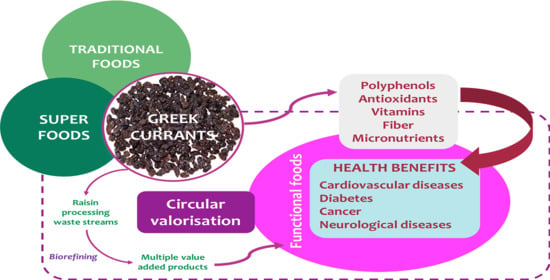
Mediterranean Raisins/Currants as Traditional Superfoods: Processing, Health Benefits, Food Applications and Future Trends within the Bio-Economy Era
Abstract
1. Introduction
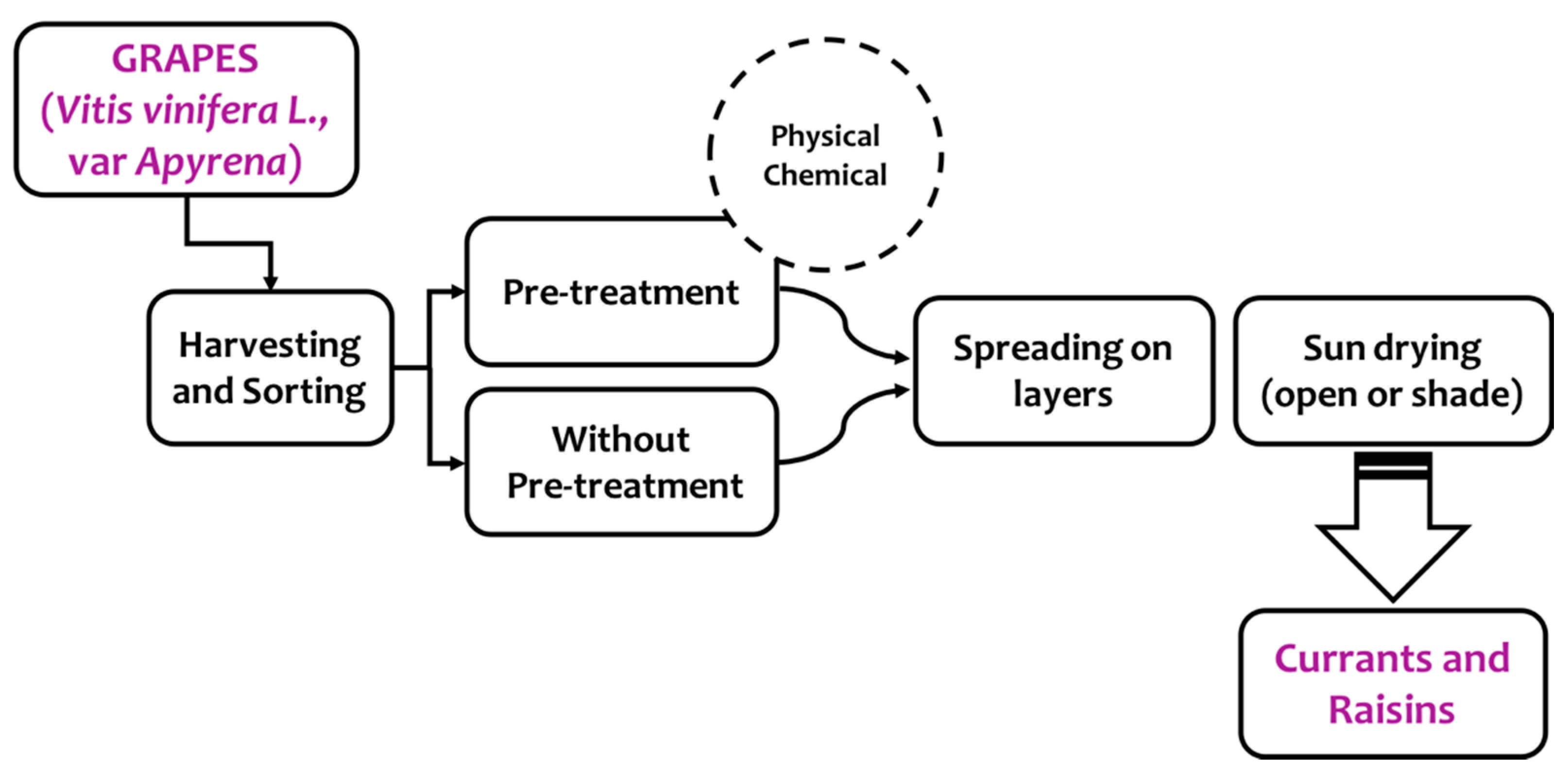
2. Production and Processing Of Raisins: Effect on Final Quality
3. Greek Dried Grapes: Source of Bioactive Compounds
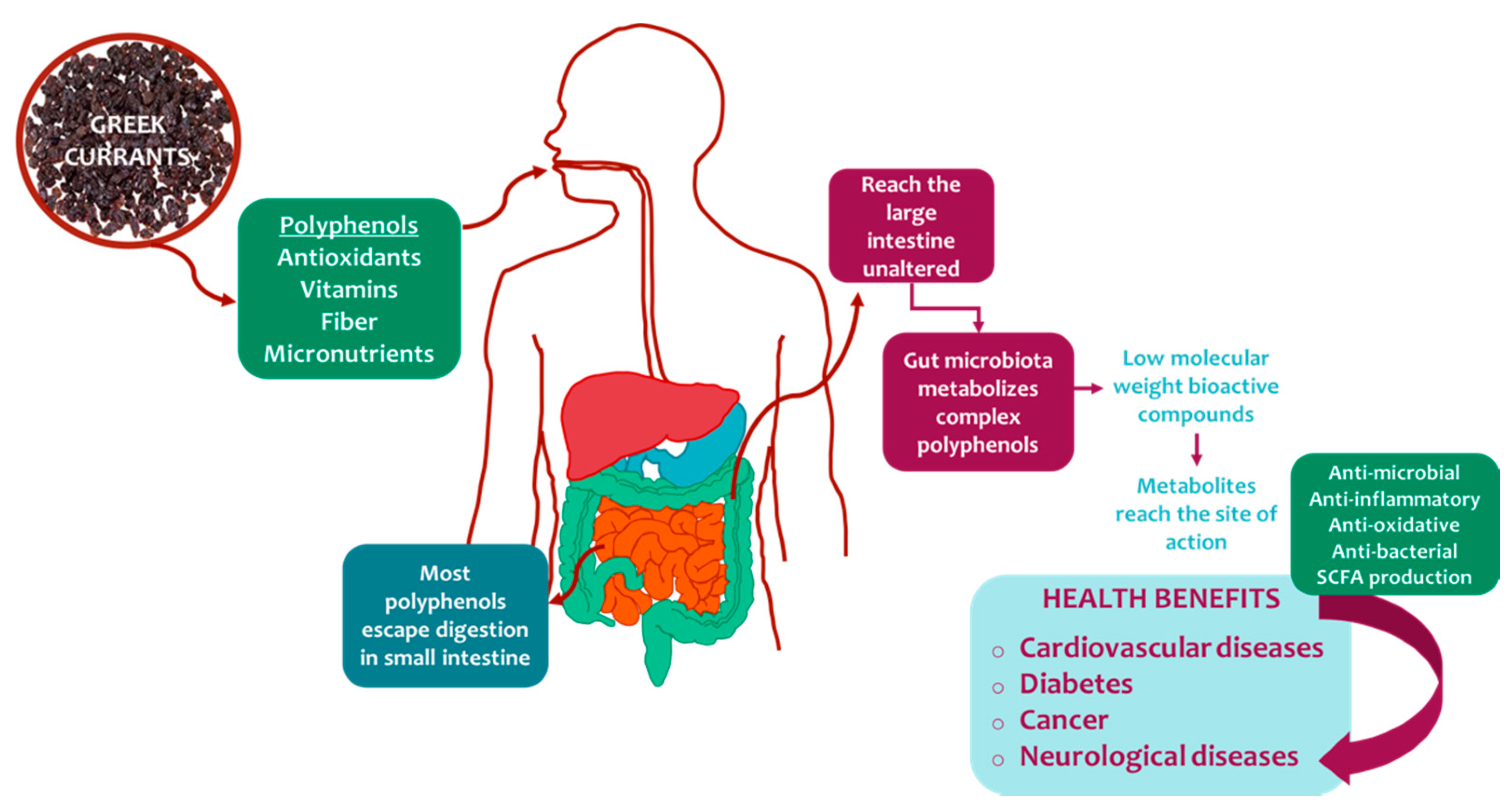
3.1. Nutritional Profile and Bioactive Compounds
