Honey as an Adjuvant in the Treatment of COVID-19 Infection: A Review
Abstract
1. Introduction
2. Methodology
3. SARS-CoV-2 Overview and Vaccines Mechanism of Actions
3.1. RNA-Dependent Inhibitors
3.2. Protease Inhibitors
3.3. Prevention of Cell Membrane/Virus Fusion
3.4. Immunomodulators
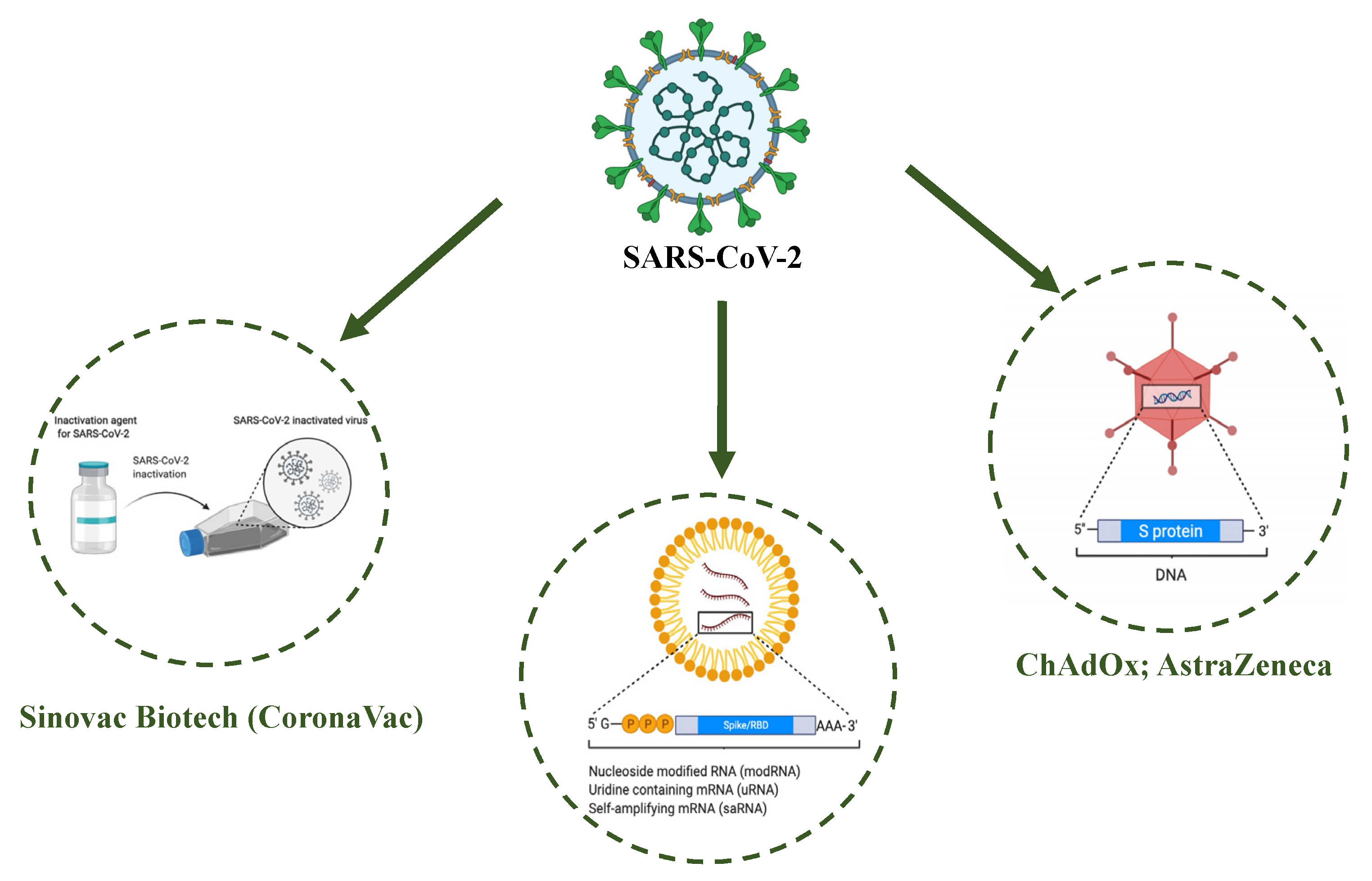
3.5. Vaccines
3.5.1. Inactivated and Protein Subunit Vaccines
3.5.2. Viral Vector Vaccines
3.5.3. mRNA Vaccines
4. Honey as a Co-Adjuvant of SARS-CoV-2 Infection Treatment
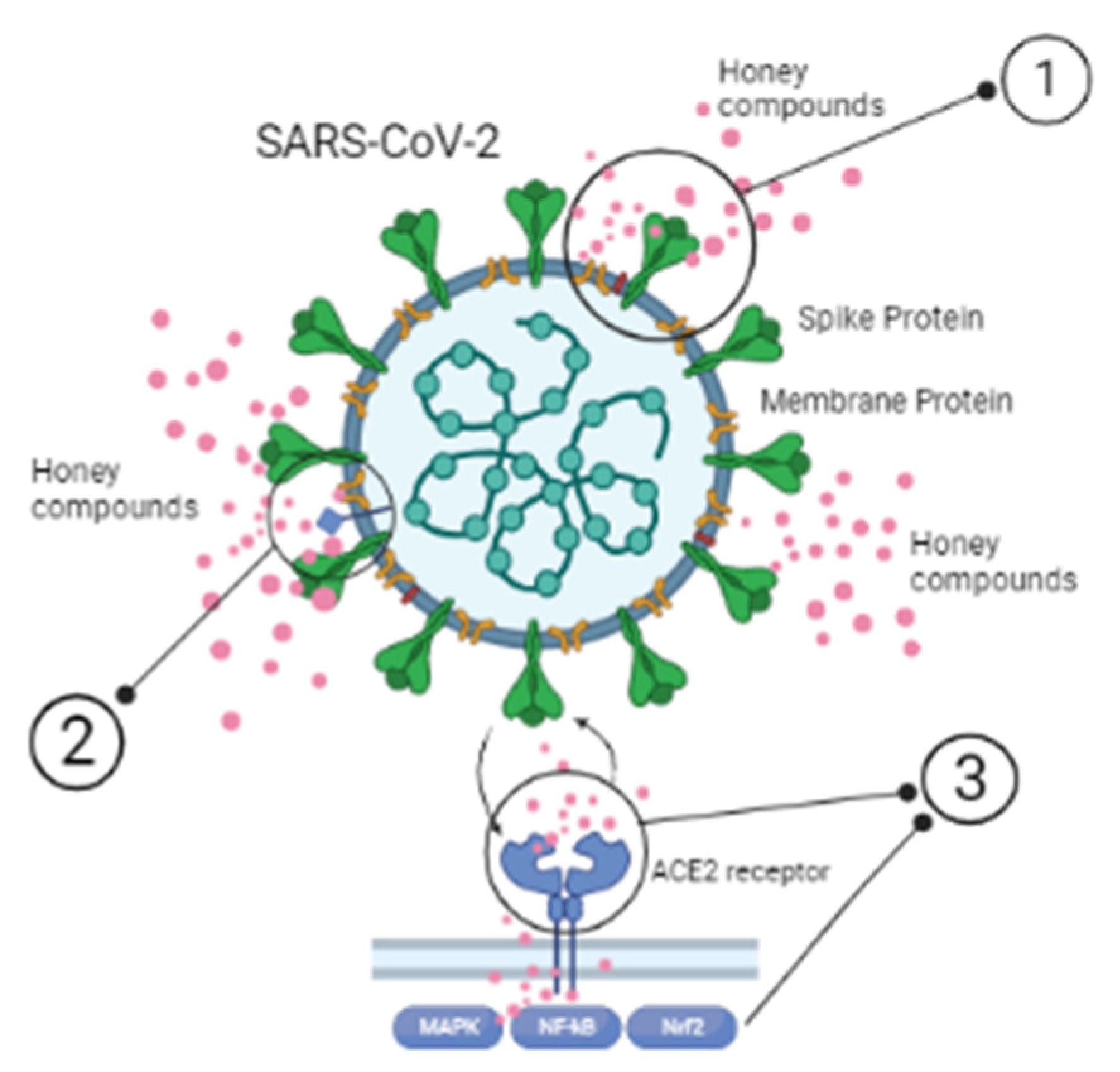
4.1. Direct Virucidal Properties
4.2. Regulation/Boosting of Host Immune Signaling Pathways
4.3. Cure and/or Improve Comorbid Conditions
5. Conclusions and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Oryan, A.; Alemzadeh, E.; Moshiri, A. Biological properties and therapeutic activities of honey in wound healing: A narrative review and meta-analysis. J. Tissue Viability 2016, 25, 98–118. [Google Scholar] [CrossRef]
- Samarghandian, S.; Farkhondeh, T.; Samini, F. Honey and health: A review of recent clinical research. Pharmacogn. Res. 2017, 9, 121–127. [Google Scholar]
- Soares, S.; Amaral, J.S.; Oliveira, M.B.P.P.; Mafra, I. A comprehensive review on the main honey authentication issues: Production and origin. Compr. Rev. Food Sci. 2017, 16, 1072–1100. [Google Scholar] [CrossRef]
- Burlando, B.; Cornara, L. Honey in dermatology and skin care: A review. J. Cosmet. Dermatol. 2013, 12, 306–313. [Google Scholar] [CrossRef]
- Saranraj, P.; Sivasakthi, S.; Feliciano, G.D. Pharmacology of honey: A review. Adv. Biol. Res. 2016, 10, 271–289. [Google Scholar]
- Ajibola, A.; Chamunorwa, J.P.; Erlwanger, K.H. Nutraceutical values of natural honey and its contribution to human health and wealth. Nutr. Metab. 2012, 9, 61. [Google Scholar] [CrossRef]
- Tomás, A.; Falcão, S.I.; Russo-Almeida, P.; Vilas-Boas, M. Potentialities of beebread as a food supplement and source of nutraceuticals: Botanical origin, nutritional composition and antioxidant activity. J. Apic. Res. 2017, 56, 219–230. [Google Scholar] [CrossRef]
- Codex Alimentarius. Standard for Honey, CXS 12-19811 Adopted in 1981. Revised in 1987, 2001. Amended in 2019. Available online: https://www.fao.org (accessed on 28 June 2022).
- Anklam, E. A review of the analytical methods to determine the geographical and botanical origin of honey. Food Chem. 1998, 63, 549–562. [Google Scholar] [CrossRef]
- Schievano, E.; Morelato, E.; Facchin, C.; Mammi, S. Characterization of markers of botanical origin and other compounds extracted from unifloral honeys. J. Agric. Food Chem. 2013, 61, 1747–1755. [Google Scholar] [CrossRef]
- Missio da Silva, P.; Gonzaga, L.V.; Biluca, F.C.; Schulz, M.; Vitali, L.; Micke, G.A.; Oliveira Costa, A.C.; Fett, R. Stability of brazilian apis mellifera l. Honey during prolonged storage: Physicochemical parameters and bioactive compounds. LWT–Food Sci. Technol. 2020, 129, 109521. [Google Scholar] [CrossRef]
- Sousa, J.M.B.d.; Souza, E.L.d.; Marques, G.; Benassi, M.d.T.; Gullón, B.; Pintado, M.M.; Magnani, M. Sugar profile, physicochemical and sensory aspects of monofloral honeys produced by different stingless bee species in brazilian semi-arid region. LWT–Food Sci. Technol. 2016, 65, 645–651. [Google Scholar] [CrossRef]
- Seraglio, S.K.T.; Bergamo, G.; Brugnerotto, P.; Gonzaga, L.V.; Fett, R.; Costa, A.C.O. Aliphatic organic acids as promising authenticity markers of bracatinga honeydew honey. Food Chem. 2021, 343, 128449. [Google Scholar] [CrossRef]
- Sun, Z.; Zhao, L.; Cheng, N.; Xue, X.; Wu, L.; Zheng, J.; Cao, W. Identification of botanical origin of chinese unifloral honeys by free amino acid profiles and chemometric methods. J. Pharm. Anal. 2017, 7, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Suto, M.; Kawashima, H.; Nakamura, Y. Determination of organic acids in honey by liquid chromatography with tandem mass spectrometry. Food Anal. Methods 2020, 13, 2249–2257. [Google Scholar] [CrossRef]
- Tan, T.Y.; Basheer, C.; Low, K.M.; Lee, H.K. Electro membrane extraction of organic acids in undiluted honey with ion chromatographic analysis. Microchem. J. 2018, 143, 234–242. [Google Scholar] [CrossRef]
- Zhao, H.; Cheng, N.; Zhang, Y.; Sun, Z.; Zhou, W.; Wang, Y.; Cao, W. The effects of different thermal treatments on amino acid contents and chemometric-based identification of overheated honey. LWT–Food Sci. Technol. 2018, 96, 133–139. [Google Scholar] [CrossRef]
