Effect of Chronic Exposure to Pesticide Methomyl on Antioxidant Defense System in Testis of Tilapia (Oreochromis niloticus) and Its Recovery Pattern
Abstract
1. Introduction
2. Materials and Methods
2.1. Fish and Chemicals
2.2. Experimental Design
2.3. Biochemical Analysis
2.4. Statistical Analysis
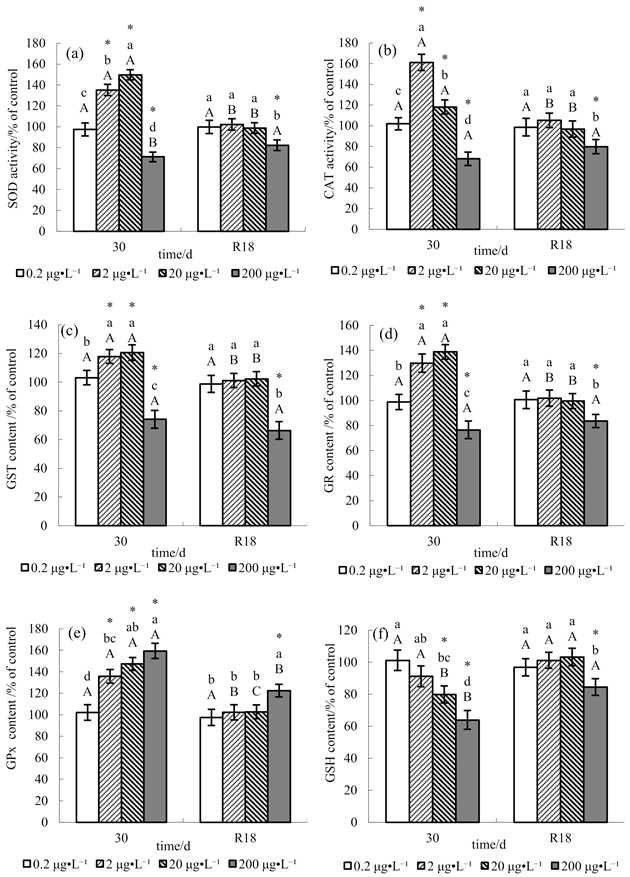
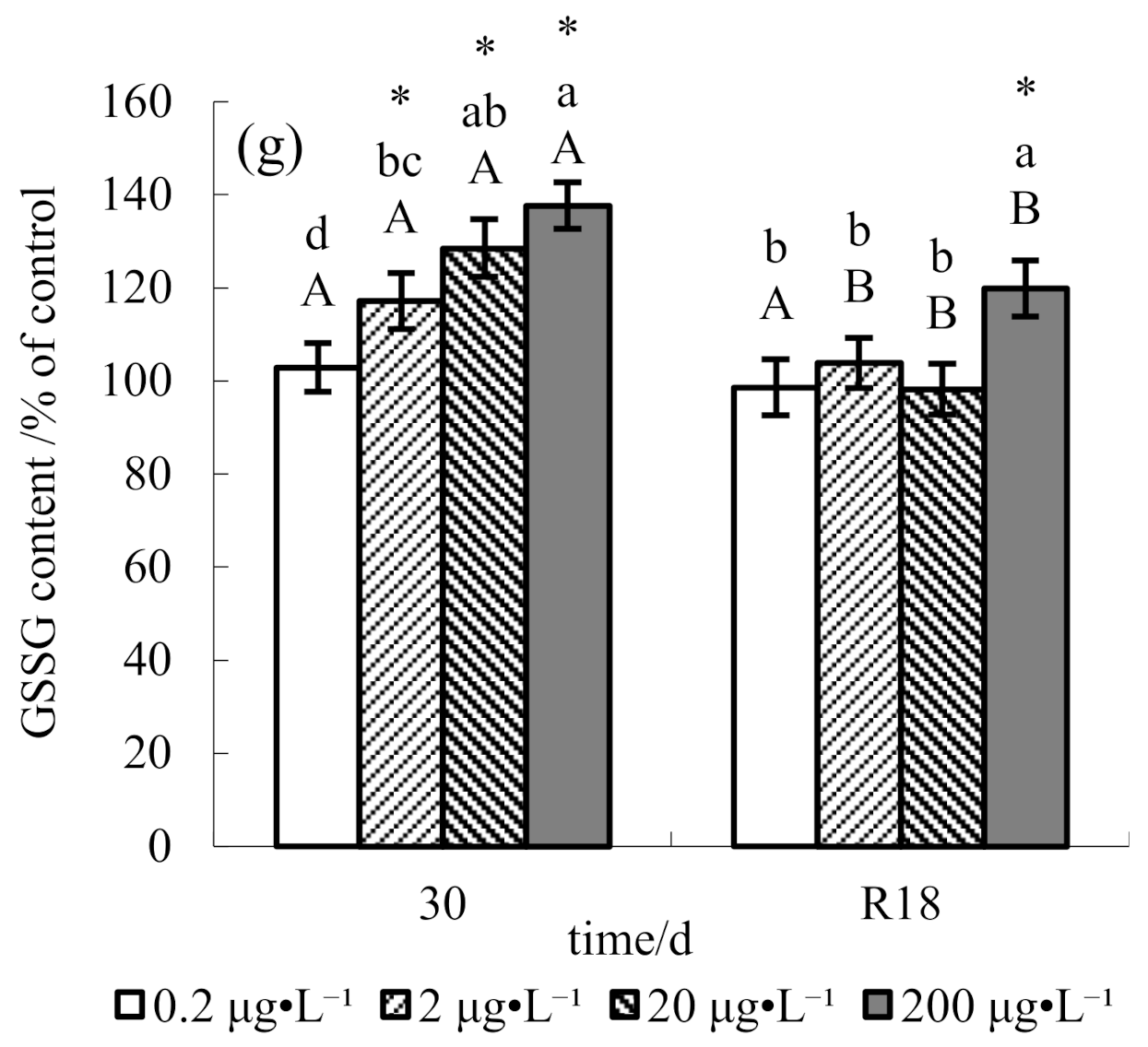
3. Results
4. Discussion
4.1. Effect of Methomyl on Antioxidant Defenses in Tilapia Testis
4.2. Recovery Pattern
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kongphonprom, K.; Burakham, R. Determination of carbamate insecticides in water, fruit, and vegetables by ultrasound-assisted dispersive liquid-liquid micro extraction and high-performance liquid chromatography. Anal. Lett. 2016, 49, 753–767. [Google Scholar] [CrossRef]
- Van Scoy, A.R.; Yue, M.; Deng, X.; Tjeerdema, R.S. Environmental fate and toxicology of methomyl. Rev. Environ. Contam. Toxicol. 2013, 222, 93–109. [Google Scholar] [PubMed]
- Guo, X.B. Environmental Health; Peking University Medical Press: Beijing, China, 2006. [Google Scholar]
- Meng, S.L.; Hu, G.D.; Qiu, L.P.; Song, C.; Fan, L.M.; Chen, J.Z.; Xu, P. Effects of chronic exposure of methomyl on the antioxidant system in kidney of tilapia (Oreochromis niloticus) and recovery pattern. J. Toxicol. Environ. Health Part A 2013, 76, 937–943. [Google Scholar] [CrossRef] [PubMed]
- Meng, S.L.; Qiu, L.P.; Hu, G.D.; Fan, L.M.; Song, C.; Zheng, Y.; Wu, W.; Qu, J.H.; Li, D.D.; Chen, J.Z.; et al. Effects of methomyl on steroidogenic gene transcription of the hypothalamic-pituitary-gonad-liver axis in male tilapia. Chemosphere 2016, 165, 152–162. [Google Scholar] [CrossRef]
- Meng, S.L.; Qiu, L.P.; Hu, G.D.; Fan, L.M.; Song, C.; Zheng, Y.; Wu, W.; Qu, J.H.; Li, D.D.; Chen, J.Z.; et al. Effect of methomyl on sex steroid hormone and vitellogenin levels in serum of male tilapia (Oreochromis niloticus) and recovery pattern. Environ. Toxicol. 2017, 32, 1869–1877. [Google Scholar] [CrossRef]
- Zhang, J.; Shen, H.; Wang, X.; Wu, J.C.; Xue, Y.Q. Effects of chronic exposure of 2,4-dichlorophenol on the antioxidant system in liver of freshwater fish Carassius auratus. Chemosphere 2004, 55, 167–174. [Google Scholar] [CrossRef]
- Livingstone, D.R. Contaminant-stimulated reactive oxygen species production and oxidative damage in aquatic organisms. Mar. Pollut. Bull. 2001, 42, 656–666. [Google Scholar] [CrossRef]