3.2. Health Benefits of Greek Dried Grapes (In Vitro, In Vivo, and Clinical Trial Studies)
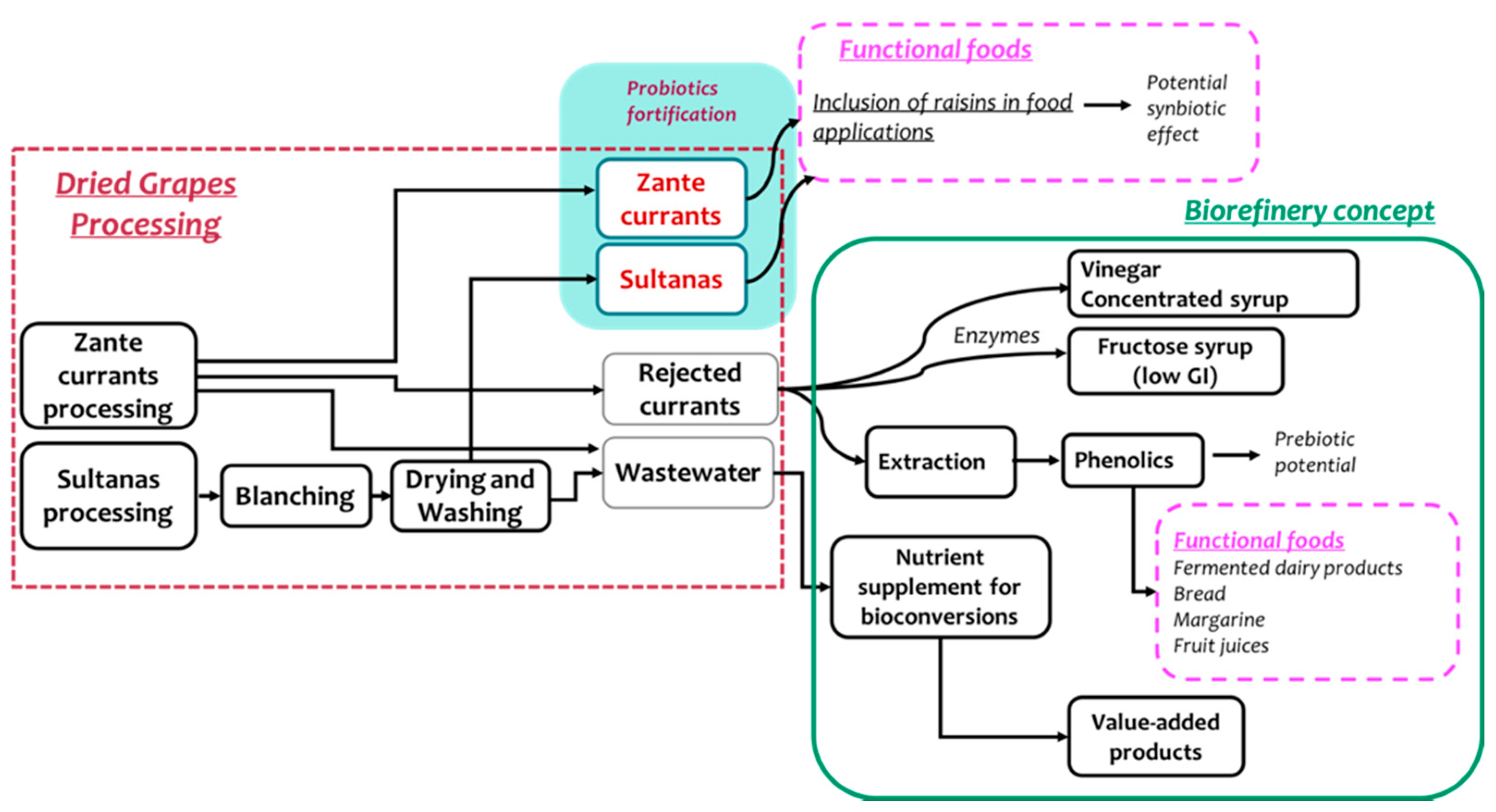
4. Development of Functional Food Products
5. Future Trends and Perspectives: Raisins and Currants Production within the Bio-Economy Era
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- WHO. World Health Organization. Obesity and Overweight. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 20 September 2019).
