Inactivation of Listeria monocytogenes and Salmonella on Stainless Steel by a Piezoelectric Cold Atmospheric Plasma Generator
Abstract
Featured Application
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Culture Conditions
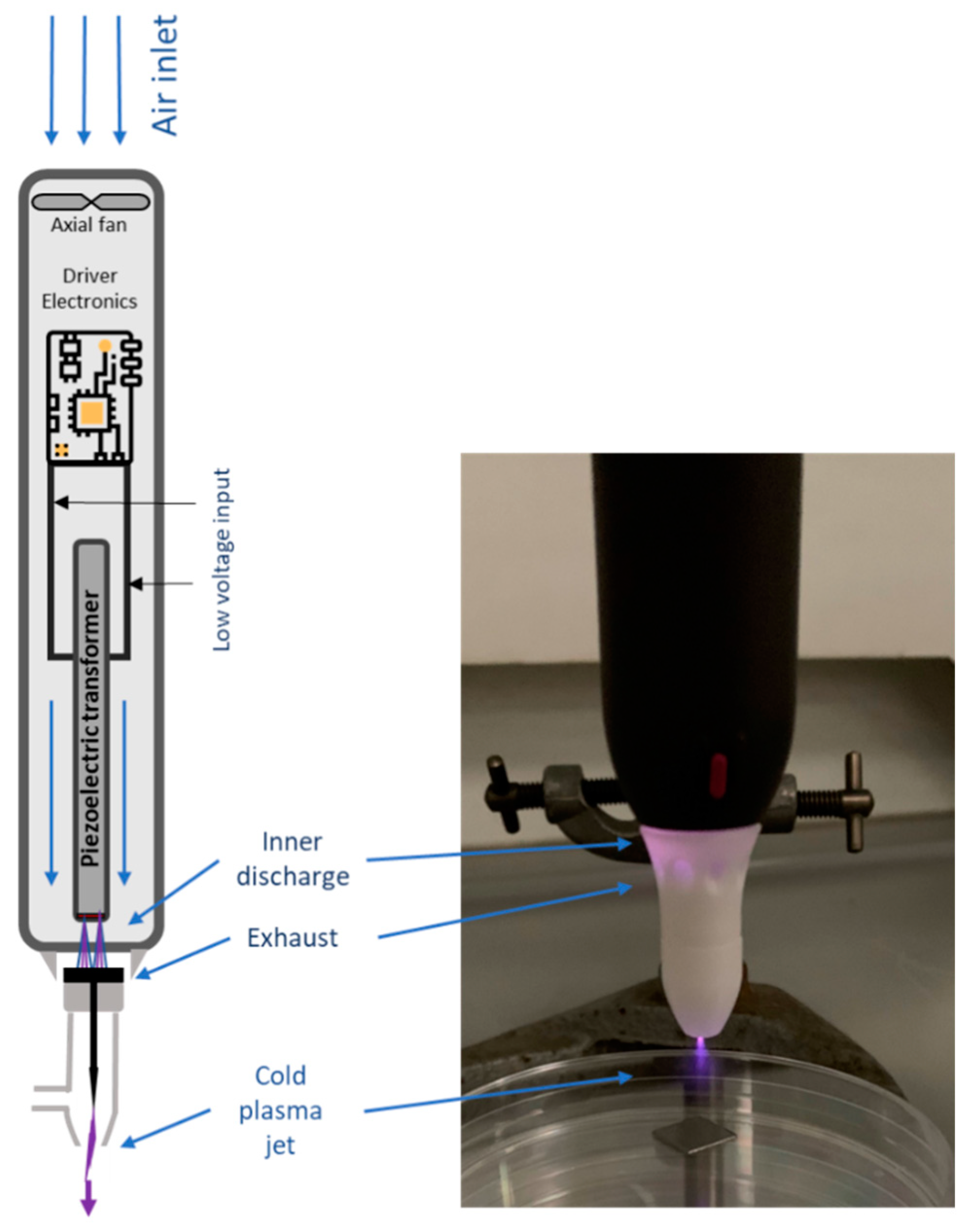
2.2. Cold Plasma System
2.3. Inoculation and Treatment of Clean and Protein-Soiled Stainless Steel
2.4. Recovery and Quantification of Salmonella and Listeria monocytogenes Cells
2.5. Statistical Analysis
2.6. Inactivation Modeling
3. Results and Discussion
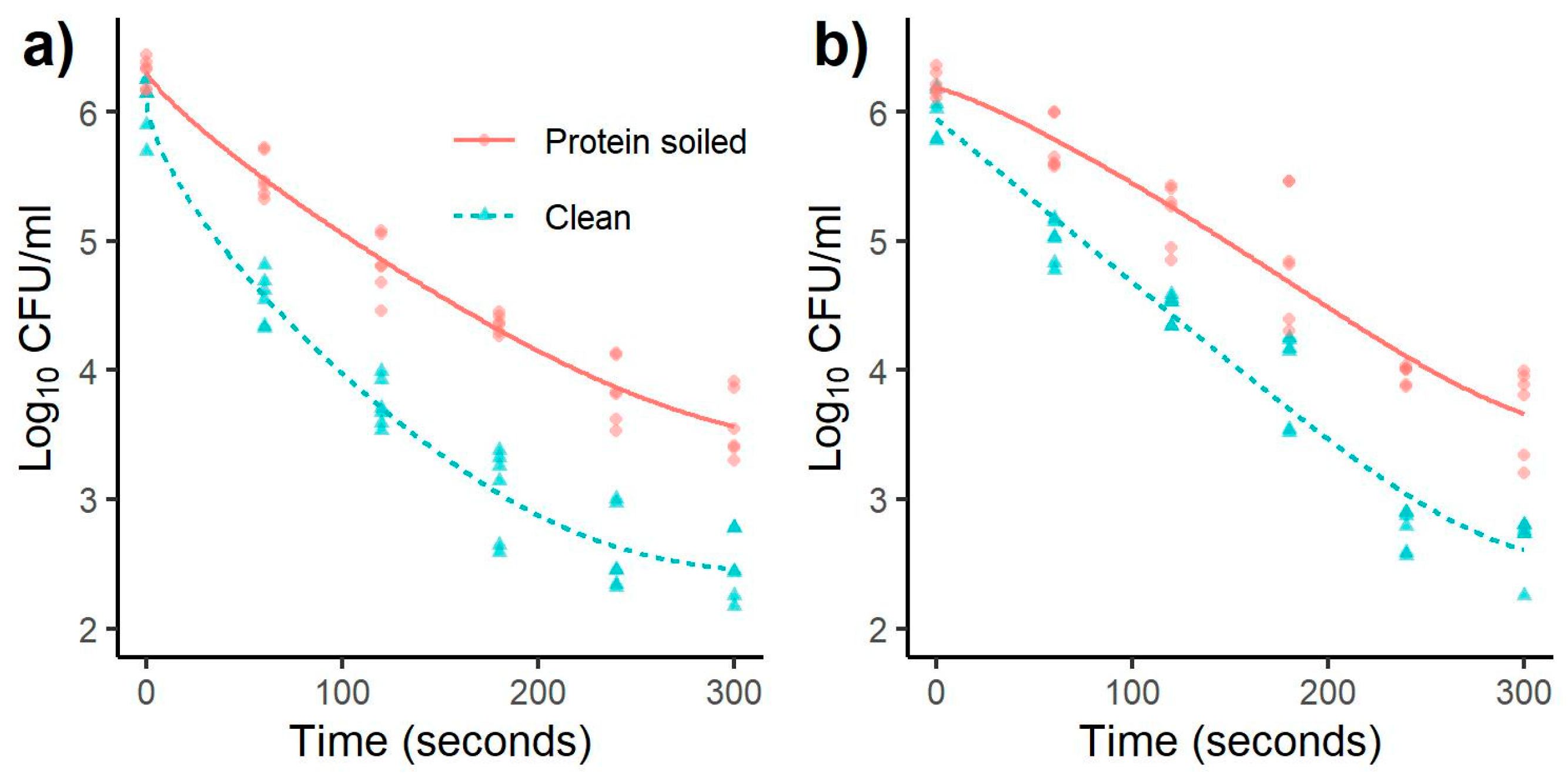
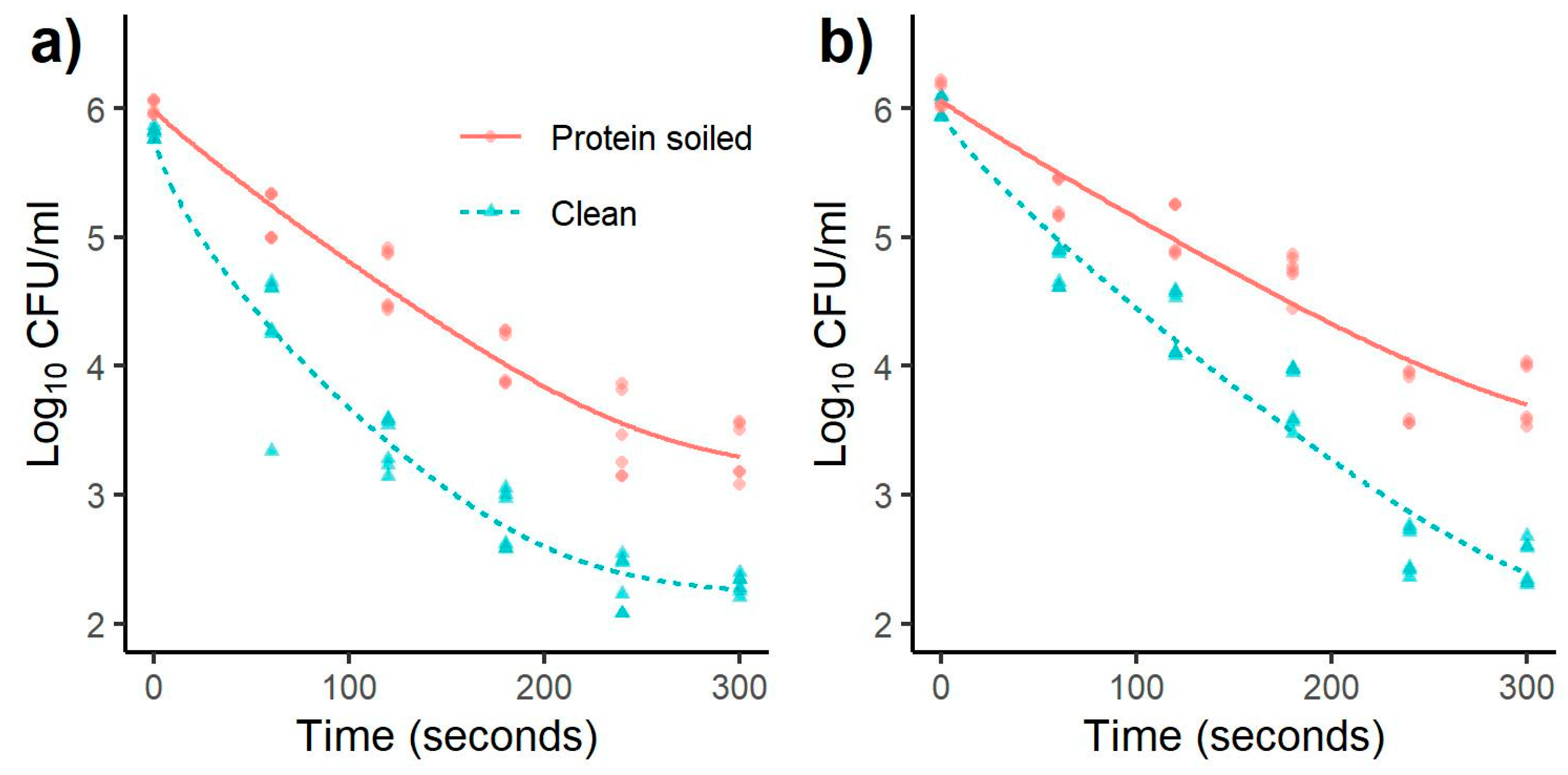
3.1. Inactivation of Salmonella and Listeria monocytogenes
3.2. Modeling Inactivation Kinetics
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- WHO. WHO Estimates of the Global Burden of Foodborne Diseases: Foodborne Diseases Burden Epidemiology Reference Group 2007–2015; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- Boelaert, F.; Amore, G.; Van der Stede, Y.; Nagy, K.; Rizzi, V.; Mirena, I.; Stoicescu, A.; Riolo, F.; Gervelmeyer, A.; Niskanen, T.; et al. The European Union summary report on trends and sources of zoonoses, zoonotic agents and food-borne outbreaks in 2015. EFSA J. 2016, 14, e04634. [Google Scholar] [CrossRef]
