Evaluation of Cement Performance Using Industrial Byproducts Such as Nano MgO and Fly Ash from Greece
Abstract
:1. Introduction
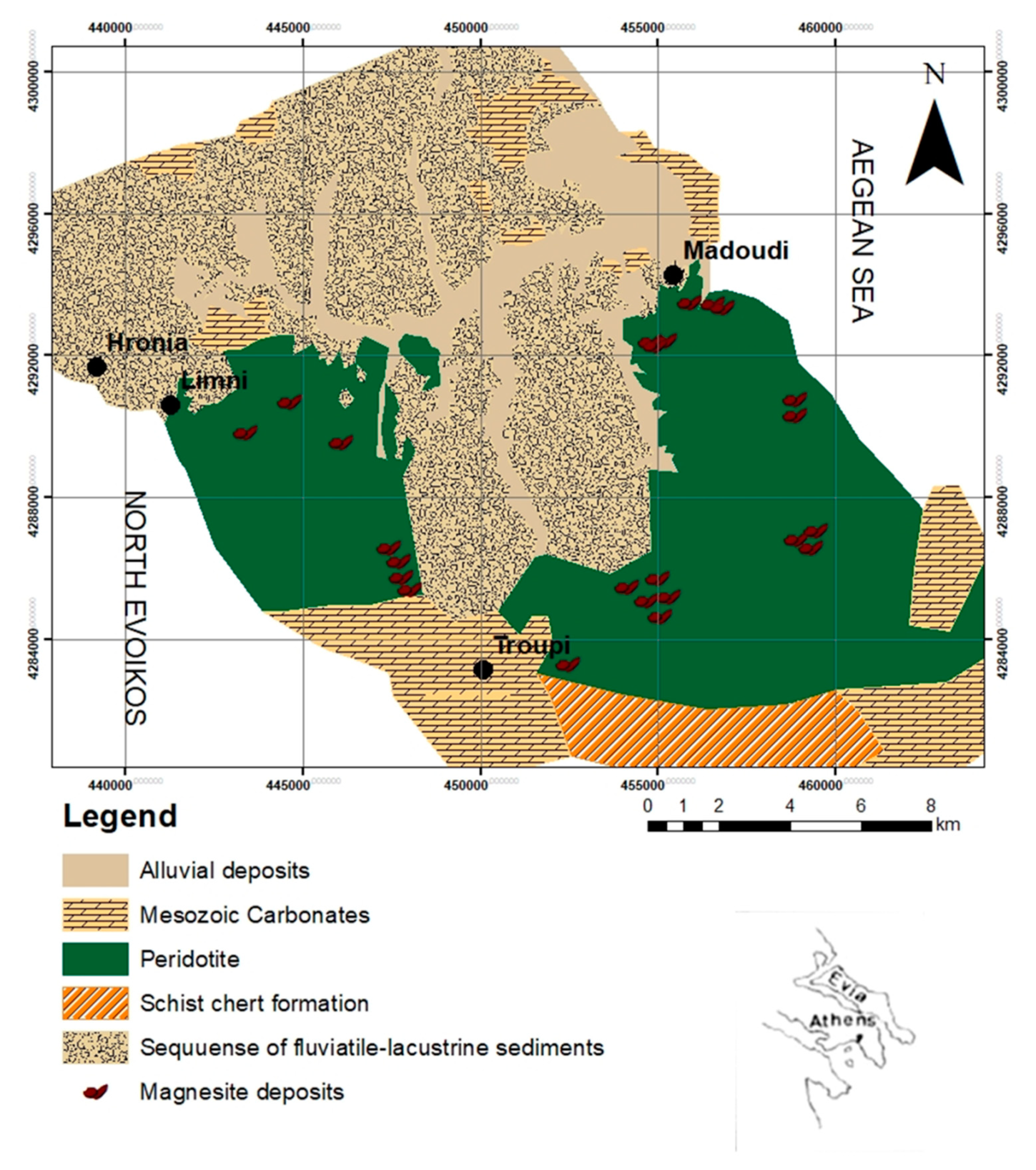
2. Geological Setting of Magnesite Tailings
3. Materials and Methods
3.1. Materials
3.2. Methods
3.2.1. Methods for Raw Materials
3.2.2. Methods for the Produced Cement
4. Results and Discussion
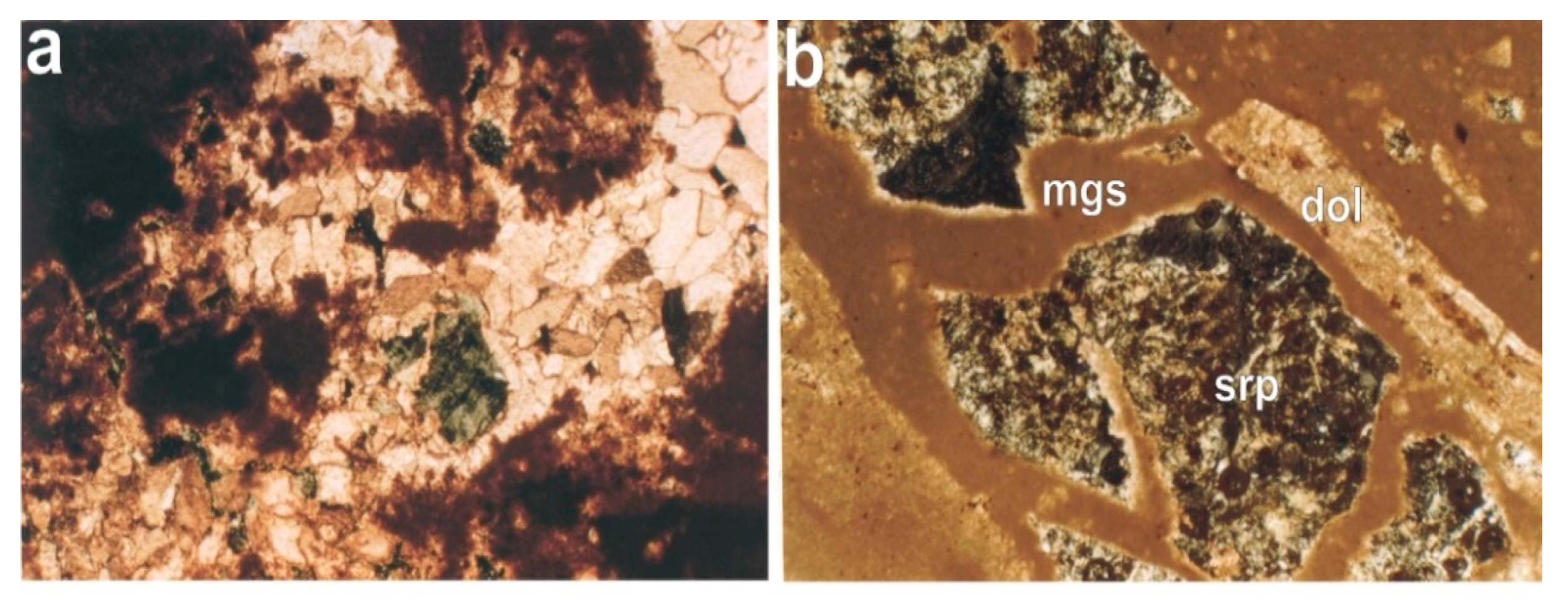