- Stara, A.; Kristan, J.; Zuskova, E. Effect of chronic exposure to prometryne on oxidative stress and antioxidant response in common carp (Cyprinus carpio L.). Pestic. Biochem. Phys. 2013, 105, 18–23. [Google Scholar] [CrossRef]
- Van der Oost, R.; Beyer, J.; Vermeulen, N.P.E. Fish bioaccumulation and biomarkers in environmental risk assessment: A review. Environ. Toxicol. Pharmacol. 2003, 13, 57–149. [Google Scholar] [CrossRef]
- Meng, S.L.; Chen, J.Z.; Hu, G.D.; Song, C.; Fan, L.M.; Qiu, L.P.; Xu, P. Effects of chronic exposure of methomyl on the antioxidant system in liver of Nile tilapia (Oreochromis niloticus). Ecotoxicol. Environ. Saf. 2014, 101, 1–6. [Google Scholar] [CrossRef]
- Meng, S.L.; Liu, T.; Song, C.; Zhang, C.; Qiu, L.P.; Chen, J.Z.; Xu, P. Determination of Methomyl Residue in Water by Ultra Performance Liquid Chromatography-Tandem Mass Spectrometry. J. Anhui Agric. Sci. 2018, 46, 166–167. [Google Scholar]
- Hafeman, D.G.; Sunde, R.A.; Hoekstra, W.G. Effect of dietary selenium on erythrocyte and liver glutathione peroxidase in the rat. J. Nutr. 1974, 104, 580–587. [Google Scholar] [CrossRef]
- Xu, J.B.; Yuan, X.F.; Lang, P.Z. Determination of catalase activity and catalase inhibition by ultraviolet spectrophotometry. Chin. Environ. Chem. 1997, 16, 73–76. [Google Scholar]
- Habig, W.H.; Pabst, M.J.; Jakoby, W.B. Glutathione S-transferases. J. Biol. Chem. 1974, 249, 7130–7139. [Google Scholar] [CrossRef]
- Carlberg, I.; Mannervik, B. Purification and characterization of flavoenzyme glutathione reductase from rat liver. J. Biol. Chem. 1975, 250, 5475–5480. [Google Scholar] [CrossRef]
- Marklund, S.; Marklund, G. Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur. J. Biochem. 1974, 47, 469–474. [Google Scholar] [CrossRef]
- Hissin, P.J.; Hil, R. A fluorometric method for determination of oxidized and reduced glutathione in tissues. Anal. Biochem. 1976, 74, 214–226. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Hidalgo, M.C.; Expósito, A.; Palma, J.M.; Manuel, D.L. Oxidative stress generated by dietary Zn-deficiency: Studies in rainbow trout (Oncorhynchus mykiss). Int. J. Biochem. Cell Biol. 2002, 34, 183–193. [Google Scholar] [CrossRef]
