Current Progress in Femtosecond Laser Ablation/Ionisation Time-of-Flight Mass Spectrometry
Abstract
1. Introduction
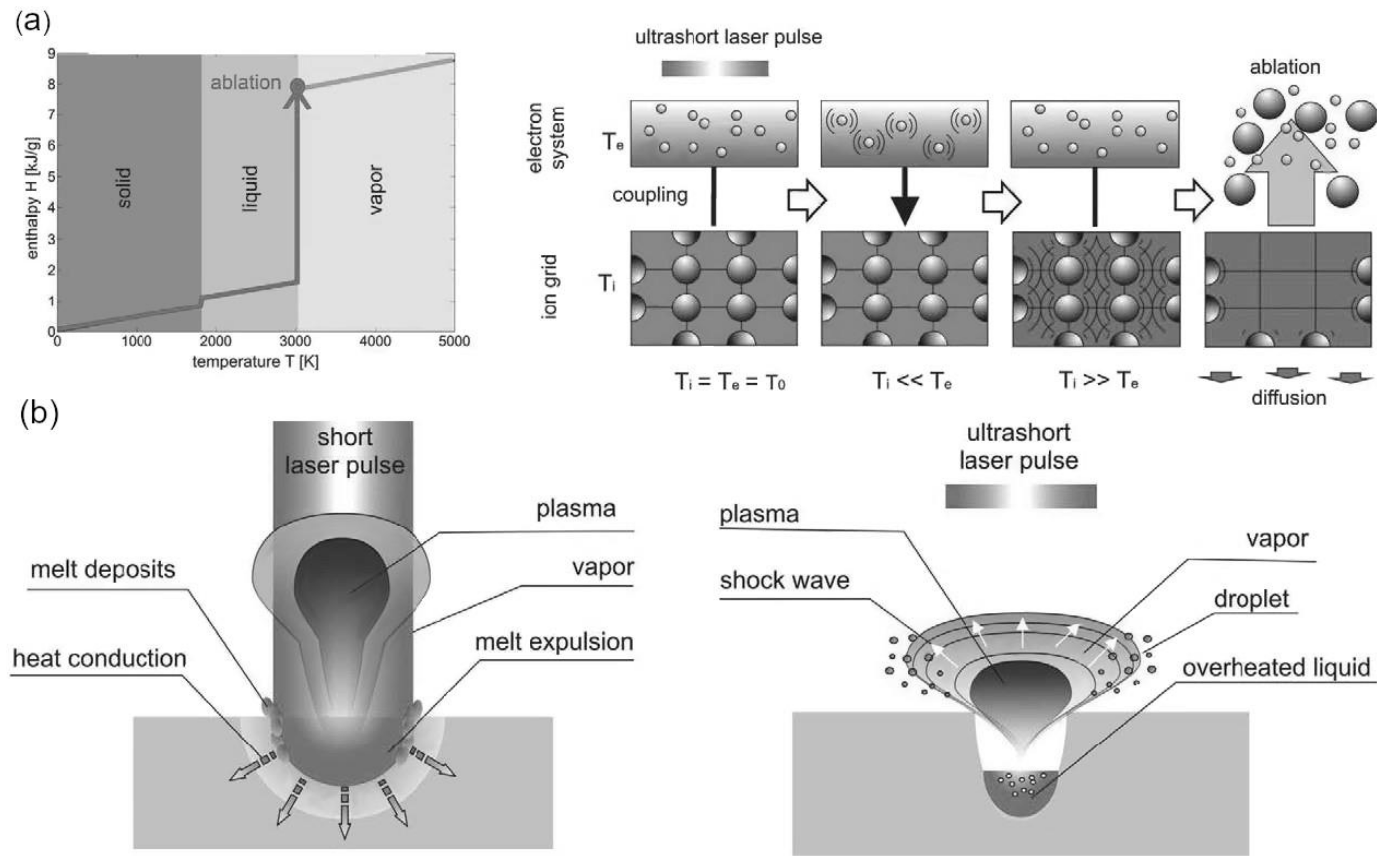
2. Laser-Matter Interaction in the Femtosecond Regime
2.1. Laser Ablation and Plasma Formation
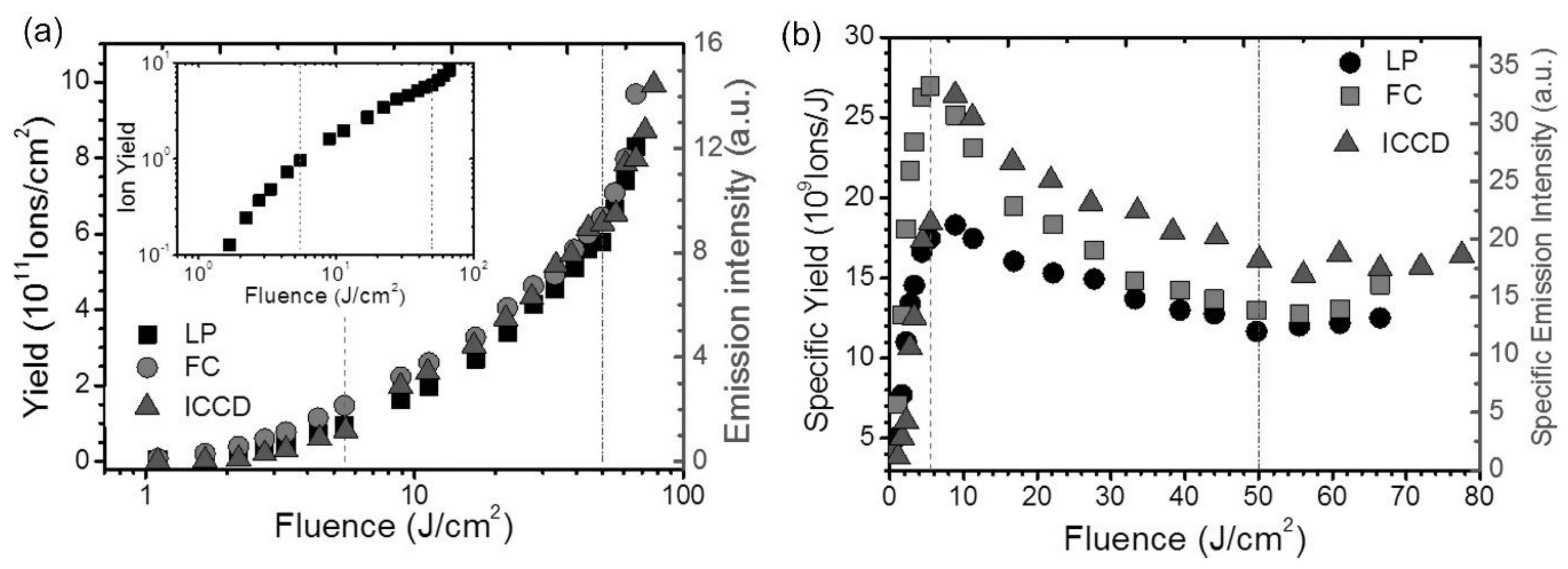
2.2. Diagnostics of the Ion Formation in fs-Laser Ablation
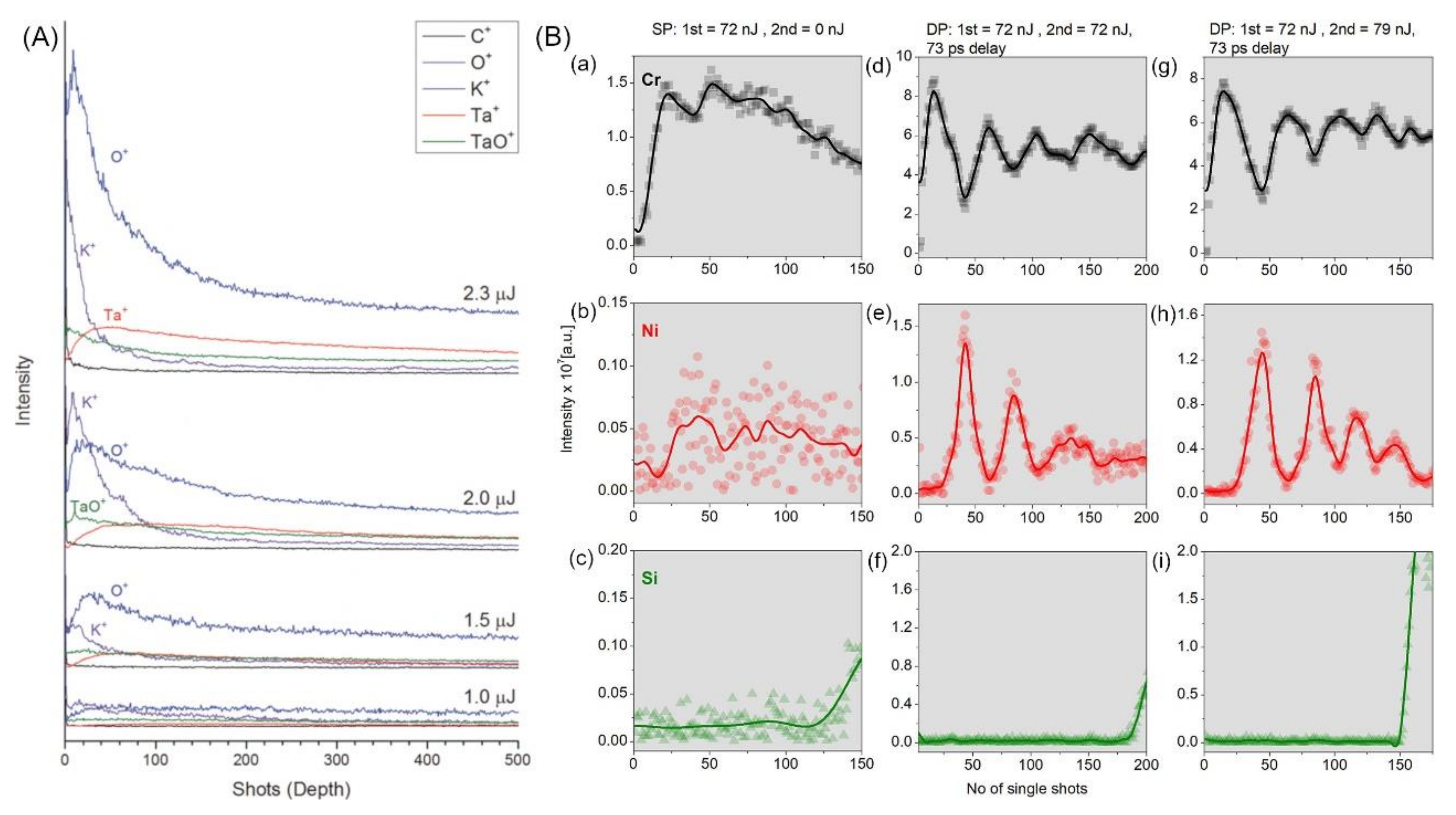
2.3. Double Pulse fs-Laser Ablation and Ion Yields
3. LIMS System: fs-Laser Ablation Ion Source and Time-of-Flight Mass Analyser
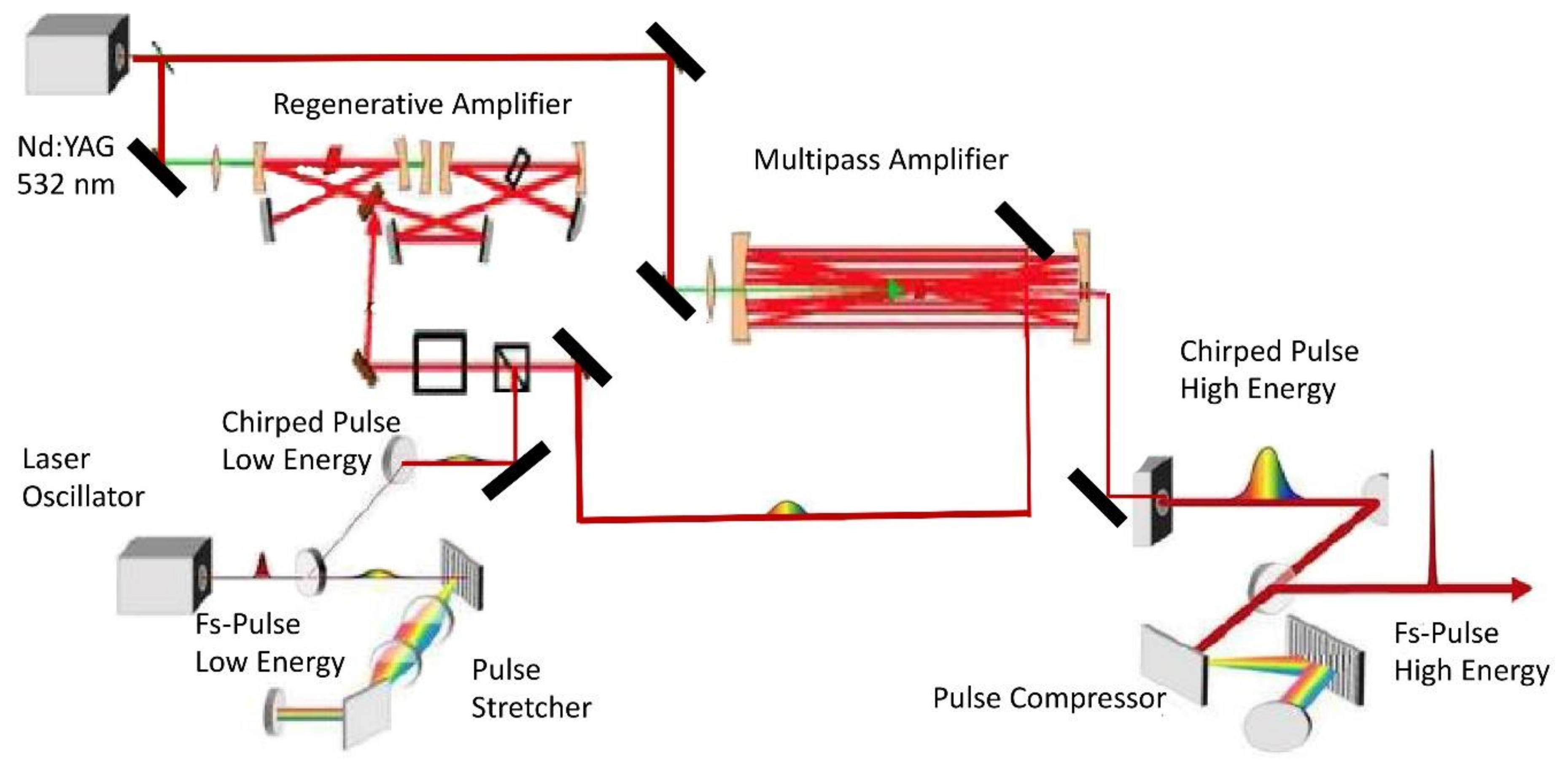
3.1. Femtosecond Laser Radiation
3.1.1. Laser Pulse Shaping
3.1.2. Generation of fs-Laser Radiation with Shorter Wavelength
3.1.3. Femtosecond Laser Optics
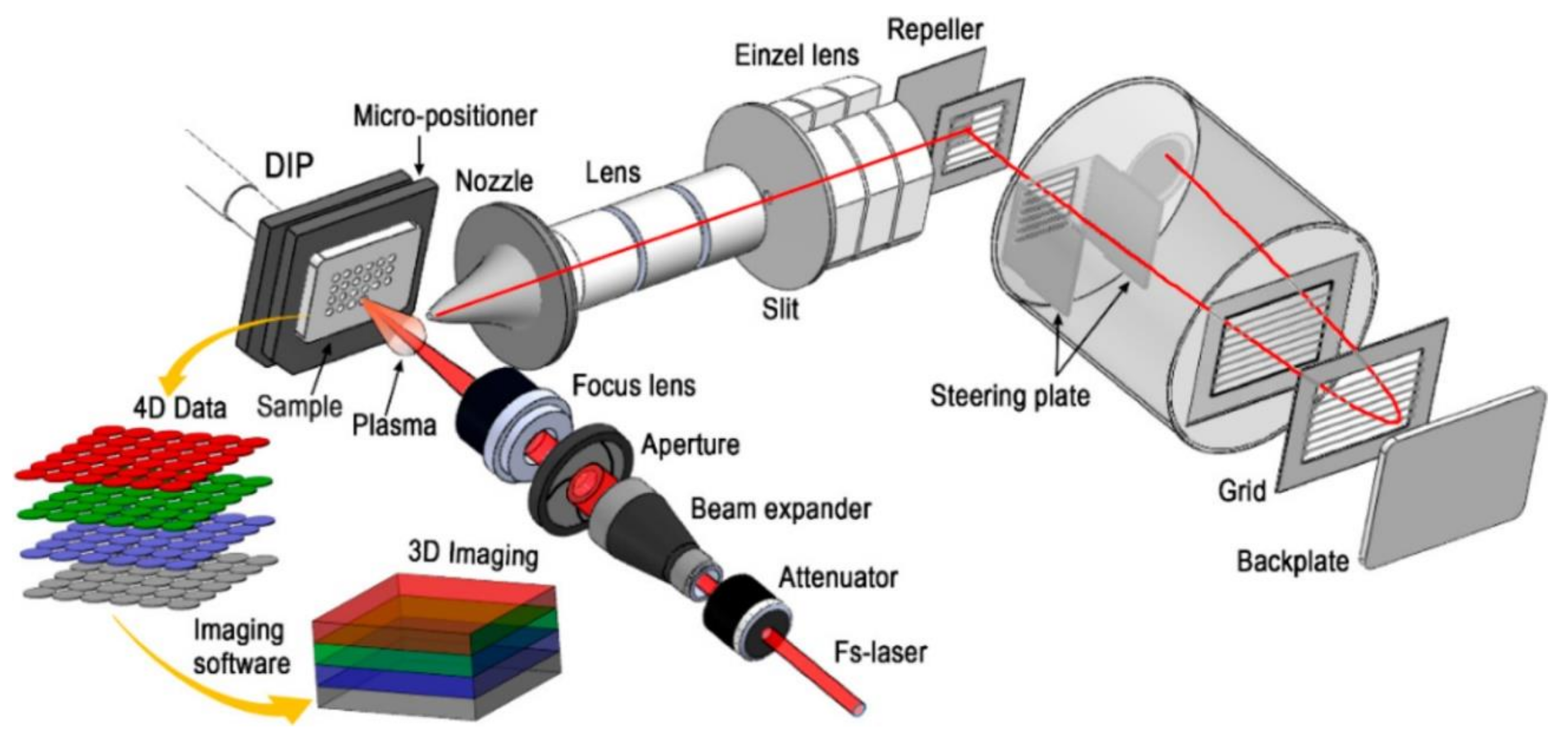
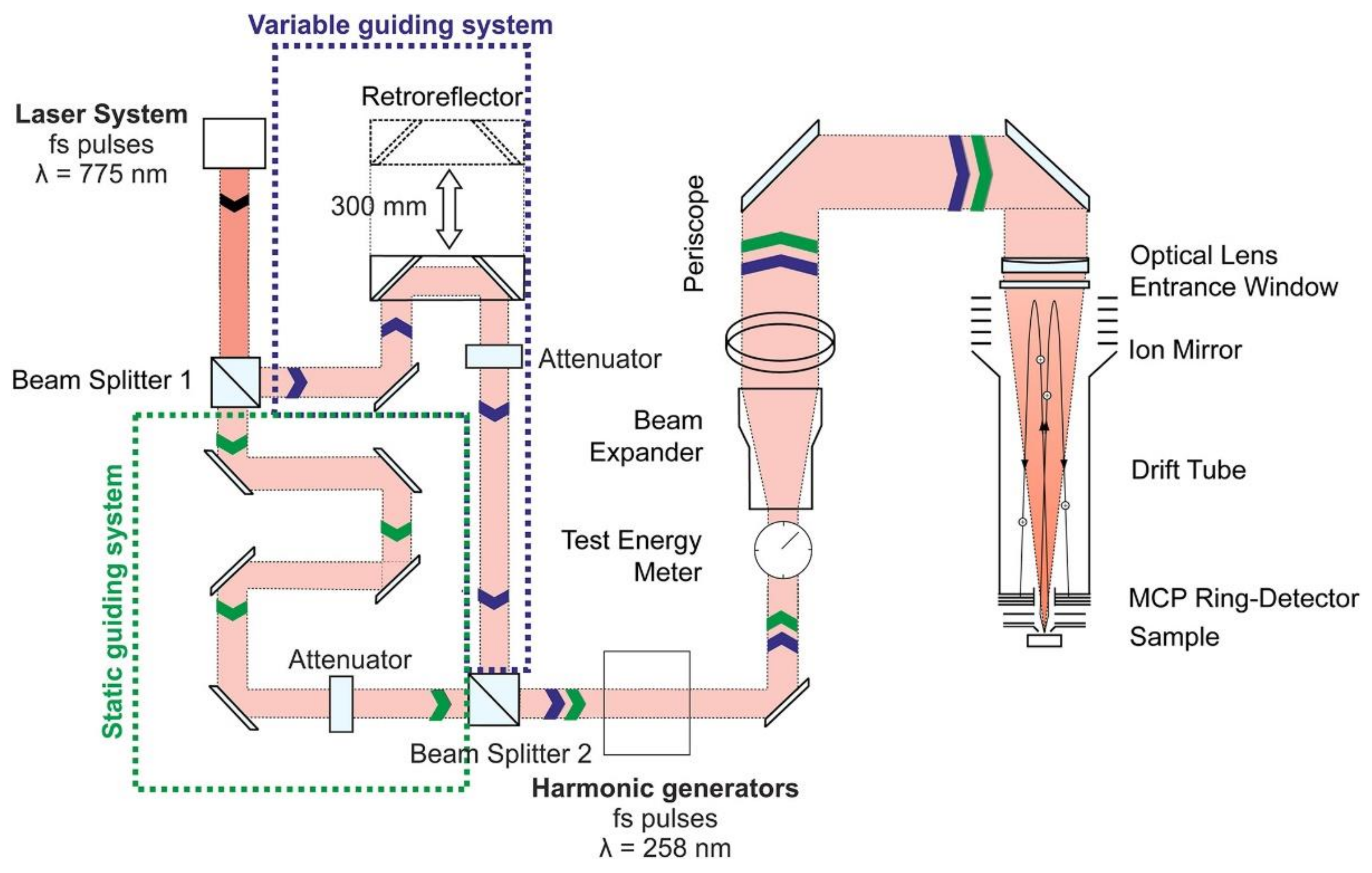
3.1.4. Optical Arrangement of Laser Ablation Ion Source
3.2. TOF Mass Analyser
3.3. Selected Femtosecond-LIMS Systems
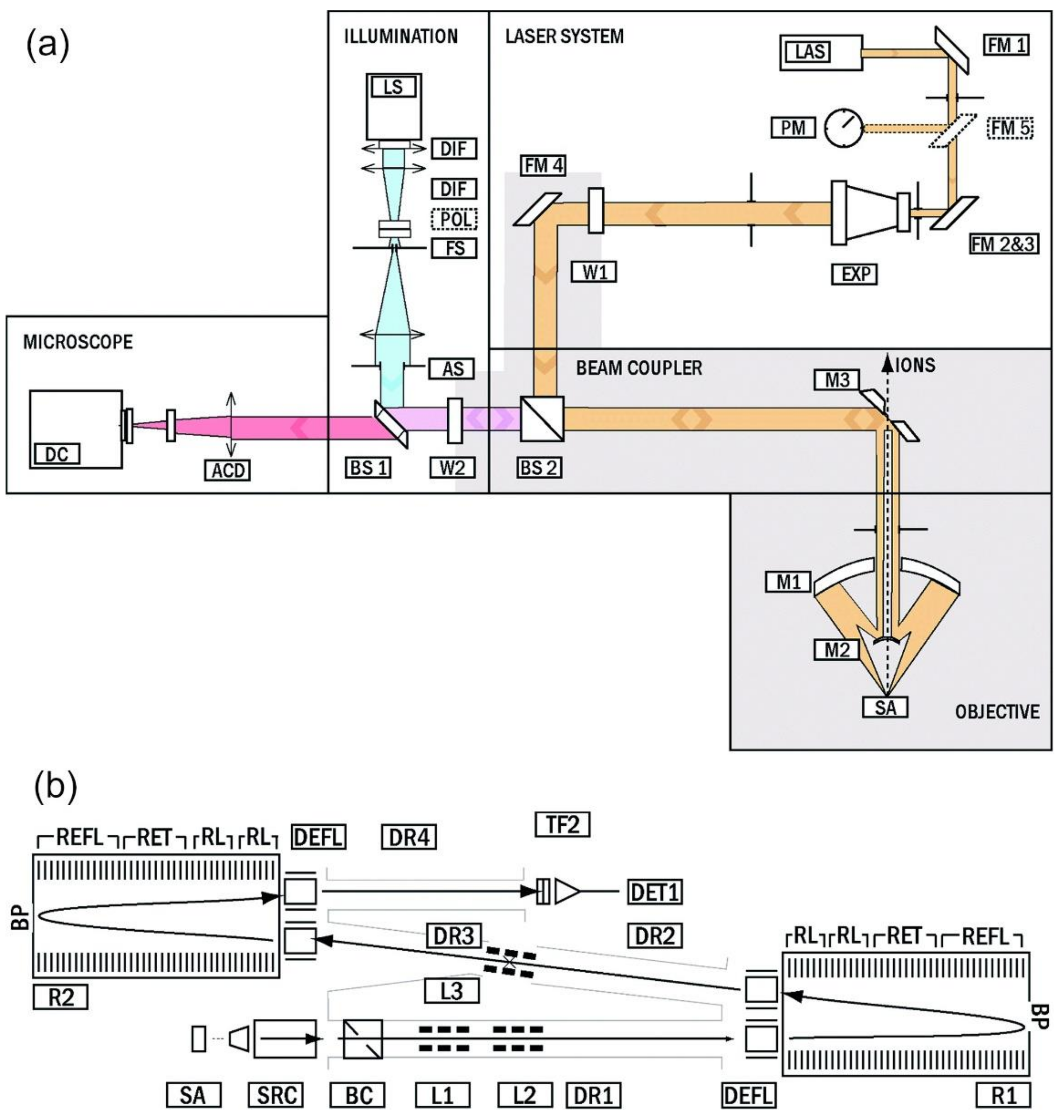
3.3.1. Laboratory: LI-O-TOFMS
3.3.2. Miniature Femtosecond-LIMS: LMS
3.3.3. High-Resolution fs-LIMS: LMS-GT
4. Performance of fs-LIMS Instruments in the Element and Isotope Analysis
4.1. Time of Flight, Mass Range, and Mass Resolution
4.2. Dynamic Range, Detection Sensitivity, and Limit of Detection
4.3. Chemometrics and Analysis Protocols
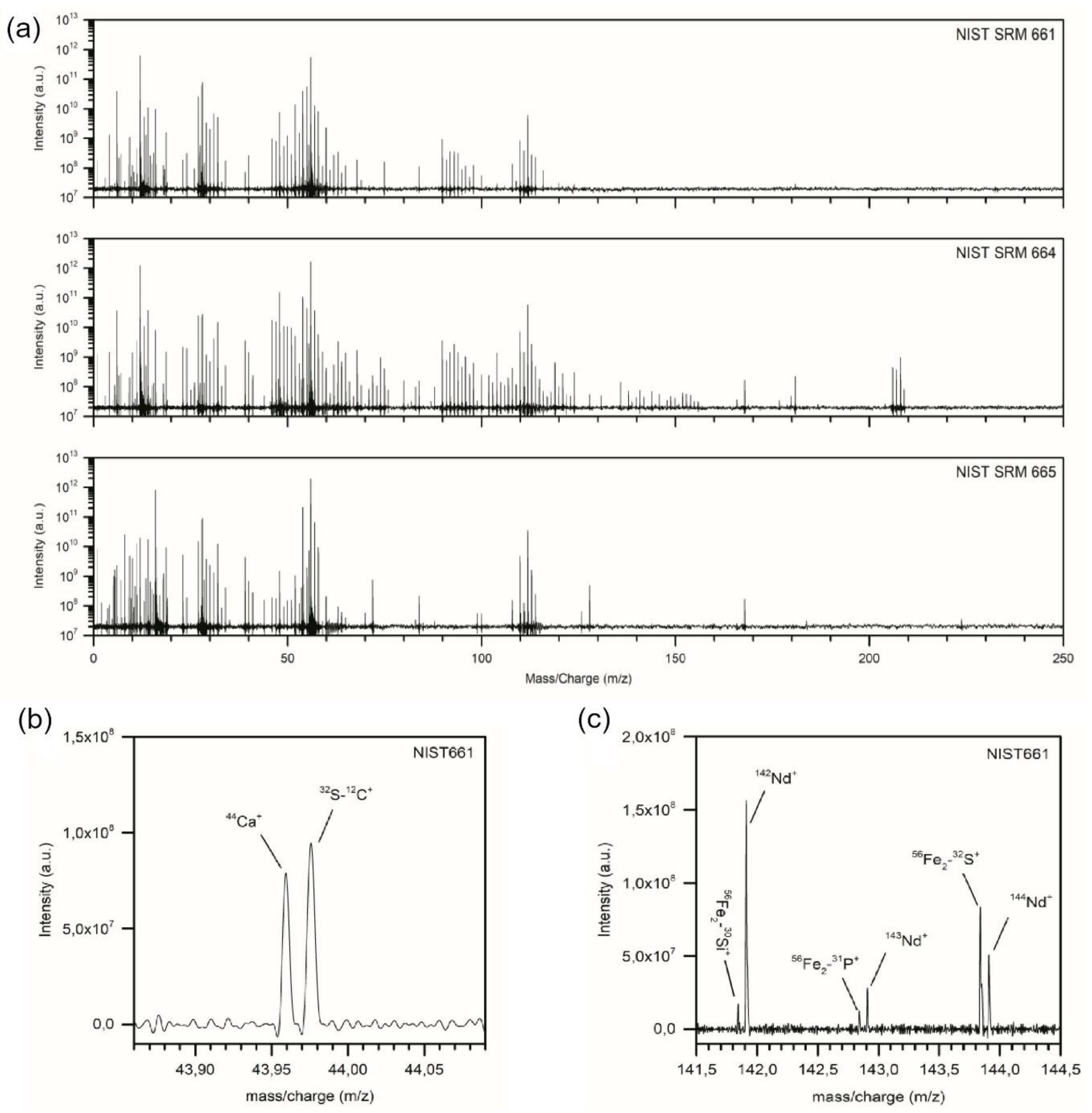
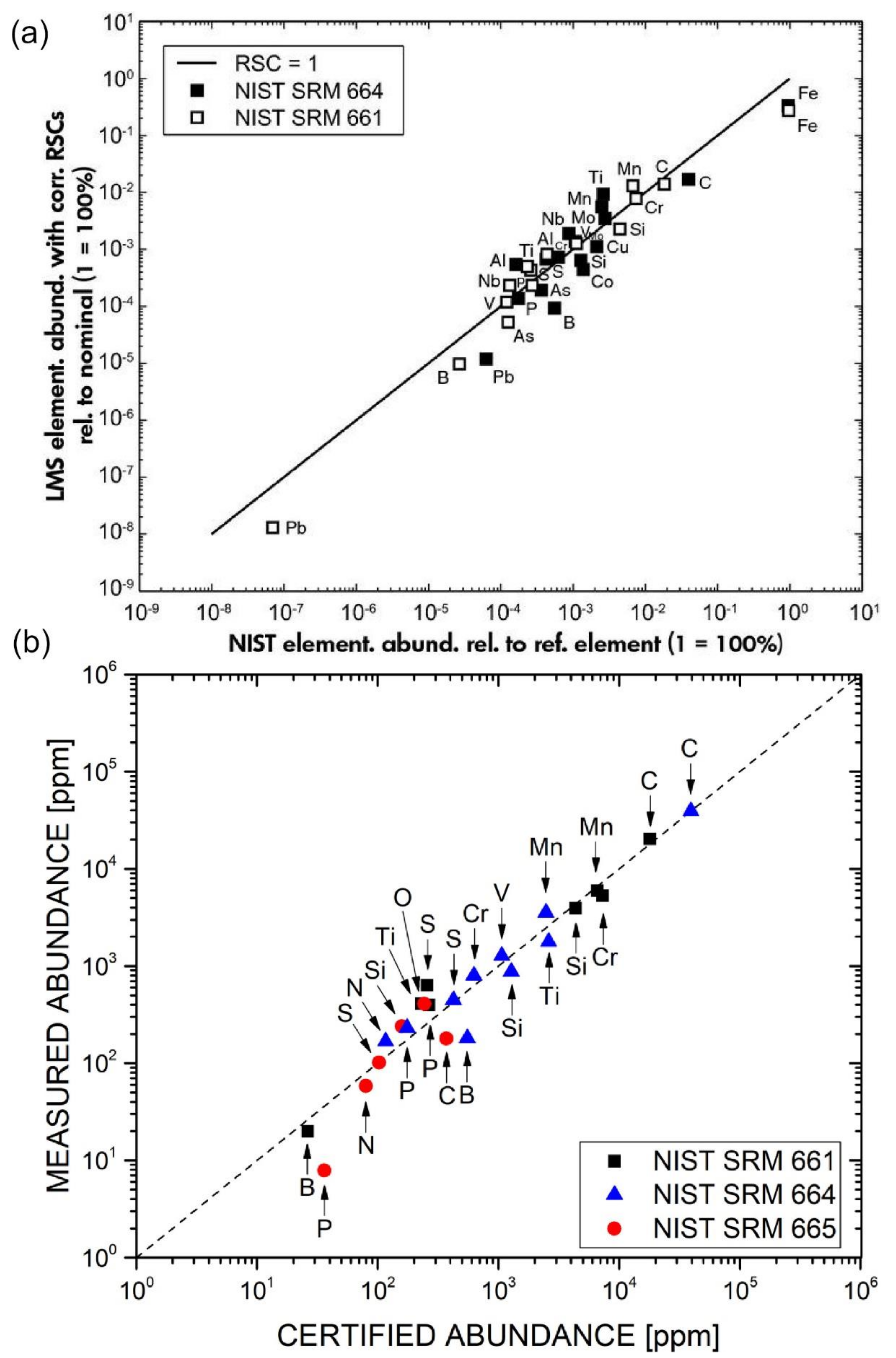
4.4. Measurements of Elements and Isotopes
4.4.1. Measurements of Elements
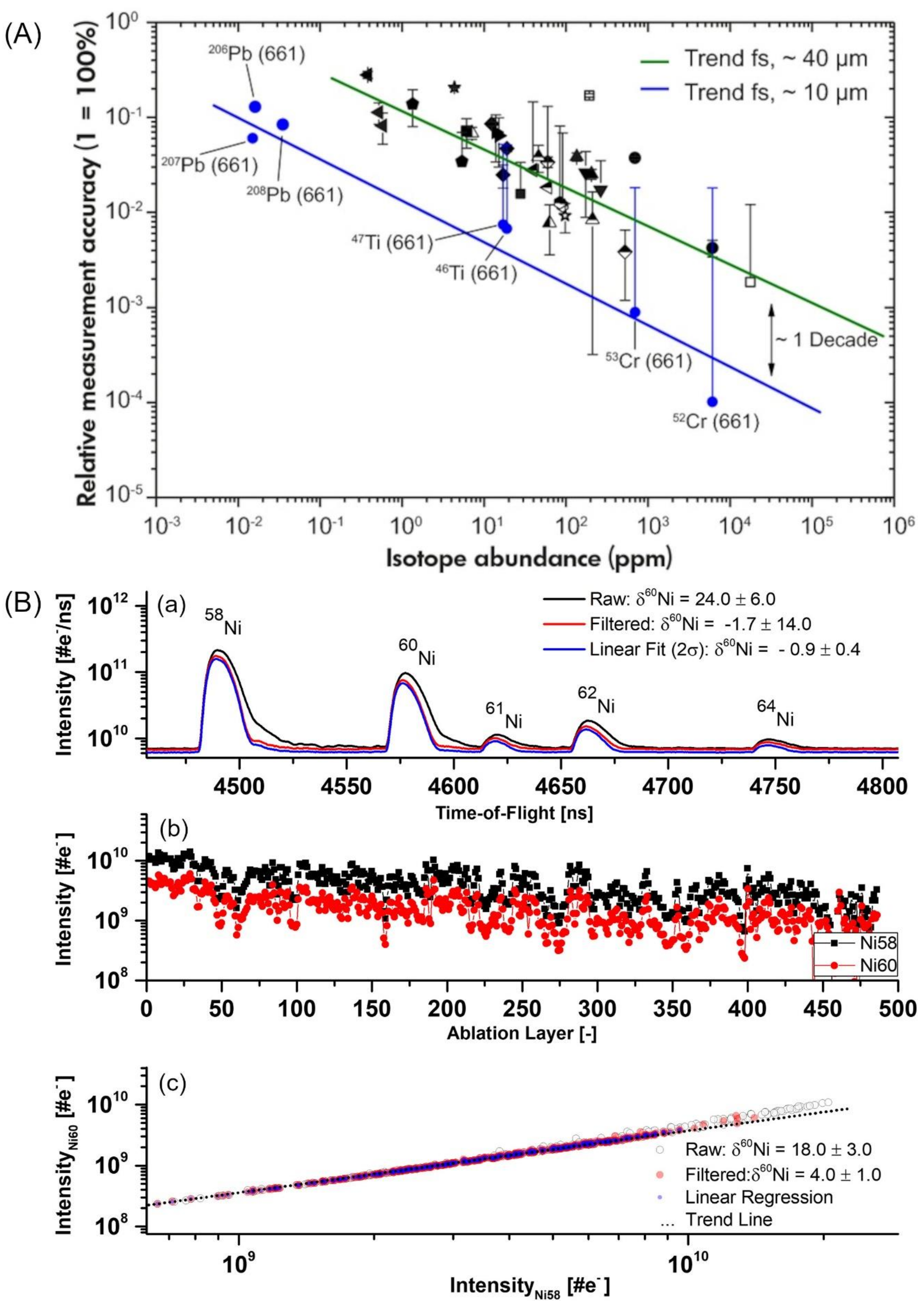
4.4.2. Measurements of Isotopes
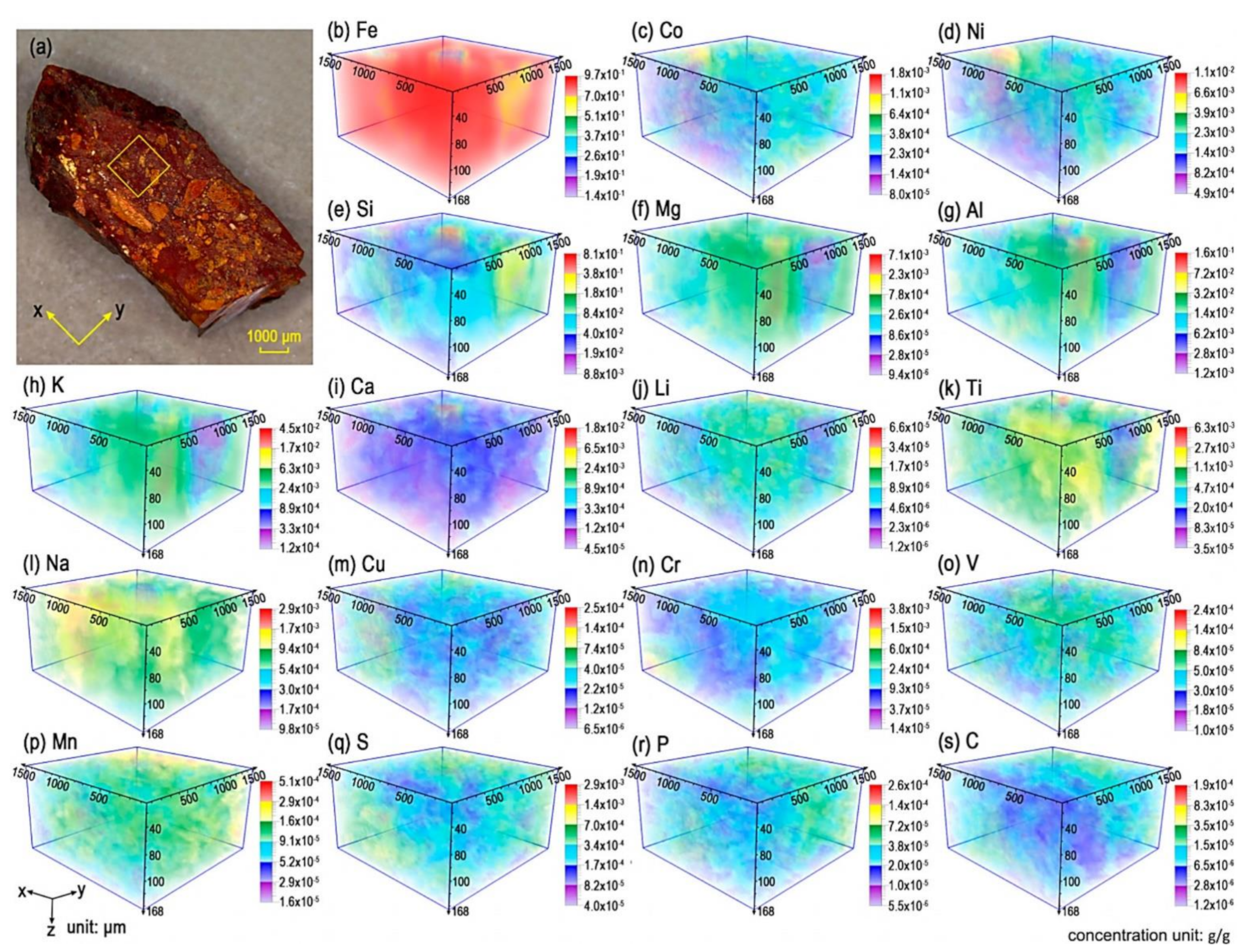
4.5. Depth Profiling, Chemical Imaging, and 3D Chemical Analysis
5. Applications
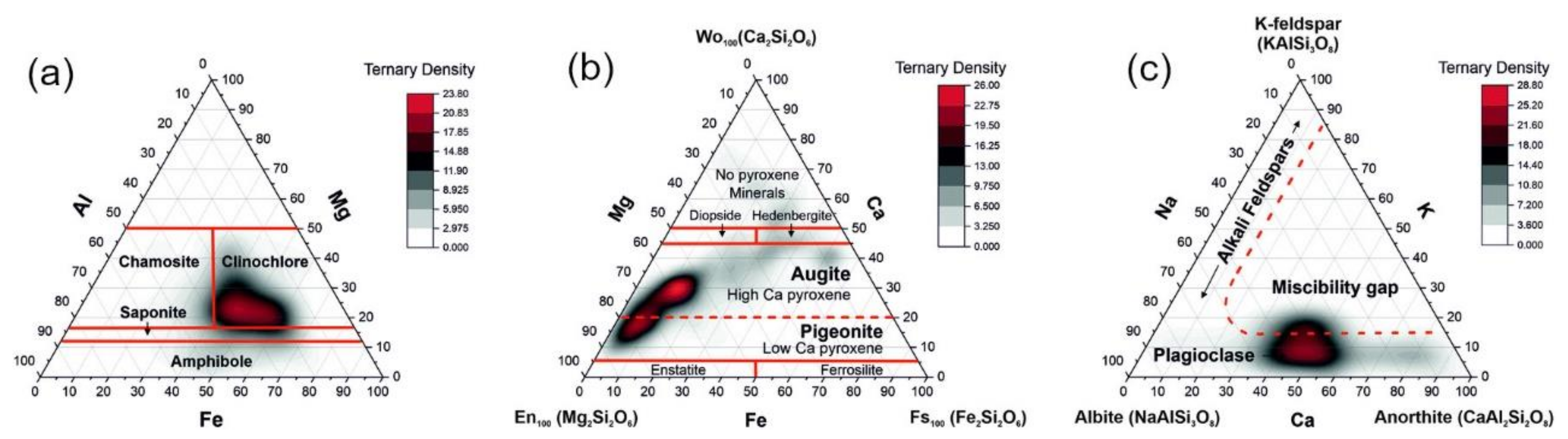
5.1. Geological and Geochemical Samples
5.2. Meteoritic Samples
5.3. Material Science
5.3.1. Semiconductor Industry
5.3.2. Materials for Electrochemistry
5.3.3. Ceramic Materials
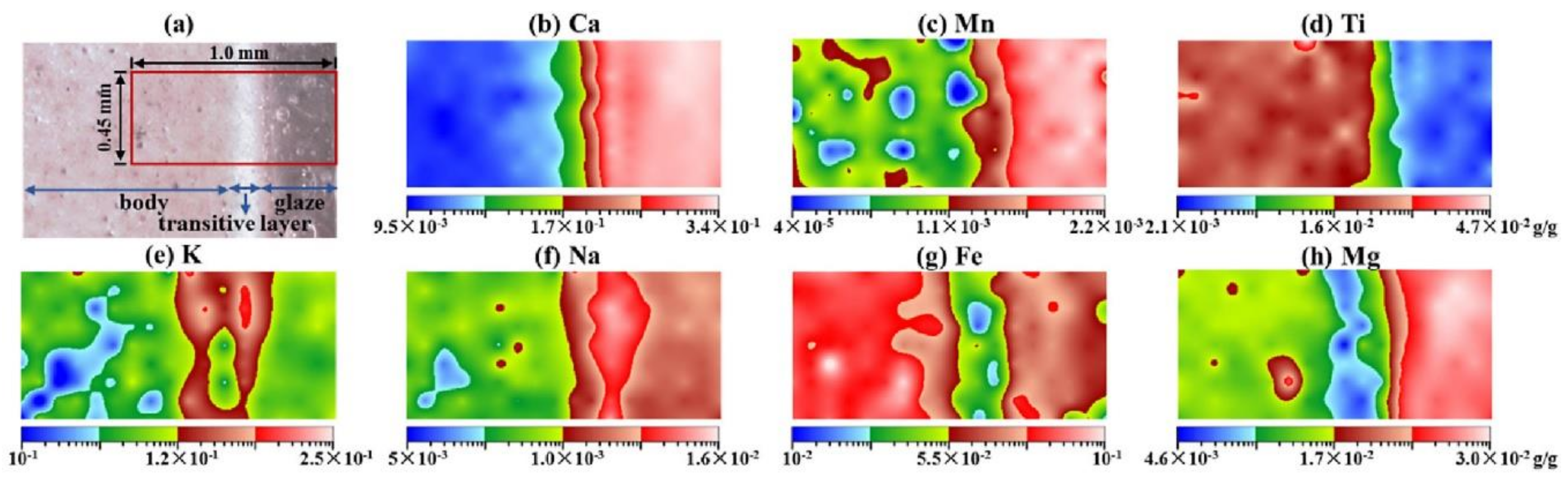
5.4. Bioanalytics
6. Summary and Outlook
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Azov, V.A.; Mueller, L.; Makarov, A.A. Laser Ionization Mass Spectrometry at 55: Quo Vadis? Mass Spectrom. Rev. 2020, 21669, 1–52. [Google Scholar] [CrossRef] [PubMed]
- Becker, J.S.; Dietze, H.-J. Laser Ionization Mass Spectrometry in Inorganic Trace Analysis. Anal. Bioanal. Chem. 1992, 344, 69–86. [Google Scholar] [CrossRef]
- Lin, Y.; Yu, Q.; Hang, W.; Huang, B. Progress of Laser Ionization Mass Spectrometry for Elemental Analysis—A Review of the past Decade. Spectrochim. Acta Part B At. Spectrosc. 2010, 65, 871–883. [Google Scholar] [CrossRef]
- Vogt, H.; Heinen, H.J.; Meier, S.; Wechsung, R. LAMMA 500 Principle and Technical Description of the Instrument. Anal. Bioanal. Chem. 1981, 308, 195–200. [Google Scholar] [CrossRef]
- Honig, R.E. Laser-Induced Emission of Electrons and Positive Ions from Metals and Semiconductors. Appl. Phys. Lett. 1963, 3, 8–11. [Google Scholar] [CrossRef]
- Hillenkamp, F.; Unsöld, E.; Kaufmann, R.; Nitsche, R. A High-Sensitivity Laser Microprobe Mass Analyzer. Appl. Phys. A 1975, 8, 341–348. [Google Scholar] [CrossRef]
- Bernal, E.; Levine, L.P.; Ready, J.F. Time-of-Flight Spectrometer for Laser Surface Interaction Studies. Rev. Sci. Instrum. 1966, 37, 938–941. [Google Scholar]
- Fenner, N.C.; Daly, N.R. Laser Used for Mass Analysis. Rev. Sci. Instrum. 1966, 37, 1068–1070. [Google Scholar] [CrossRef]
- Durrant, S.F. Laser Ablation Inductively Coupled Plasma Mass Spectrometry: Achievements, Problems, Prospects. J. Anal. At. Spectrom. 1999, 14, 1385–1403. [Google Scholar] [CrossRef]
- Gunther, D.; Jackson, S.E.; Longerich, H.P. Laser Ablation and Arc/Spark Solid Sample Introduction into Inductively Coupled Plasma Mass Spectrometers. Spectrochim Acta B 1999, 54, 381–409. [Google Scholar] [CrossRef]
- Moreno-García, P.; Grimaudo, V.; Riedo, A.; Tulej, M.; Wurz, P.; Broekmann, P. Towards Matrix-Free Femtosecond-Laser Desorption Mass Spectrometry for in Situ Space Research. Rapid Commun. Mass Spectrom. 2016, 30, 1031–1036. [Google Scholar] [CrossRef] [PubMed]
- Ligterink, N.F.W.; Grimaudo, V.; Moreno-García, P.; Lukmanov, R.; Tulej, M.; Leya, I.; Lindner, R.; Wurz, P.; Cockell, C.S.; Ehrenfreund, P.; et al. Origin: A Novel and Compact Laser Desorption—Mass Spectrometry System for Sensitive in Situ Detection of Amino Acids on Extraterrestrial Surfaces. Sci. Rep. 2020, 10, 9641. [Google Scholar] [CrossRef] [PubMed]
- Corrigan, C.M.; Brinckerhoff, W.B.; Cornish, T.; Ecelberger, S. In Situ Laser Desorption Mass Spectrometry of Meteoritic Samples as Planetary Analogs. Meteorit Planet Sci. 2007, 42, A33. [Google Scholar]
