef]
- Berente, B.; De la Calle García, D.; Reichenbächer, M.; Danzer, K. Method development for the determination of anthocyanins in red wines by high-performance liquid chromatography and classification of German red wines by means of multivariate statistical methods. J. Chromatogr. A 2000, 871, 95–103. [Google Scholar] [CrossRef]
- ICH Harmonised Tripartite Guideline. Validation of Analytical Procedures: Text and Methodology Q2 (R1). Available online: https://database.ich.org/sites/default/files/Q2%28R1%29%20Guideline.pdf (accessed on 13 August 2022).
- de Souza Dias, F.; Lovillo, M.P.; Barroso, C.G.; David, J.M. Optimization and validation of a method for the direct determination of catechin and epicatechin in red wines by HPLC/fluorescence. Microchem. J. 2010, 96, 17–20. [Google Scholar] [CrossRef]
- Vladimir-Knežević, S.; Blažeković, B.; Štefan, M.B.; Alegro, A.; Köszegi, T.; Petrik, J. Antioxidant activities and polyphenolic contents of three selected Micromeria species from Croatia. Molecules 2011, 16, 1454–1470. [Google Scholar] [CrossRef]
- Patel, A.; Patel, A.; Patel, A.; Patel, N.M. Determination of polyphenols and free radical scavenging activity of Tephrosia purpurea Linn leaves (Leguminosae). Pharmacogn. Res. 2010, 2, 152–158. [Google Scholar] [CrossRef]
- Houghton, P.J.; Zarka, R.; de las Heras, B.; Hoult, J.R. Fixed oil of Nigella sativa and derived thymoquinone inhibit eicosanoid generation in leukocytes and membrane lipid peroxidation. Planta Med. 1995, 61, 33–36. [Google Scholar] [CrossRef]
- Ćurko, N.; Kovačević Ganić, K.; Gracin, L.; Đapić, M.; Jourdes, M.; Teissedre, P.L. Characterization of seed and skin polyphenolic extracts of two red grape cultivars grown in Croatia and their sensory perception in a wine model medium. Food Chem. 2014, 145, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Šćepanović, R.P.; Wendelin, S.; Raičević, D.; Eder, R. Characterization of the phenolic profile of commercial Montenegrin red and white wines. Eur. Food Res. Technol. 2019, 245, 2233–2245. [Google Scholar] [CrossRef]
- Zhu, F.; Du, B.; Shi, P.; Li, F. Phenolic profile and antioxidant capacity of ten dry red wines from two major wine-producing regions in China. Adv. J. Food Sci. Technol. 2014, 6, 344–349. [Google Scholar] [CrossRef]
- Zhu, J.; Hu, B.; Lu, J.; Xu, S. Analysis of metabolites in Cabernet Sauvignon and Shiraz dry red wines from Shanxi by 1H NMR spectroscopy combined with pattern recognition analysis. Open Chem. 2018, 16, 446–452. [Google Scholar] [CrossRef]
- Ivanova-Petropulos, V.; Ricci, A.; Nedelkovski, D.; Dimovska, V.; Parpinello, G.P.; Versari, A. Targeted analysis of bioactive phenolic compounds and antioxidant activity of Macedonian red wines. Food Chem. 2015, 171, 412–420. [Google Scholar] [CrossRef] [PubMed]
