Physiological Effects of Bioactive Compounds Derived from Whole Grains on Cardiovascular and Metabolic Diseases
Abstract
:1. Introduction
2. Associations of Whole Grain-Specific Polyphenols with Cardiovascular and Metabolic Diseases
2.1. Alkylresorcinols (ARs)
2.2. Avenanthramides (Avns)
2.3. Ferulic Acid (FA) and γ-Oryzanol (OZ)
3. Associations of Flavonoids with Cardiovascular and Metabolic Diseases
Rutin
4. Associations of Vitamin E with Cardiovascular and Metabolic Diseases
4.1. Tocotrienol
4.2. α-Tocopherol
5. Associations of Fiber with Cardiovascular and Metabolic Diseases
β-Glucan
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Roth, G.A.; Mensah, G.A.; Johnson, C.O.; Addolorato, G.; Ammirati, E.; Baddour, L.M.; Barengo, N.C.; Beaton, A.Z.; Benjamin, E.J.; Benziger, C.P.; et al. Global burden of cardiovascular diseases and risk factors, 1990–2019. J. Am. Coll. Cardiol. 2020, 76, 2982–3021. [Google Scholar] [CrossRef]
- Musinguzi, G.; Ndejjo, R.; Ssinabulya, I.; Bastiaens, H.; van Marwijk, H.; Wanyenze, R.K. Cardiovascular risk factor mapping and distribution among adults in mukono and buikwe districts in Uganda: Small area analysis. BMC Cardiovasc. Disord. 2020, 20, 284. [Google Scholar] [CrossRef] [PubMed]
- Olatona, F.A.; Onabanjo, O.O.; Ugbaja, R.N.; Nnoaham, K.E.; Adelekan, D.A. Dietary habits and metabolic risk factors for non-communicable diseases in a University undergraduate population. J. Health Popul. Nutr. 2018, 37, 21. [Google Scholar] [CrossRef] [PubMed]
- Grundy, S.M. Metabolic syndrome pandemic. Arter. Thromb. Vasc. Biol. 2008, 28, 629–636. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Venkatakrishnan, K.; Chiu, H.-F.; Wang, C.K. Extensive review of popular functional foods and nutraceuticals against obesity and its related complications with a special focus on randomized clinical trials. Food Funct. 2019, 10, 2313–2329. [Google Scholar] [CrossRef]
- Alkhatib, A.; Tsang, C.; Tiss, A.; Bahorun, T.; Arefanian, H.; Barake, R.; Khadir, A.; Tuomilehto, J. Functional foods and lifestyle approaches for diabetes prevention and management. Nutrients 2017, 9, 1310. [Google Scholar] [CrossRef] [Green Version]
- Trumbo, P.; Schlicker, S.; Yates, A.A.; Poos, M. Food, nutrition board of the institute of medicine TNA: Dietary reference intakes for energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein and amino acids. J. Am. Diet Assoc. 2002, 102, 1621–1630. [Google Scholar] [CrossRef]
- Jiang, H.; Zhang, J.; Du, W.; Su, C.; Zhang, B.; Wang, H. Energy intake and energy contributions of macronutrients and major food sources among Chinese adults: CHNS 2015 and CNTCS 2015. Eur. J. Clin. Nutr. 2021, 75, 314–324. [Google Scholar] [CrossRef]
- Gose, M.; Krems, C.; Heuer, T.; Hoffmann, I. Trends in food consumption and nutrient intake in Germany between 2006 and 2012: Results of the German National Nutrition Monitoring (NEMONIT). Br. J. Nutr. 2016, 115, 1498–1507. [Google Scholar] [CrossRef] [Green Version]
- Wright, J.D.; Wang, C.Y. Trends in intake of energy and macronutrients in adults from 1999–2000 through 2007–2008. NCHS Data Brief 2010, 49, 1–8. [Google Scholar]
- Sabença, C.; Ribeiro, M.; Sousa, T.; Poeta, P.; Bagulho, A.; Igrejas, G. Wheat/Gluten-Related Disorders and Gluten-Free Diet Misconceptions: A Review. Foods 2021, 10, 1765. [Google Scholar] [CrossRef]
- Slavin, J.L.; Jacobs, D.; Marquart, L.; Wiemer, K. The role of whole grains in disease prevention. J. Am. Diet. Assoc. 2001, 101, 780–785. [Google Scholar] [CrossRef]
- Drozdowski, L.A.; Reimer, R.A.; Temelli, F.; Bell, R.C.; Vasanthan, T.; Thomson, A.B. Beta-glucan extracts inhibit the in vitro intestinal uptake of long-chain fatty acids and cholesterol and down-regulate genes involved in lipogenesis and lipid transport in rats. J. Nutr. Biochem. 2010, 21, 695–701. [Google Scholar] [CrossRef] [Green Version]
- Qureshi, A.A.; Sami, S.A.; Salser, W.A.; Khan, F.A. Dose-dependent suppression of serum cholesterol by tocotrienol-rich fraction (trf25) of rice bran in hypercholesterolemic humans. Atherosclerosis 2002, 161, 199–207. [Google Scholar] [CrossRef]
- Senaphan, K.; Kukongviriyapan, U.; Sangartit, W.; Pakdeechote, P.; Pannangpetch, P.; Prachaney, P.; Greenwald, S.E.; Kukongviriyapan, V. Ferulic acid alleviates changes in a rat model of metabolic syndrome induced by high-carbohydrate, high-fat diet. Nutrients 2015, 7, 6446–6464. [Google Scholar] [CrossRef]
- Sun, T.; Zhang, Y.; Huang, H.; Wang, X.; Zhou, L.; Li, S.; Huang, S.; Xie, C.; Wen, Y.; Zhu, Y.; et al. Plasma alkylresorcinol metabolite, a biomarker of whole-grain wheat and rye intake, and risk of ischemic stroke: A case-control study. Am. J. Clin. Nutr. 2019, 109, 1–7. [Google Scholar] [CrossRef]
- Thomas, M.; Kim, S.; Guo, W.; Collins, F.W.; Wise, M.L.; Meydani, M. High levels of avenanthramides in oat-based diet further suppress high fat diet-induced atherosclerosis in Ldlr-/- Mice. J. Agric. Food Chem. 2018, 66, 498–504. [Google Scholar] [CrossRef]
- Wang, O.; Liu, J.; Cheng, Q.; Guo, X.; Wang, Y.; Zhao, L.; Zhou, F.; Ji, B. Effects of ferulic acid and gamma-oryzanol on high-fat and high-fructose diet-induced metabolic syndrome in rats. PLoS ONE 2015, 10, e0118135. [Google Scholar]
- Ganesan, K.; Xu, B. Polyphenol-rich lentils and their health promoting effects. Int. J. Mol. Sci. 2017, 18, 2390. [Google Scholar] [CrossRef] [Green Version]
- Surampudi, P.; Enkhmaa, B.; Anuurad, E.; Berglund, L. Lipid lowering with soluble dietary fiber. Curr. Atheroscler. Rep. 2016, 18, 75. [Google Scholar] [CrossRef]
