Abstract
Coffee is of interest to consumers and researchers, mainly due to its stimulating properties and positive effects on the human body. Properties of coffee infusions depend on many factors and the most important ones include the roasting process and the brew preparation method. In this paper, we analyzed infusions prepared with the use of pressure methods (professional: espresso and consumer: moka pot brew), obtained from beans roasted to the medium and dark degree, with the aim of determining the share of individual groups of ingredients in the antioxidative properties of coffee. Tested infusions showed various characteristics that were partly related to different parameters of their preparation. In general, a greater amount of phenolic compounds was extracted in moka brews. Espresso, on the other hand, exhibited greater antioxidant activity per portion of the brew and a greater ability to quench free radicals. The degree of roasting had an uneven effect on the antioxidant activity of the brews, depending on the method and its mechanism of antioxidant action. The share of activity of the particular groups of antioxidants in overall activity varied between all methods; the most pronounced changes were observed in the espresso brews. The results indicate, however, that the activity of phenols and melanoidins is based on a mixed mechanism of antioxidant action, but in the case of the latter, the mechanism of hydrogen atom transfer may have an advantage.
1. Introduction
Coffee is the fruit seed of the Coffea trees, where Coffea arabica and Coffea canephora var. Robusta are of the most economic importance [1]. For many centuries, they have been used to prepare infusions, which are more and more popular for their pleasant sensory properties and the stimulating effect associated with the presence of caffeine [2,3,4]. In general terms, the coffee processing consists of fruit harvest, separating the seeds from the fruits, roasting and packing [1]. Each of these stages has an impact on the chemical composition of the beans, but the roasting effect has undoubtedly the greatest. In this process, high temperature (180–240 °C) is applied to green beans, during which degradation of some natural compounds occurs but also the formation of compounds responsible for the color and aroma of coffee [4,5,6].
Coffee beans are rich in biologically active compounds, in addition to the most well-known, caffeine, including phenolic compounds, trigonelline, diterpenes and micro- and macroelements [7,8]. The most widely discussed coffee components in the literature are recently chlorogenic acids—a group of esters of quinic acid and derivatives of trans-cinnamic acid, mainly caffeic, p-coumaric and ferulic acids [9,10].
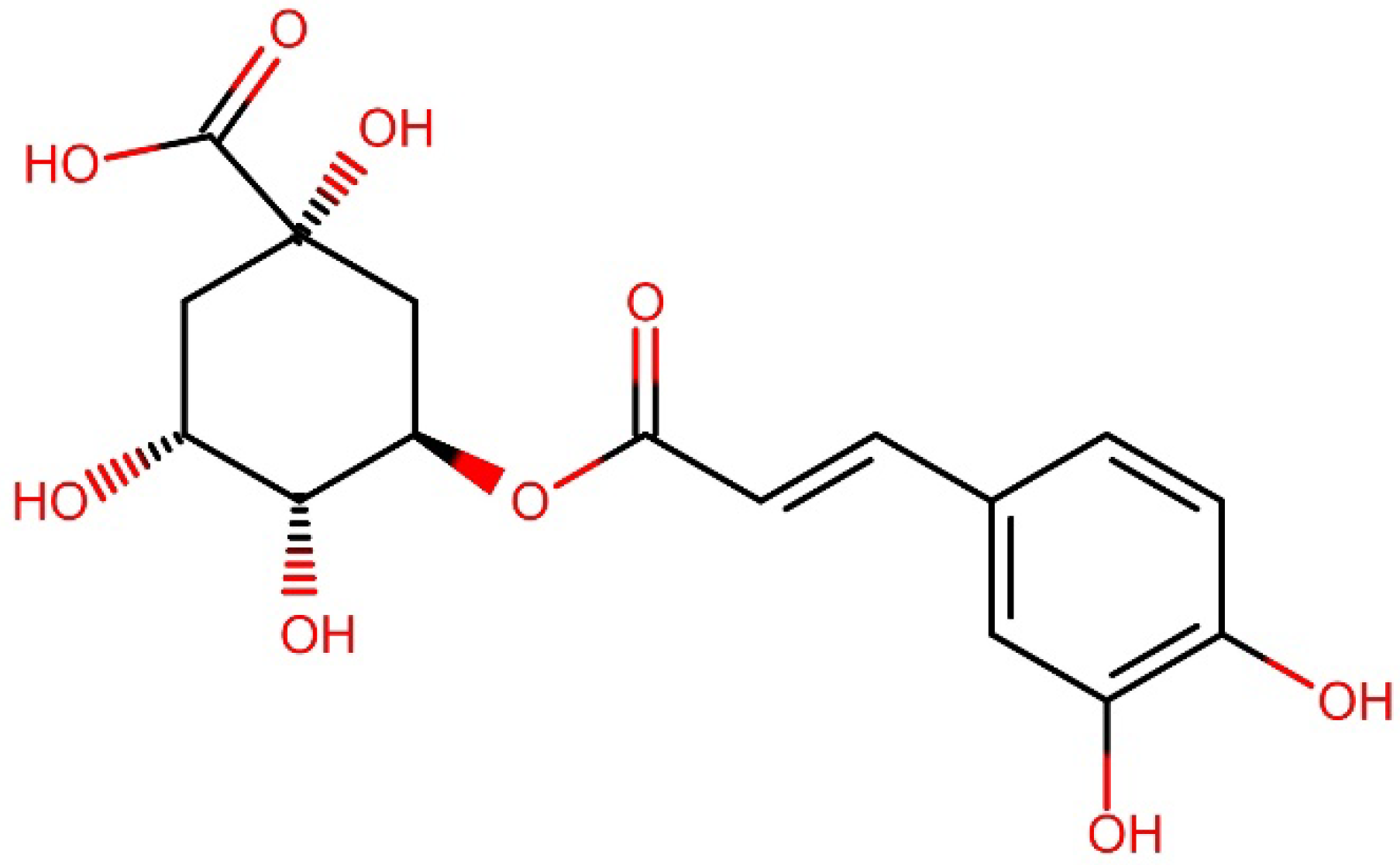
Figure 1 shows the chemical structure of 5-coffeolquinic acid, which is also known as chlorogenic acid due to its abundance in coffee. These compounds, as well as other phenolic compounds, are mainly attributed to the antioxidant activity of coffee brews; however, their content during the roasting process is reduced to as little as 10% of the initial value, and the observed decrease depends on the roasting time/degree [11]. On the other hand, during this process, melanoidins are formed—high molecular weight, water-soluble products of the Maillard reaction [6,12,13]. Numerous studies have shown that melanoidins are responsible not only for the color of coffee but also significantly contribute to its antioxidant effect [4,12].