- Majkić, T.M.; Torović, L.D.; Lesjak, M.M.; Četojević-Simin, D.D.; Beara, I.N. Activity profiling of Serbian and some other European Merlot wines in inflammation and oxidation processes. Food Res. Int. 2019, 121, 151–160. [Google Scholar] [CrossRef]
- Girelli, A.; Mele, C.; Salvagni, L.; Tarola, A.M. Polyphenol content and antioxidant activity of Merlot and Shiraz wine. Anal. Lett. 2015, 48, 1865–1880. [Google Scholar] [CrossRef]
- Fermo, P.; Comite, V.; Sredojević, M.; Ćirić, I.; Gašić, U.; Mutić, J.; Baošić, R. Elemental analysis and phenolic profiles of selected Italian wines. Foods 2021, 10, 158. [Google Scholar] [CrossRef]
- Sartor, S.; Malinovski, L.I.; Caliari, V.; da Silva, A.L.; Bordignon-Luiz, M.T. Particularities of Syrah wines from different growing regions of Southern Brazil: Grapevine phenology and bioactive compounds. J. Food Sci. Technol. 2017, 54, 1414–1424. [Google Scholar] [CrossRef]
- Alpeza, I.; Lukić, K.; Vanzo, A.; Kovačević Ganić, K. Bioactive complexity of the red wine “Portugizac”; Is younger more beneficial? Agric. Conspec. Sci. 2021, 86, 329–335. [Google Scholar]
- Atanacković Krstonošić, M.; Cvejić Hogervorst, J.; Torović, L.; Puškaš, V.; Miljić, U.; Mikulić, M.; Gojković Bukarica, L. Influence of 4 years of ageing on some phenolic compounds in red wines. Acta Aliment. 2019, 48, 449–456. [Google Scholar] [CrossRef]
- Nikfardjam, M.S.P.; Márk, L.; Avar, P.; Figler, M.; Ohmacht, R. Polyphenols, anthocyanins, and trans-resveratrol in red wines from the Hungarian Villány region. Food Chem. 2006, 98, 453–462. [Google Scholar] [CrossRef]
- Van Leeuw, R.; Kevers, C.; Pincemail, J.; Defraigne, J.O.; Dommes, J. Antioxidant capacity and phenolic composition of red wines from various grape varieties: Specificity of Pinot Noir. J. Food Compos. Anal. 2014, 36, 40–50. [Google Scholar] [CrossRef]
- Meneghetti, S.; Poljuha, D.; Frare, E.; Costacurta, A.; Morreale, G.; Bavaresco, L.; Calo, A. Inter- and intra-varietal genetic variability in Malvasia. Mol. Biotechnol. 2012, 50, 189–199. [Google Scholar] [CrossRef] [PubMed]
- Ragusa, A.; Centonze, C.; Grasso, M.E.; Latronico, M.F.; Mastrangelo, P.F.; Sparascio, F.; Fanizzi, F.P.; Maffia, M. A comparative study of phenols in Apulian Italian wines. Foods 2017, 6, 24. [Google Scholar] [CrossRef]
- Mucalo, A.; Lukšić, K.; Budić-Leto, I.; Zdunić, G. Cluster thinning improves aroma complexity of white Maraština (Vitis vinifera L.) wines compared to defoliation under Mediterranean climate. Appl. Sci. 2022, 12, 7327. [Google Scholar] [CrossRef]
