Thermal Conductivity of Nanofluids: A Review on Prediction Models, Controversies and Challenges
Abstract
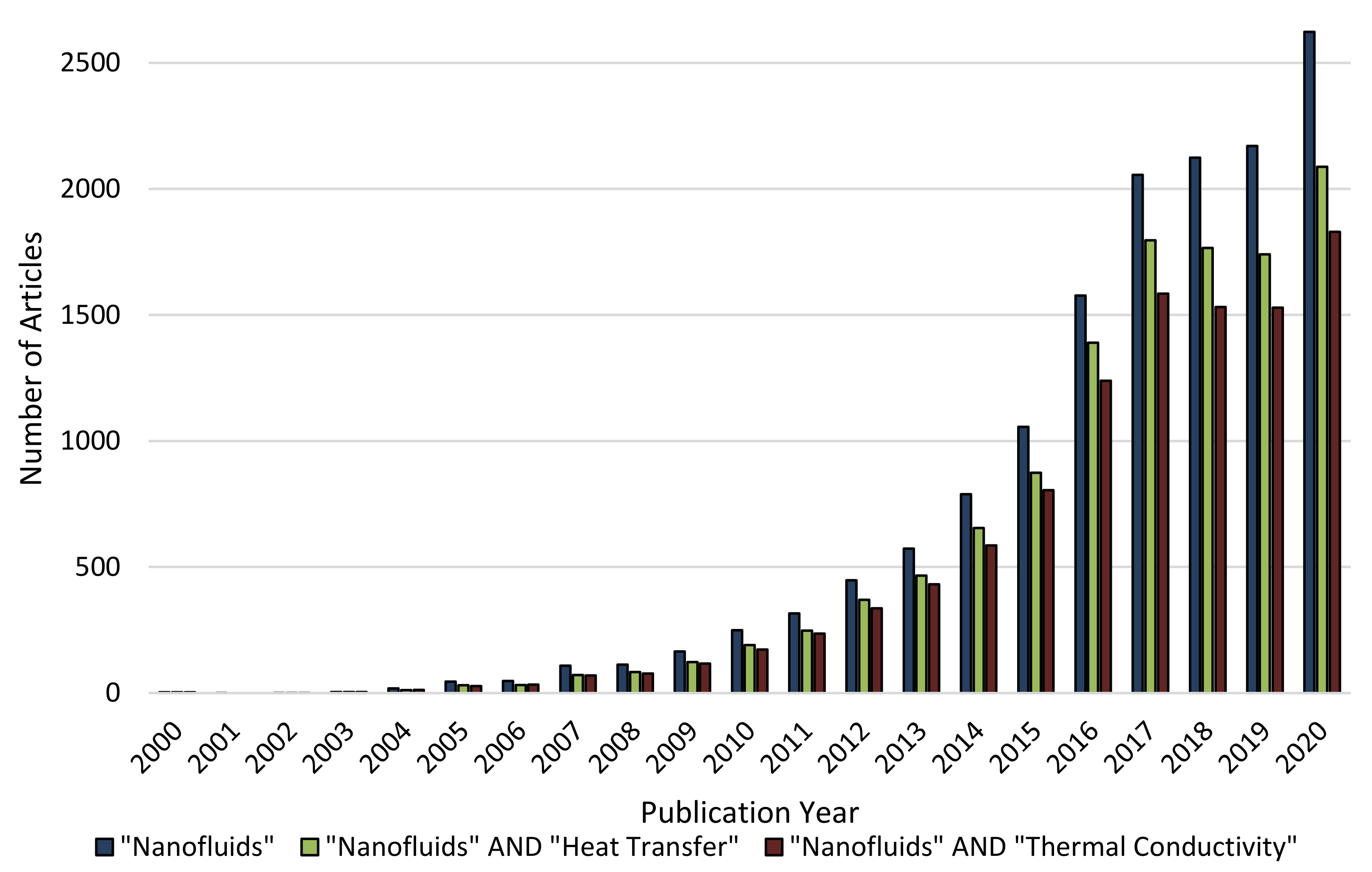
1. Introduction
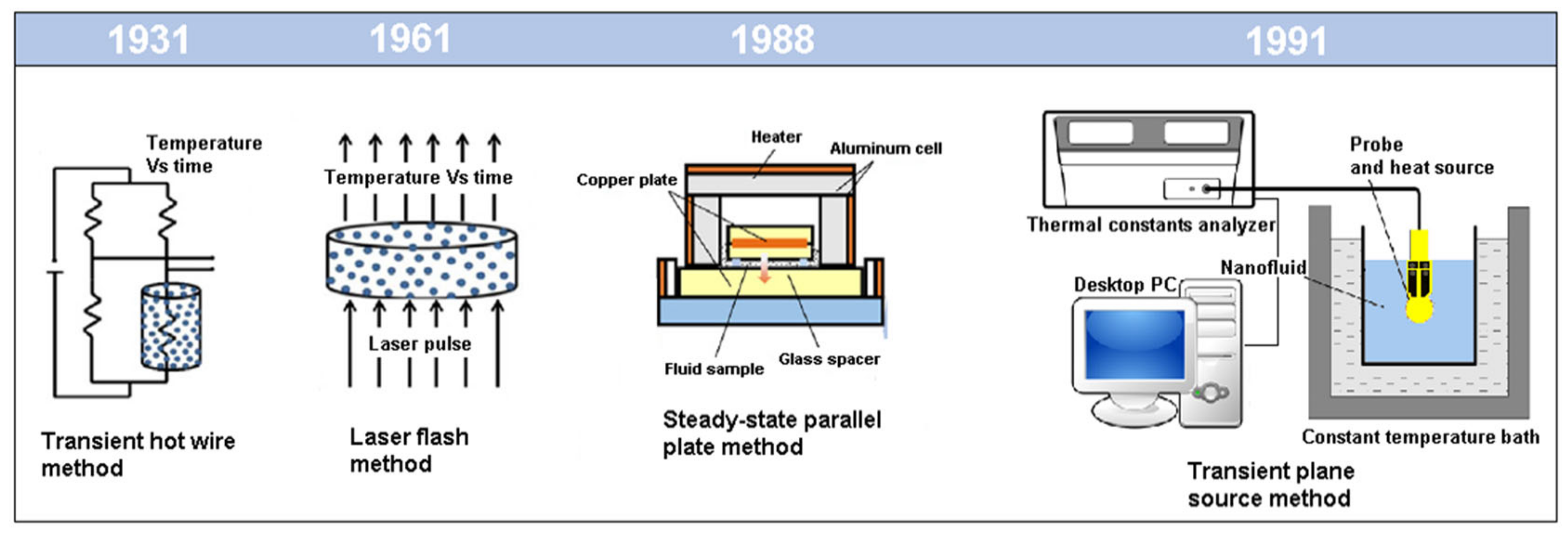
2. Thermal Conductivity Measurements Overview
2.1. Transient Hot Wire Method
2.2. Transient Plane Source Theory
2.3. Temperature Oscillation
2.4. 3ω Method
2.5. Laser Flash Method
2.6. Parallel Plate
2.7. Coaxial Cylinders
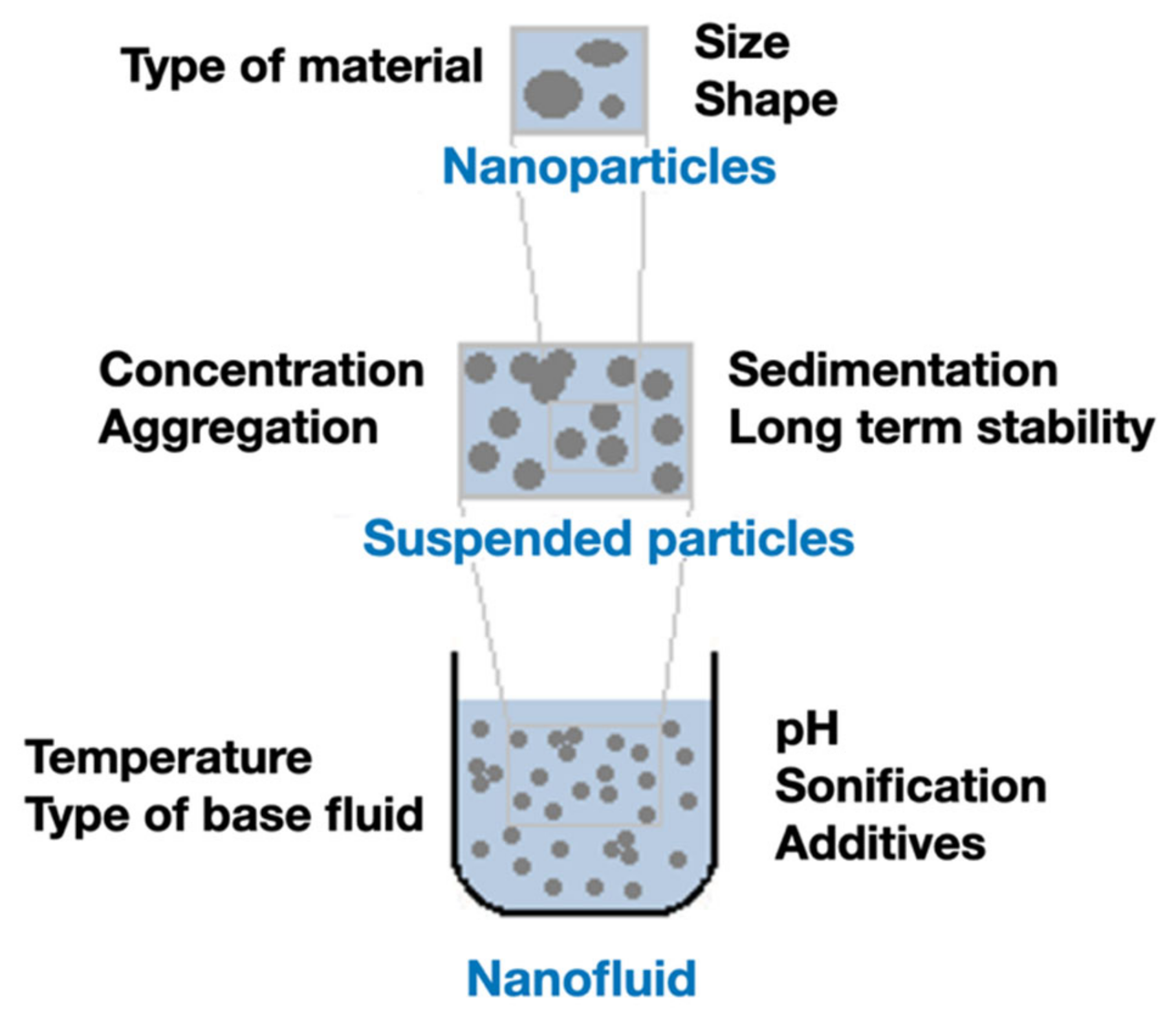
3. Parameters That Affect Thermal Conductivity
3.1. Nanoparticles (NPs)
3.1.1. Type
3.1.2. Size
3.1.3. Shape
3.2. Suspension
3.2.1. Base Fluids
3.2.2. Concentration
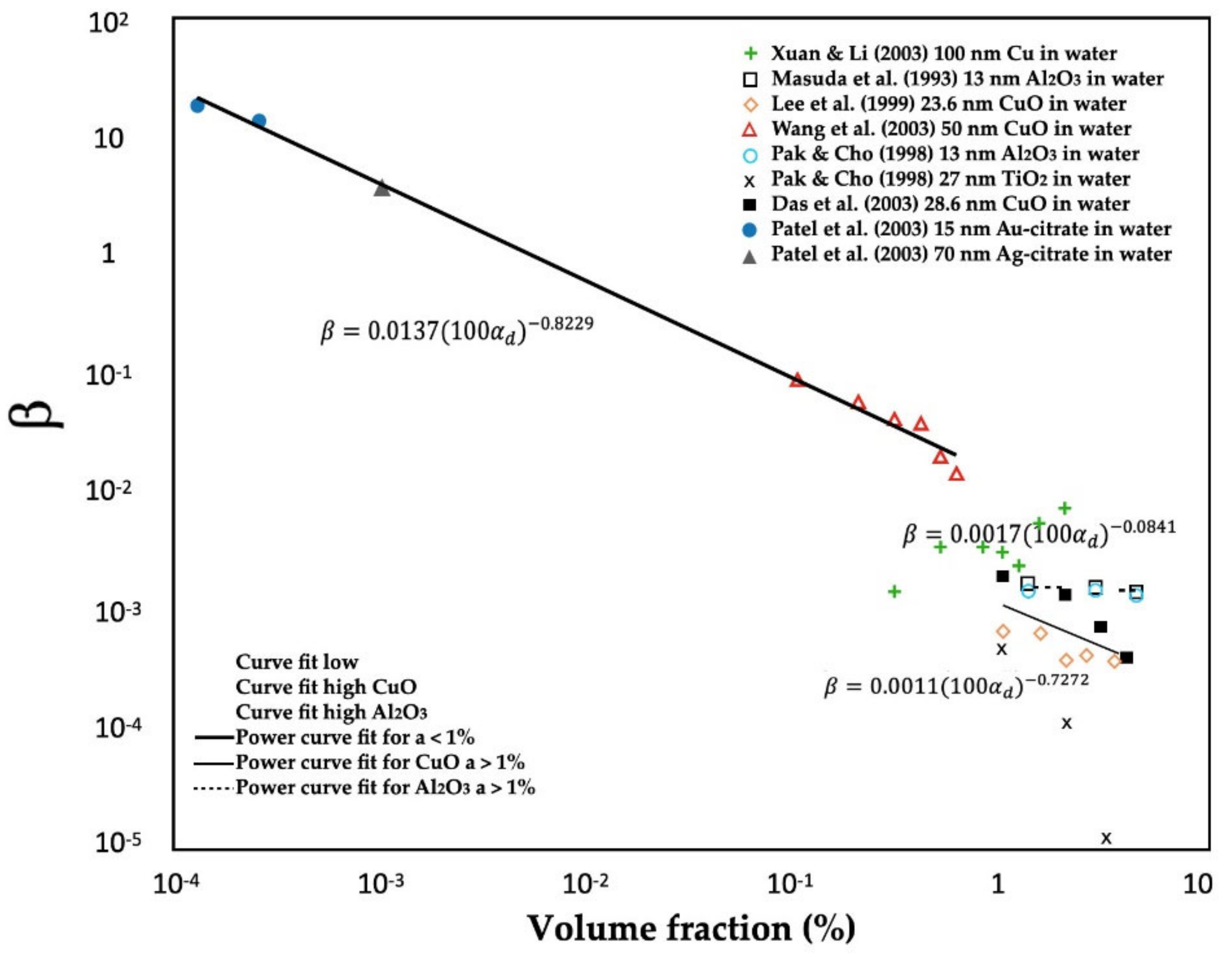
3.2.3. Agglomeration
3.3. Nanofluids
3.3.1. Preparation Methods
3.3.2. Temperature
3.3.3. pH
3.3.4. Additives, Surfactants and Solvents
3.3.5. Sonication Time
3.3.6. External Magnetic Field
3.3.7. Aggregation
4. Prediction Models for Thermal Conductivity and Other Properties of the NFs
4.1. Thermal Conductivity Empirical Models
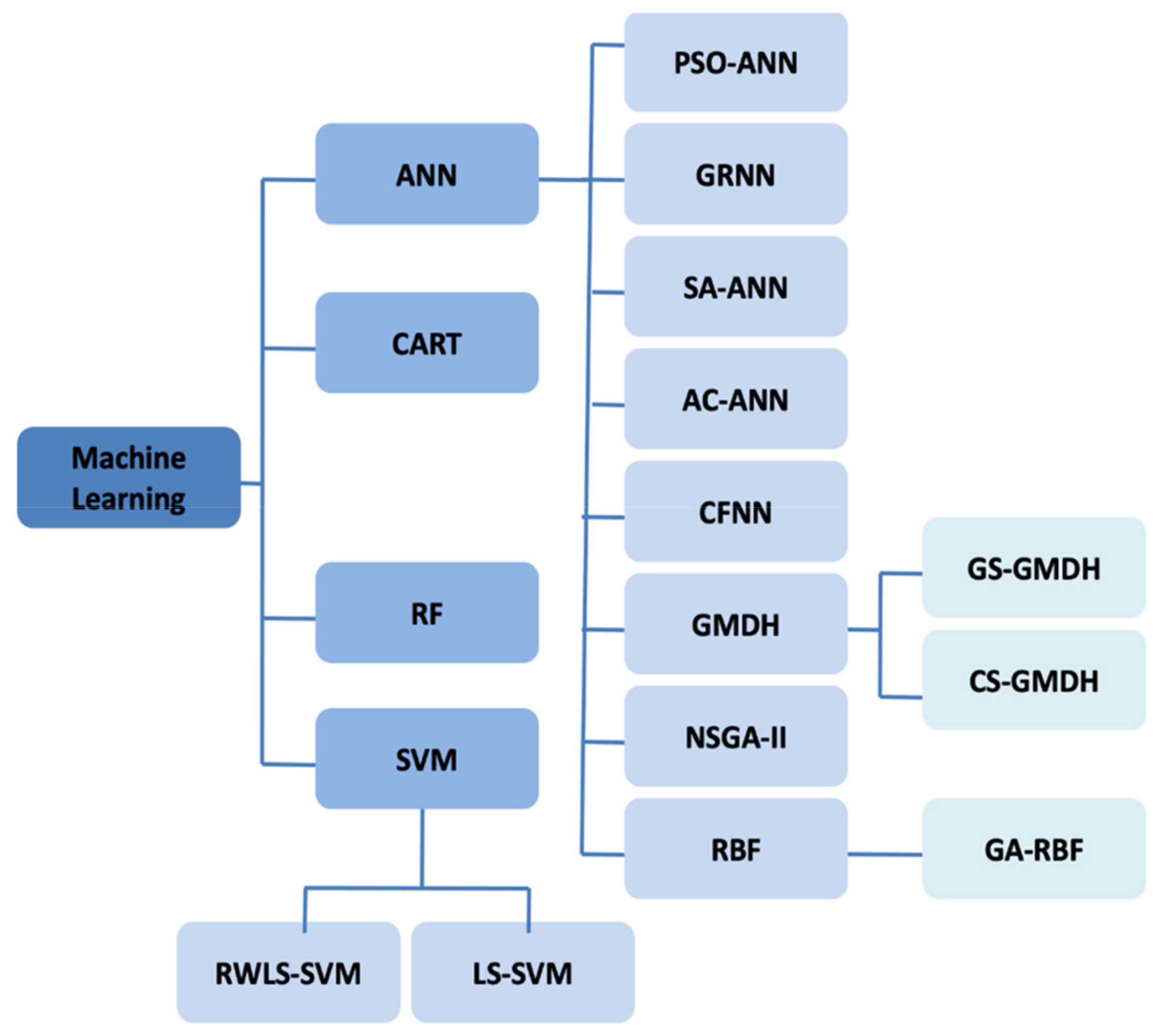
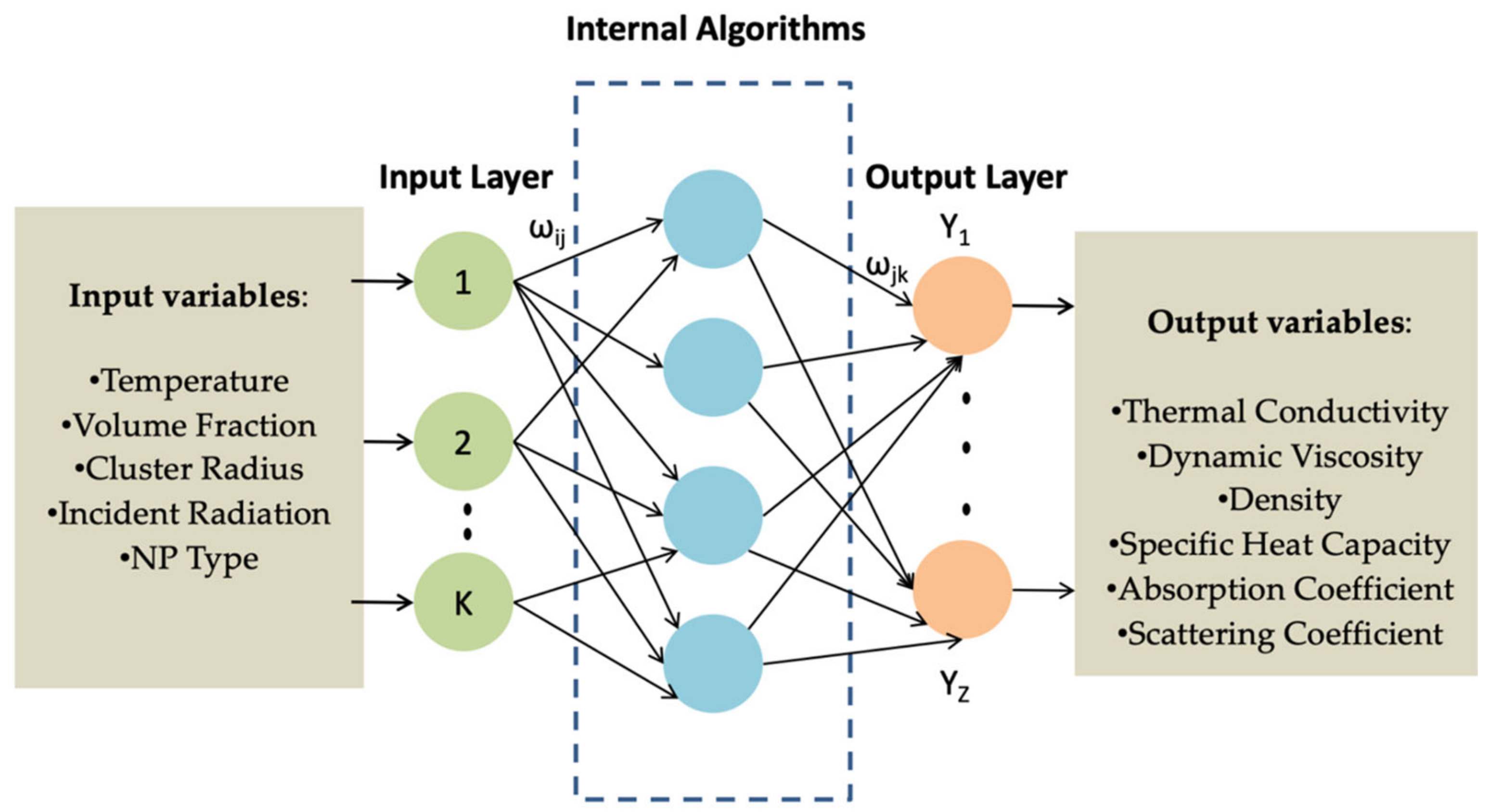
4.2. Thermal Conductivity Machine Learning-Based Models
4.3. Density
4.4. Specific Heat Capacity
4.5. Viscosity
5. Controversies in Thermal Conductivity Measurements
6. Backdraws and Future
- Complexity and high cost of NFs preparation;
- Long-term stable NFs are difficult to produce. The influence of the production should be further studied, namely the sonication time, volume fraction, and type of the NFS in order to avoid the sedimentation and agglomeration of the NPs in the BF and to achieve optimal performance;
- Lack of a common protocol for the manufacture and analysis of the thermal transport mechanisms in this type of fluids;
- Methods to scale-up production for commercialization are still in development;
- Some of the classical experimental fitting prediction models for thermal properties are not the most suitable for the estimation of the thermal conductivity, dynamic viscosity and density of the NFs;
- The classical modeling does not provide a fast prediction of the thermal properties, which may slow down the study and overall applicability of the NFs;
- Nowadays, it is clear the need to use the recent statistical data-driven machine learning models to obtain faster predictions of the thermal properties of the NFs. Those models are also more stable and sensitive than the empirical ones. However, for a in-depth knowledge on how the recent models work, the researchers must get proficient skills in machine learning methodology and in the most common data-driven models and algorithms;
- The proper machine learning model and algorithms should be chosen according to the dimension of the sample, the available computing resources, and the required prediction performance (modeling stability and sensibility).