- Schaffer, S.; Halliwell, B. Do polyphenols enter the brain and does it matter? Some theoretical and practical considerations. Genes Nutr. 2012, 7, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Sreenivasan, L.; Watson, R.R. Chapter 14—Reduction is the New Youth: The Effect of Polyphenols on Brain Aging and Diseases. In Bioactive Nutraceuticals and Dietary Supplements in Neurological and Brain Disease Prevention and Therapy; Watson, R.R., Victor, R.P., Eds.; Academic Press (Elsevier): Amsterdam, The Netherlands, 2015; pp. 137–140. [Google Scholar] [CrossRef]
- Silva Figueiredo, P.; Carla Inada, A.; Marcelino, G.; Maiara Lopes Cardozo, C.; De Cássia Freitas, K.; De Cássia Avellaneda Guimarães, R.; Pereira De Castro, A.; Aragão Do Nascimento, V.; Aiko Hiane, P. Fatty Acids Consumption: The Role Metabolic Aspects Involved in Obesity and Its Associated Disorders. Nutrients 2017, 9, 1158. [Google Scholar] [CrossRef]
- Mohamed, S. Functional foods against metabolic syndrome (obesity, diabetes, hypertension and dyslipidemia) and cardiovasular disease. Trends Food Sci. Technol. 2014, 35, 114–128. [Google Scholar] [CrossRef]
- Ashwell, M. Concepts of Functional Food. Nutr. Food Sci. 2004, 34, 47. [Google Scholar] [CrossRef]
- Küster-Boluda, I.; Vidal-Capilla, I. Consumer attitudes in the election of functional foods. Span. J. Marketing ESIC 2017, 21, 65–79. [Google Scholar] [CrossRef]
- Salanță, L.C.; Uifălean, A.; Iuga, C.-A.; Tofană, M.; Cropotova, J.; Pop, O.L.; Pop, C.R.; Rotar, M.A.; Ávila, M.B.; Velázquez González, C.V. Valuable Food Molecules with Potential Benefits for Human Health. In The Health Benefits of Foods–Current Knowledge and Further Development; Salanță, L.C., Ed.; IntechOpen: London, UK, 2020; Available online: https://www.intechopen.com/books/the-health-benefits-of-foods-current-knowledge-and-further-development/valuable-food-molecules-with-potential-benefits-for-human-health (accessed on 2 February 2021). [CrossRef]
- Technavio. Global Superfoods Market 2019–2023. Available online: https://www.technavio.com/report/global-superfoods-market-industry-analysis (accessed on 20 September 2019).
- Proestos, C. Superfoods: Recent Data on their Role in the Prevention of Diseases. Curr. Res. Nutr. Food Sci. 2018, 6. [Google Scholar] [CrossRef]
- Abuajah, C.I.; Ogbonna, A.C.; Osuji, C.M. Functional components and medicinal properties of food: A review. J. Food Sci. Technol. 2015, 52, 2522–2529. [Google Scholar] [CrossRef] [PubMed]
- Göğüş, F.; Ötles, S.; Erdoğdu, F.; Özçelik, B. Functional and Nutritional Properties of Some Turkish Traditional Foods. In Functional Properties of Traditional Foods. Integrating Food Science and Engineering Knowledge into the Food Chain; Kristbergsson, K., Ötles, S., Eds.; Springer: Boston, MA, USA, 2016; Volume 12, pp. 87–104. [Google Scholar]
- Trichopoulou, A.; Vasilopoulou, E.; Georga, K.; Soukara, S.; Dilis, V. Traditional foods: Why and how to sustain them. Trends Food Sci. Technol. 2006, 17, 498–504. [Google Scholar] [CrossRef]
- Rabelo, C.A.S.; Taarji, N.; Khalid, N.; Kobayashi, I.; Nakajima, M.; Neves, M.A. Formulation and characterization of water-in-oil nanoemulsions loaded with açaí berry anthocyanins: Insights of degradation kinetics and stability evaluation of anthocyanins and nanoemulsions. Food Res. Int. 2018, 106, 542–548. [Google Scholar] [CrossRef]
- Williamson, G.; Carughi, A. Polyphenol content and health benefits of raisins. Nutr. Res. 2010, 30, 511–519. [Google Scholar] [CrossRef]
- Kountouri, A.M.; Gioxari, A.; Karvela, E.; Kaliora, A.C.; Karvelas, M.; Karathanos, V.T. Chemopreventive properties of raisins originating from Greece in colon cancer cells. Food Funct. 2013, 4, 366–372. [Google Scholar] [CrossRef]
- Benlloch-Tinoco, M.; Carranza-Concha, J.; Camacho, M.M.; Martínez-Navarrete, N. Chapter 22—Production of Raisins and its Impact on Active Compounds. In Processing and Impact on Active Components in Food; Preedy, V., Ed.; Academic Press (Elsevier): Amsterdam, The Netherlands, 2015; pp. 181–187. [Google Scholar]
- Wang, J.; Mujumdar, A.S.; Mu, W.; Feng, J.; Zhang, X.; Zhang, Q.; Fang, X.M.; Gao, Z.J.; Xiao, H.W. Grape Drying: Current Status and Future Trends. In Grape and Wine Biotechnology; Morata, A., Loira, I., Eds.; IntechOpen: London, UK, 2016; Available online: https://www.intechopen.com/books/grape-and-wine-biotechnology/grape-drying-current-status-and-future-trends (accessed on 15 September 2019). [CrossRef]
- Khiari, R.; Zemni, H.; Mihoubi, D. Raisin Processing: Physicochemical, Nutritional and Microbiological Quality Characteristics as Affected by Drying Process. Food Rev. Int. 2019, 35, 246–298. [Google Scholar] [CrossRef]
- OIV. Statistical Report on World Vitiviniculture. World Vitiviniculture Situation. Available online: http://www.oiv.int/public/medias/6782/oiv-2019-statistical-report-on-world-vitiviniculture.pdf (accessed on 13 September 2019).
- USDA. United States Department of Agriculture, Foreign Agricultural Service. Market and Trade Data for the Worldwide Production of Raisins. 2017–2019. Available online: https://apps.fas.usda.gov/psdonline/app/index.html#/app/advQuery (accessed on 13 September 2019).
- USDA. United States Department of Agriculture, Foreign Agricultural Service. Raisins: World Markets and Trade. 2018. Available online: https://apps.fas.usda.gov/psdonline/circulars/raisins.pdf (accessed on 13 September 2019).
