The Effect of Lactic Acid Fermentation on Extraction of Phenolics and Flavonoids from Sage Leaves
Abstract
Featured Application
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Materials
2.2. Sample Preparation
2.3. Extractive Fermentation
2.4. Planar Chromatography
2.5. Post Chromatographic Derivatization
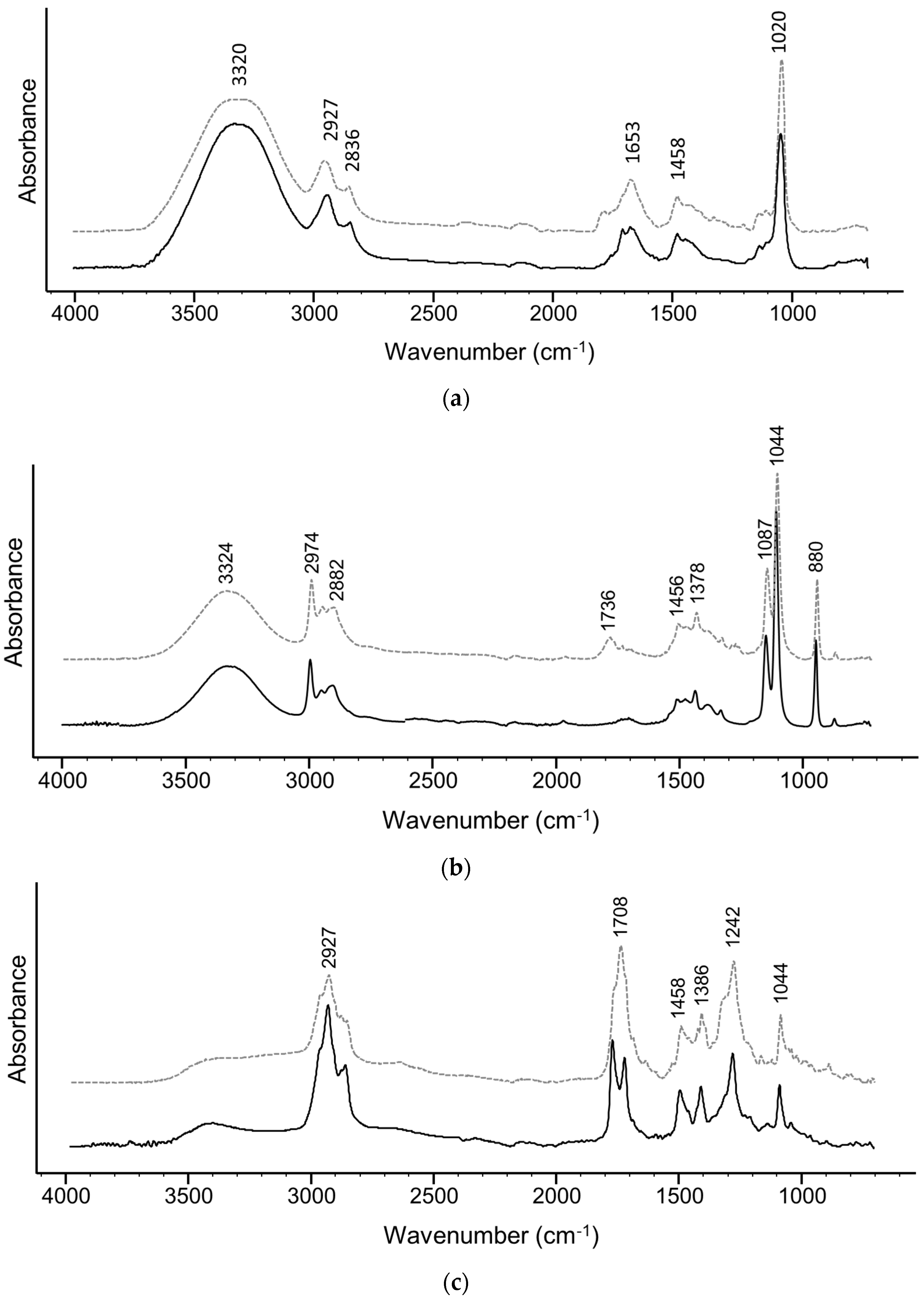
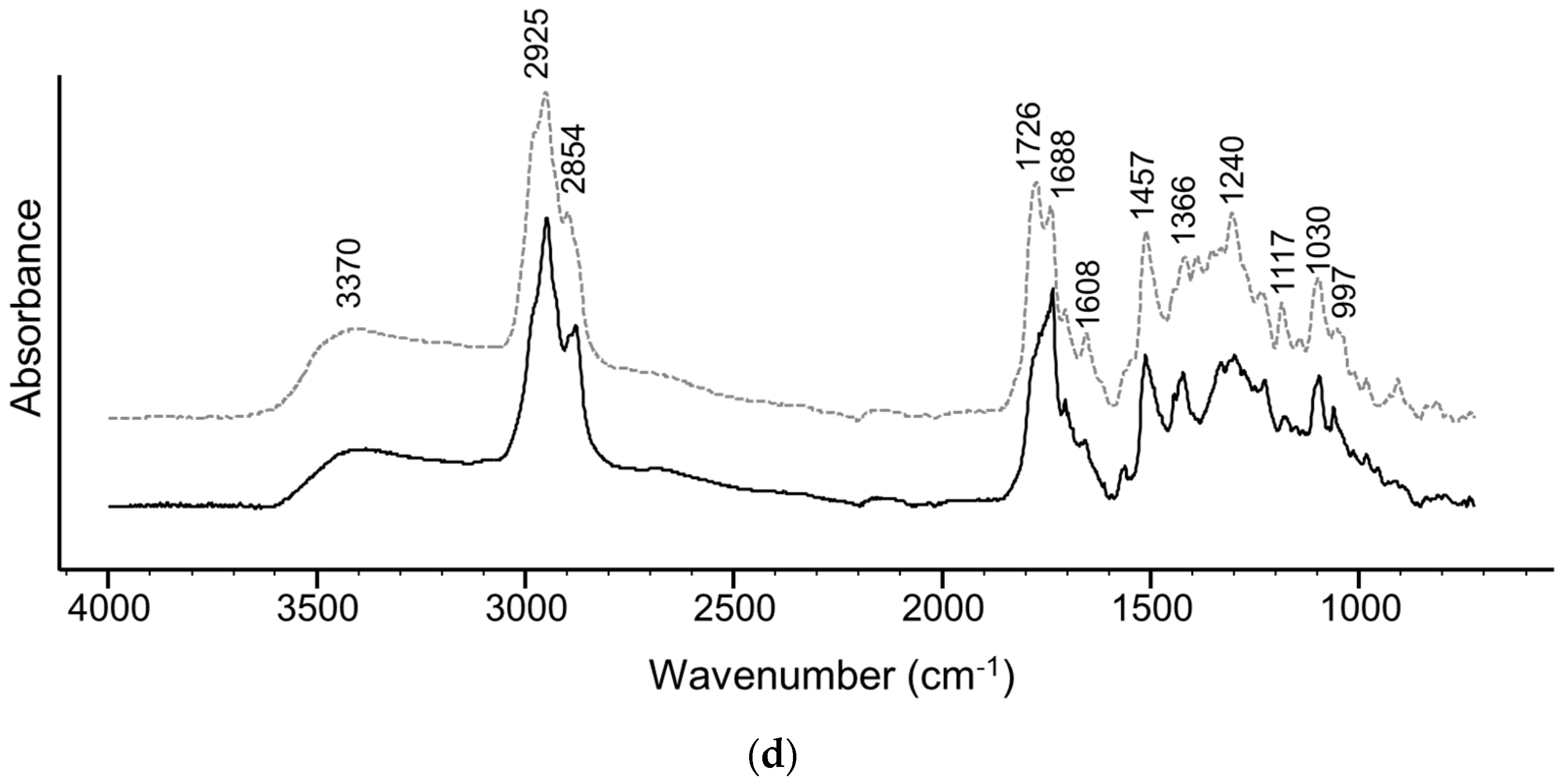
2.6. FTIR-ATR Spectra
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Frankel, E.N.; Huang, S.-W.; Aeschbach, R.; Prior, E. Antioxidant activity of a rosemary extract and its constituents, carnosic acid, carnosol, and rosmarinic acid, in bulk oil and oil-in-water emulsion. J. Agric. Food Chem. 1996, 44, 131–135. [Google Scholar] [CrossRef]
- Suhaj, M. Spice antioxidants isolation and their antiradical activity: A review. J. Food Compos. Anal. 2006, 19, 531–537. [Google Scholar] [CrossRef]
- Hamidpour, M.; Hamidpour, R.; Hamidpour, S.; Shahlari, M. Chemistry, pharmacology, and medicinal property of sage (Salvia) to prevent and cure illnesses such as obesity, diabetes, depression, dementia, lupus, autism, heart disease, and cancer. J. Tradit. Complement. Med. 2014, 4, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Farhat, M.B.; Landoulsi, A.; Chaouch-Hamada, R.; Sotomayor, J.A.; Jordán, M.J. Characterization and quantification of phenolic compounds and antioxidant properties of Salvia species growing in different habitats. Ind. Prods. Crop. 2013, 49, 904–914. [Google Scholar] [CrossRef]
- Glisic, S.; Ivanovic, J.; Ristic, M.; Skala, D. Extraction of sage (Salvia officinalis L.) by supercritical CO2: Kinetic data, chemical composition and selectivity of diterpenes. J. Supercrit. Fluids 2010, 52, 62–70. [Google Scholar] [CrossRef]