- The stability and durability of the nanofluids should be improved by optimizing the concentration of NPs and base fluid characteristics (e.g., chemical, viscosity). The stability of nanofluids should also be predicted by further analysis of the surface tension of the nanofluid vs. time;
- The general properties of the NFs should be improved by optimizing the preparation procedures (e.g., sonication time);
- The influence of the solvents should be further studied: the use of high polar solvents like the DMF (Dimethylformamide) and THF (Tetrahydrofuran) and non-polar solvents as hexane and heptane could be the right way to fully understand how polarity influences the alignment and thermal conductivity of the NFs;
- The influence of the polarity in the alignment of the NPS on the base fluid can also be assessed by the use of different surfactants with negative charge, such as for instance CATB (Cetyltrimethylamonium bromide);
- The impact of an applied magnetic field should be further studied, namely the value or range of values that make possible to reach the thermal conductivity maximum peak in less time;
- Additives for decreasing the NFs viscosity while maintaining the same level of thermal conductivity are of paramount importance to achieve the best performance of the NFs;
- The systems using nanofluids as working fluid should become more cost-effective, without the need of extra pumping power and expensive maintenance. This should be accomplished with the optimization of the microchannels configuration (e.g. number of channels, inlet/outlet positioning);
- Machine learning prediction models should be increasingly used in the future, since they provide faster and less expensive modeling of thermal properties of the NFs. Those data-driven models achieve more accurate and stable estimations than the classical ones. The path created by machine learning is beginning to clear up several doubts and backdraws, and it is a secure one for future studies and developments on the nanomaterials field of research.
7. Conclusions
- the thermal conductivity of an NF is greater than the one of the respective base fluids;
- the methods used to measure the thermal properties of NFs are still in an embryonic stage of development;
- the heat transfer coefficient of an NF is higher than the one of the respective base fluid;
- viscosity of NFs increases with the concentration of the NPs leading to higher pumping power requirements;
- NF long-term stability is mandatory for miniaturized fluidic systems, mainly mini or microchannels devices;
- the production costs for systems using nanofluids as working fluid are still high.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AC-ANN | Ant Colony Artificial Neural Network |
| ANN | Artificial Neural Network |
| CART | Category and Regression Tree |
| CS-GMDH | Group Method of Data Handling |
| CFNN | Correlation Filter Neural Network |
| GA-RBF | Genetic Algorithm Radial Basis Function |
| GMDH | Group Method of Data Handling |
| GRNN | General Regression Neural Network |
| GS-GMDH | Generalized Structure Group Method of Data Handling |
| LS-SVM | Least Square Support Vector Machine |
| NSGA II | Non-dominated Sorting Genetic Algorithm |
| PSO-ANN | Particle Sworn Optimization Artificial Neural Network |
| RBF | Radial Basis Function |
| RF | Random Forest |
| RWLS-SVM | Recursive Weighted Least Squares Support Vector Machine |
| SA-ANN | Simulate Anneal Artificial Neural Network |
| SVM | Support Vector Machine |
References
- Garimella, S.V.; Persoons, T.; Weibel, J.A.; Gektin, V. Electronics Thermal Management in Information and Communications Technologies: Challenges and Future Directions. IEEE Trans. Compon. Packag. Manuf. Technol. 2016, 7, 1191–1205. [Google Scholar] [CrossRef]
- Ghadimi, A.; Saidur, R.; Metselaar, H.S.C. A review of nanofluid stability properties and characterization in stationary conditions. Int. J. Heat Mass Transf. 2011, 54, 4051–4068. [Google Scholar] [CrossRef]
- Pang, C.; Lee, J.W.; Kang, Y.T. Review on combined heat and mass transfer characteristics in nanofluids. Int. J. Therm. Sci. 2015, 87, 49–67. [Google Scholar] [CrossRef]
- Younes, H.; Christensen, G.; Li, D.; Hong, H.; Ghaferi, A.A. Thermal Conductivity of Nanofluids: Review. J. Nanofluids 2015, 4, 107–132. [Google Scholar] [CrossRef]
- Qiu, L.; Zhu, N.; Feng, Y.; Michaelides, E.E.; Żyła, G.; Jing, D.; Zhang, X.; Norris, P.M.; Markides, C.N.; Mahian, O. A review of recent advances in thermophysical properties at the nanoscale: From solid state to colloids. Phys. Rep. 2020, 843, 1–81. [Google Scholar] [CrossRef]
- Elcock, D. Potential Impacts of Nanotechnology on Energy Transmission Applications and Needs; Environmental Science Division; Argonne National Laboratory: Argonne, Il, USA, 2007. [Google Scholar]
- Ali, H.M.; Babar, H.; Shah, T.R.; Sajid, M.U.; Qasim, M.A.; Javed, S. Prepartion Techniques of TiO2 Nanofluids and Challenges: A Review. Appl. Sci. 2018, 8, 587. [Google Scholar]
- Maia, I.; Rocha, C.; Pontes, P.; Cardoso, V.M.; Miranda, J.S.; Moita, A.; Minas, G.L.N.; Moreira, A.; Lima, R. Heat Transfer and Fluid Flow Investigations in PDMS Microchannel Heat Sinks Fabricated by Means of a Low-Cost 3D Printer. In Advances in Microfluidic Technologies for Energy and Environmental Applications; IntechOpen: London, UK, 2020. [Google Scholar]
- Freitas, E.; Pontes, P.; Cautela, R.; Bahadur, V.; Miranda, J.; Ribeiro, A.P.C.; Souza, R.R.; Oliveira, J.D.; Copetti, J.B.; Lima, R.; et al. Pool Boiling of Nanofluids on Biphilic Surfaces: An Experimental and Numerical Study. Nanomaterials 2021, 11, 125. [Google Scholar] [CrossRef] [PubMed]
- Nagpal, S. Nanofluids to be used to make new types of cameras, microdevices, and displays. Available online: https://topnews.in/nanofluids-be-used-make-new-types-cameras-microdevices-and-displays-221378 (accessed on 12 January 2021).