- Panagopoulou, E.A.; Chiou, A.; Nikolidaki, E.K.; Christea, M.; Karathanos, V.T. Corinthian raisins (Vitis vinifera L., var. Apyrena) antioxidant and sugar content as affected by the drying process: A 3 year study. J. Sci. Food Agric. 2019, 99, 915–922. [Google Scholar] [CrossRef]
- Vasilopoulou, E.; Trichopoulou, A. Greek raisins: A traditional nutritious delicacy. J. Berry Res. 2014, 4, 117–125. [Google Scholar] [CrossRef]
- Panagopoulou, E.A.; Chiou, A.; Karathanos, V.T. Water-soluble vitamin content of sun-dried Corinthian raisins (Vitis vinifera L., var. Apyrena). J. Sci. Food Agric. 2019, 99, 5327–5333. [Google Scholar] [CrossRef]
- Carranza-Concha, J.; Benlloch, M.; Camacho, M.M.; Martínez-Navarrete, N. Effects of drying and pretreatment on the nutritional and functional quality of raisins. Food Bioprod. Process. 2012, 90, 243–248. [Google Scholar] [CrossRef]
- Olivati, C.; De Oliveira Nishiyama, Y.P.; De Souza, R.T.; Janzantti, N.S.; Mauro, M.A.; Gomes, E.; Hermosín-Gutiérrez, I.; Da Silva, R.; Lago-Vanzela, E.S. Effect of the pre-treatment and the drying process on the phenolic composition of raisins produced with a seedless Brazilian grape cultivar. Food Res. Int. 2019, 116, 190–199. [Google Scholar] [CrossRef] [PubMed]
- Adiletta, G.; Russo, P.; Senadeera, W.; Di Matteo, M. Drying characteristics and quality of grape under physical pretreatment. J. Food Eng. 2016, 172, 9–18. [Google Scholar] [CrossRef]
- Deng, L.-Z.; Mujumdar, A.S.; Zhang, Q.; Yang, X.-H.; Wang, J.; Zheng, Z.-A.; Gao, Z.-J.; Xiao, H.-W. Chemical and Physical Pretreatments of Fruits and Vegetables: Effects on Drying Characteristics and Quality Attributes. A Comprehensive Review. Crit. Rev. Food Sci. Nutr. 2019, 59, 1408–1432. [Google Scholar] [CrossRef] [PubMed]
- Lydakis, D.; Fysarakis, I.; Papadimitriou, M.; Kolioradakis, G. Optimization Study of Sulfur Dioxide Application in Processing of Sultana Raisins. Int. J. Food Prop. 2003, 6, 393–403. [Google Scholar] [CrossRef]
- Bennett, L.E.; Singh, D.P.; Clingeleffer, P.R. Micronutrient mineral and folate content of Australian and imported dried fruit products. Crit. Rev. Food Sci. Nutr. 2011, 51, 38–49. [Google Scholar] [CrossRef]
- Jairaj, K.S.; Singh, S.P.; Srikant, K. A review of solar dryers developed for grape drying. Sol. Energy 2009, 83, 1698–1712. [Google Scholar] [CrossRef]
- Belessiotis, V.; Delyannis, E. Solar drying. Sol. Energy 2011, 85, 1665–1691. [Google Scholar] [CrossRef]
- Sharma, A.; Chen, C.R.; Vu Lan, N. Solar-energy drying systems: A review. Renew. Sustain. Energy Rev. 2009, 13, 1185–1210. [Google Scholar] [CrossRef]
- Kassem, A.S.; Shokr, A.Z.; El-Mahdy, A.R.; Aboukarima, A.M.; Hamed, E.Y. Comparison of drying characteristics of Thompson seedless grapes using combined microwave oven and hot air drying. J. Saudi Soc. Agric. Sci. 2011, 10, 33–40. [Google Scholar] [CrossRef]
- Esmaiili, M.; Sotudeh-Gharebagh, R.; Cronin, K.; Mousavi, M.A.E.; Rezazadeh, G. Grape Drying: A Review. Food Rev. Int. 2007, 23, 257–280. [Google Scholar] [CrossRef]
- Wang, Y.; Tao, H.; Yang, J.; An, K.; Ding, S.; Zhao, D.; Wang, Z. Effect of carbonic maceration on infrared drying kinetics and raisin qualities of Red Globe (Vitis vinifera L.): A new pre-treatment technology before drying. Innov. Food Sci. Emerg. Technol. 2014, 26, 462–468. [Google Scholar] [CrossRef]
- Singh, S.P.; Jairaj, K.S.; Kalaveerakkanavar, S. Influence of Variation in Temperature of Dipping Solution on Drying Time and Colour Parameters of Thompson Seedless Grapes. Int. J. Agric. Food Sci. 2014, 4, 36–42. [Google Scholar]
- Doymaz, I.; Altıner, P. Effect of Pretreatment Solution on Drying and Color Characteristics of Seedless Grapes. Food Sci. Biotechnol. 2012, 21, 43–49. [Google Scholar] [CrossRef]
- Bingol, G.; Roberts, J.S.; Balaban, M.O.; Devres, Y.O. Effect of Dipping Temperature and Dipping Time on Drying Rate and Color Change of Grapes. Dry. Technol. 2012, 30, 597–606. [Google Scholar] [CrossRef]
- Guiné, R.P.F.; Almeida, I.C.; Correia, A.C.; Gonçalves, F.J. Evaluation of the physical, chemical and sensory properties of raisins produced from grapes of the cultivar Crimson. J. Food Meas. Charact. 2015, 9, 337–346. [Google Scholar] [CrossRef]
- Mahmutoğlu, T.; Emír, F.; Saygi, Y.B. Sun/solar drying of differently treated grapes and storage stability of dried grapes. J. Food Eng. 1996, 29, 289–300. [Google Scholar] [CrossRef]
- Pangavhane, D.R.; Sawhney, R.L. Review of research and development work on solar dryers for grape drying. Energy Conv. Manag. 2002, 43, 45–61. [Google Scholar] [CrossRef]
- Clary, C.D.; Mejia-Meza, E.; Wang, S.; Petrucci, V.E. Improving Grape Quality Using Microwave Vacuum Drying Associated with Temperature Control. J. Food Sci. 2007, 72, E23–E28. [Google Scholar] [CrossRef]
- Zemni, H.; Sghaier, A.; Khiari, R.; Chebil, S.; Ben Ismail, H.; Nefzaoui, R.; Hamdi, Z.; Lasram, S. Physicochemical, Phytochemical and Mycological Characteristics of Italia Muscat Raisins Obtained Using Different Pre-Treatment and Drying Techniques. Food Bioproc. Technol. 2017, 10, 479–490. [Google Scholar] [CrossRef]
- Dev, S.R.S.; Padmini, T.; Adedeji, A.; Gariépy, Y.; Raghavan, G.S.V. A Comparative Study on the Effect of Chemical, Microwave, and Pulsed Electric Pretreatments on Convective Drying and Quality of Raisins. Dry. Technol. 2008, 26, 1238–1243. [Google Scholar] [CrossRef]
- Rybka, A.C.P.; De Freitas, S.T.; Netto, A.F.; Biasoto, A.C.T. Central Composite Rotatable Design Approach to Optimize Italia Raisin Drying Conditions. Comun. Sci. 2015, 6, 454–462. [Google Scholar] [CrossRef]
- Karathanos, V.T.; Belessiotis, V.G. Sun and Artificial Air Drying Kinetics of some Agricultural Products. J. Food Eng. 1997, 31, 35–46. [Google Scholar] [CrossRef]
- EFSA, European Food Safety Authority. Scientific Panel on Nutrition, Novel Foods and Food Allergens Minutes of the 88th Meeting of the Working Group on Claims, Scientific Topic 6.3 Corinthian Raisins and Lower Blood Glucose Rise after their Consumption Compared to Foods/Drinks Containing Sucrose or Glucose. Available online: http://www.efsa.europa.eu/sites/default/files/wgs/nutrition/ndaclaims.pdf (accessed on 20 December 2019).
