Mesenchymal Stem Cells: The Secret Children’s Weapons against the SARS-CoV-2 Lethal Infection
Abstract
1. Introduction
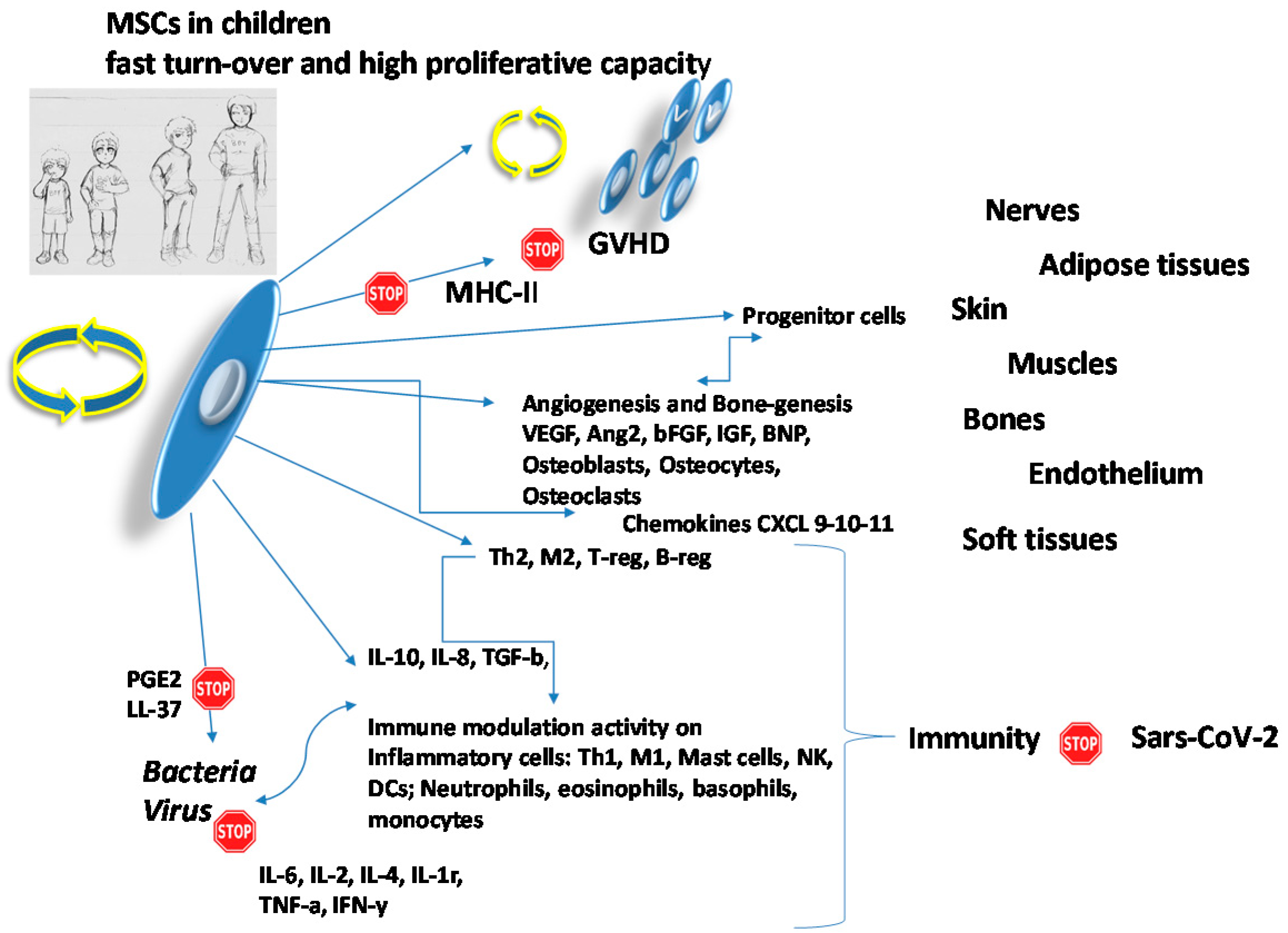
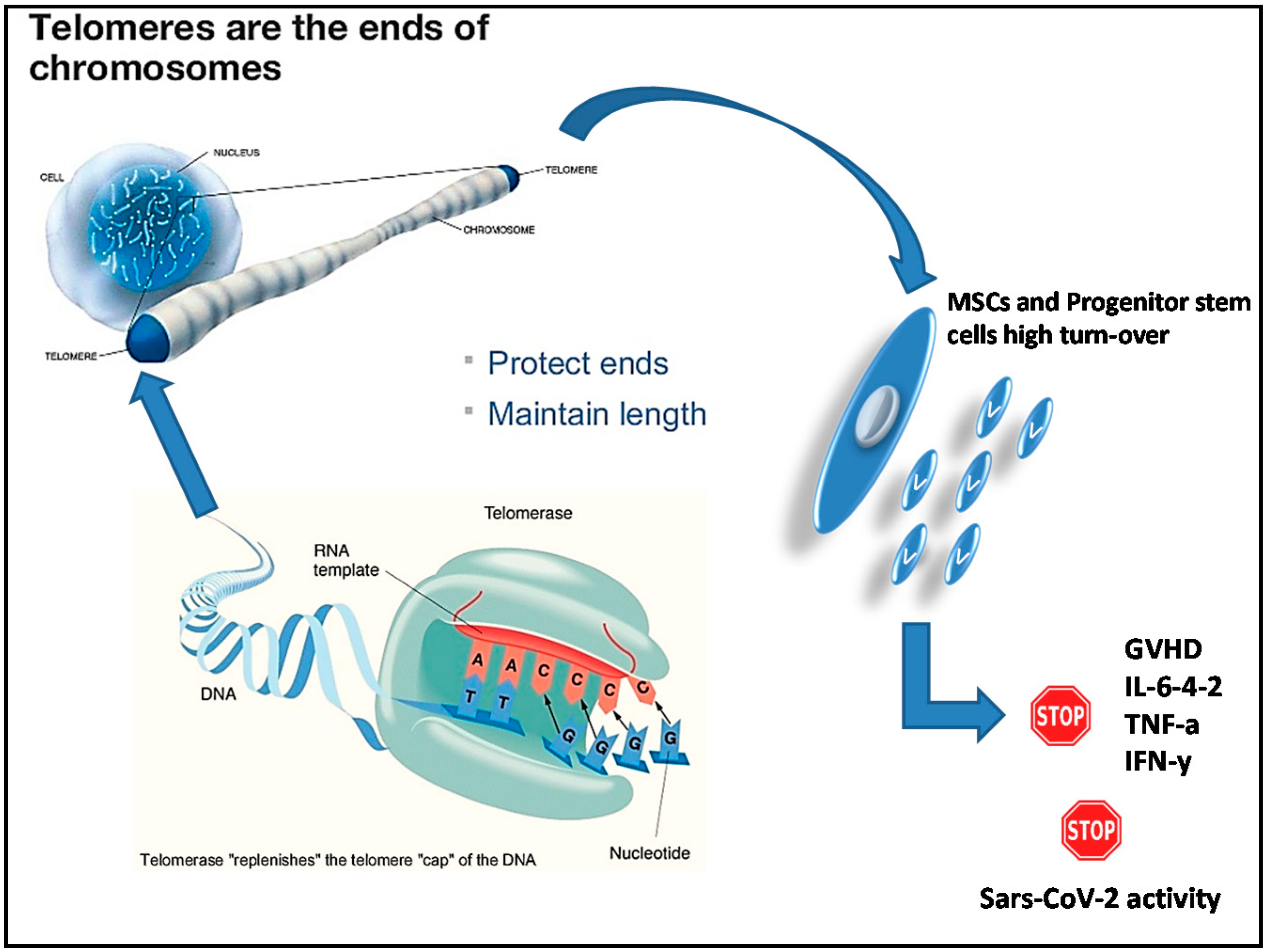
2. Hypothesis: Why Stem Cells Could Be a Possible Solution
3. Present Procedure and Future Perspectives and Limitations
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ATII | Alveolar Type II |
| DCs | Dendritic Cells |
| ESCs | Embryonic Stem Cells |
| GVHD | Graft-Versus-Host Disease |
| iPSCs | Induced Pluripotent Stem Cells |
| MHC I | Co-Stimulatory Factors CD-80 and CD-86 |
| MSCs | Mesenchymal Stem Cells |
| M2 | Macrophages Type 2 |
| NK | Natural Killer |
| SARS-CoV-2 | Severe Acute Respiratory Syndrome Coronavirus 2 |
| γ/δ T Cells | Gamma and Delta T Lymphocytes |
References
- Ludvigsson, J.F. Systematic review of COVID-19 in children show milder cases and a better prognosis than adults. Acta Paediatr. 2020, 109, 1088–1095. [Google Scholar] [CrossRef]
- Lee, P.I.; Hu, Y.L.; Chen, P.Y.; Huang, Y.C.; Hsueh, P.R. Are children less susceptible to COVID-19? J. Microbiol. Immunol. Infect. 2020, 53, 371–372. [Google Scholar] [CrossRef]
- Balzanelli, G.M.; Distratis, P.; Catucci, O.; Amatulli, F.; Cefalo, A.; Lazzaro, R.; Aityan, K.S.; Dalagni, G.; Nico, A.; De Michele, A.; et al. Clinical and diagnostic findings in COVID-19 and non-COVID-19 patients, an original research from SG Moscati Hospital in Taranto City. J. Biol. Regul. Homeost. Agents 2021, 35, 105–117. [Google Scholar]
- Charitos, I.A.; Ballini, A.; Bottalico, L.; Cantore, S.; Passarelli, P.C.; Inchingolo, F.; D’Addona, A.; Santacroce, L. Special features of SARS-CoV-2 in daily practice. World J. Clin. Cases 2020, 8, 3920–3933. [Google Scholar] [CrossRef] [PubMed]
- Santacroce, L.; Charitos, I.A.; Ballini, A.; Inchingolo, F.; Luperto, P.; De Nitto, E.; Topi, S. The Human Respiratory System and its Microbiome at a Glimpse. Biology 2020, 9, 318. [Google Scholar] [CrossRef] [PubMed]
