Abstract
Background: Human beta-defensins (hBDs) are small cationic peptides of the epithelium with broad antimicrobial and immune response-regulatory activities. hBDs are also related to oncogenesis, and their secretion profiles are affected by radiotherapy treatment. The present study aimed to investigate the oral cavity hBD 1-3 levels in head and neck cancer patients and its relation to radiotherapy treatment. Methods: Sixteen head and neck cancer patients (all with a history of smoking) were included in this study. Periodontal parameters were measured before radiotherapy, and medical information was collected from registries. Oral rinses of the patients were collected before radiotherapy; on the 1st, 3rd, and 6th weeks of radiotherapy; and the 1st month following the end of radiotherapy. hBD 1–3 levels were measured using ELISA. Results: Oral hBD-1 levels increased during radiotherapy at week 6 (p = 0.019). hBD-1 levels returned to pretreatment levels after the end of radiotherapy. No significant change was detected for hBD-2 or hBD-3 levels during or after radiotherapy. Conclusions: The constant expression of hBD-1, which is distinct from the infection and inflammation-dependent expression profiles of hBD-2 and hBD-3, may explain why this peptide is the only one affected by radiotherapy.
1. Introduction
Human beta-defensins (hBDs) are a family of small antimicrobial peptides that are produced by cells in a variety of tissues, including skin, intestine, lung, and oral cavity [1,2,3]. hBDs play a crucial role in protecting the body against various pathogens that include periodontal pathogens, such as Porphyromonas gingivalis, Prevotella intermedia, Tannerella forsythia, and Fusobacterium nucleatum, by acting as the first line of defense against infections [4,5,6,7]. In the oral cavity, hBD-1, hBD-2, and hBD-3 are produced by the epithelial cells lining the oral mucosa, including the gums, cheeks, tongue, and lips. They are also produced by the salivary gland cells and secreted into saliva [1,2,3]. hBD-1 is constitutively expressed in various tissues, including oral and mucosal tissues, and is not generally upregulated by proinflammatory factors, such as cytokines and chemokines [8]. hBD-2 and hBD-3, on the other hand, are typically inducible, meaning that their expression is activated by certain stimuli, such as bacterial infection or inflammation [9].
Periodontitis is a chronic inflammatory disease caused by a combination of factors, marked by an imbalance of bacteria in plaque biofilms and resulting in the gradual deterioration of the structures supporting the teeth [10]. The severity of periodontitis may affect the salivary levels of hBDs, although the exact relationship between the two is not fully understood. Periodontally healthy individuals generally produce hBD-1 and hBD-2 in the granular and spinous layers of the stratified squamous epithelium of the gingiva. hBD-2 is generally observed in the superficial layers of the epithelium, which indicates its interactions with oral microflora [4]. Similarly, hBD-3 is usually found in both the undifferentiated basal lamina of the stratified squamous epithelium and the junctional epithelium. In individuals with periodontal disease, hBD-3 can also be detected in the spinous and outermost layers of the gingival epithelium. It is thought that hBD-3 plays a role in facilitating communication between the gingival epithelium and connective tissue, as well as in linking innate and acquired immunity [4,5,6]. It is worth noting that the antimicrobial activity of hBDs may vary depending on the type of microorganism and the specific conditions under which they are tested. Some studies have suggested that hBDs may be more effective against certain types of bacteria than others, and the exact mechanisms with which they exert their antimicrobial activity are not fully understood [4,5,6,7].
Studies have shown that hBDs may play a role in immunomodulation in inflammatory processes. This immunomodulation has the ability to modulate both natural immunity and acquired immunity [11]. During the active stage of the disease, there is a greater abundance of immunological disturbance caused by hBDs, which is associated with the disruption of the numbers and function of peri- and intra-follicular inflammatory cells [12]. In terms of function, hBDs play a role in regulating the chemotactic activity of various innate immune cells and stimulating the release of various cytokines [13]. These findings imply that there is a complex interplay between the host’s defense mechanisms and their role in maintaining bacterial balance and responding to pathogens [12,14].
Cancer pathophysiology has been shown to be influenced by hBDs. [12,15,16,17,18,19]. Some studies have suggested that hBDs may have anti-tumor properties, including the inhibition of the growth and proliferation of cancer cells and the migration and invasion of breast cancer cells, as well as reducing the formation of new blood vessels, which is an important step in the development and progression of cancer [12,17,18]. However, other studies have suggested that hBDs may also have pro-tumorigenic properties, meaning that they may promote the development of cancer by stimulating the growth and proliferation of certain cancer cells such as cervical cancer [20,21]. The relationship between hBDs and cancer is complex and likely depends on the specific cancer type and the context in which hBDs are expressed.
Head and neck cancer occurs in the tissues in certain parts of the head, throat, and nasal cavity. It accounts for approximately 4% of all cancer types in the United States. It is more common in men than in women, and the risk of developing head and neck cancer increases with age. The risk factors for head and neck cancer include tobacco use (smoking and smokeless tobacco), alcohol consumption, and certain infections, such as human papillomavirus (HPV). The main types of this cancer are mouth, oral cavity, lip, esophagus, nasal cavity, and laryngeal cancers [22]. Previous studies have suggested a potential link between periodontal disease and an increased risk of head and neck cancers, specifically oral cancer. The inflammation and infection associated with periodontal disease may play a role in the development and progression of these types of cancers [23].
Radiotherapy is a standard treatment option for patients with head and neck cancer. Radiotherapy involves the use of high-energy radiation to kill cancer cells and shrink tumors. It can be used alone or in combination with other treatments, such as chemotherapy, to eliminate tumoral tissue and improve patient outcomes. Radiotherapy can be administered in different ways depending on the location and stage of the cancer. External beam radiotherapy is the most common type of radiotherapy for head and neck cancer and involves the use of a machine to deliver high-energy X-rays to the tumor from outside the body. While it is an effective treatment for various cancer types, including head and neck cancer, it can also have side effects, including radiotherapy-induced oral mucositis, changes in taste, dry mouth, difficulty swallowing, and changes in saliva production [24,25,26].
According to the literature, the expression of hBDs may have different characteristics in the presence of tumor tissue, and its secretory regulation may also vary depending on the type and anatomical location of the tumor [18,20,27,28,29,30,31,32,33,34]. To our knowledge, the effect of radiotherapy on oral hBD 1–3 levels have not been described previously. In the present study, we hypothesized that there are differences in the oral hBDs response against radiotherapy, which are based on their distinct expression characteristics and anti-tumorigenic properties. Therefore, we aimed to investigate oral hBD-1, hBD-2, and hBD-3 levels in individuals with head and neck cancer and the changes in their levels in relation to radiotherapy.