- Nikolidaki, E.K.; Chiou, A.; Christea, M.; Gkegka, A.P.; Karvelas, M.; Karathanos, V.T. Sun dried Corinthian currant (Vitis Vinifera, L., var. Apyrena) simple sugar profile and macronutrient characterization. Food Chem. 2017, 221, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Fabani, M.P.; Baroni, M.V.; Luna, L.; Lingua, M.S.; Monferran, M.V.; Paños, H.; Tapia, A.; Wunderlin, D.A.; Feresin, G.E. Changes in the phenolic profile of Argentinean fresh grapes during production of sun-dried raisins. J. Food Compos. Anal. 2017, 58, 23–32. [Google Scholar] [CrossRef]
- Figueiredo-González, M.; Cancho-Grande, B.; Simal-Gándara, J. Effects on colour and phenolic composition of sugar concentration processes in dried on-or-dried-off-vine grapes and their aged or not natural sweet wines. Trends Food Sci. Technol. 2013, 3, 36–54. [Google Scholar] [CrossRef]
- Chiou, A.; Karathanos, V.T.; Mylona, A.; Salta, F.N.; Preventi, F.; Andrikopoulos, N.K. Currants (Vitis vinifera L.) content of simple phenolics and antioxidant activity. Food Chem. 2007, 102, 516–522. [Google Scholar] [CrossRef]
- Chiou, A.; Panagopoulou, E.A.; Gatzali, F.; De Marchi, S.; Karathanos, V.T. Anthocyanins content and antioxidant capacity of Corinthian currants (Vitis vinifera L., var. Apyrena). Food Chem. 2014, 146, 157–165. [Google Scholar] [CrossRef]
- Kaliora, A.C.; Kountouri, A.M.; Karathanos, V.T. Antioxidant Properties of Raisins (Vitis vinifera L.). J. Med. Food 2009, 12, 1302–1309. [Google Scholar] [CrossRef]
- Narendhirakannan, R.T.; Nirmala, J.G.; Caroline, A.; Lincy, S.; Saj, M.; Durai, D. Evaluation of antibacterial, antioxidant and wound healing properties of seven traditional medicinal plants from India in experimental animals. Asian Pac. J. Trop. Biomed. 2012, 2, S1245–S1253. [Google Scholar] [CrossRef]
- Tsouko, E.; Papadaki, A.; Papapostolou, H.; Ladakis, D.; Natsia, A.; Koutinas, A.; Kampioti, A.; Eriotou, E.; Kopsahelis, N. Valorization of Zante currant side-streams for the production of phenolic-rich extract and bacterial cellulose: A novel biorefinery concept. J. Chem. Technol. Biotechnol. 2020, 95, 427–438. [Google Scholar] [CrossRef]
- Naderi, A.; Rezaei, S.; Moussa, A.; Levers, K.; Earnest, C.P. Fruit for sport. Trends Food Sci. Technol. 2018, 74, 85–98. [Google Scholar] [CrossRef]
- Salehi, B.; Vlaisavljevic, S.; Adetunji, C.O.; Adetunji, J.B.; Kregiel, D.; Antolak, H.; Pawlikowska, E.; Uprety, Y.; Mileski, K.S.; Devkota, H.P.; et al. Plants of the genus Vitis: Phenolic compounds, anticancer properties and clinical relevance. Trends Food Sci. Technol. 2019, 91, 362–379. [Google Scholar] [CrossRef]
- Carughi, A.; Lamkin, T.; Perelman, D. Health Benefits of Sun-Dried Raisins. Available online: http://www.raisins.net/Raisins_and_Health_200810.pdf (accessed on 13 July 2019).
- Gol, M.; Ghorbanian, D.; Soltanpour, N.; Faraji, J.; Pourghasem, M. Protective effect of raisin (currant) against spatial memory impairment and oxidative stress in Alzheimer disease model. Nutr. Neurosci. 2019, 22, 110–118. [Google Scholar] [CrossRef]
- Wijayabahu, A.T.; Waugh, S.G.; Ukhanova, M.; Mai, V. Dietary raisin intake has limited effect on gut microbiota composition in adult volunteers. Nutr. J. 2019, 18, 14. [Google Scholar] [CrossRef]
- Viguiliouk, E.; Jenkins, A.L.; Blanco Mejia, S.; Sievenpiper, J.L.; Kendall, C.W.C. Effect of dried fruit on postprandial glycemia: A randomized acute-feeding trial. Nutr. Diabetes 2018, 8, 59. [Google Scholar] [CrossRef]
- Zhu, R.; Fan, Z.; Dong, Y.; Liu, M.; Wang, L.; Pan, H. Postprandial glycaemic responses of dried fruit-containing meals in healthy adults: Results from a randomised trial. Nutrients 2018, 10, 694. [Google Scholar] [CrossRef] [PubMed]
- Ghorbanian, D.; Gol, M.; Pourghasem, M.; Faraji, J.; Pourghasem, K.; Soltanpour, N. Spatial memory and antioxidant protective effects of raisin (currant) in aged rats. Prev. Nutr. Food Sci. 2018, 23, 196–205. [Google Scholar] [CrossRef] [PubMed]
- Kanellos, P.T.; Kaliora, A.C.; Gioxari, A.; Christopoulou, G.O.; Kalogeropoulos, N.; Karathanos, V.T. Absorption and Bioavailability of Antioxidant Phytochemicals and Increase of Serum Oxidation Resistance in Healthy Subjects Following Supplementation with Raisins. Plant. Foods Hum. Nutr. 2013, 68, 411–415. [Google Scholar] [CrossRef] [PubMed]
- Kaliora, A.C.; Kanellos, P.T.; Gioxari, A.; Karathanos, V.T. Regulation of GIP and Ghrelin in Healthy Subjects Fed on Sun-Dried Raisins: A Pilot Study with a Crossover Trial Design. J. Med. Food 2017, 20, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Kanellos, P.T.; Kaliora, A.C.; Liaskos, C.; Tentolouris, N.K.; Perrea, D.; Karathanos, V.T. A Study of Glycemic Response to Corinthian Raisins in Healthy Subjects and in Type 2 Diabetes Mellitus Patients. Plant. Foods Hum. Nutr. 2013, 68, 145–148. [Google Scholar] [CrossRef]
- Kanellos, P.T.; Kaliora, A.C.; Tentolouris, N.K.; Argiana, V.; Perrea, D.; Kalogeropoulos, N.; Kountouri, A.M.; Karathanos, V.T. A pilot randomized controlled trial to examine the health outcomes of raisin consumption in patients with diabetes. Nutrition 2014, 30, 358–364. [Google Scholar] [CrossRef]