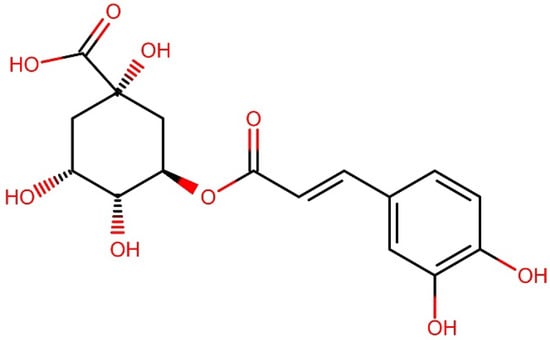
Figure 1.
Chemical structure of 5-coffeolquinic acid.
The method of preparation also affects the health and sensory quality of consumed infusions [4]. Currently, there are many methods of brewing coffee that differ in parameters and complexity. Among methods of brewing that were mostly investigated are French Press, drip/poured coffee, Turkish coffee and cold brew [11,14,15,16,17,18,19,20,21]. Much research has been performed on the influence of the brew preparation method on their characteristics, but relatively little attention has been paid to the moka pot method. During preparation, there is an increased pressure, which may explain the frequent information from producers of moka pots about the similarity of the moka infusion to the professional espresso coffee. However, the preparation of moka brew is easier and does not require the use of expensive equipment. Taking into account the above information, the aim of this study was to assess the impact of the bean roasting degree and the method of brew preparation on the characteristics and antioxidant activity with particular emphasis on the participation of various groups of antioxidants in the activity measured with different mechanisms of antioxidant action.
2. Materials and Methods
2.1. Preparation of Coffee Brews and Extracts
Coffee beans (Coffea arabica; El Morito, Guatemala; washed) were obtained from local roastery LaCava (Wolica, Poland). The beans underwent two different treatment procedures in order to obtain both medium and dark roast. Sample roaster IKAWA Pro V2 (IKAWA Ltd., London, UK) was used, and approximately 400 g of coffee per roast were prepared. Beans were ground by a local expert barista using a Mahlkönig EK 43 burr grinder (Hemro International AG, Bachenbülach, ZH, Switzerland). At the roastery, six espresso brews were prepared from a 16 g sample of ground coffee, each by a professional barista using an espresso machine (model Strada, La Marzocco, Florence, Italy), and stored separately in hermetic containers. Three moka brews were prepared using an electric moka pot designed to produce a brew equal to two espresso shots (Moka Elettrika 2, Bialetti, Coccaglio, Italy). Portions of ground coffee (10 g or 12 g for medium or dark roasted coffee, respectively) and 90 mL of hot distilled water were used in this procedure, and brewing was terminated when steam appeared in the central column of the pot.
Acetone extracts were prepared from ground coffee beans: 5 g of coffee was extracted with 50 mL of acetone solution (70%) for 24 h at 4 °C. Before and after this time, samples were shaken for 30 min at ambient temperature. The mixture was then filtered and stored at −20 °C prior to analysis. Extracts from different brews were also prepared: 30 mL of moka brews and whole shots of espresso were diluted with acetone to 100 mL. After 30 min, the solutions were filtered and stored under the same conditions. For each combination of roast degree and preparation technique, two extracts were prepared.
2.2. Characterization of Grounded Coffee Beans
The color of roasted beans was measured twice. First, the color of whole beans was analyzed using a colorimeter calibrated to SCAA standards (model CM-100, Lighttells, Zhubei, Taiwan) in order to determine roast degree. The aforementioned standard specifies three main roast degrees, namely, light, medium and dark, with values of 100–71, 70–41 and 40–0, respectively. Within these general groups, subgroups are established, e.g., extremely light, very light and light. In the laboratory, the color of ground beans was measured using a colorimeter calibrated on a standard plate (Chroma Meter CR-400, Konica Minolta Co., Ltd., Tokyo, Japan). Measurements in the laboratory were performed four times for each sample, and mean values were calculated for each color parameter.
2.3. Characterization of Coffee Brews and Extracts
Two brews from each combination were analyzed. All analyses were performed twice for each brew. The color of coffee brews was analyzed using a spectrophotometer calibrated with white and black standards (CM-3600d, Konica Minolta). The pH value was measured using a pH-meter (pH 50+ DHS, XS instruments, Carpi, Italy) calibrated on buffer solutions (pH 4 and 7) prior to analysis. The total soluble solids were determined by means of a refractometer.
The melanoidin content was determined based on a method presented by Bekedam et al. [22]. Two portions (30 mL) of moka brews and two shots of espresso were transferred to dialysis tubes (nominal molecular cut-off of 12 kDa). Dialysis was performed in water at 4 °C; water was changed every 24 h. The absorbance of discharged water was measured at 420 nm. The dialysis was performed until the water absorbance was lower than 0.015. For espresso brews, dialysis lasted 7 days and for moka brews—6 days. The solutions from the tubes were transferred to dried and weighed containers and lyophilized (Alpha 1-4 LSCplus, Martin Christ Gefriertrocknungsanlagen GmbH, Osterode am Harz, Germany; conditions: 6 mbar, 25 °C, 48 h). Samples were weighed immediately after lyophilization and stored in a desiccator.
The amount of phenolic compounds in acetonic extracts was determined by the Folin-Ciocalteu method, as described by Singleton and Rossi [23]. Briefly, in a test tube, 3.95 mL of distilled water and 250 µL of Folin-Ciocalteu reagent were mixed with 50 μL of appropriately diluted acetone extract. After 4 min, 750 µL of sodium carbonate solution (0.2 g/mL) was added. After incubation (2 h), the absorbance of samples was measured at 765 nm using a spectrophotometer (UVmini 1240, Shimadzu, Kyoto, Japan). GA was used to draw the standard curve. Analysis was performed twice for each extract. The results were calculated in mg GA per 100 g of bean dry matter (DM) and per portion of the brew equivalent to one espresso shot (32 mL for espresso and 35 mL for moka brew).