- del Barrio-Galán, R.; del Valle-Herrero, H.; Bueno-Herrera, M.; López-de-la-Cuesta, P.; Pérez-Magariño, S. Volatile and non-volatile characterization of white and rosé wines from different Spanish Protected Designations of Origin. Beverages 2021, 7, 49. [Google Scholar] [CrossRef]
- de Villiers, A.; Majek, P.; Lynen, F.; Crouch, A.; Lauer, H.; Sandra, P. Classification of South African red and white wines according to grape variety based on the non-coloured phenolic content. Eur. Food Res. Technol. 2005, 221, 520–528. [Google Scholar] [CrossRef]
- Mitić, M.N.; Obradović, M.V.; Grahovac, Z.B.; Pavlović, A.N. Antioxidant capacities and phenolic levels of different varieties of Serbian white wines. Molecules 2010, 15, 2016–2027. [Google Scholar] [CrossRef]
- Jagatić Korenika, A.-M.; Žulj, M.M.; Puhelek, I.; Plavša, T.; Jeromel, A. Study of phenolic composition and antioxidant capacity of Croatian macerated white wines. Mitt. Klostern. 2014, 64, 171–180. [Google Scholar]
- Jakobović, S.; Jero, A.; Maslov Bandić, L.; Jakobović, M. Influence of grape ripeness of Rhine Riesling on the composition of polyphenolic compounds in must and wine. J. Food Agric. Environ. 2015, 13, 29–35. [Google Scholar]
- Monagas, M.; Gómez-Cordovés, C.; Bartolomé, B.; Laureano, O.; da Silva, J.M.R. Monomeric, oligomeric, and polymeric flavan-3-ol composition of wines and grapes from Vitis vinifera L. Cv. Graciano, Tempranillo, and Cabernet Sauvignon. J. Agric. Food Chem. 2003, 51, 6475–6481. [Google Scholar] [CrossRef] [PubMed]
- Jiang, B.; Zhang, Z.-W. Comparison on phenolic compounds and antioxidant properties of Cabernet Sauvignon and Merlot wines from four wine grape-growing regions in China. Molecules 2012, 17, 8804–8821. [Google Scholar] [CrossRef] [PubMed]
- Rocchetti, G.; Gatti, M.; Bavaresco, L.; Lucini, L. Untargeted metabolomics to investigate the phenolic composition of Chardonnay wines from different origins. J. Food Compos. Anal. 2018, 71, 87–93. [Google Scholar] [CrossRef]
- Kumšta, M.; Pavloušek, P.; Kárník, P. Use of anthocyanin profiles when differentiating individual varietal wines and terroirs. Food Technol. Biotechnol. 2014, 52, 383–390. [Google Scholar] [CrossRef] [PubMed]
- de Andrade, R.H.S.; do Nascimento, L.S.; Pereira, G.E.; Hallwass, F.; Paim, A.P.S. Anthocyanic composition of Brazilian red wines and use of HPLC-UV–Vis associated to chemometrics to distinguish wines from different regions. Microchem. J. 2013, 110, 256–262. [Google Scholar] [CrossRef]
- Mark, L.; Nikfardjam, M.S.P.; Avar, P.; Ohmacht, R. A validated HPLC method for the quantitative analysis of trans-resveratrol and trans-piceid in Hungarian wines. J. Chromatogr. Sci. 2005, 43, 445–449. [Google Scholar] [CrossRef]