- Yanni, A.E.; Efthymiou, V.; Lelovas, P.; Agrogiannis, G.; Kostomitsopoulos, N.; Karathanos, V.T. Effects of dietary Corinthian currants (Vitis vinifera L., var. Apyrena) on atherosclerosis and plasma phenolic compounds during prolonged hypercholesterolemia in New Zealand White rabbits. Food Funct. 2015, 6, 963–971. [Google Scholar] [CrossRef]
- Kanellos, P.T.; Kaliora, A.C.; Protogerou, A.D.; Tentolouris, N.; Perrea, D.N.; Karathanos, V.T. The effect of raisins on biomarkers of endothelial function and oxidant damage; an open-label and randomized controlled intervention. Food Res. Int. 2017, 102, 674–680. [Google Scholar] [CrossRef]
- Kaliora, A.C.; Kountouri, A.M.; Karathanos, V.T.; Koumbi, L.; Papadopoulos, N.G.; Andrikopoulos, N.K. Effect of Greek Raisins (Vitis vinifera, L.) from Different Origins on Gastric Cancer Cell Growth. Nutr. Cancer 2008, 60, 792–799. [Google Scholar] [CrossRef]
- Kaliora, A.C.; Kokkinos, A.; Diolintzi, A.; Stoupaki, M.; Gioxari, A.; Kanellos, P.T.; Dedoussis, G.V.Z.; Vlachogiannakos, J.; Revenas, C.; Ladas, S.D.; et al. Function The effect of minimal dietary changes with raisins in NAFLD patients with non-significant fibrosis: A randomized controlled intervention. Food Funct. 2016, 7, 4533–4544. [Google Scholar] [CrossRef] [PubMed]
- Deli, C.K.; Poulios, A.; Georgakouli, K.; Papanikolaou, K.; Papoutsis, A.; Selemekou, M.; Karathanos, V.T.; Draganidis, D.; Tsiokanos, A.; Koutedakis, Y.; et al. The effect of pre-exercise ingestion of corinthian currant on endurance performance and blood redox status. J. Sports Sci. 2018, 36, 2172–2180. [Google Scholar] [CrossRef] [PubMed]
- Betoret, E.; Betoret, N.; Vidal, D.; Fito, P. Functional foods development: Trends and technologies. Trends Food Sci. Technol. 2011, 22, 498–508. [Google Scholar] [CrossRef]
- Khan, R.S.; Grigor, J.; Winger, R.; Win, A. Functional food product development—Opportunities and challenges for food manufacturers. Trends Food Sci. Technol. 2013, 30, 27–37. [Google Scholar] [CrossRef]
- Schieber, A.; Stintzing, F.; Carle, R. By-products of plant food processing as a source of functional compounds-recent developments. Trends Food Sci. Technol. 2001, 12, 401–413. [Google Scholar] [CrossRef]
- Wei, Q.; Wolf-Hall, C.; Hall, C.A., III. Application of Raisin Extracts as Preservatives in Liquid Bread and Bread Systems. J. Food Sci. 2009, 74, M177–M184. [Google Scholar] [CrossRef]
- Sabanis, D.; Tzia, C.; Papadakis, S. Effect of Different Raisin Juice Preparations on Selected Properties of Gluten-Free Bread. Food Bioproc. Technol. 2008, 1, 374–383. [Google Scholar] [CrossRef]
- Sabanis, D.; Soukoulis, C.; Tzia, C. Effect of Raisin Juice Addition on Bread Produced from Different Wheat Cultivars. Food Sci. Technol. Int. 2009, 15, 325–336. [Google Scholar] [CrossRef]
- Kim, Y.-M. Comparative study on the quality characteristics of white bread using California raisin, Sultana, and Zante currants starter. J. Korean Soc. Food Sci. Nutr. 2018, 47, 579–588. [Google Scholar] [CrossRef]
- Lara, N.; De Sousa, M.; De Pádua Gandra, F.P.; De Angelis-Pereira, M.; Carneiro, J.; Pereira, R. Development of a functional food bar containing coffee. Br. Food J. 2019, 121, 441–453. [Google Scholar] [CrossRef]
- Kumari, R.; Singh, K.; Singh, R.; Bhatia, N.; Nain, M.S. Development of healthy ready-to-eat (RTE) breakfast cereal from popped pearl millet. Indian J. Agric. Sci. 2019, 89, 877–881. [Google Scholar]
- Keshaav Krishnaa, P.; Vishnu Priya, V.; Gayathri, R. Assessment of nutritional value of a newly formulated health drink. Int. J. Res. Pharm. Sci. 2018, 9, 589–593. [Google Scholar]
- Bosnea, L.A.; Kopsahelis, N.; Kokkali, V.; Terpou, A.; Kanellaki, M. Production of a novel probiotic yogurt by incorporation of L. casei enriched fresh apple pieces, dried raisins and wheat grains. Food Bioprod. Process. 2017, 102, 62–71. [Google Scholar] [CrossRef]
- Amirah, A.S.; Nor Syazwani, S.; Shukri, R.; Radhiah, S.; Anis Shobirin, M.H.; Nor-Khaizura, M.A.R.; Wan Zunairah, W.I.; Nurul Shazini, R. Influence of raisins puree on the physicochemical properties, resistant starch, probiotic viability and sensory attributes of coconut milk yogurt. Food Res. 2020, 4, 77–84. [Google Scholar]
- Soukoulis, C.; Tzia, C. Grape, raisin and sugarcane molasses as potential partial sucrose substitutes in chocolate ice cream: A feasibility study. Int. Dairy J. 2018, 76, 18–29. [Google Scholar] [CrossRef]
- Ribeiro, C.; Freixo, R.; Silva, J.; Gibbs, P.; Morais, A.M.M.B.; Teixeira, P. Dried fruit matrices incorporated with a probiotic strains of Lactobacillus plantarum. Int. J. Food Stud. 2014, 3, 69–73. [Google Scholar] [CrossRef]
- Akman, P.K.; Uysal, E.; Ozkaya, G.U.; Tornuk, F.; Durak, M.Z. Development of probiotic carrier dried apples for consumption as snack food with the impregnation of Lactobacillus paracasei. LWT Food Sci. Technol. 2019, 103, 60–68. [Google Scholar] [CrossRef]
- Terpou, A.; Papadaki, A.; Lappa, I.K.; Kachrimanidou, V.; Bosnea, L.A.; Kopsahelis, N. Probiotics in Food Systems: Significance and Emerging Strategies Towards Improved Viability and Delivery of Enhanced Beneficial Value. Nutrients 2019, 11, 1591. [Google Scholar] [CrossRef] [PubMed]
- Athanasopoulos, N.S.; Athanasopoulos, J.S. Currant-wastewater treatment using biological and physicochemical processes. Biores. Technol. 1998, 66, 45–50. [Google Scholar] [CrossRef]