- Serrano, E.; Rus, G.; García-Martínez, J. Nanotechnology for sustainable energy. Renew. Sustain. Energy Rev. 2009, 13, 2373–2384. [Google Scholar] [CrossRef]
- Saidur, R.; Leong, K.Y.Y.; Mohammed, H.A.A. A review on applications and challenges of nanofluids. Renew. Sustain. Energy Rev. 2011, 15, 1646–1668. [Google Scholar] [CrossRef]
- Buongiorno, J.; Hu, L.-W. 8. Innovative Technologies: Two-Phase Heat Transfer in Water-Based Nanofluids for Nuclear Applications Final Report; Massachusetts Institute of Technology: Cambridge, MA, USA, 2009. [Google Scholar]
- Kuo, K.K.; Risha, G.A.; Evans, B.J.; Boyer, E. Potential Usage of Energetic Nano-sized Powders for Combustion and Rocket Propulsion. MRS Proc. 2003, 800, AA1.1. [Google Scholar] [CrossRef]
- Srikant, R.R.; Rao, D.N.; Subrahmanyam, M.S.; Krishna, P.V. Applicability of cutting fluids with nanoparticle inclusion as coolants in machining. Proc. Inst. Mech. Eng. Part J. J. Eng. Tribol. 2009, 223, 221–225. [Google Scholar] [CrossRef]
- Wilson, C.A. Experimental Investigation of Nanofluid Oscillating Heat Pipes. Ph.D. Dissertation, University of Missouri, Columbia, MO, USA, 2006. [Google Scholar]
- Grab, T.; Gross, U.; Franzke, U.; Buschmann, M.H. Operation performance of thermosyphons employing titania and gold nanofluids. Int. J. Therm. Sci. 2014, 86, 352–364. [Google Scholar] [CrossRef]
- Tyagi, H.; Phelan, P.; Prasher, R. Predicted efficiency of a Low-temperature Nanofluid-based direct absorption solar collector. J. Sol. Energy Eng. Trans. ASME 2009, 131, 0410041–0410047. [Google Scholar] [CrossRef]
- Demetzos, C. Application of Nanotechnology in Imaging and Diagnostics. In Pharmaceutical Nanotechnology: Fundamentals and Practical Applications; Springer: Singapore, 2016; pp. 65–75. ISBN 978-981-10-0791-0. [Google Scholar]
- Rodrigues, R.O.; Sousa, P.; Gaspar, J.; Bañobre-López, M.; Lima, R.; Minas, G. Organ-on-a-chip: A Preclinical Microfluidic Platform for the Progress of Nanomedicine. Small 2020, 2003517. [Google Scholar] [CrossRef] [PubMed]
- Kung, C.T.; Gao, H.; Lee, C.Y.; Wang, Y.N.; Dong, W.; Ko, C.H.; Wang, G.; Fu, L.M. Microfluidic synthesis control technology and its application in drug delivery, bioimaging, biosensing, environmental analysis and cell analysis. Chem. Eng. J. 2020, 399, 125748. [Google Scholar] [CrossRef]
- Rostami, S.; Aghakhani, S.; Pordanjani, A.H.; Afrand, M.; Cheraghian, G.; Oztop, H.F.; Shadloo, M.S. A review on the Control Parameters of Natural Convection in Different Shaped Cavities with and without nanofluids. Processes 2020, 8, 1011. [Google Scholar] [CrossRef]
- Gupta, M.; Singh, V.; Kumar, R.; Said, Z. A review on thermophysical properties of nanofluids and heat transfer applications. Renew. Sustain. Energy Rev. 2017, 74, 638–670. [Google Scholar] [CrossRef]
- Xu, G.; Fu, J.; Dong, B.; Quan, Y.; Song, G. A novel method to measure thermal conductivity of nanofluids. Int. J. Heat Mass Transf. 2019, 130, 978–988. [Google Scholar] [CrossRef]
- Bhattacharya, P.; Nara, S.; Vijayan, P.; Tang, T.; Lai, W.; Phelan, P.E.E.; Prasher, R.S.S.; Song, D.W.W.; Wang, J. Characterization of the temperature oscillation technique to measure the thermal conductivity of fluids. Int. J. Heat Mass Transf. 2006, 49, 2950–2956. [Google Scholar] [CrossRef]
- Philip, J.; Shima, P.D. Thermal properties of nanofluids. Adv. Colloid Interface Sci. 2012, 183–184, 30–45. [Google Scholar] [CrossRef]
- Wang, X.; Zhu, D.; Yang, S. Investigation of pH and SDBS on enhancement of thermal conductivity in nanofluids. Chem. Phys. Lett. 2009, 470, 107–111. [Google Scholar] [CrossRef]
- Yoo, D.-H.; Hong, K.S.; Yang, H.-S. Study of thermal conductivity of nanofluids for the application of heat transfer fluids. Thermochim. Acta 2007, 455, 66–69. [Google Scholar] [CrossRef]
- Ambreen, T.; Kim, M.H. Influence of particle size on the effective thermal conductivity of nanofluids: A critical review. Appl. Energy 2020, 264. [Google Scholar] [CrossRef]
- Xu, J.; Yu, B. A new model for heat conduction of nanofluids based on fractal distributions of nanoparticles. J. Phys. D Appl. Phys. 2008, 41, 139801. [Google Scholar] [CrossRef]
- Anoop, K.B.; Sundararajan, T.; Das, S.K. Effect of particle size on the convective heat transfer in nanofluid in the developing region. Int. J. Heat Mass Transf. 2009, 52, 2189–2195. [Google Scholar] [CrossRef]
- Pil Jang, S.; Choi, S.U.S. Effects of Various Parameters on Nanofluid Thermal Conductivity. J. Heat Transfer 2006, 129, 617–623. [Google Scholar] [CrossRef]
- Timofeeva, E.; Smith, D.; Yu, W.; France, D.; Singh, D.; Routbort, J. Particle size and interfacial effects on thermo-physical and heat transfer characteristics of water-basedalpha-SiC nanofluids. Nanotechnology 2010, 21, 215703. [Google Scholar] [CrossRef] [PubMed]
- Murshed, S.M.S.; Leong, K.C.; Yang, C. Enhanced thermal conductivity of TiO2—Water based nanofluids. Int. J. Therm. Sci. 2005, 44, 367–373. [Google Scholar] [CrossRef]
- Jeong, J.; Li, C.; Kwon, Y.; Lee, J.; Kim, S.H.; Yun, R. Particle shape effect on the viscosity and thermal conductivity of ZnO nanofluids. Int. J. Refrig. 2013, 36, 2233–2241. [Google Scholar] [CrossRef]
- Timofeeva, E.V.; Routbort, J.L.; Singh, D. Particle shape effects on thermophysical properties of alumina nanofluids. J. Appl. Phys. 2009, 106, 14304. [Google Scholar] [CrossRef]
- Glory, J.; Bonetti, M.; Helezen, M.; Mayne-L’Hermite, M.; Reynaud, C. Thermal and electrical conductivities of water-based nanofluids prepared with long multiwalled carbon nanotubes. J. Appl. Phys. 2008, 103, 94309. [Google Scholar] [CrossRef]
- Gowda, R.; Sun, H.; Wang, P.; Charmchi, M.; Gao, F.; Gu, Z.; Budhlall, B. Effects of Particle Surface Charge, Species, Concentration, and Dispersion Method on the Thermal Conductivity of Nanofluids. Adv. Mech. Eng. 2010, 2, 807610. [Google Scholar] [CrossRef]
- Chen, L.; Xie, H.; Li, Y.; Yu, W. Nanofluids containing carbon nanotubes treated by mechanochemical reaction. Thermochim. Acta 2008, 477, 21–24. [Google Scholar] [CrossRef]
- Hwang, Y.-J.J.; Ahn, Y.C.; Shin, H.S.; Lee, C.G.; Kim, G.T.; Park, H.S.; Lee, J.K. Investigation on characteristics of thermal conductivity enhancement of nanofluids. Curr. Appl. Phys. 2006, 6, 1068–1071. [Google Scholar] [CrossRef]
- Esfe, M.H.; Esfandeh, S.; Afrand, M.; Rejvani, M.; Rostamian, S.H. Experimental evaluation, new correlation proposing and ANN modeling of thermal properties of EG based hybrid nanofluid containing ZnO-DWCNT nanoparticles for internal combustion engines applications. Appl. Therm. Eng. 2018, 133, 452–463. [Google Scholar] [CrossRef]
- Hemmat Esfe, M.; Yan, W.-M.; Akbari, M.; Karimipour, A.; Hassani, M. Experimental study on thermal conductivity of DWCNT-ZnO/water-EG nanofluids. Int. Commun. Heat Mass Transf. 2015, 68, 248–251. [Google Scholar] [CrossRef]
- Rostamian, S.H.; Biglari, M.; Saedodin, S.; Hemmat Esfe, M. An inspection of thermal conductivity of CuO-SWCNTs hybrid nanofluid versus temperature and concentration using experimental data, ANN modeling and new correlation. J. Mol. Liq. 2017, 231, 364–369. [Google Scholar] [CrossRef]
- Xuan, Y.; Li, Q. Heat transfer enhancement of nanofluids. Int. J. Heat Fluid Flow 2000, 21, 58–64. [Google Scholar] [CrossRef]
- Jana, S.; Salehi-Khojin, A.; Zhong, W.-H. Enhancement of fluid thermal conductivity by the addition of single and hybrid nano-additives. Thermochim. Acta 2007, 462, 45–55. [Google Scholar] [CrossRef]
- Choi, S.U.S.; Li, S.; Eastman, J.A. Measuring thermal conductivity of fluids containing oxide nanoparticles. J. Heat Transf. 1999, 121, 280–289. [Google Scholar]
- Hong, J.; Kim, D. Effects of aggregation on the thermal conductivity of alumina/water nanofluids. Thermochim. Acta 2012, 542, 28–32. [Google Scholar] [CrossRef]
- Keblinski, P.; Eastman, J.A.; Cahill, D.G. Nanofluids for thermal transport. Mater. Today 2005, 8, 36–44. [Google Scholar] [CrossRef]
- Li, Q.; Xuan, Y.; Wang, J. Experimental investigations on transport properties of magnetic fluids. Exp. Therm. Fluid Sci. 2005, 30, 109–116. [Google Scholar] [CrossRef]
- Nasiri, A.; Shariaty-Niasar, M.; Rashidi, A.; Amrollahi, A.; Khodafarin, R. Effect of dispersion method on thermal conductivity and stability of nanofluid. Exp. Therm. Fluid Sci. 2011, 35, 717–723. [Google Scholar] [CrossRef]
- Devendiran, D.K.; Amirtham, V.A. A review on preparation, characterization, properties and applications of nanofluids. Renew. Sustain. Energy Rev. 2016, 60, 21–40. [Google Scholar] [CrossRef]
- Salari, S.; Jafari, S.M. Application of nanofluids for thermal processing of food products. Trends Food Sci. Technol. 2020, 97, 100–113. [Google Scholar] [CrossRef]
- Chon, C.H.; Kihm, K.D.; Lee, S.P.; Choi, S.U.S. Empirical correlation finding the role of temperature and particle size for nanofluid (Al2O3) thermal conductivity enhancement. Appl. Phys. Lett. 2005, 87, 153107. [Google Scholar] [CrossRef]