- Luo, Y.; Su, Y.; Lin, R.; Shi, H.H.; Wang, X.R. 2-Chlorophenol induced ROS generation in fish Carassius auratus based on the EPR method. Chemosphere 2006, 65, 1064–1073. [Google Scholar] [CrossRef]
- Li, Z.H.; Zlabek, V.; Velisek, J.; Roman, G.; Jana, M.; Tomas, R. Modulation of antioxidant defence system in brain of rainbow trout (Oncorhynchus mykiss) after chronic carbamazepine treatment. Comp. Biochem. Phys. C 2010, 151, 137–141. [Google Scholar] [CrossRef]
- Li, Z.H.; Zlabek, V.; Grabic, R.; Li, P.; Randak, T. Modulation of glutathione-related antioxidant defense system of fish chronically treated by the fungicide propiconazole. Comp. Biochem. Phys. C 2010, 152, 392–398. [Google Scholar] [CrossRef]
- Oruc, E.O.; Sevgiler, Y.; Uner, N. Tissue-specific oxidative stress responses in fish exposed to 2,4-D and azinphosmethyl. Comp. Biochem. Physiol. C 2004, 137, 43–51. [Google Scholar]
- Mazmancı, B.; Çavaş, T. Antioxidant enzyme activity and lipid peroxidation in liver and gill tissues of Nile tilapia (Oreochromis niloticus) following in vivo exposure to domoic acid. Toxicon 2010, 55, 734–738. [Google Scholar] [CrossRef] [PubMed]
- Peixoto, F.; Alves-Fernandes, D.; Santos, D.; Fontaínhas-Fernandes, A. Toxicological effects of oxyfluorfen on oxidative stress enzymes in tilapia Oreochromis niloticus. Pestic. Biochem. Phys. 2006, 85, 91–96. [Google Scholar] [CrossRef]
- Nwani, C.D.; Lakra, W.S.; Nagpure, N.S.; Kumar, R.; Kushwaha, B.; Srivastava, S.K. Toxicity of the herbicide atrazine: Effects on lipid peroxidation and activities of antioxidant enzymes in the freshwater fish Channa punctatus (Bloch). Int. J. Environ. Res. Public Health 2010, 7, 3298–3312. [Google Scholar] [CrossRef] [PubMed]
- Meng, S.L.; Chen, J.Z.; Xu, P.; Qu, J.H.; Fan, L.M.; Song, C.; Qiu, L.P. Hepatic Antioxidant Enzymes SOD and CAT of Nile Tilapia (Oreochromis niloticus) in Response to Pesticide Methomyl and Recovery Pattern. Bull. Environ. Contam. Toxicol. 2014, 92, 388–392. [Google Scholar] [CrossRef] [PubMed]
- Oruç, E.Ö.; Üner, N. Combined effects of 2,4-D and azinphosmethyl on antioxidant enzymes and lipid peroxidation in liver of Oreochromis niloticus. Comp. Biochem. Phys. C 2000, 127, 291–296. [Google Scholar] [CrossRef]
- Misra, S.; Niyogi, S. Selenite causes cytotoxicity in rainbow trout (Oncorhynchus mykiss) hepatocytes by inducing oxidative stress. Toxicol. In Vitro 2009, 23, 1249–1258. [Google Scholar] [CrossRef]
