Recent Progress in Applications of Non-Thermal Plasma for Water Purification, Bio-Sterilization, and Decontamination
Abstract
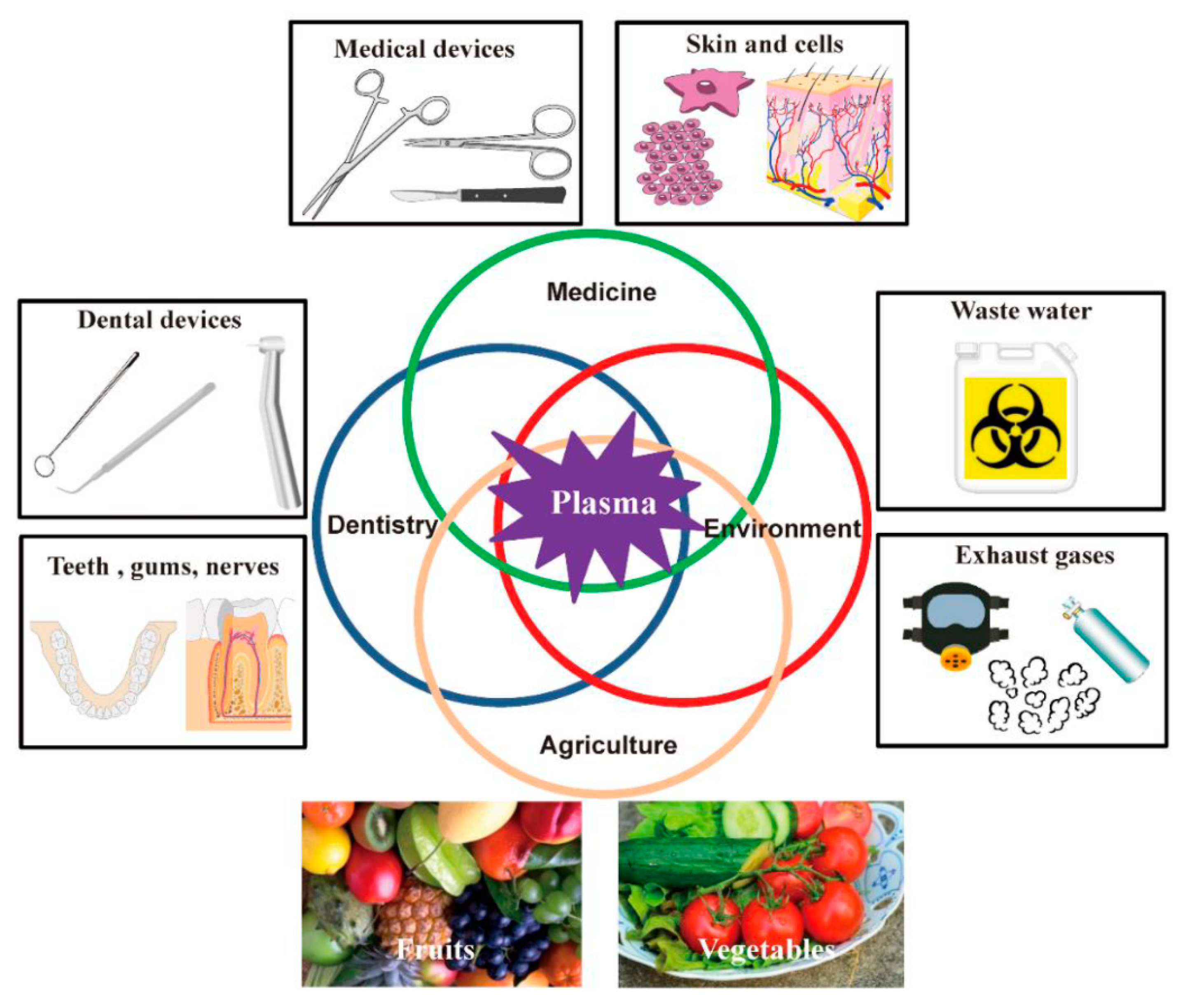
1. Introduction
- A.
- B.
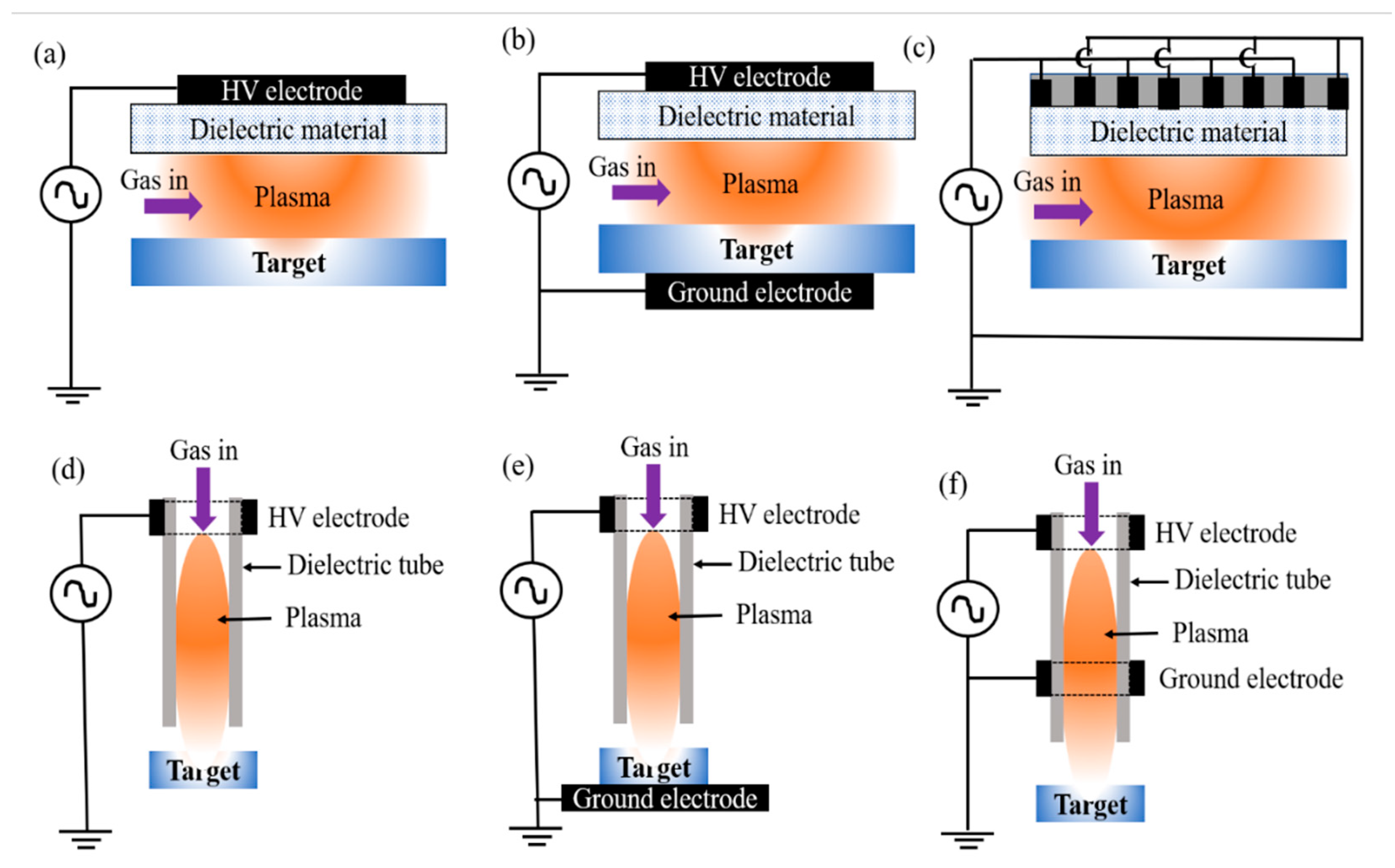
- In the indirect method, both electrodes lie above the target/water, plasma is produced above the water, and the reactive species from the plasma diffuse into the water, meaning that there is a contact surface. When the plasma is directly exposed to the water, electrons and reactive O species (ROS) are transported to the plasma–water interface and ultimately solvated into the bulk volume [34,45].