- Cazzolla, A.P.; Lovero, R.; Lo Muzio, L.; Testa, N.F.; Schirinzi, A.L.; Palmieri, G.; Pozzessere, P.; Procacci, V.; Di Comite, M.; Ciavarella, D.; et al. Taste and smell disorders in COVID-19 patients: Role of Interleukin-6. ACS Chem. Neurosci. 2020, 11, 2774–2781. [Google Scholar] [CrossRef] [PubMed]
- Klimczak, A. Perspectives on mesenchymal stem/progenitor cells and their derivates as potential therapies for lung damage caused by COVID-19. World J. Stem Cells 2020, 12, 1013–1022. [Google Scholar] [CrossRef]
- Cantore, S.; Crincoli, V.; Boccaccio, A.; Uva, A.E.; Fiorentino, M.; Monno, G.; Bollero, P.; Derla, C.; Fabiano, F.; Ballini, A.; et al. Recent Advances in Endocrine, Metabolic and Immune Disorders: Mesenchymal Stem Cells (MSCs) and Engineered Scaf-folds. Endocr. Metab. Immune. Disord. Drug Targets 2018, 18, 466–469. [Google Scholar] [CrossRef]
- Ballini, A.; Boccaccio, A.; Saini, R.; Van Pham, P.; Tatullo, M. Dental-Derived Stem Cells and Their Secretome and Interactions with Bioscaffolds/Biomaterials in Regenerative Medicine: From the In Vitro Research to Translational Applications. Stem. Cells. Int. 2017, 2017, 6975251. [Google Scholar] [CrossRef] [PubMed]
- O’Rourke, B.; Nguyen, S.; Tilles, A.W.; Bynum, J.A.; Cap, A.P.; Parekkadan, B.; Barcia, R.N. Mesenchymal stromal cell delivery via an ex vivo bioreactor preclinical test system attenuates clot formation for intravascular application. Stem Cells Transl. Med. 2021. [Google Scholar] [CrossRef]
- Cantore, S.; Ballini, A. Coronavirus Disease 2019 (COVID-19) pandemic burst and its relevant consequences in dental practice. Open Dent. J. 2020, 14, 111–112. [Google Scholar] [CrossRef]
- Maremanda, K.P.; Sundar, I.K.; Li, D.; Rahman, I. Age-Dependent Assessment of Genes Involved in Cellular Senescence, Telomere, and Mitochondrial Pathways in Human Lung Tissue of Smokers, COPD, and IPF: Associations With SARS-CoV-2 COVID-19 ACE2-TMPRSS2-Furin-DPP4 Axis. Front. Pharmacol. 2020, 11, 584637. [Google Scholar] [CrossRef] [PubMed]