- Baron, M.; Sochor, J.; Tomaskova, L.; Prusova, B.; Kumsta, M. Study on antioxidant components in rosé wine originating from the wine growing region of Moravia, Czech Republic. Erwerbs-Obstbau 2017, 59, 253–262. [Google Scholar] [CrossRef]
- Katalinić, V.; Ljubenkov, I.; Pezo, I.; Generalić, I.; Stričević, O.; Miloš, M.; Modun, D.; Boban, M. Free resveratrol monomers in varietal red and white wines from Dalmatia (Croatia). Period. Biolog. 2008, 110, 77–83. [Google Scholar]
- Valletta, A.; Iozia, L.M.; Leonelli, F. Impact of environmental factors on stilbene biosynthesis. Plants 2021, 10, 90. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Lüscher, J.; Brillante, L.; Kurtural, S.K. Flavonol profile is a reliable indicator to assess canopy architecture and the exposure of red wine grapes to solar radiation. Front. Plant Sci. 2019, 10, 10. [Google Scholar] [CrossRef] [PubMed]
- Stefenon, C.A.; Bonesi, C.D.M.; Marzarotto, V.; Barnabé, D.; Spinelli, F.R.; Webber, V.; Vanderlinde, R. Phenolic composition and antioxidant activity in sparkling wines: Modulation by the ageing on lees. Food Chem. 2014, 145, 292–2099. [Google Scholar] [CrossRef] [PubMed]
- Pozo-Bayón, M.A.; Andujar, O.; Moreno-Arribas, M.V. Scientific evidence beyond the application of inactive dry yeast preparations in winemaking. Food Res. Int. 2009, 42, 754–761. [Google Scholar] [CrossRef]
- Haseeb, S.; Alexander, B.; Lopez Santi, R.; Sosa Liprandi, A.; Baranchuk, A. What’s in wine? A clinician’s perspective. Trends Cardiovasc. Med. 2019, 29, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Platzer, M.; Kiese, S.; Tybussek, T.; Herfellner, T.; Schneider, F.; Schweiggert-Weisz, U.; Eisner, P. Radical scavenging mechanisms of phenolic compounds: A quantitative structure-property relationship (QSPR) study. Front. Nutr. 2022, 9, 882458. [Google Scholar] [CrossRef] [PubMed]
- Mervić, M.; Bival Štefan, M.; Kindl, M.; Blažeković, B.; Marijan, M.; Vladimir-Knežević, S. Comparative antioxidant, anti-acetylcholinesterase and anti-α-glucosidase activities of Mediterranean Salvia species. Plants 2022, 11, 625. [Google Scholar] [CrossRef] [PubMed]
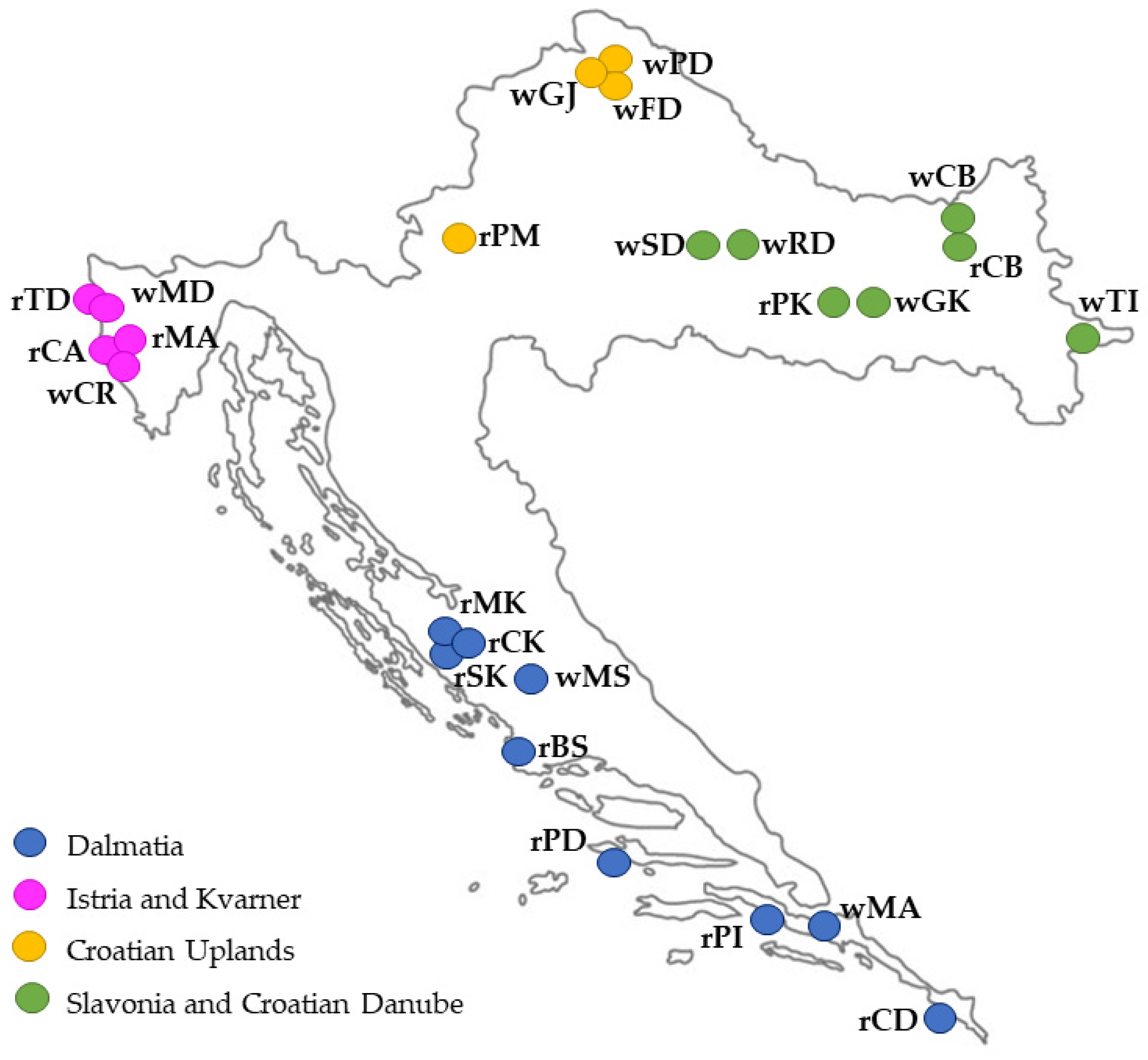
| Sample Abbrev. | Grape Variety | Croatian Wine Region | Croatian Wine Subregion | Vineyard Location |
|---|---|---|---|---|
| rPD | Plavac Mali | Dalmatia | Central and Southern Dalmatia | Ivan Dolac, (Island Hvar) |
| rPI | Plavac Mali | Dalmatia | Central and Southern Dalmatia | Postup (Pelješac Peninsula) |
| rBS | Babić | Dalmatia | Northern Dalmatia | Primošten |
| rTD | Teran | Istria and Kvarner | Istria | Dajla (Novigrad) |
| rCD | Cabernet Sauvignon | Dalmatia | Central and Southern Dalmatia | Konavle |