- Villacrés-Granda, I.; Coello, D.; Proaño, A.; Ballesteros, I.; Roubik, D.W.; Jijón, G.; Granda-Albuja, G.; Granda-Albuja, S.; Abreu-Naranjo, R.; Maza, F.; et al. Honey quality parameters, chemical composition and antimicrobial activity in twelve ecuadorian stingless bees (apidae: Apinae: Meliponini) tested against multiresistant human pathogens. LWT–Food Sci. Technol. 2021, 140, 110737. [Google Scholar] [CrossRef]
- El Sohaimy, S.A.; Masry, S.H.D.; Shehata, M.G. Physicochemical characteristics of honey from different origins. Ann. Agric. Sci. 2015, 60, 279–287. [Google Scholar] [CrossRef]
- Daniele, G.; Maitre, D.; Casabianca, H. Identification, quantification and carbon stable isotopes determinations of organic acids in monofloral honeys. A powerful tool for botanical and authenticity control. Rapid Commun. Mass Spectrom. 2012, 26, 1993–1998. [Google Scholar] [CrossRef] [PubMed]
- Julika, W.N.; Ajit, A.; Ismail, N.; Aqilah, N.; Naila, A.; Sulaiman, A.Z. Sugar profile and enzymatic analysis of stingless bee honey collected from local market in Malaysia. In IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2020; Volume 736, p. 062001. [Google Scholar]
- Starowicz, M.; Hanus, P.; Lamparski, G.; Sawicki, T. Characterizing the volatile and sensory profiles, and sugar content of beeswax, beebread, bee pollen, and honey. Molecules 2021, 26, 3410. [Google Scholar] [CrossRef] [PubMed]
- Martins, R.C.; Lopes, V.V.; Valentão, P.; Carvalho, J.C.M.F.; Isabel, P.; Amaral, M.T.; Batista, M.T.; Andrade, P.B.; Silva, B.M. Relevant principal component analysis applied to the characterisation of portuguese heather honey. Nat. Prod. Res. 2008, 22, 1560–1582. [Google Scholar] [CrossRef]
- Anjos, O.; Campos, M.G.; Ruiz, P.C.; Antunes, P. Application of ftir-atr spectroscopy to the quantification of sugar in honey. Food Chem. 2015, 169, 218–223. [Google Scholar] [CrossRef]
- Geană, E.-I.; Ciucure, C.T.; Costinel, D.; Ionete, R.E. Evaluation of honey in terms of quality and authenticity based on the general physicochemical pattern, major sugar composition and δ13c signature. Food Control. 2020, 109, 106919. [Google Scholar] [CrossRef]
- Escuredo, O.; Dobre, I.; Fernández-González, M.; Seijo, M.C. Contribution of botanical origin and sugar composition of honeys on the crystallization phenomenon. Food Chem. 2014, 149, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Pérez, R.A.; Iglesias, M.T.; Pueyo, E.; González, M.; de Lorenzo, C. Amino acid composition and antioxidant capacity of spanish honeys. J. Agric. Food Chem. 2007, 55, 360–365. [Google Scholar] [CrossRef]
- del Campo, G.; Zuriarrain, J.; Zuriarrain, A.; Berregi, I. Quantitative determination of carboxylic acids, amino acids, carbohydrates, ethanol and hydroxymethylfurfural in honey by 1H NMR. Food Chem. 2016, 196, 1031–1039. [Google Scholar] [CrossRef]
- Łozowicka, B.; Kaczyński, P.; Iwaniuk, P. Analysis of 22 free amino acids in honey from eastern europe and central asia using LC-MS/MS technique without derivatization step. J. Food Compos. Anal. 2021, 98, 103837. [Google Scholar] [CrossRef]
- Boussaid, A.; Chouaibi, M.; Rezig, L.; Hellal, R.; Donsì, F.; Ferrari, G.; Hamdi, S. Physicochemical and bioactive properties of six honey samples from various floral origins from tunisia. Arab. J. Chem. 2018, 11, 265–274. [Google Scholar] [CrossRef]
- Mouhoubi-Tafinine, Z.; Ouchemoukh, S.; Tamendjari, A. Antioxydant activity of some algerian honey and propolis. Ind. Crops Prod. 2016, 88, 85–90. [Google Scholar] [CrossRef]
- Otmani, A.; Amessis-Ouchemoukh, N.; Birinci, C.; Yahiaoui, S.; Kolayli, S.; Rodríguez-Flores, M.S.; Escuredo, O.; Seijo, M.C.; Ouchemoukh, S. Phenolic compounds and antioxidant and antibacterial activities of algerian honeys. Food Biosci. 2021, 42, 101070. [Google Scholar] [CrossRef]
- Hunter, M.; Ghildyal, R.; D’Cunha, N.M.; Gouws, C.; Georgousopoulou, E.N.; Naumovski, N. The bioactive, antioxidant, antibacterial, and physicochemical properties of a range of commercially available australian honeys. Curr. Res. Nutr. Food Sci. 2021, 4, 532–542. [Google Scholar] [CrossRef]
- Cheung, Y.; Meenu, M.; Yu, X.; Xu, B. Phenolic acids and flavonoids profiles of commercial honey from different floral sources and geographic sources. Int. J. Food Prop. 2019, 22, 290–308. [Google Scholar] [CrossRef]