- Shimamoto, A.; Yokote, K.; Tahara, H. Werner Syndrome-specific induced pluripotent stem cells: Recovery of telomere function by reprogramming. Front. Genet. 2015, 6, 10. [Google Scholar] [CrossRef]
- Gargiulo, C.; Pham, H.V.; Thuy Hai, N.; Nguyen, K.; Phuc, P.; Abe, K.; Flores, V.; Shiffman, M. Isolation and Characterization of Multipotent and Pluripotent Stem Cells from Human Peripheral Blood. Stem Cell Discov. 2015, 5, 19–32. [Google Scholar] [CrossRef]
- Gargiulo, C.; Pham, H.V.; Nguyen, C.D.K.; Trieu, V.L.H.; Duy, H.T.; Abe, K.; Aityan, S. Autologous Peripheral Blood Stem Cells Increase the Telomere Length in Patient: A Case Report of 13 Patients. J. Stem Cell Res. Ther. 2016, 6, 1–6. [Google Scholar] [CrossRef]
- Monno, R.; De Vito, D.; Losito, G.; Sibilio, G.; Costi, A.; Fumarola, L.; D’Aprile, A.; Marcuccio, P. Chlamydia pneumoniae in community-acquired pneumonia: Seven years of experience. J. Infect. 2002, 45, 135–138. [Google Scholar] [CrossRef]
- Brindicci, G.; Picciarelli, C.; Fumarola, L.; Carbonara, S.; Stano, F.; Ciracì, E.; Gramiccia, M.; Sannella, A.R.; Milella, M.; De Vito, D.; et al. Amoebic hepatic abscesses in an HIV-positive patient. AIDS Patient Care STDS 2006, 20, 606–611. [Google Scholar] [CrossRef] [PubMed]
- Pham, V.H.; Gargiulo, I.C.; Nguyen, K.C.D.; Le, S.H.; Tran, D.K.; Nguyen, Q.V.; Pham, H.T.; Aityan, S.; Pham, S.T.; Cantore, S.; et al. Rapid and sensitive diagnostic procedure for multiple detection of pandemic Coronaviridae family members SARS-CoV-2, SARS-CoV, MERS-CoV and HCoV: A translational research and cooperation between the Phan Chau Trinh University in Vietnam and University of Bari “Aldo Moro” in Italy. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 7173–7191. [Google Scholar]
- Hoffmann, C.; Casado, J.L.; Härter, G.; Vizcarra, P.; Moreno, A.; Cattaneo, D.; Meraviglia, P.; Spinner, C.D.; Schabaz, F.; Grunwald, S.; et al. Immune deficiency is a risk factor for severe COVID-19 in people living with HIV. HIV Med. 2020. [Google Scholar] [CrossRef]