- Katsigiannis, A.S.; Bayliss, D.L.; Walsh, J.L. Cold plasma decontamination of stainless steel food processing surfaces assessed using an industrial disinfection protocol. Food Control 2021, 121, 107543. [Google Scholar] [CrossRef]
- Fink, R.; Oder, M.; Stražar, E.; Filip, S. Efficacy of cleaning methods for the removal of Bacillus cereus biofilm from polyurethane conveyor belts in bakeries. Food Control 2017, 80, 267–272. [Google Scholar] [CrossRef]
- Fagerlund, A.; Moretro, T.; Heir, E.; Briandet, R.; Langsrud, S.; Møretrø, T.; Heir, E.; Briandet, R.; Langsruda, S. Cleaning and Disinfection of Biofilms Composed of Listeria monocytogenes. Appl. Environ. Microbiol. 2017, 83, 1–21. [Google Scholar] [CrossRef]
- Corcoran, M.; Morris, D.; De Lappe, N.; O’Connor, J.; Lalor, P.; Dockery, P.; Cormican, M. Commonly used disinfectants fail to eradicate Salmonella enterica biofilms from food contact surface materials. Appl. Environ. Microbiol. 2014, 80, 1507–1514. [Google Scholar] [CrossRef]
- Otto, C.; Zahn, S.; Rost, F.; Zahn, P.; Jaros, D.; Rohm, H. Physical Methods for Cleaning and Disinfection of Surfaces. Food Eng. Rev. 2011, 3, 171–188. [Google Scholar] [CrossRef]
- Skåra, T.; Rosnes, J.T. 6—Emerging Methods and Principles in Food Contact Surface Decontamination/Prevention. In Woodhead Publishing Series in Food Science, Technology and Nutrition; Leadley, C.E., Ed.; Woodhead Publishing: Sawston, UK, 2016; pp. 151–172. ISBN 978-1-78242-447-5. [Google Scholar]