- Yu, K.; Ke, M.Y.; Li, W.H.; Zhang, S.Q.; Fang, X.C. The impact of soluble dietary fibre on gastric emptying, postprandial blood glucose and insulin in patients with Type 2 diabetes. Asia Pac. J. Clin. Nutr. 2014, 23, 210–218. [Google Scholar] [PubMed]
- Jiang, Q. Natural forms of vitamin e: Metabolism, antioxidant, and anti-inflammatory activities and their role in disease prevention and therapy. Free Radic. Biol. Med. 2014, 72, 76–90. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reynolds, A.; Mann, J.; Cummings, J.; Winter, N.; Mete, E.; Morenga, L.T. Carbohydrate quality and human health: A series of systematic reviews and meta-analyses. Lancet 2019, 393, 434–445. [Google Scholar] [CrossRef] [Green Version]
- Benisi-Kohansal, S.; Saneei, P.; Salehi-Marzijarani, M.; Larijani, B.; Esmaillzadeh, A. Whole-grain intake and mortality from all causes, cardiovascular disease, and cancer: A systematic review and dose–response meta-analysis of prospective cohort studies. Adv. Nutr. 2016, 7, 1052–1065. [Google Scholar] [CrossRef] [Green Version]
- Aune, D.; Keum, N.; Giovannucci, E.; Fadnes, L.T.; Boffetta, P.; Greenwood, D.C.; Tonstad, S.; Vatten, L.J.; Riboli, E.; Norat, T. Whole grain consumption and risk of cardiovascular disease, cancer, and all cause and cause specific mortality: Systematic review and dose-response meta-analysis of prospective studies. BMJ 2016, 353, i2716. [Google Scholar] [CrossRef] [Green Version]
- Velderrain-Rodríguez, G.R.; Palafox-Carlos, H.; Wall-Medrano, A.; Ayala-Zavala, J.F.; Chen, C.Y.; Robles-Sánchez, M.; Astiazaran-García, H.; Alvarez-Parrilla, E.; González-Aguilar, G.A. Phenolic compounds: Their journey after intake. Food Funct. 2014, 5, 189–197. [Google Scholar] [CrossRef]
- Cory, H.; Passarelli, S.; Szeto, J.; Tamez, M.; Mattei, J. The Role of polyphenols in human health and food systems: A mini-review. Front. Nutr. 2018, 5, 87. [Google Scholar] [CrossRef] [Green Version]
- Tangney, C.C.; Rasmussen, H.E. Polyphenols, inflammation, and cardiovascular disease. Curr. Atheroscler. Rep. 2013, 15, 324. [Google Scholar] [CrossRef]
- Baerson, S.R.; Schröder, J.; Cook, D.; Rimando, A.M.; Pan, Z.; Dayan, F.E.; Noonan, B.P.; Duke, S.O. Alkylresorcinol biosynthesis in plants: New insights from an ancient enzyme family? Plant. Signal. Behav. 2010, 5, 1286–1289. [Google Scholar] [CrossRef] [Green Version]
- Quistad, G.B.; Staiger, L.E.; Schooley, D.A. Environmental degradation of the miticide cycloprate (Hexadecyl Cyclopro-panecarboxylate). 1. Rat metabolism. J. Agric. Food Chem. 1978, 26, 60–66. [Google Scholar] [CrossRef]
- Zhao, Z.; Moghadasian, M.H. Chemistry, natural sources, dietary intake and pharmacokinetic properties of ferulic acid: A review. Food Chem. 2008, 109, 691–702. [Google Scholar] [CrossRef]
- Scavariello, E.M.; Arellano, D.B. Gamma-oryzanol: An important component in rice brain oil. Arch. Latinoam. Nutr. 1998, 48, 7–12. [Google Scholar]
- Savolainen, O.; Lind, M.V.; Bergström, G.; Fagerberg, B.; Sandberg, A.S.; Ross, A. Biomarkers of food intake and nutrient status are associated with glucose tolerance status and development of type 2 Diabetes in older swedish women. Am. J. Clin. Nutr. 2017, 106, 1302–1310. [Google Scholar] [CrossRef] [Green Version]
- Tryggvadottir, E.A.; Halldorsson, T.I.; Landberg, R.; Hrolfsdottir, L.; Birgisdottir, B.E.; Magnusdottir, O.K.; Hreidarsdottir, I.T.; Hardardottir, H.; Gunnarsdottir, I. Higher alkylresorcinol concentrations, a consequence of whole-grain intake, are inversely associated with gestational Diabetes mellitus in Iceland. J. Nutr. 2021, 151, 1159–1166. [Google Scholar] [CrossRef]
- Sun, T.; Rong, Y.; Hu, X.; Zhu, Y.; Huang, H.; Chen, L.; Li, P.; Li, S.; Yang, W.; Cheng, J.; et al. Plasma alkylresorcinol metabolite, a biomarker of whole-grain wheat and rye intake, and risk of type 2 Diabetes and impaired glucose regulation in a Chinese population. Diabetes Care 2018, 41, 440–445. [Google Scholar] [CrossRef] [Green Version]
- Oishi, K.; Yamamoto, S.; Itoh, N.; Nakao, R.; Yasumoto, Y.; Tanaka, K.; Kikuchi, Y.; Fukudome, S.; Okita, K.; Takano-Ishikawa, Y. Wheat alkylresorcinols suppress high-fat, high-sucrose diet-induced obesity and glucose intolerance by increasing insulin sensitivity and cholesterol excretion in male mice. J. Nutr. 2015, 145, 199–206. [Google Scholar] [CrossRef] [Green Version]
- Nie, L.; Wise, M.L.; Peterson, D.M.; Meydani, M. Avenanthramide, a polyphenol from oats, inhibits vascular smooth muscle cell proliferation and enhances nitric oxide production. Atherosclerosis 2006, 186, 260–266. [Google Scholar] [CrossRef]
- Serreli, G.; Le Sayec, M.; Thou, E.; Lacour, C.; Diotallevi, C.; Dhunna, M.A.; Deiana, M.; Spencer, J.P.E.; Corona, G. Ferulic acid derivatives and avenanthramides modulate endothelial function through maintenance of nitric oxide balance in HUVEC cells. Nutrients 2021, 13, 2026. [Google Scholar] [CrossRef]
- Nie, L.; Wise, M.; Peterson, D.; Meydani, M. Mechanism by which avenanthramide-c, a polyphenol of oats, blocks cell cycle progression in vascular smooth muscle cells. Free Radic. Biol. Med. 2006, 41, 702–708. [Google Scholar] [CrossRef]
- Liu, L.; Zubik, L.; Collins, F.W.; Marko, M.; Meydani, M. The antiatherogenic potential of oat phenolic compounds. Atherosclerosis 2004, 175, 39–49. [Google Scholar] [CrossRef]
- Park, J.; Choi, H.; Abekura, F.; Lim, H.S.; Im, J.H.; Yang, W.S.; Hwang, C.W.; Chang, Y.C.; Lee, Y.C.; Park, N.G.; et al. Avenanthramide C suppresses matrix metalloproteinase-9 expression and migration through the mapk/nf-κb signaling pathway in TNF-alpha-activated HASMC cells. Front. Pharmacol. 2021, 12, 621854. [Google Scholar] [CrossRef]