- Uckert, K.; Grubisic, A.; Li, X.; Brinckerhoff, W.; Cornish, T.; Farcy, B.; Getty, S. IR Resonance-Enhanced Organic Detection with Two-Step Laser Desorption Time-of-Flight Mass Spectrometry. Icarus 2018, 299, 15–21. [Google Scholar] [CrossRef]
- Lubman, D.M. Lasers and Mass Spectrometry; Oxford University Press: New York, NY, USA, 1990. [Google Scholar]
- Vertes, A.; Gijbels, R.; Adams, F. Laser Ionization Mass spectrometry Analysis; Wiley-Interscience: New York, NY, USA, 1993. [Google Scholar]
- Yu, Q.; Chen, L.; Huang, R.; Hang, W.; Huang, B.; He, J. Laser Ionization Time-of-Flight Mass Spectrometry for Direct Elemental Analysis. TrAC Trends Anal. Chem. 2009, 28, 1174–1185. [Google Scholar] [CrossRef]
- Hergenröder, R.; Samek, O.; Hommes, V. Femtosecond Laser Ablation Elemental Mass Spectrometry. Mass Spectrom. Rev. 2006, 25, 551–572. [Google Scholar] [CrossRef]
- Cui, Y.; Moore, J.F.; Milasinovic, S.; Liu, Y.M.; Gordon, R.J.; Hanley, L. Depth profiling and imaging capabilities of an ultrashort pulse laser ablation time of flight mass spectrometer. Rev Sci Instrum 2012, 83, 093702. [Google Scholar] [CrossRef]
- Zhang, B.; He, M.; Hang, W.; Huang, B. Minimizing Matrix Effect by Femtosecond Laser Ablation and Ionization in Elemental Determination. Anal. Chem. 2013, 85, 4507–4511. [Google Scholar] [CrossRef] [PubMed]
- Riedo, A.; Neuland, M.B.; Meyer, S.; Tulej, M.; Wurz, P. Coupling of LMS with a Fs-Laser Ablation Ion Source: Elemental and Isotope Composition Measurements. J. Anal. At. Spectrom. 2013, 28, 1256–1269. [Google Scholar] [CrossRef]
- Hang, W. Laser Ionization Time-of-Flight Mass Spectrometer with an Ion Guide Collision Cell for Elemental Analysis of Solids. J. Anal. At. Spectrom. 2005, 20, 301–307. [Google Scholar] [CrossRef]
- He, M.; Li, B.; Yu, S.; Zhang, B.; Liu, Z.; Hang, W.; Huang, B. Buffer-Gas Assisted High Irradiance Femtosecond Laser Ionization Orthogonal Time-of-Flight Mass Spectrometry for Rapid Depth Profiling. J. Anal. At. Spectrom. 2013, 28, 499. [Google Scholar] [CrossRef]
- Wiesendanger, R.; Grimaudo, V.; Tulej, M.; Riedo, A.; Lukmanov, R.; Ligterink, N.; Fausch, R.; Shea, H.; Wurz, P. The LMS-GT Instrument—A New Perspective for Quantification with the LIMS-TOF Measurement Technique. J. Anal. At. Spectrom. 2019, 34, 2061–2073. [Google Scholar] [CrossRef]
- Rohner, U.; Whitby, J.A.; Wurz, P. A Miniature Laser Ablation Time-of-Flight Mass Spectrometer Forin Situplanetary Exploration. Meas. Sci. Technol. 2003, 14, 2159–2164. [Google Scholar] [CrossRef]
- Rohner, U.; Whitby, J.A.; Wurz, P.; Barabash, S. Highly Miniaturized Laser Ablation Time-of-Flight Mass Spectrometer for a Planetary Rover. Rev. Sci. Instruments 2004, 75, 1314–1322. [Google Scholar] [CrossRef]
- Tulej, M.; Riedo, A.; Neuland, M.B.; Meyer, S.; Wurz, P.; Thomas, N.; Grimaudo, V.; Moreno-García, P.; Broekmann, P.; Neubeck, A.; et al. CAMAM: A Miniature Laser Ablation Ionisation Mass Spectrometer and Microscope-Camera System forIn SituInvestigation of the Composition and Morphology of Extraterrestrial Materials. Geostand. Geoanalytical Res. 2014, 38, 441–466. [Google Scholar] [CrossRef]
- Wiesendanger, R.; Wacey, D.; Tulej, M.; Neubeck, A.; Ivarsson, M.; Grimaudo, V.; Moreno-García, P.; Cedeño-López, A.; Riedo, A.; Wurz, P. Chemical and Optical Identification of Micrometer-Sized 1.9 Billion-Year-Old Fossils by Combining a Miniature Laser Ablation Ionization Mass Spectrometry System with an Optical Microscope. Astrobiology 2018, 18, 1071–1080. [Google Scholar] [CrossRef] [PubMed]
- Managadze, G.G. Quantitative Reference-Free Express Analysis of Some Alloys on a Laser Time-of-Flight Mass Spectrometer. Tech. Phys. 1999, 44, 1253–1257. [Google Scholar] [CrossRef]
- Brinckerhoff, W.B.; Managadze, G.G.; McEntire, R.W.; Cheng, A.F.; Green, W.J. Laser Time-of-Flight Mass Spectrometry for Space. Rev. Sci. Instrum. 2000, 71, 536–545. [Google Scholar] [CrossRef]
- Sysoev, A.A.; Sysoev, A.A. Can Laser-Ionisation Time-of-Flight Mass Spectrometry Be a Promising Alternative to Laser Ablation/Inductively-Coupled Plasma Mass Spectrometry and Glow Discharge Mass Spectrometry for the Elemental Analysis of Solids? Eur. J. Mass Spectrom. 2002, 8, 213–232. [Google Scholar] [CrossRef]
- Neubeck, A.; Tulej, M.; Ivarsson, M.; Broman, C.; Riedo, A.; McMahon, S.; Wurz, P.; Bengtson, S. Mineralogical Determination in Situ of a Highly Heterogeneous Material Using a Miniaturized Laser Ablation Mass Spectrometer with High Spatial Resolution. Int. J. Astrobiol. 2016, 15, 133–146. [Google Scholar] [CrossRef]