- Kilonzo-Nthenge, A.; Liu, S.; Yannam, S.; Patras, A. Atmospheric Cold Plasma Inactivation of Salmonella and Escherichia coli on the Surface of Golden Delicious Apples. Front. Nutr. 2018, 5, 120. [Google Scholar] [CrossRef]
- Noriega, E.; Shama, G.; Laca, A.; Díaz, M.; Kong, M.G. Cold atmospheric gas plasma disinfection of chicken meat and chicken skin contaminated with Listeria innocua. Food Microbiol. 2011, 28, 1293–1300. [Google Scholar] [CrossRef]
- Šimončicová, J.; Kryštofová, S.; Medvecká, V.; Ďurišová, K.; Kaliňáková, B. Technical applications of plasma treatments: Current state and perspectives. Appl. Microbiol. Biotechnol. 2019, 103, 5117–5129. [Google Scholar] [CrossRef]
- Liao, X.; Liu, D.; Xiang, Q.; Ahn, J.; Chen, S.; Ye, X.; Ding, T. Inactivation mechanisms of non-thermal plasma on microbes: A review. Food Control 2017, 75, 83–91. [Google Scholar] [CrossRef]
- Feizollahi, E.; Misra, N.N.; Roopesh, M.S. Factors influencing the antimicrobial efficacy of Dielectric Barrier Discharge (DBD) Atmospheric Cold Plasma (ACP) in food processing applications. Crit. Rev. Food Sci. Nutr. 2020, 61, 666–689. [Google Scholar] [CrossRef] [PubMed]
