Comparison of the Biological Potential and Chemical Composition of Brazilian and Mexican Propolis
Abstract
1. Introduction
2. Methods
3. Discussion
3.1. Propolis Composition
3.2. Propolis Production in Mexico and Brazil
3.3. Propolis: Color, Botanical Sources, and Chemical Composition
3.4. Extraction Methods Commonly Used to Produce Propolis Extracts
3.5. Antibacterial Activity
3.6. Antifungal Activity
3.7. Antiviral Activity
3.8. Antiprotozoan Activity
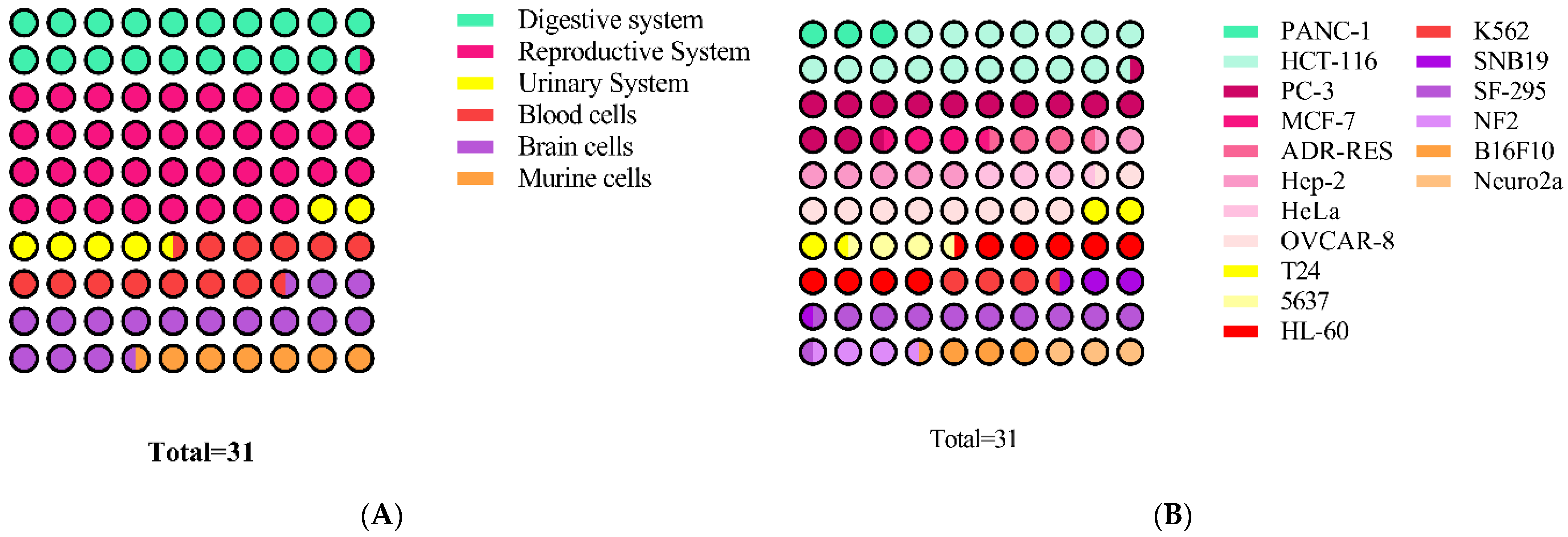
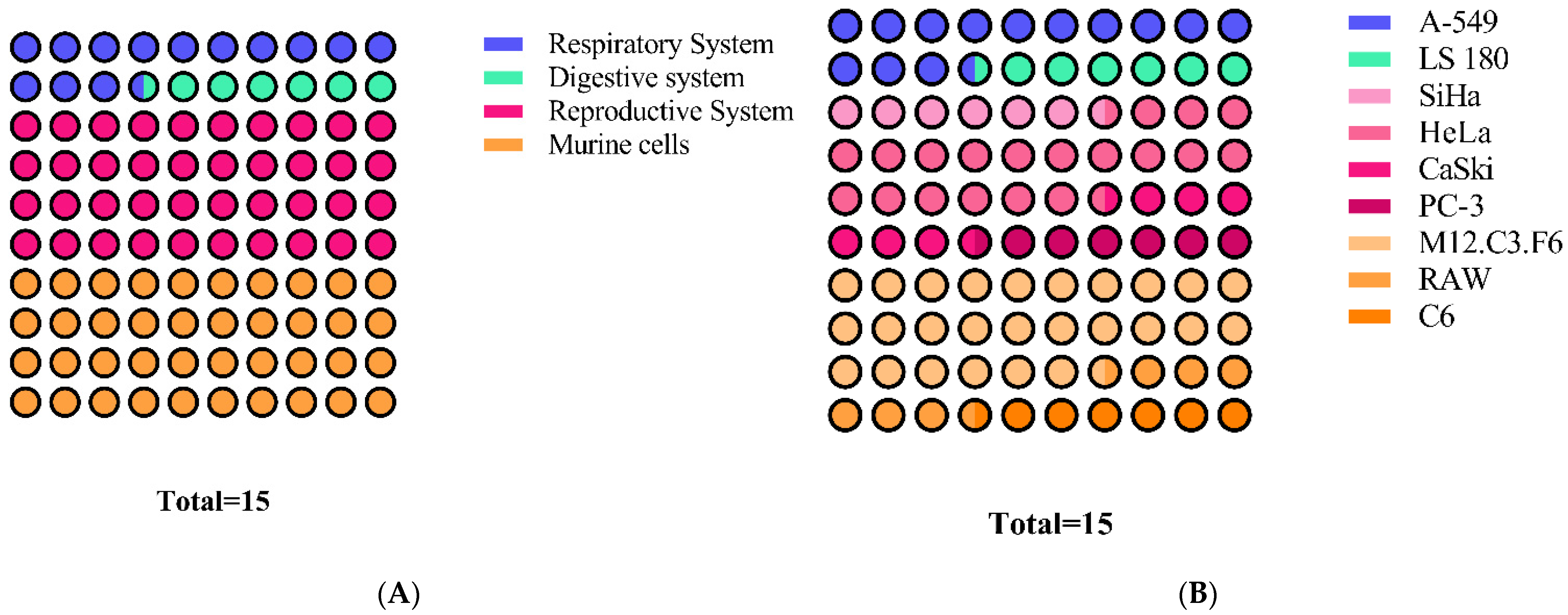
3.9. Antiproliferative and Cytotoxic Activity
3.10. Veterinary Application
4. Propolis Contamination
4.1. Xenobiotics in Propolis
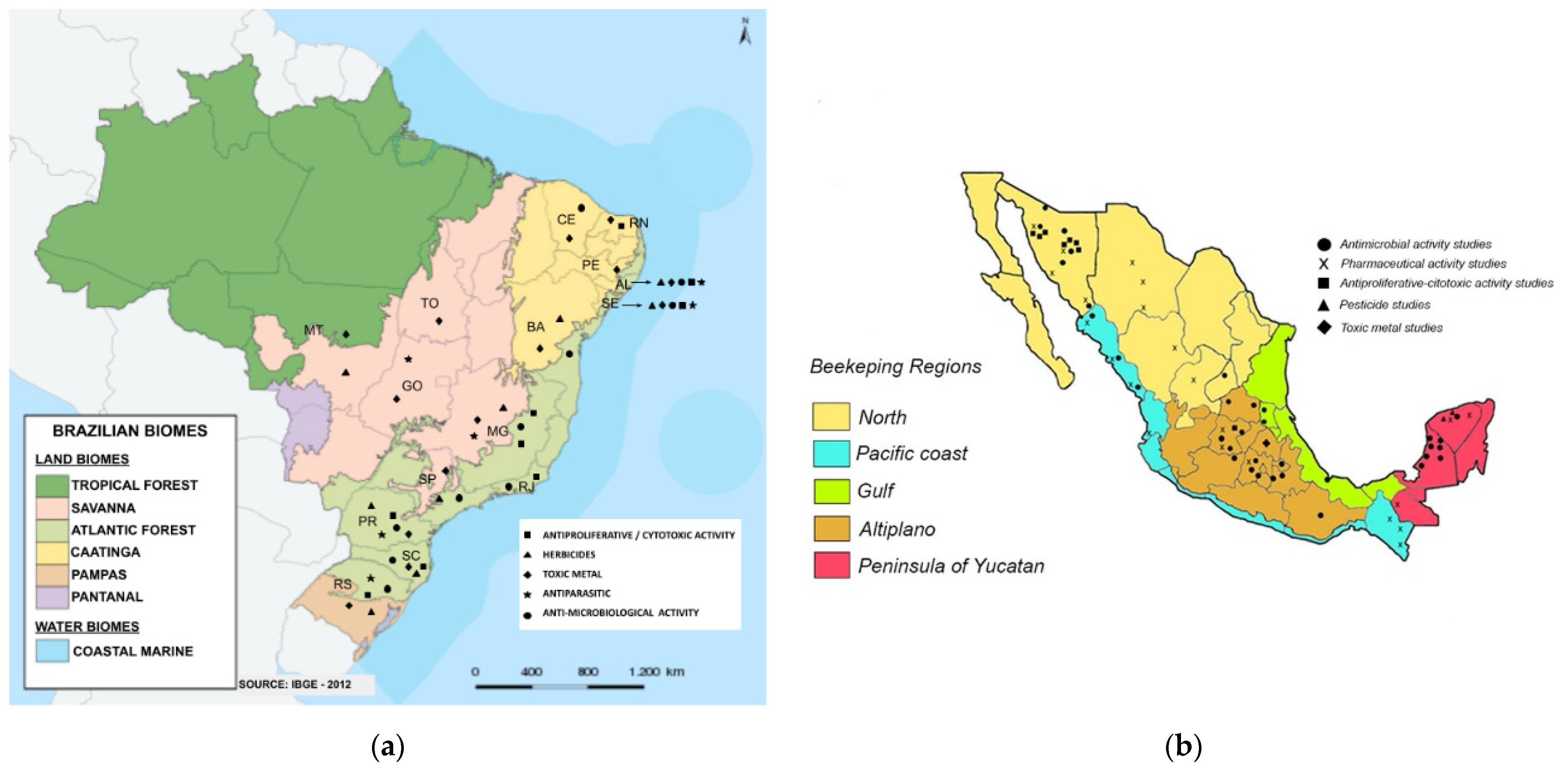
4.1.1. Pesticides in Brazilian and Mexican Propolis
4.1.2. Toxic Metals in Brazilian and Mexican Propolis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Ghisalberti, L.E. Propolis: A review. Bee World 1979, 60, 59–84. [Google Scholar] [CrossRef]
- Castaldo, S.; Capasso, F. Propolis, an old remedy used in modern medicine. Fitoterapia 2002, 73, S1–S6. [Google Scholar] [CrossRef]
- Crane, E. Bee Products; Springer: Boston, MA, USA; Tel Aviv, Israel, 1996; ISBN 9780123741448. [Google Scholar]
- Helfenberg, K.D. The analysis of beeswax and propolis. Chem. Ztg. 1908, 2, 987–988. [Google Scholar]
- Sosnowski, Z.M. Method for Extracting Propolis and Water Soluble Dry Propols Powder. U.S. Patent 4,382,886, 10 May 1983. [Google Scholar]
- Pereira, D.S.; Iberê, C.; Freitas, A.; Freitas, M.O.; Berg, J.; Agra, R. Histórico e principais usos da própolis apícola. Agropecuária CIientífica No Semi-Árido 2015, 11, 1–21. [Google Scholar]
- Quezada-Euán, J.J.G. The Past, Present, and Future of Meliponiculture in Mexico. Stingless Bees Mex. 2018, 243–269. [Google Scholar] [CrossRef]
- Norma Mexicana. Propóleos, Producción y Especificaciones para su Procesamiento. SAGARPA, NOM-003-SAG/GAN-2017. Mexico. 2017. Available online: http://www.dof.gob.mx/normasOficiales/6794/sagarpa11_C/sagarpa11_C.html (accessed on 15 August 2021).