- Kontogianni, V.G.; Tomic, G.; Nikolic, I.; Nerantzaki, A.A.; Sayyad, N.; Stosic-Grujicic, S.; Stojanovic, I.; Gerothanassis, I.P.; Tzakos, A.G. Phytochemical profile of Rosmarinus officinalis and Salvia officinalis extracts and correlation to their antioxidant and anti-proliferative activity. Food Chem. 2013, 136, 120–129. [Google Scholar] [CrossRef] [PubMed]
- Pearn, J. On “officinalis” the names of plants as one enduring history of therapeutic medicine. Vesalius Acta Int. Hist. Med. 2010, Suppl, 24–28. [Google Scholar]
- Adams, J.D.; Garcia, C. The Advantages of Traditional Chumash Healing. Evid. Based Complement. Alternat. Med. 2005, 2, 19–23. [Google Scholar] [CrossRef]
- Agatonovic-Kustrin, S.; Morton, D.W. Thin-Layer Chromatography: Fingerprint Analysis of Plant Materials. In Reference Module in Chemistry, Molecular Sciences and Chemical Engineering, Encyclopedia of Analytical Science, 3rd ed.; Worsfold, P., Townshend, A., Poole, C., Miró, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; Volume 10, pp. 43–49. [Google Scholar]
- Wang, L.; Lin, X.; Zhang, J.; Zhang, W.; Hu, X.; Li, W.; Li, C.; Liu, S. Extraction methods for the releasing of bound phenolics from Rubus idaeus L. leaves and seeds. Ind. Crops Prod. 2019, 135, 1–9. [Google Scholar] [CrossRef]
- Singh, S.; Cheng, G.; Sathitsuksanoh, N.; Wu, D.; Varanasi, P.; George, A.; Balan, V.; Gao, X.; Kumar, R.; Dale, B.E.; et al. Comparison of different biomass pretreatment techniques and their impact on chemistry and structure. Front. Energy Res. 2015, 2, 62. [Google Scholar] [CrossRef]
- Dreisbach, R.R.; Martin, R.A. Physical data on some organic compounds. Ind. Eng. Chem. 1949, 41, 2875–2878. [Google Scholar] [CrossRef]
- Quispe-Condori, S.; Foglio, M.A.; Rosa, P.T.V.; Meireles, M.A.A. Obtaining β-caryophyllene from Cordia verbenacea de Candolle by supercritical fluid extraction. J. Supercrit. Fluids 2008, 46, 27–32. [Google Scholar] [CrossRef]