- Riedo, A.; De Koning, C.; Stevens, A.H.; Cockell, C.S.; McDonald, A.; López, A.C.; Grimaudo, V.; Tulej, M.; Wurz, P.; Ehrenfreund, P. The Detection of Elemental Signatures of Microbes in Martian Mudstone Analogs Using High Spatial Resolution Laser Ablation Ionization Mass Spectrometry. Astrobiology 2020, 20, 1224–1235. [Google Scholar] [CrossRef] [PubMed]
- Tulej, M.; Lukmanov, R.; Grimaudo, V.; Riedo, A.; De Koning, C.; Ligterink, N.F.W.; Neubeck, A.; Ivarsson, M.; McMahon, S.; Wurz, P. Determination of the Microscopic Mineralogy of Inclusion in an Amygdaloidal Pillow Basalt by fs-LIMS. J. Anal. At. Spectrom. 2021, 36, 80–91. [Google Scholar] [CrossRef]
- Tulej, M.; Neubeck, A.; Ivarsson, M.; Riedo, A.; Neuland, M.B.; Meyer, S.; Wurz, P. Chemical Composition of Micrometer-Sized Filaments in an Aragonite Host by a Miniature Laser Ablation/Ionization Mass Spectrometer. Astrobiology 2015, 15, 669–682. [Google Scholar] [CrossRef] [PubMed]
- Margetic, V.; Pakulev, A.; Stockhaus, A.; Bolshov, M.; Niemax, K.; Hergenröder, R. A Comparison of Nanosecond and Femtosecond Laser-Induced Plasma Spectroscopy of Brass Samples. Spectrochim. Acta Part B At. Spectrosc. 2000, 55, 1771–1785. [Google Scholar] [CrossRef]
- Gurevich, E.L.; Hergenröder, R. Femtosecond Laser-Induced Breakdown Spectroscopy: Physics, Applications, and Perspectives. Appl. Spectrosc. 2007, 61, 233A–242A. [Google Scholar] [CrossRef] [PubMed]
- Labutin, T.A.; Lednev, V.N.; Ilyin, A.A.; Popov, A.M. Femtosecond Laser-Induced Breakdown Spectroscopy. J. Anal. At. Spectrom. 2015, 31, 90–118. [Google Scholar] [CrossRef]
- Poitrasson, F.; D’Abzac, F.-X. Femtosecond Laser Ablation Inductively Coupled Plasma Source Mass Spectrometry for Elemental and Isotopic Analysis: Are Ultrafast Lasers Worthwhile? J. Anal. At. Spectrom. 2017, 32, 1075–1091. [Google Scholar] [CrossRef]
- Povarnitsyn, M.E.; Itina, T.E.; Sentis, M.; Khishchenko, K.V.; Levashov, P.R. Material Decomposition Mechanisms in Femtosecond Laser Interactions with Metals. Phys. Rev. B 2007, 75, 235414. [Google Scholar] [CrossRef]
- Povarnitsyn, M.E.; Itina, T.E.; Khishchenko, K.V.; Levashov, P.R. Phase transitions and metastable states in ultrashort laser-metal interaction. Prog. Quantum Electron. 2013, 37, 215–323. [Google Scholar] [CrossRef]
- Gamaly, E.G.; Rode, A.V. Physics of ultra-short laser interaction with matter: From phonon excitation to ultimate transformations. Prog. Quant. Electron 2013, 37, 215–323. [Google Scholar] [CrossRef]
- Gamaly, E.G. Femtosecond Laser-Matter Interactios: Theory, Experiments and Applications; CRC Press: Cleveland, OH, USA, 2011. [Google Scholar]
- Chichkov, B.N.; Momma, C.; Nolte, S.; Von Alvensleben, F.; Tünnermann, A. Femtosecond, Picosecond and Nanosecond Laser Ablation of Solids. Appl. Phys. A 1996, 63, 109–115. [Google Scholar] [CrossRef]
- Bonse, J.; Baudach, S.; Krüger, J.; Kautek, W.; Lenzner, M. Femtosecond Laser Ablation of Silicon–Modification Thresholds and Morphology. Appl. Phys. A 2002, 74, 19–25. [Google Scholar] [CrossRef]
- Price, D.F.; More, R.M.; Walling, R.S.; Guethlein, G.; Shepherd, R.L.; Stewart, R.E.; White, W.E. Absorption of Ultrashort Laser Pulses by Solid Targets Heated Rapidly to Temperatures 1–1000 eV. Phys. Rev. Lett. 1995, 75, 252–255. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.-K.; Vallée, F.; Acioli, L.H.; Ippen, E.P.; Fujimoto, J.G. Femtosecond-Tunable Measurement of Electron Thermalization in Gold. Phys. Rev. B 1994, 50, 15337–15348. [Google Scholar] [CrossRef]
- Wellershoff, S.-S.; Hohlfeld, J.; Güdde, J.; Matthias, E. The Role of Electron–Phonon Coupling in Femtosecond Laser Damage of Metals. Appl. Phys. A 1999, 69, S99–S107. [Google Scholar] [CrossRef]
- Lorazo, P.; Lewis, L.J.; Meunier, M. Short-Pulse Laser Ablation of Solids: From Phase Explosion to Fragmentation. Phys. Rev. Lett. 2003, 91, 225502. [Google Scholar] [CrossRef] [PubMed]
- Krüger, J.; Kautek, W. Femtosecond Pulse Visible Laser Processing of Fibre Composite Materials. Appl. Surf. Sci. 1996, 106, 383–389. [Google Scholar] [CrossRef]
- Borowiec, A.; Couillard, M.; Botton, G.; Haugen, H. Sub-Surface Damage in Indium Phosphide Caused by Micromachining of Grooves with Femtosecond and Nanosecond Laser Pulses. Appl. Phys. A 2004, 79, 1887–1890. [Google Scholar] [CrossRef]
- Couillard, M.; Borowiec, A.; Haugen, H.K.; Preston, J.S.; Griswold, E.M.; Botton, G.A. Subsurface Modifications in Indium Phosphide Induced by Single and Multiple Femtosecond Laser Pulses: A Study on the for-Mation of Periodic Ripples. J. Appl. Phys. 2007, 101, 033519. [Google Scholar] [CrossRef]