- Machado, B.A.S.; Santos Cruz, L.; Baptista Nunes, S.; Andres Umsza Guez, M.; Ferreira Padilha, F. Estudo Prospectivo Da Própolis E Tecnologias Correlatas Sob O Enfoque Em Documentos De Patentes Depositados No Brasil. Rev. Gestão Inovação E Tecnol. 2012, 2, 221–235. [Google Scholar] [CrossRef]
- Fraga, É.E.A.; De Oliveira, C.R.; Da Cruz, C.A.B.; De Vasconcelos, C.R.; Almeida Paixão, A.E. Prospecção tecnológica: Um mapeamento de patentes da própolis vermelha. Cad. De Prospecção 2017, 10, 524. [Google Scholar] [CrossRef][Green Version]
- Araujo Neto, E.; da Morais, L.S.; da Cunha, A.F.S. Mapeamento Tecnológico: Uma prospecção de patentes e trabalhos científicos à própolis verde. Cad. De Prospecção 2019, 13, 268–279. [Google Scholar] [CrossRef]
- Trusheva, B.; Trunkova, D.; Bankova, V. Different extraction methods of biologically active components from propolis: A preliminary study. Chem. Cent. J. 2007, 1, 1–4. [Google Scholar] [CrossRef]
- Katekhaye, S.; Fearnley, H.; Fearnley, J.; Paradkar, A. Gaps in propolis research: Challenges posed to commercialization and the need for an holistic approach. J. Apic. Res. 2019, 58, 604–616. [Google Scholar] [CrossRef]
- Hodel, K.V.S.; MacHado, B.A.S.; Santos, N.R.; Costa, R.G.; Menezes-Filho, J.A.; Umsza-Guez, M.A. Metal Content of Nutritional and Toxic Value in Different Types of Brazilian Propolis. Sci. World J. 2020, 2020. [Google Scholar] [CrossRef]
- Umsza-Guez, M.A.; Silva-Beltrán, N.P.; Machado, B.A.S.; Balderrama-Carmona, A.P. Herbicide determination in Brazilian propolis using high pressure liquid chromatography. Int. J. Environ. Health Res. 2021, 31, 507–517. [Google Scholar] [CrossRef]
- Salatino, A.; Salatino, M.L.F. Scientific note: Often quoted, but not factual data about propolis composition. Apidologie 2021, 52, 312–314. [Google Scholar] [CrossRef]
- Papotti, G.; Bertelli, D.; Bortolotti, L.; Plessi, M. Chemical and Functional Characterization of Italian Propolis Obtained by Different Harvesting Methods. J. Agric. Food Chem. 2012, 60, 2852–2862. [Google Scholar] [CrossRef]
- Funari, C.S.; Ferro, V.O. Análise de própolis. Food Sci. Technol. 2006, 26, 171–178. [Google Scholar] [CrossRef]
- Falcão, S.I.; Lopes, M.; Vilas-Boas, M. A First Approach to the Chemical Composition and Antioxidant Potential of Guinea-Bissau Propolis. Nat. Prod. Commun. 2019, 14. [Google Scholar] [CrossRef]
- Diriba Jobir, M.; Belay, A. Comparative study of different Ethiopian propolis: In vivo wound healing, antioxidant, antibacterial, physicochemical properties and mineral profiles. J. Apitherapy 2020, 7. [Google Scholar] [CrossRef]
- Popova, M.; Lyoussi, B.; Aazza, S.; Antunes, D.; Bankova, V.; Miguel, G. Antioxidant and α-Glucosidase Inhibitory Properties and Chemical Profiles of Moroccan Propolis. Nat. Prod. Commun. 2015, 10, 1961–1964. [Google Scholar] [CrossRef]
- Brasil 2001. Brasil. Instrução Normativa n.3, de 19 de Janeiro de 2001. Ministério da Agricultura, Pecuária e Abastecimento Aprova os Regulamentos Técnicos de Identidade e Qualidade de Apitoxina, cera de Abelha, Geléia Real, Geléia Real Liofilizada, Pólen Apícola, Pró. 2001. Available online: https://www.apacame.org.br/mensagemdoce/115/artigo3.htm (accessed on 19 February 2021).
- Servicio de Información Agroalimentaria y Pesquera (SIAP). Alemania Demanda el 50% de las Exportaciones Mexicanas de miel. Available online: https://www.gob.mx/siap/articulos/mexico-pais-exportador-de-miel (accessed on 15 August 2021).
- Martinez, O.A.; Soares, A.E.E. Genetic improvement in the commercial beekeeping in production of propolis. Rev. Bras. De Saude E Prod. Anim. 2012, 13, 982–990. [Google Scholar] [CrossRef]
- Barros, K.B.N.T.; Neto, E.M.R.; de Fonteles, M.M.F. Propolis and its Cosmetic Applications: A Technological Prospection. J. Young Pharm. 2019, 11, 350–352. [Google Scholar] [CrossRef]
- Silva, R.P.D.; Machado, B.A.S.; Costa, S.S.; Barreto, G.A.; Padilha, F.F.; Umsza-Guez, M.A. Application of propolis extract in food products: A prospecting based in patent documents. Rev. Virtual Quim. 2016, 8, 1251–1261. [Google Scholar] [CrossRef]
- Vidal, M.D.F. Potencial da produção de própolis no nordeste. Escritório Técnico Estud. Econômicos Nordeste-ETENE 2021, 153, 1–9. [Google Scholar]
- México. Atlas Nacional de las Abejas y Derivados Apícolas, Instituto Nacional de Geografía y Estadística; Instituto Nacional de Estadística y Geografía (INEGI): Aguascalientes, Mexico, 2020. Available online: https://atlasnacionaldelasabejasmx.github.io/atlas/index.html (accessed on 15 August 2021).