- C.
2. NTAPP Sources and Plasma Chemistry
2.1. Non-Thermal Plasma Sources
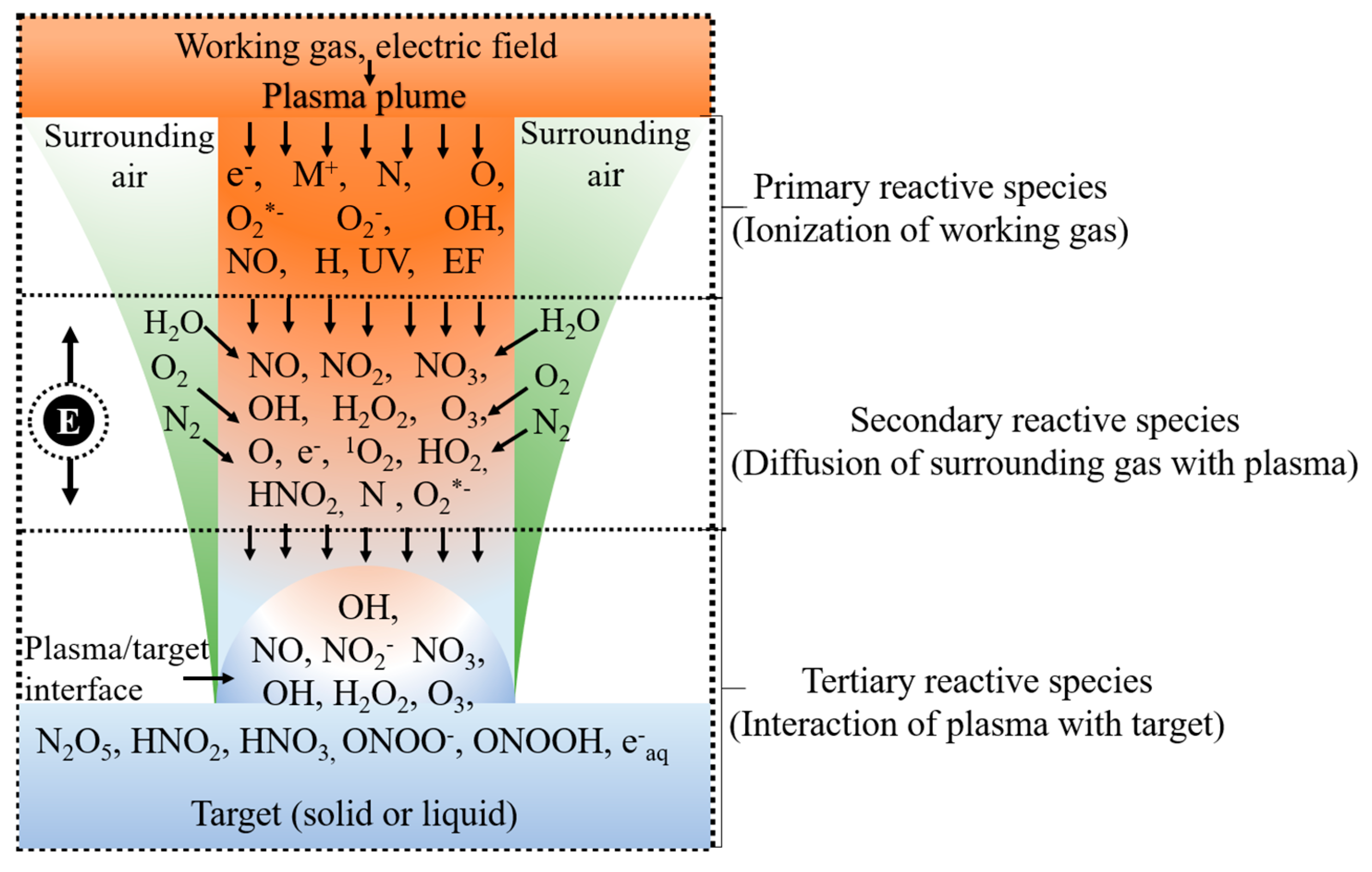
2.2. Reactive Species Formed in NTAPP Discharge
3. Water Purification
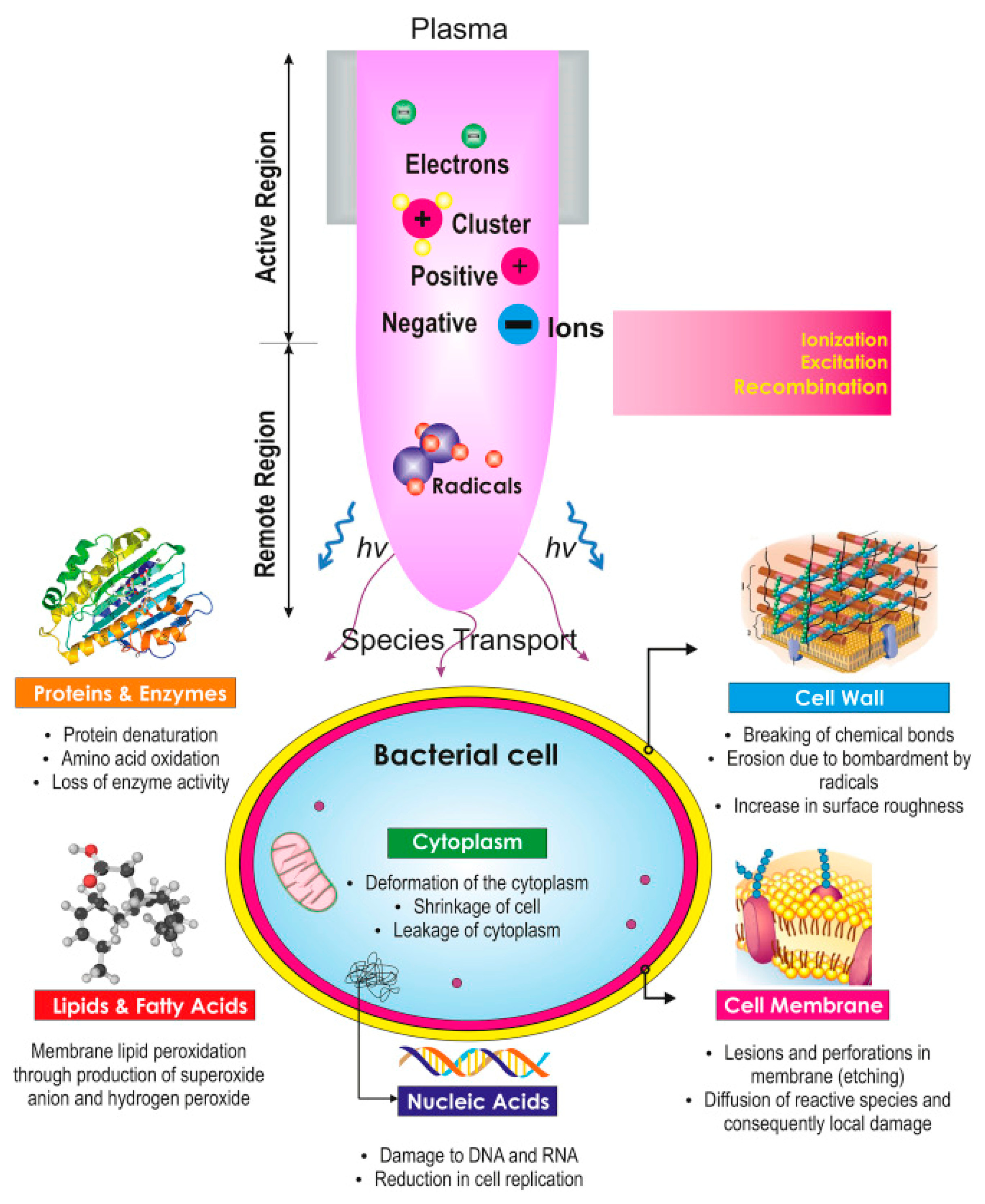
4. Water Sterilization and Disinfection
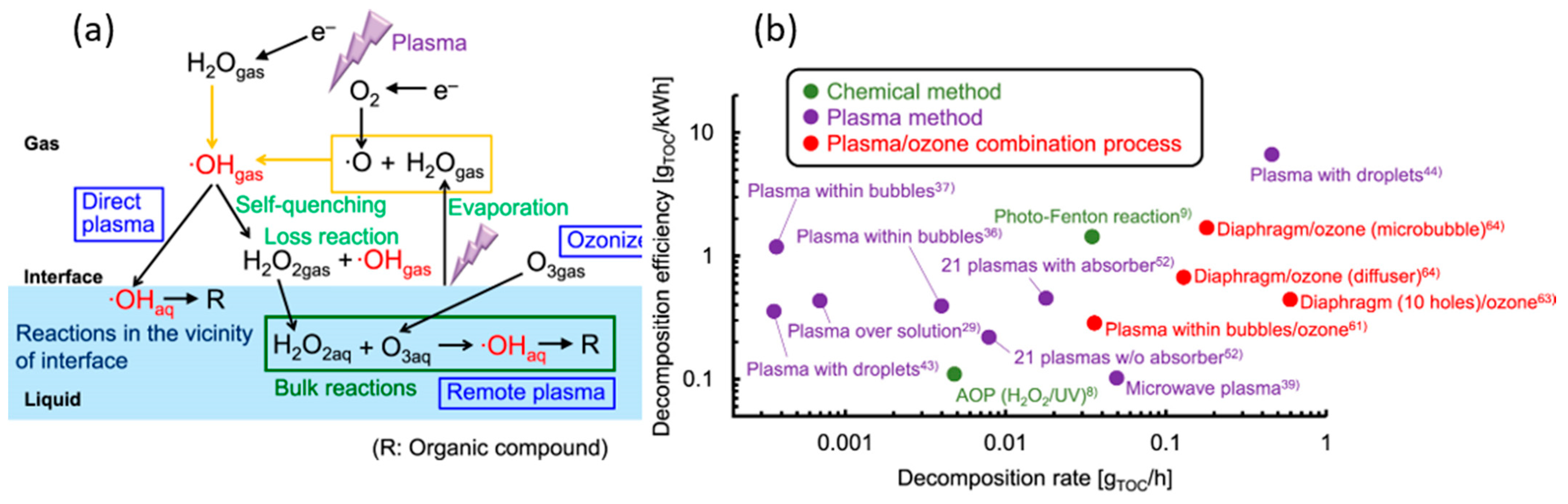
5. Decontamination of Chemical Pollutants
6. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Murugesan, P.; Moses, J.A.; Anandharamakrishnan, C. Water decontamination of using non-thermal plasma: Concepts, applications, and prospects. J. Environ. Chem. Eng. 2020, 8, 104377. [Google Scholar] [CrossRef]
- Liu, B. Non-Thermal Atmospheric Pressure Plasma Interacting with Water for Biological Applications. Ph.D Thesis, Université Paris Saclay (COmUE), Gif-sur-Yvette, France, 2019. [Google Scholar]
- Khan, R.; Bhawana, P.; Fulekar, M.H. Microbial decolorization and degradation of synthetic dyes: A review. Rev. Environ. Sci. Biol. Technol. 2013, 12, 75–97. [Google Scholar] [CrossRef]
- Inyang, M.; Dickenson, E. The potential role of biochar in the removal of organic and microbial contaminants from potable and reuse water: A review. Chemosphere 2015, 134, 232–240. [Google Scholar] [CrossRef]
- Matafonova, G.; Batoev, V. Recent progress on application of UV excilamps for degradation of organic pollutants and microbial inactivation. Chemosphere 2012, 89, 637–647. [Google Scholar] [CrossRef]
- Strong, L.C.; Gould, T.; Kasinkas, L.; Sadowsky, M.J.; Aksan, A.; Wackett, L.P. Biodegradation in waters from hydraulic fracturing: Chemistry, microbiology, and engineering. J. Environ. Eng. 2014, 140, B4013001. [Google Scholar] [CrossRef]
- Gude, V.G. Wastewater treatment in microbial fuel cells—An overview. J. Clean. Prod. 2016, 122, 287–307. [Google Scholar] [CrossRef]
- Naidoo, S.; Olaniran, A.O. Treated wastewater effluent as a source of microbial pollution of surface water resources. Int. J. Environ. Res. Public Health 2014, 11, 249–270. [Google Scholar] [CrossRef]
- Okpara, C.G.; Oparaku, N.F.; Ibeto, C.N. An overview of water disinfection in developing countries and potentials of renewable energy. J. Environ. Sci. Technol. 2011, 4, 18–30. [Google Scholar] [CrossRef]
- Deborde, M.; Von Gunten, U.R.S. Reactions of chlorine with inorganic and organic compounds during water treatment—kinetics and mechanisms: A critical review. Water Res. 2008, 42, 13–51. [Google Scholar] [CrossRef]
- Stefan, M.I. Advanced Oxidation Processes for Water Treatment: Fundamentals and Applications; IWA Publishing: London, UK, 2017. [Google Scholar]
- Mehrjouei, M.; Müller, S.; Möller, D. A review on photocatalytic ozonation used for the treatment of water and wastewater. Chem. Eng. J. 2015, 263, 209–219. [Google Scholar] [CrossRef]
- Ellis, K.; Wood, W.E. Slow sand filtration. Crit. Rev. Environ. Sci. Technol. 1985, 15, 315–354. [Google Scholar] [CrossRef]
- Burch, J.D.; Thomas, K.E. Water disinfection for developing countries and potential for solar thermal pasteurization. Sol. Energy 1998, 64, 87–97. [Google Scholar] [CrossRef]
- Lazar, M.A.; Varghese, S.; Nair, S.S. Photocatalytic water treatment by titanium dioxide: Recent updates. Catalysts 2012, 2, 572–601. [Google Scholar] [CrossRef]
- Chaplin, B.P. Critical review of electrochemical advanced oxidation processes for water treatment applications. Environ. Sci. Process. Impacts 2014, 16, 1182–1203. [Google Scholar] [CrossRef] [PubMed]
- Gehrke, I.; Geiser, A.; Somborn-Schulz, A. Innovations in nanotechnology for water treatment. Nanotechnol. Sci. Appl. 2015, 8, 1. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.K.; Ali, I.; Saleh, T.A.; Nayak, A.; Agarwal, S. Chemical treatment technologies for waste-water recycling—An overview. RSC Adv. 2012, 2, 6380–6388. [Google Scholar] [CrossRef]
- Vilhunen, S.; Sillanpää, M. Recent developments in photochemical and chemical AOPs in water treatment: A mini-review. Rev. Environ. Sci. Biol. Technol. 2010, 9, 323–330. [Google Scholar] [CrossRef]
- Miklos, D.B.; Remy, C.; Jekel, M.; Linden, K.G.; Drewes, J.E.; Hübner, U. Evaluation of advanced oxidation processes for water and wastewater treatment—A critical review. Water Res. 2018, 139, 118–131. [Google Scholar] [CrossRef]
- Deng, Y.; Zhao, R. Advanced Oxidation Processes (AOPs) in Wastewater Treatment. Curr. Pollut. Rep. 2015, 1, 167–176. [Google Scholar] [CrossRef]