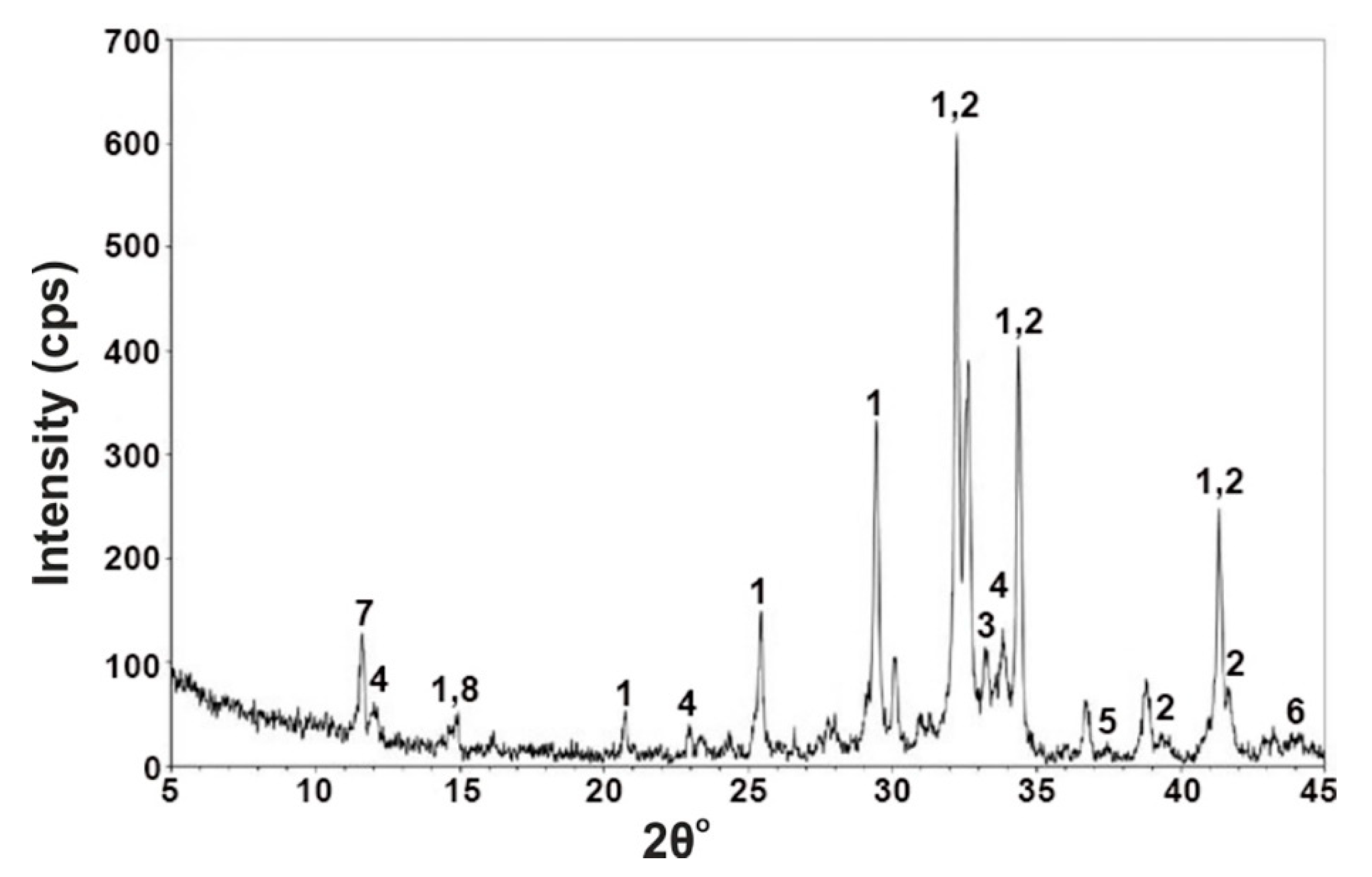
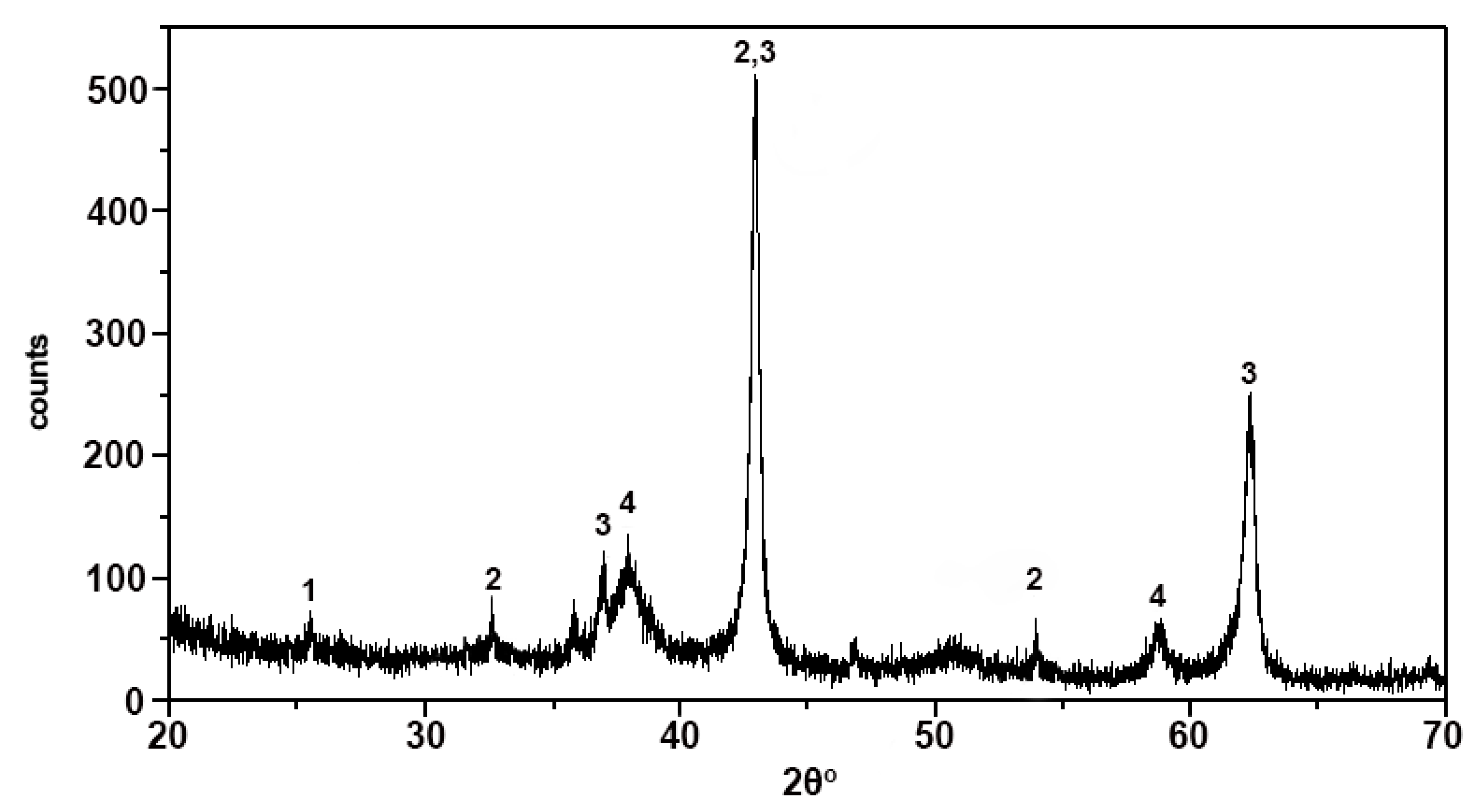
4.1. Raw Materials
4.2. Factors Influencing the Behavior of the Products
4.2.1. The Effect of Nano MgO on the Consistency of the Produced Cement
4.2.2. The Effect of Nano MgO on the Fluidity of the Produced Cement
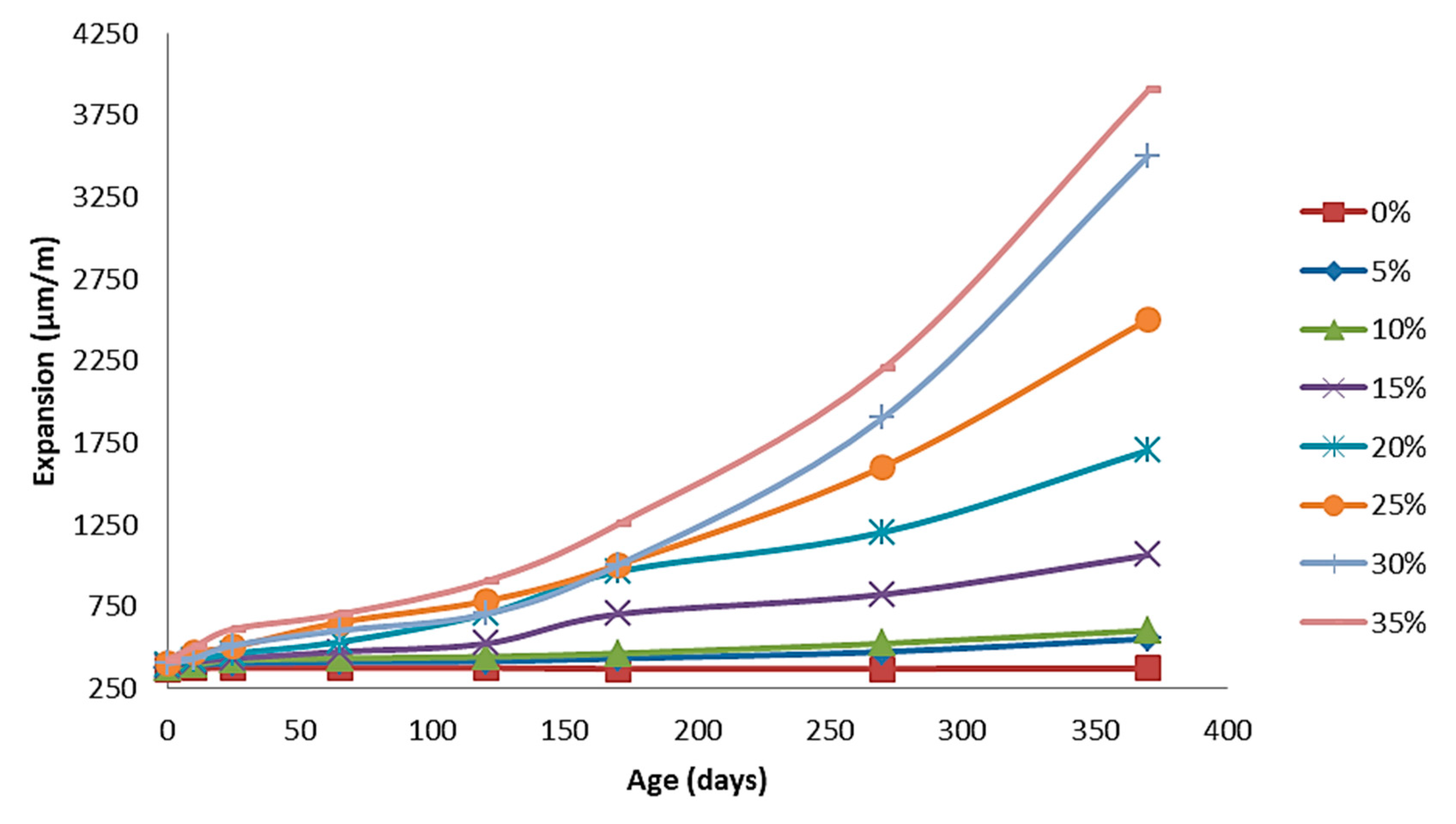