| rCK | Cabernet Sauvignon | Dalmatia | Dalmatian Hinterland | Korlat (Benkovac) |
| rCA | Cabernet Sauvignon | Istria and Kvarner | Istria | Mornarica (Poreč) |
| rCB | Cabernet Sauvignon | Slavonia and Croatian Danube | Croatian Danube | Banovo brdo (Beli Manastir) |
| rMK | Merlot | Dalmatia | Dalmatian Hinterland | Korlat (Benkovac) |
| rMA | Merlot | Istria and Kvarner | Istria | Faragude (Funtana) |
| rSK | Syrah | Dalmatia | Dalmatian Hinterland | Korlat (Benkovac) |
| rPM | Blauer Portugieser | Croatian Uplands | Plešivica Hills | Okić (Plešivica) |
| rPK | Pinot Noir | Slavonia and Croatian Danube | Slavonia | Hrnjavac (Kutjevo) |
| Sample Abbrev. | Grape Variety | Croatian Wine Region | Croatian Wine Subregion | Vineyard Location |
|---|---|---|---|---|
| wMD | Malvazija Istarska | Istria and Kvarner | Istria | Dajla (Novigrad) |
| wMA | Malvasia Bianca Lunga | Dalmatia | Central and Southern Dalmatia | Ston (Pelješac Peninsula) |
| wMS | Malvasia Bianca Lunga | Dalmatia | Northern Dalmatia | Skradin |
| wCR | Chardonnay | Istria and Kvarner | Istria | Radovani (Višnjan) |
| wCB | Chardonnay | Slavonia and Croatian Danube | Croatian Danube | Banovo brdo (Beli Manastir) |
| wGJ | Welschriesling | Croatian Uplands | Međimurje | Železna gora (Štrigova) |
| wGK | Welschriesling | Slavonia and Croatian Danube | Slavonia | Hrnjavac (Kutjevo) |
| wSD | Sauvignon Blanc | Slavonia and Croatian Danube | Slavonia | Đulovac (Daruvar) |
| wTI | Traminer | Slavonia and Croatian Danube | Croatian Danube | Principovac (Ilok) |
| wRD | Rhine Riesling | Slavonia and Croatian Danube | Slavonia | Đulovac (Daruvar) |
| wFD | Furmint | Croatian Uplands | Međimurje | Lopatinec (Sv. Juraj na Bregu) |
| wPD | Pinot Blanc | Croatian Uplands | Međimurje | Lopatinec (Sv. Juraj na Bregu) |
| Wine Sample | Gallic Acid | p-Hydroxy Benzoic Acid | Syringic Acid | o-Coumaric Acid | Chlorogenic Acid | Caffeic Acid | p-Coumaric Acid | Ferulic Acid |
|---|---|---|---|---|---|---|---|---|
| rPD | 72.5 ± 3.1 c | 7.4 ± 0.7 c,d | 5.9 ± 0.1 e,f | 0.2 ± 0.0 b,c | 0.7 ± 0.1 b,c | 7.9 ± 0.0 d | 15.7 ± 0.4 c | 2.7 ± 0.2 a |
| rPI | 90.3 ± 1.2 a | 5.7 ± 0.2 e | 7.4 ± 0.0 b | 0.2 ± 0.0 b,c | 0.5 ± 0.0 c,d | 12.3 ± 0.1 b | 21.8 ± 0.3 a | 1.5 ± 0.0 b |
| rBS | 86.4 ± 2.1 b | 7.0 ± 0.2 d | 5.4 ± 0.1 g | 0.3 ± 0.1 b | 0.5 ± 0.0 c,d | 11.2 ± 0.1 c | 18.5 ± 0.1 b | 1.5 ± 0.1 b |
| rTD | 53.0 ± 1.2 e | 3.6 ± 0.1 f | 6.4 ± 0.1 d | 0.2 ± 0.0 b,c | 0.6 ± 0.1 c | 4.9 ± 0.1 i | 8.4 ± 0.2 f | 0.8 ± 0.0 c |
| rCD | 11.8 ± 0.2 j | 1.9 ± 0.1 g,h | 5.8 ± 0.0 f,g | 0.2 ± 0.0 b,c | <LOQ | 4.1 ± 0.0 l | 4.7 ± 0.0 h | 0.1 ± 0.0 f |
