Preliminary Investigation of Geopolymer Foams as Coating Materials
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Method of Testing
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Amran, Y.M.; Alyousef, R.; Alabduljabbar, H.; El-Zeadani, M. Clean production and properties of geopolymer concrete: A review. J. Clean. Prod. 2020, 251, 119679. [Google Scholar] [CrossRef]
- Cong, P.; Cheng, Y. Advances in geopolymer materials: A comprehensive review. J. Traffic Transp. Eng. 2021, 8, 283–314. [Google Scholar] [CrossRef]
- Ma, C.K.; Awang, A.Z.; Omar, W. Structural and material performance of geopolymer concrete: A review. Constr. Build. Mater. 2018, 186, 90–102. [Google Scholar] [CrossRef]
- Gao, B.; Jang, S.; Son, H.; Lee, H.J.; Lee, H.J.; Yang, J.J.; Bae, C.J. Study on mechanical properties of kaolin-based geopolymer with various Si/Al ratio and aging time. J. Korean Ceram. Soc. 2020, 57, 709–715. [Google Scholar] [CrossRef]
- Ayeni, O.; Onwualu, A.P.; Boakye, E. Characterization and mechanical performance of metakaolin-based geopolymer for sustainable building applications. Constr. Build. Mater. 2021, 272, 121938. [Google Scholar] [CrossRef]
- Kaur, K.; Singh, J.; Kaur, M. Compressive strength of rice husk ash based geopolymer: The effect of alkaline activator. Constr. Build. Mater. 2018, 169, 188–192. [Google Scholar] [CrossRef]
- Firdous, R.; Stephan, D.; Djobo, J.N.Y. Natural pozzolan based geopolymers: A review on mechanical, microstructural and durability characteristics. Constr. Build. Mater. 2018, 190, 1251–1263. [Google Scholar] [CrossRef]
- Fan, F.; Liu, Z.; Xu, G.; Peng, H.; Cai, C.S. Mechanical and thermal properties of fly ash based geopolymers. Constr. Build. Mater. 2018, 160, 66–81. [Google Scholar] [CrossRef]
- Zhang, Y.; Xiao, R.; Jiang, X.; Li, W.; Zhu, X.; Huang, B. Effect of particle size and curing temperature on mechanical and microstructural properties of waste glass-slag-based and waste glass-fly ash-based geopolymers. J. Clean. Prod. 2020, 273, 122970. [Google Scholar] [CrossRef]
- Aziz, A.; Stocker, O.; El Hassani, I.E.E.A.; Laborier, A.P.; Jacotot, E.; El Khadiri, A.; El Bouari, A. Effect of blast-furnace slag on physicochemical properties of pozzolan-based geopolymers. Mater. Chem. Phys. 2021, 258, 123880. [Google Scholar] [CrossRef]
- Aziz, I.H.; Abdullah, M.M.A.B.; Salleh, M.M.; Azimi, E.A.; Chaiprapa, J.; Sandu, A.V. Strength development of solely ground granulated blast furnace slag geopolymers. Constr. Build. Mater. 2020, 250, 118720. [Google Scholar] [CrossRef]
- Jindal, B.B.; Sharma, R. The effect of nanomaterials on properties of geopolymers derived from industrial by-products: A state-of-the-art review. Constr. Build. Mater. 2020, 252, 119028. [Google Scholar] [CrossRef]
- Sindhunata; Van Deventer, J.S.J.; Lukey, G.C.; Xu, H. Effect of curing temperature and silicate concentration on fly-ash-based geopolymerization. Ind. Eng. Chem. Res. 2006, 45, 3559–3568. [Google Scholar] [CrossRef]