- Denes, F.S.; Reinemann, D.J.; Manolache, S.O.; Helgren, J.M. Plasma-Assisted Disinfection of Milking Machines 2005. U.S. Patent US 7,536,975, 26 May 2009. [Google Scholar]
- Cheol, M.S.; Hyun, K.J. A Method of Sterilization of Post-Packaging of Ready-to-Eat Food 2020. Korea Patent KR102113831B1, 11 May 2020. [Google Scholar]
- Potoroko, I.Y.; Naumenko, V.N.; Lejvi, Y.A.; Kalinina, I.V. Method of Grain Disinfection 2019. Russia Patent RU2707944C1, 19 July 2019. [Google Scholar]
- Butscher, D.; Zimmermann, D.; Schuppler, M.; Rudolf von Rohr, P. Plasma inactivation of bacterial endospores on wheat grains and polymeric model substrates in a dielectric barrier discharge. Food Control 2016, 60, 636–645. [Google Scholar] [CrossRef]
- Timmons, C.; Pai, K.; Jacob, J.; Zhang, G.; Ma, L.M. Inactivation of Salmonella enterica, Shiga toxin-producing Escherichia coli, and Listeria monocytogenes by a novel surface discharge cold plasma design. Food Control 2018, 84, 455–462. [Google Scholar] [CrossRef]
- Sen, Y.; Mutlu, M. Sterilization of Food Contacting Surfaces via Non-Thermal Plasma Treatment: A Model Study with Escherichia coli-Contaminated Stainless Steel and Polyethylene Surfaces. Food Bioprocess Technol. 2013, 6, 3295–3304. [Google Scholar] [CrossRef]
- Lis, K.A.; Boulaaba, A.; Binder, S.; Li, Y.; Kehrenberg, C.; Zimmermann, J.L.; Klein, G.; Ahlfeld, B. Inactivation of Salmonella Typhimurium and Listeria monocytogenes on ham with nonthermal atmospheric pressure plasma. PLoS ONE 2018, 13, e0197773. [Google Scholar] [CrossRef]