2. Materials and Methods
2.1. Study Design and Population
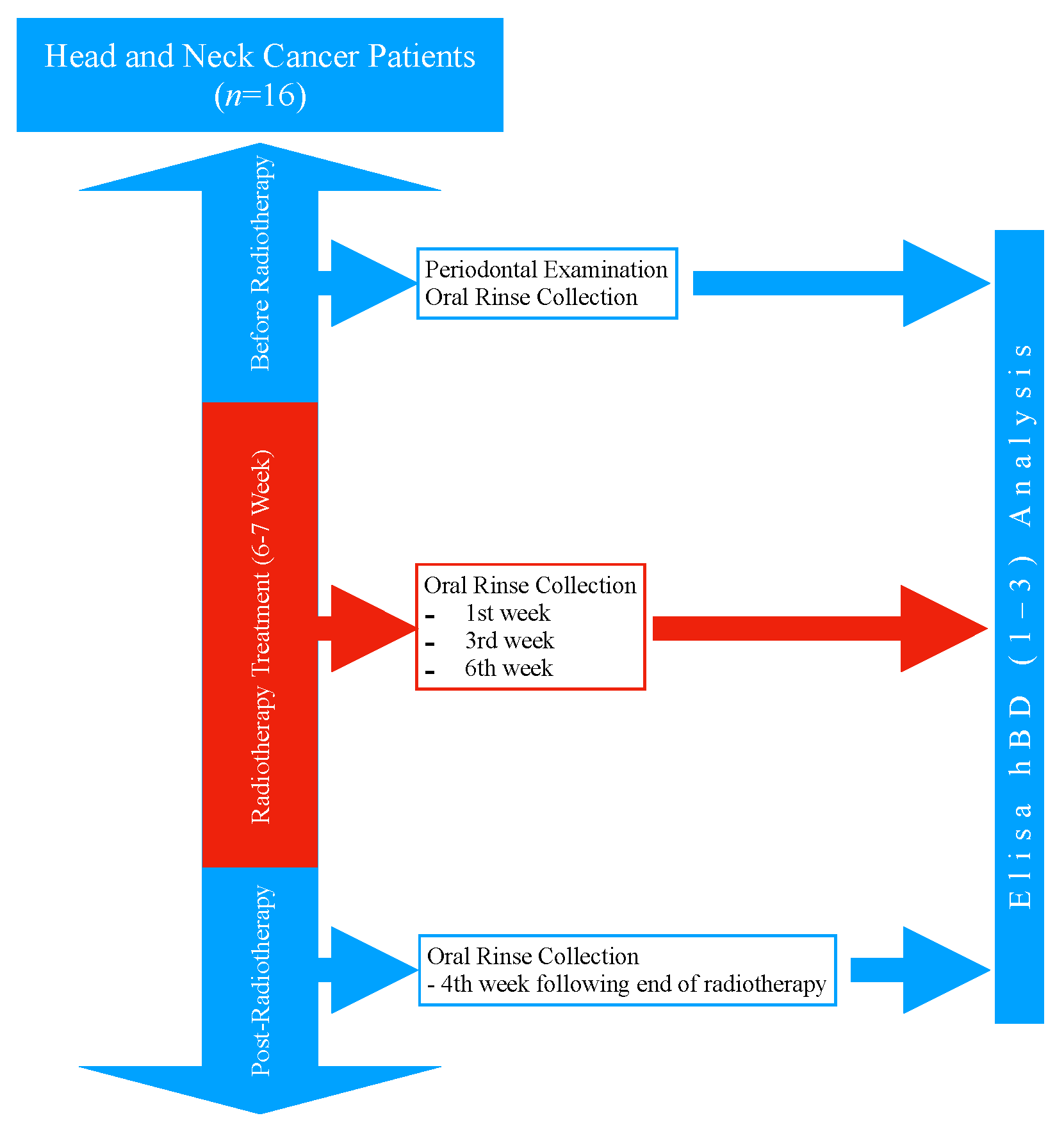
The study design is presented in Figure 1. All head and neck cancer patients who applied to the Radiation Oncology Department of Başakşehir Çam and Sakura City Hospital during June 2021 and August 2022 were invited to the study. The head and neck cancer patients who agreed to take part in the study gave their written and verbal consents (n = 16). The protocol of this study was approved by the Biruni University Ethics Committee (No: 2015-KAEK-67-22-05) and was conducted in accordance with the Helsinki principles. The systemic diseases and smoking habits of the patients were recorded through their medical reports and anamnesis.
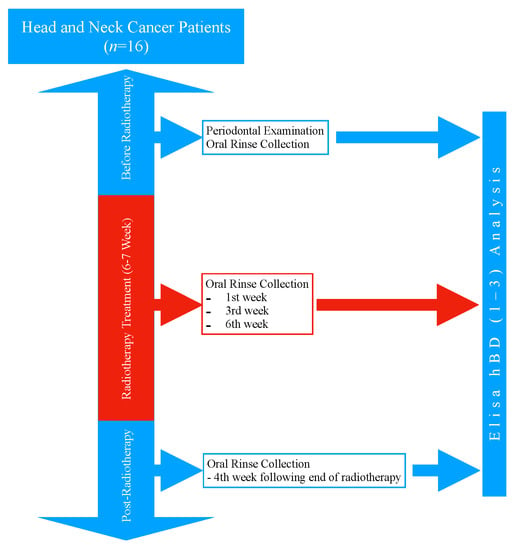
Figure 1.
Study design.
Two expert oncologists (D.K. and İ.H.) designed the treatment plans of the patients based on the pathology and radiography reports. The radiation doses were determined according to the current National Comprehensive Cancer Network® (NCCN®) guideline [35]. Each patient received a personalized chemotherapy regimen based on their medical reports, radiographic images, and NCCN® guidelines [35].
2.2. Periodontal Examination Procedure
All periodontal measurements were performed by a single periodontologist (M.K.) before the radiotherapy (baseline). Teeth that could not be examined due to various reasons (uncertain enamel-cementum junction, highly deformed teeth, teeth that were too posterior due to mouth opening problems) were excluded. Probing depths for each tooth were measured using the Michigan probe (Hu-Friedy®, Chicago, IL, USA). The probe was inserted into the periodontal pocket (the space between the tooth and gum) at each of the six surfaces (mesial, buccal/labial, distal, lingual/palatinal, mesio-lingual or palatinal, disto-buccal or labial) of the tooth, and the depth of the pocket was measured by determining how far the probe could be inserted before it reached resistance. Clinical attachment level (CAL) was used to measure the amount of support for each tooth provided by the periodontal tissue. It was determined by subtracting the depth of the pocket from the distance from the cementum-enamel junction (CEJ) to the bottom of the pocket. The CEJ was used as a reference point, and in cases where it was not visible, a specialist periodontist (MK) determined the reference point. Bleeding from probing (as a percentage) and plaque index (PI) were also measured from the four surfaces of each tooth. These measurements were used to evaluate the presence and amount of bleeding and plaque [26,36].
The periodontal disease classifications of the patients were determined as stages (I, II, III, IV) and grades (A, B, C), which was based on the 2018 Periodontal Disease Classification [10]. Staging was classified to determine the severity of the disease based on the extent of the destruction of the supporting structures of the teeth taking into account the CAL at the sites with the greatest loss, radiographic bone loss, tooth loss due to periodontitis, probing depths, pattern of bone loss, furcation lesions, number of remaining teeth, tooth mobility, ridge defects, and masticatory dysfunction:
Stage I: Mild periodontitis with minimal loss of attachment and bone.
Stage II: Moderate periodontitis with moderate loss of attachment and bone.
Stage III: Severe periodontitis with significant loss of attachment and bone.
Stage IV: Very severe periodontitis with very significant loss of attachment and bone.
The grading was classified to determine the risk of periodontal disease progression taking into account various factors, such as case phenotype, smoking, and hyperglycemia:
Grade A: Slow rate of progression
Grade B: Moderate rate of progression
Grade C: Rapid rate of progression
2.3. Oral Rinse Collection Procedure
Due to the decreased saliva secretion during radiotherapy and the procedural difficulty it may cause in patients with head and neck cancer, the oral rinse was chosen as the sample [25]. The oral rinse samples were collected before radiotherapy at the 1st week, 3rd week, and 6th week of radiotherapy, and at the 4th week following the completion of radiotherapy. All patients were advised not to eat or drink anything for 1 h before collecting the oral rinse sample. The oral rinse samples were collected as follows [25]: The patients rinsed their oral cavity with clean water for 30 s and spit out; after a 1-min resting period, they were instructed to rinse with 5 mL of sterile, distilled water for 30 s; the patients spit the rinse water into a separate container, and the oral rinse samples were transferred to eppendorf tubes with a sterile syringe; all samples were stored at −70 °C until the day of analysis.