- Ballini, A.; Dipalma, G.; Isacco, C.G.; Boccellino, M.; Di Domenico, M.; Santacroce, L.; Nguyễn, K.C.D.; Scacco, S.; Calvani, M.; Boddi, A.; et al. Oral Microbiota and Immune System Crosstalk: A Translational Research. Biology 2020, 9, 131. [Google Scholar] [CrossRef] [PubMed]
- Montagnier, L.; Aïssa, J.; Lavallée, C.; Mbamy, M.; Varon, J.; Chenal, H. Electromagnetic detection of HIV DNA in the blood of AIDS patients treated by antiretroviral therapy. Interdiscip. Sci. 2009, 1, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Bedin, A.S.; Makinson, A.; Picot, M.C.; Mennechet, F.; Malergue, F.; Pisoni, A.; Nyiramigisha, E.; Montagnier, L.; Bollore, K.; Debiesse, S.; et al. Monocyte CD169 expression as a biomarker in the early diagnosis of COVID-19. J. Infect. Dis. 2020, jiaa724. [Google Scholar] [CrossRef]
- Gargiulo, C.; Pham, V.; Thuy Hai, N.; Nguyen, K.; Duong Kim, N.; Nguyen Van, T.; Tuan, A.; Abe, K.; Flores, V.; Shiffman, M. Autologous Peripheral Blood Stem Cells and γ/δ T Cells May Improve Immunity in Treating Secondary Bacteremic Infection in HIV Infected Patient. Stem Cell Dis. 2015, 5, 48–61. [Google Scholar] [CrossRef]
- Bordea, I.R.; Xhajanka, E.; Candrea, S.; Bran, S.; Onis, F.; Inchingolo, A.D.; Malcangi, G.; Pham, V.H.; Inchingolo, A.M.; Scarano, A.; et al. Coronavirus (SARS-CoV-2) pandemic: Emerging and Future Challenges for Dental Practitioners. Mircoorganisms. 2020, 8, 1704. [Google Scholar] [CrossRef]
- Tufan, A.C. Mesenchymal stem Cells as a Treatment Strategy for Coronavirus Disease 2019 (COVID-19): Need for Authority Regulations and Clinical Guidelines. Curr. Stem Cell Res. Ther. 2020. [Google Scholar] [CrossRef]
- Harrell, C.R.; Jovicic, B.P.; Djonov, V.; Volarevic, V. Therapeutic Potential of Mesenchymal Stem Cells and Their Secretome in the Treatment of SARS-CoV-2-Induced Acute Respiratory Distress Syndrome. Anal. Cell. Pathol. 2020, 2020, 1939768. [Google Scholar] [CrossRef] [PubMed]