- Johnson, M.J.; Go, D.B. Piezoelectric transformers for low-voltage generation of gas discharges and ionic winds in atmospheric air. J. Appl. Phys. 2015, 118, 243304. [Google Scholar] [CrossRef]
- Timmermann, E.; Bansemer, R.; Gerling, T.; Hahn, V.; Weltmann, K.D.; Nettesheim, S.; Puff, M. Piezoelectric-driven plasma pen with multiple nozzles used as a medical device: Risk estimation and antimicrobial efficacy. J. Phys. D Appl. Phys. 2021, 54, 025201. [Google Scholar] [CrossRef]
- BS EN 13697:2015. Chemical Disinfectants and Antiseptics. In Quantitative Non-Porous Surface Test for the Evaluation of Bactericidal and/or Fungicidal Activity of Chemical Disinfectants Used in food, Industrial, Domestic and Institutional Areas; BSI Standards Publications: Brussels, Belgium, 2015; ISBN 978-0-539-07394-2. [Google Scholar]
- R-Core-Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019; Available online: https://www.R-project.org/ (accessed on 29 March 2021).
- Baty, F.; Delignette-Muller, M.-L. nlsMicrobio: Data Sets and Nonlinear Regression Models Dedicated to Predictive Microbiology 2014. Available online: https://CRAN.R-project.org/package=nlsMicrobio (accessed on 13 April 2021).