- Vargas Sánchez, R.D.; Martínez Benavidez, E.; Hernández, J.; Torrescano Urrutia, G.R.; Sánchez Escalante, A. Effect of physicochemical properties and phenolic compounds of bifloral propolis on antioxidant and antimicrobial capacity. Nova Sci. 2020, 12. [Google Scholar] [CrossRef]
- Teixeira, É.W.; Message, D.; Meira, R.M.S.A.; Salantino, A. Indicadores Da Origem Botânica Da Própolis Importância E Perspectivas. B Indústr.Anim. 2003, 60, 83–106. [Google Scholar]
- Park, Y.; Masaharu, I.; Alencar, S.M. de Classification of Brazilian propolis by both physicochemical methods and biological activity. Mensagem Doce 2000, 58. [Google Scholar]
- Daugsch, A.; Moraes, C.S.; Fort, P.; Park, Y.K. Brazilian red propolis-Chemical composition and botanical origin. Evid.-Based Complementary Altern. Med. 2008, 5, 435–441. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Zhang, C.-P.; Wang, K.; Li, G.Q.; Hu, F.-L. Recent Advances in the Chemical Composition of Propolis. Molecules 2014, 19, 19610–19632. [Google Scholar] [CrossRef]
- Machado, B.A.S.; Silva, R.P.D.; Barreto, G.D.A.; Costa, S.S.; Da Silva, D.F.; Brandão, H.N.; Da Rocha, J.L.C.; Dellagostin, O.A.; Henriques, J.A.P.; Umsza-Guez, M.A.; et al. Chemical composition and biological activity of extracts obtained by supercritical extraction and ethanolic extraction of brown, green and red propolis derived from different geographic regions in Brazil. PLoS ONE 2016, 11, e0145954. [Google Scholar] [CrossRef]
- Devequi-Nunes, D.; Machado, B.A.S.; De Abreu Barreto, G.; Silva, J.R.; Da Silva, D.F.; Da Rocha, J.L.C.; Brandão, H.N.; Borges, V.M.; Umsza-Guez, M.A. Chemical characterization and biological activity of six different extracts of propolis through conventional methods and supercritical extraction. PLoS ONE 2018, 13, e0207676. [Google Scholar] [CrossRef] [PubMed]
- Rufatto, L.C.; dos Santos, D.A.; Marinho, F.; Henriques, J.A.P.; Roesch Ely, M.; Moura, S. Red propolis: Chemical composition and pharmacological activity. Asian Pac. J. Trop. Biomed. 2017, 7, 591–598. [Google Scholar] [CrossRef]
- do Nascimento, T.G.; dos Santos Arruda, R.E.; da Cruz Almeida, E.T.; dos Santos Oliveira, J.M.; Basílio-Júnior, I.D.; Celerino de Moraes Porto, I.C.; Rodrigues Sabino, A.; Tonholo, J.; Gray, A.; Ebel, R.A.E.; et al. Comprehensive multivariate correlations between climatic effect, metabolite-profile, antioxidant capacity and antibacterial activity of Brazilian red propolis metabolites during seasonal study. Sci. Rep. 2019, 9, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Coelho, J.; Falcão, S.I.; Vale, N.; Almeida-Muradian, L.B.; Vilas-Boas, M. Phenolic composition and antioxidant activity assessment of southeastern and south Brazilian propolis. J. Apic. Res. 2017, 56, 21–31. [Google Scholar] [CrossRef]
- Fausto Rivero-Cruz, J.; Rodríguez de San Miguel, E.; Robles-Obregón, S.; Hernández-Espino, C.C.; Rivero-Cruz, B.E.; Pedraza-Chaverri, J.; Esturau-Escofet, N. Prediction of antimicrobial and antioxidant activities of Mexican propolis by 1H-NMR spectroscopy and chemometrics data analysis. Molecules 2017, 22, 1184. [Google Scholar] [CrossRef]
- Rivero-Cruz, J.F.; Granados-Pineda, J.; Pedraza-Chaverri, J.; Pérez-Rojas, J.M.; Kumar-Passari, A.; Diaz-Ruiz, G.; Rivero-Cruz, B.E. Phytochemical Constituents, Antioxidant, Cytotoxic, and Antimicrobial Activities of the Ethanolic Extract of Mexican Brown Propolis. Antioxidant (Basel Switz.) 2020, 9, 70. [Google Scholar] [CrossRef] [PubMed]
- Rivera-Yañez, N.; Rodriguez-Canales, M.; Nieto-Yañez, O.; Jimenez-Estrada, M.; Ibarra-Barajas, M.; Canales-Martinez, M.M.; Rodriguez-Monroy, M.A. Hypoglycaemic and Antioxidant Effects of Propolis of Chihuahua in a Model of Experimental Diabetes. Evid.-Based Complementary Altern. Med. 2018, 2018. [Google Scholar] [CrossRef]
- Li, F.; He, Y.M.; Awale, S.; Kadota, S.; Tezuka, Y. Two new cytotoxic phenylallylflavanones from mexican propolis. Chem. Pharm. Bull. 2011, 59, 1194–1196. [Google Scholar] [CrossRef]
- Park, Y.K.; Paredes-Guzman, J.F.; Aguiar, C.L.; Alencar, S.M.; Fujiwara, F.Y. Chemical Constituents in Baccharis dracunculifolia as the Main Botanical Origin of Southeastern Brazilian Propolis. J. Agric. Food Chem. 2004, 52, 1100–1103. [Google Scholar] [CrossRef]
- Lugo, E.; Martinez-Robinson, K.; Morales Ramírez, G.; De la Rosa López, R.; Noriega Rodríguez, J.A. Determinación del Origen Botánico de los Propóleos Sonorenses (CABORCA) Mediante HPLC Determination of Botanic Origen of Sonoran Propolis (CABORCA) by HPLC analysis. Invurnus 2009, 4, 24–30. [Google Scholar]
- Riani, L.R.; Silva, L.M.; da Silva, O.O.Z.; Junqueira, L.R.; Nascimento, J.W.L.; da Filho, A.A.S. Development and validation of a uhplc-esi-ms/ms method for the quantification of artepillin c in brazilian green propolis. Rev. Cienc. Farm. Basica E Appl. 2021, 42. [Google Scholar] [CrossRef]
- Guimarães, N.S.S.; Mello, J.C.; Paiva, J.S.; Bueno, P.C.P.; Berretta, A.A.; Torquato, R.J.; Nantes, I.L.; Rodrigues, T. Baccharis dracunculifolia, the main source of green propolis, exhibits potent antioxidant activity and prevents oxidative mitochondrial damage. Food Chem. Toxicol. 2012, 50, 1091–1097. [Google Scholar] [CrossRef]
- Marcucci, M.C.; Bankova, V. Chemical composition, plant origin and biological activity of Brazilian propolis. Curr. Top. Phytochem. 1999, 2, 115–123. [Google Scholar]
- Silva, B.B.; Rosalen, P.L.; Cury, J.A.; Ikegaki, M.; Souza, V.C.; Esteves, A.; Alencar, S.M. Chemical composition and botanical origin of red propolis, a new type of Brazilian propolis. Evid.-Based Complementary Altern. Med. 2008, 5, 313–316. [Google Scholar] [CrossRef]
- de Mendonça, I.C.G.; de Porto, I.C.C.M.; do Nascimento, T.G.; de Souza, N.S.; dos Oliveira, J.M.S.; dos Arruda, R.E.S.; Mousinho, K.C.; dos Santos, A.F.; Basílio-Júnior, I.D.; Parolia, A.; et al. Brazilian red propolis: Phytochemical screening, antioxidant activity and effect against cancer cells. BMC Complementary Altern. Med. 2015, 15, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Yang, B.; Wang, D.; Zhu, Y.; Miao, X.; Yang, W. The chemical composition of brazilian green propolis and its protective effects on mouse aortic endothelial cells against inflammatory injury. Molecules 2020, 25, 4612. [Google Scholar] [CrossRef]
- Park, Y.; Alencar, S.; Aguiar, S. Botanical origin and chemical composition of Brazilian propolis. J. Agric. Food Chem. 2002, 50, 2502–2506. [Google Scholar] [CrossRef] [PubMed]
- De da Freitas, A.S.; Barth, O.M.; da Luz, C.F.P. Própolis marrom da vertente atlântica do Estado do Rio de Janeiro, Brasil: Uma avaliação palinológica. Rev. Bras. Botânica 2010, 33, 343–354. [Google Scholar] [CrossRef]
- Lotti, C.; Fernandez, M.C.; Piccinelli, A.L.; Cuesta-Rubio, O.; Hernández, I.M.; Rastrelli, L. Chemical Constituents of Red Mexican Propolis. J. Agric. Food Chem. 2010, 58, 2209–2213. [Google Scholar] [CrossRef] [PubMed]
- Boisard, S.; Huynh, T.H.T.; Escalante-Erosa, F.; Hernández-Chavez, L.I.; Peña-Rodríguez, L.M.; Richomme, P. Unusual chemical composition of a Mexican propolis collected in Quintana Roo, Mexico. J. Apic. Res. 2016, 54, 350–357. [Google Scholar] [CrossRef]