4.2.3. The Effect of Nano MgO on the Expansion of the Produced Cement
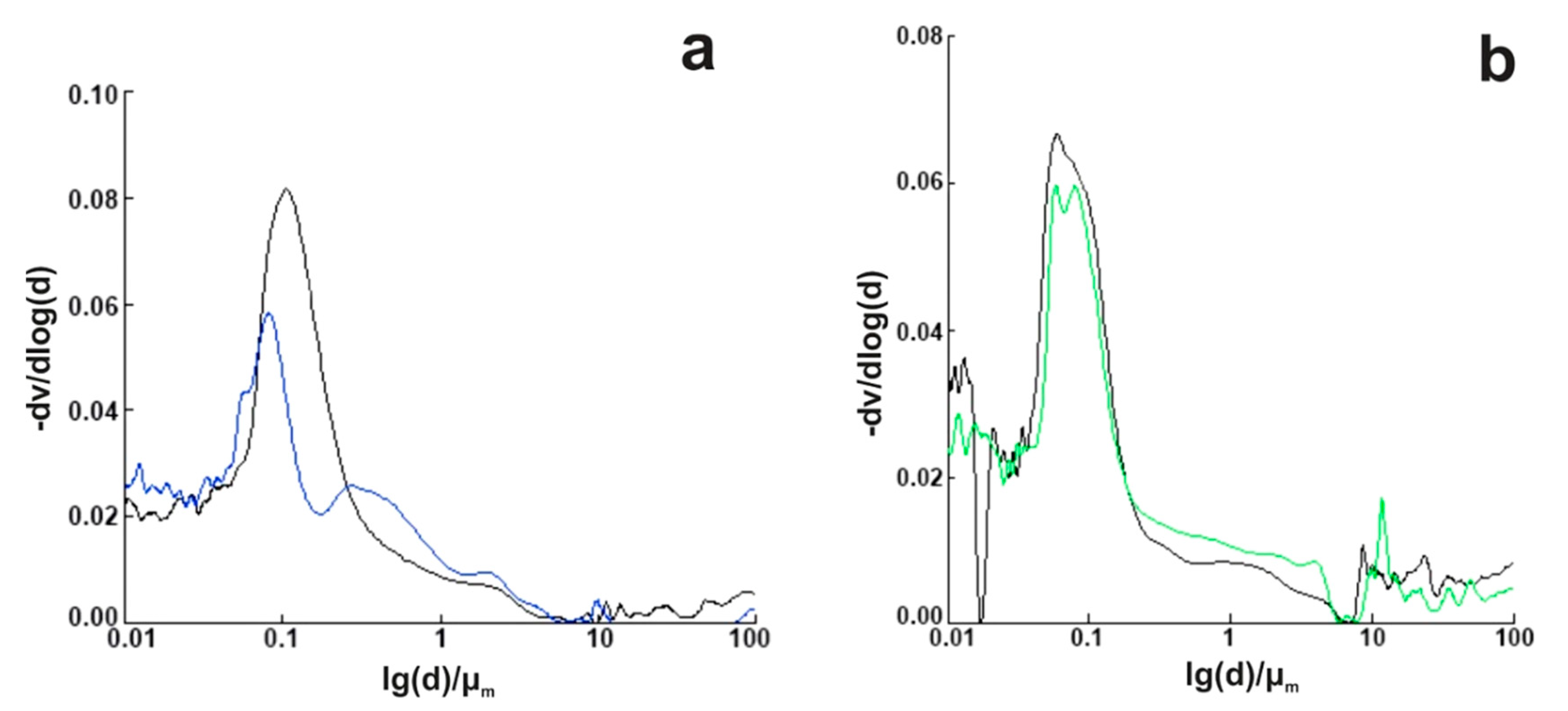
4.2.4. The Effect of Nano MgO/Fly Ash on the Cement Porosity
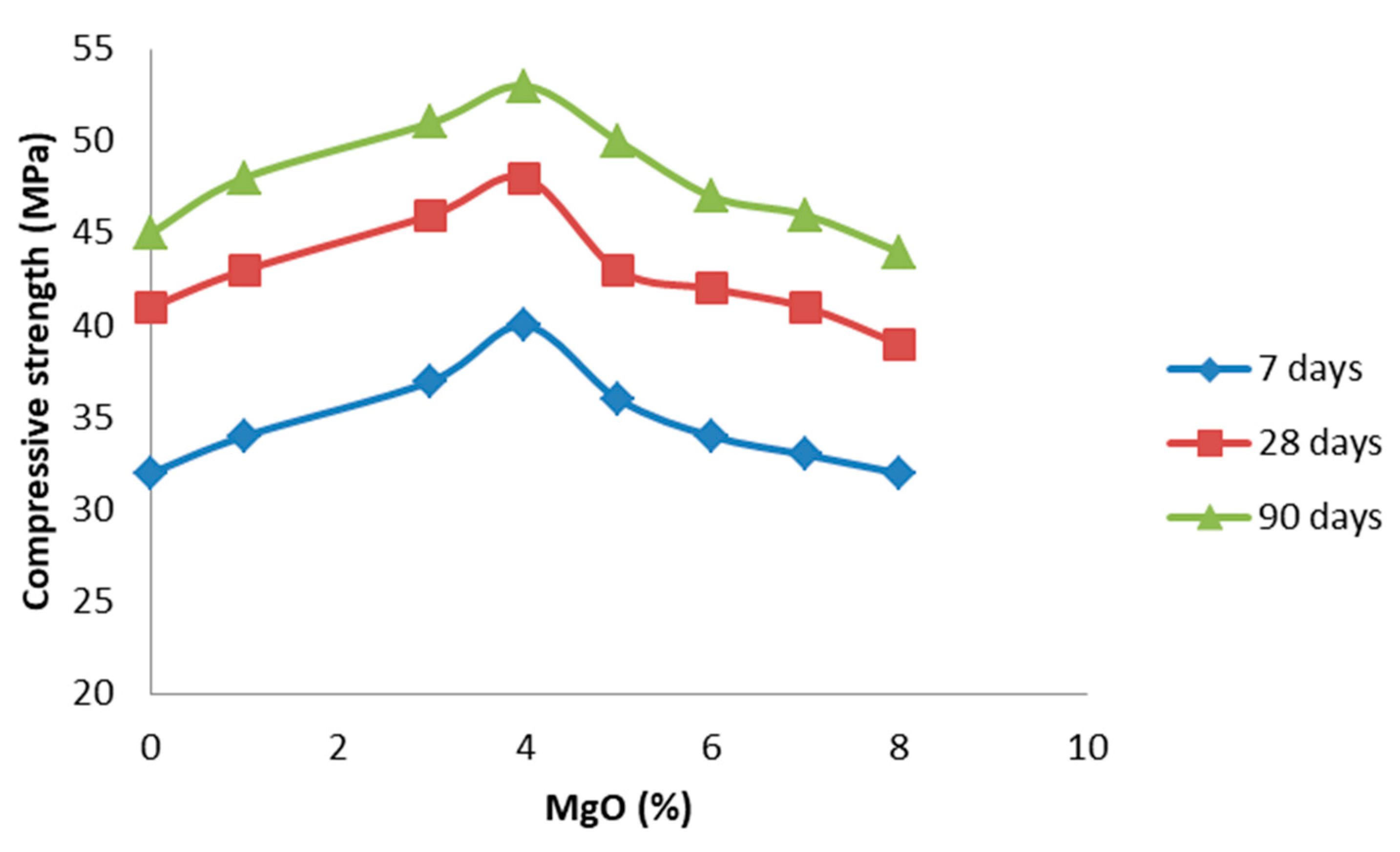
4.2.5. The Effect of Nano MgO on the Mechanical Performance of the Produced Cement
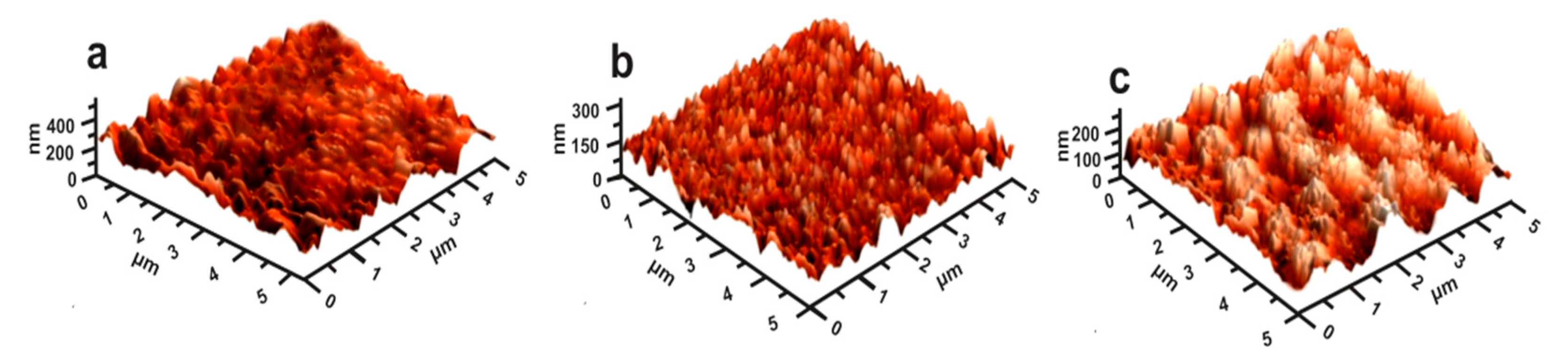
4.2.6. The Effect of Microroughness on the Cement Performance
5. Conclusions
- Byproducts from different regions of Greece, such as magnesite tailings from Evoia and fly ash from Kardia (Ptolemais), which have been combined together for first time, are suitable reinforcement additives.
- The effect of nano MgO content is the most critical parameter for the physicomechanical performance of produced cement, compared to that of the fly ash content.
- More satisfactory results in the physicomechanical properties of the produced cement were from samples of group II, containing 3–4% of nano MgO. Nano MgO content above 4% seems to have a negative influence on the compressive strength of the produced cement, simultaneously reducing its durability. Therefore, the amount of nano MgO determines the physicomechanical performance of the produced cement.