- Seydoux-Guillaume, A.-M.; Freydier, R.; Poitrasson, F.; ’Abzac, F.-X.D.; Wirth, R.; Datas, L. Dominance of Mechanical over Thermally Induced Damage during Femtosecond Laser Ablation of Monazite. Eur. J. Miner. 2010, 22, 235–244. [Google Scholar] [CrossRef]
- Haglund, F. Mechanisms of Laser-Induced Desorption and Ablation. In Laser Ablation and Desorption; Miller, J.C., Haglund, F., Eds.; Academic Press:: London, UK, 1998; pp. 15–138. [Google Scholar]
- Grossmann, F. Theoretical Femtosecond Physics; Springer: Berlin/Heidelberg, Germany, 2008; p. 214. [Google Scholar]
- Balling, P.; Schou, J. Femtosecond-Laser Ablation Dynamics of Dielectrics: Basics and Applications for Thin Films. Rep. Prog. Phys. 2013, 76, 036502. [Google Scholar] [CrossRef]
- Schmidt, V.; Husinsky, W.; Betz, G. Dynamics of Laser Desorption and Ablation of Metals at the Threshold on the Femtosecond Time Scale. Phys. Rev. Lett. 2000, 85, 3516–3519. [Google Scholar] [CrossRef]
- Momma, C.; Nolte, S.; Chichkov, B.N.; Alvensleben, F.V.; Tünnermann, A. Precise Laser Ablation with Ultrashort Pulses. Appl. Surf. Sci. 1997, 109-110, 15–19. [Google Scholar] [CrossRef]
- Nolte, S.; Momma, C.; Jacobs, H.; Tünnermann, A.; Chichkov, B.N.; Wellegehausen, B.; Welling, H. Ablation of Metals by Ultrashort Laser Pulses. J. Opt. Soc. Am. B 1997, 14, 2716–2722. [Google Scholar] [CrossRef]
- Stoian, R.; Ashkenasi, D.; Rosenfeld, A.; Campbell, E.E.B. Coulomb Explosion in Ultrashort Pulsed Laser Ablation ofAl2O3. Phys. Rev. B 2000, 62, 13167–13173. [Google Scholar] [CrossRef]
- Bulgakova, N.M.; Rosenfeld, A.; Ehrentraut, L.; Stoian, R.; Hertel, I.V. Modeling of electron dynamics in laser-irradiated solids: Progress achieved through a continuum approach and future prospects. International Conference on Lasers, Applications, and Technologies, Minsk, Belarus, 28 June, 2007, Proc. Spie. 2007, 6732, 673208. [Google Scholar] [CrossRef]
- Du, D.; Liu, X.; Korn, G.; Squier, J.; Mourou, G. Laser-Induced Breakdown by Impact Ionization in Sio2 with Pulse Widths from 7 Ns to 150 Fs. Appl. Phys. Lett. 1994, 64, 3071–3073. [Google Scholar] [CrossRef]
- Roeterdink, W.G.; Juurlink, L.B.F.; Vaughan, O.P.H.; Diez, J.D.; Bonn, M.; Kleyn, A.W. Coulomb Explosion in Femtosecond Laser Ablation of Si (111). Appl. Phys. Lett. 2003, 82, 4190. [Google Scholar] [CrossRef]
- Barthélemy, O.; Margot, J.; Chaker, M.; Sabsabi, M.; Vidal, F.; Johnston, T.; Laville, S.; Le Drogoff, B. Influence of the Laser Parameters on the Space and Time Characteristics of an Aluminum Laser-Induced Plasma. Spectrochim. Acta Part B: At. Spectrosc. 2005, 60, 905–914. [Google Scholar] [CrossRef]
- Laville, S.; Vidal, F.; Johnston, T.W.; Barthélemy, O.; Chaker, M.; Le Drogoff, B.; Margot, J.; Sabsabi, M. Fluid Modeling of the Laser Ablation Depth as a Function of the Pulse Duration for Conductors. Phys. Rev. E 2002, 66, 066415. [Google Scholar] [CrossRef] [PubMed]
- Hergenröder, R. A Model for the Generation of Small Particles in Laser Ablation ICP-MS. J. Anal. At. Spectrom. 2006, 21, 1016–1026. [Google Scholar] [CrossRef]
- Perez, D.; Lewis, L.J. Ablation of Solids under Femtosecond Laser Pulses. Phys. Rev. Lett. 2002, 89, 255504. [Google Scholar] [CrossRef] [PubMed]
- Itina, T.; Vidal, F.; Delaporte, P.; Sentis, M. Numerical Study of Ultra-short Laser Ablation of Metals and of Laser Plume Dynamics. Appl. Phys. A 2004, 79, 1089–1092. [Google Scholar] [CrossRef]
- Chen, J.K.; Beraun, J.E. Modelling of Ultrashort Laser Ablation of Gold Films in Vacuum. J. Opt. A: Pure Appl. Opt. 2003, 5, 168–173. [Google Scholar] [CrossRef]
- Itina, T.E.; Povarnitsyn, M.E.; Gouriet, K.; Noël, S.; Hermann, J. Mechanisms of Nanoparticle Formation by Short Laser Pulses. In Lasers and Applications in Science and Engineering; SPIE-Intl. Soc. Opt. Eng. 2007, 6458. [Google Scholar] [CrossRef]
- Hergenröder, R. A Model of Non-Congruent Laser Ablation as a Source of Fractionation Effects in LA-ICP-MS. J. Anal. At. Spectrom. 2006, 21, 505–516. [Google Scholar] [CrossRef]
- Mao, X.; Chan, W.-T.; Caetano, M.; Shannon, M.A.; Russo, R.E. Preferential Vaporization and Plasma Shielding during Nano-Second Laser Ablation. Appl. Surf. Sci. 1996, 96-98, 126–130. [Google Scholar] [CrossRef]
- Leitz, K.-H.; Redlingshöfer, B.; Reg, Y.; Otto, A.; Schmidt, M. Metal Ablation with Short and Ultrashort Laser Pulses. Phys. Procedia 2011, 12, 230–238. [Google Scholar] [CrossRef]
- Kawata, S.; Sun, H.-B.; Tanaka, T.; Takada, K. Finer Features for Functional Microdevices. Nat. Cell Biol. 2001, 412, 697–698. [Google Scholar] [CrossRef]