- Hayouni, E.A.; Abedrabba, M.; Bouix, M.; Hamdi, M. The effects of solvents and extraction method on the phenolic contents and biological activities in vitro of Tunisian Quercus coccifera L. and Juniperus phoenica L. fruit extracts. Food Chem. 2007, 105, 1126–1134. [Google Scholar] [CrossRef]
- Ashurst, J.V.; Nappe, T.M. Methanol toxicity. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Fu, X.; Belwal, T.; Cravotto, G.; Luo, Z. Sono-physical and sono-chemical effects of ultrasound: Primary applications in extraction and freezing operations and influence on food components. Ultrason. Sonochem. 2020, 60, 104726. [Google Scholar] [CrossRef] [PubMed]
- Cunha, S.C.; Fernandes, J.O. Extraction techniques with deep eutectic solvents. Trends Anal. Chem. 2018, 105, 225–239. [Google Scholar] [CrossRef]
- Grasel, F.D.S.; Ferrão, M.F.; Wolf, C.R. Development of methodology for identification the nature of the polyphenolic extracts by FTIR associated with multivariate analysis. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2016, 153, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Aleksovski, S.A.; Sovova, H. Supercritical CO2 extraction of Salvia officinalis L. J. Supercrit. Fluids 2007, 40, 239–245. [Google Scholar] [CrossRef]
- Silva, S.D.; Feliciano, R.P.; Boas, L.V.; Bronze, M.R. Application of FTIR-ATR to Moscatel dessert wines for prediction of total phenolic and flavonoid contents and antioxidant capacity. Food Chem. 2014, 150, 489–493. [Google Scholar] [CrossRef] [PubMed]
- Harbone, J.B. The Flavonoids: Advances in Research Since 1986; Chapman & Hall: New York, NY, USA, 1994; p. 913. [Google Scholar]
- Zhuang, J.; Li, M.; Pu, Y.; Ragauskas, A.J.; Yoo, C.G. Observation of potential contaminants in processed biomass using Fourier transform infrared spectroscopy. Appl. Sci. 2020, 10, 4345. [Google Scholar] [CrossRef]