- Shin, S.; Goh, G.; Lee, C. Predictions of compressive strength of GPC blended with GGBFS developed at varying temperatures. Constr. Build. Mater. 2019, 206, 1–9. [Google Scholar] [CrossRef]
- Yaseri, S.; Hajiaghaei, G.; Mohammadi, F.; Mahdikhani, M.; Farokhzad, R. The role of synthesis parameters on the workability, setting and strength properties of binary binder-based geopolymer paste. Constr. Build. Mater. 2017, 157, 534–545. [Google Scholar] [CrossRef]
- Lahoti, M.; Wong, K.K.; Yang, E.H.; Tan, K.H. Effects of Si/Al molar ratio on strength endurance and volume stability of metakaolin geopolymers subject to elevated temperature. Ceram. Int. 2018, 44, 5726–5734. [Google Scholar] [CrossRef]
- Nath, P.; Sarker, P.K. Flexural strength and elastic modulus of ambient-cured blended low-calcium fly ash geopolymer concrete. Constr. Build. Mater. 2017, 130, 22–31. [Google Scholar] [CrossRef]
- Kope, R.A.; Prałat, K.; Ciemnicka, J.; Buczkowska, K. Influence of the calcination temperature of synthetic gypsum on the particle size distribution and setting time of modified building materials. Energies 2020, 13, 5759. [Google Scholar] [CrossRef]
- Figiela, B.; Šimonová, H.; Korniejenko, K. State of the art, challenges, and emerging trends: Geopolymer composite reinforced by dispersed steel fibers. Rev. Adv. Mater. Sci. 2022, 61, 1–15. [Google Scholar] [CrossRef]
- Korniejenko, K.; Figiela, B.; Ziejewska, C.; Marczyk, J.; Bazan, P.; Hebda, M.; Choińska, M.; Lin, W.-T. Fracture behavior of long fiber reinforced geopolymer composites at different operating temperatures. Materials 2022, 15, 482. [Google Scholar] [CrossRef]
- Görhan, G.; Kürklü, G. The influence of the NaOH solution on the properties of the fly ash-based geopolymer mortar cured at different temperatures. Compos. Part B Eng. 2014, 58, 371–377. [Google Scholar] [CrossRef]
- Nguyen, L.; Moseson, A.J.; Farnam, Y.; Spatari, S. Effects of composition and transportation logistics on environmental, energy and cost metrics for the production of alternative cementitious binders. J. Clean. Prod. 2018, 185, 628–645. [Google Scholar] [CrossRef]
- Temuujin, J.; Minjigmaa, A.; Rickard, W.; Lee, M.; Williams, I.; Van Riessen, A. Fly Ash Based Geopolymer Thin Coatings on Metal Substrates and Its Thermal Evaluation. J. Hazard. Mater. 2010, 180, 748–752. [Google Scholar] [CrossRef] [PubMed]
- Yong, S.L.; Feng, D.W.; Lukey, G.C.; Van Deventer, J.S.J. Chemical Characterisation of The Steel Geopolymeric Gel Interface. Colloids Surf. A Physicochem. Eng. Asp. 2007, 302, 411–423. [Google Scholar] [CrossRef]
- Balaguru, P.N.; Nazier, M.; Arafa, M. Field Implementation of Geopolymer Coatings; New Jersey Department of Transportation: Ewing Township, NJ, USA, 2008. Available online: https://cait.rutgers.edu/wp-content/uploads/2018/05/fhwa-nj-2002-011.pdf (accessed on 12 August 2022).