- Coelho, A.; Alvites, R.D.; Branquinho, M.V.; Guerreiro, S.G.; Maurício, A.C. Mesenchymal Stem Cells (MSCs) as a Potential Therapeutic Strategy in COVID-19 Patients: Literature Research. Front. Cell Dev. Biol. 2020, 8, 602647. [Google Scholar] [CrossRef]
- Charitos, I.A.; Del Prete, R.; Inchingolo, F.; Mosca, A.; Carretta, D.; Ballini, A.; Santacroce, L. What we have learned for the future about COVID-19 and healthcare management of it? Acta Biomed. 2020, 91, e2020126. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Balzanelli, M.G.; Distratis, P.; Catucci, O.; Cefalo, A.; Lazzaro, R.; Inchingolo, F.; Tomassone, D.; Aityan, S.K.; Ballini, A.; Nguyen, K.C.D.; et al. Mesenchymal Stem Cells: The Secret Children’s Weapons against the SARS-CoV-2 Lethal Infection. Appl. Sci. 2021, 11, 1696. https://doi.org/10.3390/app11041696
Balzanelli MG, Distratis P, Catucci O, Cefalo A, Lazzaro R, Inchingolo F, Tomassone D, Aityan SK, Ballini A, Nguyen KCD, et al. Mesenchymal Stem Cells: The Secret Children’s Weapons against the SARS-CoV-2 Lethal Infection. Applied Sciences. 2021; 11(4):1696. https://doi.org/10.3390/app11041696
Chicago/Turabian StyleBalzanelli, Mario Giosuè, Pietro Distratis, Orazio Catucci, Angelo Cefalo, Rita Lazzaro, Francesco Inchingolo, Diego Tomassone, Sergey K. Aityan, Andrea Ballini, Kieu C. D. Nguyen, and et al. 2021. "Mesenchymal Stem Cells: The Secret Children’s Weapons against the SARS-CoV-2 Lethal Infection" Applied Sciences 11, no. 4: 1696. https://doi.org/10.3390/app11041696
APA StyleBalzanelli, M. G., Distratis, P., Catucci, O., Cefalo, A., Lazzaro, R., Inchingolo, F., Tomassone, D., Aityan, S. K., Ballini, A., Nguyen, K. C. D., & Gargiulo Isacco, C. (2021). Mesenchymal Stem Cells: The Secret Children’s Weapons against the SARS-CoV-2 Lethal Infection. Applied Sciences, 11(4), 1696. https://doi.org/10.3390/app11041696







