Microstructural, Electrochemical and In Vitro Analysis of Mg-0.5Ca-xGd Biodegradable Alloys
Abstract
1. Introduction
2. Experimental Details
2.1. Obtaining theMagnesium with Calcium and Gadolinium Alloys’ Structure and Chemical Composition Investigations
2.2. Electrochemical Analysis
2.3. Cytocompatibility Analysis
2.3.1. Alloy Sample Preparation
2.3.2. Cell Culture
2.3.3. Cell Viability
2.3.4. Cell Morphology
3. Results and Discussions
3.1. Microstructural Analysis
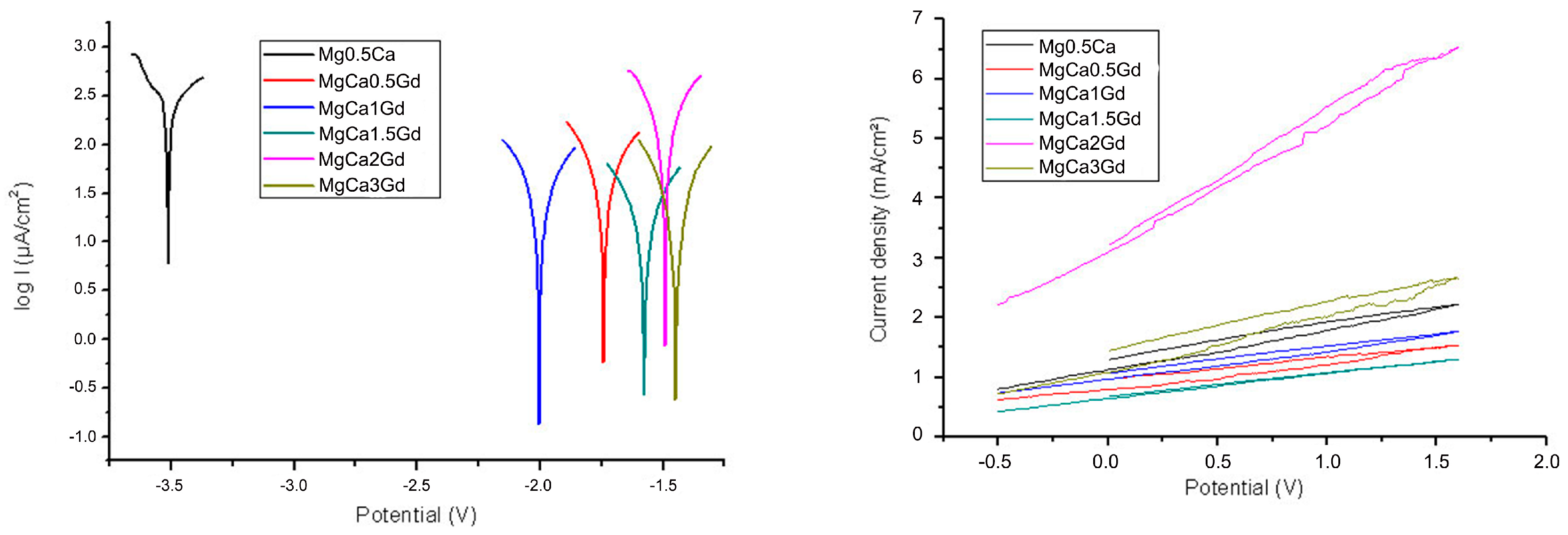
3.2. Electrochemical Corrosion Resistance Analysis
3.3. Cytocompatibility Study
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ali, Y.; Qiu, D.; Jiang, B.; Pan, F.; Zhang, M.-X. Current research progress in grain refinement of cast magnesium alloys: A review article. J. Alloys Compd. 2015, 619, 639–651. [Google Scholar] [CrossRef]
- Song, J.; She, J.; Chen, D.; Pan, F. Latest research advances on magnesium and magnesium alloys worldwide. J. Magnes. Alloy. 2020, 8, 1–41. [Google Scholar] [CrossRef]
- Kiani, F.; Wen, C.; Li, Y. Prospects and strategies for magnesium alloys as biodegradable implants from crystalline to bulk metallic glasses and composites—A review. Acta Biomater. 2020, 103, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Höhn, S.; Virtanen, S.; Boccaccini, A.R. Protein adsorption on magnesium and its alloys: A review. Appl. Surf. Sci. 2019, 464, 212–219. [Google Scholar] [CrossRef]
- Yazdimamaghani, M.; Razavi, M.; Vashaee, D.; Moharamzadeh, K.; Boccaccini, A.R.; Tayebi, L. Porous magnesium-based scaffolds for tissue engineering. Mater. Sci. Eng. 2017, 71, 1253–1266. [Google Scholar] [CrossRef] [PubMed]
- Mario, C.D.; Griffiths, H.; Goktekin, O.; Peeters, N.; Verbist, J.; Bosiers, M.; Deloose, K.; Heublein, B.; Rohde, R.; Kasese, V.; et al. Drug-eluting bioabsorbable magnesium stent. J. Interv. Cardiol. 2004, 17, 391–395. [Google Scholar] [CrossRef] [PubMed]
- Zartner, P.; Cesnjevar, R.; Singer, H.; Weyand, M. First successful implantation of a biodegradable metal stent into the left pulmonary artery of a preterm baby. Catheter. Cardiovasc. Interv. 2005, 66, 590–594. [Google Scholar] [CrossRef] [PubMed]
- Waksman, R.; Pakala, R.; Kuchulakanti, P.K.; Baffour, R.; Hellinga, D.; Seabron, R.; Tio, F.O.; Wittchow, E.; Hartwig, S.; Harder, C.; et al. Safety and efficacy of bioabsorbable magnesium alloy stents in porcine coronary arteries. Catheter. Cardiovasc. Interv. 2006, 68, 607–617. [Google Scholar] [CrossRef]
- Lim, G.B. Interventional cardiology: DREAMS of a bioabsorbable stent coming true. Nat. Rev. Cardiol. 2013, 10, 120. [Google Scholar] [CrossRef]
- Heublein, B.; Rohde, R.; Kaese, V.; Niemeyer, M.; Hartung, W.; Haverich, A. Biocorrosion of magnesium alloys: A new principle in cardiovascular implant technology. Heart 2003, 89, 651–656. [Google Scholar] [CrossRef]
- Panahi, Z.; Tamjid, E.; Rezaei, M. Surface modification of biodegradable AZ91 magnesium alloy by electrospun polymer nanocomposite: Evaluation of in vitro degradation and cytocompatibility. Surf. Coat. Technol. 2020, 386, 125461. [Google Scholar] [CrossRef]