- Raspolli Galletti, A.M.; D’Alessio, A.; Licursi, D.; Antonetti, C.; Valentini, G.; Galia, A.; Nassi o Di Nasso, N. Midinfrared FT-IR as a tool for monitoring herbaceous biomass composition and its conversion to furfural. J. Spectrosc. 2015, 2015, 719042. [Google Scholar] [CrossRef]
- Huguet, A.; del Carmen Recio, M.; Máñez, S.; Giner, R.; Rıos, J. Effect of triterpenoids on the inflammation induced by protein kinase C activators, neuronally acting irritants and other agents. Eur. J. Pharmacol. 2000, 410, 69–81. [Google Scholar] [CrossRef]
- Mitaine-Offer, A.-C.; Hornebeck, W.; Sauvain, M.; Zèches-Hanrot, M. Triterpenes and phytosterols as human leucocyte elastase inhibitors. Planta Med. 2002, 68, 930–932. [Google Scholar] [CrossRef] [PubMed]
- Sohn, K.-H.; Lee, H.-Y.; Chung, H.-Y.; Young, H.-S.; Yi, S.-Y.; Kim, K.-W. Anti-angiogenic activity of triterpene acids. Cancer Lett. 1995, 94, 213–218. [Google Scholar] [CrossRef]
- Agatonovic-Kustrin, S.; Gegechkori, V.; Morton, D.W.; Tucci, J.; Mohammed, E.U.R.; Ku, H. The bioprofiling of antibacterials in olive leaf extracts via thin layer chromatography-effect directed analysis (TLC-EDA). J. Pharm. Biomed. Anal. 2022, 219, 114916. [Google Scholar] [CrossRef]
- Boerjan, W.; Ralph, J.; Baucher, M. Lignin bioxynthesis. Annu. Rev. Plant Biol. 2003, 54, 519–546. [Google Scholar] [CrossRef] [PubMed]
- Lima, R.B.; Salvador, V.H.; dos Santos, W.D.; Bubna, G.A.; Finger-Teixeira, A.; Soares, A.R.; Marchiosi, R.; Ferrarese, M.d.L.L.; Ferrarese-Filho, O. Enhanced lignin monomer production caused by cinnamic acid and its hydroxylated derivatives inhibits soybean root growth. PLoS ONE 2013, 8, e80542. [Google Scholar] [CrossRef]
- Bors, W.; Christa, M.; Stettmaier, K.; Yinrong, L.; Foo, L.Y. Antioxidant mechanisms of polyphenolic caffeic acid oligomers, constituents of Salvia officinalis. Biol. Res. 2004, 37, 301–311. [Google Scholar] [CrossRef] [PubMed]