- Guo, W.; Wise, M.L.; Collins, F.W.; Meydani, M. Avenanthramides, polyphenols from oats, inhibit IL-1beta-induced NF-KappaB activation in endothelial cells. Free Radic. Biol. Med. 2008, 44, 415–429. [Google Scholar] [CrossRef]
- Sombutsuwan, P.; Jirattisakul, A.; Nakornsadet, A.; Akepratumchai, S.; Chumsantea, S.; Pojjanapornpun, S.; Lilitchan, S.; Krisnangkura, K.; Aryusuk, K. A simple and efficient method for synthesis and extraction of ethyl ferulate from gamma-oryzanol. J. Oleo Sci. 2021, 70, 757–767. [Google Scholar] [CrossRef]
- Raj, P.; Ames, N.; Thandapilly, S.J.; Yu, L.; Netticadan, T. The effects of oat ingredients on blood pressure in spontaneously hypertensive rats. J. Food Biochem. 2020, 44, e13402. [Google Scholar] [CrossRef]
- Jung, E.H.; Kim, S.R.; Hwang, I.K.; Ha, T.Y. Hypoglycemic effects of a phenolic acid fraction of rice bran and ferulic acid in C57BL/KsJ-db/db mice. J. Agric. Food Chem. 2007, 55, 9800–9804. [Google Scholar] [CrossRef]
- Jin Son, M.; Rico, C.W.; Hyun, N.S.; Young, K.M. Influence of Oryzanol and Ferulic acid on the lipid metabolism and antioxidative status in high fat-fed mice. J. Clin. Biochem. Nutr. 2010, 46, 150–156. [Google Scholar] [CrossRef] [Green Version]
- Wilson, T.A.; Nicolosi, R.J.; Woolfrey, B.; Kritchevsky, D. Rice bran oil and oryzanol reduce plasma lipid and lipoprotein cholesterol concentrations and aortic cholesterol ester accumulation to a greater extent than ferulic acid in hypercholesterolemic hamsters. J. Nutr. Biochem. 2007, 18, 105–112. [Google Scholar] [CrossRef]
- Guo, X.X.; Zeng, Z.; Qian, Y.Z.; Qiu, J.; Wang, K.; Wang, Y.; Ji, B.P.; Zhou, F. Wheat flour, enriched with gamma-oryzanol, phytosterol, and ferulic acid, alleviates lipid and glucose metabolism in high-fat-fructose-fed rats. Nutrients 2019, 11, 1697. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Balasubashini, M.S.; Rukkumani, R.; Menon, V.P. Protective effects of ferulic acid on hyperlipidemic diabetic rats. Acta Diabetol. 2003, 40, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Ardiansyah; Ohsaki, Y.; Shirakawa, H.; Koseki, T.; Komai, M. Novel effects of a single administration of ferulic acid on the regulation of blood pressure and the hepatic lipid metabolic profile in stroke-prone spontaneously hypertensive rats. J. Agric. Food Chem. 2008, 56, 2825–2830. [Google Scholar]
- Suzuki, A.; Yamamoto, M.; Jokura, H.; Fujii, A.; Tokimitsu, I.; Hase, T.; Saito, I. Ferulic acid restores endothelium-dependent vasodilation in aortas of spontaneously hypertensive rats. Am. J. Hypertens. 2007, 20, 508–513. [Google Scholar] [CrossRef] [PubMed]
- Kent, K.; Charlton, K.; O’Sullivan, T.; Oddy, W.H. Estimated intake and major food sources of flavonoids among australian adolescents. Eur. J. Nutr. 2020, 59, 3841–3856. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Hou, Z.; Yi, R.; Wang, Z.; Sun, P.; Li, G.; Zhao, X.; Wang, Q. Tartary buckwheat flavonoids ameliorate high fructose-induced insulin resistance and oxidative stress associated with the insulin signaling and Nrf2/HO-1 pathways in mice. Food Funct. 2017, 8, 2803–2816. [Google Scholar] [CrossRef] [PubMed]
- Hou, Z.; Hu, Y.; Yang, X.; Chen, W. Antihypertensive effects of tartary buckwheat flavonoids by improvement of vascular insulin sensitivity in spontaneously hypertensive rats. Food Funct. 2017, 8, 4217–4228. [Google Scholar] [CrossRef]
- Ghorbani, A. Mechanisms of antidiabetic effects of flavonoid rutin. Biomed. Pharmacother. 2017, 96, 305–312. [Google Scholar] [CrossRef]
- Da Silva, A.B.; Cerqueira Coelho, P.L.; das Neves Oliveira, M.; Oliveira, J.L.; Oliveira Amparo, J.A.; da Silva, K.C.; Soares, J.R.P.; Pitanga, B.P.S.; Dos Santos, S.C.; de Faria, L.G.P.; et al. The flavonoid rutin and its aglycone quercetin modulate the microglia inflammatory profile improving antiglioma activity. Brain Behav. Immun. 2020, 85, 170–185. [Google Scholar] [CrossRef]
- Cai, E.P.; Lin, J.K. Epigallocatechin gallate (egcg) and rutin suppress the glucotoxicity through activating IRS2 and AMPK signaling in rat pancreatic beta cells. J. Agric. Food Chem. 2009, 57, 9817–9827. [Google Scholar] [CrossRef]
- Wieslander, G.; Fabjan, N.; Vogrincic, M.; Kreft, I.; Janson, C.; Spetz-Nyström, U.; Vombergar, B.; Tagesson, C.; Leanderson, P.; Norbäck, D. Eating buckwheat cookies is associated with the reduction in serum levels of myeloperoxidase and cholesterol: A double blind crossover study in day-care centre staffs. Tohoku J. Exp. Med. 2011, 225, 123–130. [Google Scholar] [CrossRef] [Green Version]
- Halliwell, B.; Gutteridge, J.M. The definition and measurement of antioxidants in biological systems. Free Radic. Biol. Med. 1995, 18, 125–126. [Google Scholar] [CrossRef]
- Wang, H.; Cao, G.; Prior, R.L. Total antioxidant capacity of fruits. J. Agric. Food Chem. 1996, 44, 701–705. [Google Scholar] [CrossRef]
- Maritim, A.C.; Sanders, R.A.; Watkins, J.B., 3rd. Diabetes, oxidative stress, and antioxidants: A review. J. Biochem. Mol. Toxicol. 2003, 17, 24–38. [Google Scholar] [CrossRef]
- Siti, H.N.; Kamisah, Y.; Kamsiah, J. The role of oxidative stress, antioxidants and vascular inflammation in cardiovascular disease (A review). Vascul. Pharmacol. 2015, 71, 40–56. [Google Scholar] [CrossRef]
- Gaziano, J.M.; Hennekens, C.H. Vitamin antioxidants and cardiovascular disease. Curr. Opin. Lipidol. 1992, 3, 291–294. [Google Scholar] [CrossRef]
- Gale, C.R.; Martyn, C.N.; Winter, P.D.; Cooper, C. Vitamin C and risk of death from stroke and coronary heart disease in cohort of elderly people. BMJ 1995, 310, 1563–1566. [Google Scholar] [CrossRef] [Green Version]