- Zhang, Z.; Yao, X.; Zhu, H. Potential Application of Geopolymers as Protection Coatings for Marine Concrete I. Basic Properties. Appl. Clay Sci. 2010, 49, 67–68. [Google Scholar]
- Zhang, Z.; Yao, X.; Zhu, H. Potential application of geopolymers as protection coatings for marine concrete: II Microstructure and anticorrosion mechanism. Appl. Clay Sci. 2010, 49, 7–12. [Google Scholar] [CrossRef]
- Tian, Q.; Wang, S.; Sui, Y.; Lv, Z. Alkali-activated materials as coatings deposited on various substrates: A review. Int. J. Adhes. Adhes. 2021, 110, 102934. [Google Scholar] [CrossRef]
- Temuujin, J.; Minjigmaa, A.; Rickard, W.; Lee, M.; Williams, I.; Van Riessen, A. Preparation of metakaolin based geopolymer coatings on metal substrates as thermal barriers. Appl. Clay Sci. 2009, 46, 265–270. [Google Scholar] [CrossRef]
- McAlorum, J.; Perry, M.; Vlachakis, C.; Biondi, L.; Lavoie, B. Robotic spray coating of self-sensing metakaolin geopolymer for concrete monitoring. Autom. Constr. 2021, 121, 103415. [Google Scholar] [CrossRef]
- Lu, B.; Zhu, W.; Weng, Y.; Liu, Z.; Yang, E.H.; Leong, K.F.; Qian, S. Study of MgO-activated slag as a cementless material for sustainable spray-based 3D printing. J. Clean. Prod. 2020, 258, 120671. [Google Scholar] [CrossRef]
- Zhang, Z.; Yao, X.; Wang, H. Potential application of geopolymers as protection coatings for marine concrete III: Field experiment. Appl. Clay Sci. 2012, 67–68, 57–60. [Google Scholar] [CrossRef]
- Deshmukh, K.; Parsai, R.; Anshul, A.; Singh, A.; Bhardwaj, P.; Gupta, R.; Mishra, D.; Amritphale, S.S. Studies on fly ash basedgeopolymeric material for coating on mild steel by paint brush technique. Int. J. Adhes. Adhes. 2017, 75, 139–144. [Google Scholar] [CrossRef]