- Socrates, G. Infrared and Raman Characteristic Group Frequencies: Tables and Charts; John Wiley & Sons: Chichester, UK, 2004. [Google Scholar]
- Adler, E.; Gierer, J. The alkylation of lignin with alcoholic hydrochloric acid. Acta Chem. Scand. 1955, 9, 84–93. [Google Scholar] [CrossRef]
- Durie, R.A.; Lynch, B.M.; Sternhell, S. Comparative studies of brown coal and lignin. I. Infra-red spectra. Aust. J. Chem. 1960, 13, 156–168. [Google Scholar] [CrossRef]
- Bykov, I. Characterization of Natural and Technical Lignins using FTIR Spectroscopy. Master’s Thesis, Lulea University of Technology, Lulea, Sweden, 2008. [Google Scholar]
- Hergert, H.L. Infrared spectra of lignin and related compounds. II. Conifer lignin and model compounds1, 2. J. Org. Chem. 1960, 25, 405–413. [Google Scholar] [CrossRef]
- Stahl, E. Thin-Layer Chromatography A Laboratory Handbook, 2nd ed.; Springer: Berlin/Heidelberg, Germany, 1969. [Google Scholar]
- Pasto, D.J.; Johnson, C.R. Laboratory Text for Organic Chemistry. A Source Book of Chemical and Physical Techniques; Prentice-Hall, Inc.: Englewood Cliffs, NJ, USA, 1979. [Google Scholar]
- Rosa, L.A.; Moreno-Escamilla, J.O.; Rodrigo-Gracia, J.; Haard, N.F. Phenolic compounds. In Postharvest Physiology and Biochemistry of Fruits and Vegetables, Phenolic Compounds; Yahia, E., Carrillo-Lopez, A., Eds.; Woodhead Publishing: Duxford, UK, 2018; pp. 253–271. [Google Scholar]
- Bleiziffer, R.; Mesaros, C.; Suvar, S.; Podea, P.; Iordache, A.; Yudin, F.-D.; Culea, M. Comparative characterization of basil, mint and sage extracts. Stud. Univ. Babeş Bolyai Ser. Chem. 2017, 62, 373–385. [Google Scholar] [CrossRef]