2.4. Determination of Antioxidant Activity with CUPRAC Method
For this analysis, the original method of Özyürek et al. [24] was used. First, 1 mL of aquatic solutions of copper(II) chloride (1.704 g CuCl2*2H2O/mL) and ammonium acetate (77.08 mg/mL) and ethanolic solution of neocuproine (1.56 mg/mL) were mixed in the test tube. The diluted extract or melanoidin solution (1 mg/mL) were added in a total volume of 1.1 mL and mixed. After 30 min, the absorbance was measured using a spectrophotometer at 450 nm. The analysis was performed twice for each extract or solution (a total of four repetitions per sample). The solution of Trolox in acetone (70%) was used as a standard solution to obtain a calibration curve. The results were expressed as mg Trolox per 100 g of beans’ DM and per portion of the brew equal to one espresso shot. The standard solution of chlorogenic acid was examined and used to calculate overall antioxidant activity of phenolic compounds (as determined by the Folin-Ciocalteu method). Based on the acquired data, the contribution of coffee components (phenolics, melanoidins, others) to antioxidant activity of the whole extract was calculated.
2.5. Determination of Antioxidant Activity Using ABTS Radical Cations
According to Re et al. [25], in the test tube, 4 mL of prepared ABTS•+ solution and 40 µL of diluted extract/melanoidin solution (1 mg/mL) were mixed. After 6 min of incubation, absorbance of the samples was measured at 734 nm. The calibration curve was prepared using Trolox solutions in 70% acetone. The results were calculated in the way described for the CUPRAC method. The chlorogenic acid solution was analyzed in order to calculate the overall antioxidant activity of phenolic compounds indicated by the Folin-Ciocalteu method.
2.6. Determination of Antioxidant Activity with Crocin Bleaching Assay (CBA)
The analysis was based on the method described by Bountagkidou et al. [26] and Di Majo et al. [27]. First, 2 mL of crocin solution (12 mL of 1 mg/mL solution diluted to 100 mL with PBS) and 1 mL of diluted extract/melanoidin solution (1 mg/mL) was heated in a water bath (37 °C) for 5 min. Next, 1 mL of AAPH (in PBS; 8 mg/mL) was added and the absorbance of the sample was measured using a spectrophotometer after 75 min of incubation at 443 nm. Control and blind samples, as well as Trolox and chlorogenic acid standard solutions (in 70% acetone solution), were analyzed. The results were expressed as percentage of inhibition of crocin degradation by analyzed samples and in mg of Trolox per portion equal to one espresso. The contributions of phenols and melanoidins to the antioxidant activity of brews or extracts were also calculated.
2.7. Determination of Antioxidant Activity in the Process of Linoleic Acid Autoxidation
According to the method of Kuo et al. [28]: 9.25 mL of linoleic acid emulsion was mixed with 0.25 mL of extracts diluted four times or melanoidin solutions (1 mg/mL), and 380 µL of that mixture was heated in a water bath (37 °C) for 1 min. Oxidation was initiated by the addition of 20 µL of hemoglobin solution (0.35 mg/mL) and was terminated by the addition of 5 mL of hydrochloric acid ethanolic solution (6 mg/mL) after 5 min of reaction. In order to measure the amount of peroxides, 100 µL of ammonium thiocyanate (0.3 g/mL) and iron(II) chloride (2.54 mg/mL) solutions were added. The absorbance of the sample was measured after 5 min at 480 nm. The control sample, blind sample, Trolox and chlorogenic acid standard solutions (in 70% acetone solution) were also analyzed. Results were calculated in the same manner as described for the CBA method.
2.8. Statistical Analysis
The preparation of numerical results was performed using MS Excel 2019 and Statistica ver. 13.3. In order to evaluate the influence of tested factors on the acquired data, analysis of variance (one-way ANOVA) was performed. Statistical assumptions concerning the normal distribution of values and equality of variances were tested using Shapiro–Wilk’s and Levene’s or Bartlet’s tests, respectively. To compare mean values of every group of results, Tukey’s HSD test was used—results of this test are presented in tables by letter designations. The correlation between phenol content and antioxidant activity was described by Pearson’s (rp) or Spearman’s (rs) rank correlation coefficients, depending on the assumption that a normal distribution of numerical values is met. The significance level for all statistical analysis was p = 0.05.
2.9. Chemicals and Reagents
Folin-Ciocalteu reagent, sodium carbonate, hydrochloric acid, sodium dihydrogen phosphate dihydrate, disodium hydrogen phosphate dodecahydrate, sodium chloride, iron(II) chloride tetrahydrate, AAPH and Tween 20 were purchased from SigmaAldrich (Poznań, Poland). Chlorogenic acid, gallic acid (GA), neocuproine, ABTS, linoleic acid, hemoglobin and Trolox were purchased from Avantor Performance Materials Poland S.A. (Gliwice, Poland). Acetone, copper(II) chloride and ammonium acetate were purchased from Chempur (Piekary Śląskie, Poland). Potassium persulfate was purchased from Macherey-Nagel GmbH and Co. KG (Dueren, Germany). Crocin was purchased from VWR International (Gdańsk, Poland). Ammonium thiocyanate was purchased from AppliChem GmbH (Darmstadt, Germany). Ethanol was purchased from Alchem Group (Toruń, Poland).
Phosphate-buffered saline (PBS) was prepared by dissolving 0.148 g of sodium dihydrogen phosphate dihydrate, 1.45 g of disodium hydrogen phosphate dodecahydrate and 4.0325 g of sodium chloride in 500 mL of distilled water. The active solution for ABTS analysis (96 mg ABTS and 16.5 mg potassium persulfate for 25 mL solution) was prepared 24 h before analysis and kept in the dark. After this time, the solution was diluted with PBS to obtain the absorbance of 0.700 (±0.020) at 734 nm. The linoleic acid emulsion was prepared by homogenization of 100 mg Tween 20, 224.2 mg linoleic acid and 200 mL of PBS. The rest of the reagents were prepared by dissolution in distilled water, if not mentioned otherwise.
3. Results and Discussion
3.1. Characterization of Coffee Beans
Coffee beans were subjected to different treatments in order to acquire various roasting degrees, which was judged by the color of whole beans.. The results for coffee beans used in this study are presented in Table 1. According to the SCAA standard, coffee samples were prepared correctly, as their roasting degrees were medium and dark. The beans were then ground using different settings to obtain coffee suitable to produce espresso and moka brew; a higher grinding level indicates bigger particles of ground coffee. Since coffee for espresso needs to be ground to a smaller size than for moka brews [29], the grinder’s settings were different (Table 2). In the case of the dark roasted coffee for espresso, a higher grinding level (2.3 instead of 1.3) was used. Dark roasted coffee is more brittle and porous, so this operation was performed in order to reduce the amount of nanoparticles in the final brew [30]. The color of the ground coffee was measured in the laboratory, and the results are presented in Table 1. L* (lightness) values in all samples were in the range 30–35, and a* (red-green) values were in the range 9–14, while in case of the b* (yellow-blue) parameter, more visible changes were observed—in medium roast, positive values were acquired, while in dark roasted coffee, negative values were acquired. Overall, a significant decrease in all values with the darker roast of coffee was observed, but only in b* parameter was a shift in the color component indicated. Similar tendencies in color changes were reported by researchers [13,20,31].