- Aune, D.; Keum, N.; Giovannucci, E.; Fadnes, L.T.; Boffetta, P.; Greenwood, D.C.; Tonstad, S.; Vatten, L.J.; Riboli, E.; Norat, T. Dietary intake and blood concentrations of antioxidants and the risk of cardiovascular disease, total cancer, and all-cause mortality: A systematic review and dose–response meta-analysis of prospective studies. Am. J. Clin. Nutr. 2018, 108, 1069–1091. [Google Scholar] [CrossRef]
- Pang, K.L.; Chin, K.Y. The role of tocotrienol in protecting against metabolic diseases. Molecules 2019, 24, 923. [Google Scholar] [CrossRef] [Green Version]
- Qureshi, A.A.; Peterson, D.M.; Hasler-Rapacz, J.O.; Rapacz, J. Novel tocotrienols of rice bran suppress cholesterogenesis in hereditary hypercholesterolemic swine. J. Nutr. 2001, 131, 223–230. [Google Scholar] [CrossRef]
- Minhajuddin, M.; Beg, Z.H.; Iqbal, J. Hypolipidemic and antioxidant properties of tocotrienol rich fraction isolated from rice bran oil in experimentally induced hyperlipidemic rats. Food Chem. Toxicol. 2005, 43, 747–753. [Google Scholar] [CrossRef]
- Siddiqui, S.; Rashid, K.M.; Siddiqui, W.A. Comparative hypoglycemic and nephroprotective effects of tocotrienol rich fraction (TRF) from palm oil and rice bran oil against hyperglycemia induced nephropathy in type 1 Diabetic rats. Chem. Biol. Interact. 2010, 188, 651–658. [Google Scholar] [CrossRef]
- Siddiqui, S.; Ahsan, H.; Khan, M.R.; Siddiqui, W.A. Protective effects of tocotrienols against lipid-induced nephropathy in experimental type-2 Diabetic rats by modulation in TGF-beta expression. Toxicol. Appl. Pharmacol. 2013, 273, 314–324. [Google Scholar] [CrossRef]
- Iqbal, J.; Minhajuddin, M.; Beg, Z.H. Suppression of 7,12-Dimethylbenz[α]anthracene-induced carcinogenesis and hypercholesterolaemia in rats by tocotrienol-rich fraction isolated from rice bran oil. Eur. J. Cancer Prev. 2003, 12, 447–453. [Google Scholar] [CrossRef]
- Fukui, K.; Shirai, M.; Ninuma, T.; Kato, Y. Anti-obesity effects of tocotrienols and bran in high-fat diet-treated mice. Nutrients 2019, 11, 830. [Google Scholar] [CrossRef] [Green Version]
- Baliarsingh, S.; Beg, Z.H.; Ahmad, J. The therapeutic impacts of tocotrienols in type 2 Diabetic patients with hyperlipidemia. Atherosclerosis 2005, 182, 367–374. [Google Scholar] [CrossRef]
- Yang, C.S.; Luo, P.; Zeng, Z.; Wang, H.; Malafa, M.; Suh, N. Vitamin E and cancer prevention: Studies with different forms of tocopherols and tocotrienols. Mol. Carcinog. 2020, 59, 365–389. [Google Scholar] [CrossRef]
- Sookwong, P.; Nakagawa, K.; Murata, K.; Kojima, Y.; Miyazawa, T. Quantitation of tocotrienol and tocopherol in various rice brans. J. Agric. Food Chem. 2007, 55, 461–466. [Google Scholar] [CrossRef]
- Aggarwal, B.B.; Sundaram, C.; Prasad, S.; Kannappan, R. Tocotrienols, the vitamin E of the 21st century: Its potential against cancer and other chronic diseases. Biochem. Pharmacol. 2010, 80, 1613–1631. [Google Scholar] [CrossRef] [Green Version]
- Di Vincenzo, A.; Tana, C.; El Hadi, H.; Pagano, C.; Vettor, R.; Rossato, M. Antioxidant, anti-inflammatory, and metabolic properties of tocopherols and tocotrienols: Clinical implications for vitamin e supplementation in diabetic kidney disease. Int. J. Mol. Sci. 2019, 20, 5101. [Google Scholar] [CrossRef] [Green Version]
- Shibata, A.; Kawakami, Y.; Kimura, T.; Miyazawa, T.; Nakagawa, K. Alpha-tocopherol attenuates the triglyceride- and cholesterol-lowering effects of rice bran tocotrienol in rats fed a western diet. J. Agric. Food Chem. 2016, 64, 5361–5366. [Google Scholar] [CrossRef]
- Qureshi, A.A.; Salser, W.A.; Parmar, R.; Emeson, E.E. Novel tocotrienols of rice bran inhibit atherosclerotic lesions in C57BL/6 ApoE-deficient mice. J. Nutr. 2001, 131, 2606–2618. [Google Scholar] [CrossRef] [Green Version]
- Kanaya, Y.; Doi, T.; Sasaki, H.; Fujita, A.; Matsuno, S.; Okamoto, K.; Nakano, Y.; Tsujiwaki, S.; Furuta, H.; Nishi, M.; et al. Rice bran extract prevents the elevation of Plasma Peroxylipid in KKAy Diabetic mice. Diabetes Res. Clin. Pract. 2004, 66 (Suppl. S1), S157–S160. [Google Scholar] [CrossRef]
- Holscher, H.D. Dietary fiber and prebiotics and the Gastrointestinal Microbiota. Gut Microbes 2017, 8, 172–184. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.R.; Lineback, D.M.; Levine, M.J. Dietary reference intakes: Implications for fiber labeling and consumption: A summary of the International Life Sciences Institute North America fiber workshop, 1–2 June 2004, Washington, DC. Nutr. Rev. 2006, 64, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Stephen, A.M.; Cummings, J.H. Mechanism of action of dietary fibre in the human colon. Nature 1980, 284, 283–284. [Google Scholar] [CrossRef] [PubMed]
- Meyer, K.A.; Kushi, L.H.; Jacobs, D.R., Jr.; Slavin, J.; Sellers, T.A.; Folsom, A.R. Carbohydrates, dietary fiber, and incident type 2 Diabetes in older women. Am. J. Clin. Nutr. 2000, 71, 921–930. [Google Scholar] [CrossRef] [Green Version]
- Jenkins, D.J.; Wolever, T.M.; Leeds, A.R.; Gassull, M.A.; Haisman, P.; Dilawari, J.; Goff, D.V.; Metz, G.L.; Alberti, K.G. Dietary fibres, fibre analogues, and glucose tolerance: Importance of viscosity. Br. Med. J. 1978, 1, 1392–1394. [Google Scholar] [CrossRef] [Green Version]
- Jesch, E.D.; Carr, T.P. Food ingredients that inhibit cholesterol absorption. Prev. Nutr. Food Sci. 2017, 22, 67–80. [Google Scholar]
- Schulze, M.B.; Liu, S.; Rimm, E.B.; Manson, J.E.; Willett, W.C.; Hu, F.B. Glycemic index, glycemic load, and dietary fiber intake and incidence of type 2 Diabetes in younger and middle-aged women. Am. J. Clin. Nutr. 2004, 80, 348–356. [Google Scholar] [CrossRef]