| rCK | 43.3 ± 0.7 f | 8.0 ± 0.2 b,c | 7.0 ± 0.1 b, c | 0.6 ± 0.1 a | 0.6 ± 0.1 c | 5.4 ± 0.0 h | 7.4 ± 0.1 g | 0.5 ± 0.0 d |
| rCA | 35.2 ± 0.7 g | 2.6 ± 0.1 g | 3.6 ± 0.0 h | 0.1 ± 0.0 c | 0.3 ± 0.0 d | 13.1 ± 0.1 a | 10.2 ± 0.1 e | 0.5 ± 0.0 d |
| rCB | 30.9 ± 0.4 h | 4.0 ± 0.6 f | 4.0 ± 0.5 h | 0.2 ± 0.0 b,c | 1.1 ± 0.1 a | 4.4 ± 0.0 k | 3.3 ± 0.1 i | 0.3 ± 0.1 d,e,f |
| rMK | 38.9 ± 0.8 g | 8.9 ± 0.0 b | 9.2 ± 0.1 a | 0.5 ± 0.0 a | 0.9 ± 0.0 a,b | 5.9 ± 0.0 g | 18.0 ± 0.3 b | 0.5 ± 0.0 d |
| rMA | 37.0 ± 0.6 g | 2.6 ± 0.3 g | 3.0 ± 0.1 i | 0.2 ± 0.0 b,c | 0.6 ± 0.1 c | 6.4 ± 0.0 f | 10.1 ± 0.2 e | 0.4 ± 0.0 d,e |
| rSK | 30.8 ± 0.6 h | 11.2 ± 0.3 a | 6.9 ± 0.1 c | 0.5 ± 0.1 a | 1.0 ± 0.1 a | 4.5 ± 0.0 k | 12.9 ± 0.2 d | 0.5 ± 0.0 d |
| rPM | 25.1 ± 0.4 i | 2.5 ± 0.0 g | 7.4 ± 0.1 b | 0.3 ± 0.0 b | <LOQ | 6.6 ± 0.0 e | 4.7 ± 0.0 h | 0.2 ± 0.0 e,f |
| rPK | 57.9 ± 1.6 d | 1.5 ± 0.2 h | 6.3 ± 0.1 d,e | 0.2 ± 0.0 b,c | <LOQ | 4.7 ± 0.0 j | 3.4 ± 0.0 i | 0.3 ± 0.0 d,e,f |
| wMD | <LOQ | 2.1 ± 0.1 b | 0.3 ± 0.0 b | n.d. | 0.6 ± 0.1 b,c | 1.2 ± 0.0 i | 1.4 ± 0.0 g,h | 0.7 ± 0.0 c |
| wMA | <LOQ | 1.3 ± 0.1 d | 0.3 ± 0.0 b | n.d. | 0.6 ± 0.2 b,c | 1.2 ± 0.0 i | 1.2 ± 0.1 h | 0.3 ± 0.0 g |
| wMS | 2.3 ± 0.1 c | 1.7 ± 0.1 b,c,d | 0.5 ± 0.0 a | 0.5 ± 0.0 | <LOQ | 0.6 ± 0.0 j | 0.1 ± 0.0 i | 0.2 ± 0.0 h |
| wCR | <LOQ | 1.9 ± 0.0 b,c | <LOQ | n.d. | 1.1 ± 0.0 a | 8.7 ± 0.0 a | 4.4 ± 0.1 a | 0.5 ± 0.0 e |
| wCB | <LOQ | 2.1 ± 0.2 b | <LOQ | n.d. | 0.9 ± 0.1 a,b | 4.6 ± 0.0 c | 2.0 ± 0.0 c,d | 0.4 ± 0.0 f |
| wGJ | <LOQ | 1.8 ± 0.1 b,c | n.d. | n.d. | <LOQ | 3.3 ± 0.0 f | 1.6 ± 0.2 e,f,g | 0.4 ± 0.1 f |
| wGK | <LOQ | 0.8 ± 0.2 e | <LOQ | n.d. | n.d. | 1.5 ± 0.0 h | 1.5 ± 0.0 f,g | 0.3 ± 0.0 g |
| wSD | 4.0 ± 0.3 a | 0.7 ± 0.2 e | n.d. | n.d. | <LOQ | 4.9 ± 0.0 b | 2.7 ± 0.0 b | 0.6 ± 0.0 d |
| wTI | 2.4 ± 0.0 c | 0.5 ± 0.2 e | <LOQ | n.d. | <LOQ | 3.3 ± 0.0 f | 1.7 ± 0.0 e,f | 1.1 ± 0.0 a |
| wRD | 3.4 ± 0.1 b | 2.0 ± 0.3 b | <LOQ | n.d. | n.d. | 3.6 ± 0.0 e | 1.8 ± 0.0 d,e | 0.9 ± 0.0 b |
| wFD | <LOQ | 3.3 ± 0.1 a | n.d. | n.d. | 0.3 ± 0.0 c | 2.8 ± 0.0 g | 2.1 ± 0.0 c | 0.2 ± 0.0 h |
| wPD | <LOQ | 1.5 ± 0.1 c,d | n.d. | n.d. | 1.1 ± 0.2 a | 4.0 ± 0.0 d | 1.5 ± 0.0 f,g | 0.2 ± 0.0 h |
| Wine Sample | Catechin | Epicatechin | Procyanindin B1 | Procyanindin B2 | Delphinidin-3-Glucoside | Malvidin-3-Glucoside | cis-Resveratrol | trans-Resveratrol |
|---|---|---|---|---|---|---|---|---|
| rPD | 25.3 ± 0.9 c | 9.5 ± 0.4 e | 56.7 ± 4.1 b | 6.4 ± 0.8 c | 0.2 ± 0.0 d | 6.0 ± 1.1 b | <LOQ | 2.9 ± 0.1 c |
| rPI | 28.1 ± 1.5 b | 15.8 ± 2.6 c | 55.3 ± 3.5 b | 8.8 ± 0.3 b | 0.1 ± 0.0 e | 4.4 ± 0.3 b,c,d | <LOQ | 1.8 ± 0.0 e |