- Toong, D.W.Y.; Ng, J.C.K.; Huang, Y.; Wong, P.E.H.; Leo, H.L.; Venkatraman, S.S.; Ang, H.Y. Bioresorbable metals in cardiovascular stents: Material insights and progress. Materialia 2020, 12, 100727. [Google Scholar] [CrossRef]
- Witte, F.; Hort, N.; Vogt, C.; Cohen, S.; Kainer, K.U.; Willumeit, R.; Feyerabend, F. Degradable biomaterials based on magnesium corrosion. Curr. Opin. Solid State Mater. Sci. 2008, 12, 63–72. [Google Scholar] [CrossRef]
- Waizy, H.; Seitz, J.M.; Reifenrath, J.; Weizbauer, A.; Bach, F.-W.; Lindenberg, A.M.; Denkena, B.; Henning, W. Biodegradable magnesium implants for orthopedic applications. J. Mater. Sci. 2013, 48, 39–50. [Google Scholar] [CrossRef]
- Song, G.L.; Atrens, A. Corrosion Mechanisms of Magnesium Alloys. Adv. Eng. Mater. 1999, 1, 11–33. [Google Scholar] [CrossRef]
- Zheng, Y.F.; Gu, X.N.; Witte, F. Biodegradable metals. Mater. Sci. Eng. R. Rep. 2014, 77, 1–34. [Google Scholar] [CrossRef]
- Wu, G.; Wang, C.; Sun, M.; Ding, W. Recent developments and applications on high-performance cast magnesium rare-earth alloys. J. Magnes. Alloy. 2020. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, S.; Wu, R.; Hou, L.; Zhang, M. Recent developments in high-strength Mg-RE-based alloys: Focusing on Mg-Gd and Mg-Y systems. J. Magnes. Alloy. 2018, 6, 277–291. [Google Scholar] [CrossRef]
- Liu, J.; Bian, D.; Zheng, Y.; Chu, X.; Lin, Y.; Wang, M.; Lin, Z.; Li, M.; Zhang, Y.; Guan, S. Comparative in vitro study on binary Mg-RE (Sc, Y, La, Ce, Pr, Nd, Sm, Eu, Gd, Tb, Dy, Ho, Er, Tm, Yb and Lu) alloy systems. Acta Biomater. 2020, 102, 508–528. [Google Scholar] [CrossRef]
- Tekumalla, S.; Seetharaman, S.; Almajid, A.; Gupta, M. Mechanical Properties of Magnesium-Rare Earth Alloy Systems: A Review. Metals 2015, 5, 1–39. [Google Scholar] [CrossRef]
- Feyerabend, F.; Fischer, J.; Holtz, J.; Witte, F.; Willumeit, R.; Drucker, H.; Vogt, C.; Hort, N. Evaluation of short-term effects of rare earth and other elements used in magnesium alloys on primary cells and cell lines. Acta Biomater. 2010, 6, 1834–1842. [Google Scholar] [CrossRef] [PubMed]
- Hort, N.; Huang, Y.; Fechner, D.; Störmer, M.; Blawert, C.; Witte, F.; Vogt, C.; Drücker, H.; Willumeit, R.; Kainer, K.U.; et al. Magnesium alloys as implant materials—Principles of property design for Mg–RE alloys. Acta Biomater. 2010, 6, 1714–1725. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.; Chen, R.S.; Han, E.H. Effects of rare-earth elements Gd and Y on the solid solution strengthening of mg alloys. J. Alloys Compd. 2009, 481, 379–384. [Google Scholar] [CrossRef]
- Peng, Q.; Wu, Y.; Fang, D.; Meng, J.; Wang, L. Microstructures and properties of melt-spun and as-cast Mg-20Gd binary alloy. J. Rare Earths 2006, 24, 466–470. [Google Scholar] [CrossRef]
- Nodooshan, H.R.J.; Liu, W.C.; Wu, G.H.; Rao, Y.; Zhou, C.X.; He, S.P.; Ding, W.J.; Mahmudi, R. Effect of Gd content on microstructure and mechanical properties of Mg–Gd–Y–Zr alloys under peak-aged condition. Mater. Sci. Eng. A 2014, 615, 79–86. [Google Scholar] [CrossRef]
- Xiaobo, Z.; Zhixin, B.; Zhangzhong, W.; Yujuan, W.; Yajun, X. Effect of LPSO structure on mechanical properties and corrosion behavior of as-extruded GZ51K magnesium alloy. Mater. Lett. 2016, 163, 250–253. [Google Scholar]