- Oturan, M.A.; Aaron, J.-J. Advanced oxidation processes in water/wastewater treatment: Principles and applications. A review. Crit. Rev. Environ. Sci. Technol. 2014, 44, 2577–2641. [Google Scholar] [CrossRef]
- Ribeiro, A.R.; Nunes, O.C.; Pereira, M.F.R.; Silva, A.M.T. An overview on the advanced oxidation processes applied for the treatment of water pollutants defined in the recently launched Directive 2013/39/EU. Environ. Int. 2015, 75, 33–51. [Google Scholar] [CrossRef]
- Wang, J.L.; Xu, L.J. Advanced oxidation processes for wastewater treatment: Formation of hydroxyl radical and application. Crit. Rev. Environ. Sci. Technol. 2012, 42, 251–325. [Google Scholar] [CrossRef]
- Burm, K. Plasma: The fourth state of matter. Plasma Chem. Plasma Process. 2012, 32, 401–407. [Google Scholar] [CrossRef]
- López, M.; Calvo, T.; Prieto, M.; Múgica-Vidal, R.; Muro-Fraguas, I.; Alba-Elías, F.; Alvarez-Ordóñez, A. A review on non-thermal atmospheric plasma for food preservation: Mode of action, determinants of effectiveness, and applications. Front. Microbiol. 2019, 10, 622. [Google Scholar] [CrossRef]
- Lieberman, M.A.; Lichtenberg, A.J. Principles of Plasma Discharges and Materials Processing; John Wiley & Sons: Hoboken, NJ, USA, 2005. [Google Scholar]
- Schütze, A.; Jeong, J.Y.; Babayan, S.E.; Park, J.; Selwyn, G.S.; Hicks, R.F. The atmospheric-pressure plasma jet: A review and comparison to other plasma sources. IEEE Trans. Plasma Sci. 1998, 26, 1685–1694. [Google Scholar] [CrossRef]
- Setsuhara, Y. Low-temperature atmospheric-pressure plasma sources for plasma medicine. Arch. Biochem. Biophys. 2016, 605, 3–10. [Google Scholar] [CrossRef]
- Ghimire, B.; Lamichhane, P.; Lim, J.S.; Min, B.; Paneru, R.; Weltmann, K.; Choi, E.H. An atmospheric pressure plasma jet operated by injecting natural air. Appl. Phys. Lett. 2018, 113, 194101. [Google Scholar] [CrossRef]
- Lamichhane, P.; Adhikari, B.C.; Nguyen, L.N.; Paneru, R.; Ghimire, B.; Mumtaz, S.; Lim, J.S.; Hong, Y.J.; Choi, E.H. Sustainable nitrogen fixation from synergistic effect of photo-electrochemical water splitting and atmospheric pressure N2 plasma. Plasma Sources Sci. Technol. 2020, 29, 045026. [Google Scholar] [CrossRef]
- Toth, J.R.; Abuyazid, N.H.; Lacks, D.J.; Renner, J.N.; Sankaran, R.M. A plasma-water droplet reactor for process-intensified, continuous nitrogen fixation at atmospheric pressure. ACS Sustain. Chem. Eng. 2020, 8, 14845–14854. [Google Scholar] [CrossRef]
- Lamichhane, P.; Ghimire, B.; Mumtaz, S.; Paneru, R.; Ki, S.H.; Choi, E.H. Control of hydrogen peroxide production in plasma activated water by utilizing nitrification. J. Phys. D Appl. Phys. 2019, 52, 265206. [Google Scholar] [CrossRef]
- Lamichhane, P.; Paneru, R.; Nguyen, L.N.; Lim, J.S.; Bhartiya, P.; Adhikari, B.C.; Mumtaz, S.; Choi, E.H. Plasma-assisted nitrogen fixation in water with various metals. React. Chem. Eng. 2020. [Google Scholar] [CrossRef]
- Nguyen, L.N.; Kaushik, N.; Lamichhane, P.; Mumtaz, S.; Paneru, R.; Bhartiya, P.; Kwon, J.S.; Mishra, Y.K.; Nguyen, L.Q.; Kaushik, N.K.; et al. In situ plasma-assisted synthesis of polydopamine-functionalized gold nanoparticles for biomedical applications. Green Chem. 2020, 22, 6588–6599. [Google Scholar] [CrossRef]
- Kelly, S.; Turner, M.M. Atomic oxygen patterning from a biomedical needle-plasma source. J. Appl. Phys. 2013, 114, 123301. [Google Scholar] [CrossRef]
- Joshi, R.P.; Thagard, S.M. Streamer-like electrical discharges in water: Part I. Fundamental mechanisms. Plasma Chem. Plasma Process. 2013, 33, 1–15. [Google Scholar] [CrossRef]
- Graves, D.B. The emerging role of reactive oxygen and nitrogen species in redox biology and some implications for plasma applications to medicine and biology. J. Phys. D Appl. Phys. 2012, 45, 263001. [Google Scholar] [CrossRef]
- Gilmour, C.R. Water Treatment Using Advanced Oxidation Processes: Application Perspectives. Master’s Thesis, Western University, London, ON, Canada, 2012. [Google Scholar]
- Ganzenko, O.; Huguenot, D.; Van Hullebusch, E.D.; Esposito, G.; Oturan, M.A. Electrochemical advanced oxidation and biological processes for wastewater treatment: A review of the combined approaches. Environ. Sci. Pollut. Res. 2014, 21, 8493–8524. [Google Scholar] [CrossRef] [PubMed]
- Foster, J.; Sommers, B.S.; Gucker, S.N.; Blankson, I.M.; Adamovsky, G. Perspectives on the interaction of plasmas with liquid water for water purification. IEEE Trans. Plasma Sci. 2012, 40, 1311–1323. [Google Scholar] [CrossRef]