- Myha, M.; Koshovyi, O.; Gamulya, O.; Ilina, T.; Borodina, N.; Vlasova, I. Phytochemical study of Salvia grandiflora and Salvia officinalis leaves for establishing prospects for use in medical and pharmaceutical practice. ScienceRise Pharm. Sci. 2020, 1, 23–28. [Google Scholar] [CrossRef]
- Nguyen, C.-H.; Augis, L.; Fourmentin, S.; Barratt, G.; Legrand, F.-X. Deep eutectic solvents for innovative pharmaceutical formulations. In Deep Eutectic Solvents for Medicine, Gas Solubilization and Extraction of Natural Substances; Fourmentin, S., Gomes, M.C., Eric Lichtfouse, E., Eds.; Springer: Cham, Switzerland, 2021; pp. 41–102. [Google Scholar]


| Plant Species | Plant Material | Solvent | Yield 1/% |
|---|---|---|---|
| Salvia officinalis | Ground dried leaves | Methanol | 15.33 |
| Ground dried leaves | Ethanol | 7.33 | |
| Ground dried leaves | Ethyl acetate | 6 | |
| Fermented ground dried leaves | Ethyl acetate | 3.17 | |
| Salvia apiana | Ground dried leaves | Methanol | 14.47 |
| Ground dried leaves | Ethanol | 10.32 | |
| Ground dried leaves | Ethyl acetate | 9.65 | |
| Fermented ground dried leaves | Ethyl acetate | 5.64 |
| Linear Regression | RSD (%) | LOD (µg) | LOQ (µg) | Linearity (Analytical Range) | |
|---|---|---|---|---|---|
| DPPH• | y = 131,547x + 72,883 R2 = 0.95 | 3.20–5.09 | 0.11 | 0.35 | 0.2–6.0 |
| y = −16,690x2 + 217,442x + 27,344 R2 = 0.985 | |||||
| FeCl3 | y = 21,093x + 15,402 R2 = 0.96 | 2.42–5.41 | 0.22 | 0.74 | 0.5–10 |
| y = 1252x2 + 32,564x − 1086.2 R2 = 0.99 | |||||
| AlCl3 | y = 84,307x + 37,984 R2 = 0.97 | 2.06–2.35 | 0.35 | 1.19 | 1.0–7.0 |
| y = 713.27x2 + 13,883x + 30,954 R2 = 0.99 | |||||
| ASA | y = 554.5x + 13,011 R2 = 0.95 | 3.28–6.86 | 0.44 | 1.48 | 0.5–6.0 |
| y = 766.54x2 +10,599x + 7959.9 R2 = 0.98 |
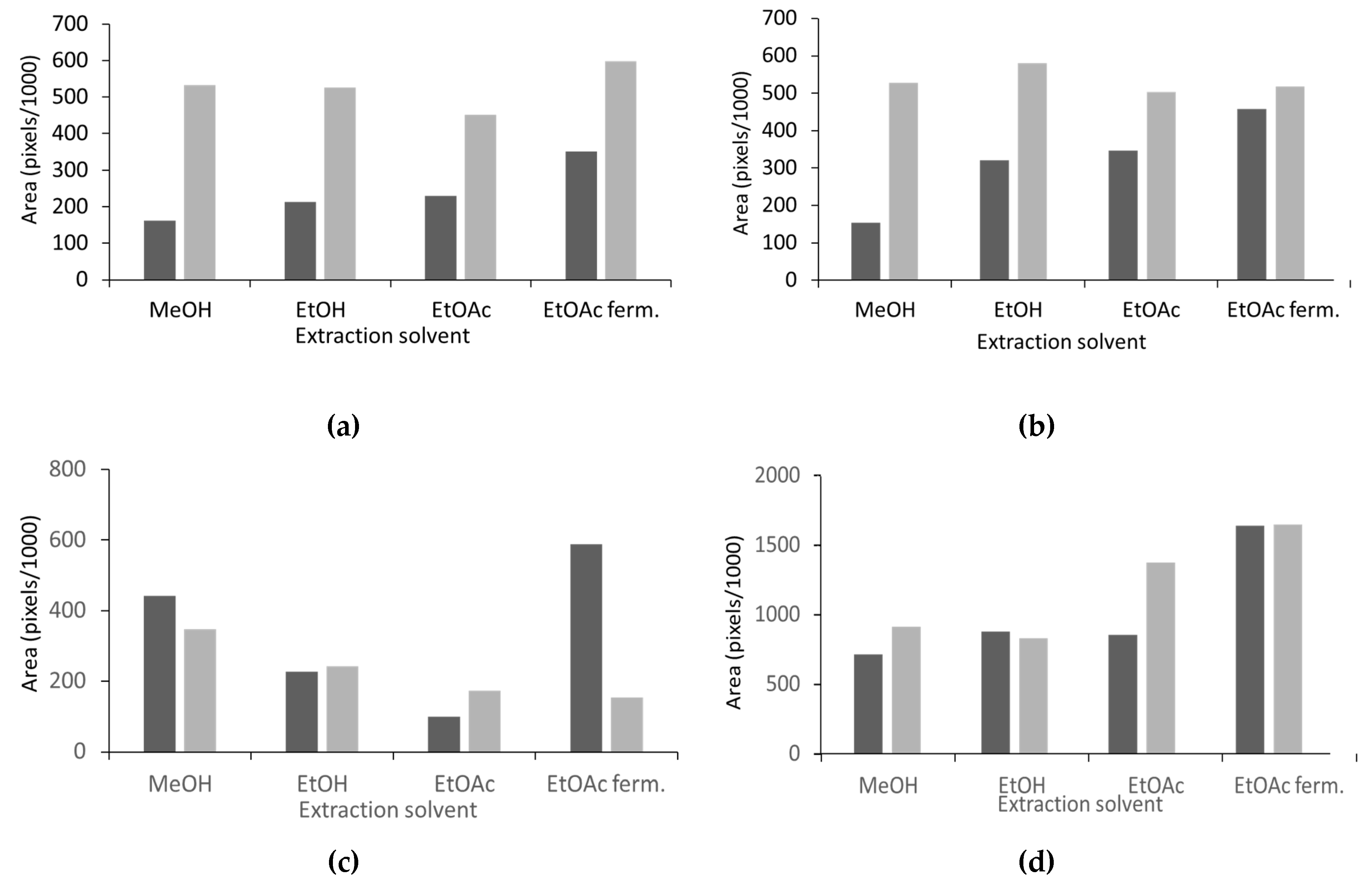
| Antioxidant Activity (DPPH• Assay) | Phenolics (FeCl3) | Total Flavonoids (AlCl3) | Natural Products (ASA) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Area (Pixels) | GAE /20 μL | Area (Pixels) | GAE /20 μL | Area (Pixels) | RE /20 μL | Area (Pixels) | SE /20 μL | ||