- Sugioka, K.; Cheng, Y. Femtosecond Laser 3D Micromachining for Microfluidic and Optofluidic Applications; Springer International Publishing: New York, NY, USA, 2014. [Google Scholar]
- Samek, O.; Hommes, V.; Hergenroder, R.; Kukhlevsky, S.V. Femtosecond Pulse Shaping Using a Liquid–Crystal Display: Applications to Depth Profiling Analysis. Rev. Sci. Instrum. 2005, 76, 086104. [Google Scholar] [CrossRef]
- Amoruso, S.; Wang, X.; Altucci, C.; de Lisio, C.; Armenante, M.; Bruzzese, R.; Spinelli, N.; Velotta, R. Double-Peak Distribution of Electron and Ion Emission Profile during Femtosecond Laser Ablation of Metals. Appl. Surf. Sci. 2002, 186, 358–363. [Google Scholar] [CrossRef]
- Anisimov, S.I.; Bauerle, D.; Luk’Yanchuk, B.S. Gas Dynamics and Film Profiles in Pulsed-Laser Deposition of Materials. Phys. Rev. B 1993, 48, 12076–12081. [Google Scholar] [CrossRef] [PubMed]
- Anoop, K.K.; Polek, M.P.; Bruzzese, R.; Amoruso, S.; Harilal, S.S. Multidiagnostic Analysis of Ion Dynamics in Ultrafast Laser Ablation of Metals over a Large Fluence Range. J. Appl. Phys. 2015, 117, 083108. [Google Scholar] [CrossRef]
- Colombier, J.P.; Combis, P.; Bonneau, F.; Le Harzic, R.; Audouard, E. Hydrodynamic Simulations of Metal Ablation by Femtosecond Laser Irradiation. Phys. Rev. B 2005, 71. [Google Scholar] [CrossRef]
- López, A.C.; Grimaudo, V.; Riedo, A.; Tulej, M.; Wiesendanger, R.; Lukmanov, R.; Moreno-García, P.; Lörtscher, E.; Wurz, P.; Broekmann, P. Three-Dimensional Composition Analysis of SnAg Solder Bumps Using Ultraviolet Femtosecond Laser Ablation Ionization Mass Spectrometry. Anal. Chem. 2019, 92, 1355–1362. [Google Scholar] [CrossRef] [PubMed]
- Grimaudo, V.; Tulej, M.; Riedo, A.; Lukmanov, R.; Ligterink, N.F.; De Koning, C.; Wurz, P. UV Post-Ionization Laser Ablation Ionization Mass Spectrometry for Improved nm-Depth Profiling Resolution on Cr/Ni Reference Standard. Rapid Commun. Mass Spectrom. 2020, 34, e8803. [Google Scholar] [CrossRef]
- Garrelie, F.; Bourquard, F.; Loir, A.-S.; Donnet, C.; Colombier, J.-P. Control of Femtosecond Pulsed Laser Ablation and Deposition by Temporal Pulse Shaping. Opt. Laser Technol. 2016, 78, 42–51. [Google Scholar] [CrossRef]
- Höhm, S.; Rosenfeld, A.; Krüger, J.; Bonse, J. Laser-Induced Periodic Surface Structures on Zinc Oxide Crystals upon Two-Colour Femtosecond Double-Pulse Irradiation. Phys. Scr. 2017, 92, 034003. [Google Scholar] [CrossRef]
- Lopez-Quintas, I.; Rebollar, E.; Ávila-Brande, D.; Izquierdo, J.G.; Bañares, L.; Díaz-Guerra, C.; Urbieta, A.; Castillejo, M.; De Nalda, R.; Martín, M. Femtosecond Double-Pulse Laser Ablation and Deposition of Co-Doped ZnS Thin Films. Nanomaterials 2020, 10, 2229. [Google Scholar] [CrossRef]
- Piñon, V.; Fotakis, C.; Nicolas, G.; Anglos, D. Double Pulse Laser-Induced Breakdown Spectroscopy with Femtosecond Laser Pulses. Spectrochim. Acta Part B At. Spectrosc. 2008, 63, 1006–1010. [Google Scholar] [CrossRef]
- Riedo, A.; Lukmanov, R.; Grimaudo, V.; de Koening, C.; Lighterink, N.F.W.; Tulej, M.; Wurz, P. Improved plasma stoichiometry recorded by LIMS by using a double-pulse femtosecond laser ablation ion source. Rapid Commun. Mass Sp. 2020. under review. [Google Scholar] [CrossRef]
- Tulej, M.; Wiesendanger, R.; Riedo, A.; Knopp, G.; Wurz, P. Mass Spectrometric Analysis of the MG Plasma Produced by Double-Pulse Femtosecond Laser Irradiation. J. Anal. At. Spectrom. 2018, 33, 1292–1303. [Google Scholar] [CrossRef]
- Povarnitsyn, M.E.; Itina, T.E.; Khishchenko, K.V.; Levashov, P.R. Suppression of Ablation in Femtosecond Double-Pulse Experiments. Phys. Rev. Lett. 2009, 103, 195002. [Google Scholar] [CrossRef]
- Povarnitsyn, M.E.; Itina, T.E.; Levashov, P.R.; Khishchenko, K.V. Simulation of Ultrashort Double-Pulse Laser Ablation. Appl. Surf. Sci. 2011, 257, 5168–5171. [Google Scholar] [CrossRef]
- Semerok, A.; Dutouquet, C. Ultrashort Double Pulse Laser Ablation of Metals. Thin Solid Film. 2004, 453-454, 501–505. [Google Scholar] [CrossRef]
- Donnelly, T.; Lunney, J.G.; Amoruso, S.; Bruzzese, R.; Wang, X.; Ni, X. Double Pulse Ultrafast Laser Ablation of Nickel in Vacuum. J. Appl. Phys. 2009, 106, 013304. [Google Scholar] [CrossRef]
- Noël, S.; Hermann, J. Reducing Nanoparticles in Metal Ablation Plumes Produced by Two Delayed Short Laser Pulses. Appl. Phys. Lett. 2009, 94, 53120. [Google Scholar] [CrossRef]
- Maine, P.; Strickland, D.; Bado, P.; Pessot, M.; Mourou, G. Generation of Ultrahigh Peak Power Pulses by Chirped Pulse Amplification. IEEE J. Quantum Electron. 1988, 24, 398–403. [Google Scholar] [CrossRef]