- Flanjak, I.; Strelec, I.; Kenjerić, D.; Primorac, L. Croatian produced unifloral honey characterized according to the protein and proline content and enzyme activities. J. Apic. Sci. 2016, 60, 39–48. [Google Scholar] [CrossRef]
- Debela, H.; Belay, A. Caffeine, invertase enzyme and triangle test sensory panel used to differentiate coffea arabica and vernonia amygdalina honey. Food Control 2021, 123, 107857. [Google Scholar] [CrossRef]
- Cianciosi, D.; Forbes-Hernandez, T.Y.; Alvarez-Suarez, J.M.; Ansary, J.; Quinzi, D.; Amici, A.; Navarro-Hortal, M.D.; Esteban-Muñoz, A.; Quiles, J.L.; Battino, M.; et al. Anti-inflammatory activities of italian chestnut and eucalyptus honeys on murine raw 264.7 macrophages. J. Funct. Foods 2021, 87, 104752. [Google Scholar] [CrossRef]
- Estevinho, L.M.; Feás, X.; Seijas, J.A.; Pilar Vázquez-Tato, M. Organic honey from trás-os-montes region (portugal): Chemical, palynological, microbiological and bioactive compounds characterization. Food Chem. Toxicol. 2012, 50, 258–264. [Google Scholar] [CrossRef]
- Soares, S.; Pinto, D.; Rodrigues, F.; Alves, R.C.; Oliveira, M. Portuguese honeys from different geographical and botanical origins: A 4-year stability study regarding quality parameters and antioxidant activity. Molecules 2017, 22, 1338. [Google Scholar] [CrossRef]
- Oroian, M.; Ropciuc, S. Honey authentication based on physicochemical parameters and phenolic compounds. Comput. Electron. Agric. 2017, 138, 148–156. [Google Scholar] [CrossRef]
- Guzelmeric, E.; Ciftci, I.; Yuksel, P.I.; Yesilada, E. Importance of chromatographic and spectrophotometric methods in determining authenticity, classification and bioactivity of honey. LWT–Food Sci. Technol. 2020, 132, 109921. [Google Scholar] [CrossRef]
- Conti, M.E.; Finoia, M.G.; Fontana, L.; Mele, G.; Botrè, F.; Iavicoli, I. Characterization of argentine honeys on the basis of their mineral content and some typical quality parameters. Chem. Cent. J. 2014, 8, 44. [Google Scholar] [CrossRef] [PubMed]
- Atanassova, J.; Pavlova, D.; Lazarova, M.; Yurukova, L. Characteristics of honey from serpentine area in the eastern rhodopes mt., bulgaria. Biol. Trace Elem. Res. 2016, 173, 247–258. [Google Scholar] [CrossRef]
- Sager, M.; Maleviti, E. Elemental composition of honeys from greece-possible use as environmental indicators. Nutr. Food Sci. 2014, S8, 2. [Google Scholar]
- Sajid, M.; Yamin, M.; Asad, F.; Yaqub, S.; Ahmad, S.; Mubarik, M.A.M.S.; Ahmad, B.; Ahmad, W.; Qamer, S. Comparative study of physio-chemical analysis of fresh and branded honeys from pakistan. Saudi J. Biol. Sci. 2020, 27, 173–176. [Google Scholar] [CrossRef]
- Lanjwani, M.F.; Channa, F.A. Minerals content in different types of local and branded honey in Sindh, Pakistan. Heliyon 2019, 5, e02042. [Google Scholar] [CrossRef] [PubMed]
- Madejczyk, M.; Baralkiewicz, D. Characterization of polish rape and honeydew honey according to their mineral contents using ICP-MS and F-AAS/AES. Anal. Chim. Acta 2008, 617, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Kováčik, J.; Grúz, J.; Biba, O.; Hedbavny, J. Content of metals and metabolites in honey originated from the vicinity of industrial town Košice (Eastern Slovakia). Environ. Sci. Pollut. Res. 2016, 23, 4531–4540. [Google Scholar] [CrossRef]
- Kılıç Altun, S.; Dinç, H.; Paksoy, N.; Temamoğulları, F.K.; Savrunlu, M. Analyses of mineral content and heavy metal of honey samples from south and east region of turkey by using icp-ms. Int. J. Anal. Chem. 2017, 2017, 6391454. [Google Scholar] [CrossRef]
- Citak, D.; Silici, S.; Tuzen, M.; Soylak, M. Determination of toxic and essential elements in sunflower honey from thrace region, turkey. Int. J. Food Sci. Technol. 2012, 47, 107–113. [Google Scholar] [CrossRef]
- Alimohamadi, Y.; Sepandi, M.; Taghdir, M.; Hosamirudsari, H. Determine the most common clinical symptoms in COVID-19 patients: A systematic review and meta-analysis. J. Prev. Med. Hyg. 2020, 61, E304–E312. [Google Scholar]
- Hu, B.; Guo, H.; Zhou, P.; Shi, Z.-L. Characteristics of SARS-CoV-2 and COVID-19. Nat. Rev. Microbiol. 2021, 19, 141–154. [Google Scholar] [CrossRef] [PubMed]