- Guzmán-Gutiérrez, S.L.; Nieto-Camacho, A.; Castillo-Arellano, J.I.; Huerta-Salazar, E.; Hernández-Pasteur, G.; Silva-Miranda, M.; ello-Nájera, O.A.; Sepúlveda-Robles, O.; Espitia, C.I.; Reyes-Chilpa, R. Mexican propolis: A source of antioxidants and anti-inflammatory compounds, and isolation of a novel chalcone and ε-caprolactone derivative. Molecules 2018, 23, 334. [Google Scholar] [CrossRef]
- Valencia, D.; Alday, E.; Robles-Zepeda, R.; Garibay-Escobar, A.; Galvez-Ruiz, J.C.; Salas-Reyes, M.; Jiménez-Estrada, M.; Velazquez-Contreras, E.; Hernandez, J.; Velazquez, C. Seasonal effect on chemical composition and biological activities of Sonoran propolis. Food Chem. 2012, 131, 645–651. [Google Scholar] [CrossRef]
- Bankova, V.; Trusheva, B.; Popova, M. Propolis extraction methods: A review. J. Apic. Res. 2021, 1–10. [Google Scholar] [CrossRef]
- Oliveira, A.; França, H.; Kuster, R.; Teixeira, L.; Rocha, L. Chemical composition and antibacterial activity of Brazilian propolis essential oil. J. Venom. Anim. Toxins 2010, 16, 121–130. [Google Scholar] [CrossRef]
- Silva, C.S.R.; Villaça, C.L.P.B.; de Peixoto, R.M.; Mota, R.A.; de Ribeiro, M.F.; da Costa, M.M. Antibacterial effect of Brazilian brown propolis in different solvents against staphylococcus spp. isolated from caprine mastitis. Cienc. Anim. Bras. 2012, 13, 247–251. [Google Scholar] [CrossRef]
- Sauri-Duch, E.; Gutiérrez-Canul, C.; Cuevas-Glory, L.F.; Ramón-Canul, L.; Pérez-Pacheco, E.; Moo-Huchin, V.M. Determination of quality characteristics, phenolic compounds and antioxidant activity of propolis from southeastern Mexico. J. Apic. Sci. 2021, 65, 109. [Google Scholar] [CrossRef]
- Machado, B.A.S.; Pereira, C.G.; Nunes, S.B.; Padilha, F.F.; Umsza-Guez, M.A. Supercritical Fluid Extraction Using CO2: Main Applications and Future Perspectives. Sep. Sci. Technol. 2013, 48, 2741–2760. [Google Scholar] [CrossRef]
- Machado, B.A.S.; De Abreu Barreto, G.; Costa, A.S.; Costa, S.S.; Silva, R.P.D.; Da Silva, D.F.; Brandao, H.N.; Da Rocha, J.L.C.; Nunes, S.B.; Umsza-Guez, M.A.; et al. Determination of parameters for the supercritical extraction of antioxidant compounds from green propolis using carbon dioxide and ethanol as co-solvent. PLoS ONE 2015, 10, e0134489. [Google Scholar] [CrossRef]
- De Oliveira Reis, J.H.; De Abreu Barreto, G.; Cerqueira, J.C.; Dos Anjos, J.P.; Andrade, L.N.; Padilha, F.F.; Druzian, J.I.; MacHado, B.A.S. Evaluation of the antioxidant profile and cytotoxic activity of red propolis extracts from different regions of northeastern Brazil obtained by conventional and ultrasoundassisted extraction. PLoS ONE 2019, 14, e0219063. [Google Scholar] [CrossRef]
- Margeretha, I.; Fatma Suniarti, D.; Herda, E.; Mas’ud, Z.A. Optimization and comparative study of different extraction methods of biologically active components of Indonesian propolis Trigona spp. J. Nat. Prod. 2012, 5, 233–242. [Google Scholar]
- Orsat, V.; Routray, W. Microwave-Assisted Extraction of Flavonoids. Water Extr. Bioact. Compd. Plants Drug Dev. 2017, 221–244. [Google Scholar] [CrossRef]
- Saito, É.; Sacoda, P.; Paviani, L.C.; Paula, J.T.; Cabral, F.A. Conventional and supercritical extraction of phenolic compounds from Brazilian red and green propolis. Sep. Sci. Technol. 2020, 1–8. [Google Scholar] [CrossRef]
- Xie, M.; Fan, D.; Zhao, Z.; Li, Z.; Li, G.; Chen, Y.; He, X.; Chen, A.; Li, J.; Lin, X.; et al. Nano-curcumin prepared via supercritical: Improved anti-bacterial, anti-oxidant and anti-cancer efficacy. Int. J. Pharm. 2015, 496, 732–740. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wu, J.Z.; Lin, Z.X.; Yuan, Q.J.; Li, Y.C.; Liang, J.L.; Zhan, J.Y.X.; Xie, Y.L.; Su, Z.R.; Liu, Y.H. Ameliorative effect of supercritical fluid extract of Chrysanthemum indicum Linnén against D-galactose induced brain and liver injury in senescent mice via suppression of oxidative stress, inflammation and apoptosis. J. Ethnopharmacol. 2019, 234, 44–56. [Google Scholar] [CrossRef] [PubMed]
- You, G.S.G.S.; Lin, S.C.; Chen, C.R.; Tsai, W.C.; Chang, C.M.J.; Huang, W.W. Supercritical carbon dioxide extraction enhances flavonoids in water-soluble propolis. J. Chin. Inst. Chem. Eng. 2002, 33, 233–241. [Google Scholar]
- Garcia-Mendoza, M.P.; Paula, J.T.; Paviani, L.C.; Cabral, F.A.; Martinez-Correa, H.A. Extracts from mango peel by-product obtained by supercritical CO2 and pressurized solvent processes. LWT-Food Sci. Technol. 2015, 62, 131–137. [Google Scholar] [CrossRef]
- Chen, C.R.; Shen, C.T.; Wu, J.J.; Yang, H.L.; Hsu, S.L.; Chang, C.M.J. Precipitation of sub-micron particles of 3,5-diprenyl-4-hydroxycinnamic acid in Brazilian propolis from supercritical carbon dioxide anti-solvent solutions. J. Supercrit. Fluids 2009, 50, 176–182. [Google Scholar] [CrossRef]
- De Zordi, N.; Cortesi, A.; Kikic, I.; Moneghini, M.; Solinas, D.; Innocenti, G.; Portolan, A.; Baratto, G.; Dall’Acqua, S. The supercritical carbon dioxide extraction of polyphenols from Propolis: A central composite design approach. J. Supercrit. Fluids 2014, 95, 491–498. [Google Scholar] [CrossRef]
- Monroy, Y.M.; Rodrigues, R.A.F.; Rodrigues, M.V.N.; Sant’Ana, A.S.; Silva, B.S.; Cabral, F.A. Brazilian green propolis extracts obtained by conventional processes and by processes at high pressure with supercritical carbon dioxide, ethanol and water. J. Supercrit. Fluids 2017, 130, 189–197. [Google Scholar] [CrossRef]
- Bastos, E.M.A.F.; Simone, M.; Jorge, D.M.; Soares, A.E.E.; Spivak, M. In vitro study of the antimicrobial activity of Brazilian propolis against Paenibacillus larvae. J. Invertebr. Pathol. 2008, 97, 273–281. [Google Scholar] [CrossRef]
- Botteon, C.E.A.; Silva, L.B.; Ccana-Ccapatinta, G.V.; Silva, T.S.; Ambrosio, S.R.; Veneziani, R.C.S.; Bastos, J.K.; Marcato, P.D. Biosynthesis and characterization of gold nanoparticles using Brazilian red propolis and evaluation of its antimicrobial and anticancer activities. Sci. Rep. 2021, 11, 1–16. [Google Scholar] [CrossRef]
- Vasconcelos, W.A.; Braga, N.M.A.; Chitarra, V.R.; Santos, V.R.; Andrade, Â.L.; Domingues, R.Z. Bioactive Glass-Green and Red Propolis Association: Antimicrobial Activity Against Oral Pathogen Bacteria. Nat. Prod. Chem. Res. 2014, 2. [Google Scholar] [CrossRef]
- Bueno-Silva, B.; Marsola, A.; Ikegaki, M.; Alencar, S.M.; Rosalen, P.L. The effect of seasons on Brazilian red propolis and its botanical source: Chemical composition and antibacterial activity. Nat. Prod. Res. 2017, 31, 1318–1324. [Google Scholar] [CrossRef]
- Kalil, M.A.; Santos, L.M.; Barral, T.D.; Rodrigues, D.M.; Pereira, N.P.; da Sá, M.C.A.; Umsza-Guez, M.A.; Machado, B.A.S.; Meyer, R.; Portela, R.W. Brazilian Green Propolis as a Therapeutic Agent for the Post-surgical Treatment of Caseous Lymphadenitis in Sheep. Front. Vet. Sci. 2019, 6. [Google Scholar] [CrossRef]
- Martins, M.L.; Monteiro, A.S.N.; Ferreira-Filho, J.C.C.; Vieira, T.I.; de Guimarães, M.B.C.T.; Farah, A.; Romanos, M.T.V.; Maia, L.C.; Cavalcanti, Y.W.; Fonseca-Gonçalves, A. Antibacterial and cytotoxic potential of a brazilian red propolis. Pesqui. Bras. Em Odontopediatria E Clin. Integr. 2019, 19, 1–9. [Google Scholar] [CrossRef]