- Myrissa, A.; Braeuer, S.; Martinelli, E.; Willumeit-Römer, R.; Goessler, W.; Weinberg, A.M. Gadolinium accumulation inorgans of Sprague-Dawley® rats after implantation of abiodegradable magnesium-gadolinium alloy. Acta Biomater. 2017, 48, 521. [Google Scholar] [CrossRef]
- Xuenan, G.; Yufeng, Z.; Yan, C.; Shengping, Z.; Tingfei, X. In vitro corrosion and biocompatibility of binary magnesium alloys. Biomaterials 2009, 30, 484–498. [Google Scholar] [CrossRef]
- Wang, X.; Dong, L.H.; Li, J.T.; Li, X.L.; Ma, X.L.; Zheng, Y.F. Microstructure, mechanical property and corrosion behavior of interpenetrating (HA+β-TCP)/MgCa composite fabricated by suction casting. Mater. Sci. Eng. C 2013, 33, 4266–4273. [Google Scholar] [CrossRef]
- Salahshosor, M.; Li, C.; Liu, Z.Y.; Fang, X.Y.; Guo, Y.B. Surface integrity and corrosion performance of biomedical magnesiumcalcium alloy processed by hybrid dry cutting-finish burnishing. J. Mech. Behav. Biomed. Mater. 2018, 78, 246–253. [Google Scholar] [CrossRef]
- Staiger, M.P.; Pietak, A.M.; Huadmai, J.; Dias, G. Magnesium and its alloys as orthopedic biomaterials: A review. Biomaterials 2006, 27, 1728–1734. [Google Scholar] [CrossRef] [PubMed]
- Zeng, R.C.; Dietzel, W.; Witte, F.; Hort, N.; Blawert, C. Progress and challenge for magnesium alloys as biomaterials. Adv. Eng. Mater. 2008, 10, B3–B14. [Google Scholar] [CrossRef]
- Li, Z.; Gu, X.; Lou, S.; Zheng, Y. The development of binary Mg-Ca alloys for use as biodegradable materials within bone. Biomaterials 2008, 29, 1329–1344. [Google Scholar] [CrossRef] [PubMed]
- Atrens, A.; Song, G.L.; Liu, M.; Shi, Z.; Cao, F.; Dargusch, M.S. Review of recent developments in the field of magnesium corrosion. Adv. Eng. Mater. 2015, 17, 400–453. [Google Scholar] [CrossRef]
- Yu, H.D.; Zhang, Z.Y.; Win, K.Y.; Chan, J.; Teoh, S.H.; Han, M.Y. Bioinspired fabrication of 3D hierarchical porous nanomicrostructures of calcium carbonate for bone regeneration. Chem. Commun. 2010, 35, 6578. [Google Scholar] [CrossRef] [PubMed]
- Maradze, D.; Capel, A.; Martin, N.; Lewis, M.P.; Zheng, Y.; Liu, Y. In vitro investigation of cellular effects of magnesium and magnesium-calcium alloy corrosion products on skeletal muscle regeneration. J. Mater. Sci. Technol. 2019, 35, 2503–2512. [Google Scholar] [CrossRef]
- Schwander, M.; Leu, M.; Stumm, M.; Dorchies, O.M.; Ruegg, U.T.; Schittny, J.; Müller, U. β1 Integrins Regulate Myoblast Fusion and Sarcomere Assembly. Dev. Cell 2003, 4, 673–685. [Google Scholar] [CrossRef]
- Zeng, R.C.; Qi, W.C.; Cui, H.Z.; Zhang, F.; Li, S.Q.; Han, E.H. In vitro corrosion of as-extruded Mg–Ca alloys—The influence of Ca concentration. Corros. Sci. 2015, 96, 23–31. [Google Scholar] [CrossRef]
- Salahshoor, M.; Guo, Y. Biodegradable Orthopedic Magnesium-Calcium (MgCa) Alloys, Processing, and Corrosion Performance. Materials 2012, 5, 135–155. [Google Scholar] [CrossRef]
- Jeong, Y.S.; Kim, W.J. Enhancement of mechanical properties and corrosion resistance of Mg–Ca alloys through microstructural refinement by indirect extrusion. Corros. Sci. 2014, 82, 392–403. [Google Scholar] [CrossRef]
- Master Alloys Supplier Website. Available online: http://www.hbnewmaterial.com/supplier-129192-master-alloy (accessed on 15 June 2020).