| S. officinalis | MeOH | 162,764 | 0.68 | 153,566 | 6.55 | 442,707 | 48.01 | 715,681 | 126.72 |
| EtOH | 213,722 | 1.07 | 321,111 | 14.49 | 228,069 | 22.55 | 878,126 | 156.01 | |
| EtOAc | 230,009 | 1.19 | 347,442 | 15.74 | 100,748 | 7.44 | 856,353 | 152.08 | |
| EtOAc f. | 351,167 | 2.12 | 458,563 | 21.01 | 588,739 | 65.33 | 1,640,941 | 293.57 | |
| S. apiana | MeOH | 532,673 | 3.50 | 528,201 | 24.31 | 347,686 | 36.74 | 913,075 | 162.31 |
| EtOH | 526,481 | 3.45 | 580,221 | 26.78 | 242,396 | 24.25 | 829,756 | 147.29 | |
| EtOAc | 451,561 | 2.88 | 503,363 | 23.13 | 173,947 | 16.13 | 1,373,288 | 245.31 | |
| EtOAc f. | 598,410 | 3.99 | 518,137 | 23.83 | 154,577 | 13.83 | 1,646,589 | 294.59 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Agatonovic-Kustrin, S.; Gegechkori, V.; Kustrin, E.; Morton, D.W. The Effect of Lactic Acid Fermentation on Extraction of Phenolics and Flavonoids from Sage Leaves. Appl. Sci. 2022, 12, 9959. https://doi.org/10.3390/app12199959
Agatonovic-Kustrin S, Gegechkori V, Kustrin E, Morton DW. The Effect of Lactic Acid Fermentation on Extraction of Phenolics and Flavonoids from Sage Leaves. Applied Sciences. 2022; 12(19):9959. https://doi.org/10.3390/app12199959
Chicago/Turabian StyleAgatonovic-Kustrin, Snezana, Vladimir Gegechkori, Ella Kustrin, and David W. Morton. 2022. "The Effect of Lactic Acid Fermentation on Extraction of Phenolics and Flavonoids from Sage Leaves" Applied Sciences 12, no. 19: 9959. https://doi.org/10.3390/app12199959
APA StyleAgatonovic-Kustrin, S., Gegechkori, V., Kustrin, E., & Morton, D. W. (2022). The Effect of Lactic Acid Fermentation on Extraction of Phenolics and Flavonoids from Sage Leaves. Applied Sciences, 12(19), 9959. https://doi.org/10.3390/app12199959