- Li, H.; Jiang, H.; Gao, X.; Wang, X.; Qu, W.; Lin, R.; Chen, J. Acute toxicity of the pesticide methomyl on the topmouth gudgeon (Pseudorasbora parva): Mortality and effects on four biomarkers. Fish Physiol. Biochem. 2008, 34, 209–216. [Google Scholar] [CrossRef]
- Puerto, M.; Pichardo, S.; Jos, Á.; Prieto, A.I.; Sevilla, E.; Frías, J.E.; Cameán, A.M. Differential oxidative stress responses to pure Microcystin-LR and Microcystin-containing and non-containing cyanobacterial crude extracts on Caco-2 cells. Toxicon 2010, 55, 514–522. [Google Scholar] [CrossRef]
- Meng, S.L.; Liu, T.; Chen, X.; Qiu, L.P.; Hu, G.D.; Song, C.; Fan, L.M.; Zheng, Y.; Chen, J.Z.; Xu, P. Effect of Chronic Exposure to Methomyl on Tissue Damage and Apoptosis in Testis of Tilapia (Oreochromis niloticus) and Recovery Pattern. Bull. Environ. Contam. Toxicol. 2019, 102, 371–376. [Google Scholar] [CrossRef]
- Meng, S.L.; Qu, J.H.; Song, C.; Fan, L.M.; Qiu, L.P.; Chen, J.Z.; Xu, P. Joint toxicity of pesticides methomyl and phoxim to tilapia (oreochromis niloticus). J. Agro-Environ. Sci. 2014, 33, 257–263. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meng, S.; Chen, X.; Song, C.; Fan, L.; Qiu, L.; Zheng, Y.; Chen, J.; Xu, P. Effect of Chronic Exposure to Pesticide Methomyl on Antioxidant Defense System in Testis of Tilapia (Oreochromis niloticus) and Its Recovery Pattern. Appl. Sci. 2021, 11, 3332. https://doi.org/10.3390/app11083332
Meng S, Chen X, Song C, Fan L, Qiu L, Zheng Y, Chen J, Xu P. Effect of Chronic Exposure to Pesticide Methomyl on Antioxidant Defense System in Testis of Tilapia (Oreochromis niloticus) and Its Recovery Pattern. Applied Sciences. 2021; 11(8):3332. https://doi.org/10.3390/app11083332
Chicago/Turabian StyleMeng, Shunlong, Xi Chen, Chao Song, Limin Fan, Liping Qiu, Yao Zheng, Jiazhang Chen, and Pao Xu. 2021. "Effect of Chronic Exposure to Pesticide Methomyl on Antioxidant Defense System in Testis of Tilapia (Oreochromis niloticus) and Its Recovery Pattern" Applied Sciences 11, no. 8: 3332. https://doi.org/10.3390/app11083332
APA StyleMeng, S., Chen, X., Song, C., Fan, L., Qiu, L., Zheng, Y., Chen, J., & Xu, P. (2021). Effect of Chronic Exposure to Pesticide Methomyl on Antioxidant Defense System in Testis of Tilapia (Oreochromis niloticus) and Its Recovery Pattern. Applied Sciences, 11(8), 3332. https://doi.org/10.3390/app11083332