- Tessari, P.; Lante, A. A Multifunctional bread rich in beta glucans and low in starch improves metabolic control in type 2 Diabetes: A controlled trial. Nutrients 2017, 9, 297. [Google Scholar] [CrossRef]
- Yao, B.; Fang, H.; Xu, W.; Yan, Y.; Xu, H.; Liu, Y.; Mo, M.; Zhang, H.; Zhao, Y. Dietary fiber intake and risk of type 2 Diabetes: A dose–response analysis of prospective studies. Eur. J. Epidemiol. 2014, 29, 79–88. [Google Scholar] [CrossRef]
- US Food and Drug Administration. Food labeling: Health claims; oats and coronary heart disease. Final rule. Fed. Regist 1997, 62, 3583–3601. [Google Scholar]
- Bell, S.; Goldman, V.M.; Bistrian, B.R.; Arnold, A.H.; Ostroff, G.; Forse, R.A. Effect of beta-glucan from oats and yeast on serum lipids. Crit. Rev. Food Sci. Nutr. 1999, 39, 189–202. [Google Scholar] [CrossRef]
- Biörklund, M.; van Rees, A.; Mensink, R.P.; Onning, G. Changes in serum lipids and postprandial glucose and insulin concentrations after consumption of beverages with beta-glucans from oats or barley: A randomised dose-controlled trial. Eur. J. Clin. Nutr. 2005, 59, 1272–1281. [Google Scholar] [CrossRef] [Green Version]
- Braaten, J.T.; Wood, P.J.; Scott, F.W.; Wolynetz, M.S.; Lowe, M.K.; Bradley-White, P.; Collins, M.W. Oat beta-glucan reduces blood cholesterol concentration in hypercholesterolemic subjects. Eur. J. Clin. Nutr. 1994, 48, 465–474. [Google Scholar]
- Theuwissen, E.; Mensink, R.P. Simultaneous intake of beta-glucan and plant stanol esters affects lipid metabolism in slightly hypercholesterolemic subjects. J. Nutr. 2007, 137, 583–588. [Google Scholar] [CrossRef] [Green Version]
- Dong, J.; Cai, F.; Shen, R.; Liu, Y. Hypoglycaemic effects and inhibitory effect on intestinal disaccharidases of oat beta-glucan in streptozotocin-induced diabetic mice. Food Chem. 2011, 129, 1066–1071. [Google Scholar] [CrossRef]
- Zhang, P.P.; Hu, X.Z.; Zhen, H.M.; Xu, C.; Fan, M.T. Oat beta-glucan increased atpases activity and energy charge in small intestine of rats. J. Agric. Food Chem. 2012, 60, 9822–9827. [Google Scholar] [CrossRef]
- Maki, K.C.; Galant, R.; Samuel, P.; Tesser, J.; Witchger, M.S.; Ribaya-Mercado, J.D.; Blumberg, J.B.; Geohas, J. Effects of consuming foods containing oat beta-glucan on blood pressure, carbohydrate metabolism and biomarkers of oxidative stress in men and women with elevated blood pressure. Eur. J. Clin. Nutr. 2007, 61, 786–795. [Google Scholar] [CrossRef] [Green Version]
- Mäkeläinen, H.; Anttila, H.; Sihvonen, J.; Hietanen, R.M.; Tahvonen, R.; Salminen, E.; Mikola, M.; Sontag-Strohm, T. The effect of beta-glucan on the glycemic and insulin index. Eur. J. Clin. Nutr. 2007, 61, 779–785. [Google Scholar] [CrossRef]
- Guo, H.; Ding, J.; Liang, J.; Zhang, Y. Associations of whole grain and refined grain consumption with metabolic syndrome. A meta-analysis of observational studies. Front. Nutr. 2021, 8, 695620. [Google Scholar] [CrossRef]
- Vitale, M.; Hanhineva, K.; Koistinen, V.; Auriola, S.; Paananen, J.; Costabile, G.; Della Pepa, G.; Rivellese, A.A.; Riccardi, G.; Giacco, R. Putative metabolites involved in the beneficial effects of wholegrain cereal: Nontargeted metabolite profiling approach. Nutr. Metab. Cardiovasc. Dis. 2021, 31, 1156–1165. [Google Scholar] [CrossRef]
- Kristensen, M.; Bügel, S. A Diet rich in oat bran improves blood lipids and hemostatic factors and reduces apparent energy digestibility in young healthy volunteers. Eur. J. Clin. Nutr. 2011, 65, 1053–1058. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qiu, J.; Liu, Y.; Yue, Y.; Qin, Y.; Li, Z. Dietary tartary buckwheat intake attenuates insulin resistance and improves lipid profiles in patients with type 2 Diabetes: A randomized controlled trial. Nutr. Res. 2016, 36, 1392–1401. [Google Scholar] [CrossRef] [PubMed]
- Vitaglione, P.; Mennella, I.; Ferracane, R.; Rivellese, A.A.; Giacco, R.; Ercolini, D.; Gibbons, S.M.; La Storia, A.; Gilbert, J.A.; Jonnalagadda, S.; et al. Whole-grain wheat consumption reduces inflammation in a randomized controlled trial on overweight and obese subjects with unhealthy dietary and lifestyle behaviors: Role of polyphenols bound to cereal dietary fiber. Am. J. Clin. Nutr. 2015, 101, 251–261. [Google Scholar] [CrossRef] [PubMed] [Green Version]
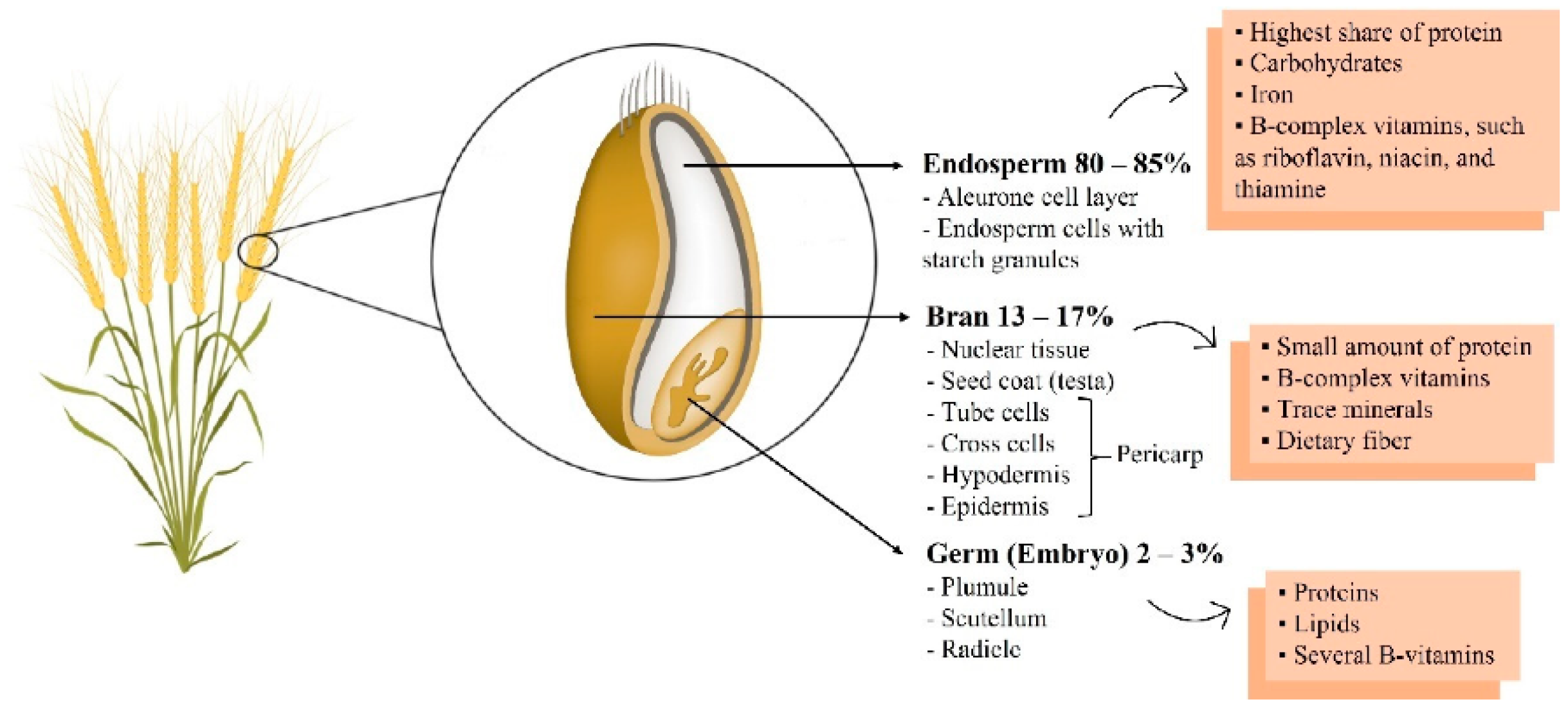
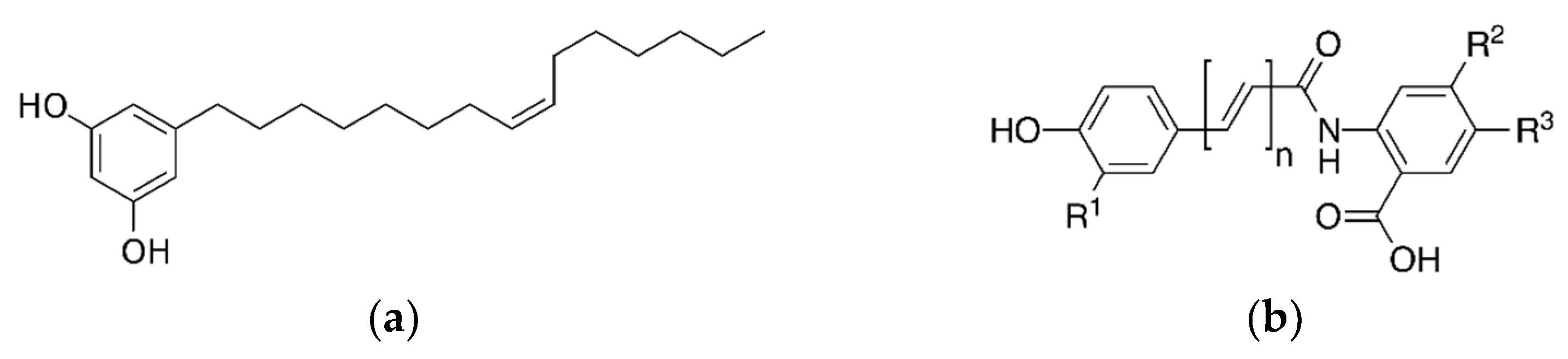
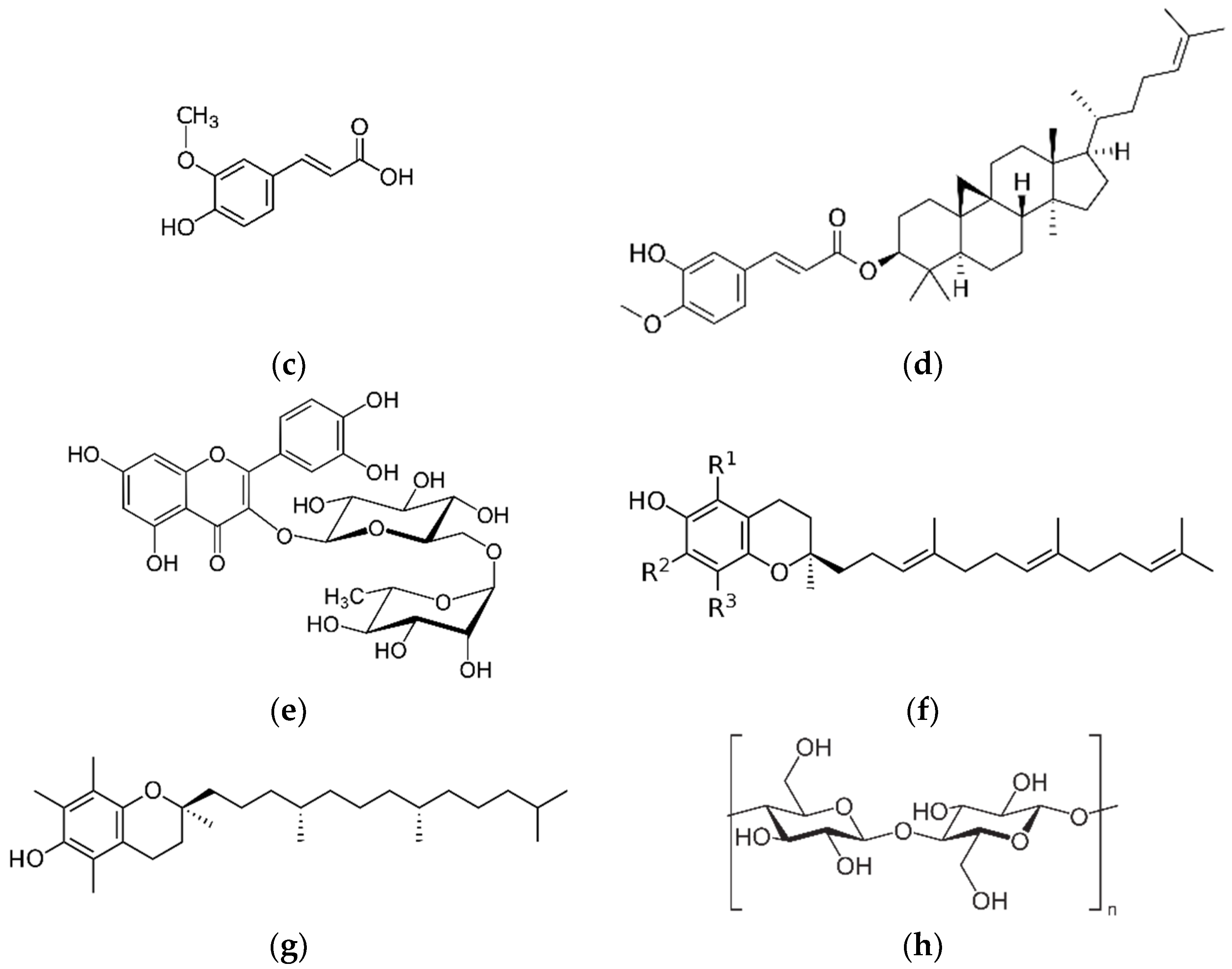
| Bioactive Compounds | Food Source | Study Subject or Model | Treatment or Intervention | Results | References |
|---|---|---|---|---|---|
| Alkylresorcinols (ARs) | |||||
| ARs | Whole grain | Human study - Nested case control study - 64 years, women | - 5 years follow-up - concentration of ARs | - Concentration of AR homologs C17 and C19 lower in subjects with IGT than that in normal subjects | [33] |
| ARs | Whole grain | Human study - Prospective cohort study - Pregnant women (gestational weeks 11–14) | - follow-up during pregnancy - total plasma AR Q1 (median 66 nmol/L) vs. Q4 (median 706 nmol/L) - weekly whole grain consumption Q1 (median 1.2 times/week) vs. Q4 (median 14.5 times/week) | - Frequency of whole-grain consumption lower in subjects with GDM than in subjects without GDM - Median concentration of ARs lower in subjects with GDM than in subjects without GDM - Highest ARs concentration quartile with an RR of 0.50 compared to lowest quartile | [34] |
| DHPPA [3-(3,5-dihydroxyphenyl)-1-propanoic acid] | Whole grain | Human study - Case-control study - Subjects with T2D and IGT - ≥30 years | - plasma DHPPA concentrations Q1 (<6.56 nmol/L) vs. Q4 (≥17.98 nmol/L) | - Negatively correlated with DHPPA concentrations and odds of T2D and IGT | [35] |