- The increased microroughness of the cements is in accordance with the mechanical behavior of the produced cements.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
| AFM | Atomic force microscopy |
| ASTM | International standards worldwide |
| dol | Dolomite |
| EDS | Energy dispersive spectrometer |
| MEA | MgO expansion agent |
| mgs | Magnesite |
| OP | Ordinary Portland |
| PC | Portland cement |
| srp | Serpentine |
| XRD | X-ray diffraction |
| XRF | X-ray fluorescence |
| XRPD | X-ray powdered diffraction |
References
- Freedonia. World Construction Aggregates-Demand and Sales Forecasts, Market, Share, Market, Size, Market, Leaders. In Industry Study No. 3389; The Freedonia Group: Cleveland, OH, USA, 2016; p. 390. [Google Scholar]
- USGS. Commodity Statistics and Information Mineral. In Yearbooks; USA Geological Survey: Washington, DC, USA, 2015. [Google Scholar]
- Petrounias, P.; Rogkala, A.; Giannakopoulou, P.P.; Lampropoulou, P.; Xanthopoulou, V.; Koutsovitis, P.; Koukouzas, N.; Lagogiannis, I.; Lykokanellos, G.; Golfinopoulos, A. An Innovative Experimental Petrographic Study of Concrete Produced by Animal Bones and Human Hair Fibers. Sustainability 2021, 13, 8107. [Google Scholar] [CrossRef]
- Petrounias, P.; Giannakopoulou, P.P.; Rogkala, A.; Lampropoulou, P.; Tsikouras, B.; Rigopoulos, I.; Hatzipanagiotou, K. Petrographic and Mechanical Characteristics of Concrete Produced by Different Type of Recycled Materials. Geosciences 2019, 9, 264. [Google Scholar] [CrossRef] [Green Version]
- José, N.; Ahmed, H.; Miguel, B.; Luís, E.; Jorge, D.B. Magnesia (MgO) Production and Characterization, and Its Influence on the Performance of Cementitious Materials: A Review. Materials 2020, 13, 4752. [Google Scholar] [CrossRef]
- Ye, Y.; Liu, Y.; Shi, T.; Hu, Z.; Zhong, L.; Wang, H.; Chen, Y. Effect of Nano-Magnesium Oxide on the Expansion Performance and Hydration Process of Cement-Based Materials. Materials 2021, 14, 3766. [Google Scholar] [CrossRef]
- Shayanfar, M.A.; Farnia, S.M.H.; Ghanooni-Bagha, M.; Massoudi, M.S. The Effect of Crack Width On Chloride Threshold Reaching Time in Reinforced Concrete Members. Asian J. Civ. Eng. 2020, 21, 625–637. [Google Scholar] [CrossRef]
- Jackson, N. Civil Engineering Materials; Macmillan Press Ltd.: London, UK, 1981. [Google Scholar]
- Neville, A.M. Properties of Concrete, ELSB, 5th ed.; Pearson Education Publishing Ltd.: London, UK, 2005. [Google Scholar]
- Kabir, H.; Hooton, R.D. Evaluating Soundness of Concrete Containing Shrinkage-Compensating MgO Admixtures. Constr. Build. Mater. 2020, 253, 119141. [Google Scholar] [CrossRef]
- Ye, Q.; Chen, H.; Wang, Y.; Wang, S.; Lou, Z. Effect of MgO and gypsum content on long-term expansion of low heat Portland slag cement with slight expansion. Cem. Concr. Compos. 2004, 26, 331–337. [Google Scholar] [CrossRef]
- Lou, Z.; Ye, Q.; Chen, H.; Wang, Y.; Shen, J. Hydration of MgO in clinker and its expansion property. J. Chin. Ceram. Soc. 1998, 26, 430–436. [Google Scholar] [CrossRef]
- Walling, S.; Provis, J. Magnesia-based cements: A journey of 150 years, and cements for the future? Chem. Rev. 2016, 116, 4170–4204. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Wang, S.; Huang, J.; Wei, Z.; Guan, B.; Fang, J. Experimental Investigation on the Properties and Microstructure of Magnesium Oxychloride Cement Prepared with Caustic Magnesite and Dolomite. Constr. Build. Mater. 2015, 85, 247–255. [Google Scholar] [CrossRef]
- Dimopoulos, I.; Anastassakis, G. Recovery of magnesite from fine waste material rejected before hand-sorting. In Proceedings of the XV Balkan Mineral Processing Congress, Sozopol, Bulgaria, 12–16 June 2013; Volume 1, pp. 213–216. [Google Scholar]