2.4. The Radiotherapy Treatment Procedure
Radiation therapy was carried out in accordance with the NCCN® guidelines [35]. A personalized, patient-specific, thermoplastic mask was crafted for immobilization during treatment, and the region between 1 cm above the frontal sinus and the manubrium stern was simulated to be within the range of a 3-mm cross-section using a Toshiba Aquilian computed tomography (CT) simulator. The obtained CT images were then transferred to the Monaco treatment planning system for further analysis. To determine the treatment areas, the CT images were fused with PET/CT scans taken for the disease’s staging. An expert in radiation oncology contoured the fused images to establish target areas and critical structures in the head and neck region.
For the patients with head and neck cancer, treatment of the primary tumor and affected lymph nodes was scheduled at 1.8–2.0 Gy/day and a total of 70–72 Gy. In many head and neck cancers, 1.8–2 Gy/day for regional lymph nodes and a total of 50.4–54 Gy radiotherapy were planned, as comprising the entire neck. In the case of lymph node involvement in the imaging, the dose was increased to 66 Gy. In postoperative cases, 60 Gy was targeted for R0 resection, 66 Gy for R1 resection, and 70–72 Gy for R2 resection for primary tumor targets. A total of 50.4–54 Gy radiotherapy treatment plans were scheduled for all neck lymphatics after surgery at 1.8–2 Gy/day. In cases of pathological lymph node involvement, the dose was elevated to 60 Gy. The doses were limited for high-risk organs to preserve normal structures.
For the treatment, the IMRT plans were approved by the radiation oncologist, which permitted RT to be delivered to clinical target volumes while ensuring that the high doses did not exceed 95% of the dose defined in the treatment area, and that areas hotter than 7–10% of the total dose were not formed. Each patient was set up on the Elekta Synergy linear accelerator, and the patients were treated with 6 MV energy and photons.
2.5. Beta-Defensin Analysis
Commercial ELISA kits were used to determine the concentrations of human beta-defensins 1–3 (Peprotech, catalog numbers 900-M202, 900-M172, 900-M210). Standards for hBDs 1–3 were provided in the ELISA kit. The analyses were performed according to the manufacturer’s instructions with the exception that samples and standards were assayed in duplicates, not triplicates, due to the low volume of the samples. A 1:30 dilution was used for the hBD-1 analysis, and hBD-2 and hBD-3 were analyzed from undiluted samples. The absorbances of the samples were read at 405 nm wavelength with a Multiskan FC Microplate photometer (Thermo Fisher Scientific catalog number 51119000), and the data were treated with SkanIt software V.3.1.0.4 (Thermo Fisher Scientific catalog number 5187139). Readings were obtained at 5-min intervals as instructed by the manufacturer, and readings from 25 min were chosen for further analysis. In order to account for any levels of hBD-1, hBD-2, and hBD-3 that were below the limit of detection (LOD) of the assay, values under the LOD were substituted with half of the LOD of each ELISA kit [37]. Specifically, hBD-1 levels below the LOD were substituted with 2 pg/mL, hBD-2 levels below the LOD with 8 pg/mL, and hBD-3 levels below the LOD with 31 pg/mL.
2.6. Statistical Analyses
The statistical analyses were performed using SPSS V26.0 (IBM, Armonk, North Castle, NY, USA). The statistical differences in the hBD 1–3 levels between visits were analyzed with the Friedman test. The Wilcoxon signed-rank test was used for the post-hoc comparisons of the hBD-1 levels between each visit. Five outliers (6.25%) were removed during the Wilcoxon tests. A p value of <0.05 was accepted as significant.