Table 1.
Characteristics of coffee beans.

Table 2.
Characteristics of coffee brews.
3.2. Characterization of Coffee Brews and Extracts
The coffee samples were used to prepare espresso and moka brews. The manufacturer’s information regarding similarity between moka pot brew and a specific number of espressos was confirmed based on approximate volumes of the analyzed brews; however, significant differences in their properties have emerged (Table 2). The values of the L* parameter for the espresso were lower than 20 each time, and the b* parameter was less than 35, while for the moka brews, these values were above 40 and above 65, respectively. Additionally, the darker roast degree significantly reduced the determinants mentioned above. More pronounced changes were observed in espresso brews, where a decrease greater than 70% occurred. Changes in the values of the a* parameter were not consistent and did not fulfill statistical assumptions.
The pH values of all brews were lower than 6, and espresso exhibited a more acidic character in comparison with moka brews (Table 2). The higher roast resulted in a pH increase and a further reduction of the acidic character. These changes are similar to transformations described for color parameters and conformances found in the literature [16,19,32]; however, while comparing brews prepared with the French Press and chemex methods, the first one exhibited a lower pH value. It was suggested that longer brewing time resulted in more acidic compound extraction [11]. When filtered coffee and espresso were compared, the latter one had a lower pH value [17]. It might suggest that even if pressure is applied during moka preparation, brews prepared that way are similar to those prepared in conventional ways. More soluble matter was extracted during espresso preparation; the roast degree had various impacts depending on the type of brew (Table 2). It is well known that smaller particle size can enhance extraction, which could be used to explain higher values of total soluble solids in the espresso [11]. The greater pressure in the case of espresso brews also could have an effect on soluble solid extraction [21]. Moreover, the amount of coffee used for the preparation of moka brews (10 g and 12 g for medium and dark roast, respectively) could alter the effect of roast degree observed here. Even if espresso showed a greater amount of soluble solids, its melanoidin content was lower (Table 2). Longer extraction time and greater coffee:water ratio might explain these differences. The darker roast degree resulted in higher melanoidin content, which is in accordance with the theory behind their formation during Maillard’s reactions [12] and previously reported results [30,33]. Table 3 contains results from the phenolic compound measurement using Folin’s method for bean extracts and different brews. Both preparation methods extracted the most examined compounds from coffee beans; considering the amount of phenolic compounds calculated per DM, only 64–74% of total phenols were extracted from espresso, while for moka brews it was more than 85% (Table 3). However in portions equivalent to espresso shots, the opposite trend is noticeable (Table 3). Identical observations were reported in the literature [16]. A greater coffee:water ratio and longer time of preparation might be advantageous for the purpose of better extraction. However, moka brews are much more diluted than espresso shots; hence, while comparing identical portions, the latter has a greater amount of phenolic compounds. Ludwig et al. [17] reported that during the first 8 s of the espresso preparation process, phenol elution is the most efficient, but for filtered coffee, constant extraction was observed. It might suggest that applied pressure has a stronger effect on the extraction results. The previously conducted studies show opposite tendencies—shorter contact time and higher pressure enhanced phenolic compound extraction; however, when results referred to brew volume, the same tendencies as observed in this research appeared [18,34]. A general decreasing tendency with the darker roast can be seen, although its significance is proven mostly for results presented per coffee portion. This trend is in agreement with results obtained by other researchers [31,32,33,34,35,36,37].

Table 3.
Content of phenolic compounds and antioxidant activity measured by SET-based methods.
3.3. Antioxidant Activity of Coffee Extracts
The antioxidant properties of coffee were investigated using four methods that differ in the reaction mechanism. All methods used in this study reflect the ability to break the chain of oxidation reactions by scavenging or inhibiting the creation of free radicals but through different mechanisms. Antioxidant compounds react with active radicals to stabilize them by single electron transfer (SET) or hydrogen atom transfer (HAT) mechanisms. In both cases, the antioxidant compounds react more rapidly with the oxidative agent, thus, protecting the potentially exposed molecules. The antioxidant activity based entirely on the SET mechanism is tested with the use of the complex of neocuproine with copper(II) ions. As a result of the tested compound’s reducing power, the electron is transferred to the complex and the copper ion is being reduced. In the case of the method based entirely on the HAT mechanism, crocin was used in our study as a molecular probe. Crocin is a carotenoid that undergoes decomposition (and discoloration) only in the presence of free radicals, losing hydrogen atoms upon the reaction. Most oxidation reactions, however, follow a mixed mechanism, and to investigate them, different methods are used, i.e., the ABTS method (advantage of SET mechanism) and the most complex method based on autoxidation of linoleic acid (advantage of HAT mechanism) [38].
The results per 100 g DM show that espresso was much less active than moka brews, and both brews were less active than bean extracts (Table 3). In the CUPRAC method, the activity of espresso in relation to the total activity of the beans was 71.6% and 57.8%, while the activity of moka brews was 81.7% and 82.4% for medium and dark roasted beans, respectively. In the ABTS method, the statistical analysis showed no significant effect of the roasting degree on the antioxidant activity, as well as no significant differences between the activity of moka brews and bean extracts, which indicates a high extraction of compounds active against ABTS•+ in this preparation method. Taking into account the values per portion corresponding to an espresso shot, an inverse relationship is visible—espresso exhibited higher antioxidant activity in the method based entirely on the SET mechanism (Table 3). The values for the ABTS method did not meet statistical assumptions, and non-parametric analysis did not indicate differences between analyzed samples. In a method based entirely on the HAT mechanism (CBA method), espresso brews showed a capacity higher than the moka brews to scavenge free radicals and protect crocin from degradation (Table 4). It is worth noting that the medium-roasted espresso brew showed even higher activity than bean extract, which may be due to better extraction of active compounds during its preparation. As mentioned before, the method using the autoxidation of linoleic acid is based on a combined mechanism with a predominance of HAT. Due to the specific conditions of this technique, i.e., the use of compounds found in the human body, the reaction temperature and pH value of the environment, it is said that this method best reflects the conditions of the oxidation process in the human body. These method’s results are similar to those obtained with the CBA method. When the results of those two methods were converted based on standard solutions and presented in mg of Trolox per portion, the influence of the preparation method was ambiguous—in the CBA method, moka brews were significantly different from espresso brews, while in the method using the autoxidation reaction, both espresso samples were similar to the respective moka brews (Table 4). As indicated by statistical analysis, the effect of the degree of roast in those methods was unclear as well.