- Bhardwaj, P.; Gupta, R.; Deshmukh, K.; Mishra, D. Optimization studies and characterization of advanced geopolymer coatings for the fabrication of mild steel substrate by spin coating technique. Indian J. Chem. Technol. 2021, 28, 59–76. Available online: https://op.niscair.res.in/index.php/IJCT/article/view/31721 (accessed on 12 August 2022).
- Provis, J.L.; Van Deventer, J.S. Geopolymers—Structure, Processing, Properties, and Industrial Application; Woodhead Publishing in Materials: Oxford, UK, 2009; pp. 310–311. [Google Scholar]
- Lizcano, M.; Gonzalez, A.; Basu, S.; Lozano, K.; Radovic, M. Effects of water content and chemical composition on structural properties of alkaline activated metakaolin-based geopolymers. J. Am. Ceram. Soc. 2012, 95, 2169–2177. [Google Scholar] [CrossRef]
- Perera, D.S.; Vance, E.; Finnie, K.S.; Blackford, M.G.; Cassidy, D.J. Disposition of water in Metakaolinite based geopolymers. Adv. Ceram. Matrix Compos. 2006, 75, 225–236. [Google Scholar]
- Song, Z.L.; Ma, L.Q.; Wu, Z.J.; He, D.P. Effects of viscosity on the cellular structure of foamed aluminum in the foaming process. J. Mater. Sci. 2000, 35, 15–20. [Google Scholar] [CrossRef]
- Chindaprasirt, P.; Chareerat, T.; Sirivivatnanon, V. Workability, and strength of coarse high calcium fly ash geopolymer. Cem. Concr. Compos. 2007, 29, 224–229. [Google Scholar] [CrossRef]