3. Results
3.1. The Demographic and Periodontal Characteristics of the Patients
This study included 16 head and neck cancer patients with a mean age of 51.8 ± 14.03 (3 female and 13 male). All patients had a history of smoking at least 10 cigarettes per day for a minimum of 5 years. Table 1 presents the systemic conditions and medications used by the patients. It was reported that seven of the patients did not have any systemic diseases besides their oncological conditions. Four of the patients had diabetes, three had chronic obstructive pulmonary disease, two had cardiovascular disease, and two had hypothyroidism. The medications used by the patients were as follows: Four of the patients used Metformin hydrochloride, three used ipratropium bromide, three used acetylsalicylic acid, two used levothyroxine sodium, one used atorvastatin, and one used metoprolol.

Table 1.
Patient characteristics.
The patients were exposed to a mean dose of 6481.75 (±567.21) cGy of radiation therapy. The patients received a mean of 31.12 (±3.44) days of radiation therapy. Eight of the patients (50%) received chemotherapy in addition to radiotherapy. Cisplatin was used in concurrent chemotherapy with radiotherapy. Neo-adjuvant chemotherapy, including gemcitabine and three cycles of Cisplatin, was administered to the patients with nasopharyngeal cancer.
The initial clinical periodontal parameters of the patients are specified in Table 2.

Table 2.
The initial clinical periodontal parameters of the patients. Abbreviations: SD: Standard deviation.
3.2. Oral hBD-1, hBD-2, and hBD-3 Levels during and after the Radiotherapy
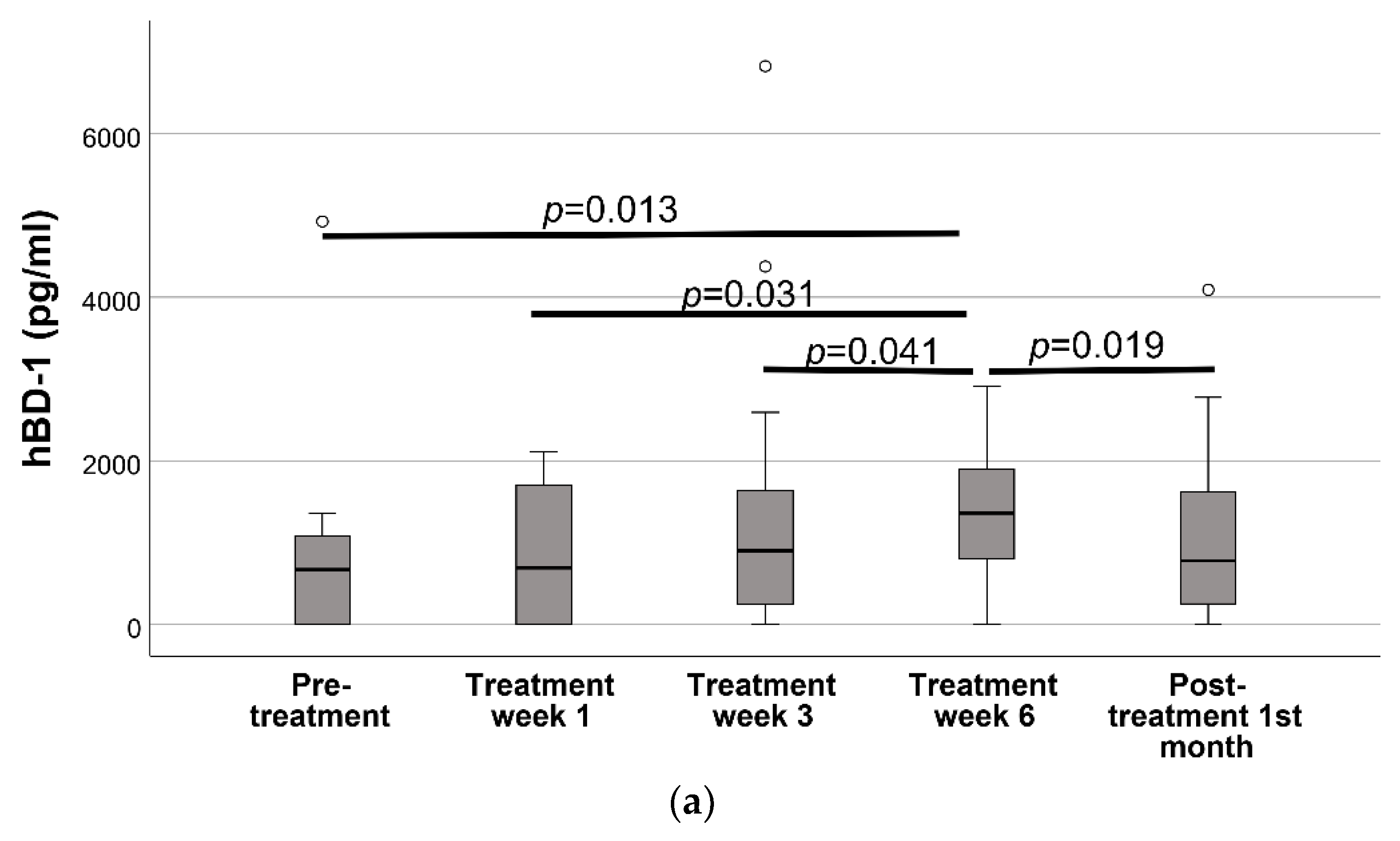
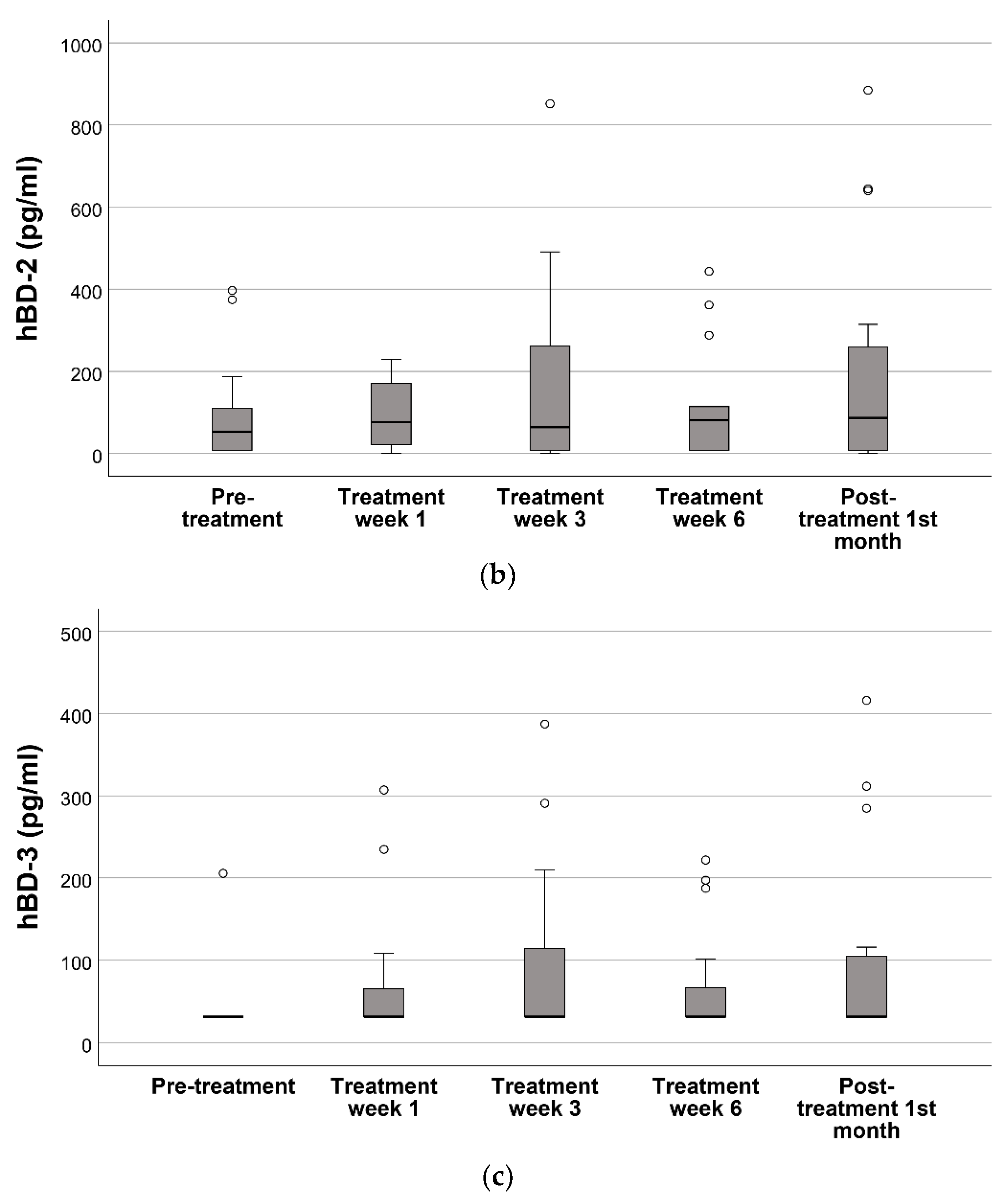
Out of a total of 80 samples, the hBD-1 and hBD-2 levels of 23 samples (28.7%) and hBD-3 levels of 61 samples (76.3%) were below the LOD. Oral hBD-1 levels increased constantly with the initiation of the radiotherapy and reached its peak levels at treatment week 6 (p = 0.013). These levels decreased after the completion of the radiotherapy (p = 0.019) (Figure 2). No difference was observed in the oral hBD-2 and hBD-3 levels during or after the radiotherapy (p > 0.05). According to the Friedman test, only the hBD-1 levels differed between the groups (p = 0.034).
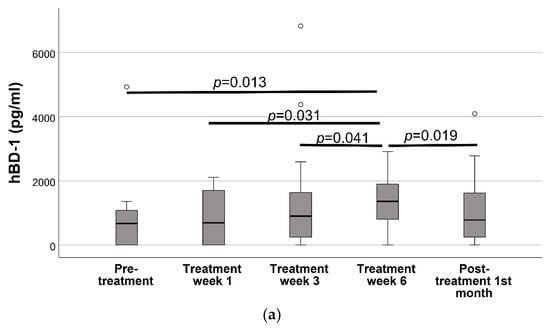
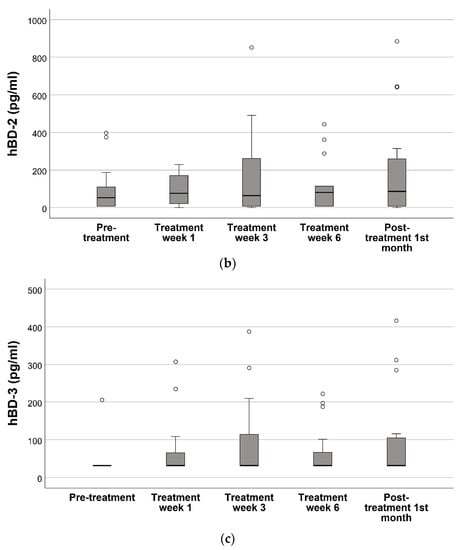
Figure 2.
Oral cavity hBD levels before radiotherapy, 1st week of radiotherapy treatment, 3rd week of radiotherapy treatment, 6th week of radiotherapy treatment, and 1 month following the end of radiotherapy treatment *. (a) hBD-1, (b) hBD-2, (c) hBD-3. * Statistical differences in the hBD 1–3 levels between visits were analyzed with the Friedman test. The Wilcoxon signed-rank test was used for the post-hoc comparisons of the hBD-1 levels between each visit. As the Friedman test did not indicate any statistical difference for hBD-2 and hBD-3, pairwise analysis was not performed.
4. Discussion
Various studies indicated that the expressions of hBDs, which play a role in the health of periodontal tissues and the maintenance of oral mucosal integrity, can be suppressed during the development and progression of cancer [1,9,30,34,38,39,40,41]. The characterization of hBDs in the oral cavity is important in the presence of head and neck cancer that is directly or indirectly related to the oral region, as well as in disease therapeutic processes, such as radiotherapy with a local irritant effect. The present study is, to the best of our knowledge, the first one to investigate the relationship between hBD levels in oral fluids and radiotherapy in head and neck cancer cases.