- Mintsa, H.A.; Roy, G.; Nguyen, C.T.; Doucet, D. New temperature dependent thermal conductivity data for water-based nanofluids. Int. J. Therm. Sci. 2009, 48, 363–371. [Google Scholar] [CrossRef]
- Shalkevich, N.; Escher, W.; Bürgi, T.; Michel, B.; Si-Ahmed, L.; Poulikakos, D. On the Thermal Conductivity of Gold Nanoparticle Colloids. Langmuir 2010, 26, 663–670. [Google Scholar] [CrossRef] [PubMed]
- Duangthongsuk, W.; Wongwises, S. Measurement of temperature-dependent thermal conductivity and viscosity of TiO2-water nanofluids. Exp. Therm. Fluid Sci. 2009, 33, 706–714. [Google Scholar] [CrossRef]
- Krishnakumar, T.S.; Viswanath, S.P.; Varghese, S.M. Experimental studies on thermal and rheological properties of Al2O3–ethylene glycol nanofluid. Int. J. Refrig. 2018, 89, 122–130. [Google Scholar]
- Li, X.F.; Zhu, D.S.; Wang, X.J.; Wang, N.; Gao, J.W.; Li, H. Thermal conductivity enhancement dependent pH and chemical surfactant for Cu-H2O nanofluids. Thermochim. Acta 2008, 469, 98–103. [Google Scholar] [CrossRef]
- Wang, X.; Li, X.; Yang, S. Influence of pH and SDBS on the Stability and Thermal Conductivity of Nanofluids. Energy Fuels 2009, 23, 2684–2689. [Google Scholar] [CrossRef]
- Xie, H.; Yu, W.; Li, Y.; Chen, L. Discussion on the thermal conductivity enhancement of nanofluids. Nanoscale Res. Lett. 2011, 6, 124. [Google Scholar] [CrossRef] [PubMed]
- Wamkam, C.T.; Opoku, M.K.; Hong, H.; Smith, P. Effects of ph on heat transfer nanofluids containing ZrO2 and TiO2 nanoparticles. J. Appl. Phys. 2011, 109, 024305. [Google Scholar] [CrossRef]
- Zhu, D.; Li, X.; Wang, N.; Wang, X.; Gao, J.; Li, H. Dispersion behavior and thermal conductivity characteristics of Al2O3–H2O nanofluids. Curr. Appl. Phys. 2009, 9, 131–139. [Google Scholar] [CrossRef]
- Eastman, J.A.; Choi, S.U.S.; Li, S.; Yu, W.; Thompson, L.J. Anomalously increased effective thermal conductivities of ethylene glycol-based nanofluids containing copper nanoparticles. Appl. Phys. Lett. 2001, 78, 718–720. [Google Scholar] [CrossRef]
- Younes, H.; Christensen, G.; Luan, X.; Hong, H.; Smith, P. Effects of alignment, ph, surfactant, on heat transfer nanofluids containing Fe2O3 and CuO nanoparticles. J. Appl. Phys. 2012, 111, 064308. [Google Scholar] [CrossRef]
- Hong, T.-K.K.; Yang, H.-S.S.; Choi, C.J. Study of the enhanced thermal conductivity of Fe nanofluids. J. Appl. Phys. 2005, 97, 64311. [Google Scholar] [CrossRef]
- Garg, P.; Alvarado, J.L.; Marsh, C.; Carlson, T.A.; Kessler, D.A.; Annamalai, K. An experimental study on the effect of ultrasonication on viscosity and heat transfer performance of multi-wall carbon nanotube-based aqueous nanofluids. Int. J. Heat Mass Transf. 2009, 52, 5090–5101. [Google Scholar] [CrossRef]
- Asadi, A.; Asadi, M.; Siahmargoi, M.; Asadi, T.; Gholami Andarati, M. The effect of surfactant and sonication time on the stability and thermal conductivity of water-based nanofluid containing Mg(OH)2 nanoparticles: An experimental investigation. Int. J. Heat Mass Transf. 2017, 108, 191–198. [Google Scholar] [CrossRef]
- Hong, H.; Wright, B.; Wensel, J.; Jin, S.; Ye, X.R.; Roy, W. Enhanced thermal conductivity by the magnetic field in heat transfer nanofluids containing carbon nanotube. Synthetic Metals 2007, 157, 437–440. [Google Scholar] [CrossRef]
- Wright, B.; Thomas, D.; Hong, H.; Groven, L.; Puszynski, J.; Duke, E.; Ye, X.; Jin, S. Magnetic field enhanced thermal conductivity in heat transfer nanofluids Ni coated single wall carbon nanotubes. Appl. Phys. Lett. 2007, 91, 173116. [Google Scholar] [CrossRef]
- Wensel, J.; Wright, B.; Thomas, D.; Douglas, W.; Mannhalter, B.; Cross, W.; Hong, H.; Kellar, J.; Smith, P.; Roy, W. Enhanced thermal conductivity by aggregation in heat transfer nanofluids containing metal oxide nanoparticles and carbon nanotubes. Appl. Phys. Lett. 2008, 92, 023110. [Google Scholar] [CrossRef]
- Das, P.K. A review based on the effect and mechanism of thermal conductivity of normal nanofluids and hybrid nanofluids. J. Mol. Liq. 2017, 240, 420–446. [Google Scholar] [CrossRef]
- Lamas, B.; Abreu, B.; Fonseca, A.; Martins, N.; Oliveira, M. Critical analysis of the thermal conductivity models for CNT based nanofluids. Int. J. Therm. Sci. 2014, 78, 65–76. [Google Scholar] [CrossRef]
- Maxwell, J.C. A Treatise on Electricity and Magnetism; Cambridge University Press: Cambridge, UK, 1873; ISBN 9780511709340. [Google Scholar]
- Wasp, E.J.; Kenny, J.P.; Gandhi, R.L. Solid-liquid flow: Slurry pipeline transportation. Pumps, valves, mechanical equipment, economics. Ser. Bulk Mater.Handl. 1977, 1, 106804135. [Google Scholar]
- Xuan, Y.; Li, Q.; Hu, W. Aggregation structure and thermal conductivity of nanofluids. AIChE J. 2003, 49, 1038–1043. [Google Scholar] [CrossRef]
- Koo, J.; Kleinstreuer, C. A new thermal conductivity model for nanofluids. J. Nanoparticle Res. 2004, 6, 577–588. [Google Scholar] [CrossRef]
- Xie, H.; Fujii, M.; Zhang, X. Effect of interfacial nanolayer on the effective thermal conductivity of nanoparticle-fluid mixture. Int. J. Heat Mass Transf. 2005, 48, 2926–2932. [Google Scholar] [CrossRef]
- Avsec, J.; Oblak, M. The calculation of thermal conductivity, viscosity and thermodynamic properties for nanofluids on the basis of statistical nanomechanics. Int. J. Mass Transf. 2007, 50, 4331–4341. [Google Scholar] [CrossRef]
- Pak, B.C.; Cho, Y.I. Hydrodynamic and heat transfer study of dispersed fluids with submicron metallic oxide particles. Exp. Heat Transf. 1998, 11, 151–170. [Google Scholar] [CrossRef]
- Yu, W.; Choi, S.U.S. The role of interfacial layers in the enhanced thermal conductivity of nanofluids: A renovated Maxwell model. J. Nanopart Res. 2003, 5, 167–171. [Google Scholar] [CrossRef]
- Wang, W.; Lin, L.; Zhou, X.; Wang, S. A Comprehensive Model for the Enhanced Thermal Conductivity of Nanofluids. J. Adv. Res. Phys. 2012, 3, 1–5. [Google Scholar]
- Chandrasekar, M.; Suresh, S.; Srinivasan, R.; Bose, A.C. New Analytical Models to Investigate Thermal Conductivity of Nanofluids. J. Nanosci. Nanotechnol. 2009, 9, 533–538. [Google Scholar] [CrossRef] [PubMed]
- Corcione, M. Empirical correlating equations for predicting the effective thermal conductivity and dynamic viscosity of nanofluids. Energy Convers Manag. 2011, 52, 789–793. [Google Scholar] [CrossRef]
- Rocha, C.D.S. Study of the Cooling in Heat Sinks with the Recourse to Innovative Nanofluids. Master’s Thesis, Department of Mechanical Engineering, Universidade do Minho, Braga, Portugal, 2018. [Google Scholar]
- Zhang, Y.; Xu, X. Predicting the thermal conductivity enhancement of nanofluids using computacional intelligence. Phys. Lett. A 2020, 384, 126500. [Google Scholar] [CrossRef]
- Sahaluddin, M.; Alade, I.O.; Oyedeji, M.O.; Aliyu, U.S. A machine learnig-based model to estimate the density of nanofluids of nitrides in ethylene glycol. J. Appl. Phys. 2020, 127, 205105. [Google Scholar] [CrossRef]
- Alade, I.O.; Rahman, M.A.A.; Hassan, A.; Saleh, T.A. Modeling the viscosity of nanofluids using artificial neural network and Bayesan support vector regression. J. Appl. Phys. 2020, 128, 085306. [Google Scholar] [CrossRef]
- Shateri, M.; Sobhanigavgani, Z.; Alinasab, A.; Varamesh, A.; Hemmati-Sarapardeh, A.; Mosavi, A.; Shahab, S. Comparative Analysis of Machine Learning Models for Nanofluids Viscosity Assessment. Nanomaterials 2020, 10, 1767. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, X. Machine learning glass transition temperature of polyacrylamides using quantum chemical descriptors. Polym. Chem. 2021, 12, 843–851. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, X. Machine learning glass transition temperature of polymers. Heliyon 2020, 6, e05055. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, X. Machine Learning Decomposition Onset Temperature of Lubricant Additives. J. Mater. Eng. Perform. 2020, 29, 6605. [Google Scholar] [CrossRef]
- Ma, T.; Guo, Z.; Lin, M.; Wang, Q. Recent trends on nanofluid heat transfer machine learning research applied to renewable energy. Renew. Sustain. Energy Rev. 2021, 138, 110494. [Google Scholar] [CrossRef]
- Zhao, N.B.; Li, S.Y.; Yang, J.L. A review on nanofluids: Data-driven modeling of thermalphysical properties and the application in automotive radiator. Renew. Sustain. Energy Rev. 2016, 66, 596–616. [Google Scholar] [CrossRef]
- Ramezanizadeh, M.; Ahmadi, M.H.; Nazari, M.A.; Sadeghzadeh, M.; Chen, L.G. A review on the utilized machine learning approaches for modeling the dynamic viscosity of nanofluids. Renew. Sustain. Energy Rev. 2019, 114, 109345. [Google Scholar] [CrossRef]
- Bahiraei, M.; Heshmatian, S.; Moayedi, H. Artificial intelligence in the field of nanofluids: A review on applications and potencial future directions. Powder Technol. 2019, 353, 276–301. [Google Scholar] [CrossRef]
- Guo, Z. A review on heat transfer enhancement with nanofluids. J. Enhanc. Heat Transf. 2020, 27, 1–70. [Google Scholar] [CrossRef]