- Ercoli, R.; Laskowska, D.; Nguyen, V.V.; Le, V.S.; Louda, P.; Łoś, P.; Ciemnicka, J.; Prałat, K.; Renzulli, A.; Paris, E.; et al. Mechanical and thermal properties of geopolymer foams (GFs) doped with by-products of the secondary aluminum industry. Polymers 2022, 14, 703. [Google Scholar] [CrossRef]
- Nguyen, V.V.; Le, V.S.; Louda, P.; Szczypiński, M.M.; Ercoli, R.; Vojtěch, R.; Łoś, P.; Prałat, K.; Plaskota, P.; Pacyniak, T.; et al. Low-density geopolymer composites for the construction industry. Polymers 2022, 14, 304. [Google Scholar] [CrossRef]
- Novais, R.M.; Ascensão, G.; Buruberri, L.H.; Senff, L.; Labrincha, J.A. Influence of blowing agent on the fresh- and hardened-state properties of lightweight geopolymers. Mater. Des. 2016, 108, 551–559. [Google Scholar] [CrossRef]
- Łach, M. Geopolymer Foams—Will They Ever Become a Viable Alternative to Popular Insulation Materials?—A Critical Opinion. Materials 2021, 14, 3568. [Google Scholar] [CrossRef] [PubMed]
- Łach, M.; Pławecka, K.; Bąk, A.; Lichocka, K.; Korniejenko, K.; Cheng, A.; Lin, W.-T. Determination of the influence of hydraulic additives on the foaming process and stability of the produced geopolymer foams. Materials 2021, 14, 5090. [Google Scholar] [CrossRef] [PubMed]
- Wongsa, A.; Sata, V.; Nuaklong, P.; Chindaprasirt, P. Use of crushed clay brick and pumice aggregates in lightweight geopolymer concrete. Constr. Build. Mater. 2018, 188, 1025–1034. [Google Scholar] [CrossRef]
- Demirboǧa, R.; Gül, R. The effects of expanded perlite aggregate, silica fume and fly ash on the thermal conductivity of lightweight concrete. Cem. Concr. Res. 2003, 33, 723–727. [Google Scholar] [CrossRef]
- Cui, C.; Wang, D. Effects of Water on Pore Structure and Thermal Conductivity of Fly Ash-Based Foam Geopolymers. Adv. Mater. Sci. Eng. 2019, 2019, 3202794. [Google Scholar] [CrossRef]