The prevalence of head and neck cancer in the general population is quite low; therefore, the number of participants was limited. Although a relatively small number of patients (n = 16) were included in this study, all patients were followed up during and after radiotherapy, which is the main strength of the present study. A periodontally healthy control group was not included into the study design. Since periodontal status may have an effect on intraoral hBD levels, the clinical periodontal parameters of the patients were measured before treatment. Clinical periodontal parameters after radiotherapy were not included in this study since the study covered an average radiotherapy period of 6 weeks, which is a relatively short period of time for the significant progression of periodontal disease. A decreased salivary flow due to the radiotherapy process is frequently encountered in patients with head and neck cancer [42]. Gingival crevicular fluid, another intraoral fluid, can only provide information about the site of the tooth from which the sample is collected and its practically unsuitable for head and neck cancer patients due to its procedural difficulty. Therefore, considering the ease of application and standardization advantages, oral rinse analysis was preferred in our study.
Upon examination of our study cohort, it is apparent that the participants utilize a plethora of medications based on their systemic conditions. There is a scarcity of research on the impact of these medications on human beta-defensin (hBD) secretion. According to the literature, specific medications may alter hBD levels. For instance, the utilization of metformin, a medication commonly used in diabetic circumstances, may enhance hBD expression [43]. Nevertheless, further studies are needed to comprehensively understand the influence of these and other medications on hBD secretion.
In our study, a statistically significant increase in oral hBD-1 secretion was observed during the radiotherapy. It is an interesting finding that the hBD-1 secretion, which is generally stable and unaffected by inflammation [1,8], increased during the radiotherapy. Although some studies have been conducted to examine the effects of radiation on hBD expression, studies directly examining the effects of radiation therapy on hBD expression are quite limited [44,45,46,47]. Koerdt et al. showed that radiation therapy applied to oral cancer patients reduced cutaneous hBD secretion [44]. In this study, the decrease in cutaneous hBD expression was interpreted as potentially increasing the risk of microbial infection [44]. On the other hand, there are also studies in the literature showing that ultraviolet B (UVB) radiation can directly induce hBD secretion from keratinocytes [45,46,47,48]. When our study’s findings are evaluated together with the literature data, it becomes clear that radiation creates a complex background that can affect oral hBD-1 expression by changing the host–microbial balance and through direct effects on tissues. Considering the anti-tumorigenic effects of hBD-1 [49], there is a need for further studies investigating the radiotherapy-stimulated hBD-1 activation as part of cancer treatment.
According to our findings, there were a high number of samples that were under the LOD of the applied analysis protocols (hBD-1 28.7%, hBD-2 28.7%, and hBD-3 76.3%). hBDs are the predominant form of antimicrobial peptides found in periodontal tissues. The quantitative detection of hBDs in oral fluids has been applied in various oral conditions, and the loss of hBD is not routinely encountered [41,50,51,52,53,54,55]. Studies have shown that individuals with periodontitis could have higher levels of hBDs in their saliva compared to individuals without periodontitis [52,53,54,56,57]. Additionally, research has also shown that there is a positive correlation between the severity of periodontitis in diabetic individuals and hBD levels, meaning that as the severity of periodontitis increases, so do hBD levels [56,57]. Pereira et al. found that patients with periodontitis displayed elevated levels of HBD-2 and hBD-3 in their oral fluids compared to healthy individuals [52,53]. Vardar-Sengul et al. also reported that mRNA gene expression of hBD-1 is up-regulated in patients with chronic periodontitis [54]. When we evaluate together the literature data and the periodontal condition of our own study group, we are interpreting the values detected below the LOD as being a loss (absence or very low levels of hBD expression). Our study raises the potential that the loss of hBD in oral fluids, including before radiotherapy, may be caused by cancer. There are various studies showing that there is a loss of hBD in oral cancers [18,29,30,40,58,59,60,61]. It was demonstrated that hBD-1 expression is significantly lower in oral squamous cell carcinoma, a common type of head and neck cancer, compared to healthy gingival tissue [18,29,30,60]. Besides oral cancer, hBD-1 release was found to be significantly down-regulated in epithelial renal cancer, prostate cancers, hepatocellular cancer, lung cancer, cutaneous squamous cell carcinoma, and other cancer types including basal cell of the skin [40,58,61,62,63,64]. Donald et al., 2003 concluded that hBD-1 is not expressed in 90% of renal cancer patients and 82% of prostate cancer patients [40]. hBD-1 mRNA is known to be ubiquitously actively secreted in healthy individuals including mucosal tissues [34,65]. Similar to hBD-1, hBD-2 has also been shown to have lower expression in oral squamous cell carcinoma (OSCC) than in healthy oral cells [30]. Meyer et al., 2004 found that hBD-2 concentrations may decrease in patients with tonsillar cancer in the head and neck cancer group when compared to normal tissues in a study on patients with tonsillar cancer in the head and neck cancer group [59]. On the other hand, the expression of hBD-3 in cancer cases is contradictory. Although some studies involving squamous cell oral cancers have shown that hBD-3 is released more than hBD-1 and hBD-2, there are also opposite findings in the literature. [20,28,29,30,31]. Among the study participants of ours, a significant loss of hBD-3 was found (76.3%) in the pre-radiotherapy stage. It is suggested that human hBD-3 helps to facilitate communication between the gingival epithelium and connective tissue, as well as plays a role in linking innate and acquired immunity [45]. It has been shown that hBD-3 can inhibit the development of periodontitis by suppressing the inflammatory response in macrophages and reducing cytokine secretion [45,64,65]. In addition to playing important roles in the preservation of periodontal and mucosal health, the therapeutic contribution of hBD-3 in the treatment of periodontal disease has been studied for years [46,66,67,68]. Transplantation of hBD3-expressing periodontal ligament cell sheets may have the ability to stimulate the growth of new bone, making it a potential agent for promoting bone repair and regeneration [46]. Li et al. found that modified human periodontal ligament cells with the hBD-3 gene showed strong expression of genes related to bone formation and high levels of calcium deposition [68]. These findings also suggest that the loss of hBD-3 that may occur as a result of cancer can increase the risk of periodontal disease progression and tissue deterioration. Finally, hBD expression in cancer cases varies depending on the type and location of the cancer, and hBD suppression may be caused by a variety of complex mechanisms. While DNA methylation is involved in the secretory regulation of hBD-2 in people with oral cancer, the Nuclear factor kappa b (NF-κB) pathway may be involved in the regulation of hBD-2 in people with esophageal cancer [32,67]. hBD-1 is suppressed by the EGFR-ERK-MYC pathway in colon cancers, while hBD-1 is suppressed by the PAX-2 transcriptional pathway in prostate cancers [66,68]. While the EGFR pathway for hBD-3 is effective in OSCC cases, the tumor-suppressor p-53 protein may be involved in HPV-related oropharyngeal cancer cases [31,39]. When our findings are combined with the data from the literature, it is possible to conclude that the presence of cancer may play a role in the suppression of hBD release in a broader area, including the surrounding tissues, and that highly complex mechanisms are involved in this process.
5. Conclusions
In the limitations of this study, radiotherapy in patients with head and neck cancer regulates oral hBD levels differently. The constant expression of anti-tumorigenic hBD-1, which differs from the infection and inflammation-dependent expression profiles of hBD-2 and hBD-3, may partly explain why only this peptide is affected by radiotherapy.
Author Contributions
Study concept, M.K. and U.K.G.; study design, M.K., D.K., İ.H. and U.K.G.; data acquisition, M.K., E.S. and U.K.G.; quality control of data and algorithms, U.K.G. and E.S., data analysis and interpretation, U.K.G., M.K. and E.S.; statistical analysis, U.K.G. and E.S.; manuscript preparation, M.K., U.K.G. and E.S.; manuscript editing, T.S., D.K., U.K.G. and İ.H.; manuscript review, U.K.G. and T.S. All authors have read and agreed to the published version of the manuscript.