- Mafart, P.; Couvert, O.; Gaillard, S.; Leguerinel, I. On calculating sterility in thermal preservation methods: Application of the Weibull frequency distribution model. Acta Hortic. 2001, 566, 107–114. [Google Scholar] [CrossRef]
- van Boekel, M.A.J.S. On the use of the Weibull model to describe thermal inactivation of microbial vegetative cells. Int. J. Food Microbiol. 2002, 74, 139–159. [Google Scholar] [CrossRef]
- Albert, I.; Mafart, P. A modified Weibull model for bacterial inactivation. Int. J. Food Microbiol. 2005, 100, 197–211. [Google Scholar] [CrossRef]
- Lis, K.A.; Kehrenberg, C.; Boulaaba, A.; von Köckritz-Blickwede, M.; Binder, S.; Li, Y.; Zimmermann, J.L.; Pfeifer, Y.; Ahlfeld, B. Inactivation of multidrug-resistant pathogens and Yersinia enterocolitica with cold atmospheric-pressure plasma on stainless-steel surfaces. Int. J. Antimicrob. Agents 2018, 52, 811–818. [Google Scholar] [CrossRef]
- Suwal, S.; Coronel-Aguilera, C.P.; Auer, J.; Applegate, B.; Garner, A.L.; Huang, J.-Y. Mechanism characterization of bacterial inactivation of atmospheric air plasma gas and activated water using bioluminescence technology. Innov. Food Sci. Emerg. Technol. 2019, 53, 18–25. [Google Scholar] [CrossRef]
- Brun, P.; Bernabè, G.; Marchiori, C.; Scarpa, M.; Zuin, M.; Cavazzana, R.; Zaniol, B.; Martines, E. Antibacterial efficacy and mechanisms of action of low power atmospheric pressure cold plasma: Membrane permeability, biofilm penetration and antimicrobial sensitization. J. Appl. Microbiol. 2018, 125, 398–408. [Google Scholar] [CrossRef]
- Takai, E.; Kitano, K.; Kuwabara, J.; Shiraki, K. Protein Inactivation by Low-temperature Atmospheric Pressure Plasma in Aqueous Solution. Plasma Process. Polym. 2012, 9, 77–82. [Google Scholar] [CrossRef]
- Arjunan, K.P.; Sharma, V.K.; Ptasinska, S. Effects of Atmospheric Pressure Plasmas on Isolated and Cellular DNA—A Review. Int. J. Mol. Sci. 2015, 16, 2971–3016. [Google Scholar] [CrossRef]
- Thirumdas, R.; Sarangapani, C.; Annapure, U.S. Cold Plasma: A novel Non-Thermal Technology for Food Processing. Food Biophys. 2015, 10, 1–11. [Google Scholar] [CrossRef]
- Liao, X.; Forghani, F.; Liu, D.; Ding, T. Cumulative Damages by Nonthermal Plasma (NTP) Exceed Defense Barrier of Multiple Antibiotic Resistant Staphylococcus aureus: A Key to Achieve Completed Inactivation. Food Qual. Saf. 2021, 1–14. [Google Scholar] [CrossRef]
- Hasan, M.I.; Walsh, J.L. Numerical investigation of the spatiotemporal distribution of chemical species in an atmospheric surface barrier-discharge. J. Appl. Phys. 2016, 119, 203302. [Google Scholar] [CrossRef]
- Dickenson, A.; Britun, N.; Nikiforov, A.; Leys, C.; Hasan, M.I.; Walsh, J.L. The generation and transport of reactive nitrogen species from a low temperature atmospheric pressure air plasma source. Phys. Chem. Chem. Phys. 2018, 20, 28499–28510. [Google Scholar] [CrossRef]