Table 4.
Content of phenolic compounds and antioxidant activity measured by HAT-based methods.
Infusions commonly referred to as “coffee” are prepared in many ways today. The choice of method is mainly influenced by the taste preferences of consumers. Two methods were used in the research, in which increased pressure is used—espresso and the method with the use of a moka pot. The dependence of the antioxidant properties on the method of preparing the brew is not clear. In the case of SET-based methods, the changes in the properties tested can be easily explained by the different characteristics of the preparation methods, mainly different pressure and coffee:water ratio, resulting in a different volume of final infusions. The research of Ludwig et al. [17] confirms the supposition about the importance of the preparation method—comparing filtered coffee and espresso, the latter had a higher activity per mL of infusion each time. In the research by Janda et al. [39], espresso brews showed lower activity than drip coffee, and brews prepared with aeropress were the most active. In that research, however, espresso brews were prepared with a different coffee:water ratio than is normally established [29]. Even so, it was proven that higher pressure used during the preparation of the brew might improve the extractability of active compounds from coffee beans. The results, expressed in mg of Trolox per portion, also indicate that the common information presented to consumers about moka brews being comparable to a specific number of espresso shots is misleading. When using the methods based on the HAT mechanism, espresso once more showed higher antioxidant activity; however, no significant effect of brew preparation was seen after conversion of the results using standard solutions. It might indicate that in those methods, a nonlinear dependency of activity occurs. Furthermore, the phenolic profile obtained by different roasting and brewing methods may not behave in a similar way in antioxidant experiments based on different mechanisms. Regarding the roasting degree effect on the antioxidant activity of coffee brews, the information in the literature is not explicit. Vignoli et al. [33], examining the activity of aqueous coffee extracts, showed both an increase and a decrease in the values with the darker degree of roasting in various analytical methods. Similar observations were reported in later publications [11,31,33,34,35,40]. Initial changes regarding antioxidant activity might be connected to the release of low-molecular-weight phenolic compounds in the early stage of roasting, while the decrease observed later might be associated with their degradation and/or structural changes [5,30,37]. Liao et al. [36] also observed a decrease in the antioxidant activity of coffee with a darker roast. They also observed the formation of some phenolic compounds during roasting, e.g., gallic acid, pyrogallol or purpurogallin, and suggested that it might compensate for the decrease in antioxidant activity of coffee from the loss of chlorogenic acid. Studies using hot and cold brews [19] showed that the degree of roasting had a significant negative effect only in the case of cold brew. In hot coffee brews, an increase in antioxidant activity with a higher degree of roast was reported using 2,2,6,6-tetramethyl-1-piperidin-1-oxyl stabilized radical with electron paramagnetic resonance spectroscopy. The issue of factors influencing the antioxidant activity of coffee is, therefore, difficult to unequivocally solve. Not only the issues related to the coffee bean and its treatment but also the choice of the analytical method can have a significant influence on the final result.
Using statistical analysis, the correlation of the results of antioxidant activity with the content of phenolic compounds in coffee was checked (the content of melanoidins was omitted due to the small amount of data). In the case of the results expressed as mg Trolox per 100 g DM, a strong positive correlation was found between the content of phenolic compounds and the results obtained with the CUPRAC and ABTS methods (rp = 0.8282 and rp = 0.8975, respectively; p ≤ 0.001). Furthermore, the results obtained with the CUPRAC and ABTS methods, as well as the CBA method and the method based on the autoxidation of linoleic acid, were strongly correlated with each other (rp = 0.8943 and rs = 0.9510, respectively; p ≤ 0.001), which may result from the identical mechanism of the reactions used. The weak correlation between the content of phenolic compounds and the results of the CBA method (rs = 0.6154; p = 0.033) and the lack of this correlation with results of the method using the autoxidation reaction may indicate that in these methods, other compounds with antioxidant activity, e.g., melanoidins, were of greater importance. In the case of the results per portion of the brew, a strong correlation was demonstrated between the content of phenolic compounds and the results of the CUPRAC, ABTS and CBA methods (rp = 0.9805, rs = 0.8095 and rp = 0.8070, respectively; p ≤ 0.015). Moreover, the results obtained by the CUPRAC method were correlated with the results of the ABTS (rs = 0.8810; p < 0.004) and CBA methods (rp = 0.8538; p = 0.007). There were no correlations with the results obtained with the method of linoleic acid autoxidation in this case, however. This may be related to the high degree of complexity of the reaction. The differences between the correlation coefficients obtained for results expressed in different units might be explained by various proportions of the individual results after recalculations, being, among others, an effect of the nonlinear dependence of the HAT-based methods’ results, as mentioned earlier.