- Orozco, A.L.; Acevedo, J.G.A.; Martinez, M.M.C.; Delgado, C.T.H.; Serrano, P.R.; Ortiz, C.M.F.; Diaz, A.D.; Carrillo, J.G.P.; Tovar, C.G.G.; Sanchez, T.A.C. Antibacterial comparative study between extracts of mexican propolis and of three plants which use apis mellifera for its production. J. Anim. Vet. Adv. 2010, 9, 1250–1254. [Google Scholar] [CrossRef]
- Carrillo, M.L.; Castillo, L.N.; Mauricio, R. Evaluación de la Actividad Antimicrobiana de Extractos de Propóleos de la Huasteca Potosina (México). Inf. Tecnol. 2011, 22, 21–28. [Google Scholar] [CrossRef]
- Enciso-Díaz, O.J.; Méndez-Gutiérrez, A.; De Jesús, L.H.; Sharma, A.; Villarreal, M.L.; Taketa, A.C. Antibacterial Activity of Bougainvillea Glabra, Eucalyptus Globulus, Gnaphalium Attenuatum, and Propolis Collected in Mexico. Pharmacol. Pharm. 2012, 03, 433–438. [Google Scholar] [CrossRef]
- Navarro-Navarro, M.; Lugo-Sepúlveda, R.E.; del García-Moraga, M.C.; de la Rosa-López, R.; Robles-Zepada, R.E.; Ruis-Bustos, E.; Veláquez-Contreras, C.A. Antibacterial and antioxidant activities of propolis methanolic extracts from Magdalena de Kino and Sonoyta, Sonora. Rev. Cienc. Biológicas Salud 2012, 14, 9–15. [Google Scholar]
- Rodríguez Pérez, B.; Canales Martínez, M.M.; Penieres Carrillo, J.G.; Cruz Sánchez, T.A. Composición química, propiedades antioxidantes y actividad antimicrobiana de propóleos mexicanos. Acta Univ. 2020, 30, 1–30. [Google Scholar] [CrossRef]
- Tolosa Cañizares, E.L. Obtención, caracterización y evaluación de la actividad antimicrobiana de extractos de propóleos de Campeche. ARS Pharm. 2002, 187–204. [Google Scholar]
- Velazquez, C.; Navarro, M.; Acosta, A.; Angulo, A.; Dominguez, Z.; Robles, R.; Robles-Zepeda, R.; Lugo, E.; Goycoolea, F.M.; Velazquez, E.F.; et al. Antibacterial and free-radical scavenging activities of Sonoran propolis. J. Appl. Microbiol. 2007, 103, 1747–1756. [Google Scholar] [CrossRef]
- Navarro-Navarro, M.; Ruiz-Bustos, P.; Valencia, D.; Robles-Zepeda, R.; Ruiz-Bustos, E.; Virués, C.; Hernandez, J.; Domínguez, Z.; Velazquez, C. Antibacterial activity of sonoran propolis and some of its constituents against clinically significant vibrio species. Foodborne Pathog. Dis. 2013, 10, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Popova, M.; Giannopoulou, E.; Skalicka-Woźniak, K.; Graikou, K.; Widelski, J.; Bankova, V.; Kalofonos, H.; Sivolapenko, G.; Gaweł-Bęben, K.; Antosiewicz, B.; et al. Characterization and Biological Evaluation of Propolis from Poland. Molecules 2017, 22, 1159. [Google Scholar] [CrossRef]
- Herrera-López, M.G.; Rubio-Hernández, E.I.; Leyte-Lugo, M.A.; Schinkovitz, A.; Richomme, P.; Calvo-Irabién, L.M.; Peña-Rodríguez, L.M. Botanical origin of triterpenoids from Yucatecan propolis. Phytochem. Lett. 2019, 29, 25–29. [Google Scholar] [CrossRef]
- Koru, O.; Toksoy, F.; Acikel, C.H.; Tunca, Y.M.; Baysallar, M.; Uskudar Guclu, A.; Akca, E.; Ozkok Tuylu, A.; Sorkun, K.; Tanyuksel, M.; et al. In vitro antimicrobial activity of propolis samples from different geographical origins against certain oral pathogens. Anaerobe 2007, 13, 140–145. [Google Scholar] [CrossRef] [PubMed]
- Vardar-Ünlü, G.; Silici, S.; Ünlü, M. Composition and in vitro antimicrobial activity of Populus buds and poplar-type propolis. World J. Microbiol. Biotechnol. 2008, 24, 1011–1017. [Google Scholar] [CrossRef]
- Kim, Y.H.; Chung, H.J. The effects of Korean propolis against foodborne pathogens and transmission electron microscopic examination. New Biotechnol. 2011, 28, 713–718. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.C.; Rodrigues, S.; Feás, X.; Estevinho, L.M. Antimicrobial activity, phenolic profile and role in the inflammation of propolis. Food Chem. Toxicol. 2012, 50, 1790–1795. [Google Scholar] [CrossRef]
- Silici, S.; Kutluca, S. Chemical composition and antibacterial activity of propolis collected by three different races of honeybees in the same region. J. Ethnopharmacol. 2005, 99, 69–73. [Google Scholar] [CrossRef]
- Mohammadzadeh, S.; Shariatpanahi, M.; Hamedi, M.; Ahmadkhaniha, R.; Samadi, N.; Ostad, S.N. Chemical composition, oral toxicity and antimicrobial activity of Iranian propolis. Food Chem. 2007, 103, 1097–1103. [Google Scholar] [CrossRef]
- Santos, L.M.; Rodrigues, D.M.; Kalil, M.A.; Azevedo, V.; Meyer, R.; Umsza-Guez, M.A.; Machado, B.A.; Seyffert, N.; Portela, R.W. Activity of Ethanolic and Supercritical Propolis Extracts in Corynebacterium pseudotuberculosis and Its Associated Biofilm. Front. Vet. Sci. 2021, 965. [Google Scholar] [CrossRef] [PubMed]
- Sawaya, A.C.H.F.; Cunha, I.B.S.; Marcucci, M.C.; De Oliveira Rodrigues, R.F.; Eberlin, M.N. Brazilian Propolis of Tetragonisca angustula and Apis mellifera 1. Apidologie 2006, 37, 398–407. [Google Scholar] [CrossRef]
- Ibitoye, O.B.; Ajiboye, T.O. Ferulic acid potentiates the antibacterial activity of quinolone-based antibiotics against Acinetobacter baumannii. Microb. Pathog. 2019, 126, 393–398. [Google Scholar] [CrossRef] [PubMed]
- Borges, A.; Ferreira, C.; Saavedra, M.J.; Simões, M. Antibacterial activity and mode of action of ferulic and gallic acids against pathogenic bacteria. Microb. Drug Resist. 2013, 19, 256–265. [Google Scholar] [CrossRef]
- Yoshimasu, Y.; Ikeda, T.; Sakai, N.; Yagi, A.; Hirayama, S.; Morinaga, Y.; Furukawa, S.; Nakao, R. Rapid bactericidal action of propolis against Porphyromonas gingivalis. J. Dent. Res. 2018, 97, 928–936. [Google Scholar] [CrossRef]
- Vasconcelos, N.G.; Croda, J.; Simionatto, S. Antibacterial mechanisms of cinnamon and its constituents: A review. Microb. Pathog. 2018, 120, 198–203. [Google Scholar] [CrossRef]
- Wu, M.; Brown, A.C. Applications of catechins in the treatment of bacterial infections. Pathogens 2021, 10, 546. [Google Scholar] [CrossRef]
- Bezerra, C.R.F.; Borges, K.R.A.; de Alves, R.N.S.; Teles, A.M.; Rodrigues, I.V.P.; da Silva, M.A.C.N.; do Nascimento, M.D.S.B.; de Bezerra, G.F.B. Highly efficient antibiofilm and antifungal activity of green propolis against Candida species in dentistry materials. PLoS ONE 2020, 15, e0228828. [Google Scholar] [CrossRef] [PubMed]
- Deegan, K.R.; Fonseca, M.S.; Oliveira, D.C.P.; Santos, L.M.; Fernandez, C.C.; Hanna, S.A.; Machado, B.A.S.; Umsza-Guez, M.A.; Meyer, R.; Portela, R.W. Susceptibility of Malassezia pachydermatis Clinical Isolates to Allopathic Antifungals and Brazilian Red, Green, and Brown Propolis Extracts. Front. Vet. Sci. 2019, 6. [Google Scholar] [CrossRef]
- Londoño Orozco, A.; Guillermo Penieres Carrillo, J.; Gerardo García Tovar, C.; Carrillo, L.M.; Leonor Quintero Mora, M.; Elvira García Vázquez, S.; Antonio Mendoza Saavedra, M.; natiuh Alejandro Cruz Sánchez, T.; Orozco, L.; Carrillo, P.; et al. Palabras clave Estudio de la actividad antifúngica de un extracto de propóleo de la abeja Apis mellifera proveniente del estado de México. Rev. Tecnol. Marcha 2008, 21, ág-49. [Google Scholar]
- Ramón-Sierra, J.; Peraza-López, E.; Rodríguez-Borges, R.; Yam-Puc, A.; Madera-Santana, T.; Ortiz-Vázquez, E. Partial characterization of ethanolic extract of Melipona beecheii propolis and in vitro evaluation of its antifungal activity. Rev. Bras. Farmacogn. 2019, 29, 319–324. [Google Scholar] [CrossRef]