- Niemira, B.A.; Boyd, G.; Sites, J. Cold Plasma Inactivation of Escherichia coli O157:H7 Biofilms. Front. Sustain. Food Syst. 2018, 2, 47. [Google Scholar] [CrossRef]
- Yong, H.I.; Kim, H.J.; Park, S.; Choe, W.; Oh, M.W.; Jo, C. Evaluation of the treatment of both sides of raw chicken breasts with an atmospheric pressure plasma jet for the inactivation of escherichia coli. Foodborne Pathog. Dis. 2014, 11, 652–657. [Google Scholar] [CrossRef]
- Maresca, P.; Ferrari, G. Modeling of the microbial inactivation by high hydrostatic pressure freezing. Food Control 2017, 73, 8–17. [Google Scholar] [CrossRef]
- Pan, Y.; Cheng, J.H.; Lv, X.; Sun, D.W. Assessing the inactivation efficiency of Ar/O2 plasma treatment against Listeria monocytogenes cells: Sublethal injury and inactivation kinetics. LWT 2019, 111, 318–327. [Google Scholar] [CrossRef]
- De Oliveira, T.L.C.; Soares, R.; de Araújo Soares, R.; Piccoli, R.H. A Weibull model to describe antimicrobial kinetics of oregano and lemongrass essential oils against Salmonella Enteritidis in ground beef during refrigerated storage. Meat Sci. 2013, 93, 645–651. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.; Lee, T.; Puligundla, P.; Mok, C. Effect of relative humidity on the inactivation of foodborne pathogens by corona discharge plasma jet (CDPJ). LWT 2020, 127, 109379. [Google Scholar] [CrossRef]
- Govaert, M.; Smet, C.; Baka, M.; Ećimović, B.; Walsh, J.L.; Van Impe, J. Resistance of L. monocytogenes and S. Typhimurium towards cold atmospheric plasma as function of biofilm age. Appl. Sci. 2018, 8, 2702. [Google Scholar] [CrossRef]
| Bacterial Species | Treatment Time (s) | Reduction (log CFU/mL) (Mean ± SD) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| 10 mm Distance | 20 mm Distance | ||||||||
| Protein-Soiled SS | Clean SS | Protein-Soiled SS | Clean SS | ||||||
| Listeria | 60 | 0.81 a | 0.21 | 1.51 a | 0.29 | 0.48 a | 0.22 | 1.01 a | 0.24 |
| monocytogenes | 120 | 1.49 b | 0.26 | 2.33 b | 0.29 | 1.02 b | 0.26 | 1.52 b | 0.21 |
| 180 | 1.95 c | 0.13 | 3.01 c | 0.41 | 1.34 b | 0.51 | 2.03 c | 0.39 | |
| 240 | 2.46 d | 0.27 | 3.47 d | 0.38 | 2.25 c | 0.12 | 3.23 d | 0.24 | |
| 300 | 2.73 d | 0.28 | 3.59 d | 0.34 | 2.52 c | 0.35 | 3.32 d | 0.28 | |
| Salmonella | 60 | 0.84 a | 0.20 | 1.52 a | 0.50 | 0.80 a | 0.18 | 1.25 a | 0.17 |
| Typhimurium | 120 | 1.34 b | 0.24 | 2.42 b | 0.20 | 1.04 a | 0.22 | 1.68 b | 0.27 |
| 180 | 1.94 c | 0.22 | 3.01 c | 0.23 | 1.38 b | 0.18 | 2.25 c | 0.25 | |
| 240 | 2.56 d | 0.33 | 3.49 d | 0.22 | 2.36 c | 0.23 | 3.44 d | 0.20 | |
| 300 | 2.67 d | 0.23 | 3.51 d | 0.08 | 2.32 c | 0.26 | 3.54 d | 0.19 | |
| Bacteria | Distance (mm) | Cleanliness | Weibull | Weibull + Tail | ||