- Lupescu, S.; Istrate, B.; Munteanu, C.; Minciuna, M.G.; Focsaneanu, S.; Earar, K. Characterization of Some Master Mg-X System (Ca, Mn, Zr, Y) Alloys Used in Medical Applications. Rev. Chim. 2017, 68, 1408–1413. [Google Scholar] [CrossRef]
- Istrate, B.; Munteanu, C.; Lupescu, S.; Chelariu, R.; Vlad, M.D.; Vizureanu, P. Electrochemical Analysis and In Vitro Assay of Mg-0.5Ca-xY Biodegradable Alloys. Materials 2020, 13, 3082. [Google Scholar] [CrossRef] [PubMed]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Vlad, M.D.; Valle, L.J.; Poeată, I.; Barracó, M.; López, J.; Torres, R.; Fernández, E. Injectable iron-modified apatitic bone cement intended for kyphoplasty: Cytocompatibility study. J. Mater. Sci. Mater. Med. 2008, 19, 3575–3583. [Google Scholar] [CrossRef]
- Vlad, M.D.; Valle, L.J.; Poeată, I.; López, J.; Torres, R.; Barracó, M.; Fernández, E. Biphasic calcium sulfate dihydrate/iron-modified alpha-tricalcium phosphate bone cement for spinal applications: In vitro study. Biomed. Mater. 2010, 5, 025006. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Schmutz, P.; Uggowitzer, P.; Song, G.; Atrens, A. The influence of Y (Y) on the corrosion of Mg–Y binary alloys. Corros. Sci. 2010, 52, 3687–3701. [Google Scholar] [CrossRef]
- Tong, X.; Zai, L.; You, G.; Wu, H.; Wen, H.; Long, S. Effects of bonding temperature on microstructure and mechanical properties of diffusion-bonded joints of as-cast Mg–Gd alloy. Mater. Sci. Eng. A 2019, 767, 138408. [Google Scholar] [CrossRef]
- Kang, L.; Liang, Z.; Guohua, W.; Wencai, L.; Wenjian, D. Effect of Y and Gd content on the microstructure and mechanical properties of Mg–Y–RE alloys. J. Magnes. Alloy. 2019, 7, 345–354. [Google Scholar]
- Junxiu, C.; Lili, T.; Xiaoming, Y.; Ke, Y. Effect of minor content of Gd on the mechanical and degradable properties of as-cast Mg-2Zn-xGd-0.5Zr alloys. J. Mater. Sci. Technol. 2019, 35, 503–511. [Google Scholar]
- Sudholz, A.D.; Gusieva, K.; Chen, X.B.; Muddle, B.; Gibson, M.; Birbilis, N. Electrochemical behaviour and corrosion of Mg–Y alloys. Corros. Sci. 2011, 53, 2277–2282. [Google Scholar] [CrossRef]
- Südholz, A.D.; Kirkland, N.T.; Buchheit, R.G.; Birbilis, N. Electrochemical Properties of Intermetallic Phases and Common Impurity Elements in Magnesium Alloys. Electrochem. Solid-State Lett. 2011, 14, C5–C7. [Google Scholar] [CrossRef]
- Li, Z.; Sun, S.; Chen, M.; Fahlman, B.D.; Liu, D.; Bi, H. In vitro and in vivo corrosion, mechanical properties and biocompatibility evaluation of MgF2-coated Mg-Zn-Zr alloy as cancellous screws. Mater. Sci. Eng. C 2017, 75, 1268–1280. [Google Scholar] [CrossRef] [PubMed]
- Gilles, R.; Belkhir, M.; Compere, P.; Libioulle, C.; Thiry, M. Effect of high osmolarity acclimation on tolerance to hyperosmotic shocks in L929 cultured cells. Tissue Cell 1995, 27, 679–687. [Google Scholar] [CrossRef]
- Esmaily, M.; Svensson, J.E.; Fajardo, S.; Birbilis, N.; Frankel, G.S.; Virtanen, S.; Arrabal, R.; Thomas, S.; Johansson, L.G. Fundamentals and advances in magnesium alloy corrosion. Prog. Mater. Sci. 2017, 89, 92–193. [Google Scholar] [CrossRef]
- ISO 10993-5:2009—Biological Evaluation of Medical Devices—Part 5: Tests for In Vitro Cytotoxicity. Available online: http://nhiso.com/wp-content/uploads/2018/05/ISO-10993-5-2009.pdf (accessed on 25 September 2020).