- Wu, J.; Zhang, Z.; Zhang, Y.; Li, D. Preparation and characterization of ultra-lightweight foamed geopolymer (UFG) based on fly ash-metakaolin blends. Constr. Build. Mater. 2018, 168, 771–779. [Google Scholar] [CrossRef]
- Zhang, Z.; Provis, L.J.; Reid, A.; Wang, H. Mechanical, thermal insulation, thermal resistance and acoustic absorption properties of geopolymer foam concrete. Cem. Concr. Compos. 2015, 62, 97–105. [Google Scholar] [CrossRef]
- Kurtuluş, C.; Baspinar, M.S. Development of Efflorescence Control Methods of Fly Ash Based Foam Geopolymers. AKU J. Sci. Eng. 2020, 20, 129–137. [Google Scholar] [CrossRef]
- Krzywoń, R.; Dawczyński, S. Strength Parameters of Foamed Geopolymer Reinforced with GFRP Mesh. Materials 2021, 14, 689. [Google Scholar] [CrossRef]
- Huang, Z.; Zhang, T.; Wen, Z. Proportioning and characterization of Portland cement-based ultra-lightweight foam concretes. Constr. Build. Mater. 2015, 79, 390–396. [Google Scholar] [CrossRef]
- Pławecka, K.; Bazan, P.; Lin, W.-T.; Korniejenko, K.; Sitarz, M.; Nykiel, M. Development of Geopolymers Based on Fly Ashes from Different Combustion Processes. Polymers 2022, 14, 1954. [Google Scholar] [CrossRef] [PubMed]
- Kang, X.; Gan, Y.; Chen, R.; Zhang, C. Sustainable eco-friendly bricks from slate tailings through geopolymerization: Synthesis and characterization analysis. Constr. Build. Mater. 2021, 278, 122337. [Google Scholar] [CrossRef]
- Su, Z.; Hou, W.; Sun, Z.; Lv, W. Study of In Situ Foamed Fly Ash Geopolymer. Materials 2020, 13, 4059. [Google Scholar] [CrossRef] [PubMed]