- Plitnick, J.; Griesemer, S.; Lasek-Nesselquist, E.; Singh, N.; Lamson, D.M.; George, K.S.; Dekker, J.P. Whole-genome sequencing of SARS-CoV-2: Assessment of the Ion Torrent AmpliSeq Panel and Comparison with the Illumina MiSeq ARTIC protocol. J. Clin. Microbiol. 2021, 59, e00649-21. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Yang, C.; Xu, X.-f.; Xu, W.; Liu, S.-w. Structural and functional properties of SARS-Cov-2 spike protein: Potential antivirus drug development for COVID-19. Acta Pharmacol. Sin. 2020, 41, 1141–1149. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Jedrzejczak, R.; Maltseva, N.I.; Wilamowski, M.; Endres, M.; Godzik, A.; Michalska, K.; Joachimiak, A. Crystal structure of Nsp15 endoribonuclease NendoU from SARS-CoV-2. Protein. Sci. 2020, 29, 1596–1605. [Google Scholar] [CrossRef]
- Sun, S.H.; Chen, Q.; Gu, H.J.; Yang, G.; Wang, Y.X.; Huang, X.Y.; Liu, S.S.; Zhang, N.N.; Li, X.F.; Xiong, R.; et al. A mouse model of SARS-CoV-2 infection and pathogenesis. Cell Host Microbe 2020, 28, 124–133.e124. [Google Scholar] [CrossRef]
- Harrison, A.G.; Lin, T.; Wang, P. Mechanisms of SARS-CoV-2 transmission and pathogenesis. Trends Immunol. 2020, 41, 1100–1115. [Google Scholar] [CrossRef] [PubMed]
- Tai, W.; He, L.; Zhang, X.; Pu, J.; Voronin, D.; Jiang, S.; Zhou, Y.; Du, L. Characterization of the receptor-binding domain (RBD) of 2019 novel coronavirus: Implication for development of RBD protein as a viral attachment inhibitor and vaccine. Cell Mol. Immunol. 2020, 17, 613–620. [Google Scholar] [CrossRef]
- Nishiga, M.; Wang, D.W.; Han, Y.; Lewis, D.B.; Wu, J.C. COVID-19 and cardiovascular disease: From basic mechanisms to clinical perspectives. Nat. Rev. Cardiol. 2020, 17, 543–558. [Google Scholar] [CrossRef]
- Barton, M.I.; MacGowan, S.A.; Kutuzov, M.A.; Dushek, O.; Barton, G.J.; van der Merwe, P.A. Effects of common mutations in the SARS-CoV-2 spike RBD and its ligand, the human ACE2 receptor on binding affinity and kinetics. Elife 2021, 10, e70658. [Google Scholar] [CrossRef]
- Oroojalian, F.; Haghbin, A.; Baradaran, B.; Hemmat, N.; Shahbazi, M.-A.; Baghi, H.B.; Mokhtarzadeh, A.; Hamblin, M.R. Novel insights into the treatment of SARS-CoV-2 infection: An overview of current clinical trials. Int. J. Biol. Macromol. 2020, 165, 18–43. [Google Scholar] [CrossRef]
- Mulangu, S.; Dodd, L.; Davey, R.; Mbaya, O.; Proschan, M.; Mukadi, D.; Manzo, M.; Nzolo, D.; Oloma, A.; Ibanda, A.; et al. A randomized, controlled trial of Ebola virus disease therapeutics. N. Engl. J. Med. 2019, 381, 2293–2303. [Google Scholar] [CrossRef] [PubMed]
- Study to Evaluate the Safety and Antiviral Activity of Remdesivir (GS-5734™) in Participants with Severe Coronavirus Disease (COVID-19). Available online: https://www.clinicaltrials.gov/ct2/show/NCT04292899 (accessed on 27 June 2022).
- Sheahan, T.P.; Sims, A.C.; Leist, S.R.; Schäfer, A.; Won, J.; Brown, A.J.; Montgomery, S.A.; Hogg, A.; Babusis, D.; Clarke, M.O.; et al. Comparative therapeutic efficacy of remdesivir and combination lopinavir, ritonavir, and interferon beta against MERS-CoV. Nat. Commun. 2020, 11, 222. [Google Scholar] [CrossRef] [PubMed]
- Furuta, Y.; Komeno, T.; Nakamura, T. Favipiravir (t-705), a broad spectrum inhibitor of viral rna polymerase. Proc. Jpn. Acad. Ser. B 2017, 93, 449–463. [Google Scholar] [CrossRef] [PubMed]
- Joshi, S.; Parkar, J.; Ansari, A.; Vora, A.; Talwar, D.; Tiwaskar, M.; Patil, S.; Barkate, H. Role of favipiravir in the treatment of COVID-19. Int. J. Infect. Dis. 2021, 102, 501–508. [Google Scholar] [CrossRef] [PubMed]
- Chuah, M.K.L.; Vandendriessche, T.; Morgan, R.A. Anti-viral strategies. In Molecular and Cell Biology of Human Gene Therapeutics; Dickson, G., Ed.; Springer: Dordrecht, The Netherlands, 1995; pp. 175–194. [Google Scholar]
- Yilmaz, N.K.; Swanstrom, R.; Schiffer, C.A. Improving viral protease inhibitors to counter drug resistance. Trends Microbiol. 2016, 24, 547–557. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Montero, J.V.; Barreiro, P.; Soriano, V. HIV protease inhibitors: Recent clinical trials and recommendations on use. Expert Opin. Pharmacother. 2009, 10, 1615–1629. [Google Scholar] [CrossRef]
- Rowe, I.A.; Mutimer, D.J. Protease inhibitors for treatment of genotype 1 hepatitis C virus infection. BMJ 2011, 343, d6972. [Google Scholar] [CrossRef]
- Anderson, J.; Schiffer, C.; Lee, S.-K.; Swanstrom, R. Viral protease inhibitors. In Antiviral Strategies; Kräusslich, H.-G., Bartenschlager, R., Eds.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 85–110. [Google Scholar]
- FDA, US Food and Drug Administration. Why You should Not Use Ivermectin to Treat or Prevent COVID-19. Available online: https://www.fda.gov/consumers/consumer-updates/why-you-should-not-use-ivermectin-treat-or-prevent-covid-19 (accessed on 27 June 2022).