- Mbele, G.O.; Deloulme, J.C.; Gentil, B.J.; Delphin, C.; Ferro, M.; Garin, J.; Takahashi, M.; Baudier, J. The zinc and calcium-binding S100B interacts and co-localizes with IQGAP1 during dynamic rearrangement of cell membranes. J. Biol. Chem. 2002, 277, 49998–50007. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Hort, N.; Laipple, D.; Höche, D.; Huang, Y.; Kainer, K.U.; Willumeit, R.; Feyerabend, F. Element distribution in the corrosion layer and cytotoxicity of alloy Mg–10Dy during in vitro biodegradation. Acta Biomater. 2013, 9, 8475–8487. [Google Scholar] [CrossRef] [PubMed]
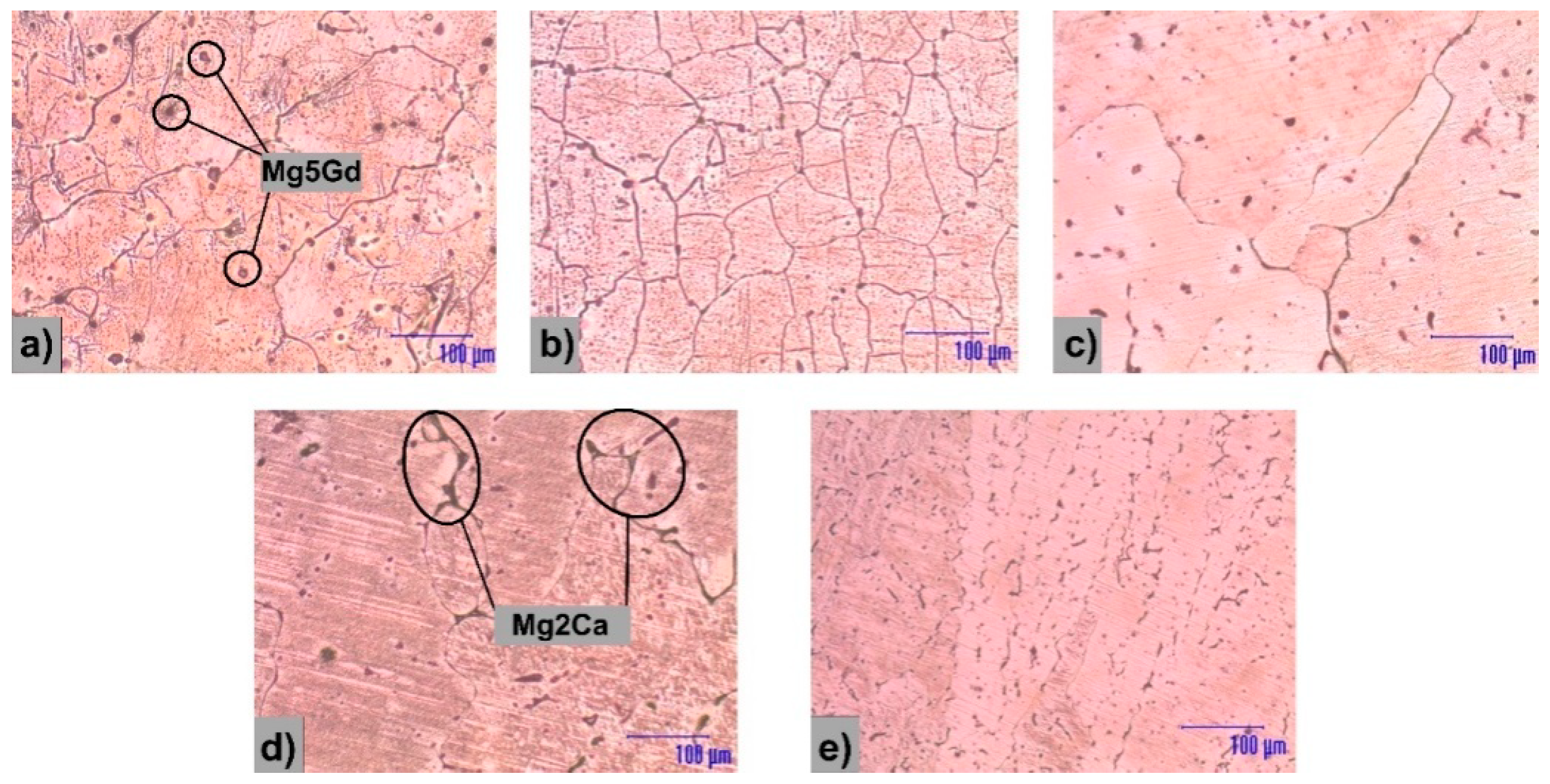

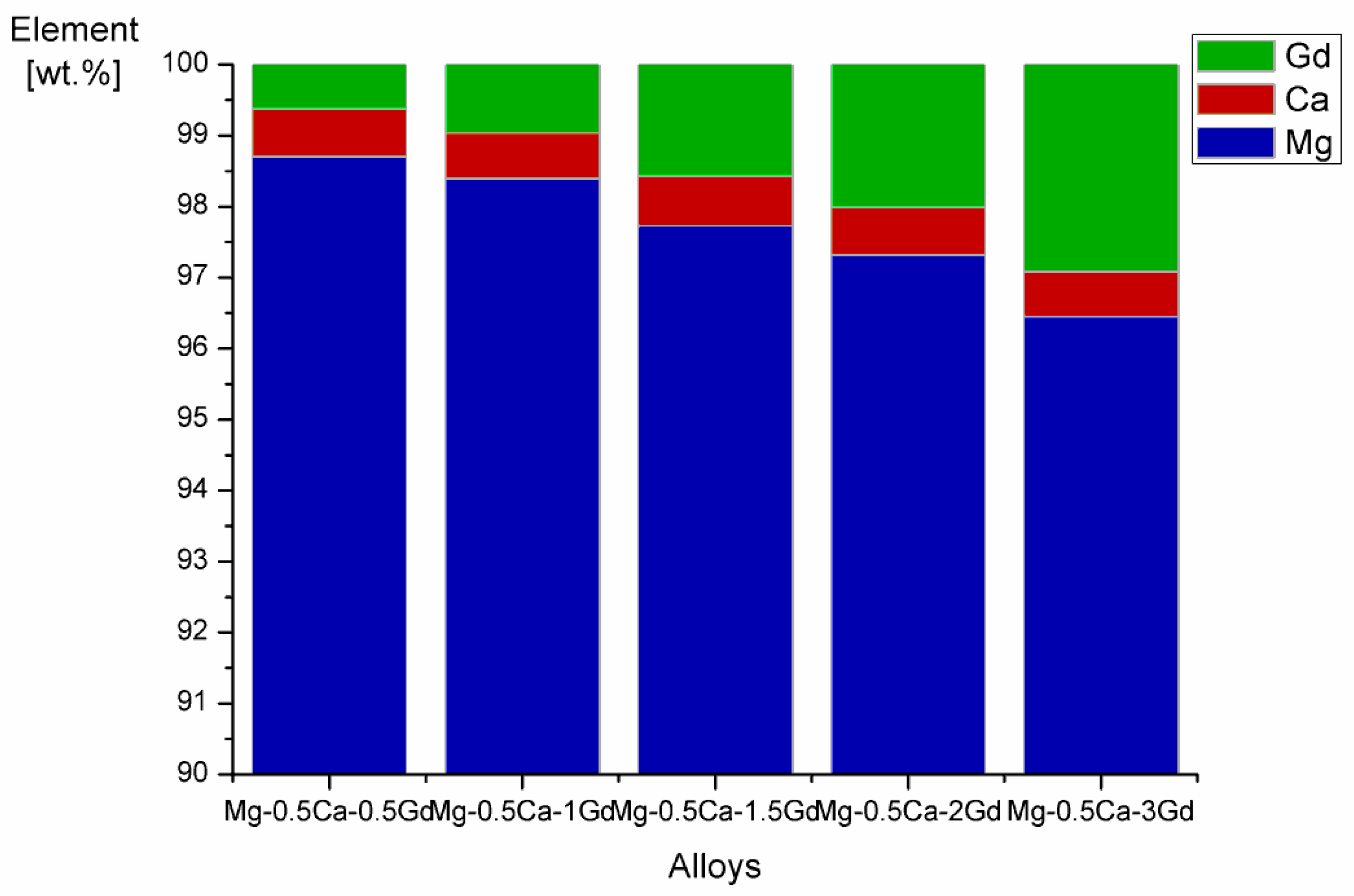
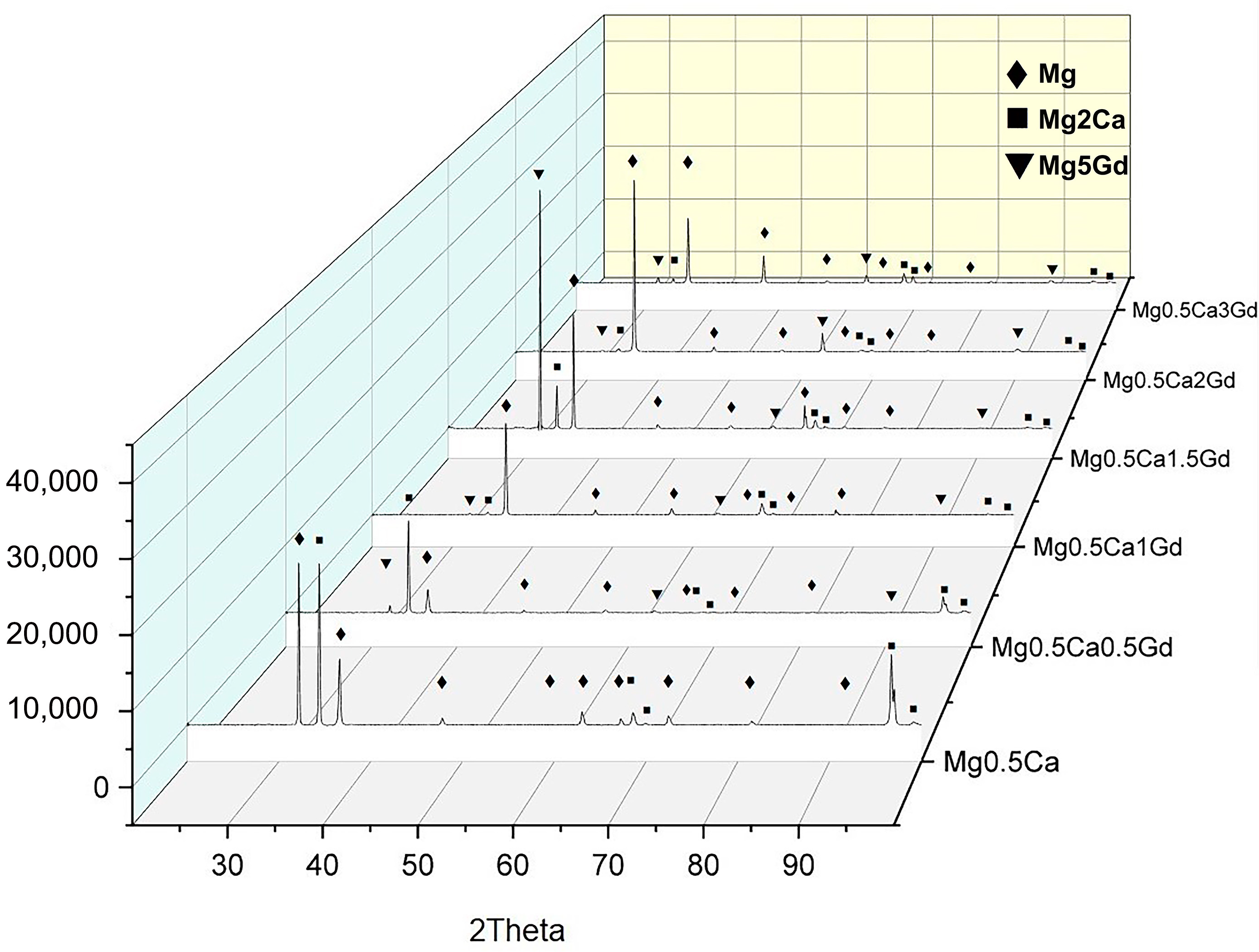



| Materials | Mg/Ca/Gd (wt.%) | Fe (wt.%) | Ni (wt.%) | Cu (wt.%) | Si (wt.%) | Al (wt.%) |
|---|---|---|---|---|---|---|
| Pure Mg | Mg (99 wt.%) | 0.15–0.2 | 0.17–0.2 | 0.14–0.2 | 0.15–0.2 | 0.16–0.2 |
| Mg15Ca | Ca (15.29 wt.%) | 0.004 | 0.001 | 0.003 | 0.013 | 0.011 |
| Mg30Gd | Gd (28.05 wt.%) | 0.010 | 0.001 | 0.001 | 0.006 | 0.011 |
| Specimens | Chemical Composition | Mg [Grams] | Mg–15Ca (Grams) | Mg–30Gd (Grams) |
|---|---|---|---|---|
| MgCa0.5Gd | Mg-0.5%Ca-0.5%Gd | 21.82 | 0.77 | 0.41 |