- Shank, C.V. Subpicosecond Kilowatt Pulses from a MoDe-locked Cw Dye Laser. Appl. Phys. Lett. 1974, 24, 373. [Google Scholar] [CrossRef]
- Moulton, P. Tunable Solid-State Lasers. Proc. IEEE 1992, 80, 348–364. [Google Scholar] [CrossRef]
- Strickland, D.; Mourou, G. Compression of Amplified Chirped Optical Pulses. Opt. Commun. 1985, 56, 219–221. [Google Scholar] [CrossRef]
- Backus, S.; Durfee, C.G.; Murnane, M.M.; Kapteyn, H.C. High Power Ultrafast Lasers. Rev. Sci. Instrum. 1998, 69, 1207–1223. [Google Scholar] [CrossRef]
- Dong, L.; Samson, B. Fiber Lasers, Basics, Technology, and Applications; CRC Press/Taylor and Francis: Boca Raton, FL, USA; New York, NY, USA; London, UK, 2017. [Google Scholar]
- Herrmann, J. Theory of Kerr-Lens Mode Locking: Role of Self-Focusing and Radially Varying Gain. J. Opt. Soc. Am. B 1994, 11, 498–512. [Google Scholar] [CrossRef]
- Roullier, C. Femtosecond Laser Pulses, 2nd ed.; Springer US: New York, NY, USA, 2005. [Google Scholar]
- Wollenhaupt, M.; Assion, A.; Baumert, T. Short und Ultrashort Laser Pulses. In Springer Handbook of Lasers and Optics; Springer-Verlag: Berlin/Heidelberg, Germany, 2012. [Google Scholar] [CrossRef]
- Stoian, R.; Bulgakova, N.M.; Mermillod-Blondin, A.; Rosenfeld, A.; Spyridaki, M.; Koudoumas, E.; Fotakis, C.; Hertel, I.V. Adaptive Control of Ion Beams Produced by Ultrafast Laser Ablation of Silicon (Invited Paper). Integr. Optoelectron. Devices 2005, 5725, 329–343. [Google Scholar] [CrossRef]
- Dachraoui, H.; Husinsky, W. Thresholds of Plasma Formation in Silicon Identified by Optimizing the Ablation Laser Pulse Form. Phys. Rev. Lett. 2006, 97, 107601. [Google Scholar] [CrossRef] [PubMed]
- Wharton, K.B.; Boley, C.D.; Komashko, A.M.; Rubenchik, A.M.; Zweiback, J.; Crane, J.; Hays, G.; Cowan, T.E.; Ditmire, T. Effects of Nonionizing Prepulses in High-Intensity Laser-Solid Interactions. Phys. Rev. E 2001, 64, 025401. [Google Scholar] [CrossRef] [PubMed]
- Chin, S.L. Femtosecond Laser Filamentation; Springer International Publishing: New York, NY, USA, 2010; Volume 55. [Google Scholar]
- Jiang, L.; Wang, A.-D.; Li, B.; Cui, T.-H.; Lu, Y.-F. Electrons Dynamics Control by Shaping Femtosecond Laser Pulses in Micro/Nanofabrication: Modeling, Method, Measurement and Application. Light. Sci. Appl. 2018, 7, 17134. [Google Scholar] [CrossRef] [PubMed]
- Gross, J.M.; Hanley, L.; Veryovkin, I.V. Comparing Flat-Top and Gaussian Femtosecond Laser Ablation of Silicon at Normal and Oblique Angles of Incidence. In Proceedings of the Laser Beam Shaping XIX. SPIE-Intl Soc. Opt. Eng. 2019, 11107, 111070I. [Google Scholar]
- Sidick, E.; Dienes, A.; Knoesen, A. Ultrashort-Pulse Second-Harmonic Generation II Non-Transform-Limited Fundamental Pulses. J. Opt. Soc. Am. B 1995, 12, 1713–1722. [Google Scholar] [CrossRef]
- Sidick, E.; Knoesen, A.; Dienes, A. Ultrashort-Pulse Second-Harmonic Generation I Transform-Limited Fundamental Pulses. J. Opt. Soc. Am. B 1995, 12, 1704–1712. [Google Scholar] [CrossRef]
- Cerullo, G.; De Silvestri, S. Ultrafast Optical Parametric Amplifiers. Rev. Sci. Instrum. 2003, 74, 1–18. [Google Scholar] [CrossRef]
- Wiesendanger, R.; Tulej, M.; Grimaudo, V.; Cedeño-López, A.; Lukmanov, R.; Riedo, A.; Wurz, P. A Method for Improvement of Mass Resolution and Isotope Accuracy for Laser Ablation Time-of-Flight Mass Spectrometers. J. Chemom. 2018, 33, e3081. [Google Scholar] [CrossRef]
- Clark-MXR Inc. Manufacturer of ultrafast laser and instrumentation for various applications. Available online: https://www.cmxr.com/wp-content/uploads/2018/01/STORC-201801-01.pdf (accessed on 8 March 2021).
- Lin, Y.; Yu, Q.; Huang, R.; Hang, W.; He, J.; Huang, B. Characterization of Laser Ablation and Ionization in Helium and Argon: A Comparative Study by Time-of-Flight Mass Spectrometry. Spectrochim. Acta Part B At. Spectrosc. 2009, 64, 1204–1211. [Google Scholar] [CrossRef]
- He, J.; Huang, R.F.; Yu, Q.; Hang, W.; Huang, B.L. Standardless Semi-quantitative Elemental Analysis of Steel Using La-ser Ablation Time-of-flight Mass Spectrometry. Chem J. Chin. U 2009, 30, 57–59. [Google Scholar]
- McSween, H.Y.J.; Huss, G.R.; McSween, J.H.Y. Cosmochemistry; Cambridge University Press: Cambridge, UK, 2010. [Google Scholar]
- Simon, P.; Ihlemann, J. Machining of Submicron Structures on Metals and Semiconductors by Ultrashort UV-Laser Pulses. Appl. Phys. A 1996, 63, 505–508. [Google Scholar] [CrossRef]
- Website of Optical Engineering Company Offering Various Tools for Optical Modelling. Available online: www.zemax.com (accessed on 8 March 2021).