- Ghimire, B.; Lee, G.J.; Mumtaz, S.; Choi, E.H. Scavenging effects of ascorbic acid and mannitol on hydroxyl radicals generated inside water by an atmospheric pressure plasma jet. AIP Adv. 2018, 8, 075021. [Google Scholar] [CrossRef]
- Xu, H.; Ma, R.; Zhu, Y.; Du, M.; Zhang, H.; Jiao, Z. A systematic study of the antimicrobial mechanisms of cold atmospheric-pressure plasma for water disinfection. Sci. Total Environ. 2020, 703, 134965. [Google Scholar] [CrossRef]
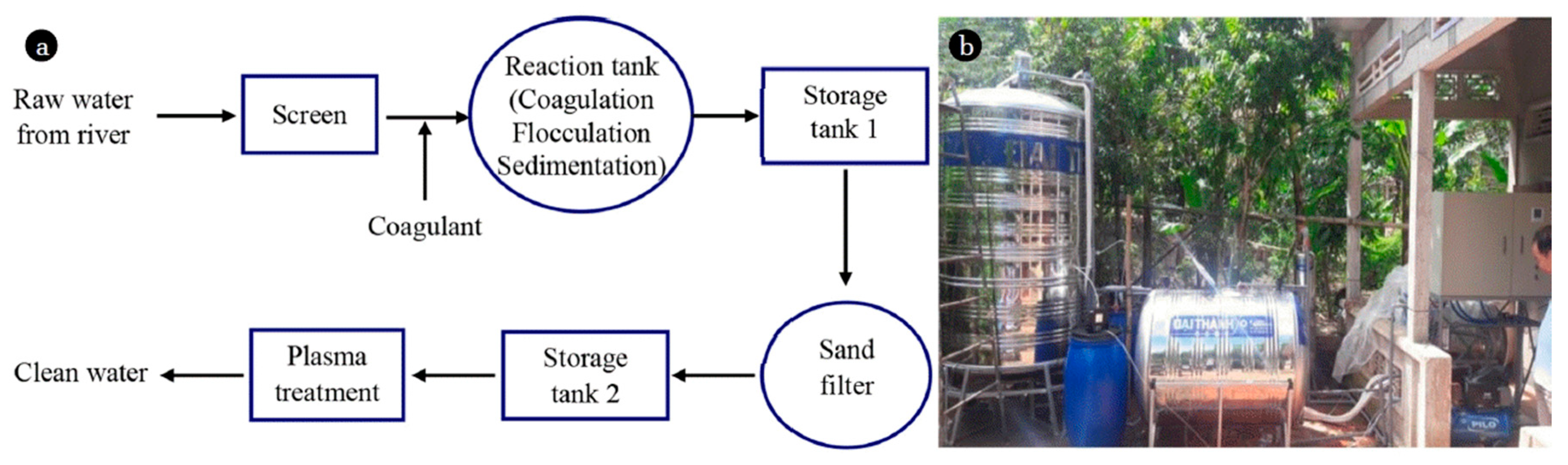
- Van Nguyen, D.; Ho, P.Q.; Van Pham, T.; Van Nguyen, T.; Kim, L. Treatment of surface water using cold plasma for domestic water supply. Environ. Eng. Res. 2019, 24, 412–417. [Google Scholar] [CrossRef]
- Mujovic, S. The Plasma Water Reactor: A Geometric Approach to Scaling Electric Discharges for Water Treatment. Ph.D. Thesis, University of Michigan, Ann Arbor, MI, USA, 2019. [Google Scholar]
- Ghimire, B.; Szili, E.J.; Lamichhane, P.; Short, R.D.; Lim, J.S.; Attri, P.; Masur, K.; Weltmann, K.-D.; Hong, S.-H.; Choi, E.H. The role of UV photolysis and molecular transport in the generation of reactive species in a tissue model with a cold atmospheric pressure plasma jet. Appl. Phys. Lett. 2019, 114, 093701. [Google Scholar] [CrossRef]
- Foster, J.E. Plasma-based water purification: Challenges and prospects for the future. Phys. Plasmas 2017, 24, 55501. [Google Scholar] [CrossRef]
- Horikoshi, S.; Serpone, N. In-liquid plasma: A novel tool in the fabrication of nanomaterials and in the treatment of wastewaters. RSC Adv. 2017, 7, 47196–47218. [Google Scholar] [CrossRef]
- Zuluaga-Gomeza, J.; Bonaverib, P.; Zuluagab, D.; Álvarez-Peñaa, C.; Ramirez-Ortiza, N. Techniques for water disinfection, decontamination and desalinization: A review. Desalin. WATER Treat. 2020, 181, 47–63. [Google Scholar] [CrossRef]
- Ma, S.; Kim, K.; Huh, J.; Hong, Y. Characteristics of microdischarge plasma jet in water and its application to water purification by bacterial inactivation. Sep. Purif. Technol. 2017, 188, 147–154. [Google Scholar] [CrossRef]
- Sakudo, A.; Yagyu, Y.; Onodera, T. Disinfection and Sterilization Using Plasma Technology: Fundamentals and Future Perspectives for Biological Applications. Int. J. Mol. Sci. 2019, 20, 5216. [Google Scholar] [CrossRef]
- Malik, M.A. Water purification by plasmas: Which reactors are most energy efficient? Plasma Chem. Plasma Process. 2010, 30, 21–31. [Google Scholar] [CrossRef]
- Iervolino, G.; Vaiano, V.; Palma, V. Enhanced removal of water pollutants by dielectric barrier discharge non-thermal plasma reactor. Sep. Purif. Technol. 2019, 215, 155–162. [Google Scholar] [CrossRef]
- Kogelschatz, U. Filamentary, patterned, and diffuse barrier discharges. IEEE Trans. Plasma Sci. 2002, 30, 1400–1408. [Google Scholar] [CrossRef]
- Kanazawa, S.; Kogoma, M.; Moriwaki, T.; Okazaki, S. Stable glow plasma at atmospheric pressure. J. Phys. D Appl. Phys. 1988, 21, 838. [Google Scholar] [CrossRef]
- Selwyn, G.S.; Herrmann, H.W.; Park, J.; Henins, I. Materials Processing Using an Atmospheric Pressure, RF-Generated Plasma Source. Contrib. Plasma Phys. 2001, 41, 610–619. [Google Scholar] [CrossRef]
- Paneru, R.; Lamichhane, P.; Chandra Adhikari, B.; Ki, S.H.; Choi, J.; Kwon, J.S.; Choi, E.H. Surface modification of PVA thin film by nonthermal atmospheric pressure plasma for antifogging property. AIP Adv. 2019, 9, 75008. [Google Scholar] [CrossRef]
- Paneru, R.; Ki, S.H.; Lamichhane, P.; Nguyen, L.N.; Adhikari, B.C.; Jeong, I.J.; Mumtaz, S.; Choi, J.; Kwon, J.S.; Choi, E.H. Enhancement of antibacterial and wettability performances of polyvinyl alcohol/chitosan film using non-thermal atmospheric pressure plasma. Appl. Surf. Sci. 2020, 532, 147339. [Google Scholar] [CrossRef]
- Lu, X.; Naidis, G.V.; Laroussi, M.; Ostrikov, K. Guided ionization waves: Theory and experiments. Phys. Rep. 2014, 540, 123–166. [Google Scholar] [CrossRef]
- Reuter, S.; Tresp, H.; Wende, K.; Hammer, M.U.; Winter, J.; Masur, K.; Schmidt-bleker, A.; Weltmann, K. From RONS to ROS: Tailoring Plasma Jet Treatment of Skin Cells. IEEE Trans. Plasma Sci. 2012, 40, 2986–2993. [Google Scholar] [CrossRef]
- Ito, T.; Uchida, G.; Nakajima, A.; Takenaka, K.; Setsuhara, Y. Control of reactive oxygen and nitrogen species production in liquid by nonthermal plasma jet with controlled surrounding gas. Jpn. J. Appl. Phys. 2016, 56, 01AC06. [Google Scholar] [CrossRef]