| rBS | 34.5 ± 0.9 a | 29.7 ± 1.6 a | 63.8 ± 0.7 a | 12.0 ± 0.8 a | 0.1 ± 0.0 e | 3.2 ± 0.1 c,d,e,f | <LOQ | 2.4 ± 0.0 d |
| rTD | 15.8 ± 0.3 e | 9.7 ± 0.2 e | 25.4 ± 1.3 e,f | 4.0 ± 0.6 d, e | 0.1 ± 0.0 e | 3.7 ± 0.4 b,c,d,e,f | 0.6 ± 0.0 c | 1.3 ± 0.0 f |
| rCD | 10.5 ± 0.1 f | 3.0 ± 0.0 f | 21.5 ± 0.7 e,f | <LOQ | 0.5 ± 0.0 a | 20.2 ± 0.5 a | n.d. | <LOQ |
| rCK | 19.3 ± 0.6 d | 4.2 ± 0.1 f | 44.6 ± 1.7 c,d | 3.2 ± 0.8 e | 0.4 ± 0.0 b | 3.2 ± 0.3 c,d,e,f | <LOQ | 1.4 ± 0.1 f |
| rCA | 15.5 ± 1.2 e | 11.0 ± 1.2 d,e | 26.0 ± 3.4 e,f | 4.2 ± 0.1 d, e | 0.1 ± 0.0 e | 4.1 ± 1.6 b,c,d | <LOQ | <LOQ |
| rCB | 10.9 ± 0.4 f | 2.6 ± 0.2 f | 13.7 ± 1.2 g | <LOQ | 0.1 ± 0.0 e | 1.4 ± 0.0 f | n.d. | <LOQ |
| rMK | 19.0 ± 0.5 d | 2.7 ± 0.0 f | 39.0 ± 1.8 d | <LOQ | 0.2 ± 0.0 d | 1.6 ± 0.2 e,f | <LOQ | 1.8 ± 0.1 e |
| rMA | 12.5 ± 0.4 f | 4.3 ± 0.0 f | 20.6 ± 1.1 f | <LOQ | n.d. | 2.1 ± 0.2 d,e f | 1.4 ± 0.0 b | 3.2 ± 0.0 b |
| rSK | 12.4 ± 0.8 f | <LOQ | 26.2 ± 1.5 e,f | <LOQ | 0.3 ± 0.0 c | 3.9 ± 0.4 b,c,d,e | <LOQ | 1.4 ± 0.2 f |
| rPM | 25.0 ± 0.9 c | 13.5 ± 0.5 c,d | 27.8 ± 2.2 e | 5.7 ± 0.7 c,d | 0.2 ± 0.0 d | 21.4 ± 1.9 a | 1.6 ± 0.1 a | 6.8 ± 0.1 a |
| rPK | 20.6 ± 0.6 d | 21.5 ± 0.0 b | 45.9 ± 1.7 c | 13.2 ± 0.8 a | 0.1 ± 0.0 e | 4.6 ± 0.3 b,c | <LOQ | 1.8 ± 0.1 e |
| wMD | <LOQ | <LOQ | n.d. | <LOQ | n.d. | n.d. | n.d. | n.d |
| wMA | <LOQ | n.d. | n.d. | <LOQ | n.d. | n.d. | n.d. | n.d |
| wMS | <LOQ | n.d. | <LOQ | 3.8 ± 1.0 b | n.d. | n.d. | n.d. | n.d |
| wCR | 5.4 ± 0.6 a,b | <LOQ | <LOQ | 3.5 ± 0.8 b | n.d. | n.d. | n.d. | n.d |
| wCB | <LOQ | <LOQ | <LOQ | 3.3 ± 0.3 b | n.d. | n.d. | n.d. | n.d |
| wGJ | <LOQ | <LOQ | 5.5 ± 0.3 b | 3.5 ± 0.3 b | n.d. | n.d. | n.d. | <LOQ |
| wGK | <LOQ | <LOQ | <LOQ | 3.0 ± 0.0 b | n.d. | n.d. | n.d. | n.d |
| wSD | <LOQ | <LOQ | n.d. | <LOQ | n.d. | n.d. | n.d. | n.d |
| wTI | 5.1 ± 0.1 b | <LOQ | <LOQ | <LOQ | n.d. | n.d. | n.d. | n.d |
| wRD | <LOQ | n.d. | <LOQ | 8.1 ± 0.2 a | n.d. | n.d. | n.d. | n.d |
| wFD | <LOQ | <LOQ | <LOQ | 3.6 ± 0.2 b | n.d. | n.d. | n.d. | n.d |
| wPD | 6.2 ± 0.2 a | <LOQ | 6.8 ± 0.7 a | <LOQ | n.d. | n.d. | n.d. | n.d |
| Wine Sample | Naringenin | Apigenin | Luteolin | Myricetin | Kaempferol | Quercetin | Rutin | Quercitrin |
|---|---|---|---|---|---|---|---|---|
| rPD | 0.1 ± 0.0 c | 0.5 ± 0.0 d | <LOQ | 11.5 ± 0.2 b | 2.2 ± 0.0 b | 14.3 ± 0.2 d | 0.1 ± 0.0 h | 0.9 ± 0.0 a |
| rPI | 0.1 ± 0.0 c | 0.4 ± 0.0 e | <LOQ | 6.9 ± 0.0 e | 1.1 ± 0.1 d | 7.7 ± 0.1 f | 0.1 ± 0.0 h | 0.6 ± 0.0 d |
| rBS | 0.2 ± 0.0 b | 0.2 ± 0.0 f | <LOQ | 12.1 ± 0.2 a | 0.5 ± 0.0 e | 12.2 ± 0.1 e | 0.1 ± 0.0 h | 0.8 ± 0.0 b |
| rTD | n.d. | 0.2 ± 0.0 f | n.d. | 5.3 ± 0.2 g | <LOQ | 4.7 ± 0.1 g | 0.2 ± 0.0 g | 0.3 ± 0.0 f |