- Pavlik, M. The Dependence of Suspension Viscosity on Particle Size, Shear Rate, and Solvent Viscosity. Ph.D. Thesis, DePaul University, Chicago, IL, USA, 2009. [Google Scholar]
- Brinkman, H.C. The viscosity of concentrated suspensions and solutions. J. Chem. Phys. 1952, 20, 571. [Google Scholar] [CrossRef]
- Mooney, M. The viscosity of a concentrated suspension of spherical particles. J. Colloid Sci. 1951, 6, 162–170. [Google Scholar] [CrossRef]
- Vajjha, R.S.; Das, D.K. Specific Heat Measurement of Three Nanofluids and Development of New Correlations. J. Heat Transf. 2009, 131, 071601. [Google Scholar] [CrossRef]
- Nieh, H.-M.; Teng, T.-P.; Yu, C.-C. Enhanced heat dissipation of a radiator using oxide nano-coolant. Int. J. Therm. Sci. 2014, 77, 252–261. [Google Scholar] [CrossRef]
- Lee, J.; Mudawar, I. Assessment of the effectiveness of nanofluids for single-phase and two-phase heat transfer in micro-channels. Int. J. Heat Mass Transf. 2007, 50, 452–463. [Google Scholar] [CrossRef]
- Bergman, T.L. Effect of reduced specific heats of nanofluids on single phase, laminar internal forced convection. Int. J. Heat Mass Transf. 2009, 52, 1240–1244. [Google Scholar] [CrossRef]
- Ho, C.J.; Wei, L.C.; Li, Z.W. An experimental investigation of forced convective cooling performance of a microchannel heat sink with Al2O3/water nanofluid. Appl. Therm. Eng. 2010, 30, 96–103. [Google Scholar] [CrossRef]
- Al-Waeli, A.H.A.; Chaichan, M.T.; Kazem, H.A.; Sopian, K. Comparative study to use nano-(Al2O3, CuO, and SiC) with water to enhance photovoltaic thermal PV/T collectors. Energy Convers. Manag. 2017, 148, 963–973. [Google Scholar] [CrossRef]
- Roberts, N.A.; Walker, D.G. Convective performance of nanofluids in commercial electronics cooling systems. Appl. Therm. Eng. 2010, 30, 2499–2504. [Google Scholar] [CrossRef]
- Khaleduzzaman, S.S.; Sohel, M.R.; Saidur, R.; Mahbubul, I.M.; Shahrul, I.M.; Akash, B.A.; Selvaraj, J. Energy and exergy analysis of alumina-water nanofluid for an electronic liquid cooling system. Int. Commun. Heat Mass Transf. 2014, 57, 118–127. [Google Scholar] [CrossRef]
- Li, C.H.; Peterson, G.P. Experimental investigation of temperature and volume fraction variations on the effective thermal conductivity of nanoparticle suspensions (nanofluids). J. Appl. Phys. 2006, 99, 084314. [Google Scholar] [CrossRef]
- Li, C.H.; Peterson, G.P. The effect of particle size on the effective thermal conductivity of Al2O3-water nanofluids. J. Appl. Phys. 2007, 101, 44312. [Google Scholar] [CrossRef]
- Zhang, X.; Gu, H.; Fujii, M. Effective thermal conductivity and thermal diffusivity of nanofluids containing spherical and cylindrical nanoparticles. Exp. Therm. Fluid Sci. 2007, 31, 593–599. [Google Scholar] [CrossRef]
- Beck, M.P.; Yuan, Y.; Warrier, P.; Teja, A.S. The effect of particle size on the thermal conductivity of alumina nanofluids. J. Nanoparticle Res. 2009, 11, 1129–1136. [Google Scholar] [CrossRef]
- Buonomo, B.; Manca, O.; Marinelli, L.; Nardini, S. Effect of temperature and sonication time on nanofluid thermal conductivity measurements by nano-flash method. Appl. Therm. Eng. 2015, 91, 181–190. [Google Scholar] [CrossRef]
- Chandrasekar, M.; Suresh, S.; Chandra Bose, A. Experimental investigations and theoretical determination of thermal conductivity and viscosity of Al2O3/water nanofluid. Exp. Therm. Fluid Sci. 2010, 34, 210–216. [Google Scholar] [CrossRef]
- Kim, S.H.; Choi, S.R.; Kim, D. Thermal Conductivity of Metal-Oxide Nanofluids: Particle Size Dependence and Effect of Laser Irradiation. J. Heat Transf. 2006, 129, 298–307. [Google Scholar] [CrossRef]
- Kumar, N.; Sonawane, S.S.; Sonawane, S.H. Experimental study of thermal conductivity, heat transfer and friction factor of Al2O3 based nanofluid. Int. Commun. Heat Mass Transf. 2018, 90, 1–10. [Google Scholar] [CrossRef]
- Beck, M.P.; Yuan, Y.; Warrier, P.; Teja, A.S. The thermal conductivity of alumina nanofluids in water, ethylene glycol, and ethylene glycol + water mixtures. J. Nanoparticle Res. 2010, 12, 1469–1477. [Google Scholar] [CrossRef]
- Oh, D.-W.W.; Jain, A.; Eaton, J.K.; Goodson, K.E.; Lee, J.S. Thermal conductivity measurement and sedimentation detection of aluminum oxide nanofluids by using the 3ω method. Int. J. Heat Fluid Flow 2008, 29, 1456–1461. [Google Scholar] [CrossRef]
- Usri, N.A.; Azmi, W.H.; Mamat, R.; Hamid, K.A.; Najafi, G. Thermal Conductivity Enhancement of Al2O3 Nanofluid in Ethylene Glycol and Water Mixture; Elsevier: Amsterdam, The Netherlands, 2015; Volume 79. [Google Scholar]
- Khaleduzzaman, S.S.; Mahbubul, I.M.; Sohel, M.R.; Saidur, R.; Selvaraj, J.; Ward, T.A.; Niza, M.E. Experimental analysis of energy and friction factor for titanium dioxide nanofluid in a water block heat sink. Int. J. Heat Mass Transf. 2017, 115, 77–85. [Google Scholar] [CrossRef]
- Narendran, G.; Bhat, M.M.; Akshay, L.; Arumuga Perumal, D. Experimental analysis on exergy studies of flow through a minichannel using Tio2/Water nanofluids. Therm. Sci. Eng. Prog. 2018, 8, 93–104. [Google Scholar] [CrossRef]
- Zhang, J.; Diao, Y.; Zhao, Y.; Zhang, Y. Experimental study of TiO2-water nanofluid flow and heat transfer characteristics in a multiport minichannel flat tube. Int. J. Heat Mass Transf. 2014, 79, 628–638. [Google Scholar] [CrossRef]
- Xia, G.D.; Liu, R.; Wang, J.; Du, M. The characteristics of convective heat transfer in microchannel heat sinks using Al2O3 and TiO2 nanofluids. Int. Commun. Heat Mass Transf. 2016, 76, 256–264. [Google Scholar] [CrossRef]
- Gan, Y.Y.; Ong, H.C.; Ling, T.C.; Zulkifli, N.W.M.; Wang, C.T.; Yang, Y.C. Thermal conductivity optimization and entropy generation analysis of titanium dioxide nanofluid in evacuated tube solar collector. Appl. Therm. Eng. 2018, 145, 155–164. [Google Scholar] [CrossRef]
- He, Y.; Jin, Y.; Chen, H.; Ding, Y.; Cang, D.; Lu, H. Heat transfer and flow behavior of aqueous suspensions of TiO2 nanoparticles (nanofluids) flowing upward through a vertical pipe. Int. J. Heat Mass Transf. 2007, 50, 2272–2281. [Google Scholar] [CrossRef]
- Krishnakumar, T.S.; Sheeba, A.; Mahesh, V.; Jose Prakash, M. Heat transfer studies on ethylene glycol/water nanofluid containing TiO2 nanoparticles. Int. J. Refrig. 2019, 102, 55–61. [Google Scholar] [CrossRef]
- Khedkar, R.S.; Shrivastava, N.; Sonawane, S.S.; Wasewar, K.L. Experimental investigations and theoretical determination of thermal conductivity and viscosity of TiO2–ethylene glycol nanofluid. Int. Commun. Heat Mass Transf. 2016, 73, 54–61. [Google Scholar] [CrossRef]
- Murshed, S.M.S.; Leong, K.C.; Yang, C. Investigations of thermal conductivity and viscosity of nanofluids. Int. J. Therm. Sci. 2008, 47, 560–568. [Google Scholar] [CrossRef]
- Turgut, A.; Tavman, I.; Chirtoc, M.; Schuchmann, H.P.; Sauter, C.; Tavman, S. Thermal Conductivity and Viscosity Measurements of Water-Based TiO2 Nanofluids. Int. J. Thermophys. 2009, 30, 1213–1226. [Google Scholar] [CrossRef]
- Al-Waeli, A.H.A.; Sopian, K.; Chaichan, M.T.; Kazem, H.A.; Hasan, H.A.; Al-Shamani, A.N. An experimental investigation of SiC nanofluid as a base-fluid for a photovoltaic thermal PV/T system. Energy Convers. Manag. 2017, 142, 547–558. [Google Scholar] [CrossRef]
- Khedkar, R.S.; Sonawane, S.S.; Wasewar, K.L. Influence of CuO nanoparticles in enhancing the thermal conductivity of water and monoethylene glycol based nanofluids. Int. Commun. Heat Mass Transf. 2012, 39, 665–669. [Google Scholar] [CrossRef]
- Agarwal, R.; Verma, K.; Agrawal, N.K.; Duchaniya, R.K.; Singh, R. Synthesis, characterization, thermal conductivity and sensitivity of CuO nanofluids. Appl. Therm. Eng. 2016, 102, 1024–1036. [Google Scholar] [CrossRef]
- Hwang, Y.; Lee, J.K.; Lee, C.H.; Jung, Y.M.; Cheong, S.I.; Lee, C.G.; Ku, B.C.; Jang, S.P. Stability and thermal conductivity characteristics of nanofluids. Thermochim. Acta 2007, 455, 70–74. [Google Scholar] [CrossRef]
- Naik, B.A.K.; Vinod, A.V. Heat transfer enhancement using non-Newtonian nanofluids in a shell and helical coil heat exchanger. Exp. Therm. Fluid Sci. 2018, 90, 132–142. [Google Scholar] [CrossRef]
- Keyvani, M.; Afrand, M.; Toghraie, D.; Reiszadeh, M. An experimental study on the thermal conductivity of cerium oxide/ethylene glycol nanofluid: Developing a new correlation. J. Mol. Liq. 2018, 266, 211–217. [Google Scholar] [CrossRef]
- Li, H.; Wang, L.; He, Y.; Hu, Y.; Zhu, J.; Jiang, B. Experimental investigation of thermal conductivity and viscosity of ethylene glycol based ZnO nanofluids. Appl. Therm. Eng. 2015, 88, 363–368. [Google Scholar] [CrossRef]
- Yu, W.; Xie, H.; Chen, L.; Li, Y. Enhancement of thermal conductivity of kerosene-based Fe3O4 nanofluids prepared via phase-transfer method. Colloids Surf. A Physicochem. Eng. Asp. 2010, 355, 109–113. [Google Scholar] [CrossRef]
- Abareshi, M.; Goharshadi, E.K.; Mojtaba Zebarjad, S.; Khandan Fadafan, H.; Youssefi, A. Fabrication, characterization and measurement of thermal conductivity of Fe3O4 nanofluids. J. Magn. Magn. Mater. 2010, 322, 3895–3901. [Google Scholar] [CrossRef]