- Caly, L.; Druce, J.D.; Catton, M.G.; Jans, D.A.; Wagstaff, K.M. The FDA-approved drug ivermectin inhibits the replication of SARS-CoV-2 in vitro. Antivir. Res. 2020, 178, 104787. [Google Scholar] [CrossRef]
- Popp, M.; Stegemann, M.; Metzendorf, M.I.; Gould, S.; Kranke, P.; Meybohm, P.; Skoetz, N.; Weibel, S. Ivermectin for preventing and treating COVID-19. Cochrane Database Syst. Rev 2021, CD015017. [Google Scholar] [CrossRef]
- Eckert, D.M.; Kim, P.S. Mechanisms of viral membrane fusion and its inhibition. Annu. Rev. Biochem. 2001, 70, 777–810. [Google Scholar] [CrossRef] [PubMed]
- Musarrat, F.; Chouljenko, V.; Dahal, A.; Nabi, R.; Chouljenko, T.; Jois, S.D.; Kousoulas, K.G. The anti-hiv drug nelfinavir mesylate (viracept) is a potent inhibitor of cell fusion caused by the sarscov-2 spike (s) glycoprotein warranting further evaluation as an antiviral against COVID-19 infections. J. Med. Virol. 2020, 92, 2087–2095. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Cao, R.; Zhang, L.; Yang, X.; Liu, J.; Xu, M.; Shi, Z.; Hu, Z.; Zhong, W.; Xiao, G. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-ncov) in vitro. Cell Res. 2020, 30, 269–271. [Google Scholar] [CrossRef] [PubMed]
- Yao, X.; Ye, F.; Zhang, M.; Cui, C.; Huang, B.; Niu, P.; Liu, X.; Zhao, L.; Dong, E.; Song, C.; et al. In vitro antiviral activity and projection of optimized dosing design of hydroxychloroquine for the treatment of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Clin. Infect. Dis. 2020, 71, 732–739. [Google Scholar] [CrossRef] [PubMed]
- Rolain, J.-M.; Colson, P.; Raoult, D. Recycling of chloroquine and its hydroxyl analogue to face bacterial, fungal and viral infections in the 21st century. Int. J. Antimicrob. Agents 2007, 30, 297–308. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Li, M.-X.; Lu, G.-D.; Shen, H.-M.; Zhou, J. Hydroxychloroquine/chloroquine as therapeutics for COVID-19: Truth under the mystery. Int. J. Biol. Sci. 2021, 17, 1538–1546. [Google Scholar] [CrossRef]
- Feuillet, V.; Canard, B.; Trautmann, A. Combining antivirals and immunomodulators to fight COVID-19. Trends Immunol. 2021, 42, 31–44. [Google Scholar] [CrossRef] [PubMed]
- Ngamprasertchai, T.; Kajeekul, R.; Sivakorn, C.; Ruenroegnboon, N.; Luvira, V.; Siripoon, T.; Luangasanatip, N. Efficacy and safety of immunomodulators in patients with covid-19: A systematic review and network meta-analysis of randomized controlled trials. Infect Dis. Ther. 2021, 11, 231–248. [Google Scholar] [CrossRef] [PubMed]
- Nile, S.H.; Nile, A.; Qiu, J.; Li, L.; Jia, X.; Kai, G. COVID-19: Pathogenesis, cytokine storm and therapeutic potential of interferons. Cytokine Growth Factor Rev. 2020, 53, 66–70. [Google Scholar] [CrossRef] [PubMed]
- Brattsand, R.; Linden, M. Cytokine modulation by glucocorticoids: Mechanisms and actions in cellular studies. Aliment. Pharmacol. Ther. 1996, 10, 81–90. [Google Scholar] [CrossRef]
- Rello, J.; Waterer, G.W.; Bourdiol, A.; Roquilly, A. COVID-19, steroids and other immunomodulators: The jigsaw is not complete. Anaesth. Crit. Care Pain Med. 2020, 39, 699–701. [Google Scholar] [CrossRef]
- Bhat, S.A.; Rather, S.A.; Iqbal, A.; Qureshi, H.A.; Islam, N. Immunomodulators for curtailing COVID-19: A positive approach. J. Drug Deliv. Ther. 2020, 10, 286–294. [Google Scholar] [CrossRef]
- Dilokthornsakul, W.; Kosiyaporn, R.; Wuttipongwaragon, R.; Dilokthornsakul, P. Potential effects of propolis and honey in COVID-19 prevention and treatment: A systematic review of in silico and clinical studies. J. Integr. Med. 2022, 20, 114–125. [Google Scholar] [CrossRef] [PubMed]
- Iqbal Yatoo, M.; Hamid, Z.; Rather, I.; Nazir, Q.U.A.; Bhat, R.A.; Ul Haq, A.; Magray, S.N.; Haq, Z.; Sah, R.; Tiwari, R.; et al. Immunotherapies and immunomodulatory approaches in clinical trials—A mini review. Hum. Vaccines Immunother. 2021, 17, 1897–1909. [Google Scholar] [CrossRef] [PubMed]
- Market, M.; Angka, L.; Martel, A.B.; Bastin, D.; Olanubi, O.; Tennakoon, G.; Boucher, D.M.; Ng, J.; Ardolino, M.; Auer, R.C. Flattening the COVID-19 curve with natural killer cell based immunotherapies. Front. Immunol. 2020, 11, 1512. [Google Scholar] [CrossRef]
- Al-Hatamleh, M.; Hatmal, M.M.; Sattar, K.; Ahmad, S.; Mustafa, M.Z.; Carvalho Bittencourt, M.; Mohamud, R. Antiviral and Immunomodulatory Effects of Phytochemicals from Honey against COVID-19: Potential Mechanisms of Action and Future Directions. Molecules 2020, 25, 5017. [Google Scholar] [CrossRef]
- Major, J.; Crotta, S.; Llorian, M.; McCabe, T.M.; Gad, H.H.; Priestnall, S.L.; Hartmann, R.; Wack, A. Type i and iii interferons disrupt lung epithelial repair during recovery from viral infection. Science 2020, 369, 712–717. [Google Scholar] [CrossRef] [PubMed]
- Creech, C.B.; Walker, S.C.; Samuels, R.J. SARS-CoV-2 vaccines. JAMA 2021, 325, 1318–1320. [Google Scholar] [CrossRef]
- Baraniuk, C. What do we know about China’s COVID-19 vaccines? BMJ 2021, 373, n912. [Google Scholar] [CrossRef]
- Ura, T.; Okuda, K.; Shimada, M. Developments in viral vector-based vaccines. Vaccines 2014, 2, 624–641. [Google Scholar] [CrossRef]
- Pardi, N.; Hogan, M.J.; Porter, F.W.; Weissman, D. Mrna vaccines—A new era in vaccinology. Nat. Rev. Drug Discov. 2018, 17, 261–279. [Google Scholar] [CrossRef]
- Moderna COVID-19 Vaccine VRBPAC Briefing Document. Available online: https://www.fda.gov/media/159189/download (accessed on 28 June 2022).
- US Food and Drug Administration. Briefing Document, Moderna COVID-19 Vaccine. 2020. Available online: https://www.fda.gov›media›download (accessed on 28 June 2022).
- Turner, J.S.; O’Halloran, J.A.; Kalaidina, E.; Kim, W.; Schmitz, A.J.; Zhou, J.Q.; Lei, T.; Thapa, M.; Chen, R.E.; Case, J.B.; et al. SARS-CoV-2 mrna vaccines induce persistent human germinal centre responses. Nature 2021, 596, 109–113. [Google Scholar] [CrossRef]
- Alvarez-Suarez, J.M.; Gasparrini, M.; Forbes-Hernández, T.Y.; Mazzoni, L.; Giampieri, F. The composition and biological activity of honey: A focus on manuka honey. Foods 2014, 3, 420–432. [Google Scholar] [CrossRef] [PubMed]
- Ranneh, Y.; Akim, A.M.; Hamid, H.A.; Khazaai, H.; Fadel, A.; Mahmoud, A.M. Stingless bee honey protects against lipopolysaccharide induced-chronic subclinical systemic inflammation and oxidative stress by modulating nrf2, nf-κb and p38 mapk. Nutr. Metab. 2019, 16, 15. [Google Scholar] [CrossRef] [PubMed]