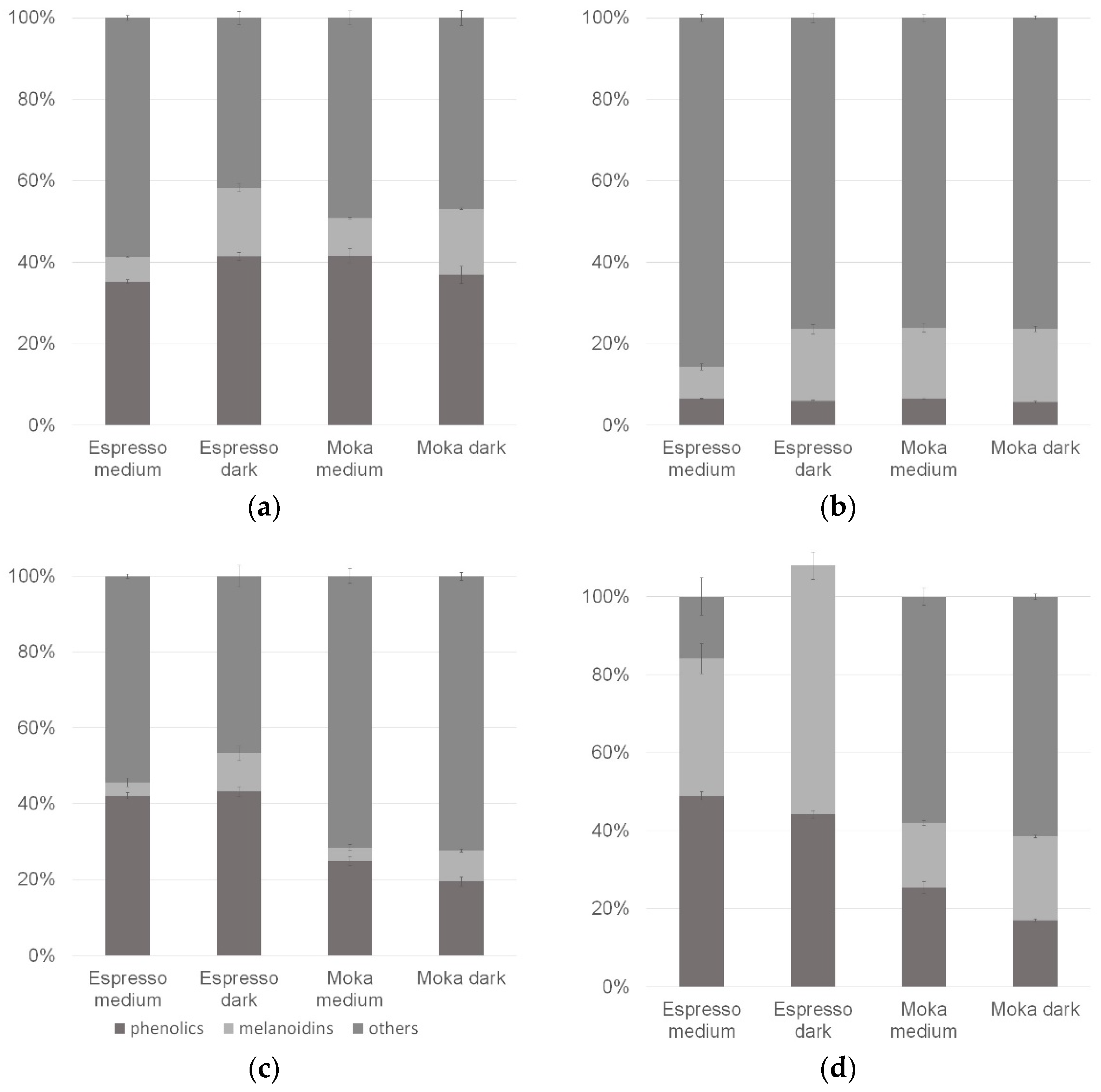
In order to better understand the importance of analyzed compounds for the antioxidant activity of the whole brew, the obtained results were presented as a percentage of the contribution of phenolic compounds and melanoidins for the antioxidant activity of coffee infusions (Figure 1). The amount of phenolic compounds was acquired by calculating Folin-Ciocalteu results with chlorogenic acid results, analyzed in every method. The values for each test sample add up to 100% so that the portion size is ignored; only quantitative proportions are shown. In the case of the method based only on the SET mechanism, the participation of phenolic compound activity is similar in all brews, and the melanoidin share was greater in infusions prepared from darker roasted beans (Figure 2a). In the ABTS method, melanoidins were of greater importance compared to phenolic compounds; however, their total participation in brew activity was less than 25% (Figure 2b). In methods based entirely or partially on the HAT mechanism (Figure 2c,d), the effect of the preparation method was evident—espresso each time showed a greater total share of the tested compounds in the entire brew activity. In the case of the autoxidation of linoleic acid, the total activity of phenolics and melanoidins tested separately was even greater than the overall activity of the coffee beans (lack of additivity in this experiment). In Figure 2c,d, the darker roast degree increased the share of melanoidin activity and slightly decreased the share of phenolic compound activity.
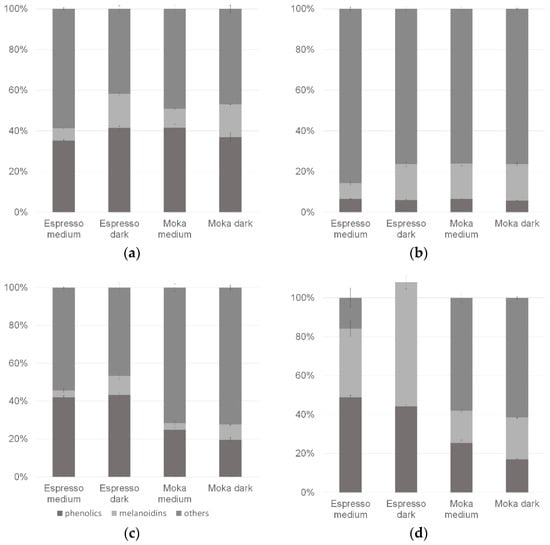
Figure 2.
Activity of common classes of coffee compounds in the CUPRAC method (a), the ABTS method (b), the CBA method (c) and the method based on autoxidation of linoleic acid (d).
As is well known, during the roasting process, numerous changes occur in the chemical composition of coffee beans, mainly, the degradation of thermolabile substances and the formation of new compounds, e.g., as a result of the Maillard reaction. Observed differences regarding the different proportions of the activity of phenolic compounds may result from their partial degradation in the roasting process or their different extractives during the preparation of infusions. However, the differences in the proportion of melanoidins are more pronounced and suggest that their activity depends significantly on the degree of roast. It is worth noting that the activity of melanoidins is also influenced by the phenolic compounds attached to their chain [12,31,37]. The obtained results clearly show that Maillard reaction products can at least compensate for the loss of phenolic compounds with their antioxidant activity. Compounds defined as “others” also deserve attention. Across all studied methods, their share in the total activity of brews ranged from 16–86%. This suggests that other compounds with antioxidant activity are present in the coffee bean; these can be naturally contained compounds or low-molecular-weight Maillard reaction products.
4. Conclusions
This research aimed at checking the influence of the roasting degree and method of preparation on the antioxidant activity of coffee brews and their main antioxidant classes, with special attention to the share of the analyzed groups of antioxidants in overall activity. Four methods of antioxidant activity determination differing in the reaction mechanism (pure reduction ability, pure hydrogen donation, mixed-mechanism radical deactivation, mixed-mechanism autoxidation inhibition) were utilized. In terms of general characteristics and antioxidant activity, espresso and moka brews turned out to be significantly different. It was shown that the activities of infusions were influenced not only by factors related to the treatment of coffee beans but also by the applied analytical method. In general, espresso was characterized by greater activity in terms of the consumed portion. Increasing the degree of roasting led to a decrease in the content of phenolic compounds and an increase in the amount of melanoidins in brews (as well as the share of their activity), a phenomenon which was also observed by other authors and well explained.