- Rodríguez Pérez, B.; Penieres Carrillo, J.G.; Canales Martínez, M.M.; Luna Mora, R.A.; Cruz Sánchez, T.A. Potencialización de la actividad antifúngica de propóleos mexicanos con la adición de chalconas sintetizadas. Biotecnia 2019, 21, 76–85. [Google Scholar] [CrossRef]
- Pascoal, A.; Feás, X.; Dias, T.; Dias, L.G.; Estevinho, L.M. The Role of Honey and Propolis in the Treatment of Infected Wounds. Microbiol. Surg. Infect. Diagn. Progn. Treat. 2014, 221–234. [Google Scholar] [CrossRef]
- Kurek-Górecka, A.; Górecki, M.; Rzepecka-Stojko, A.; Balwierz, R.; Stojko, J. Bee Products in Dermatology and Skin Care. Molecules 2020, 25, 556. [Google Scholar] [CrossRef]
- González-Búrquez, M.D.J.; González-Díaz, F.R.; García-Tovar, C.G.; Carrillo-Miranda, L.; Soto-Zárate, C.I.; Canales-Martínez, M.M.; Penieres-Carrillo, J.G.; Crúz-Sánchez, T.A.; Fonseca-Coronado, S. Comparison between in Vitro Antiviral Effect of Mexican Propolis and Three Commercial Flavonoids against Canine Distemper Virus. Evid.-Based Complementary Altern. Med. 2018, 2018. [Google Scholar] [CrossRef] [PubMed]
- Amoros, M.; Simõs, C.M.O.; Girre, L.; Sauvager, F.; Cormier, M. Synergistic Effect of Flavones and Flavonols Against Herpes Simplex Virus Type 1 in Cell Culture. Comparison with the Antiviral Activity of Propolis. J. Nat. Prod. 2004, 55, 1732–1740. [Google Scholar] [CrossRef]
- Anjum, S.I.; Ullah, A.; Khan, K.A.; Attaullah, M.; Khan, H.; Ali, H.; Bashir, M.A.; Tahir, M.; Ansari, M.J.; Ghramh, H.A.; et al. Composition and functional properties of propolis (bee glue): A review. Saudi J. Biol. Sci. 2019, 26, 1695–1703. [Google Scholar] [CrossRef]
- Bachevski, D.; Damevska, K.; Simeonovski, V.; Dimova, M. Back to the basics: Propolis and COVID-19. Dermatol. Ther. 2020, 33, e13780. [Google Scholar] [CrossRef] [PubMed]
- Silva-Beltrán, N.P.; Balderrama-Carmona, A.P.; Umsza-Guez, M.A.; Souza Machado, B.A. Antiviral effects of Brazilian green and red propolis extracts on Enterovirus surrogates. Environ. Sci. Pollut. Res. 2020, 27, 28510–28517. [Google Scholar] [CrossRef]
- Silveira, M.A.D.; De Jong, D.; Berretta, A.A.; dos Galvão, E.B.S.; Ribeiro, J.C.; Cerqueira-Silva, T.; Amorim, T.C.; da Conceição, L.F.M.R.; Gomes, M.M.D.; Teixeira, M.B.; et al. Efficacy of Brazilian green propolis (EPP-AF®) as an adjunct treatment for hospitalized COVID-19 patients: A randomized, controlled clinical trial. Biomed. Pharmacother. 2021, 138, 111526. [Google Scholar] [CrossRef]
- Lobo-Galo, N.; Gálvez-Ruíz, J.C.; Balderrama-Carmona, A.P.; Silva-Beltrán, N.P.; Ruiz-Bustos, E. Recent biotechnological advances as potential intervention strategies against COVID-19. 3 Biotech 2021, 11. [Google Scholar] [CrossRef]
- Rebouças-Silva, J.; Celes, F.S.; Lima, J.B.; Barud, H.S.; De Oliveira, C.I.; Berretta, A.A.; Borges, V.M. Parasite Killing of Leishmania (V) braziliensis by Standardized Propolis Extracts. Evid.-Based Complementary Altern. Med. 2017, 2017. [Google Scholar] [CrossRef]
- Forma, E.; Bryś, M. Anticancer Activity of Propolis and Its Compounds. Nutrients 2021, 13, 2594. [Google Scholar] [CrossRef] [PubMed]
- Marcucci, M.C.; Rodriguez, J.; Ferreres, F.; Bankova, V.; Groto, R.; Popov, S. Chemical composition of Brazilian propolis from Sao Paulo State. Z. Nat.-Sect. C J. Biosci. 1998, 53, 117–119. [Google Scholar] [CrossRef]
- Marcucci, M.C.; Ferreres, F.; García-Viguera, C.; Bankova, V.S.; De Castro, S.L.; Dantas, A.P.; Valente, P.H.; Paulino, N. Phenolic compounds from Brazilian propolis with pharmacological activities. J. Ethnopharmacol. 2001, 74, 105–112. [Google Scholar] [CrossRef]
- Franchi, G.C.; Moraes, C.S.; Toreti, V.C.; Daugsch, A.; Nowill, A.E.; Park, Y.K. Comparison of effects of the ethanolic extracts of brazilian propolis on human leukemic cells as assessed with the MTT assay. Evid.-Based Complementary Altern. Med. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- de Carvalho, F.M.A.; Schneider, J.K.; de Jesus, C.V.F.; de Andrade, L.N.; Amaral, R.G.; David, J.M.; Krause, L.C.; Severino, P.; Soares, C.M.F.; Bastos, E.C.; et al. Brazilian red propolis: Extracts production, physicochemical characterization, and cytotoxicity profile for antitumor activity. Biomolecules 2020, 10, 726. [Google Scholar] [CrossRef] [PubMed]
- Dantas Silva, R.P.; Machado, B.A.S.; de Barreto, G.A.; Costa, S.S.; Andrade, L.N.; Amaral, R.G.; Carvalho, A.A.; Padilha, F.F.; Barbosa, J.D.V.; Umsza-Guez, M.A. Antioxidant, antimicrobial, antiparasitic, and cytotoxic properties of various Brazilian propolis extracts. PLoS ONE 2017, 12, e0172585. [Google Scholar] [CrossRef]
- Silva, F.R.G.; Matias, T.M.S.; Souza, L.I.O.; Matos-Rocha, T.J.; Fonseca, S.A.; Mousinho, K.C.; Santos, A.F. Phytochemical screening and in vitro antibacterial, antifungal, antioxidant and antitumor activities of the red propolis Alagoas. Braz. J. Biol. 2018, 79, 452–459. [Google Scholar] [CrossRef]
- Awale, S.; Li, F.; Onozuka, H.; Esumi, H.; Tezuka, Y.; Kadota, S. Constituents of Brazilian red propolis and their preferential cytotoxic activity against human pancreatic PANC-1 cancer cell line in nutrient-deprived condition. Bioorg. Med. Chem. 2008, 16, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Ishiai, S.; Tahara, W.; Yamamoto, E.; Yamamoto, R.; Nagai, K. Histone deacetylase inhibitory effect of Brazilian propolis and its association with the antitumor effect in Neuro2a cells. Food Sci. Nutr. 2014, 2, 565–570. [Google Scholar] [CrossRef]
- Bonamigo, T.; Campos, J.F.; Oliveira, A.S.; Torquato, H.F.V.; Balestieri, J.B.P.; Cardoso, C.A.L.; Paredes-Gamero, E.J.; de Souza, K.P.; Santos, E.L. dos Antioxidant and cytotoxic activity of propolis of Plebeia droryana and Apis mellifera (Hymenoptera, Apidae) from the Brazilian Cerrado biome. PLoS ONE 2017, 12, e0183983. [Google Scholar] [CrossRef] [PubMed]
- Messerli, S.M.; Ahn, M.-R.; Kunimasa, K.; Yanagihara, M.; Tatefuji, T.; Hashimoto, K.; Mautner, V.; Uto, Y.; Hori, H.; Kumazawa, S.; et al. Artepillin C (ARC) in Brazilian green propolis selectively blocks oncogenic PAK1 signaling and suppresses the growth of NF tumors in mice. Phytother. Res. 2009, 23, 423–427. [Google Scholar] [CrossRef] [PubMed]
- Yasukawa, K.; Yu, S.Y.; Tsutsumi, S.; Kurokawa, M.; Park, Y.K. Inhibitory effects of Brazilian propolis on tumor promotion in two-stage mouse skin carcinogenesis. J. Pharm. Nutr. Sci. 2012, 2, 71–76. [Google Scholar] [CrossRef]
- Conti, B.J.; Santiago, K.B.; Búfalo, M.C.; Herrera, Y.F.; Alday, E.; Velazquez, C.; Hernandez, J.; Sforcin, J.M. Modulatory effects of propolis samples from Latin America (Brazil, Cuba and Mexico) on cytokine production by human monocytes. J. Pharm. Pharmacol. 2015, 67, 1431–1438. [Google Scholar] [CrossRef]
- Silveira, M.A.D.; Teles, F.; Berretta, A.A.; Sanches, T.R.; Rodrigues, C.E.; Seguro, A.C.; Andrade, L. Effects of Brazilian green propolis on proteinuria and renal function in patients with chronic kidney disease: A randomized, double-blind, placebo-controlled trial. BMC Nephrol. 2019, 20, 1–12. [Google Scholar] [CrossRef]