| rCD | 0.1 ± 0.0 c | 0.2 ± 0.1 f | n.d. | 6.3 ± 0.0 f | n.d. | <LOQ | 0.5 ± 0.0 e | n.d. |
| rCK | 0.2 ± 0.0 b | 0.9 ± 0.0 b | <LOQ | 9.5 ± 0.1 c | 2.5 ± 0.1 a | 21.7 ± 0.3 a | 2.3 ± 0.1 b | 0.4 ± 0.0 e |
| rCA | 0.1 ± 0.0 c | 0.1 ± 0.0 f | n.d. | 3.7 ± 0.1 h | <LOQ | 2.3 ± 0.0 i | 0.2 ± 0.0 g | 0.2 ± 0.0 g |
| rCB | n.d. | 0.6 ± 0.0 c | n.d. | 8.9 ± 0.1 d | 0.6 ± 0.0 e | 8.0 ± 0.1 f | 1.2 ± 0.1 d | <LOQ |
| rMK | 0.2 ± 0.0 b | 0.5 ± 0.0 d | <LOQ | 7.0 ± 0.3 e | 2.3 ± 0.0 b | 20.8 ± 0.2 b | 6.1 ± 0.2 a | 0.7 ± 0.0 c |
| rMA | 0.2 ± 0.0 b | 0.2 ± 0.0 f | n.d. | 3.7 ± 0.1 h | <LOQ | 3.1 ± 0.0 h | 0.5 ± 0.1 e | 0.2 ± 0.0 g |
| rSK | 0.1 ± 0.0 c | 1.2 ± 0.1 a | <LOQ | 9.6 ± 0.3 c | 1.5 ± 0.1 c | 18.3 ± 0.7 c | 1.3 ± 0.1 c | 0.4 ± 0.0 e |
| rPM | 0.3 ± 0.0 a | 0.2 ± 0.1 f | <LOQ | 3.9 ± 0.2 h | n.d. | <LOQ | 0.4 ± 0.1 f | <LOQ |
| rPK | 0.1 ± 0.0 c | 0.1 ± 0.0 f | <LOQ | 2.9 ± 0.0 i | n.d. | <LOQ | 0.2 ± 0.0 g | <LOQ |
| Phenolic Compound | DPPH• Scavenging Activity | NO• Scavenging Activity | Lipid Peroxidation Inhibition |
|---|---|---|---|
| Quercetin | 0.87 ± 0.04 d,e | 20.50 ± 0.60 d | 2.06 ± 0.26 d |
| Myricetin | 0.82 ± 0.07 d,e | 20.77 ± 0.63 d | 1.68 ± 0.10 d |
| Gallic acid | 0.33 ± 0.01 e | 12.36 ± 0.93 e | 11.27 ± 0.90 a,b |
| Caffeic acid | 0.62 ± 0.04 d,e | 18.79 ± 0.42 d | 141.76 ± 15.60 * |
| Catechin | 1.34 ± 0.01 c,d | 31.75 ± 0.92 c | 13.05 ± 1.2 a |
| Epicatechin | 1.90 ± 0.01 b,c | 21.26 ± 0.26 d | 10.12 ± 0.57 b |
| Resveratrol | 7.56 ± 0.52 a | 35.08 ± 1.32 b | 5.02 ± 0.34 c |
| Trolox | 2.50 ± 0.75 b | 60.87 ± 1.51 a | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kropek, M.; Štefan, M.B.; Rajkovača, K.; Petković, T.; Cvetnić, M.; Bolanča, T.; Vladimir-Knežević, S. Comparative Phenolic Profiles of Monovarietal Wines from Different Croatian Regions. Appl. Sci. 2023, 13, 3031. https://doi.org/10.3390/app13053031
Kropek M, Štefan MB, Rajkovača K, Petković T, Cvetnić M, Bolanča T, Vladimir-Knežević S. Comparative Phenolic Profiles of Monovarietal Wines from Different Croatian Regions. Applied Sciences. 2023; 13(5):3031. https://doi.org/10.3390/app13053031
Chicago/Turabian StyleKropek, Matija, Maja Bival Štefan, Katarina Rajkovača, Tea Petković, Matija Cvetnić, Tomislav Bolanča, and Sanda Vladimir-Knežević. 2023. "Comparative Phenolic Profiles of Monovarietal Wines from Different Croatian Regions" Applied Sciences 13, no. 5: 3031. https://doi.org/10.3390/app13053031
APA StyleKropek, M., Štefan, M. B., Rajkovača, K., Petković, T., Cvetnić, M., Bolanča, T., & Vladimir-Knežević, S. (2023). Comparative Phenolic Profiles of Monovarietal Wines from Different Croatian Regions. Applied Sciences, 13(5), 3031. https://doi.org/10.3390/app13053031