- Ghimire, B.; Sornsakdanuphap, J.; Hong, Y.J.; Uhm, H.S.; Weltmann, K.D.; Choi, E.H. The effect of the gap distance between an atmospheric-pressure plasma jet nozzle and liquid surface on OH and N2species concentrations. Phys. Plasmas 2017, 24. [Google Scholar] [CrossRef]
- Ghimire, B.; Szili, E.; Patenall, B.; Lamichhane, P.; Gaur, N.; Robson, A.; Trivedi, D.; Thet, N.T.; Jenkins, T.; Choi, E.H.; et al. Enhancement of hydrogen peroxide production from an atmospheric pressure argon plasma jet and implications to the antibacterial activity of plasma activated water. Plasma Sources Sci. Technol. 2021. [Google Scholar] [CrossRef]
- Yue, Y.; Xian, Y.; Pei, X.; Lu, X. The effect of three different methods of adding O2 additive on O concentration of atmospheric pressure plasma jets (APPJs). Phys. Plasmas 2016, 23, 123503. [Google Scholar] [CrossRef]
- Lim, J.S.; Kim, R.H.; Hong, Y.J.; Lamichhane, P.; Adhikari, B.C.; Choi, J.; Choi, E.H. Interactions between atmospheric pressure plasma jet and deionized water surface. Results Phys. 2020, 19, 103569. [Google Scholar] [CrossRef]
- Nguyen, T.S.; Rond, C.; Vega, A.; Duten, X.; Forget, S. Investigation of Hydrogen Peroxide Formation After Underwater Plasma Discharge. Plasma Chem. Plasma Process. 2020, 40, 955–969. [Google Scholar] [CrossRef]
- Lu, X.; Keidar, M.; Laroussi, M.; Choi, E.; Szili, E.J.; Ostrikov, K. Transcutaneous plasma stress: From soft-matter models to living tissues. Mater. Sci. Eng. R Rep. 2019, 138, 36–59. [Google Scholar] [CrossRef]
- Attri, P.; Kim, Y.H.; Park, D.H.; Park, J.H.; Hong, Y.J.; Uhm, H.S.; Kim, K.-N.; Fridman, A.; Choi, E.H. Generation mechanism of hydroxyl radical species and its lifetime prediction during the plasma-initiated ultraviolet (UV) photolysis. Sci. Rep. 2015, 5, 9332. [Google Scholar] [CrossRef] [PubMed]
- Kondeti, V.S.S.K.; Phan, C.Q.; Wende, K.; Jablonowski, H.; Gangal, U.; Granick, J.L.; Hunter, R.C.; Bruggeman, P.J. Long-lived and short-lived reactive species produced by a cold atmospheric pressure plasma jet for the inactivation of Pseudomonas aeruginosa and Staphylococcus aureus. Free Radic. Biol. Med. 2018, 124, 275–287. [Google Scholar] [CrossRef]
- Judée, F.; Simon, S.; Bailly, C.; Dufour, T. Plasma-activation of tap water using DBD for agronomy applications: Identification and quantification of long lifetime chemical species and production/consumption mechanisms. Water Res. 2018, 133, 47–59. [Google Scholar] [CrossRef] [PubMed]
- Conrads, H.; Schmidt, M. Plasma generation and plasma sources. Plasma Sources Sci. Technol. 2000, 9, 441. [Google Scholar] [CrossRef]
- Mumtaz, S.; Bhartiya, P.; Kaushik, N.; Adhikari, M.; Lamichhane, P.; Lee, S.-J.; Kumar Kaushik, N.; Ha Choi, E. Pulsed high-power microwaves do not impair the functions of skin normal and cancer cells in vitro: a short-term biological evaluation. J. Adv. Res. 2019. [Google Scholar] [CrossRef] [PubMed]
- Van Gaens, W.; Bogaerts, A. Kinetic modelling for an atmospheric pressure argon plasma jet in humid air. J. Phys. D Appl. Phys. 2013, 46, 275201. [Google Scholar] [CrossRef]
- Maheux, S.; Duday, D.; Belmonte, T.; Penny, C.; Cauchie, H.-M.; Clément, F.; Choquet, P. Formation of ammonium in saline solution treated by nanosecond pulsed cold atmospheric microplasma: A route to fast inactivation of E. coli bacteria. RSC Adv. 2015, 5, 42135–42140. [Google Scholar] [CrossRef]
- Gibalov, V.I.; Pietsch, G.J. Dynamics of dielectric barrier discharges in coplanar arrangements. J. Phys. D Appl. Phys. 2004, 37, 2082. [Google Scholar] [CrossRef]
- Takeuchi, N.; Yasuoka, K. Review of plasma-based water treatment technologies for the decomposition of persistent organic compounds. Jpn. J. Appl. Phys. 2020, 60, SA0801. [Google Scholar] [CrossRef]
- Kodama, S.; Matsumoto, S.; Wang, D.; Namihira, T.; Akiyama, H. Persistent Organic Pollutants Treatment in Wastewater Using Nanosecond Pulsed Non-Thermal Plasma. Int. J. Plasma Environ. Sci. Technol. 2018, 11, 138–143. [Google Scholar]
- Berger, A.; Ziv-Gal, A.; Cudiamat, J.; Wang, W.; Zhou, C.; Flaws, J.A. The effects of in utero bisphenol A exposure on the ovaries in multiple generations of mice. Reprod. Toxicol. 2016, 60, 39–52. [Google Scholar] [CrossRef] [PubMed]
- Morris, A. Reproductive endocrinology: Exposure to pesticide residues linked to adverse pregnancy outcomes. Nat. Rev. Endocrinol. 2018, 14, 4. [Google Scholar] [CrossRef]