- Begnini, K.R.; Moura De Leon, P.M.; Thurow, H.; Schultze, E.; Campos, V.F.; Martins Rodrigues, F.; Borsuk, S.; Dellagostin, O.A.; Savegnago, L.; Roesch-Ely, M.; et al. Brazilian red propolis induces apoptosis-like cell death and decreases migration potential in bladder cancer cells. Evid.-Based Complementary Altern. Med. 2014, 2014. [Google Scholar] [CrossRef]
- da Silva Frozza, C.O.; Garcia, C.S.C.; Gambato, G.; de Souza, M.D.O.; Salvador, M.; Moura, S.; Padilha, F.F.; Seixas, F.K.; Collares, T.; Borsuk, S.; et al. Chemical characterization, antioxidant and cytotoxic activities of Brazilian red propolis. Food Chem. Toxicol. 2013, 52, 137–142. [Google Scholar] [CrossRef] [PubMed]
- da Frozza, C.O.S.; Santos, D.A.; Rufatto, L.C.; Minetto, L.; Scariot, F.J.; Echeverrigaray, S.; Pich, C.T.; Moura, S.; Padilha, F.F.; Borsuk, S.; et al. Antitumor activity of Brazilian red propolis fractions against Hep-2 cancer cell line. Biomed. Pharmacother. 2017, 91, 951–963. [Google Scholar] [CrossRef]
- Costa, A.G.; Yoshida, N.C.; Garcez, W.S.; Perdomo, R.T.; de Matos, M.F.C.; Garcez, F.R. Metabolomics Approach Expands the Classification of Propolis Samples from Midwest Brazil. J. Nat. Prod. 2020, 83, 333–343. [Google Scholar] [CrossRef]
- Hernandez, J.; Goycoolea, F.M.; Quintero, J.; Acosta, A.; Castañeda, M.; Dominguez, Z.; Robles, R.; Vazquez-Moreno, L.; Velazquez, E.F.; Astiazaran, H.; et al. Sonoran Propolis: Chemical Composition and Antiproliferative Activity on Cancer Cell Lines. Planta Med. 2007, 73, 1469–1474. [Google Scholar] [CrossRef] [PubMed]
- Alday, E.; Valencia, D.; Garibay-Escobar, A.; Domínguez-Esquivel, Z.; Piccinelli, A.L.; Rastrelli, L.; Monribot-Villanueva, J.; Guerrero-Analco, J.A.; Robles-Zepeda, R.E.; Hernandez, J.; et al. Plant origin authentication of Sonoran Desert propolis: An antiproliferative propolis from a semi-arid region. Sci. Nat. 2019, 106. [Google Scholar] [CrossRef]
- Mendez-Pfeiffer, P.; Alday, E.; Carreño, A.L.; Hernández-Tánori, J.; Montaño-Leyva, B.; Ortega-García, J.; Valdez, J.; Garibay-Escobar, A.; Hernandez, J.; Valencia, D.; et al. Seasonality modulates the cellular antioxidant activity and antiproliferative effect of sonoran desert propolis. Antioxidants 2020, 9, 1294. [Google Scholar] [CrossRef]
- Li, F.; Awale, S.; Tezuka, Y.; Esumi, H.; Kadota, S. Study on the constituents of mexican propolis and their cytotoxic activity against PANC-1 human pancreatic cancer cells. J. Nat. Prod. 2010, 73, 623–627. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Awale, S.; Tezuka, Y.; Kadota, S. Cytotoxic constituents from Brazilian red propolis and their structure-activity relationship. Bioorg. Med. Chem. 2008, 16, 5434–5440. [Google Scholar] [CrossRef]
- Alday-Provencio, S.; Diaz, G.; Rascon, L.; Quintero, J.; Alday, E.; Robles-Zepeda, R.; Garibay-Escobar, A.; Astiazaran, H.; Hernandez, J.; Velazquez, C. Sonoran propolis and some of its chemical constituents inhibit in vitro growth of Giardia lamblia trophozoites. Planta Med. 2015, 81, 742–747. [Google Scholar] [CrossRef] [PubMed]
- Kimoto, T.; Arai, S.; Kohguchi, M.; Aga, M.; Nomura, Y.; Micallef, M.; Kurimoto, M.; Mito, K. Apoptosis and suppression of tumor growth by artepillin C extracted from Brazilian propolis. Cancer Detect. Prev. 1998, 22, 506–515. [Google Scholar] [CrossRef]
- Matsumoto, K.; Akao, Y.; Kobayashi, E.; Ito, T.; Ohguchi, K.; Tanaka, T.; Iinuma, M.; Nozawa, Y. Cytotoxic benzophenone derivatives from Garcinia species display a strong apoptosis-inducing effect against human leukemia cell lines. Biol. Pharm. Bull. 2003, 26, 569–571. [Google Scholar] [CrossRef]
- Chen, W.; Sun, Y.; Lu, C.; Chao, C. Thermal cycling as a novel thermal therapy to synergistically enhance the anticancer effect of propolis on PANC-1 cells. Int. J. Oncol. 2019, 55, 617–628. [Google Scholar] [CrossRef]
- Santos, L.M.; Fonseca, M.S.; Sokolonski, A.R.; Deegan, K.R.; Araújo, R.P.C.; Umsza-Guez, M.A.; Barbosa, J.D.V.; Portela, R.D.; Machado, B.A.S. Propolis: Types, composition, biological activities, and veterinary product patent prospecting. J. Sci. Food Agric. 2020, 100, 1369–1382. [Google Scholar] [CrossRef]
- Estrada, S.T.A.; López, G.P.A.; Autran, Z.C.I.; Pérez, M.M.; Londoño, V. V Use of Propolis for Topical Treatment of Dermatophytosis in Dog. Open J. Vet. Med. 2014, 4, 239–245. [Google Scholar] [CrossRef]
- del Rodríguez, I.S.F.; Monteagudo, M.M.; Orozco, A.L.; Sánchez, T.A.C. Use of Mexican Propolis for the Topical Treatment of Dermatomycosis in Horses. Open J. Vet. Med. 2016, 06, 1–8. [Google Scholar] [CrossRef]
- Aguilar-Ayala, F.J.; Rejón-Peraza, M.E.; Cauich-Rodríguez, J.V.; Borges-Argáez, R.; Pinzón-Te, A.L.; González-Alam, C.J.; Aguilar-Perez, F.J. Biophysicochemical study of propolis and its clinical and radiographic assessment in dental pulpectomy. Drug Invent. Today 2019, 12, 2928–2933. [Google Scholar]
- Walkwork-Baber, M.; Ferenbaugh, R.; Glandney, E. The use of honey bees as monitor of environment pollution. Am. Bee J. 1982, 112, 770–772. [Google Scholar]
- Celli, G.; Maccagnani, B. Honey bees as bioindicators of environmental pollution. Bull. Insectology 2003, 56, 137–139. [Google Scholar] [CrossRef]
- Borg, D.; Attard, E. Honeybees and theis products as bioindicators for heavy metal pollution in Malta. Acta Bras. 2020, 4, 60–69. [Google Scholar] [CrossRef]
- Johnson, R.M. Honey bee toxicology. Annu. Rev. Entomol. 2015, 60, 415–434. [Google Scholar] [CrossRef]
- Mizrahi, A.; Lensky, Y. Bee Products; Springer: Berlin/Heidelberg, Germany, 1997. [Google Scholar]
- Navntoft, S.; Strandberg, B.; Nimgaard, R.; Esbjerg, P.; Axelsen, J. Effects of Herbicide-Free Field Margins on Bumblebee and Butterfly Diversity in and along Hedgerows; Environmental Protection Agency: Copenhagen, Denmark, 2011. [Google Scholar]
- Moreira, J.C.; Peres, F.; Simões, A.C.; Pignati, W.A.; de Dores, E.C.; Vieira, S.N.; Strüssmann, C.; Mott, T. Contaminação de águas superficiais e de chuva por agrotóxicos em uma região do estado do Mato Grosso. Ciência Saúde Coletiva 2012, 17, 1557–1568. [Google Scholar] [CrossRef]
- Kogan, W.R.; Kogam, M.; Parada, A.M. Phytotoxic activity of root absorbed glyphosate in corn seedlings (Zea mays L.). Weed Biol. Manag. 2003, 3, 228–232. [Google Scholar] [CrossRef]
- Nandula, V.K.; Vencill, W.K. Herbicide Absorption and Translocation in Plants using Radioisotopes. Weed Sci. 2015, 63, 140–151. [Google Scholar] [CrossRef]
- Brian, P.W. How Herbicides Work; Alberta Agriculture and Rural Development: Edmonton, AB, Canada, 1999; Volume 246, ISBN 0773261311. [Google Scholar]
- Medina-Dzul, K.; Muñoz-Rodríguez, D.; Moguel-Ordoñez, Y.; Carrera-Figueiras, C. Application of mixed solvents for elution of organophosphate pesticides extracted from raw propolis by matrix solid-phase dispersion and analysis by GC-MS. Chem. Pap. 2014, 68, 1474–1481. [Google Scholar] [CrossRef]
- González-Martín, M.I.; Revilla, I.; Vivar-Quintana, A.M.; Betances Salcedo, E.V. Pesticide residues in propolis from Spain and Chile. An approach using near infrared spectroscopy. Talanta 2017, 165, 533–539. [Google Scholar] [CrossRef] [PubMed]