- Kristály, F.; Szabó, R.; Mádai, F.; Debreczeni, Á.; Mucsi, G. Lightweight composite from fly ash geopolymer and glass foam. J. Sustain. Cem. Based Mater. 2021, 10, 1–22. [Google Scholar] [CrossRef]


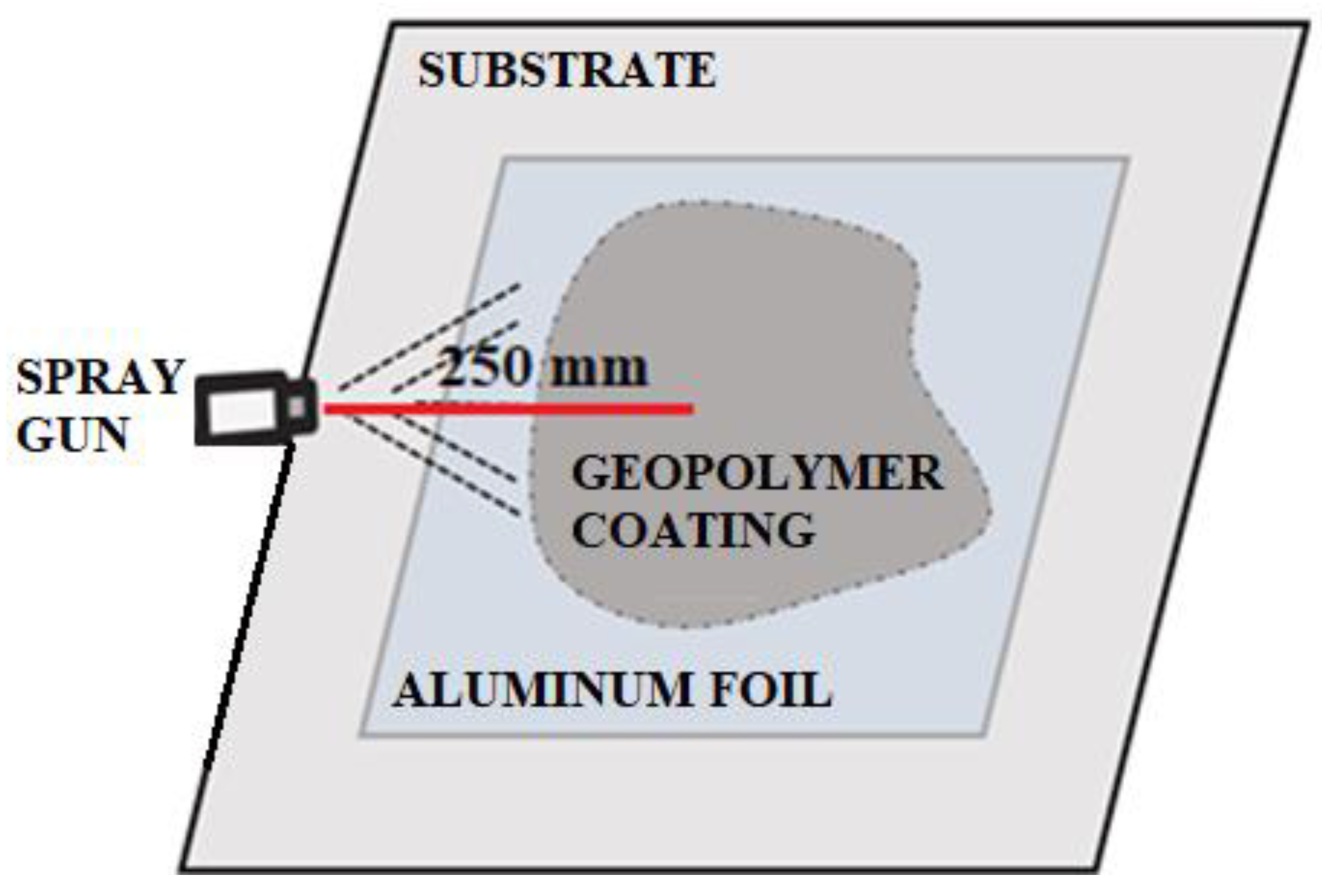

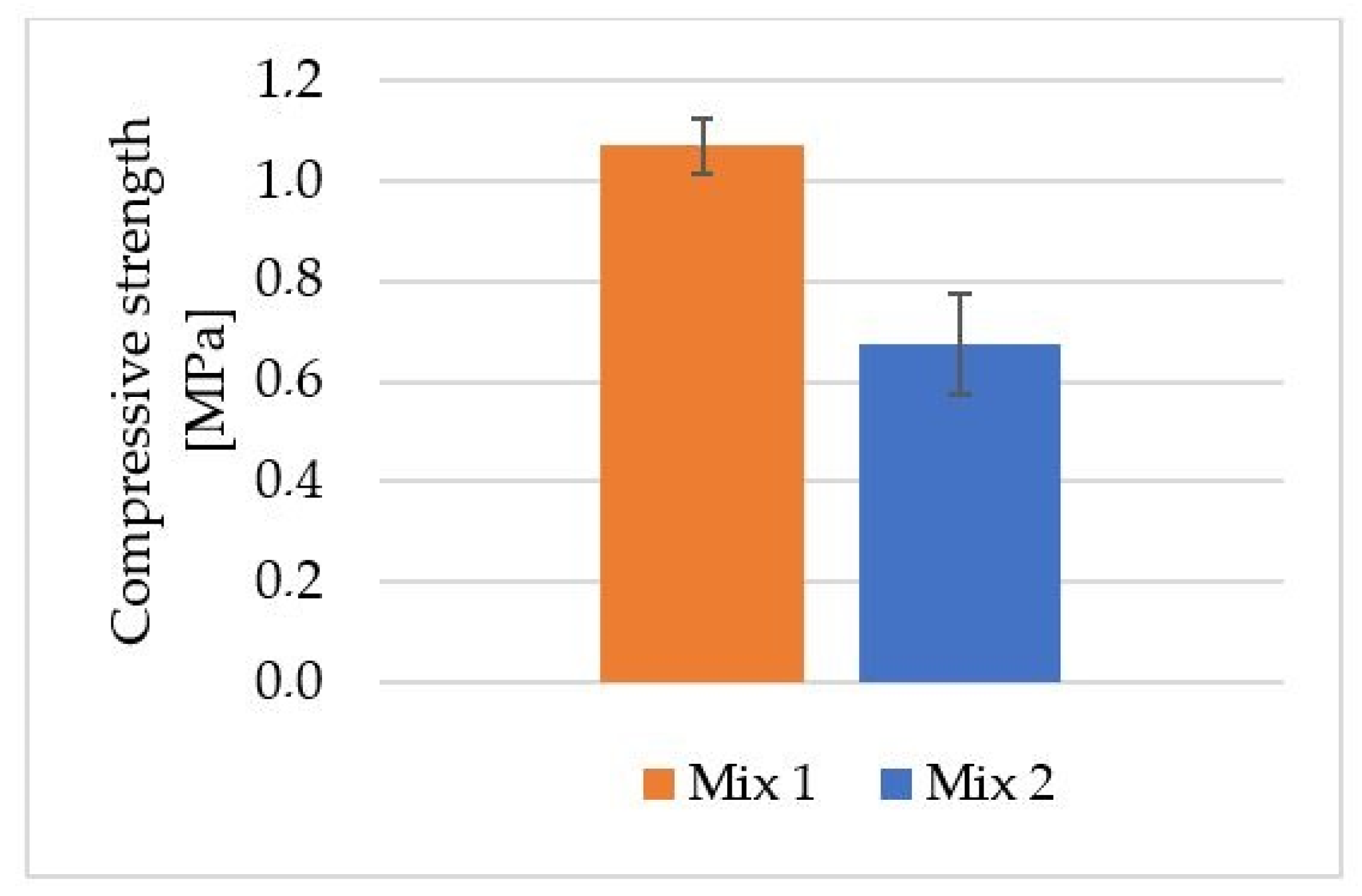
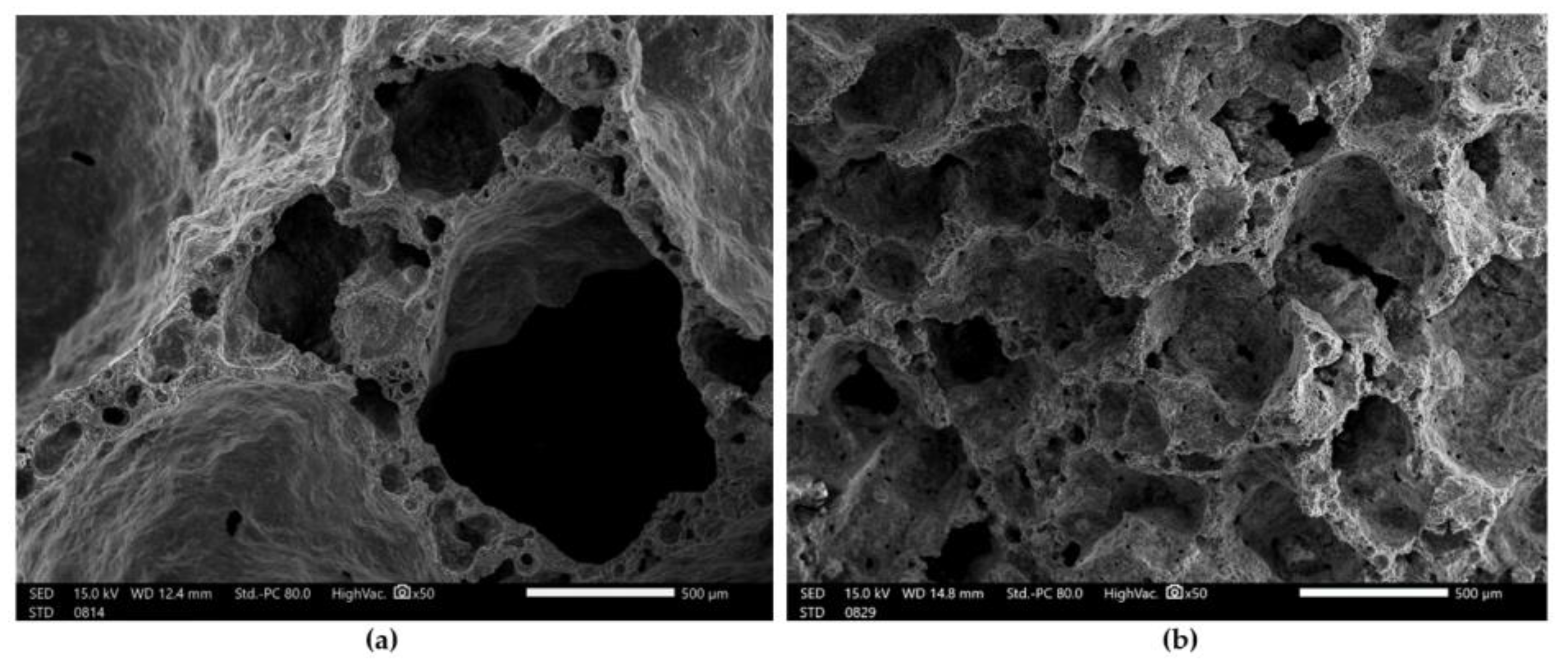
| Index | Description | Alkaline Activator | Foaming Agent | Mix Proportion (L/S/F) |
|---|---|---|---|---|
| Mix 1 | Geopolymer based on fly ash and river sand (weight ratio: 1:0.1) | 10 M NaOH + sodium water glass (weight ratio: 1:2.5) | H2O2 | 1:2.18:0.17 |
| Mix 2 | Geopolymer based on fly ash and river sand (weight ratio: 1:0.1) | 10 M NaOH + sodium water glass (weight ratio: 1:2.5) | Aluminum powder | 1:1.87:0.004 |
| Type | Manufacturer | Tank Capacity (m3) | Nozzle Diameter (m) | Working Pressure (MPa) | Diameter of Compressed Air Supply hose (m) |
|---|---|---|---|---|---|
| Spray gun—AD-8030P | ADLER | 0.005 | 0.004 | 0.4–0.8 | 0.008 |
| Spray gun—AD-8030P | ADLER | 0.005 | 0.006 | 0.4–0.8 | 0.008 |
| Spray gun—AD-8030P | ADLER | 0.005 | 0.008 | 0.4–0.8 | 0.008 |
| Spray Gun Mode | Pressure Range (MPa) |
|---|---|
| Low pressure | 0.4–0.5 |
| Medium pressure | 0.51–0.65 |
| High pressure | 0.66–0.8 |
| Oxide Composition | SiO2 | Al2O3 | Fe2O3 | K2O | CaO | MgO | TiO2 | Na2O | P2O5 | SO2 |
|---|---|---|---|---|---|---|---|---|---|---|
| Fly ash | 49.69% | 24.84% | 7.20% | 3.59% | 3.77% | 1.52% | 1.13% | 1.88% | 0.33% | 0.96% |
| D-Values | Fly Ash | River Sand |
|---|---|---|
| D10 (µm) | 2.208 | 400.776 |
| D50 (µm) | 12.268 | 647.689 |
| D90 (µm) | 36.175 | 1036.757 |