Funding
The present study was funded by the Altınbaş University Research Foundation (Grant No: PB2020-SHMYO-3 2021/02).
Institutional Review Board Statement
Protocols of this study were approved by the Biruni University Ethics Committee (No: 2015-KAEK-67-22-05) and were conducted in accordance with the Helsinki principles.
Informed Consent Statement
Written acceptance of informed consent was obtained from all subjects involved in the study.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Gursoy, U.K.; Könönen, E. Understanding the roles of gingival beta-defensins. J. Oral Microbiol. 2012, 4, 15127. [Google Scholar] [CrossRef]
- Bonass, W.A.; High, A.S.; Owen, P.J.; Devine, D.A. Expression of beta-defensin genes by human salivary glands. Oral Microbiol. Immunol. 1999, 14, 371–374. [Google Scholar] [CrossRef] [PubMed]
- Dunsche, A.; Açil, Y.; Siebert, R.; Harder, J.; Schröder, J.-M.; Jepsen, S. Expression profile of human defensins and antimicrobial proteins in oral tissues. J. Oral Pathol. Med. 2001, 30, 154–158. [Google Scholar] [CrossRef] [PubMed]
- Schibli, D.J.; Hunter, H.N.; Aseyev, V.; Starner, T.D.; Wiencek, J.M.; McCray, P.B., Jr.; Tack, B.F.; Vogel, H.J. The Solution Structures of the Human β-Defensins Lead to a Better Understanding of the Potent Bactericidal Activity of HBD3 against Staphylococcus aureus. J. Biol. Chem. 2002, 277, 8279–8289. [Google Scholar] [CrossRef]
- Ji, S.; Hyun, J.; Park, E.; Lee, B.-L.; Kim, K.-K.; Choi, Y. Susceptibility of various oral bacteria to antimicrobial peptides and to phagocytosis by neutrophils. J. Periodontal Res. 2007, 42, 410–419. [Google Scholar] [CrossRef] [PubMed]
- Ouhara, K.; Komatsuzawa, H.; Yamada, S.; Shiba, H.; Fujiwara, T.; Ohara, M.; Sayama, K.; Hashimoto, K.; Kurihara, H.; Sugai, M. Susceptibilities of periodontopathogenic and cariogenic bacteria to antibacterial peptides, β-defensins and LL37, produced by human epithelial cells. J. Antimicrob. Chemother. 2005, 55, 888–896. [Google Scholar] [CrossRef]
- Lee, S.-H.; Jun, H.-K.; Lee, H.-R.; Chung, C.-P.; Choi, B.-K. Antibacterial and lipopolysaccharide (LPS)-neutralising activity of human cationic antimicrobial peptides against periodontopathogens. Int. J. Antimicrob. Agents 2010, 35, 138–145. [Google Scholar] [CrossRef]
- Williams, W.M.; Torres, S.; Siedlak, S.L.; Castellani, R.J.; Perry, G.; Smith, M.A.; Zhu, X. Antimicrobial peptide β-defensin-1 expression is upregulated in Alzheimer’s brain. J. Neuroinflammation 2013, 10, 898. [Google Scholar] [CrossRef]
- Ganz, T. Defensins: Antimicrobial peptides of innate immunity. Nat. Rev. Immunol. 2003, 3, 710–720. [Google Scholar] [CrossRef]
- Papapanou, P.N.; Sanz, M.; Buduneli, N.; Dietrich, T.; Feres, M.; Fine, D.H.; Flemmig, T.F.; Garcia, R.; Giannobile, W.V.; Graziani, F.; et al. Periodontitis: Consensus report of workgroup 2 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J. Periodontol. 2018, 89, S173–S182. [Google Scholar] [CrossRef]
- Kim, J.; Yang, Y.L.; Jang, S.-H.; Jang, Y.-S. Human β-defensin 2 plays a regulatory role in innate antiviral immunity and is capable of potentiating the induction of antigen-specific immunity. Virol. J. 2018, 15, 124. [Google Scholar] [CrossRef]
- Agarwal, S.; Chauhan, A.; Singh, K.; Kumar, K.; Kaur, R.; Masih, M.; Gautam, P.K. Immunomodulatory effects of β-defensin 2 on macrophages induced immuno-upregulation and their antitumor function in breast cancer. BMC Immunol. 2022, 23, 53. [Google Scholar] [CrossRef]
- Swanson, K.; Gorodetsky, S.; Good, L.; Davis, S.; Musgrave, D.; Stelwagen, K.; Farr, V.; Molenaar, A. Expression of a β-Defensin mRNA, Lingual Antimicrobial Peptide, in Bovine Mammary Epithelial Tissue Is Induced by Mastitis. Infect. Immun. 2004, 72, 7311–7314. [Google Scholar] [CrossRef]
- Agier, J.; Brzezińska-Błaszczyk, E.; Różalska, S.; Wiktorska, M.; Wawrocki, S.; Żelechowska, P. β-Defensin Strengthens Antimicrobial Peritoneal Mast Cell Response. J. Immunol. Res. 2020, 2020, 5230172. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Lu, W. Defensins: A Double-Edged Sword in Host Immunity. Front. Immunol. 2020, 11, 764. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.Q.; Arnold, R.; Fernandez-Golarz, C.; Parrish, A.B.; Almekinder, T.; He, J.; Ho, S.-M.; Svoboda, P.; Pohl, J.; Marshall, F.F.; et al. Human β-Defensin-1, a Potential Chromosome 8p Tumor Suppressor: Control of Transcription and Induction of Apoptosis in Renal Cell Carcinoma. Cancer Res. 2006, 66, 8542–8549. [Google Scholar] [CrossRef] [PubMed]
- Bullard, R.S.; Gibson, W.; Bose, S.K.; Belgrave, J.K.; Eaddy, A.C.; Wright, C.J.; Hazen-Martin, D.J.; Lage, J.M.; Keane, T.E.; Ganz, T.A.; et al. Functional analysis of the host defense peptide Human Beta Defensin-1: New insight into its potential role in cancer. Mol. Immunol. 2008, 45, 839–848. [Google Scholar] [CrossRef]
- Han, Q.; Wang, R.; Sun, C.; Jin, X.; Liu, D.; Zhao, X.; Wang, L.; Ji, N.; Li, J.; Zhou, Y.; et al. Human Beta-Defensin-1 Suppresses Tumor Migration and Invasion and Is an Independent Predictor for Survival of Oral Squamous Cell Carcinoma Patients. PLoS ONE 2014, 9, e91867. [Google Scholar] [CrossRef]
- Bindra, G.K.; Williams, S.A.; Lay, F.T.; Baxter, A.A.; Poon, I.K.H.; Hulett, M.D.; Phan, T.K. Human β-Defensin 2 (HBD-2) Displays Oncolytic Activity but Does Not Affect Tumour Cell Migration. Biomolecules 2022, 12, 264. [Google Scholar] [CrossRef]
- Kesting, M.R.; Loeffelbein, D.J.; Hasler, R.J.; Wolff, K.-D.; Rittig, A.; Schulte, M.; Hirsch, T.; Wagenpfeil, S.; Jacobsen, F.; Steinstraesser, L. Expression Profile of Human Beta-Defensin 3 in Oral Squamous Cell Carcinoma. Cancer Investig. 2009, 27, 575–581. [Google Scholar] [CrossRef]
- Xu, D.; Zhang, B.; Liao, C.; Zhang, W.; Wang, W.; Chang, Y.; Shao, Y. Human beta-defensin 3 contributes to the carcinogenesis of cervical cancer via activation of NF-κB signaling. Oncotarget 2016, 7, 75902–75913. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef]