- Tsolcha, O.N.; Tekerlekopoulou, A.G.; Akratos, C.S.; Aggelis, G.; Genitsaris, S.; Moustaka-Gouni, M.; Vayenas, D.V. Biotreatment of raisin and winery wastewaters and simultaneous biodiesel production using a Leptolyngbya-based microbial consortium. J. Clean. Prod. 2017, 148, 185–193. [Google Scholar] [CrossRef]
- Tsolcha, O.N.; Tekerlekopoulou, A.G.; Akratos, C.S.; Aggelis, G.; Genitsaris, S.; Moustaka-Gouni, M.; Vayenas, D.V. Agroindustrial wastewater treatment with simultaneous biodiesel production in attached growth systems using a mixed microbial culture. Water 2018, 10, 1693. [Google Scholar] [CrossRef]
- García-Vargas, M.C.; Contreras, M.D.M.; Castro, E. Avocado-Derived Biomass as a Source of Bioenergy and Bioproducts. Appl. Sci. 2020, 10, 8195. [Google Scholar] [CrossRef]
- Gibson, G.R.; Hutkins, R.; Sanders, M.E.; Prescott, S.L.; Reimer, R.A.; Salminen, S.J.; Scott, K.; Stanton, C.; Swanson, K.S.; Cani, P.D.; et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 491–502. [Google Scholar] [CrossRef] [PubMed]
- Tomasik, P.; Tomasik, P. Probiotics, Non-Dairy Prebiotics and Postbiotics in Nutrition. Appl. Sci. 2020, 10, 1470. [Google Scholar] [CrossRef]
- Lappa, I.K.; Papadaki, A.; Kachrimanidou, V.; Terpou, A.; Koulougliotis, D.; Eriotou, E.; Kopsahelis, N. Cheese Whey Processing: Integrated Biorefinery Concepts and Emerging Food Applications. Foods 2019, 8, 347. [Google Scholar] [CrossRef]
- Bekatorou, A.; Plioni, I.; Sparou, K.; Maroutsiou, R.; Tsafrakidou, P.; Petsi, T.; Kordouli, E. Bacterial Cellulose Production Using the Corinthian Currant Finishing Side-Stream and Cheese Whey: Process Optimization and Textural Characterization. Foods 2019, 8, 193. [Google Scholar] [CrossRef]
- Colombo, F.; Restani, P.; Biella, S.; Di Lorenzo, C. Botanicals in Functional Foods and Food Supplements: Tradition, Efficacy and Regulatory Aspects. Appl. Sci. 2020, 10, 2387. [Google Scholar] [CrossRef]
| Zante Currant 1 | Corinthian Currant 2 | |
|---|---|---|
| Nutrient | Value per 100 g | Value per 100 g |
| Water (g) | 17.6 | NR 3 |
| Protein (g) | 3.43 | 2.5 |
| Total lipid (g) | 0.22 | 0.4 |
| Carbohydrate (g) | 76.98 | 77.5 |
| Sugars (total, g) | 62.28 | NR 3 |
| Fiber (total dietary, g) | 4.4 | 6.7 |
| Minerals | ||
| Calcium (mg) | 88 | 10 |
| Iron (mg) | 1.88 | 4 |
| Magnesium (mg) | 36 | 30 |
| Phosphorus (mg) | 99 | 180 |
| Potassium (mg) | 777 | 710 |
| Sodium (mg) | 43 | NR 3 |
| Zinc (mg) | 0.37 | NR 3 |
| Sub-Varieties | Region in Greece | Total Phenolic Content (mg/g) a | Reference |
|---|---|---|---|
| Vostizza | northern Peloponesse | 2.446 b | [55] |
| Vostizza | northern Peloponesse | 1.55–2.46 | [53] |
| Vostizza | northern Peloponesse (conventional cultivation) | 2.30–2.55 | [54] |
| Vostizza | northern Peloponesse (organic cultivation) | 2.61–2.66 | [54] |
| Messinia | western Peloponesse | 2.41 b | [55] |
| Cretan Sultanas | Crete island | 0.662 b | [55] |
| Nemea | northern Peloponesse | 0.592 b | [55] |
| Provincial | western Peloponesse | 2.01–2.05 | [53] |
| Provincial | western Peloponesse | 2.28–2.64 | [54] |
| Provincial | Zante island | 2.11 | [53] |
| Provincial | Zante island | 2.17–2.43 | [54] |
| Gulf | northern Peloponesse | 1.67–1.90 | [53] |
| Gulf | northern Peloponesse | 2.33–2.35 | [54] |
| Zante (discarded during processing) | Zante island | 1.92–3.36 | [57] |
| Raisin Variety or Sub-Variety | Potential Effect | Trial Conditions | Primary Findings | Reference |
|---|---|---|---|---|
| Dried raisins and Sultanas | Decrease postprandial glycemia | Human study: 10 healthy volunteers 18–75 years, 25 g raisins to displace half of the available carbohydrates in white bread | The combination of dried fruits with high glycemic index foods by displacing available carbohydrate reduces the glycemic response of these foods | [63] |
| Raisins (Vitis vinifera Linn.) | Decrease postprandial glycemia | Human study: 11 healthy volunteers 18–25 years, 75.2 g raisins | The combination of raisins with nuts prevented hyper- or hypo-glycaemia episodes | [64] |
| Corinthian | Decrease postprandial glycemia | Human study: 15 healthy volunteers 20–30 years and 15 volunteers with type 2 diabetes mellitus 55–68 years, 74 g raisins or 50 g glucose (reference food) | Raisins consumption decreased glycemic and insulinemic responses compared to reference food | [68] |
| Corinthian | Bioavailability of phytochemicals | Human study: 15 healthy volunteers 20–40 years, 144 g raisins | The bioavailability confirmed by the identification of several phytochemicals in plasma | [66] |
| Corinthian | Improve biological markers of patients with type 2 diabetes mellitus (T2DM) | Human study: 48 volunteers with T2DM 40–65 years, 36 g raisins daily, 24 weeks | Raisins consumption decreased diastolic blood pressure and increased plasma antioxidant capacity | [69] |
| Dried raisins | Appetite regulation | Human study: 10 healthy volunteers 26.3 ± 0.8 years, 74 g raisins or 50 g glucose (reference food) | The hormones ghrelin and also ghrelin/obestatin ratio was lower in raisins than in glucose consumption, which led to a balanced appetite | [67] |
| Corinthian | Improve biological markers of healthy smokers | Human study: 10 healthy volunteers smoking > 10 cigarettes/day, 20–40 years, 90 g raisins daily, 4 weeks | Blood pressure, total cholesterol and LDL-cholesterol were reduced only in female participants. The quantity of raisins was insufficient to cause a significant effect on smokers | [71] |