- Suraj, M.; Anuradha, T. Plasma Based Water Purifier: Design and Testing of Prototype with Different Samples of Water. Preprints 2018. [Google Scholar] [CrossRef]
- Richmonds, C.; Witzke, M.; Bartling, B.; Lee, S.W.; Wainright, J.; Liu, C.-C.; Sankaran, R.M. Electron-transfer reactions at the plasma–liquid interface. J. Am. Chem. Soc. 2011, 133, 17582–17585. [Google Scholar] [CrossRef]
- Lee, S.W.; Zamani, H.; Feng, P.X.-L.; Mohan Sankaran, R. Extraction of a low-current discharge from a microplasma for nanoscale patterning applications at atmospheric pressure. J. Vac. Sci. Technol. B Nanotechnol. Microelectron. Mater. Process. Meas. Phenom. 2012, 30, 10603. [Google Scholar] [CrossRef]
- Ali, F.; Lestari, D.L.; Putri, M.D. Application of Ozone Plasma Technology for Treating Peat Water into Drinking Water. IOP Conf. Ser. Earth Environ. Sci. 2019, 385, 12056. [Google Scholar] [CrossRef]
- Kim, H.-S.; Wright, K.; Piccioni, J.; Cho, D.J.; Cho, Y.I. Inactivation of bacteria by the application of spark plasma in produced water. Sep. Purif. Technol. 2015, 156, 544–552. [Google Scholar] [CrossRef]
- Barillas, L. Design of a prototype of water purification by plasma technology as the foundation for an industrial wastewater plant. J. Phys. Conf. Ser. 2015, 591, 12057. [Google Scholar] [CrossRef]
- Onga, L.; Kornev, I.; Preis, S. Oxidation of reactive azo-dyes with pulsed corona discharge: Surface reaction enhancement. J. Electrostat. 2020, 103, 103420. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, D.; Wang, H.; Liu, L.; Wang, S.; Yang, S. Highly effective inactivation of Pseudomonas sp HB1 in water by atmospheric pressure microplasma jet array. Plasma Chem. Plasma Process. 2012, 32, 949–957. [Google Scholar] [CrossRef]
- Ziuzina, D.; Patil, S.; Cullen, P.J.; Keener, K.M.; Bourke, P. Atmospheric cold plasma inactivation of E scherichia coli in liquid media inside a sealed package. J. Appl. Microbiol. 2013, 114, 778–787. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.; Xu, Y.; Huang, H.; Zhang, Z.; Liu, Z.; Yan, K.; Zhu, A. Water disinfection by pulsed atmospheric air plasma along water surface. AIChE J. 2013, 59, 1458–1467. [Google Scholar] [CrossRef]
- Kehrer, J.P. The Haber--Weiss reaction and mechanisms of toxicity. Toxicology 2000, 149, 43–50. [Google Scholar] [CrossRef]
- Takai, E.; Ikawa, S.; Kitano, K.; Kuwabara, J.; Shiraki, K. Molecular mechanism of plasma sterilization in solution with the reduced pH method: Importance of permeation of HOO radicals into the cell membrane. J. Phys. D Appl. Phys. 2013, 46, 295402. [Google Scholar] [CrossRef]
- Oehmigen, K.; Hähnel, M.; Brandenburg, R.; Wilke, C.; Weltmann, K.-D.; Von Woedtke, T. The role of acidification for antimicrobial activity of atmospheric pressure plasma in liquids. Plasma Process. Polym. 2010, 7, 250–257. [Google Scholar] [CrossRef]
- Kaushik, N.K.; Ghimire, B.; Li, Y.; Adhikari, M.; Veerana, M.; Kaushik, N.; Jha, N.; Adhikari, B.; Lee, S.-J.; Masur, K.; et al. Biological and medical applications of plasma-activated media, water and solutions. Biol. Chem. 2018, 400, 39–62. [Google Scholar] [CrossRef]
- Ikawa, S.; Kitano, K.; Hamaguchi, S. Effects of pH on bacterial inactivation in aqueous solutions due to low-temperature atmospheric pressure plasma application. Plasma Process. Polym. 2010, 7, 33–42. [Google Scholar] [CrossRef]
- Rashmei, Z.; Bornasi, H.; Ghoranneviss, M. Evaluation of treatment and disinfection of water using cold atmospheric plasma. J. Water Health 2016, 14, 609–616. [Google Scholar] [CrossRef]
- Foster, J.E.; Mujovic, S.; Groele, J.; Blankson, I.M. Towards high throughput plasma based water purifiers: Design considerations and the pathway towards practical application. J. Phys. D Appl. Phys. 2018, 51, 293001. [Google Scholar] [CrossRef]
- Misra, N.N.; Jo, C. Applications of cold plasma technology for microbiological safety in meat industry. Trends Food Sci. Technol. 2017, 64, 74–86. [Google Scholar] [CrossRef]
- Hijosa-Valsero, M.; Molina, R.; Bayona, J.M. Assessment of a dielectric barrier discharge plasma reactor at atmospheric pressure for the removal of bisphenol A and tributyltin. Environ. Technol. 2014, 35, 1418–1426. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Bai, Y.; Yu, H.; Zhang, C.; Chen, J. Degradation of selected organophosphate pesticides in wastewater by dielectric barrier discharge plasma. Bull. Environ. Contam. Toxicol. 2013, 91, 314–319. [Google Scholar] [CrossRef]