Based on the obtained results of antioxidant activity in various reactions, it was concluded that phenolic compounds and melanoidins act through a mixed antioxidant mechanism, but in the case of the latter, the HAT mechanism may have an advantage. Taking into account only results from SET-based reactions would lead to underestimation of the role of melanoidins in the antioxidant activity of coffee brews and misunderstanding of their potential. We found that a large proportion of the total activity cannot be attributed to the two most well-known and mentioned groups of antioxidants. It is, therefore, necessary to look for other antioxidants relevant to the total balance of coffee activity.
Author Contributions
Conceptualization, R.W. and P.P.; methodology, R.W.; software, P.P.; validation, R.W. and P.P.; formal analysis, P.P.; investigation, P.P.; resources, R.W. and M.J.; data curation, P.P.; writing—original draft preparation, P.P.; writing—review and editing, P.P., R.W., B.D. and M.J.; visualization, P.P.; supervision, R.W. and B.D.; project administration, R.W.; funding acquisition, R.W. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
We would like to show our gratitude to Alek Smęk, Hear Coffee Roaster at LaCava roastery, for help with acquiring the sample material and for sharing his knowledge and experience in coffee processing and brew preparation methods.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Farah, A.; dos Santos, T.F. The Coffee Plant and Beans: An Introduction. In Coffee in Health and Disease Prevention, 1st ed.; Preedy, V.R., Ed.; Academic Press: Cambridge, UK, 2015; pp. 5–10. ISBN 978-0-12-409517-5. [Google Scholar]
- Hatzold, T. Introduction. In Coffee: Emerging Health Effects and Disease Prevention; Chu, Y.F., Ed.; John Wiley & Sons Incorporated: Hoboken, NJ, USA, 2012; pp. 1–20. ISBN 978-0-470-95878-0. [Google Scholar]
- Samoggia, A.; Riedel, B. Consumers’ perceptions of coffee health benefits and motives for coffee consumption and purchasing. Nutrients 2019, 11, 653. [Google Scholar] [CrossRef] [PubMed]
- Trandafir, I.; Nour, V.; Ionica, M.E. Antioxidant capacity, phenolic acids and caffeine contents of some commercial coffees available on the Romanian market. Arch Lat. Nutr. 2013, 63, 87–94. [Google Scholar]
- Chung, H.S.; Kim, D.H.; Youn, K.S.; Lee, J.B.; Moon, K.D. Optimization of roasting conditions according to antioxidant activity and sensory quality of coffee brews. Food Sci. Biotechnol. 2013, 22, 23–29. [Google Scholar] [CrossRef]
- Olechno, E.; Puścion-Jakubik, A.; Markiewicz-Żukowska, R.; Socha, K. Impact of Brewing Methods on Total Phenolic Content (TPC) in Various Types of Coffee. Molecules 2020, 25, 5274. [Google Scholar] [CrossRef] [PubMed]
- Poisson, L.; Blank, I.; Andreas, D.A.; Hofmann, T. The chemistry of roasting—Decoding flavor formation. In The Craft and Science of Coffee, 1st ed.; Folmer, B., Ed.; Academic Press: Cambridge, UK, 2017; pp. 273–310. ISBN 978-0-128-03558-0. [Google Scholar]
- Heide, J.; Czech, H.; Ehlert, S.; Koziorowski, T.; Zimmermann, R. Toward Smart Online Coffee Roasting Process Control: Feasibility of Real-Time Prediction of Coffee Roast Degree and Brew Antioxidant Capacity by Single-Photon Ionization Mass Spectrometric Monitoring of Roast Gases. J. Agric. Food Chem. 2020, 68, 4752–4759. [Google Scholar] [CrossRef] [PubMed]
- Corrêa, C.L.O.; Penha, E.M.; dos Anjos, M.R.; Pacheco, S.; Freitas-Silva, O.; Luna, A.S.; Gottschalk, L.M.F. Use of asparaginase for acrylamide mitigation in coffee and its influence on the content of caffeine, chlorogenic acid, and caffeic acid. Food Chem. 2021, 338, 128045. [Google Scholar] [CrossRef] [PubMed]
- Muzykiewicz-Szymańska, A.; Nowak, A.; Wira, D.; Klimowicz, A. The Effect of Brewing Process Parameters on Antioxidant Activity and Caffeine Content in Infusions of Roasted and Unroasted Arabica Coffee Beans Originated from Different Countries. Molecules 2021, 26, 3681. [Google Scholar] [CrossRef]
- Bilge, G. Investigating the effects of geographical origin, roasting degree, particle size and brewing method on the physicochemical and spectral properties of Arabica coffee by PCA analysis. J. Food Sci. Technol. 2020, 57, 3345–3354. [Google Scholar] [CrossRef] [PubMed]
- Moreira, A.; Nunes, F.M.; Domingues, M.R.; Coimbra, M. Coffee melanoidins: Structures, mechanisms of formation and potential health impacts. Food Funct. 2012, 3, 903–915. [Google Scholar] [CrossRef]
- Ripper, B.; Kaiser, C.R.; Perrone, D. Use of NMR techniques to investigate the changes on the chemical composition of coffee melanoidins. J. Food Compos. Anal. 2020, 87, 103399. [Google Scholar] [CrossRef]
- Cordoba, C.N.; Pataquiva, L.; Osorio, C.; Moreno, F.; Ruiz, R. Effect of grinding, extraction time and type of coffee on the physicochemical and flavor characteristics of cold brew coffee. Sci. Rep. 2019, 9, 8440. [Google Scholar] [CrossRef]
- Derossi, A.; Ricci, I.; Caporizzi, R.; Fiore, A.; Severini, C. How grinding level and brewing method (Espresso, American, Turkish) could affect the antioxidant activity and bioactive compounds in a coffee cup: Effect of grinding level and brewing methods on healthy compounds of coffee brew. J. Sci. Food Agric. 2017, 98, 3198–3207. [Google Scholar] [CrossRef]
- Gloess, A.N.; Schonbachler, B.; Klopprogge, B.; D’Ambrosio, L.; Chatelain, K.; Bongartz, A.; Strittmatter, A.; Rast, M.; Yeretzian, C. Comparison of nine common coffee extraction methods: Instrumental and sensory analysis. Eur. Food Res. Technol. 2013, 236, 607–627. [Google Scholar] [CrossRef]