| Vostizza, Nemea, Messinia and Cretan Sultanas | Prevention of gastric cancer | In vitro study: methanolic extracts from raisins (1:10, solid:liquid) were tested to AGS human epithelial cells from stomach | Raisins’ extracts inhibited cell proliferation, induced cell death and prevented inflammation | [72] |
| Corinthian and Sultanas | Prevention of colon cancer cells | In vitro study: methanolic extracts from raisins (1:10, solid:liquid) were tested to HT29 human epithelial cells from colon | Raisins’ extracts inhibited cell proliferation and exhibited anti-radical activity | [16] |
| Corinthian | Improve biological markers of patients with non-alcoholic fatty liver disease (NAFLD) | Human study: 50 volunteers with NAFLD disease, around 50 years, 36 g raisins daily, 24 weeks | Minimal dietary changes in patients of non-significant fibrosis resulted in improvements in fasting glucose, inflammation and fibrosis stage | [73] |
| Corinthian and Vostizza | Reduce atheroma development | Animal study: 30 white rabbits, diet supplemented with 10% raisins or 10% raisins and 0.5% cholesterol for 8 weeks | The atherosclerotic lesion formation was retarded in hypercholesterolemic rabbits | [70] |
| Corinthian | Raisins as pre-exercise supplement instead of a glucose drink | Human study: 11 healthy volunteers, 21–45 years, 1.5 g carbohydrates per kg of body weight, 30 min before exercise | Raisins were equally effective to the glucose drink | [74] |
| Sun-dried raisins | Improve athletes’ performance | Human study: 10 males endurance-trained cyclists & triathletes, g raisins during a 10-km cycling time trial | Improvement in performance following a pre-exhaustive bout of exercise and improved Hedonic scores, which implies the viability of an alternative source of carbohydrate | [58] |
| Sun-dried raisins | Modulation of gut microbiota | Human study: 13 healthy volunteers 18–59 years, 28.3 g raisins (contain 2 g of dietary fiber) daily, 14 days | The population of beneficial bacteria was increased (Faecalibacterium prausnitzii, Bacteroidetes sp. and Ruminococcus sp.) and opportunistic pathogens were decreased (Klebsiella sp.) | [62] |
| Sun-dried black raisins (Maviz) | Neuroprotective | Animal study: 12 old rats, 6 g raisins per day for 90 days | Spatial memory was improved and cognitive and motor function were promoted | [65] |
| Sun-dried black raisins | Neuroprotective | Animal study: 24 rats with Alzheimer’s disease, 6 g raisins per day for 60 days | Spatial memory was improved | [61] |
| Type of Raisin-Based Substrate | Food Product | Food Properties | Reference |
|---|---|---|---|
| Raisin juice concentrate and raisin paste | Bread | Increased shelf life, due to the antifungal and antibacterial properties | [78] |
| Concentrate raisin juice and dried raisin juice | Gluten-free bread | Improved loaf volume, color and increased crust and crumb softness | [79] |
| Concentrate raisin juice and dried raisin juice | Bread and durum wheat flour dough | Improved loaf volume, flavor, color, increased crust and crumb softness | [80] |
| California raisins, Sultanas and Zante currants | White bread | Increased bread volume, gumminess, brittleness and hardness | [81] |
| Raisins | Cereal bars | Increased phenolic content and antioxidant activity | [82] |
| Raisins | Ready to eat cereal | Increased nutritional profile | [83] |
| Raisins | Health drink | Lower carbohydrate content | [84] |
| Corinthian raisins | Enriched probiotic yogurt | Improved probiotic viability, sensory and nutritional profile, and syneresis phenomenon | [85] |
| Raisin puree | Coconut milk yogurt | Improved probiotic viability and nutritional profile | [86] |
| Concentrate raisin juice | Chocolate ice-cream | Improved flow properties (highly creamy and gummy) and melting resistance, balanced chocolate flavor | [87] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Papadaki, A.; Kachrimanidou, V.; Lappa, I.K.; Eriotou, E.; Sidirokastritis, N.; Kampioti, A.; Kopsahelis, N. Mediterranean Raisins/Currants as Traditional Superfoods: Processing, Health Benefits, Food Applications and Future Trends within the Bio-Economy Era. Appl. Sci. 2021, 11, 1605. https://doi.org/10.3390/app11041605
Papadaki A, Kachrimanidou V, Lappa IK, Eriotou E, Sidirokastritis N, Kampioti A, Kopsahelis N. Mediterranean Raisins/Currants as Traditional Superfoods: Processing, Health Benefits, Food Applications and Future Trends within the Bio-Economy Era. Applied Sciences. 2021; 11(4):1605. https://doi.org/10.3390/app11041605
Chicago/Turabian StylePapadaki, Aikaterini, Vasiliki Kachrimanidou, Iliada K. Lappa, Effimia Eriotou, Nikolaos Sidirokastritis, Adamantia Kampioti, and Nikolaos Kopsahelis. 2021. "Mediterranean Raisins/Currants as Traditional Superfoods: Processing, Health Benefits, Food Applications and Future Trends within the Bio-Economy Era" Applied Sciences 11, no. 4: 1605. https://doi.org/10.3390/app11041605
APA StylePapadaki, A., Kachrimanidou, V., Lappa, I. K., Eriotou, E., Sidirokastritis, N., Kampioti, A., & Kopsahelis, N. (2021). Mediterranean Raisins/Currants as Traditional Superfoods: Processing, Health Benefits, Food Applications and Future Trends within the Bio-Economy Era. Applied Sciences, 11(4), 1605. https://doi.org/10.3390/app11041605