| MgCa1Gd | Mg-0.5%Ca-1%Gd | 21.42 | 0.77 | 0.82 |
| MgCa1.5Gd | Mg-0.5%Ca-1.5%Gd | 21.00 | 0.77 | 1.23 |
| MgCa2Gd | Mg-0.5%Ca-2%Gd | 20.59 | 0.77 | 1.64 |
| MgCa3Gd | Mg-0.5%Ca-3%Gd | 19.77 | 0.77 | 2.46 |
| Electrolyte Ions (mmol/dm3) | Na+ | K+ | Mg2+ | Ca2+ | Cl− | HCO3− | HPO42− | SO42− |
|---|---|---|---|---|---|---|---|---|
| Simulated body fluid | 142 | 5 | 1.5 | 2.5 | 147.8 | 4.2 | 1 | 0.5 |
| Human blood plasma | 142 | 5 | 1.5 | 2.5 | 103 | 27 | 1 | 0.5 |
| Compound | Space Group | Crystal System | a (Å) | b (Å) | c (Å) | α (°) | β (°) | γ (°) | Cell Volume (106 µm3) | RIR |
|---|---|---|---|---|---|---|---|---|---|---|
| Mg | P63/mmc | Hexagonal | 3.2089 | 3.2089 | 5.2101 | 90 | 90 | 120 | 46.46 | 4.01 |
| Mg2Ca | P63/mmc | Hexagonal | 6.2250 | 6.2250 | 10.1800 | 90 | 90 | 120 | 341.63 | 2.30 |
| Mg5Gd | F43m | Cubic | 3.9500 | 3.9500 | 3.9500 | 90 | 90 | 90 | 61.63 | 18.05 |
| Sample | E0 mV | ba mV | bc mV | Rp ohm·cm2 | Jcor µA/cm2 | Vcor mm/Y | OCP V |
|---|---|---|---|---|---|---|---|
| Mg–0.5Ca | −3511.2 | 217.8 | −188.6 | 0.114 | 170.11 | 4.2 | −1.72 |
| Mg–Ca–0.5Gd | −1740.8 | 283.8 | −240.4 | 0.993 | 42.42 | 1.0 | −1.56 |