- Liang, Z.; Zhang, S.; Li, X.; Wang, T.; Huang, Y.; Hang, W.; Yang, Z.; Li, J.; Tian, Z. Tip-Enhanced Ablation and Ionization Mass Spectrometry for Nanoscale Chemical Analysis. Sci. Adv. 2017, 3, eaaq1059. [Google Scholar] [CrossRef] [PubMed]
- Boesl, U. Time-of-Flight Mass Spectrometry: Introduction to the Basics. Mass Spectrom. Rev. 2016, 36, 86–109. [Google Scholar] [CrossRef] [PubMed]
- Radionova, A.; Filippov, I.; Derrick, P.J. In Pursuit of Resolution in Time-of-Flight Mass Spectrometry: A Historical Perspective. Mass Spectrom. Rev. 2016, 35, 738–757. [Google Scholar] [CrossRef] [PubMed]
- Cotter, R.J. Peer Reviewed: The New Time-of-Flight Mass Spectrometry. Anal. Chem. 1999, 71, 445A–451A. [Google Scholar] [CrossRef] [PubMed]
- Wollnik, H. History of Mass Measurements in Time-of-Flight Mass Analyzers. Int. J. Mass Spectrom. 2013, 349-350, 38–46. [Google Scholar] [CrossRef]
- Standing, K.; Vestal, M.L. Time-of-Flight Mass Spectrometry (TOFMS): From Niche to Mainstream. Int. J. Mass Spectrom. 2015, 377, 295–308. [Google Scholar] [CrossRef]
- Hofer, L.; Wurz, P.; Buch, A.; Cabane, M.; Coll, P.; Coscia, D.; Gerasimov, M.; Lasi, D.; Sapgir, A.; Szopa, C.; et al. Prototype of the Gas Chromatograph–Mass Spectrometer to Investigate Volatile Species in the Lunar Soil for the Luna-Resurs Mission. Planet. Space Sci. 2015, 111, 126–133. [Google Scholar] [CrossRef]
- Mamyrin, B.A.; Shmikk, D.V. Linear Mass-Reflectron. Zh Eksp. Teor. Fiz. 1979, 76, 1500–1505. [Google Scholar]
- Scherer, S.; Altwegg, K.; Balsiger, H.; Fischer, J.; Jäckel, A.; Korth, A.; Mildner, M.; Piazza, D.; Reme, H.; Wurz, P. A Novel Principle for an Ion Mirror Design in Time-of-Flight Mass Spectrometry. Int. J. Mass Spectrom. 2006, 251, 73–81. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, S.; Cheng, X.; Zhu, E.; Hang, W.; Huang, B. Ion Kinetic Energy Distributions in Laser-Induced Plasma. Spectrochim. Acta Part B: At. Spectrosc. 2014, 99, 101–114. [Google Scholar] [CrossRef]
- Li, L.; Zhang, B.; Huang, R.; Hang, W.; He, J.; Huang, B. Laser Ionization Orthogonal Time-of-Flight Mass Spectrometry for Simultaneous Determination of Nonmetallic Elements in Solids. Anal. Chem. 2010, 82, 1949–1953. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Gonin, M.; Fuhrer, K.; Dodonov, A.; Su, C.; Wollnik, H. Orthogonal Electron Impact Source for a Time-of-Flight Mass Spectrometer with High Mass Resolving Power. Int. J. Mass Spectrom. 1999, 185–187, 221–226. [Google Scholar] [CrossRef]
- Peng, D.; He, J.; Yu, Q.; Chen, L.; Hang, W.; Huang, B. Parametric Evaluation of Laser Ablation and Ionization Time-of-Flight Mass Spectrometry with Ion Guide Cooling Cell. Spectrochim. Acta Part B At. Spectrosc. 2008, 63, 868–874. [Google Scholar] [CrossRef]
- Riedo, A.; Bieler, A.; Neuland, M.B.; Tulej, M.; Wurz, P. Performance Evaluation of a Miniature Laser Ablation Time-of-Flight Mass Spectrometer Designed Forin Situinvestigations in Planetary Space Research. J. Mass Spectrom. 2013, 48, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.F.; Lin, Y.M.; Li, L.F.; Hang, W.; He, J.; Huang, B.L. Two-Dimensional Separation in Laser Ionization Orthogonal Time-of-Flight Mass Spectrometry. Anal. Chem. 2010, 82, 3077–3080. [Google Scholar] [CrossRef]
- He, M.; Meng, Y.; Yan, S.; Hang, W.; Zhou, W.; Huang, B. Three-Dimensional Elemental Imaging of Nantan Meteorite via Femtosecond Laser Ionization Time-of-Flight Mass Spectrometry. Anal. Chem. 2017, 89, 565–570. [Google Scholar] [CrossRef] [PubMed]
- Tulej, M.; Iakovleva, M.; Leya, I.; Wurz, P. A Miniature Mass Analyser for in-Situ Elemental Analysis of Planetary Material–Performance Studies. Anal. Bioanal. Chem. 2011, 399, 2185–2200. [Google Scholar] [CrossRef]
- Riedo, A.; Tulej, M.; Rohner, U.; Wurz, P. High-Speed Microstrip Multi-Anode Multichannel Plate Detector System. Rev. Sci. Instrum. 2017, 88, 045114. [Google Scholar] [CrossRef] [PubMed]
- Wiesendanger, R.; Tulej, M.; Riedo, A.; Frey, S.; Shea, H.; Wurz, P. Improved Detection Sensitivity for Heavy Trace Elements Using a Miniature Laser Ablation Ionisation Mass Spectrometer. J. Anal. At. Spectrom. 2017, 32, 2182–2188. [Google Scholar] [CrossRef]
- Website of Ion Optical Engineering Company Offering Various Tools for Optical Modelling. Available online: www.simion.com (accessed on 8 March 2021).
- Wurz, P.; Schletti, R. Optical signal coupling in microchannel plate detectors with a subnanosecond performance. Rev. Sci. Instrum. 2001, 72, 3225–3229. [Google Scholar] [CrossRef]
- Schletti, R.; Wurz, P.; Scherer, S.; Siegmund, O.H. Fast Microchannel Plate Detector with an Impedance Matched Anode in Suspended Substrate Technology. Rev. Sci. Instrum. 2001, 72, 1634. [Google Scholar] [CrossRef]
- Koning, C.P.; Riedo, A.; Wiesendanger, R.; Grimaudo, V.; Lukmanov, R.; Lighterink, N.F.W.; Tulej, M.; Wurz, P. Quantitative Elemental Analysis with the LMS-GT; A Next Generation LIMS-TOF Instrument. Int. J. Mass Spectrom. 2021. submitted. [Google Scholar]
- Brenton, A.G.; Godfrey, A.R. Accurate Mass Measurement: Terminology and Treatment of Data. J. Am. Soc. Mass Spectrom. 2010, 21, 1821–1835. [Google Scholar] [CrossRef] [PubMed]
- Managadze, G.G.; Wurz, P.; Sagdeev, R.Z.; Chumikov, A.E.; Tuley, M.; Yakovleva, M.; Bondarenko, A.L. Study of the Main Geochemical Characteristics of Phobos’ Regolith Using Laser Time-of-Flight Mass Spectrometry. Sol. Syst. Res. 2010, 44, 376–384. [Google Scholar] [CrossRef]
- Spengler, B.; Kirsch, D.; Kaufmann, R.; Cotter, R.J. Metastable Decay of Peptides and Proteins in Matrix-Assisted Laser-Desorption Mass Spectrometry. Rapid Commun. Mass Spectrom. 1991, 5, 198–202. [Google Scholar] [CrossRef]
- Li, X.; Brinckerhoff, W.; Managadze, G.; Pugel, D.; Corrigan, C.; Doty, J. Laser Ablation Mass Spectrometer (LAMS) as a Standoff Analyzer in Space Missions for Airless Bodies. Int. J. Mass Spectrom. 2012, 323-324, 63–67. [Google Scholar] [CrossRef]
- Anderson, F.S.; Levine, J.; Whitaker, T.J. Rb-Sr Resonance Ionization Geochronology of the Duluth Gabbro: A Proof of Concept for in Situ Dating on the Moon. Rapid Commun. Mass Spectrom. 2015, 29, 1457–1464. [Google Scholar] [CrossRef]
- Riedo, A.; Meyer, S.; Heredia, B.; Neuland, M.; Bieler, A.; Tulej, M.; Leya, I.; Iakovleva, M.; Mezger, K.; Wurz, P. Highly Accurate Isotope Composition Measurements by a Miniature Laser Ablation Mass Spectrometer Designed for in Situ in-Vestigations on Planetary Surfaces. Planet. Space Sci. 2013, 87, 1–13. [Google Scholar] [CrossRef]
- Russo, E.R.; Mao, X.; Borisov, O.V. Laser Ablation Sampling. TrAC Trends Anal. Chem. 1998, 17, 461–469. [Google Scholar] [CrossRef]
- Zhang, S.; Zhang, B.; Hang, W.; Huang, B. Chemometrics and Theoretical Approaches for Evaluation of Matrix Effect in Laser Ablation and Ionization of Metal Samples. Spectrochim. Acta Part B: At. Spectrosc. 2015, 107, 17–24. [Google Scholar] [CrossRef]
- Neuland, M.B.; Grimaudo, V.; Mezger, K.; Moreno-García, P.; Riedo, A.; Tulej, M.; Wurz, P. Quantitative Measurement of the Chemical Composition of Geological Standards with a Miniature Laser Ablation/Ionization Mass Spectrometer Designed Forin Situapplication in Space Research. Meas. Sci. Technol. 2016, 27, 035904. [Google Scholar] [CrossRef]
- Meyer, S.; Riedo, A.; Neuland, M.B.; Tulej, M.; Wurz, P. Fully Automatic and Precise Data Analysis Developed for Time-of-Flight Mass Spectrometry. J. Mass Spectrom. 2017, 52, 580–590. [Google Scholar] [CrossRef] [PubMed]
- Tulej, M.; Neubeck, A.; Riedo, A.; Lukmanov, R.; Grimaudo, V.; Ligterink, N.F.; Ivarsson, M.; Bach, W.; De Koning, C.; Wurz, P. Isotope Abundance Ratio Measurements Using Femtosecond Laser Ablation Ionization Mass Spectrometry. J. Mass Spectrom. 2020, 55, e4660. [Google Scholar] [CrossRef] [PubMed]
- Fietzke, J.; Liebetrau, V.; Günther, D.; Gürs, K.; Hametner, K.; Zumholz, K.; Hansteen, T.H.; Eisenhauer, A. An Alternative Data Acquisition and Evaluation Strategy for Improved Isotope Ratio Precision Using LA-MC-LCP-MS Applied to Stable and Radiogenic Strontium Isotopes in Carbonates. J. Anal. At. Spectrom. 2008, 23, 955–961. [Google Scholar] [CrossRef]
- Zhang, S.; He, M.; Yin, Z.; Zhu, E.; Hang, W.; Huang, B. Elemental Fractionation and Matrix Effects in Laser Sampling Based Spectrometry. J. Anal. At. Spectrom. 2016, 31, 358–382. [Google Scholar] [CrossRef]
- Becker, J.S. Inorganic Mass Spectrometry: Principles and Applications; John Wiley & Sons, Ltd: Chichester, West Sussex, UK, 2007. [Google Scholar]
- Anderson, F.S.; Alexander, A.; Crow, C.; Whitaker, T.J.; Levine, J. Improved Precision for the Chemistry and Dating Experiment Using Fs-Laser Ablation. In Proceedings of the 82nd Annual Meeting of The Meteoritical Society, Sapporo, Hokkaido, Japan, 7–12 July 2019. [Google Scholar]
- Anderson, F.S.; Levine, J.; Whitaker, T. Dating a Martian Meteorite with 20 Myr Precision Using a Prototype in-Situ Dating Instrument. Planet. Space Sci. 2020, 191, 105007. [Google Scholar] [CrossRef]
- Stephan, T.; Trappitsch, R.; Davis, A.M.; Pellin, M.J.; Rost, D.; Savina, M.R.; Yokochi, R.; Liu, N. CHILI—The Chicago Instrument for Laser Ionization—A New Tool for Isotope Measurements in Cosmochemistry. Int. J. Mass Spectrom. 2016, 407, 1–15. [Google Scholar] [CrossRef]
- Grimaudo, V.; Moreno-García, P.; Riedo, A.; Meyer, S.; Tulej, M.; Neuland, M.B.; Mohos, M.; Gütz, C.; Waldvogel, S.R.; Wurz, P.; et al. Toward Three-Dimensional Chemical Imaging of Ternary Cu–Sn–Pb Alloys Using Femtosecond Laser Ablation/Ionization Mass Spectrometry. Anal. Chem. 2017, 89, 1632–1641. [Google Scholar] [CrossRef] [PubMed]
- Lukmanov, R.A.; Tulej, M.; Wiesendanger, R.; Riedo, A.; Grimaudo, V.; Lighterink, N.F.W.; de Koening, C.; Neubeck, A.; Wacey, D.; Wurz, P. Multiwavelength Abaltion/Ionisation and Mass Spectrometric Analysis of 1.88 Ga Gunflint Chert. Astrobiology 2020. submitted. [Google Scholar]
- Song, K.; Cha, H.K.; Kim, D.Y.; Min, K.Y. Determination of the Isotope Ratio for Metal Samples Using a Laser Ablation/Ionization Time-of-Flight Mass Spectrometry. B Korean Chem. Soc. 2004, 25, 101–105. [Google Scholar]
- Song, K.; Park, Y.; Kim, W. Application of Non-resonant Laser Ionization Mass Spectrometry for a Fast Isotope Analysis of Metal Microparticles. Int. J. Mass Spectrom. 2006, 254, 122–126. [Google Scholar] [CrossRef]