- Martek, I.; Hosseini, M.R.; Shrestha, A.; Edwards, D.J.; Durdyev, S. Barriers inhibiting the transition to sustainability within the Australian construction industry: An investigation of technical and social interactions. J. Clean. Prod. 2019, 211, 2812–2892. [Google Scholar] [CrossRef]
- Kuppig, V.D.; Cook, Y.C.; Carter, D.A.; Larson, N.J.; Williams, R.E.; Dvorak, B.I. Implementation of sustainability improvements at the facility level: Motivations and barriers. J. Clean. Prod. 2016, 139, 15291–15538. [Google Scholar] [CrossRef]
- Murtagh, N.; Scott, L.; Fan, J.L. Sustainable and resilient construction: Current status and future challenges. J. Clean. Prod. 2020, 268, 10. [Google Scholar] [CrossRef]
- Vishwakarma, V.; Uthaman, S. Environmental impact of sustainable green concrete. In Smart Nanoconcretes and Cement-Based Materials: Properties, Modelling and Applications; Elsevier: Amsterdam, The Netherlands, 2019; pp. 241–255. [Google Scholar]
- Robertson, A.H.E. Origin and emplacement of an inferred late Jurassic subduction-accretion complex, Euboea, eastern Greece. Geol. Mag. 1991, 128, 27–41. [Google Scholar] [CrossRef]
- Katsikatsos, G. Geological Map, Sheet LIMNI; Scale 1:50.000,1.G.M.E.; Institute of Geology and Mineral Exploration: Athens, Greece, 1980. [Google Scholar]
- Spray, J.G.; Roddick, J.C. Petrology and 40Ar/39Ar Geochronology of some Hellenic Sub-Ophiolite Metamorphic Rocks. Contrib. Mineral. Petrol. 1980, 72, 43–55. [Google Scholar] [CrossRef]
- Thuizat, R.; Whitechurch, H.; Montigny, R.; Juteau, Τ. K-Ar dating of some infra-ophiolitic metamorphic soles from the Eastern Mediterranean: New evidence for oceanic thrustings before obduction. Earth Planet. Sci. Lett. 1981, 52, 302–310. [Google Scholar] [CrossRef]
- Gartzos, E. Carbon and oxygen irotope constraints on the origin of magnesite deposits, North Evia (Greece). Schweiz. Mineral. Petrogr. Mitt. 1990, 70, 67–72. [Google Scholar]
- Mehta, P.K. Pozzolanic and cementitious byproducts as mineral admixtures for concrete––A critical review. In Proceedings of the 1st International Conference on the Use of Fly Ash, Silica Fume, Slag, and Natural Pozzolans in Concrete, ACI SP-79, Detroit, MI, USA, July 1983; p. 1. [Google Scholar]
- Rai, P.; Qiu, W.; Pei, H.; Chen, J.; Ai, X.; Liu, Y.; Ahmad, M. Effect of Fly Ash and Cement on the Engineering Characteristic of Stabilized Subgrade Soil: An Experimental Study. Geofluids 2021, 2021, 1–11. [Google Scholar] [CrossRef]
- Koukouzas, N.K.; Zeng, R.; Perdikatsis, V.; Xu, W.; Kakaras, E.K. Mineralogy and geochemistry of Greek and Chinese coal fly ash. Fuel 2006, 85, 2301–2309. [Google Scholar] [CrossRef]
- Gao, P.; Lu, X.; Geng, F.; Li, X.; Hou, J.; Lin, H.; Shi, N. Production of MgO-type Expansive Agent in Dam Concrete by Use of Industrial By-Products. Build. Environ. 2008, 43, 453–457. [Google Scholar] [CrossRef]
- Wang, X. Investigations of Nano-Materials Modified Cement Slurry(Stone) Structure and Properties. Master’s Thesis, China University of Petroleum, Beijing, China, 2016; pp. 37–38. [Google Scholar]
- Ding, W. Study on the Properties of Cement Paste and Mortar with MgO Expansive Admixture. Master’s Thesis, Southwest University of Science and Technology, Mianyang, China, 2016; pp. 36–37. [Google Scholar]
- Mo, L.; Deng, M. Thermal behavior of cement matrix with high-volume mineral admixtures at early hydration age. Cem. Concr. Res. 2006, 36, 1992–1998. [Google Scholar]