| Mg–Ca–1 Gd | −2002.8 | 307.4 | −265.0 | 1.54 | 32.2 | 0.8 | −1.62 |
| Mg–Ca–1.5 Gd | −1576.0 | 264.1 | −250.2 | 2.55 | 16.53 | 0.41 | −1.68 |
| Mg–Ca–2 Gd | −1491.9 | 196.9 | −179.8 | 2.76 | 12.71 | 0.28 | −1.61 |
| Mg–Ca–3 Gd | −1447.7 | 256.1 | −236.3 | 2.25 | 5.92 | 0.16 | −1.58 |
| Mg–0.5Ca | Chemical elements | Mg | Ca | Gd | O | Cl | Na |
| wt.% | wt.% | wt.% | wt.% | wt.% | wt.% | ||
| Surface with oxides | 81 | 0.8 | - | 18.2 | - | - | |
| Mg–0.5Ca–0.5Gd | Chemical elements | Mg | Ca | Gd | O | Cl | Na |
| wt.% | wt.% | wt.% | wt.% | wt.% | wt.% | ||
| Surface with oxides | 70.5 | 0.8 | 0.8 | 27.9 | - | - | |
| Mg–0.5Ca–1Gd | Chemical elements | Mg | Ca | Gd | O | Cl | Na |
| wt.% | wt.% | wt.% | wt.% | wt.% | wt.% | ||
| Surface with oxides | 77.7 | 1.1 | 1.2 | 18.7 | - | 1.3 | |
| Mg–0.5Ca–1.5Gd | Chemical elements | Mg | Ca | Gd | O | Cl | Na |
| wt.% | wt.% | wt.% | wt.% | wt.% | wt.% | ||
| Surface with oxides | 72.2 | 0.8 | 0.8 | 23.3 | 2.2 | 0.6 | |
| Mg–0.5Ca–2Gd | Chemical elements | Mg | Ca | Gd | O | Cl | Na |
| wt.% | wt.% | wt.% | wt.% | wt.% | wt.% | ||
| Surface with oxides | 65.3 | 0.9 | 0.6 | 32.2 | 0.3 | 0.7 | |
| Mg–0.5Ca–3Gd | Chemical elements | Mg | Ca | Gd | O | Cl | Na |