- Doganay, S.; Turgut, A.; Cetin, L. Magnetic field dependent thermal conductivity measurements of magnetic nanofluids by 3ω method. J. Magn. Magn. Mater. 2019, 474, 199–206. [Google Scholar] [CrossRef]
- Karimi, A.; Sadatlu, M.A.A.; Saberi, B.; Shariatmadar, H.; Ashjaee, M. Experimental investigation on thermal conductivity of water based nickel ferrite nanofluids. Adv. Powder Technol. 2015, 26, 1529–1536. [Google Scholar] [CrossRef]
- Hemmat Esfe, M.; Afrand, M.; Karimipour, A.; Yan, W.-M.; Sina, N. An experimental study on thermal conductivity of MgO nanoparticles suspended in a binary mixture of water and ethylene glycol. Int. Commun. Heat Mass Transf. 2015, 67, 173–175. [Google Scholar] [CrossRef]
- Liu, M.-S.; Lin, M.C.-C.; Tsai, C.Y.; Wang, C.-C. Enhancement of thermal conductivity with Cu for nanofluids using chemical reduction method. Int. J. Heat Mass Transf. 2006, 49, 3028–3033. [Google Scholar] [CrossRef]
- Putnam, S.A.; Cahill, D.G.; Braun, P.V.; Ge, Z.; Shimmin, R.G. Thermal conductivity of nanoparticle suspensions. J. Appl. Phys. 2006, 99, 84308. [Google Scholar] [CrossRef]
- Carbajal-Valdéz, R.; Rodríguez-Juárez, A.; Jiménez-Pérez, J.L.; Sánchez-Ramírez, J.F.; Cruz-Orea, A.; Correa-Pacheco, Z.N.; Macias, M.; Luna-Sánchez, J.L. Experimental investigation on thermal properties of Ag nanowire nanofluids at low concentrations. Thermochim. Acta 2019, 671, 83–88. [Google Scholar] [CrossRef]
- Parametthanuwat, T.; Bhuwakietkumjohn, N.; Rittidech, S.; Ding, Y. Experimental investigation on thermal properties of silver nanofluids. Int. J. Heat Fluid Flow 2015, 56, 80–90. [Google Scholar] [CrossRef]
- Alirezaie, A.; Hajmohammad, M.H.; Hassani Ahangar, M.R.; Hemmat Esfe, M. Price-performance evaluation of thermal conductivity enhancement of nanofluids with different particle sizes. Appl. Therm. Eng. 2018, 128, 373–380. [Google Scholar] [CrossRef]
- Lee, S.W.; Park, S.D.; Kang, S.; Bang, I.C.; Kim, J.H. Investigation of viscosity and thermal conductivity of SiC nanofluids for heat transfer applications. Int. J. Heat Mass Transf. 2011, 54, 433–438. [Google Scholar] [CrossRef]
- Li, X.; Zou, C.; Zhou, L.; Qi, A. Experimental study on the thermo-physical properties of diathermic oil based SiC nanofluids for high temperature applications. Int. J. Heat Mass Transf. 2016, 97, 631–637. [Google Scholar] [CrossRef]
- Ranjbarzadeh, R.; Moradikazerouni, A.; Bakhtiari, R.; Asadi, A.; Afrand, M. An experimental study on stability and thermal conductivity of water/silica nanofluid: Eco-friendly production of nanoparticles. J. Clean. Prod. 2019, 206, 1089–1100. [Google Scholar] [CrossRef]
- Mousavi, S.M.; Esmaeilzadeh, F.; Wang, X.P. A detailed investigation on the thermo-physical and rheological behavior of MgO/TiO2 aqueous dual hybrid nanofluid. J. Mol. Liq. 2019, 282, 323–339. [Google Scholar] [CrossRef]
- Sarafraz, M.M.; Yang, B.; Pourmehran, O.; Arjomandi, M.; Ghomashchi, R. Fluid and heat transfer characteristics of aqueous graphene nanoplatelet (GNP) nanofluid in a microchannel. Int. Commun. Heat Mass Transf. 2019, 107, 24–33. [Google Scholar] [CrossRef]
- Yu, W.; Xie, H.; Wang, X.; Wang, X. Significant thermal conductivity enhancement for nanofluids containing graphene nanosheets. Phys. Lett. A 2011, 375, 1323–1328. [Google Scholar] [CrossRef]
- Arzani, H.K.; Amiri, A.; Kazi, S.N.; Chew, B.T.; Badarudin, A. Experimental and numerical investigation of thermophysical properties, heat transfer and pressure drop of covalent and noncovalent functionalized graphene nanoplatelet-based water nanofluids in an annular heat exchanger. Int. Commun. Heat Mass Transf. 2015, 68, 267–275. [Google Scholar] [CrossRef]
- Bhattad, A.; Sarkar, J.; Ghosh, P. Experimentation on effect of particle ratio on hydrothermal performance of plate heat exchanger using hybrid nanofluid. Appl. Therm. Eng. 2019, 162, 114309. [Google Scholar] [CrossRef]
- Poongavanam, G.K.; Panchabikesan, K.; Murugesan, R.; Duraisamy, S.; Ramalingam, V. Experimental investigation on heat transfer and pressure drop of MWCNT—Solar glycol based nanofluids in shot peened double pipe heat exchanger. Powder Technol. 2019, 345, 815–824. [Google Scholar] [CrossRef]
- Rehman, W.U.; Merican, Z.M.A.; Bhat, A.H.; Hoe, B.G.; Sulaimon, A.A.; Akbarzadeh, O.; Khan, M.S.; Mukhtar, A.; Saqib, S.; Hameed, A.; et al. Synthesis, characterization, stability and thermal conductivity of multi-walled carbon nanotubes (MWCNTs) and eco-friendly jatropha seed oil based nanofluid: An experimental investigation and modeling approach. J. Mol. Liq. 2019, 293, 111534. [Google Scholar] [CrossRef]
- Ding, Y.; Alias, H.; Wen, D.; Williams, R.A. Heat transfer of aqueous suspensions of carbon nanotubes (CNT nanofluids). Int. J. Heat Mass Transf. 2006, 49, 240–250. [Google Scholar] [CrossRef]
- Assael, M.J.; Metaxa, I.N.; Arvanitidis, J.; Christofilos, D.; Lioutas, C. Thermal Conductivity Enhancement in Aqueous Suspensions of Carbon Multi-Walled and Double-Walled Nanotubes in the Presence of Two Different Dispersants. Int. J. Thermophys. 2005, 26, 647–664. [Google Scholar] [CrossRef]
- Xing, M.; Yu, J.; Wang, R. Experimental investigation and modeling on the thermal conductivity of CNTs based nanofluids. Int. J. Therm. Sci. 2016, 104, 404–411. [Google Scholar] [CrossRef]
- Fakoor Pakdaman, M.; Akhavan-Behabadi, M.A.; Razi, P. An experimental investigation on thermo-physical properties and overall performance of MWCNT/heat transfer oil nanofluid flow inside vertical helically coiled tubes. Exp. Therm. Fluid Sci. 2012, 40, 103–111. [Google Scholar] [CrossRef]
- Liu, M.-S.; Ching-Cheng Lin, M.; Huang, I.-T.; Wang, C.-C. Enhancement of thermal conductivity with carbon nanotube for nanofluids. Int. Commun. Heat Mass Transf. 2005, 32, 1202–1210. [Google Scholar] [CrossRef]
- Harish, S.; Ishikawa, K.; Einarsson, E.; Aikawa, S.; Chiashi, S.; Shiomi, J.; Maruyama, S. Enhanced thermal conductivity of ethylene glycol with single-walled carbon nanotube inclusions. Int. J. Heat Mass Transf. 2012, 55, 3885–3890. [Google Scholar] [CrossRef]

| Researchers | Mathematical Expressions | Included Parameters/Observations |
|---|---|---|
| Maxwell [73] | Includes the thermal conductivities of BF and NPs | |
| Hamilton and Crosser [72] | Includes the particle shape and composition and the thermal conductivities of BF and NPs | |
| Wasp [74] | Includes the thermal conductivities of BF and NPs and the sphericity of the particles assumption | |
| Xue [71] | Includes the particle shape and composition and the logarithmic progressions of the thermal conductivities of BF and NPs | |
| Xuan [75] | Includes the temperature and viscosity of the BF, the average radius and viscosity of the clusters, and the Brownian motion | |
| Kleinstreuer [76] | Includes the temperature and viscosity of the BF, the volume fraction and type of NPs, and the Brownian diffusion | |
| Xie [77] | Includes the nanolayer thickness, the NPs volume fraction and radius and the thermal conductivity ratio between the NPs and the BF | |
| Avsec et al. [78] | Includes the liquid layer thickness, thermal conductivities of BF and NPs, but not the particle size and the interface between the particles | |
| Jang and Choi [32] | Includes the thermal condutivity and the diameter of the molecules of the BF, the particle fraction and diameter, the thermal conductivity of the NPs involving the Kapitza resistance (surface resistance),and the numbers of Reynolds and Prandtl | |
| Pak and Cho [79] | Includes the geometry, diameter, and the surface resistance of the NPs | |
| Timofeeva et al. [33] | Includes the geometry, agglomeration state, and the surface resistance of the NPs | |
| Yu and Choi et al. [80] | Modified Maxwell model.Includes the nanolayer thickness | |
| Wang et al. [81] | Includes nanolayer thickness, particle size, temperature, volume fraction, and interaction between adjacent particles | |
| Chandrasekar et al. [82] | Model I is applicable over a wide range of particlenature, fraction and size, and different base fluids. Model II help to determine the contribution layer thickness, particle shape, and Brownian motion | |
| Corcione [83] | Applicable for the temperature range of 294–324 K, nanoparticle diameter of 10–150 nm and volume fraction of 0.002–0.9 |
| Reference | Work Focus | Machine Learning Involviment |
|---|---|---|
| Zhao et al., 2016 [93] | Prediction of thermal conductivity and viscosity based on ANN and applications in automotive radiators | ANN data-driven modeling |
| Ramezanizadeh et al., 2019 [94] | Characteristics of different machine learning methods including MLP-ANN, GMDH, ANFIS, RBF, and LS-SVM combined with GA, PSO, and ICA. Applications of machine learning methods to dynamic viscosity modeling of nanofluids | Machine learning for viscosity prediction |