| Mean size (µm) | 16.975 | 730.226 |
| Mix 1 | Mix 2 | |
|---|---|---|
| Low pressure | No spraying of the geopolymer coating is possible. | No spraying of the geopolymer coating is possible. |
| Medium pressure |  |  |
| High pressure |  |  |
| Material | Mean Temp. (K) | Delta Temp. (K) | Thermal Conductivity (W/(m∙K)) | Load Pressure (kPa) |
|---|---|---|---|---|
| Cardboard | 283.15 | 20.0 | 0.04169 | 2.0 |
| Geopolymer layer (Mix 2) on the cardboard | 283.15 | 20.0 | 0.15545 | 0.6 |
| Material | Mean Temp. (K) | Delta Temp. (K) | Thermal Conductivity [W/(m∙K)) | Load Pressure (kPa) |
|---|---|---|---|---|
| Mix 1 | 283.15 | 20.0 | 0.12493 | 2.2 |
| Mix 2 | 283.15 | 20.0 | 0.12581 | 2.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaczmarski, K.; Pławecka, K.; Kozub, B.; Bazan, P.; Łach, M. Preliminary Investigation of Geopolymer Foams as Coating Materials. Appl. Sci. 2022, 12, 11205. https://doi.org/10.3390/app122111205
Kaczmarski K, Pławecka K, Kozub B, Bazan P, Łach M. Preliminary Investigation of Geopolymer Foams as Coating Materials. Applied Sciences. 2022; 12(21):11205. https://doi.org/10.3390/app122111205
Chicago/Turabian StyleKaczmarski, Krzysztof, Kinga Pławecka, Barbara Kozub, Patrycja Bazan, and Michał Łach. 2022. "Preliminary Investigation of Geopolymer Foams as Coating Materials" Applied Sciences 12, no. 21: 11205. https://doi.org/10.3390/app122111205
APA StyleKaczmarski, K., Pławecka, K., Kozub, B., Bazan, P., & Łach, M. (2022). Preliminary Investigation of Geopolymer Foams as Coating Materials. Applied Sciences, 12(21), 11205. https://doi.org/10.3390/app122111205