- Joseph, M.; Sivakumar, N.; Manoravi, P.; Balasubramanian, R. Determination of Boron Isotope Ratio in Boron Carbide Using a Laser Mass Spectrometric Method. Rapid Commun. Mass Spectrom. 2004, 18, 231–234. [Google Scholar] [CrossRef] [PubMed]
- Manoravi, P.; Joseph, M.; Sivakumar, N.; Balasubramanian, R. Determination of Isotopic Ratio of Boron in Boric Acid Using Laser Mass Spectrometry. Anal. Sci. 2005, 21, 1453–1455. [Google Scholar] [CrossRef]
- Frey, S.; Wiesendanger, R.; Tulej, M.; Neuland, M.; Riedo, A.; Grimaudo, V.; Moreno-García, P.; López, A.C.; Mohos, M.; Hofmann, B.; et al. Chemical Analysis of a Lunar Meteorite by Laser Ablation Mass Spectrometry. Planet. Space Sci. 2020, 182, 104816. [Google Scholar] [CrossRef]
- Epov, V.N.; Berail, S.; Jimenez-Moreno, M.; Perrot, V.; Pécheyran, C.; Amouroux, D.; Donard, O.F.X. Approach to Measure Isotopic Ratios in Species Using Multicollector-ICPMS Coupled with Chromatography. Anal. Chem. 2010, 82, 5652–5662. [Google Scholar] [CrossRef]
- Shirk, M.D.; Molian, P.A. A Review of Ultrashort Pulsed Laser Ablation of Materials. J. Laser Appl. 1998, 10, 18–28. [Google Scholar] [CrossRef]
- Preuss, S.; Demchuk, A.; Stuke, M. Sub-Picosecond UV Laser Ablation of Metals. Appl. Phys. A 1995, 61, 33–37. [Google Scholar] [CrossRef]
- Margetic, V.; Niemax, K.; Hergenröder, R. Application of Femtosecond Laser Ablation Time-of-Flight Mass Spectrometry to In-Depth Multilayer Analysis. Anal. Chem. 2003, 75, 3435–3439. [Google Scholar] [CrossRef] [PubMed]
- Margetic, V.; Bolshov, M.A.; Stockhaus, A.; Niemax, K.; Hergenroder, R. Depth Profiling of Multi-Layer Samples Using Femtosecond Laser Ablation. J. Anal. At. Spectrom. 2001, 16, 616–621. [Google Scholar] [CrossRef]
- Riedo, A.; Grimaudo, V.; Morenogarcia, P.; Neuland, M.B.; Tulej, M.; Wurz, P.; Broekmann, P. High Depth-Resolution Laser Ablation Chemical Analysis of Additive-Assisted Cu Electroplating for Microchip Architectures. J. Anal. At. Spectrom. 2015, 30, 2371–2374. [Google Scholar] [CrossRef]
- Grimaudo, V.; Moreno-García, P.; Riedo, A.; Neuland, M.B.; Tulej, M.; Broekmann, P.; Wurz, P. High-Resolution Chemical Depth Profiling of Solid Material Using a Miniature Laser Ablation/Ionization Mass Spectrometer. Anal. Chem. 2015, 87, 2037–2041. [Google Scholar] [CrossRef]
- Grimaudo, V.; Moreno-García, P.; López, A.C.; Riedo, A.; Wiesendanger, R.; Tulej, M.; Gruber, C.; Lörtscher, E.; Wurz, P.; Broekmann, P. Depth Profiling and Cross-Sectional Laser Ablation Ionization Mass Spectrometry Studies of Through-Silicon-Vias. Anal. Chem. 2018, 90, 5179–5186. [Google Scholar] [CrossRef]
- López, A.C.; Grimaudo, V.; Moreno-García, P.; Riedo, A.; Tulej, M.; Wiesendanger, R.; Wurz, P.; Broekmann, P. Towards Femtosecond Laser Ablation Ionization Mass Spectrometric Approaches for Chemical Depth-Profiling Analysis of Lead-Free SN Solder Bumps with Minimized Side-wall Contributions. J. Anal. At. Spectrom. 2018, 33, 283–293. [Google Scholar] [CrossRef]
- Käser, D.; Hendriks, L.; Koch, J.; Günther, D. Depth Profile Analyses with sub 100-nm Depth Resolution of a Metal Thin Film by Femtosecond—Laser Ablation—Inductively Coupled Plasma—Time-of-Flight Mass Spectrometry. Spectrochim. Acta Part B: At. Spectrosc. 2018, 149, 176–183. [Google Scholar] [CrossRef]
- Riedo, A.; Grimaudo, V.; López, A.C.; Tulej, M.; Wurz, P.; Broekmann, P. Novel 2D Binning Approach for Advanced LIMS Depth Profiling Analysis. J. Anal. At. Spectrom. 2019, 34, 1564–1570. [Google Scholar] [CrossRef]
- Zou, D.-X.; Yin, Z.-B.; Zhang, B.-C.; Hang, W.; Huang, B.-L. Elemental Analysis of Porcelain Shards by Laser Ionization Time-of-Flight Mass Spectrometry. Chin. J. Anal. Chem. (CHINESE VERSION) 2012, 40, 498–502. [Google Scholar] [CrossRef]
- Lukmanov, R.; Tulej, M.; Lighterink, N.F.W.; de Koening, C.; Riedo, A.; Grimaudo, V.; Neubeck, A.; Wacey, D.; Wurz, P. Chemical Identification of Microfossils from the 1.88 Ga Gunflint Chert. Towards Emirical Biosignatures with LIMS Data. Proc. Nat. Acad. Sci. USA 2020. submitted. [Google Scholar]
- Tong, Q.; Yu, Q.; Jin, X.; He, J.; Hang, W.; Huang, B. Semi-quantitative Analysis of Geological Samples Using Laser Plasma Time-of-Flight Mass Spectrometry. J. Anal. At. Spectrom. 2009, 24, 228–231. [Google Scholar] [CrossRef]
- Sysoev, A.A.; Poteshin, S.S.; Kuznetsov, G.B.; Kovalev, I.A.; Yushkov, E.S. Analysis of Bulk and Powdered Samples Using a LAMAS-10M Laser Ionization Time-of-Flight Mass Spectrometer. J. Anal. Chem. 2002, 57, 811–820. [Google Scholar] [CrossRef]
- Xu, Z.; Hang, L.; Hang, W.; Huang, B. Rapid Analysis of Soil Samples by Laser Ionization Mass Spectrometry. At. Spectrosc. 2020, 41, 147–152. [Google Scholar] [CrossRef]
- Neuland, M.B.; Meyer, S.; Mezger, K.; Riedo, A.; Tulej, M.; Wurz, P. Probing the Allende Meteorite with a Miniature Laser-Ablation Mass Analyser for Space Application. Planet. Space Sci. 2014, 101, 196–209. [Google Scholar] [CrossRef]
- Neuland, M.; Metzger, K.; Riedo, A.; Tulej, M.; Wurz, P. The Chemical Composition and Homogeneity of the Allende Matrix. Planet Space Sci. 2021. submitted. [Google Scholar]
- Grimaudo, V.; Moreno-García, P.; Riedo, A.; López, A.C.; Tulej, M.; Wiesendanger, R.; Wurz, P.; Broekmann, P. Review—Laser Ablation Ionization Mass Spectrometry (LIMS) for Analysis of Electrodeposited Cu Interconnects. J. Electrochem. Soc. 2019, 166, D3190–D3199. [Google Scholar] [CrossRef]
- Moreno-García, P.; Grimaudo, V.; Riedo, A.; Tulej, M.; Neuland, M.B.; Wurz, P.; Broekmann, P. Towards Structural Analysis of Polymeric Contaminants in Electrodeposited Cu films. Electrochimica Acta 2016, 199, 394–402. [Google Scholar] [CrossRef]
- Moreno-García, P.; Grimaudo, V.; Riedo, A.; López, A.C.; Wiesendanger, R.; Tulej, M.; Gruber, C.; Lörtscher, E.; Wurz, P.; Broekmann, P. Insights into Laser Ablation Processes of Heterogeneous Samples: Toward Analysis of Through-Silicon-Vias. Anal. Chem. 2018, 90, 6666–6674. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Xiao, Y.; Zhang, S.; Liu, R.; Hang, W.; Huang, B. Composition Analysis of Ancient Celadon via Femtosecond Laser Ionization Time-of-Flight Mass Spectrometry. Appl. Surf. Sci. 2015, 351, 624–634. [Google Scholar] [CrossRef]
- Chen, L.; Lin, L.; Yu, Q.; Yan, X.; Hang, W.; He, J.; Huang, B. Semiquantitative Multielemental Analysis of Biological Samples by a Laser Ionization Orthogonal Time-of-Flight Mass Spectrometer. J. Am. Soc. Mass Spectrom. 2009, 20, 1355–1358. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gao, Y.; Lin, Y.; Zhang, B.; Zou, D.; He, M.; Dong, B.; Hang, W.; Huang, B. Single-Cell Elemental Analysis via High Irradiance Femtosecond Laser Ionization Time-of-Flight Mass Spectrometry. Anal. Chem. 2013, 85, 4268–4272. [Google Scholar] [CrossRef]
- Bleiner, D.; Trottmann, M.; Cabas-Vidani, A.; Wichser, A.; Romanyuk, Y.E.; Tiwari, A.N. Correction to: XUV Laser Mass Spectrometry for Nano-Scale 3d Elemental Profiling of Functional Thin Films. Appl. Phys. A 2020, 126, 1. [Google Scholar] [CrossRef]
- Kim, J.; Lee, S.; Lee, S.; Jung, Y.; Oh, K. Bessel Beam Generating Hybrid Polymer-Fiber Lens under Various Fabricating Structures. In Proceedings of the 2009 Conference on Lasers & Electro Optics & The Pacific Rim Conference on Lasers and Electro-Optics, Shangai, China, 30 August–9 September 2009; Volumes 1 and 2, pp. 1–2. [Google Scholar]
- Kim, J.K.; Kim, J.; Jung, Y.; Ha, W.; Jeong, Y.S.; Lee, S.; Tünnermann, A.; Oh, K. Compact All-Fiber Bessel Beam Generator Based on Hollow Optical Fiber Combined with a Hybrid Polymer Fiber Lens. Opt. Lett. 2009, 34, 2973–2975. [Google Scholar] [CrossRef] [PubMed]


| Element | A (u) | Tm (K) | R (%) | IP (eV) | Y0 (1011ions/cm2) |
|---|---|---|---|---|---|
| Mg | 24.30 | 923 | … | 7.65 | 5.7 |
| Al | 26.98 | 933 | 86.8 | 5.99 | 4.9 |
| Cr | 51.99 | 2180 | 56.8 | 6.77 | 3.8 |
| Fe | 55.84 | 1811 | 56.12 | 7.90 | 4.5 |
| Cu | 63.54 | 1357 | 96.3 | 7.73 | 1.9 |
| W | 183.84 | 3695 | 49.6 | 7.86 | 2.4 |
| Ti:Sapphire [19] (Spectra-Physics) | Ti:Sapphire [21] (Clark-MXR, Inc.) | Ytterbium-Doped Semiconductor Laser S-Pulse, Amplitude Systems [20] | |
|---|---|---|---|
| wavelength | 800 nm | 775 nm SHG, THG | 1030 nm |
| duration | 45 fs (effect. 75 fs) | 180 fs | 500 fs |
| repetition | 1 kHz | 1 kHz | 10 Hz |
| output, max. | 2 mJ | 1.2 mJ | 1 mJ |
| Spot dia. | 8 μm | 5–40 μm | 40 μm |
| DIN 1716 | LMS | ICP-MS | LA-ICP-MS | ||
|---|---|---|---|---|---|
| CuSn10Pb10 | Cu | 78.0–82.0 | 78.61 ± 11.87 | 82.26 ± 4.13 | 76.99 ± 0.75 |
| Sn | 9.0–11.0 | 9.94 ± 2.45 | 6.69 ± 0.79 | 10.91 ± 0.19 | |
| Pb | 8.0–11.0 | 12.25 ± 3.28 | 11.13 ± 0.63 | 8.56 ± 0.47 | |
| CuSn7Pb15 | Cu | 75.0–79.0 | 77.93 ± 11.77 | 75.55 ± 3.73 | 70.67 ± 1.03 |
| Sn | 7.0–9.0 | 5.65 ± 1.39 | 6.56 ± 0.40 | 7.30 ± 0.06 | |
| Pb | 13.0–17.0 | 14.89 ± 3.99 | 19.45 ± 0.57 | 18.42 ± 1.04 | |
| CuSn5Pb20 | Cu | 69.0–76.0 | 76.37 ± 11.53 | 75.63 ± 3.39 | 74.94 ± 0.68 |
| Sn | 4.0–6.0 | 5.99 ± 1.48 | 7.08 ± 0.73 | 6.92 ± 0.11 | |
| Pb | 18.0–23.0 | 15.63 ± 4.19 | 20.59 ± 1.25 | 16.59 ± 0.62 |
| Metal | Threshold Fluence (mJ/cm2) | Depth per Pulse (nm) |
|---|---|---|
| Ni | 100 | 15 |
| In | 125 | 90 |
| Cu | 175 | 8 |
| Mo | 150 | 6 |
| Au | 210 | 17 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tulej, M.; Ligterink, N.F.W.; de Koning, C.; Grimaudo, V.; Lukmanov, R.; Keresztes Schmidt, P.; Riedo, A.; Wurz, P. Current Progress in Femtosecond Laser Ablation/Ionisation Time-of-Flight Mass Spectrometry. Appl. Sci. 2021, 11, 2562. https://doi.org/10.3390/app11062562
Tulej M, Ligterink NFW, de Koning C, Grimaudo V, Lukmanov R, Keresztes Schmidt P, Riedo A, Wurz P. Current Progress in Femtosecond Laser Ablation/Ionisation Time-of-Flight Mass Spectrometry. Applied Sciences. 2021; 11(6):2562. https://doi.org/10.3390/app11062562
Chicago/Turabian StyleTulej, Marek, Niels F.W. Ligterink, Coenraad de Koning, Valentine Grimaudo, Rustam Lukmanov, Peter Keresztes Schmidt, Andreas Riedo, and Peter Wurz. 2021. "Current Progress in Femtosecond Laser Ablation/Ionisation Time-of-Flight Mass Spectrometry" Applied Sciences 11, no. 6: 2562. https://doi.org/10.3390/app11062562
APA StyleTulej, M., Ligterink, N. F. W., de Koning, C., Grimaudo, V., Lukmanov, R., Keresztes Schmidt, P., Riedo, A., & Wurz, P. (2021). Current Progress in Femtosecond Laser Ablation/Ionisation Time-of-Flight Mass Spectrometry. Applied Sciences, 11(6), 2562. https://doi.org/10.3390/app11062562