| Bahiraei et al., 2019 [95] | AI algorithms inluding ANNs, fuzzy logic optimization methods and hybrid AI algorithms used for prediction and optimization of thermal properties of nanofluids | Machine learning algorithms for prediction and optimization |
| Guo 2020 [96] | Overview on measured thermal properties, enhancement mechanisms, models for properties and heat transfer characteristics and applications of nanofluids to cooling, renewable energy, and energy and building technologies | ANN model for thermal conductivity prediction |
| Sahaluddin et al., 2020 [86] | Development of a machine learning model for density prediction of nitrides in ethylene glycol. The developed is much more accurate than the Pak and Cho empirical model | SVM model for density prediction |
| Zhang and Xu 2020 [89] | Machine learning glass transition temperature of polymers prediction using a GPR (Gauss Process Regression) data-driven model | GPR model for glass transition temperature prediction |
| Zhang and Xu 2020 [91] | Machine learning decomposition onset temperature of lubricant additives prediction using a GPR data-driven model | GPR model for temperature prediction |
| Shateri et al., 2020 [88] | CMIS (Comittee Machine Intelligent System) machine learning model for nanofluid viscosity estimation | CMIS model for viscosity prediction |
| Alade et al., 2020 [87] | BSVR (Bayesian Support Vector Regression) and ANN machine learning models for nanofluid viscosity prediction | BSVR and ANN models for viscosity prediction |
| Ma et al., 2021 [92] | Nanofluid heat transfer machine learning research applied to renewable energy | Machine learning description and applications |
| NPs | BF | Size [nm] | Concentration | Keff Increase [%] | Method | Ref. |
|---|---|---|---|---|---|---|
| Water | 33 | 1 and 2 vol.% | 5.4 | Theory | [104] | |
| 30–60 | 0.5, 1.0, 2, 3 and 4 vol.% | 1.96 | THW | [105] | ||
| 10, 20–30 and 150 | Up to 1.5 vol.% | 23 | Theory | [106] | ||
| 13 | 0.1, 0.15, 0.20 and 0.25 vol.% | 6.40 for 0.25 vol.% NF | THW | [107] | ||
| 41 | 18 vol.% | 31 | THW | [54] | ||
| 36 | 10 vol.% | 30 | SSCB | [108] | ||
| 36 | 6 vol.% | 28 | SSCB | [109] | ||
| 20 | 14.6 vol.% | 22 | TSHW | [110] | ||
| 282 | 4 vol.% | 17.7 | THW | [111] | ||
| 40 | 4 vol.% | 14.4 | Flash | [112] | ||
| 15–50 | 4 vol.% | 10.1 | TPS | [61] | ||
| 43 | 3 vol.% | 9.7 | THW | [113] | ||
| 11 | 1 vol.% | 9 | THW | [53] | ||
| 38 | 3 vol.% | 8 | THW | [114] | ||
| 43 | 2 vol.% | 7.52 | THW | [113] | ||
| 10 | 0.08 vol.% | 7.1 | THW | [115] | ||
| 12 | 4 vol.% | 5.4 | THW | [116] | ||
| 10 | 0.05 vol.% | 4.7 | THW | [115] | ||
| 43 | 0.75 vol.% | 3.28 | THW | [113] | ||
| 10 | 0.04 vol.% | 3.1 | THW | [115] | ||
| 43 | 0.33 vol.% | 1.64 | THW | [113] | ||
| EG | 10 | 0.08 vol.% | 22 | THW | [115] | |
| 10 | 0.06 vol.% | 17.3 | THW | [115] | ||
| 282 | 3 vol.% | 16.3 | THW | [111] | ||
| 12 | 4 vol.% | 14.3 | THW | [116] | ||
| 38 | 3 vol.% | 10.6 | THW | [114] | ||
| 45 | 4 vol.% | 9.7 | 3ω | [117] | ||
| EG/Water | 13 | 2 vol.% | 12.6 | THW | [118] | |
| 10 | 3 vol.% | 11.3 | THW | [116] | ||
| 50 | 3 vol.% | 10.4 | THW | [116] | ||
| 13 | 2 vol.% | 8.4 | THW | [118] | ||
| 13 | 2 vol.% | 16.2 | THW | [118] | ||
| DI | 45 | 4 vol.% | 13.3 | 3ω | [117] | |
| 48 | 1 vol.% | 4 | THW | [28] | ||
| Water | 21 | 0.1 vol% | About 11.1 | THW | [119] | |
| 100 | 0.10, 0.15, 0.21 and 0.25 vol.% | 16.7 for TiO2-0.25% | THW | [120] | ||
| 10, 30, and 50 | 0.005, 0.01, 0.1, 0.5, and 1 vol.% | 0.4 | Theory | [121] | ||
| 5 | 0, 0.1, 0.5 and 1.0 vol.% | 6.55 | TPS | [122] | ||
| 21 | 0.1–0.5 vol.% | 7.28 | [123] | |||
| 10 | 3 vol.% | 11.4 | THW | [114] | ||
| 34 | 3 vol.% | 8.7 | THW | [114] | ||
| 21 | 2 vol.% | 7 | THW | [56] | ||
| 40 | 2.6 vol.% | 6.5 | THW | [110] | ||
| 70 | 3 vol.% | 6.4 | THW | [114] | ||
| 20 | 2 vol.% | 4.2 | THW | [124] | ||
| Water:EG | 40 | 0.2 to 0.8 vol.% | 24 at VF 0.8% and temperature 50 °C | THW | [125] | |
| EG | 5 | 7 vol.% | 19.52 | THW | [126] | |
| 15 | 5 vol.% | 18 | THW | [127] | ||
| 10 | 3 vol.% | 14.4 | THW | [114] | ||
| 34 | 3 vol.% | 12.3 | THW | [114] | ||
| 70 | 3 vol.% | 7.5 | THW | [114] | ||
| DI | Ø10×40 | 5 vol.% | 33 | THW | [34] | |
| 15 | 5 vol.% | 30 | THW | [34] | ||
| 20.5 | 1 vol.% | 14.4 | THW | [28] | ||
| 21 | 3 vol.% | 7.2 | 3ω | [128] | ||
| Water | 45–65 | 1, 1.5, 2, 3, and 4 wt% | 8.2 | THW | [129] | |
| 45–65 | 0.5, 1.0, 2, 3 and 4 vol.% | 4.8 | THW | [105] | ||
| CuO | Water | 35–45 | 0.5, 1.0, 2, 3 and 4 vol.% | 3.42 | THW | [105] |
| 29 | 6 vol.% | 52 | SSCB | [108] | ||
| 25 | 7.5 vol.% | 32.3 | THW | [130] | ||
| 55–66 | 2 vol.% | 24 | THW | [129] | ||
| 33 | 4.68 vol.% | 16.5 | TSHW | [131] | ||
| 33 | 1 vol.% | 5 | THW | [131] | ||
| Water + 0.5 wt% CMC | 40 | 0.2–1.0 wt% | 29% | THW | [132] | |
| EG | 55–66 | 2 vol.% | 21 | THW | [130] | |
| 33 | 1 vol.% | 9 | THW | [40] | ||
| MEG | 25 | 7.5 vol.% | 21.3 | THW | [129] | |
| EO | 55–66 | 2 vol.% | 14 | THW | [130] | |
| CeO2 | EG | 10–30 | 2.5 vol.% | 22 | THW | [132] |
| ZnO | Water | 10 | 3 vol.% | 14.2 | THW | [133] |
| 60 | 3 vol.% | 7.3 | THW | [134] | ||
| EG | 30 | 3 vol.% | 21 | THW | [113] | |
| 50 | 2.4 vol.% | 13 | THW | [135] | ||
| WO3 | EG | 38 | 0.3 vol.% | 13.8 | THW | [28] |
| Fe3O4 | Kerosene | 15 | 1 vol.% | 34.6 | THW | [136] |
| Water | 15–23 | 3 vol.% | 11.5 | THW | [137] | |
| 15–20 | 4.8 vol.% | 2.9 | 3ω | [138] | ||
| 15–20 | 1 vol.% | 1.1 | 3ω | [138] | ||
| NiFe2O4 | DI | 8 | 2 vol.% | 17.2 | THW | [139] |
| MgO | EG/Water | 40 | 3 vol.% | 34.43 | THW | [140] |
| Cu | Water | 75–100 | 0.1 vol.% | 23.8 | THW | [141] |
| Au | Toluene | 1.65 | 0.003 vol.% | 8 | TSHW | [110] |
| 2 | 0.024 vol.% | 1.4 | MSBD | [142] | ||
| Ethanol | 4 | 0.018 vol.% | 1.3 | MSBD | [142] | |
| Ag | Water | 96 | 1.7×10−5 vol.% | 20.8 | TWRC | [143] |
| 96 | 3.5×10−6 vol.% | 4 | TWRC | [143] | ||
| DI | 5–25 | 0.5 vol.% | 16 | THW | [144] | |
| Fe | EG | 20 | 4 vol.% | 38.8 | Theory | [145] |
| 10 | 0.55 vol.% | 18 | THW | [64] | ||
| 10 | 0.3 vol.% | 16.5 | THW | [28] | ||
| 50 | 2 vol.% | 15.5 | Theory | [145] | ||
| Al | EG | 80 | 5 vol.% | 45 | THW | [127] |
| SiC (sphere) | DI | 100 | 3 vol.% | 7.2 | THW | [146] |
| DO | 30 | 0.8 vol.% | 7.36 | THW | [147] | |
| SiO2 | Water | 40–50 | 3 vol.% | 38.2 | THW | [148] |
| 12 | 1 vol.% | 3.2 | THW | [132] | ||
| 12 | 1 vol.% | 3 | THW | [40] | ||
| Water | 10–45 | 50:50, 80:20, 20:80, 60:40 and 40:60 wt% | 21.8 at 50 °C | THW | [149] | |
| Graphene nanoplatelet (GFnanopl) | Water | 0.025 to 0.1 wt% | 27 | THW | [150] | |
| EG | 0.7–1.3 | 0.05vol.% | 86 | TSHW | [151] | |
| Graphene Oxide | EG | 0.7–1.4 | 0.05vol.% | 61 | TSHW | [151] |
| GFnanopl – SDBS | Water | 0.025, 0.05 and 0.1 wt.% | About 11.56 | Theory | [152] | |
| GFnanopl – COOH | Water | 0.025, 0.05 and 0.1 wt.% | About 21.03 | Theory | [152] | |
| Water | 10–100 () | 0.01 vol.% | 10.85 for MWCNT (0:5) NFs | THW | [153] | |
| MWCNT | Solar glycol | 20–30 | 0.2, 0.4, and0.6 vol.% | 30.59 with MWCNT volume of 0.6% | THW | [154] |
| Jatropha seed oil | Length × OD = 2.5–20 μm × 6–13nm | 0.2–0.8 wt.% | 6.76 | THW | [155] | |
| Water | Ø40 | 0.49 vol.% | 80 | THW | [156] | |
| Ø130 | 0.6 vol.% | 34 | THW | [157] | ||
| Ø10–30 | 1 vol.% | 7 | THW | [132] | ||
| Ø10–30 | 0.48 vol.% | 5 | THW | [158] | ||
| HTO | Ø5–20 | 2 vol.% | 15 | THW | [159] | |
| EG | Ø20–50 | 1 vol.% | 12.4 | THW | [160] | |
| EO | Ø20–50 | 1 vol.% | 8.5 | THW | [160] | |
| DWCNT | Water | Ø5 | 1 vol.% | 8 | THW | [157] |
| SWCNT | Water | Ø1-2×5000-30,000 | 0.48 vol.% | 16.2 | THW | [157] |
| Ø1-2×1000–3000 | 0.48 vol.% | 8.1 | THW | [158] | ||
| EG | 100–600 | 0.21 vol.% | 15.5 | THW | [161] | |
| C60-C70 fullerenes | Toluene | – | 0.378 vol.% | 0.816 | MSBD | [143] |
| MO | 10 | 5 vol.% | 6 | THW | [132] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gonçalves, I.; Souza, R.; Coutinho, G.; Miranda, J.; Moita, A.; Pereira, J.E.; Moreira, A.; Lima, R. Thermal Conductivity of Nanofluids: A Review on Prediction Models, Controversies and Challenges. Appl. Sci. 2021, 11, 2525. https://doi.org/10.3390/app11062525
Gonçalves I, Souza R, Coutinho G, Miranda J, Moita A, Pereira JE, Moreira A, Lima R. Thermal Conductivity of Nanofluids: A Review on Prediction Models, Controversies and Challenges. Applied Sciences. 2021; 11(6):2525. https://doi.org/10.3390/app11062525
Chicago/Turabian StyleGonçalves, Inês, Reinaldo Souza, Gonçalo Coutinho, João Miranda, Ana Moita, José Eduardo Pereira, António Moreira, and Rui Lima. 2021. "Thermal Conductivity of Nanofluids: A Review on Prediction Models, Controversies and Challenges" Applied Sciences 11, no. 6: 2525. https://doi.org/10.3390/app11062525
APA StyleGonçalves, I., Souza, R., Coutinho, G., Miranda, J., Moita, A., Pereira, J. E., Moreira, A., & Lima, R. (2021). Thermal Conductivity of Nanofluids: A Review on Prediction Models, Controversies and Challenges. Applied Sciences, 11(6), 2525. https://doi.org/10.3390/app11062525