- Dżugan, M.; Tomczyk, M.; Sowa, P.; Grabek-Lejko, D. Antioxidant activity as biomarker of honey variety. Molecules 2018, 23, 2069. [Google Scholar] [CrossRef]
- Ali, A.M.; Kunugi, H. Propolis, bee honey, and their components protect against coronavirus disease 2019 (COVID-19): A review of in silico, in vitro, and clinical studies. Molecules 2021, 26, 1232. [Google Scholar] [CrossRef] [PubMed]
- Abedi, F.; Ghasemi, S.; Farkhondeh, T.; Azimi-Nezhad, M.; Shakibaei, M.; Samarghandian, S. Possible potential effects of honey and its main components against COVID-19 infection. Dose Response 2021, 19, 1559325820982423. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Sulaiman, S.A.; Baig, A.A.; Ibrahim, M.; Liaqat, S.; Fatima, S.; Jabeen, S.; Shamim, N.; Othman, N.H. Honey as a potential natural antioxidant medicine: An insight into its molecular mechanisms of action. Oxid. Med. Cell. Longev. 2018, 2018, 8367846. [Google Scholar] [CrossRef] [PubMed]
- Shahzad, A.; Cohrs, R.J. In vitro antiviral activity of honey against varicella zoster virus (vzv): A translational medicine study for potential remedy for shingles. Transl. Biomed. 2012, 3, 2. [Google Scholar] [CrossRef]
- Zeina, B.; Othman, O.; Al-Assad, S. Effect of honey versus thyme on rubella virus survival in vitro. J. Altern. Complement. Med. 1996, 2, 345–348. [Google Scholar] [CrossRef]
- Watanabe, K.; Rahmasari, R.; Matsunaga, A.; Haruyama, T.; Kobayashi, N. Anti-influenza viral effects of honey in vitro: Potent high activity of manuka honey. Arch. Med. Res. 2014, 45, 359–365. [Google Scholar] [CrossRef]
- Abdel-Naby Awad, O.G.; Hamad, A.H. Honey can help in herpes simplex gingivostomatitis in children: Prospective randomized double blind placebo controlled clinical trial. Am. J. Otolaryngol. 2018, 39, 759–763. [Google Scholar] [CrossRef] [PubMed]
- Abuelgasim, H.; Albury, C.; Lee, J. Effectiveness of honey for symptomatic relief in upper respiratory tract infections: A systematic review and meta-analysis. BMJ Evid. Based Med. 2021, 26, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Obossou, E.K.; Shikamoto, Y.; Hoshino, Y.; Kohno, H.; Ishibasi, Y.; Kozasa, T.; Taguchi, M.; Sakakibara, I.; Tonooka, K.; Shinozuka, T.; et al. Effect of manuka honey on human immunodeficiency virus type 1 reverse transcriptase activity. Nat. Prod. Res. 2021, 36, 1552–1557. [Google Scholar] [CrossRef] [PubMed]
- Ranneh, Y.; Akim, A.M.; Hamid, H.A.; Khazaai, H.; Fadel, A.; Zakaria, Z.A.; Albujja, M.; Bakar, M.F.A. Honey and its nutritional and anti-inflammatory value. BMC Complement. Altern. Med. 2021, 21, 30. [Google Scholar] [CrossRef] [PubMed]
- Hussein, S.Z.; Mohd Yusoff, K.; Makpol, S.; Mohd Yusof, Y.A. Gelam Honey Inhibits the Production of Proinflammatory, Mediators NO, PGE(2), TNF-α, and IL-6 in Carrageenan-Induced Acute Paw Edema in Rats. Evid. Based Complement. Altern. Med. 2012, 2012, 109636. [Google Scholar] [CrossRef] [PubMed]
- Rossi, M.; Marrazzo, P. The potential of honeybee products for biomaterial applications. Biomimetics 2021, 6, 6. [Google Scholar] [CrossRef] [PubMed]
- Elmahallawy, E.K.; Mohamed, Y.; Abdo, W.; El-Gohary, F.A.; Ahmed Awad Ali, S.; Yanai, T. New insights into potential benefits of bioactive compounds of bee products on COVID-19: A review and assessment of recent research. Front. Mol. Biosci. 2020, 7, 618318. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.-W.; Ducroux, A.; Jeang, K.-T.; Neuveut, C. Impact of cellular autophagy on viruses: Insights from hepatitis b virus and human retroviruses. J. Biomed. Sci. 2012, 19, 92. [Google Scholar] [CrossRef]
- Curuțiu, C.; Dițu, L.M.; Grumezescu, A.M.; Holban, A.M. Polyphenols of honeybee origin with applications in dental medicine. Antibiotics 2020, 9, 856. [Google Scholar] [CrossRef]
- Mandal, M.D.; Mandal, S. Honey: Its medicinal property and antibacterial activity. Asian Pac. J. Trop. Biomed. 2011, 1, 154–160. [Google Scholar] [CrossRef]
- Al-Waili, N.S. Topical honey application vs. Acyclovir for the treatment of recurrent herpes simplex lesions. Med. Sci. Monit. 2004, 10, Mt94-8. [Google Scholar]
- Charyasriwong, S.; Haruyama, T.; Kobayashi, N. In vitro evaluation of the antiviral activity of methylglyoxal against influenza b virus infection. Drug Discov. Ther. 2016, 10, 201–210. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hossain, K.S.; Hossain, M.G.; Moni, A.; Rahman, M.M.; Rahman, U.H.; Alam, M.; Kundu, S.; Rahman, M.M.; Hannan, M.A.; Uddin, M.J. Prospects of honey in fighting against COVID-19: Pharmacological insights and therapeutic promises. Heliyon 2020, 6, e05798. [Google Scholar] [CrossRef]
- Combarros-Fuertes, P.; Estevinho, L.M.; Dias, L.G.; Castro, J.M.; Tomás-Barberán, F.A.; Tornadijo, M.E.; Fresno-Baro, J.M. Bioactive components and antioxidant and antibacterial activities of different varieties of honey: A screening prior to clinical application. J. Agric. Food Chem. 2019, 67, 688–698. [Google Scholar] [CrossRef]
- Corrêa, T.A.F.; Rogero, M.M. Polyphenols regulating micrornas and inflammation biomarkers in obesity. Nutrition 2019, 59, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Güler, H.İ.; Ay Şal, F.; Can, Z.; Kara, Y.; Yildiz, O.; Beldüz, A.O.; Çanakçi, S.; Kolayli, S. Targeting cov-2 spike rbd and ace-2 interaction with flavonoids of anatolian propolis by in silico and in vitro studies in terms of possible COVID-19 therapeutics. Turk. J. Biol. 2021, 45, 530–548. [Google Scholar] [CrossRef] [PubMed]
- Shaldam, M.A.; Yahya, G.; Mohamed, N.H.; Abdel-Daim, M.M.; Al Naggar, Y. In silico screening of potent bioactive compounds from honeybee products against COVID-19 target enzymes. Environ. Sci. Pollut. Res. Int. 2021, 28, 40507–40514. [Google Scholar] [CrossRef] [PubMed]
- Hussain, F.; Jahan, N.; Rahman, K.-u.; Sultana, B.; Jamil, S. Identification of hypotensive biofunctional compounds of Coriandrum sativum and evaluation of their angiotensin-converting enzyme (ace) inhibition potential. Oxid. Med. Cell. Longev. 2018, 2018, 4643736. [Google Scholar] [CrossRef]
- Kalediene, L.; Baz, M.; Liubaviciute, A.; Biziuleviciene, G.; Grabauskyte, I.; Bieliauskiene, R.; Jovaisas, P.; Jurjonas, N. Antiviral effect of honey extract camelyn against SARS-CoV-2. J. Adv. Biotechnol. Exp. Ther. 2021, 4, 290–297. [Google Scholar] [CrossRef]