|---|---|---|---|---|---|---|
| RMSE | R2 | RMSE | R2 | |||
| Listeria | 10 | Clean | 0.28 | 0.95 | 0.27 | 0.96 |
| 10 | Protein-soiled | 0.20 | 0.96 | 0.20 | 0.96 | |
| 20 | Clean | 0.29 | 0.94 | 0.29 | 0.94 | |
| 20 | Protein-soiled | 0.30 | 0.89 | 0.31 | 0.89 | |
| Salmonella | 10 | Clean | 0.29 | 0.95 | 0.27 | 0.96 |
| 10 | Protein-soiled | 0.24 | 0.94 | 0.24 | 0.94 | |
| 20 | Clean | 0.28 | 0.95 | 0.29 | 0.95 | |
| 20 | Protein-soiled | 0.27 | 0.90 | 0.27 | 0.90 | |
| Weibull | Weibull + Tail | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Bacteria | Distance (mm) | Cleanliness | δ ± SE | p ± SE | Predicted Log Reduction (300 s) | δ ± SE | p ± SE | Nres ± SE | Predicted Log Reduction (300 s) |
| L. monocytogenes | 10 | Clean | 23.14 ± 5.47 | 0.52 ± 0.05 | 3.77 | 33.29 ± 6.61 | 0.67 ± 0.08 | 2.38 ± 0.15 | 3.61 |
| 10 | Protein-soiled | 70.31 ± 9.02 | 0.71 ± 0.06 | 2.82 | 75.57 ± 9.1 | 0.82 ± 0.1 | 3.32 ± 0.26 | 2.74 | |
| 20 | Clean | 69.4 ± 10.38 | 0.85 ± 0.08 | 3.45 | 78.78 ± 11.16 | 0.99 ± 0.13 | 2.4 ± 0.29 | 3.34 | |
| 20 | Protein-soiled | 122.47 ± 16.44 | 1.06 ± 0.14 | 2.59 | 127.93 ± 15.97 | 1.23 ± 0.25 | 3.39 ± 0.45 | 2.52 | |
| Salmonella | 10 | Clean | 20.49 ± 5.34 | 0.49 ± 0.05 | 3.73 | 32.48 ± 6.45 | 0.68 ± 0.05 | 2.22 ± 0.12 | 3.55 |
| 10 | Protein-soiled | 73.42 ± 11.14 | 0.73 ± 0.07 | 2.81 | 82.88 ± 10.63 | 0.92 ± 0.13 | 3.16 ± 0.2 | 2.69 | |
| 20 | Clean | 55.69 ± 8.82 | 0.77 ± 0.07 | 3.63 | 61.2 ± 10.02 | 0.84 ± 0.11 | 2.04 ± 0.47 | 3.57 | |
| 20 | Protein-soiled | 104.23 ± 15.68 | 0.83 ± 0.1 | 2.41 | 109.41 ± 15.15 | 0.95 ± 0.19 | 3.36 ± 0.54 | 2.36 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gonzalez-Gonzalez, C.R.; Hindle, B.J.; Saad, S.; Stratakos, A.C. Inactivation of Listeria monocytogenes and Salmonella on Stainless Steel by a Piezoelectric Cold Atmospheric Plasma Generator. Appl. Sci. 2021, 11, 3567. https://doi.org/10.3390/app11083567
Gonzalez-Gonzalez CR, Hindle BJ, Saad S, Stratakos AC. Inactivation of Listeria monocytogenes and Salmonella on Stainless Steel by a Piezoelectric Cold Atmospheric Plasma Generator. Applied Sciences. 2021; 11(8):3567. https://doi.org/10.3390/app11083567
Chicago/Turabian StyleGonzalez-Gonzalez, Cid R., Bethan J. Hindle, Saliha Saad, and Alexandros Ch. Stratakos. 2021. "Inactivation of Listeria monocytogenes and Salmonella on Stainless Steel by a Piezoelectric Cold Atmospheric Plasma Generator" Applied Sciences 11, no. 8: 3567. https://doi.org/10.3390/app11083567
APA StyleGonzalez-Gonzalez, C. R., Hindle, B. J., Saad, S., & Stratakos, A. C. (2021). Inactivation of Listeria monocytogenes and Salmonella on Stainless Steel by a Piezoelectric Cold Atmospheric Plasma Generator. Applied Sciences, 11(8), 3567. https://doi.org/10.3390/app11083567