- Irani, S.; Barati, I.; Badiei, M. Periodontitis and oral cancer-current concepts of the etiopathogenesis. Oncol. Rev. 2020, 14, 465. [Google Scholar] [CrossRef]
- Karam, S.D.; Raben, D. Radioimmunotherapy for the treatment of head and neck cancer. Lancet Oncol. 2019, 20, e404–e416. [Google Scholar] [CrossRef] [PubMed]
- Keskin, M.; Lähteenmäki, H.; Rathnayake, N.; Räisänen, I.T.; Tervahartiala, T.; Pärnänen, P.; Şenışık, A.M.; Karaçetin, D.; Balkanay, A.Y.; Heikkilä, P.; et al. Active matrix metalloproteinase-8 and interleukin-6 detect periodontal degeneration caused by radiotherapy of head and neck cancer: A pilot study. Expert Rev. Proteom. 2020, 17, 777–784. [Google Scholar] [CrossRef] [PubMed]
- Keskin, M.; Kompuinen, J.; Harmankaya, I.; Karaçetin, D.; Nissilä, V.; Gürsoy, M.; Sorsa, T.; Gürsoy, U.K. Oral Cavity Calprotectin and Lactoferrin Levels in Relation to Radiotherapy. Curr. Issues Mol. Biol. 2022, 44, 4439–4446. [Google Scholar] [CrossRef] [PubMed]
- Abiko, Y.; Mitamura, J.; Nishimura, M.; Muramatsu, T.; Inoue, T.; Shimono, M.; Kaku, T. Pattern of expression of beta-defensins in oral squamous cell carcinoma. Cancer Lett. 1999, 143, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Hussaini, H.M.; Lim, P.K.; Rahman, R.A.; Abraham, T.; Cheong, C.S.; Zain, R.B.; Ismail, M.S.; Nam, A.N. Expression of human b-defensin-3 in oral squamous cell carcinoma using tissue microarray—A preliminary study. J. Oral Pathol. Med. 2006, 35, 432–433. [Google Scholar]
- Wenghoefer, M.; Pantelis, A.; Dommisch, H.; Reich, R.; Martini, M.; Allam, J.-P.; Novak, N.; Bergé, S.; Jepsen, S.; Winter, J. Decreased gene expression of human β-defensin-1 in the development of squamous cell carcinoma of the oral cavity. Int. J. Oral Maxillofac. Surg. 2008, 37, 660–663. [Google Scholar] [CrossRef]
- Joly, S.; Compton, L.M.; Pujol, C.; Kurago, Z.B.; Guthmiller, J.M. Loss of human β-defensin 1, 2, and 3 expression in oral squamous cell carcinoma. Oral Microbiol. Immunol. 2009, 24, 353–360. [Google Scholar] [CrossRef]
- Shuyi, Y.; Feng, W.; Jing, T.; Hongzhang, H.; Haiyan, W.; Pingping, M.; Liwu, Z.; Zwahlen, R.A.; Hongyu, Y. Human beta-defensin-3 (hBD-3) upregulated by LPS via epidermal growth factor receptor (EGFR) signaling pathways to enhance lymphatic invasion of oral squamous cell carcinoma. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontology 2011, 112, 616–625. [Google Scholar] [CrossRef] [PubMed]
- Kamino, Y.; Kurashige, Y.; Uehara, O.; Sato, J.; Nishimura, M.; Yoshida, K.; Arakawa, T.; Nagayasu, H.; Saitoh, M.; Abiko, Y. hBD-2 is downregulated in oral carcinoma cells by DNA hypermethylation, and increased expression of hBD-2 by DNA demethylation and gene transfection inhibits cell proliferation and invasion. Oncol. Rep. 2014, 32, 462–468. [Google Scholar] [CrossRef]
- Gao, C.; Yue, W.; Tian, H.; Li, L.; Li, S.; Si, L. Human beta-defensin 2 promotes the proliferation of lung cancer cells through ATP-binding cassette transporter G2. Int. J. Clin. Exp. Pathol. 2016, 9, 5944–5949. [Google Scholar]
- Ghosh, S.K.; McCormick, T.S.; Weinberg, A. Human Beta Defensins and Cancer: Contradictions and Common Ground. Front. Oncol. 2019, 9, 341. [Google Scholar] [CrossRef] [PubMed]
- National Comprehensive Cancer Network® (NCCN®) Guideline (V.1.2021). Available online: https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&cad=rja&uact=8&ved=2ahUKEwin-q-Zvtb8AhVrQ_EDHS0IByoQFnoECBUQAQ&url=https%3A%2F%2Fwww.nccn.org%2Fguidelines%2Fguidelines-process%2Ftransparency-process-and-recommenda-tions%2FGetFileFromFileManager%3FfileManagerId%3D11180&usg=AOvVaw2gJrBKrcFg5BTpeJRmkcvn (accessed on 16 December 2022).
- Ainamo, J.; Bay, I. Problems and proposals for recording gingivitis and plaque. Int. Dent. J. 1975, 25, 229–235. [Google Scholar]
- Lubin, J.H.; Colt, J.S.; Camann, D.; Davis, S.; Cerhan, J.; Severson, R.K.; Bernstein, L.; Hartge, P. Epidemiologic Evaluation of Measurement Data in the Presence of Detection Limits. Environ. Health Perspect. 2004, 112, 1691–1696. [Google Scholar] [CrossRef] [PubMed]
- Chan, L.-P.; Wang, L.-F.; Chiang, F.-Y.; Lee, K.-W.; Kuo, P.-L.; Liang, C.-H. IL-8 promotes HNSCC progression on CXCR1/2-meidated NOD1/RIP2 signaling pathway. Oncotarget 2016, 7, 61820–61831. [Google Scholar] [CrossRef]
- DasGupta, T.; Nweze, E.I.; Yue, H.; Wang, L.; Jin, J.; Ghosh, S.K.; Kawsar, H.I.; Zender, C.; Androphy, E.J.; Weinberg, A.; et al. Human papillomavirus oncogenic E6 protein regulates human β-defensin 3 (hBD3) expression via the tumor suppressor protein p53. Oncotarget 2016, 7, 27430–27444. [Google Scholar] [CrossRef]
- Donald, C.D.; Sun, C.Q.; Lim, S.D.; Macoska, J.; Cohen, C.; Amin, M.B.; Young, A.N.; Ganz, T.A.; Marshall, F.F.; Petros, J.A. Cancer-Specific Loss of β-Defensin 1 in Renal and Prostatic Carcinomas. Lab. Investig. 2003, 83, 501–505. [Google Scholar] [CrossRef]
- Güncü, G.N.; Yilmaz, D.; Könönen, E.; Gursoy, U.K. Salivary Antimicrobial Peptides in Early Detection of Periodontitis. Front. Cell. Infect. Microbiol. 2015, 5, 99. [Google Scholar] [CrossRef]
- Chambers, M.S.; Tomsett, K.L.; Artopoulou, I.I.; Garden, A.S.; El-Naggar, A.K.; Martin, J.W.; Keene, H.J. Salivary flow rates measured during radiation therapy in head and neck cancer patients: A pilot study assessing salivary sediment formation. J. Prosthet. Dent. 2008, 100, 142–146. [Google Scholar] [CrossRef]
- Rodriguez-Carlos, A.; Miramontes, C.E.V.; Marin-Luevano, P.; González-Curiel, I.; Enciso-Moreno, J.A.; Rivas-Santiago, B. Metformin promotes Mycobacterium tuberculosis killing and increases the production of human β-defensins in lung epithelial cells and macrophages. Microbes Infect. 2020, 22, 111–118. [Google Scholar] [CrossRef]
- Koerdt, S.; Steinstraesser, L.; Stoeckelhuber, M.; Wales, C.; Rohleder, N.; Babaryka, G.; Steiner, T.; Wolff, K.-D.; Loeffelbein, D.; Muecke, T.; et al. Radiotherapy for oral cancer decreases the cutaneous expression of host defence peptides. J. Cranio-Maxillofacial Surg. 2016, 44, 882–889. [Google Scholar] [CrossRef]