- Al Naggar, Y.; Giesy, J.P.; Abdel-Daim, M.M.; Javed Ansari, M.; Al-Kahtani, S.N.; Yahya, G. Fighting against the second wave of COVID-19: Can honeybee products help protect against the pandemic? Saudi J. Biol. Sci. 2021, 28, 1519–1527. [Google Scholar] [CrossRef]
- Smilowitz, N.R.; Kunichoff, D.; Garshick, M.; Shah, B.; Pillinger, M.; Hochman, J.S.; Berger, J.S. C-reactive protein and clinical outcomes in patients with covid-19. Eur. Heart J. 2021, 42, 2270–2279. [Google Scholar] [CrossRef]
- Chua, L.S.; Rahaman, N.L.A.; Adnan, N.A.; Eddie Tan, T.T. Antioxidant activity of three honey samples in relation with their biochemical components. J. Anal. Methods Chem. 2013, 2013, 313798. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, T.; Ishitsuka, Y.; Nakamura, Y.; Okiyama, N.; Watanabe, R.; Fujisawa, Y.; Fujimoto, M. Honey and chamomile activate keratinocyte antioxidative responses via the keap1/nrf2 system. Clin. Cosmet. Investig. Dermatol. 2020, 13, 657–660. [Google Scholar] [CrossRef] [PubMed]
- Manyi-loh, C.E.; Clarke, A.M.; Ndip, R.N. An overview of honey: Therapeutic properties and contribution in nutrition and human health. Afr. J. Microbiol. Res. 2011, 5, 844–852. [Google Scholar]
- Fukuda, M.; Kobayashi, K.; Hirono, Y.; Miyagawa, M.; Ishida, T.; Ejiogu, E.C.; Sawai, M.; Pinkerton, K.E.; Takeuchi, M. Jungle honey enhances immune function and antitumor activity. Evid. Based Complement. Alternat. Med. 2011, 2011, 908743. [Google Scholar] [CrossRef] [PubMed]
- Bíliková, K.; Šimúth, J. New criterion for evaluation of honey: Quantification of royal jelly protein apalbumin 1 in honey by Elisa. J. Agric. Food Chem. 2010, 58, 8776–8781. [Google Scholar] [CrossRef] [PubMed]
- Tonks, A.J.; Dudley, E.; Porter, N.G.; Parton, J.; Brazier, J.; Smith, E.L.; Tonks, A. A 5.8-kda component of manuka honey stimulates immune cells via tlr4. J. Leukoc. Biol. 2007, 82, 1147–1155. [Google Scholar] [CrossRef]
- Mesaik, M.A.; Azim, M.K.; Mohiuddin, S. Honey modulates oxidative burst of professional phagocytes. Phytother. Res. 2008, 22, 1404–1408. [Google Scholar] [CrossRef]
- Klappan, A.K.; Hones, S.; Mylonas, I.; Brüning, A. Proteasome inhibition by quercetin triggers macroautophagy and blocks mtor activity. Histochem. Cell Biol. 2012, 137, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Parveen, A.; Do, M.H.; Kang, M.C.; Yumnam, S.; Kim, S.Y. Molecular mechanisms of methylglyoxal-induced aortic endothelial dysfunction in human vascular endothelial cells. Cell Death Dis. 2020, 11, 403. [Google Scholar] [CrossRef]
- Erejuwa, O.O.; Sulaiman, S.A.; Wahab, M.S.A. Fructose might contribute to the hypoglycemic effect of honey. Molecules 2012, 17, 1900–1915. [Google Scholar] [CrossRef]
- Meo, S.A.; Ansari, M.J.; Sattar, K.; Chaudhary, H.U.; Hajjar, W.; Alasiri, S. Honey and diabetes mellitus: Obstacles and challenges—Road to be repaired. Saudi J. Biol. Sci. 2017, 24, 1030–1033. [Google Scholar] [CrossRef] [PubMed]
- Ruan, Q.; Yang, K.; Wang, W.; Jiang, L.; Song, J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from wuhan, china. Int. Care Med. 2020, 46, 846–848. [Google Scholar] [CrossRef] [PubMed]
- Bt Hj Idrus, R.; Sainik, N.Q.A.V.; Nordin, A.; Saim, A.B.; Sulaiman, N. Cardioprotective effects of honey and its constituent: An evidence-based review of laboratory studies and clinical trials. Int. J. Environ. Res. Public Health 2020, 17, 3613. [Google Scholar] [CrossRef] [PubMed]
- Alagwu, E.A.; Okwara, J.E.; Nneli, R.O.; Osim, E.E. Effect of honey intake on serum cholesterol, triglycerides and lipoprotein levels in albino rats and potential benefits on risks of coronary heart disease. Niger J. Physiol. Sci. 2011, 26, 161–165. [Google Scholar] [PubMed]
- Afroz, R.; Tanvir, E.M.; Karim, N.; Hossain, M.S.; Alam, N.; Gan, S.H.; Khalil, M.I. Sundarban honey confers protection against isoproterenol-induced myocardial infarction in wistar rats. BioMed. Res. Int. 2016, 2016, 6437641. [Google Scholar] [CrossRef]
- Rakha, M.K.; Nabil, Z.I.; Hussein, A.A. Cardioactive and vasoactive effects of natural wild honey against cardiac malperformance induced by hyperadrenergic activity. J. Med. Food 2008, 11, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Busserolles, J.; Gueux, E.; Rock, E.; Mazur, A.; Rayssiguier, Y. Substituting honey for refined carbohydrates protects rats from hypertriglyceridemic and prooxidative effects of fructose. J. Nutr. 2002, 132, 3379–3382. [Google Scholar] [CrossRef]

| Geographical Origin | Sugars (g/100 g) | Organic Acids (mg/kg) | Amino Acids (mg/kg) | Ref. | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Glucose | Fructose | Sucrose | Trehalose | Melezitose | Turanose | Maltose | Gluconic Acid | Tartaric Acid | Malic Acid | Citric Acid | Succinic Acid | Quinic Acid | Pyroglutamic Acid | Lactic Acid | Formic Acid | |||
| Brasil | 29.74–31.89 | 39.74–43.94 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [11] |
| Brasil | 37.7–45.4 | 50.0–59.2 | 0.7–3.9 | - | - | - | nd | - | - | - | - | - | - | - | - | - | - | [12] |
| Brasil | - | - | - | - | - | - | - | 3309.6–18,737.3 | - | <LOD–2861.2 | <LOD–1322.3 | <LOD–2292.0 | - | - | <LOD–3063.6 | <LOD–341.2 | - | [13] |
| China | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 394.7–1572.9 | [14] |
| China | - | - | - | - | - | - | - | 649.0–1682.9 | - | 15.6–262.6 | 31.9–58.3 | 11.7–34.8 | - | - | - | - | - | [15] |
| China | - | - | - | - | - | - | - | - | - | 7.6–32.0 | - | 0.8–38.9 | - | - | - | 1.1–151.8 | [16] | |
| China | 30.2–30.3 | 40.5–40.6 | 2.2–3.5 | - | - | - | - | - | - | - | - | - | - | - | - | - | 1192–1688 | [17] |
| Ecuador | 26.00–38.26 | 34.77–44.57 | 2.63–5.14 | - | - | - | - | - | - | - | 0.3–6.8 | - | - | - | 0.4–7.2 | - | - | [18] |
| Egypt | 10.63–26.54 | 4.48–50.78 | 1.34–3.59 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [19] |
| France | - | - | - | - | - | - | - | 1857–12725 | - | - | 44–434 | - | 54–1779 | 217–1962 | 125–752 | 19–1897 | - | [20] |
| Japan | - | - | - | - | - | - | - | 1337.7–6475.1 | 15 | 10.3–1724.4 | 5.7–307.4 | 7.9–91.9 | - | - | - | - | - | [15] |
| Malaysia | 12.17–40.9 | 15.03–48.44 | <0.01–7.29 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [21] |
| New Zeland | - | - | - | - | - | - | - | 1842.1–5448.9 | 2.8-7.2 | 40.0–267.6 | 6.2–288.6 | 5.1–58.6 | - | - | - | - | - | [15] |