- Mo, L.; Min, D.; Tang, M. Effects of calcination condition on expansion property of MgO-type expansive agent used in cement-based materials. Cem. Concr. Res. 2010, 40, 437–446. [Google Scholar] [CrossRef]
- Zhang, X. Microscopic Characterization and Early age Performance of MgO and Nano MgO High Performance Concrete. Master’s Thesis, Shandong University, Jinan, China, 2019; pp. 33–34. [Google Scholar]
- Ding, W.; Tan, K.; Liu, L.; Tang, K.; Zhao, C. Influence research of light-burned magnesia on autogenous shrinkage and pore structure of steam-cured cement paste. China Concr. Cem. Prod. 2016, 5, 21–26. [Google Scholar]
- Ye, Q.; Yu, S.; Zhang, Z.; Zhang, Q.; Shi, T. Effect of Nano-MgO on the expansion and strength of hardened cement paste. J. Build. Mater. 2017, 20, 765–769. [Google Scholar]
- Zhou, L.; Wang, Y.; Zhang, G.; Zhang, F. Research progress in the effect of Nano-MgO on the properties of cement-based materials. China Concr. Cem. Prod. 2019, 5, 13–18. [Google Scholar]
- Chen, H. Expansive property and pore structure characterisitics of magnesium oxide slight expansive cement- fly ash binding material. J. Chin. Ceram. Soc. 2005, 33, 516–519. [Google Scholar]




| Sample Code | OP Cement | Fly Ash | Nano MgO | Water |
|---|---|---|---|---|
| Fk1 | 345 | 151 | 0 | 140 |
| Fk2 | 340 | 149 | 5 | 140 |
| Fk3 | 336 | 147 | 10 | 140 |
| Fk4 | 330 | 145 | 15 | 140 |
| Fk5 | 325 | 143 | 20 | 140 |
| Fk6 | 323 | 141 | 25 | 140 |
| Fk7 | 320 | 139 | 30 | 140 |
| Fk8 | 310 | 137 | 35 | 140 |
| Material | Chemical Compositions | ||||||
|---|---|---|---|---|---|---|---|
| CaO | MgO | Al2O3 | SiO2 | Fe2O3 | Loss | Total | |
| Magnesite tailing | 2.85 | 40.35 | 0.03 | 5.00 | 0.05 | 50.75 | 99.03 |
| Material | Chemical Compositions | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| CaO | MgO | Al2O3 | SiO2 | Fe2O3 | SO3 | Na2O | K2O | Loss | Total | |
| Cement | 63.16 | 1.43 | 4.75 | 19.47 | 3.43 | 2.68 | 0.28 | 0.62 | 3.26 | 99.08 |
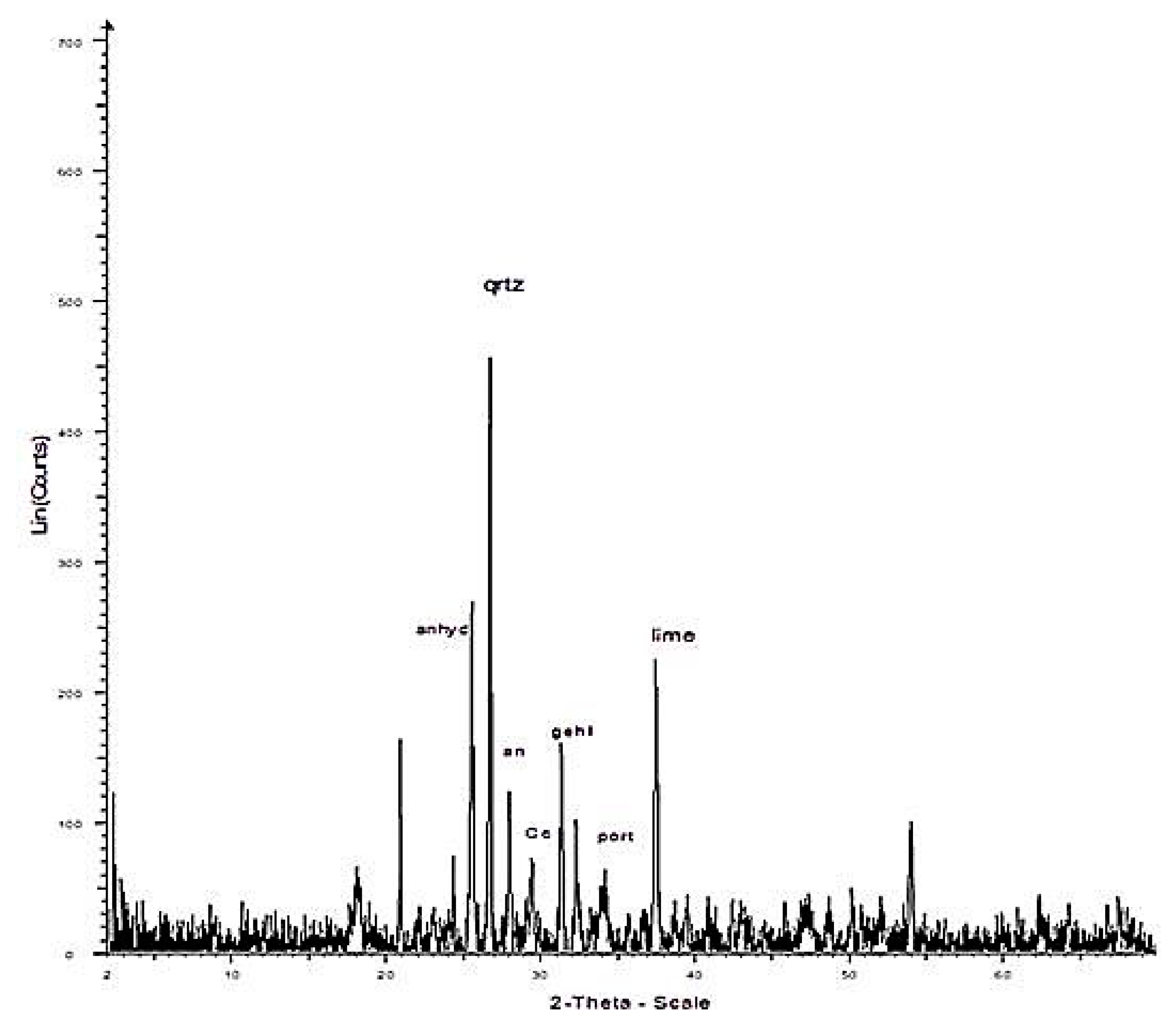
| Kardia | |
|---|---|
| Minerals | (%) |
| Quartz | 8 |
| Anhydrite | 15 |
| Lime | 30 |
| Plagioclase | 3 |
| Calcite | 14 |
| Gehlenite | 10 |
| Hematite | 2 |
| Mica | 1 |
| Clay minerals | - |
| Amorphous | 17 |
| Samples | Kardia |
|---|---|
| Oxides | |
| CaO | 53.00 |
| SiO2 | 17.00 |
| Fe2O3 | 12.05 |
| Al2O3 | 5.40 |
| SO3 | 8.00 |
| K2O | 2.02 |
| MgO | 0.60 |
| TiO2 | 1.00 |
| Cr2O3 | 0.19 |
| P2O5 | 0.26 |
| MnO | 0.14 |
| SrO | 0.09 |
| NiO | 0.09 |
| Na2O | Bdl |
| ZrO2 | 0.06 |
| CuO | 0.04 |
| ZnO | 0.03 |
| Rb2O | 0.02 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giannakopoulou, P.P.; Rogkala, A.; Lampropoulou, P.; Kalpogiannaki, M.; Petrounias, P. Evaluation of Cement Performance Using Industrial Byproducts Such as Nano MgO and Fly Ash from Greece. Appl. Sci. 2021, 11, 11601. https://doi.org/10.3390/app112411601
Giannakopoulou PP, Rogkala A, Lampropoulou P, Kalpogiannaki M, Petrounias P. Evaluation of Cement Performance Using Industrial Byproducts Such as Nano MgO and Fly Ash from Greece. Applied Sciences. 2021; 11(24):11601. https://doi.org/10.3390/app112411601
Chicago/Turabian StyleGiannakopoulou, Panagiota P., Aikaterini Rogkala, Paraskevi Lampropoulou, Maria Kalpogiannaki, and Petros Petrounias. 2021. "Evaluation of Cement Performance Using Industrial Byproducts Such as Nano MgO and Fly Ash from Greece" Applied Sciences 11, no. 24: 11601. https://doi.org/10.3390/app112411601
APA StyleGiannakopoulou, P. P., Rogkala, A., Lampropoulou, P., Kalpogiannaki, M., & Petrounias, P. (2021). Evaluation of Cement Performance Using Industrial Byproducts Such as Nano MgO and Fly Ash from Greece. Applied Sciences, 11(24), 11601. https://doi.org/10.3390/app112411601