| wt.% | wt.% | wt.% | wt.% | wt.% | wt.% | ||
| Surface with oxides | 83.1 | 0.5 | 1.3 | 15.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Istrate, B.; Munteanu, C.; Cimpoesu, R.; Cimpoesu, N.; Popescu, O.D.; Vlad, M.D. Microstructural, Electrochemical and In Vitro Analysis of Mg-0.5Ca-xGd Biodegradable Alloys. Appl. Sci. 2021, 11, 981. https://doi.org/10.3390/app11030981
Istrate B, Munteanu C, Cimpoesu R, Cimpoesu N, Popescu OD, Vlad MD. Microstructural, Electrochemical and In Vitro Analysis of Mg-0.5Ca-xGd Biodegradable Alloys. Applied Sciences. 2021; 11(3):981. https://doi.org/10.3390/app11030981
Chicago/Turabian StyleIstrate, Bogdan, Corneliu Munteanu, Ramona Cimpoesu, Nicanor Cimpoesu, Oana Diana Popescu, and Maria Daniela Vlad. 2021. "Microstructural, Electrochemical and In Vitro Analysis of Mg-0.5Ca-xGd Biodegradable Alloys" Applied Sciences 11, no. 3: 981. https://doi.org/10.3390/app11030981
APA StyleIstrate, B., Munteanu, C., Cimpoesu, R., Cimpoesu, N., Popescu, O. D., & Vlad, M. D. (2021). Microstructural, Electrochemical and In Vitro Analysis of Mg-0.5Ca-xGd Biodegradable Alloys. Applied Sciences, 11(3), 981. https://doi.org/10.3390/app11030981