- Chieosilapatham, P.; Ogawa, H.; Niyonsaba, F. Current insights into the role of human β-defensins in atopic dermatitis. Clin. Exp. Immunol. 2017, 190, 155–166. [Google Scholar] [CrossRef]
- Seo, S.J.; Ahn, S.W.; Hong, C.K.; Ro, B.I. Expressions of β-defensins in human keratinocyte cell lines. J. Dermatol. Sci. 2001, 27, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Horrell, E.W.; D’Orazio, J. UV-independent induction of beta defensin 3 in neonatal human skin explants. F1000Research 2015, 3, 288. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.J.; Rho, Y.K.; Lee, H.I.; Jeong, M.S.; Li, K.; Seo, S.J.; Kim, M.N.; Hong, C.K. The Effect of Calcipotriol on the Expression of HumanβDefensin-2 and LL-37 in Cultured Human Keratinocytes. J. Immunol. Res. 2009, 2009, 645898. [Google Scholar] [CrossRef]
- Sun, C.Q.; Arnold, R.S.; Hsieh, C.-L.; Dorin, J.R.; Lian, F.; Li, Z.; Petros, J.A. Discovery and mechanisms of host defense to oncogenesis: Targeting the β-defensin-1 peptide as a natural tumor inhibitor. Cancer Biol. Ther. 2019, 20, 774–786. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, D.; Yilmaz, N.; Polat, R.; Nissilä, V.; Aydın, E.G.; Rautava, J.; Gürsoy, M.; Gürsoy, U.K. Salivary levels of hBDs in children and adolescents with type 1 diabetes mellitus and gingivitis. Clin. Oral Investig. 2022, 26, 4897–4904. [Google Scholar] [CrossRef]
- Yilmaz, D.; Topcu, A.O.; Akcay, E.U.; Altındis, M.; Gursoy, U.K. Salivary human beta-defensins and cathelicidin levels in relation to periodontitis and type 2 diabetes mellitus. Acta Odontol. Scand. 2020, 78, 327–331. [Google Scholar] [CrossRef]
- Pereira, A.G.; Costa, L.C.M.; Soldati, K.R.; De Abreu, M.H.N.G.; Costa, F.O.; Zandim-Barcelos, D.L.; Cota, L.O.M. Gingival Crevicular Fluid Levels of Human Beta-defensin 2 and 3 in Healthy and Diseased Sites of Individuals with and without Periodontitis. J. Int. Acad. Periodontol. 2020, 22, 90–99. [Google Scholar]
- Pereira, A.L.; Franco, G.C.; Cortelli, S.C.; Aquino, D.R.; Costa, F.; Raslan, S.A.; Cortelli, J.R. Influence of Periodontal Status and Periodontopathogens on Levels of Oral Human β-Defensin-2 in Saliva. J. Periodontol. 2013, 84, 1445–1453. [Google Scholar] [CrossRef] [PubMed]
- Vardar-Sengul, S.; Demirci, T.; Sen, B.H.; Erkizan, V.; Kurulgan, E.; Baylas, H. Human β defensin-1 and -2 expression in the gingiva of patients with specific periodontal diseases. J. Periodontal Res. 2007, 42, 429–437. [Google Scholar] [CrossRef] [PubMed]
- Atalay, N.; Balci, N.; Toygar, H.U.; Yardimci, G.; Gürsoy, U.K. Serum, saliva, and gingival tissue human β-defensin levels in relation to retinoic acid use. J. Periodontol. 2022. [Google Scholar] [CrossRef] [PubMed]
- Komesu, M.C.; Ribeiro, A.E.R.A.; Lourenco, A.G.; Motta, A.C.F. Influence of periodontal condition on levels of human beta defensins 1 and 2 in saliva. J. Microbiol. Exp. 2018, 6, 40–44. [Google Scholar] [CrossRef]
- Xiao, Z.; Ding, W.; Wen, C.; Ge, C.; Liu, L.; Xu, K.; Cao, S. Correlation between salivary HBD-2 and LL-37 expression levels with blood glucose and periodontal status in patients with type 2 diabetes mellitus. Am. J. Transl. Res. 2022, 14, 3286–3291. [Google Scholar]
- Young, A.N.; Amin, M.B.; Moreno, C.S.; Lim, S.D.; Cohen, C.; Petros, J.A.; Marshall, F.F.; Neish, A.S. Expression Profiling of Renal Epithelial Neoplasms: A Method for Tumor Classification and Discovery of Diagnostic Molecular Markers. Am. J. Pathol. 2001, 158, 1639–1651. [Google Scholar] [CrossRef]
- Meyer, J.E.; Harder, J.; Görögh, T.; Weise, J.B.; Schubert, S.; Janssen, D.; Maune, S. Human beta-defensin-2 in oral cancer with opportunistic Candida infection. Anticancer. Res. 2004, 24, 1025–1030. [Google Scholar]
- Wang, X.; Jiang, W.; Duan, N.; Qian, Y.; Zhou, Q.; Ye, P.; Jiang, H.; Bai, Y.; Zhang, W.; Wang, W. NOD1, RIP2 and Caspase12 are potentially novel biomarkers for oral squamous cell carcinoma development and progression. Int. J. Clin. Exp. Pathol. 2014, 7, 1677–1686. [Google Scholar]
- Li, X.; Song, W.; Zhang, M.; Zhao, P. Human β-defensin 1 Functions as a Tumor Suppressor via ER Stress-triggered JNK pathway in Hepatocellular Carcinoma. J. BUON 2021, 26, 1365–1372. [Google Scholar]
- Gambichler, T.; Skrygan, M.; Huyn, J.; Bechara, F.G.; Sand, M.; Altmeyer, P.; Kreuter, A. Pattern of mRNA expression of β-defensins in basal cell carcinoma. BMC Cancer 2006, 6, 163. [Google Scholar] [CrossRef]
- Scola, N.; Gambichler, T.; Saklaoui, H.; Bechara, F.; Georgas, D.; Stücker, M.; Gläser, R.; Kreuter, A. The expression of antimicrobial peptides is significantly altered in cutaneous squamous cell carcinoma and precursor lesions. Br. J. Dermatol. 2012, 167, 591–597. [Google Scholar] [CrossRef] [PubMed]
- Ling, Y.-M.; Chen, J.-Y.; Guo, L.; Wang, C.-Y.; Tan, W.-T.; Wen, Q.; Zhang, S.-D.; Deng, G.-H.; Lin, Y.; Kwok, H.F. β-defensin 1 expression in HCV infected liver/liver cancer: An important role in protecting HCV progression and liver cancer development. Sci. Rep. 2017, 7, 13404. [Google Scholar] [CrossRef] [PubMed]
- Krisanaprakornkit, S.; Weinberg, A.; Perez, C.N.; Dale, B.A. Expression of the Peptide Antibiotic Human β-Defensin 1 in Cultured Gingival Epithelial Cells and Gingival Tissue. Infect. Immun. 1998, 66, 4222–4228. [Google Scholar] [CrossRef]
- Bonamy, C.; Sechet, E.; Amiot, A.; Alam, A.; Mourez, M.; Fraisse, L.; Sansonetti, P.J.; Sperandio, B. Expression of the human antimicrobial peptide β-defensin-1 is repressed by the EGFR-ERK-MYC axis in colonic epithelial cells. Sci. Rep. 2018, 8, 18043. [Google Scholar] [CrossRef]
- Shi, N.; Jin, F.; Zhang, X.; Clinton, S.K.; Pan, Z.; Chen, T. Overexpression of human β-defensin 2 promotes growth and invasion during esophageal carcinogenesis. Oncotarget 2014, 5, 11333–11344. [Google Scholar] [CrossRef] [PubMed]
- Bose, S.K.; Gibson, W.; Bullard, R.S.; Donald, C.D. PAX2 oncogene negatively regulates the expression of the host defense peptide human beta defensin-1 in prostate cancer. Mol. Immunol. 2009, 46, 1140–1148. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).