- Ludwig, I.A.; Sancher, L.; Caemmerer, B.; Kroh, L.W.; De Pena, M.P.; Cid, C. Extraction of coffee antioxidants: Impact of brewing time and method. Food Res. Int. 2012, 48, 57–64. [Google Scholar] [CrossRef]
- Perez-Martiner, M.; Caemmerer, B.; De Pena, M.P.; Cid, C.; Kroh, L.W. Influence of brewing method and acidity regulators on the antioxidant capacity of coffee brews. J. Agric. Food Chem. 2010, 58, 2958–2965. [Google Scholar] [CrossRef] [PubMed]
- Rao, N.Z.; Fuller, M.; Grim, M.D. Physiochemical Characteristics of Hot and Cold Brew Coffee Chemistry: The Effects of Roast Level and Brewing Temperature on Compound Extraction. Foods 2020, 9, 902. [Google Scholar] [CrossRef] [PubMed]
- Seninde, D.R.; Chambers, E.I.; Chambers, D. Determining the impact of roasting degree, coffee to water ratio and brewing method on the sensory characteristics of cold brew Ugandan coffee. Food Res. Int. 2020, 137, 109667. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, X.; Manickavasagan, A.; Lim, L.T. Extraction and physicochemical characteristics of high pressure-assisted cold brew coffee. Future Foods 2022, 5, 100113. [Google Scholar] [CrossRef]
- Bekedam, E.K.; Schols, H.A.; van Boekel, M.A.J.S.; Smit, G. High Molecular Weight Melanoidins from Coffee Brew. J. Agric. Food Chem. 2006, 54, 7658–7666. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Özyürek, M.; Güçlü, K.; Apak, R. The main and modified CUPRAC methods of antioxidant measurement. TrAC Trends Anal. Chem. 2011, 30, 652–664. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free. Radic Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Bountagkidou, O.; Ordoudi, S.; Tsimidou, M. Structure–antioxidant activity relationship study of natural hydroxybenzaldehydes using in vitro assays. Food Res. Int. 2010, 43, 2014–2019. [Google Scholar] [CrossRef]
- Di Majo, D.; Neve, L.; Guardia, M.; Casuccio, A.; Giammanco, M. The influence of two different pH levels on the antioxidant properties of flavonols, flavan-3-ols, phenolic acids and aldehyde compounds analysed in synthetic wine and in a phosphate buffer. J. Food Compos. Anal. 2011, 24, 265–269. [Google Scholar] [CrossRef]
- Kuo, J.M.; Yeh, D.B.; Pan, B.S. Rapid photometric assay evaluating antioxidative activity in edible plant material. J. Agric. Food Chem. 1999, 47, 3206–3209. [Google Scholar] [CrossRef]
- Mestdagh, F.; Glabasnia, A.; Giuliano, P. The brew—Extracting for excellence. In The Craft and Science of Coffee, 1st ed.; Folmer, B., Ed.; Academic Press: Cambridge, UK, 2017; pp. 355–380. ISBN 978-0-128-03558-0. [Google Scholar]
- Herawati, D.; Giriwono, P.E.; Dewi, F.N.A.; Kashiwagi, T.; Andarwulan, N. Critical roasting level determines bioactive content and antioxidant activity of Robusta coffee beans. Food Sci. Biotechnol. 2019, 28, 7–14. [Google Scholar] [CrossRef]
- Song, J.L.; Asare, T.S.; Kang, M.Y.; Lee, S.C. Changes in bioactive compounds and antioxidant capacity of coffee under different roasting conditions. Korean J. Plant Res. 2018, 31, 704–713. [Google Scholar] [CrossRef]
- Varady, M.; Hruskova, T.; Popelka, P. Effect of preparation method and roasting temperature on total polyphenol content in coffee beverages on total polyphenol content in coffee beverages. Czech J. Food Sci. 2020, 38, 417–421. [Google Scholar] [CrossRef]
- Vignoli, J.A.; Viegas, M.C.; Bassoli, D.G.; Benassi, M.T. Roasting process affects differently the bioactive compounds and the antioxidant activity of arabica and robusta coffees. Food Res. Int. 2014, 61, 279–285. [Google Scholar] [CrossRef]
- Górecki, M.; Hallmann, E. The antioxidant content of coffee and its in vitro activity as an effect of its production method and roasting and brewing time. Antioxidants 2020, 9, 308. [Google Scholar] [CrossRef]
- Cammerer, B.; Kroh, L.W. Antioxidant activity of coffee brews. Eur. Food Res. Technol. 2006, 223, 469–474. [Google Scholar] [CrossRef]
- Liao, Y.C.; Kim, T.; Silva, J.L.; Hu, W.Y.; Chen, B.Y. Effects of roasting degrees on phenolic compounds and antioxidant activity in coffee beans from different geographic origins. LWT Food Sci. Technol. 2022, 168, 113965. [Google Scholar] [CrossRef]
- Schouten, M.A.; Tappi, S.; Angeloni, S.; Cortese, M.; Capriolo, G.; Vittori, S.; Romani, S. Acrylamide formation and antioxidant activity in coffee during roasting—A systematic study. Food Chem. 2021, 334, 128514. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Yang, C.; Tsao, R. Nomenclature and general classification of antioxidant activity/capacity assays. In Measurement of Antioxidant Activity & Capacity, 1st ed.; Apak, R., Capanoglu, E., Shahidi, F., Eds.; Wiley-Blackwell: Hoboken, NJ, USA, 2018; pp. 1–20. ISBN 978-1-119-13538-8. [Google Scholar]
- Janda, K.; Jakubczyk, K.; Baranowska-Bosiacka, I.; Kapczuk, P.; Kochman, J.; Rębacz-Maron, E.; Gutowska, I. Mineral Composition and Antioxidant Potential of Coffee Beverages Depending on the Brewing Method. Foods 2020, 9, 121. [Google Scholar] [CrossRef]
- Bobkova, A.; Hudacek, M.; Jakabova, S.; Belej, L.; Capcarova, M.; Curlej, J.; Bobko, M.; Arvay, J.; Jakab, I.; Capla, J.; et al. The effect of roasting on the total polyphenols and antioxidant activity of coffee. J. Environ. Sci. Health Part B 2020, 25, 2574–2588. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).