- Xu, L.; Li, H.; Rashid, S.; Shen, C.; Wen, Y.; He, T. Treatment of saline dye wastewater using glow discharge plasma. FEB-FRESENIUS Environ. Bull. 2016, 82, 2466. [Google Scholar]
- Petrovic, M.; Barceló, D. LC-MS for identifying photodegradation products of pharmaceuticals in the environment. TrAC Trends Anal. Chem. 2007, 26, 486–493. [Google Scholar] [CrossRef]
- Loraine, G.A.; Pettigrove, M.E. Seasonal variations in concentrations of pharmaceuticals and personal care products in drinking water and reclaimed wastewater in southern California. Environ. Sci. Technol. 2006, 40, 687–695. [Google Scholar] [CrossRef]
- Goldstein, A.H.; Galbally, I.E. Known and Unexplored Organic Constituents in the Earth’s Atmosphere; ACS Publications: Washington, DC, USA, 2007. [Google Scholar]
- Abdullahi, M.E.; Abu Hassan, M.A.; Zainon Noor, Z.; Raja Ibrahim, R.K. Integrated air stripping and non-thermal plasma system for the treatment of volatile organic compounds from wastewater: Statistical optimization. Desalin. Water Treat. 2016, 57, 16066–16077. [Google Scholar] [CrossRef]
- Lukes, P.; Locke, B.R.; Brisset, J.-L. Aqueous-phase chemistry of electrical discharge plasma in water and in gas-liquid environments. Plasma Chem. Catal. Gases Liq. 2012, 1, 243–308. [Google Scholar]
- Banaschik, R.; Lukes, P.; Jablonowski, H.; Hammer, M.U.; Weltmann, K.-D.; Kolb, J.F. Potential of pulsed corona discharges generated in water for the degradation of persistent pharmaceutical residues. Water Res. 2015, 84, 127–135. [Google Scholar] [CrossRef]
- Magureanu, M.; Mandache, N.B.; Parvulescu, V.I. Degradation of pharmaceutical compounds in water by non-thermal plasma treatment. Water Res. 2015, 81, 124–136. [Google Scholar] [CrossRef]
- Aziz, K.H.H.; Miessner, H.; Mueller, S.; Kalass, D.; Moeller, D.; Khorshid, I.; Rashid, M.A.M. Degradation of pharmaceutical diclofenac and ibuprofen in aqueous solution, a direct comparison of ozonation, photocatalysis, and non-thermal plasma. Chem. Eng. J. 2017, 313, 1033–1041. [Google Scholar] [CrossRef]
- Zhou, R.; Zhou, R.; Zhuang, J.; Zong, Z.; Zhang, X.; Liu, D.; Bazaka, K.; Ostrikov, K. Interaction of atmospheric-pressure air microplasmas with amino acids as fundamental processes in aqueous solution. PLoS ONE 2016, 11, e0155584. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Qu, G.; Zhang, L.; Wang, T.; Sun, Q.; Liang, D.; Hu, S. Humic acid removal from micro-polluted source water using gas phase surface discharge plasma at different grounding modes. Sep. Purif. Technol. 2017, 180, 36–43. [Google Scholar] [CrossRef]
- Zhou, R.; Zhou, R.; Yu, F.; Xi, D.; Wang, P.; Li, J.; Wang, X.; Zhang, X.; Bazaka, K.; Ostrikov, K.K. Removal of organophosphorus pesticide residues from Lycium barbarum by gas phase surface discharge plasma. Chem. Eng. J. 2018, 342, 401–409. [Google Scholar] [CrossRef]
- Qi, Z.H.; Yang, L.; Xia, Y.; Ding, Z.F.; Niu, J.H.; Liu, D.P.; Zhao, Y.; Ji, L.F.; Song, Y.; Lin, X.S. Removal of dimethyl phthalate in water by non-thermal air plasma treatment. Environ. Sci. Water Res. Technol. 2019, 5, 920–930. [Google Scholar] [CrossRef]
- Gulkowska, A.; He, Y.; So, M.K.; Yeung, L.W.Y.; Leung, H.W.; Giesy, J.P.; Lam, P.K.S.; Martin, M.; Richardson, B.J. The occurrence of selected antibiotics in Hong Kong coastal waters. Mar. Pollut. Bull. 2007, 54, 1287–1293. [Google Scholar] [CrossRef]
- Lindsey, M.E.; Meyer, M.; Thurman, E.M. Analysis of trace levels of sulfonamide and tetracycline antimicrobials in groundwater and surface water using solid-phase extraction and liquid chromatography/mass spectrometry. Anal. Chem. 2001, 73, 4640–4646. [Google Scholar] [CrossRef]
- Kolpin, D.W.; Furlong, E.T.; Meyer, M.T.; Thurman, E.M.; Zaugg, S.D.; Barber, L.B.; Buxton, H.T. Pharmaceuticals, hormones, and other organic wastewater contaminants in US streams, 1999–2000: A national reconnaissance. Environ. Sci. Technol. 2002, 36, 1202–1211. [Google Scholar] [CrossRef]
- Of, E.; Gestagens, S.; Fish, O.N. Pharmaceuticals and personal care products in the environment. Environ. Toxicol. 2009, 28, 2663–2670. [Google Scholar]
- Rong, S.; Sun, Y. Wetted-wall corona discharge induced degradation of sulfadiazine antibiotics in aqueous solution. J. Chem. Technol. Biotechnol. 2014, 89, 1351–1359. [Google Scholar] [CrossRef]
- He, D.; Sun, Y.; Xin, L.; Feng, J. Aqueous tetracycline degradation by non-thermal plasma combined with nano-TiO2. Chem. Eng. J. 2014, 258, 18–25. [Google Scholar] [CrossRef]