| ARs | Wheat bran | In vivo - 3-week-old, C57BL/6J mice fed with HFHS | - 10 weeks - 0.4% ARs containing diet | - Suppressed increases in bodyweight and hepatic TG accumulation - Suppressed increases in blood insulin and leptin concentration - Reduced levels of fasting and postprandial glucose and improved glucose intolerance and insulin resistance - Increased phosphorylation of insulin-stimulated hepatic serine/threonine protein kinase B - Increased fecal excretion of cholesterol and reduced blood cholesterol concentration - upregulated expression of hepatic cholesterol synthetic genes (Srebf2, Hmgcs1) | [36] |
| DHPPA (3-(3,5-dihydroxyphenyl)-1-propanoic acid) | Whole grain | Human study - Case-control study - Subjects with ischemic stroke - ≥35 years | - Concentration of DHPPA | - Negatively correlated with DHPPA concentrations and odds of ischemic stroke | [16] |
| Avenanthramides (Avns) | |||||
| Avns | Oats | In vivo - 5-week-old, Ldlr-/- mice fed with HFD | - 16 weeks - oat bran with low concentrations of Avns (8.8 g/kg, HFLA) or oat bran with high concentrations of Avns (480 g/kg, HFHA) diet | - Both oat-based diets decreased HFD-induced atheroma lesions in the aortic valve - HFHA administration for atheroma lesions is more effective than HFLA - TC level similarly reduced in both oat-based diets | [17] |
| Avn-A, B, C | Oats | In vitro - HUVECs | - Avn-A, B, C 1 μM | - Increased NO and cGMP levels - Increased Akt1 and eNOS expression | [38] |
| Avn-c | Oats | In vitro - Human aortic SMCs and HAECs | - Avn-c 40, 80, 120 μM | - Inhibited SMC proliferation - Increased NO secretion in both SMC and HAEC - Upregulated eNOS mRNA expression in both SMC and HAEC | [37] |
| Avn-c | Oats | In vitro - A10 rat embryonic aortic SMCs | - Avn-c 40, 80, 120 μM | - Arrested SMC proliferation at G1 phase - Decreased phosphorylation of pRb - Decreased cyclin D1 expression - Increased cyclin-dependent kinase inhibitor p21cip1 expression - Increased expression and stability of p53 protein | [39] |
| Avns | Oats | In vitro - IL-1β-stimulated HAECs | - Avns 4, 20, 40 ng/mL | - Decreased U937 monocytic cells adhesion in IL-1β-stimulated HAEC - Suppressed expressions of ICAM-1, VCAM-1, and E-selectin - Suppressed secretion of proinflammatory cytokines IL-6, IL-8, and MCP-1 | [40] |
| Avn-c | Oats | In vitro - TNF-α-stimulated HASMCs | - Avn-c 50, 100 ng/mL | - Inhibited cell migration - Suppressed increasing MMP-9 protein and mRNA levels - Inhibited MMP-9 enzyme activity - Reduced IL-6 level - Suppressed nuclear protein translocation of nuclear factor kappa B (NF-κB) - Reduced expression of ERK, JNK, and p38 phosphorylation | [41] |
| Avns | Oats | In vitro - IL-1β-stimulated HAECs - IL-1β-stimulated HUVECs | - AvnsO (oat extract) 4, 20, 40 mg/mL - Avn-c (Avn-c) 1, 10, 20, 40, 100 µM - CH3-Avn-c (methyl ester of Avn-c) 1, 10, 40, 100 µM | - AvnsO, Avn-c, and CH3-Avn-c suppressed NF-κB p50 DNA binding activity - CH3-Avn-c decreased expression of IL-6, IL-8, and MCP-1 - AvnsO, Avn-c, and CH3-Avn-c inhibited NF-κB-dependent reporter gene expression activated by TNFR-associated factor 2 and 6 (TRAF2, TRAF6) and NF-κB-inducing kinase - Decreased phosphorylation of IKKβ and IκB and stabilized IκB protein | [42] |
| Ferulic acid (FA) and γ-oryzanol (OZ) | |||||
| FA and OZ | Rice bran | In vivo - 4-week-old, C57BL/6 mice fed with HFD | - 7 weeks - 0.5% FA or OZ containing diet | - Decreased bodyweight gain in both groups - Decreased blood TG, TC, and increased plasma HDL-C in both groups - Decreased hepatic TG and TC in both groups - Decreased ME and FAS in both groups—increased GSH-Px, CAT, GR, and PON1 in both groups - Increased fecal excretion of TC in OZ treated group | [46] |
| FA and OZ | Rice bran | In vivo - F1B Golden Syrian hamsters fed with HCD | - 10 weeks - 0.5% FA or OZ containing diet | - Decreased blood TC, and non-HDL-C in both groups - Decreased plasma lipid hydroperoxides in both groups - Decreased aortic TC, and TG accumulation in both groups - Decreased blood TG and increased HDL-C in OZ treated group - Increased fecal excretion of TC and coprostanol in OZ treated group - Increased plasma vitamin E in FA treated group | [47] |
| FA and OZ | Whole grain wheat | In vivo - SD rats (weighing 180–200 g) fed with HFFD | In vivo - 16 weeks - 0.24% FA or 0.71% OZ containing diet | In vivo - Decreased body weight in both groups - Decreased blood TG, TC, LDL-C, and increased HDL-C in both groups - Decreased fasting blood glucose and HOMA-IR in both groups - Decreased glucose AUC in FA treated group - Increased AMPK and Akt phosphorylation in liver tissues in both groups | |
| In vitro - HepG2 cell line - FFA induced lipid/glucose metabolic dysfunction cell model | In vitro - FA or OZ 100 μM | In vitro - Decreased TG accumulation in both groups - Decreased MDA level in FA treated group - Decreased AMPK phosphorylation and increased Akt phosphorylation in both groups - Decreased DGAT1 expression in both groups | [48] | ||
| FA and OZ | Rice bran | In vivo - SD rats (weighing 350–360 g) with HFFD | In vivo - 13 weeks - 0.05% FA or 0.16% OZ containing diet | In vivo - Decreased body weight and fat index in both groups - Decreased blood TC, TG, LDL-C, and FFA in both groups - Decreased hepatic TC in both groups - Decreased glucose, insulin, glucose AUC, and HOMA-IR in both groups - Increased TAC and decreased MDA in both groups - Decreased CRP and IL-6 and increased adiponectin in OZ treated group - Decreased TNF-α in FA treated group | [18] |