| Poland | 26.32 | 27.6 | 0.1 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [22] |
| Portugal | 21.00–36.00 | 33.40–48.80 | 0.0–3.00 | 0.004–0.80 | 0.18–1.20 | 0.001–0.69 | 0.93–7.83 | - | - | - | - | - | - | - | - | - | - | [23] |
| Portugal | 18.1–31.1 | 25.5–45.3 | <LOQ–2.0 | <LOQ–0.77 | <LOQ–6.9 | 1.2–4.0 | <LOQ–3.0 | - | - | - | - | - | - | - | - | - | - | [24] |
| Romania | 25.7–39.1 | 34.5–41.9 | nd–0.06 | - | - | - | 1.0-3.7 | - | - | - | - | - | - | - | - | - | - | [25] |
| Spain, Romania | 23.2–38.7 | 32.9–42.3 | 0.2–2.3 | - | nd–0.2 | - | 1.2–2.9 | - | - | - | - | - | - | - | - | - | - | [26] |
| Spain | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 56.08–141.65 | [27] |
| Spain | - | - | - | - | - | - | - | 2877.7–3250.3 | 44.3–52.2 | 78.9–89.1 | 14.4 | - | - | - | - | - | [15] | |
| Spain | 27.4–32.8 | 34.6–40.8 | - | - | - | - | - | - | 47.1–241 | - | 7.1–69.7 | - | - | <LOQ-51.7 | 27.5–150.6 | - | [28] | |
| Several Countries | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 127.7–1523.2 | [29] |
| Tunisia | 31.07–36.58 | 35.78–37.84 | 0.20–4.6 | - | - | - | 1.36–4.34 | - | - | - | - | - | - | - | - | - | - | [30] |
| Geographical Origin | Total Protein (mg/g) | Enzymes | TPC (mg GAE)/kg) | TFC | Bioactive Compounds (mg/kg Honey) | Carotenoids (mg β-Carotene/100 g) | Ref. | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diastase (Amylase) | Invertase (Saccharase) | p-Coumaric | Gallic Acid | Caffeic Acid | Syringic Acid | Vanillic Acid | Chlorogenic Acid | Quercetin | ||||||
| Algeria | - | - | - | - | - | - | - | - | - | - | - | - | 0.03–0.101 | [31] |
| Algeria | - | - | - | 640–2000 | 30–280 mg/kg | - | - | - | - | - | - | - | - | [32] |
| Australia | - | - | - | 30.9–66.3 | 4160–9640 mg CE/kg | - | - | - | - | - | - | - | 1.74–6.96 | [33] |
| Brasil | - | 11.14–22.69 DN | - | - | - | 0.22–1.43 | - | nd–0.46 | - | nd–0.90 | - | nd–1.58 | - | [11] |
| Brasil | - | - | - | 73.93 | 2.03 mg/kg | 30.9 | nd | nd | nd | nd | - | - | [34] | |
| Brasil | 2.0–5.0 | - | - | - | - | - | - | - | - | - | - | - | - | [12] |
| Canada | - | - | - | 136.87 | 6.26 mg/kg | - | 32.1 | nd | nd | nd | nd | - | - | [34] |
| China | - | - | - | 22.90–159.04 | nd-4.42 mg/kg | - | nd–66.2 | nd–23.6 | nd–4.9 | nd | nd–21.2 | - | - | [34] |
| Croatia | - | 7.5–37.3 DN | 26.4–277.9 U/kg | - | - | - | - | - | - | - | - | - | - | [35] |
| Ecuador | 0.02–0.37 | 8.33–40 DN | - | - | - | - | - | - | - | - | - | - | - | [18] |
| Egypt | 1.69–4.67 | - | - | - | - | - | - | - | - | - | - | - | - | [19] |
| Ethiopia | - | 7.64–12.5 DN | - | - | - | - | - | - | - | - | - | - | - | [36] |
| Germany | - | - | - | 105.13–135.22 | 2.72–6.83 mg/kg | - | 29.3–45.5 | nd–14.8 | nd–3.2 | nd | nd | - | - | [34] |
| Italy | - | - | - | 400–730 | 20.66–30.56 mgCE/kg | - | - | - | - | - | - | - | - | [37] |
| Italy | - | - | - | 81.29–148.75 | 1.59–12.35 mg/kg | - | 21.0–55.9 | nd–20.3 | nd–18.4 | nd | nd–19.3 | - | - | [34] |
| Malaysia | - | - | 0.27–4.94 IN | - | - | - | - | - | - | - | - | - | - | [21] |
| Portugal | - | 15.2–15.6 DN | - | 678.4–698.1 | 494.4–563.3 mg/kg | - | - | - | - | - | - | - | - | [38] |
| Portugal | - | 16.15–37.77 DN | - | 139.52–591.87 | 16.46–112.83 mg/kg | - | - | - | - | - | - | - | - | [39] |
| Romania | - | - | - | - | - | 3.6-29.1 | 0.5–0.9 | 0.2–7.5 | - | - | - | - | - | [40] |
| Spain | 0.83–0.93 | - | - | - | - | - | - | - | - | - | - | - | - | [27] |
| Spain | - | - | - | 74.57–254.03 | 2.66–41.65 mg/kg | - | 28.5–63.6 | nd–20.9 | nd–87.7 | nd–5.1 | nd–19.6 | - | - | [34] |
| Tunisia | 0.13–0.16 | - | 46.25–184.68 U/kg | 32.17–119.42 | 9.58–22.45 mg CE/kg | - | - | - | - | - | - | - | 1.16–4.72 | [30] |
| Turkey | - | - | - | 1813–60510 | 7660–28,750 mg/kg | - | nd–390 | 60–4610 | - | - | nd–1140 | nd–1430 | - | [41] |
| Geographical Origin | Minerals and Trace Elements (mg/kg) | Ref. | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Fe | Zn | Cu | Mn | Na | K | Ca | Mg | ||
| Argentina | 2.07–4.5 | 0.51–2.75 | 0.09–1.19 | 0.14–8.84 | 4.88–105.95 | 134.1–2813.3 | 1.97–18.97 | 3.01–75.38 | [42] |
| Bulgaria | 0.71–19.25 | 0.71–1.71 | 0.05–0.49 | 0.31–4.7 | 6.3–20.2 | 136–1900 | 24–94 | 8.3–48 | [43] |
| Ecuador | nd | nd | nd | nd | 9.0–23.0 | 7.0–133.0 | 15.0–31.0 | 4.0–11.0 | [13] |
| Greece | 1.03–6.30 | 0.89–1.81 | 0.14–0.52 | 0.15–1.46 | 10.3–42.0 | 391–2494 | 18.0–78.0 | 6.8–63.6 | [44] |
| Pakistan | 0.04–0.19 | - | - | 1.05–3.11 | 211.6–579.6 | 166.5–465.66 | 0.5–0.73 | - | [45] |
| Pakistan | 2.98–16.2 | 1.11–4.1 | 0.08–0.33 | 0.12–0.95 | 77.5–200 | 225–439 | 46.1–98.1 | 31.3–73.8 | [46] |
| Poland | nd–16.1 | nd–9.93 | nd–1.82 | - | 0.38–89.6 | 7.7–2612.2 | 3.3–159.2 | 0.07–19.83 | [47] |
| Slovakia | 1.02–5.14 | 0.16–1.30 | 0.045–2.01 | 0.44–15.1 | 8.49–10.3 | 0.33–3.71 | 20.3–36.6 | 12.5–65.0 | [48] |
| Tunisia | 0.83–3.54 | 0.42–2.06 | 0.12–0.34 | - | 251.34–521.22 | 172.48–976.75 | 113.85–221.07 | 37.32–78.12 | [30] |
| Turkey | <0.001–7.25 | <0.001–0.24 | <0.001–0.93 | <0.001–0.27 | 0.48–13.1 | 1.18–268 | 0.77–4.5 | - | [49] |
| Turkey | BDL–14.0 | BDL–1.98 | BDL–0.46 | BDL–0.82 | - | - | - | - | [50] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Soares, S.; Bornet, M.; Grosso, C.; Ramalhosa, M.J.; Gouvinhas, I.; Garcia, J.; Rodrigues, F.; Delerue-Matos, C. Honey as an Adjuvant in the Treatment of COVID-19 Infection: A Review. Appl. Sci. 2022, 12, 7800. https://doi.org/10.3390/app12157800
Soares S, Bornet M, Grosso C, Ramalhosa MJ, Gouvinhas I, Garcia J, Rodrigues F, Delerue-Matos C. Honey as an Adjuvant in the Treatment of COVID-19 Infection: A Review. Applied Sciences. 2022; 12(15):7800. https://doi.org/10.3390/app12157800
Chicago/Turabian StyleSoares, Sónia, Mélina Bornet, Clara Grosso, Maria João Ramalhosa, Irene Gouvinhas, Juliana Garcia, Francisca Rodrigues, and Cristina Delerue-Matos. 2022. "Honey as an Adjuvant in the Treatment of COVID-19 Infection: A Review" Applied Sciences 12, no. 15: 7800. https://doi.org/10.3390/app12157800
APA StyleSoares, S., Bornet, M., Grosso, C., Ramalhosa, M. J., Gouvinhas, I., Garcia, J., Rodrigues, F., & Delerue-Matos, C. (2022). Honey as an Adjuvant in the Treatment of COVID-19 Infection: A Review. Applied Sciences, 12(15), 7800. https://doi.org/10.3390/app12157800