- Gerrity, D.; Stanford, B.D.; Trenholm, R.A.; Snyder, S.A. An evaluation of a pilot-scale nonthermal plasma advanced oxidation process for trace organic compound degradation. Water Res. 2010, 44, 493–504. [Google Scholar] [CrossRef] [PubMed]
- Krishna, S.; Ceriani, E.; Marotta, E.; Giardina, A.; Špatenka, P.; Paradisi, C. Products and mechanism of verapamil removal in water by air non-thermal plasma treatment. Chem. Eng. J. 2016, 292, 35–41. [Google Scholar] [CrossRef]
- Magureanu, M.; Dobrin, D.; Mandache, N.B.; Bradu, C.; Medvedovici, A.; Parvulescu, V.I. The mechanism of plasma destruction of enalapril and related metabolites in water. Plasma Process. Polym. 2013, 10, 459–468. [Google Scholar] [CrossRef]
- Khetan, S.K.; Collins, T.J. Human pharmaceuticals in the aquatic environment: A challenge to green chemistry. Chem. Rev. 2007, 107, 2319–2364. [Google Scholar] [CrossRef]
- Liu, Y.; Mei, S.; Iya-Sou, D.; Cavadias, S.; Ognier, S. Carbamazepine removal from water by dielectric barrier discharge: Comparison of ex situ and in situ discharge on water. Chem. Eng. Process. Process Intensif. 2012, 56, 10–18. [Google Scholar] [CrossRef]
- Magureanu, M.; Mandache, N.B.; Bradu, C.; Parvulescu, V.I. High efficiency plasma treatment of water contaminated with organic compounds. Study of the degradation of ibuprofen. Plasma Process. Polym. 2018, 15, 1700201. [Google Scholar] [CrossRef]
- Back, J.O.; Obholzer, T.; Winkler, K.; Jabornig, S.; Rupprich, M. Combining ultrafiltration and non-thermal plasma for low energy degradation of pharmaceuticals from conventionally treated wastewater. J. Environ. Chem. Eng. 2018, 6, 7377–7385. [Google Scholar] [CrossRef]

| Water Disinfection Method | Advantages | Disadvantages | Refs. |
|---|---|---|---|
| Chlorination | Effective and inexpensive | Reaction with organic materials in water and generation of carcinogenic materials | [9,10,11] |
| Ozonation | Minimal odor and taste, reduced formation of by-products, oxidation of organic contaminants | Expensive | [9,12] |
| UV radiation | Protection against microbes and chemicals, no residual effects | Low penetration power, loss of power | [9] |
| Filtration | Provision of immediate access to drinking water without adding an unpleasant taste | Expensive, not suitable for removing virus particles | [13] |
| Boiling | Provision of drinkable water for at least 3 min | Time consuming, expensive | [14] |
| AOP | Eco-friendly, no need to add chemical agents | Expensive | [19] |
| Plasma Source | Chemical Pollutants | Refs. |
|---|---|---|
| DBD reactor | Bisphenol A and tributyltin | [98] |
| DBD device | Organophosphate pesticide(dimethoate and dichlorvos) | [99] |
| Glow discharge | Saline and azo dye (AR 73) | [100] |
| Combination of air stripping and NTAPP | M-xylene and toluene | [104] |
| Microplasma-based AOP process | DMP removal | [112] |
| DBD reactor | Verapamil oxidation | [120] |
| Low-power ex situ plasma treatment | carbamazepine | [123] |
| Pulsed corona discharge NTAPP | Ibuprofen degradation | [124] |
| Combination of ultrafiltration and an NTAPP system | Slurry wastewater treatment | [125] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barjasteh, A.; Dehghani, Z.; Lamichhane, P.; Kaushik, N.; Choi, E.H.; Kaushik, N.K. Recent Progress in Applications of Non-Thermal Plasma for Water Purification, Bio-Sterilization, and Decontamination. Appl. Sci. 2021, 11, 3372. https://doi.org/10.3390/app11083372
Barjasteh A, Dehghani Z, Lamichhane P, Kaushik N, Choi EH, Kaushik NK. Recent Progress in Applications of Non-Thermal Plasma for Water Purification, Bio-Sterilization, and Decontamination. Applied Sciences. 2021; 11(8):3372. https://doi.org/10.3390/app11083372
Chicago/Turabian StyleBarjasteh, Azadeh, Zohreh Dehghani, Pradeep Lamichhane, Neha Kaushik, Eun Ha Choi, and Nagendra Kumar Kaushik. 2021. "Recent Progress in Applications of Non-Thermal Plasma for Water Purification, Bio-Sterilization, and Decontamination" Applied Sciences 11, no. 8: 3372. https://doi.org/10.3390/app11083372
APA StyleBarjasteh, A., Dehghani, Z., Lamichhane, P., Kaushik, N., Choi, E. H., & Kaushik, N. K. (2021). Recent Progress in Applications of Non-Thermal Plasma for Water Purification, Bio-Sterilization, and Decontamination. Applied Sciences, 11(8), 3372. https://doi.org/10.3390/app11083372