| In vitro - HepG2 cell line | In vitro - FA and OZ 50 mM | In vitro - Decreased TG accumulation in OZ treated group - Decreased FAS, ACC, SREBP-2, HMGCR expression in both groups - Decreased stearoyl coA desaturase-1 expression in OZ treated group | |||
| Phenolic acid fraction (ethyl acetate fraction, EAE) and FA | Rice bran | In vivo - 8-week-old, C57BL/KsJ-db/db mice | - 17 days - EAE 0.2 g/kg or FA 0.05 g/kg - oral administration | - Decreased glucose and increased insulin - Increased hepatic glycogen synthesis and glucokinase activity - Decreased TC and LDL-C | [45] |
| FA | Whole grain and plant materials | In vivo - STZ-induced diabetic rats (weighing 160–170 g) | - 45 days - FA 10, 40 mg/kg - intragastric intubation | - Decreased blood glucose - Decreased blood FFA, TG, TC, and phospholipids | [49] |
| FA | Rice bran and plant materials | In vivo - SD rats (weighing 220–250 g) fed with HCHF diet and 15% fructose in drinking water | - 6 weeks - FA 30, 60 mg/kg - oral administration | - Decreased fasting blood glucose, glucose AUC, HOMA-IR, TC, TG and increased HDL-C - Decreased SBP, DBP, MAP, HR, HVR and increased HBF - Decreased plasma MDA and p47phox protein expression - Increased nitrate/nitrite levels and eNOS protein expression - Decreased mesenteric arterial wall thickness, media to lumen ratio, cross-sectional area of the media layer, and lumen area | [15] |
| FA | Whole grain oats | In vitro - HUVECs | - FA, FA derivatives (isoferulic acid, hydroferulic acid, ferulic acid 4-O-glucuronide, isoferulic acid 3-O-sulfate, dihydroferulic acid 4-O-glucuronide) 1 μM | - Increased NO and cGMP levels - Decreased superoxide levels - Increased Akt1 and eNOS expression | [38] |
| FA | Plant materials | Ex vivo - 20 to 24-week-old, SHRs | - FA 0.01, 0.1, 1 mmol/ml | - Relaxed phenylephrine-induced contraction - Relaxation partially supressed by removing endothelium or by pretreatment with L-NAME - Decreased TXB2 production - Decreased NADPH-dependent superoxide anion levels - Increased ACh-induced vascular relaxation | [51] |
| FA | Wheat bran and plant materials | In vivo - 6-week-old, SHRs | - single dose - FA 9.5 mg/kg - oral administration | - Decreased SBP - Decreased blood TC, TG, ACE activity | [50] |
| Bioactive Compounds | Food Source | Study Subject or Model | Treatment or Intervention | Results | References |
|---|---|---|---|---|---|
| Flavonoid fraction | |||||
| Flavonoid fraction | Tartary buckwheat | In vivo - Mice fed high fructose in drinking water (weighing 18–22 g) | - 8 weeks - Tartary buckwheat flavonoids 200, 400, and 800 mg/kg/day in drinking water | - Improved insulin sensitivity and glucose tolerance - Reversed and attenuated of insulin action on IRS1 phosphorylation, Akt and PI3K, and GLUT4 translocation in insulin-resistant liver | [53] |
| Flavonoid fraction | Tartary buckwheat | In vivo - 6-week-old, SHRs | - 8 weeks - Tartary buckwheat flavonoids 100 mg/kg/day - oral administration | - Reduced SBP - Increased vasodilator response to insulin and reduced IRS-1 phosphorylation at serine 307 - Attenuated hypertension development by reducing vascular oxidative stress | [54] |
| Rutin | |||||
| Rutin | N/A | In vitro - Insulinoma pancreatic β cells of RIN-m5F rat | - EGCG or buckwheat flavonoid Rutin 0.1, 10 μM | - Stimulated IRS2 metabolic pathway in pancreatic β cells of rat - Activated AMPK pathway to suppress activation of lipogenic enzyme | [55] |
| Rutin | Buckwheat cookies | Human study - RCT, crossover - Female adults - mean age 46 years | - 2 weeks - Common buckwheat (low rutin content, 16.5 mg rutin equivalents/day) vs. Tartary buckwheat (high rutin content, 359.7 mg rutin equivalents/day) cookies | - Reduced total serum cholesterol and HDL-C and improved lung vital capacity - Improved MPO, an indicator of inflammation | [58] |
| Bioactive Compounds | Food Source | Study Subject or Model | Treatment or Intervention | Results | References |
|---|---|---|---|---|---|
| Tocotrienol | |||||
| TRF | N/A | In vivo - 4-month-old, Hypercholesterolemic swine | - 6 weeks - Corn-soybean control diet vs. control diet added with 50 μg of d-P21-tocotrienol, d-P25- tocotrienol, γ-tocotrienol, or TRF | - Reduced serum TG, LDL-C, TC, and apolipoprotein B - Reduced glucose level - Level of insulin greater in treatment group | [67] |
| TRF | RBO | In vivo - Hyperlipidemic albino male rats fed with atherogenic diet (weighing 175–200 g) | - 1 week - TRF derived from rice bran oil with concentrations of 0, 4, 8, 12, 25 or 50 mg TRF/kg bw/day - mouth intubation | - Decreased lipid parameters including TC, LDL-C, and TG in a dose-dependent manner - Attenuated HMG-CoA reductase activity | [68] |
| TRF | RBO, PO | In vivo - 9 to 10-week-old, STZ-induced diabetic rats (weighing 250 g) | - 8 weeks - PO and RBO derived TRF at dose of 200 mg/kg bw/day by gavage | - Lowered blood glucose and HbA1c levels - Decreased protein in urine, serum NO, TBARS, and MDA level and increased SOD and catalase level | [69] |
| TRF | RBO, PO | In vivo - T2D rats (weighing 175–200g) | - 16 weeks - Supplementation with TRF of RBO at a dose of 400 mg/kg bw/day and PO at a dose of 200 mg/kg bw/day by gavage | - Improved glycemic status and lipid parameters by reducing blood glucose, HbA1c, VLDL-C, LDL-C, TC, and TG levels - Ameliorated lipid induced nephropathy by its anti-hyperglycemic, anti-hyperlipidemic, and activities of antioxidant as well as by modulation of TGF-β | [70] |
| TRF | RBO | In vivo - Rats treated with the chemical carcinogen DMBA (weighing 120 g) | - 6 months- 10mg TRF/kg/day - gastric intubation | - Decreased LDL-C and TC levels induced by DMBA compared to normal control levels - Decreased ALP activities and maintained low GST activities in liver | [71] |
| Tocotrienol | Bran | In vivo - 4-week-old, HF diet treated mice | - 8 weeks - HF diet supplemented with or without 10 mg of tocotrienols and 5% bran | - Co-treatment with bran and tocotrienols significantly supressed bodyweight gain in HFD-fed mice | [72] |
| TRF | Rice bran | Human study - RCT, parallel - Hypercholesterolemic adults - males <50 years and females <40 years) | - 35 days - AHA Step-1 diet + TRF of rice bran 25, 50, 100, 200 mg/day | - Dose of 100 mg/day of TRF decreased serum TC, LDL, TG, and apolipoprotein B | [14] |
| Tocotrienol | RBO | Human study - RCT, crossover - T2D with hyperlipidemia - mean age 48.5–52.6 years | - 60 days - TRF capsules (2 capsules, 3 mg TRF/kg bw) vs. placebo (100 mg of TRF-free RBO/kg bw) - Group A: TRF in the first phase and color matched placebo capsules in the second phase - Group B: TRF in both first and second phases | - TRF supplement showed reduction of 23 to 42% in serum lipid | [73] |
| α-tocopherol and torotrienol | |||||
| α-tocopherol and torotrienol | Rice bran | In vivo - 6-week-old, F344/slc male rats | - 21 days - Group 1, normal chow diet; Group 2, WD; Group 3, WD with α-Toc 50 mg/day; Group 4, WD with rice bran tocotrienol 11.1 mg/day; Group 5, WD with α-tocopherol 50 mg/day and rice bran tocotrienol 11.1 mg/day - oral administration | - α-tocopherol attenuated lowering effects of TC and TG rice bran tocotrienol - α-tocopherol alone did not attenuate hyperlipidemic effects - Rice bran tocotrienol-induced gene expression of CPT-1a and Cyp7a1 reduced by α-tocopherol | [78] |
| α-tocopherol and torotrienol | Rice bran | In vivo - C57BL/6 ApoE-deficient mice | - 24 weeks - α-tocopherol, TRF, didesmethyl tocotrienol (d-P25-tocotrienol) with low or high fat diets | - Reduced TC and LDL-C in TRF25 and d-P25-T3 group - Reduced atherosclerotic lesion size | [79] |
| α-tocopherol and torotrienol | Rice bran | In vivo - 6-week-old, KKAy diabetic mice | - 6 weeks - normal diet (DM group), a diet including 0.1% Ricetrienol (RT group), non-diabetic C57BL mice (C group) - 0.1% Ricetrienol (crude lipophilic rice bran extract) contains α-tocopherol, tocotrienol, phytosterol | - Rice bran treatment did not alter bodyweight, lipid levels, or glucose metabolism - Elevation of the level of MDA in plasma significantly attenuated by rice bran - The level of α-tocopherol in plasma of RT group was significantly higher compared to that in DM group | [80] |
| Bioactive Compounds | Food Source | Study Subject or Model | Treatment or Intervention | Results | References |
|---|---|---|---|---|---|
| β-glucan | |||||
| β-glucan | Whole grain oats | Human study - RCT, crossover - Mildly hypercholesterolemic subjects - 18–65 years | - 4 weeks - β-glucan 5 g | - Decreased TC, LDL-C, TC/HDL-C ratio - Decreased lathosterol and campesterol | [94] |
| β-glucan | Oat gum (80% β-glucan) | Human study - RCT, crossover - Hypercholesterolemic subjects | - 4 weeks - oat gum (2.9 g β-glucan) | - Decreased TC and LDL-C | [93] |
| β-glucan | Whole grain oats and barley | In vitro - Rat tissue (small intestine) | - 0.5% high-purity (HP)/high-viscosity (HV) barley, HP/low-viscosity (LV) barley, HP/HV oat, HP/LV oat, medium-purity (MP)/HV barley | - Decreased intestinal uptake of long chain fatty acid - Decreased expression of ACC, FAS, i-FABP, and FATP4 | [13] |
| β-glucan | Whole grain oats | Human study - RCT, parallel - Hypercholesterolemic subjects - 18–70 years | - 3 weeks - β-glucan 5 mg | - Decreased TC - Decreased postprandial glucose and insulin | [92] |
| β-glucan | Whole grain oats | In vivo - SD rats (weighing 150–170 g) | - 4 weeks - β-glucan 312.5 mg/kg - oral administration | - Decreased bodyweight gain - Decreased fasting blood glucose - Increased ISI and NEFA - Increased portion of Bifidobacterium and Lactobacillus in colon contents | [96] |
| β-glucan | Whole grain oats | In vivo - 4-week-old, HSHF diet and STZ-induced diabetic mice | - 6 weeks - β-glucan 2000, 1200, or 800 mg/kg - oral administration | - Decreased fasting glucose and fructosamine - Increased fasting insulin and decreased IAI - Decreased activity of disaccharidase (sucrose, lactase, maltase) in small intestine mucosa | [95] |
| β-glucan | Whole grain oat | Human study - RCT, parallel - Subjects with elevated blood pressure - ≥40 years | - 12 weeks - Foods containing oat β-glucan (7.7 g/serving) | - Decreased SBP in subjects with BMI above the median (31.5 kg/m2) - Decreased glucose AUC, fasting insulin, peak insulin, and insulin AUC | [97] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chung, S.; Hwang, J.-T.; Park, S.-H. Physiological Effects of Bioactive Compounds Derived from Whole Grains on Cardiovascular and Metabolic Diseases. Appl. Sci. 2022, 12, 658. https://doi.org/10.3390/app12020658
Chung S, Hwang J-T, Park S-H. Physiological Effects of Bioactive Compounds Derived from Whole Grains on Cardiovascular and Metabolic Diseases. Applied Sciences. 2022; 12(2):658. https://doi.org/10.3390/app12020658
Chicago/Turabian StyleChung, Sangwon, Jin-Taek Hwang, and Soo-Hyun Park. 2022. "Physiological Effects of Bioactive Compounds Derived from Whole Grains on Cardiovascular and Metabolic Diseases" Applied Sciences 12, no. 2: 658. https://doi.org/10.3390/app12020658
APA StyleChung, S., Hwang, J.-T., & Park, S.-H. (2022). Physiological Effects of Bioactive Compounds Derived from Whole Grains on Cardiovascular and Metabolic Diseases. Applied Sciences, 12(2), 658. https://doi.org/10.3390/app12020658