- González-Martín, M.I.; Revilla, I.; Betances-Salcedo, E.V.; Vivar-Quintana, A.M. Pesticide residues and heavy metals in commercially processed propolis. Microchem. J. 2018, 143, 423–429. [Google Scholar] [CrossRef]
- Gérez, N.; Pérez-Parada, A.; Cesio, M.V.; Heinzen, H. Occurrence of pesticide residues in candies containing bee products. Food Control 2017, 72, 293–299. [Google Scholar] [CrossRef]
- Acosta-Tejada, G.M.; Medina-Peralta, S.; Moguel-Ordóñez, Y.B.; Muñoz-Rodríguez, D. Matrix solid-phase dispersion extraction of organophosphoruspesticides from propolis extracts and recovery evaluationby GCM. Anal. Bioanal. Chem. 2011, 400, 885–891. [Google Scholar] [CrossRef]
- de Orsi, R.O.; Barreto, L.M.R.C.; Gomes, S.M.A.; Kadri, S.M. Pesticidas na própolis do Estado de São Paulo, Brasil. Acta Sci.-Anim. Sci. 2012, 34, 433–436. [Google Scholar] [CrossRef][Green Version]
- Valdovinos-Flores, C.; Alcantar-Rosales, V.M.; Gaspar-Ramírez, O.; Saldaña-Loza, L.M.; Dorantes-Ugalde, J.A. Agricultural pesticide residues in honey and wax combs from Southeastern, Central and Northeastern Mexico. J. Apic. Res. 2017, 56, 667–679. [Google Scholar] [CrossRef]
- Tette, P.A.S.; Da Silva Oliveira, F.A.; Pereira, E.N.C.; Silva, G.; De Abreu Glória, M.B.; Fernandes, C. Multiclass method for pesticides quantification in honey by means of modified QuEChERS and UHPLC-MS/MS. Food Chem. 2016, 211, 130–139. [Google Scholar] [CrossRef]
- Orso, D.; Floriano, L.; Ribeiro, L.C.; Bandeira, N.M.G.; Prestes, O.D.; Zanella, R. Simultaneous Determination of Multiclass Pesticides and Antibiotics in Honey Samples Based on Ultra-High Performance Liquid Chromatography-Tandem Mass Spectrometry. Food Anal. Methods 2016, 9, 1638–1653. [Google Scholar] [CrossRef]
- Woodcock, B.A.; Isaac, N.J.B.; Bullock, J.M.; Roy, D.B.; Garthwaite, D.G.; Crowe, A.; Pywell, R.F. Impacts of neonicotinoid use on long-term population changes in wild bees in England. Nat. Commun. 2016. [Google Scholar] [CrossRef]
- Sánchez-Bayo, F.; Wyckhuys, K.A.G. Worldwide decline of the entomofauna: A review of its drivers. Biol. Conserv. 2019, 232, 8–27. [Google Scholar] [CrossRef]
- Grab, H.; Branstetter, M.G.; Amon, N.; Urban-Mead, K.R.; Park, M.G.; Gibbs, J.; Blitzer, E.J.; Poveda, K.; Loeb, G.; Danforth, B.N. Agriculturally dominated landscapes reduce bee phylogenetic diversity and pollination services. Science 2019, 363, 282–284. [Google Scholar] [CrossRef] [PubMed]
- Martinello, M.; Manzinello, C.; Borin, A.; Avram, L.E.; Dainese, N.; Giuliato, I.; Gallina, A.; Mutinelli, F. A survey from 2015 to 2019 to investigate the occurrence of pesticide residues in dead honeybees and other matrices related to honeybee mortality incidents in Italy. Diversity 2020, 12, 15. [Google Scholar] [CrossRef]
- Banks, J.E.; Banks, H.T.; Myers, N.; Laubmeier, A.N.; Bommarco, R. Lethal and sublethal effects of toxicants on bumble bee populations: A modelling approach. Ecotoxicology 2020, 29, 237–245. [Google Scholar] [CrossRef]
- Naiara Gomes, I.; Ingred Castelan Vieira, K.; Moreira Gontijo, L.; Canto Resende, H. Honeybee survival and flight capacity are compromised by insecticides used for controlling melon pests in Brazil. Ecotoxicology 2020, 29, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Tomé, H.V.V.; Schmehl, D.R.; Wedde, A.E.; Godoy, R.S.M.; Ravaiano, S.V.; Guedes, R.N.C.; Martins, G.F.; Ellis, J.D. Frequently encountered pesticides can cause multiple disorders in developing worker honey bees. Environ. Pollut. 2020, 256. [Google Scholar] [CrossRef]
- Sgolastra, F.; Medrzycki, P.; Bortolotti, L.; Maini, S.; Porrini, C.; Simon-Delso, N.; Bosch, J. Bees and pesticide regulation: Lessons from the neonicotinoid experience. Biol. Conserv. 2020, 241. [Google Scholar] [CrossRef]
- Orsi, R.O.; Barros, D.C.B.; Silva, R.C.M.; Queiroz, J.V.; Araújo, W.L.P.; Shinohara, A.J. Toxic metals in the crude propolis and its transfer rate to the ethanolic extract. Sociobiology 2018, 65, 640–644. [Google Scholar] [CrossRef]
- Schmidt, E.M.; Stock, D.; Chada, F.J.; Finger, D.; Christine Helena Frankland Sawaya, A.; Eberlin, M.N.; Felsner, M.L.; Quináia, S.P.; Monteiro, M.C.; Torres, Y.R. A comparison between characterization and biological properties of brazilian fresh and aged propolis. BioMed Res. Int. 2014, 2014. [Google Scholar] [CrossRef]
- Finger, D.; Filho, I.K.; Torres, Y.R.; Quináia, S.P. Propolis as an indicator of environmental contamination by metals. Bull. Environ. Contam. Toxicol. 2014, 92, 259–264. [Google Scholar] [CrossRef]
- Divan, A.M.; de Oliveira, P.L.; Perry, C.T.; Atz, V.L.; Azzarini-Rostirola, L.N.; Raya-Rodriguez, M.T. Using wild plant species as indicators for the accumulation of emissions from a thermal power plant, Candiota, South Brazil. Ecol. Indic. 2009, 9, 1156–1162. [Google Scholar] [CrossRef]
- Zoffoli, H.J.O.; do Amaral-Sobrinho, N.M.B.; Zonta, E.; Luisi, M.V.; Marcon, G.; Tolón-Becerra, A. Inputs of heavy metals due to agrochemical use in tobacco fields in Brazil’s Southern Region. Environ. Monit. Assess. 2012, 185, 2423–2437. [Google Scholar] [CrossRef]
- Sattler, J.A.G.; Melo, A.A.M.; do Nacimento, K.S.; de Melo, I.L.P.; Mancini-Filho, J.; Sattler, A.; de Almeida-Muradian, L.B. Essential minerals and inorganic contaminants (barium, cadmium, lithium, lead and vanadium) in dried bee pollen produced in Rio Grande do Sul State, Brazil. Food Sci. Technol. 2016, 36, 505–509. [Google Scholar] [CrossRef]
- Pierini, G.D.; Pistonesi, M.F.; Di Nezio, M.S.; Centurión, M.E. A pencil-lead bismuth film electrode and chemometric tools for simultaneous determination of heavy metals in propolis samples. Microchem. J. 2016, 125, 266–272. [Google Scholar] [CrossRef]
- Brasil RESOLUÇÃO-RDC No 42, DE 29 DE AGOSTO DE. 2013. Available online: https://bvsms.saude.gov.br/bvs/saudelegis/anvisa/2013/rdc0042_29_08_2013.html (accessed on 15 August 2021).
- Montiel, J.; Marmolejo, Y.; Castellanos, I.; Pérez, F.; Prieto, F.; Gaytán, J.; Fonseca, M. Niveles de cadmio, cromo y plomo en abejas (Apis mellifera) y sus productos en Hidalgo, México. Rev. Iberoam. Cienc. 2020, 7, 57–58. [Google Scholar]
- Cuba 1994. Norma Cubana (1994) Propóleos Materia Prima. Especificaciones, NRAG-1135-94. Ministerio de Agricultura. 1994. Available online: https://www.minag.gob.cu/ (accessed on 9 October 2020).
- El Salvador. Norma Salvadoreña (2003) Calidad del Propólero Crudo, Norma NSO 65.19.02:03. Diario Oficial.Tomo 360. San Salvador. 2003. Available online: https://www.diariooficial.gob.sv/ (accessed on 28 October 2020).
- Argentina 2008. Instituto Argentino de Normalización y Certificación (2008) Productos del NOA (Noroeste Argentino). Propóleos. Parte 1-Propóleos en Bruto. Norma IRAM-INTA 15935-1. Available online: https://catalogo.iram.org.ar/#/normas/detalles/9314 (accessed on 19 February 2021).
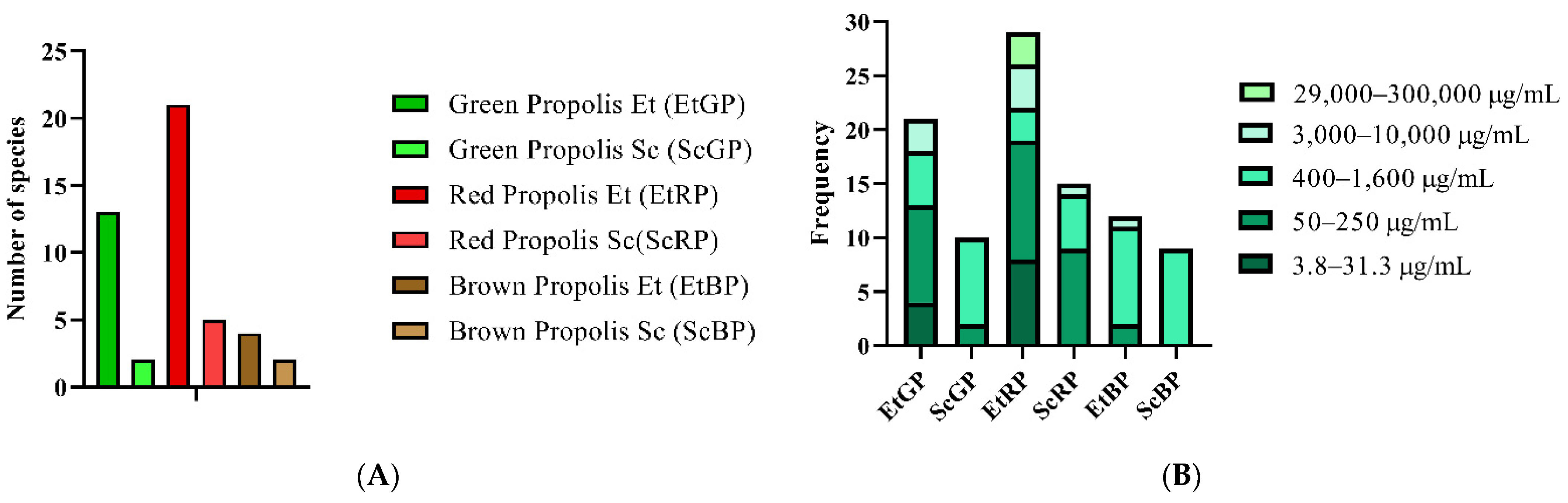
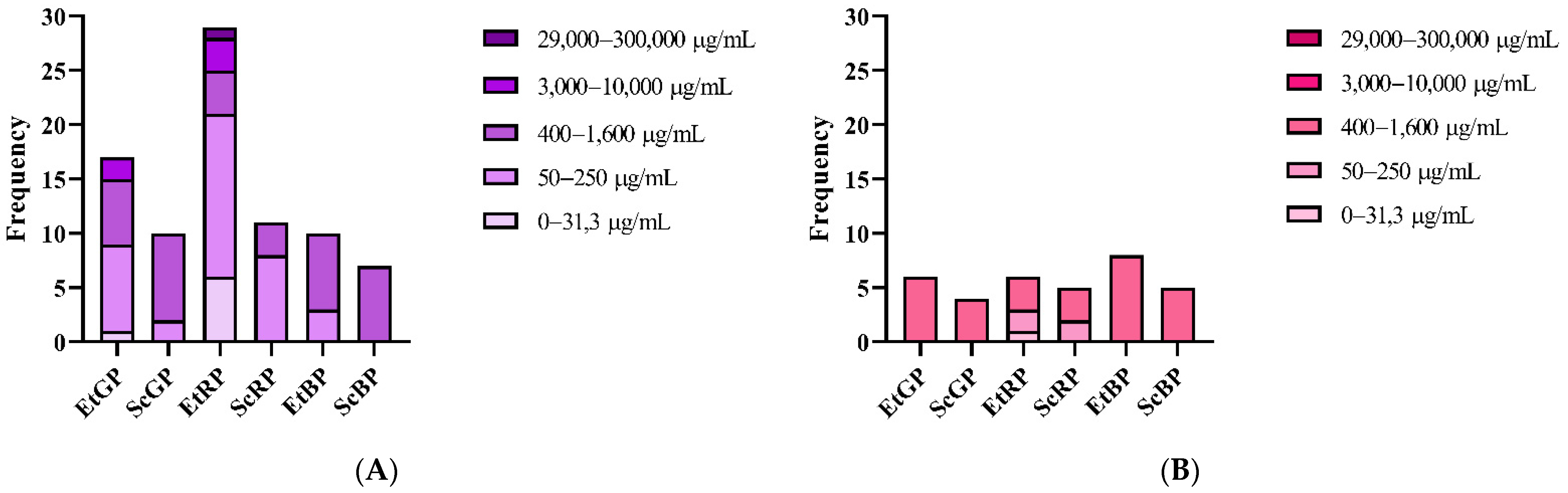

| Distribution of Brazilian Propolis Minimal Inhibitory Concentrations | |||||
| Type of Propolis Extract | Concentration (µg/mL) | ||||
| 0–31.3 | 50–250 | 400–1600 | 3000–10,000 | 29,000–300,000 | |
| Ethanolic Extract of Green Propolis (EtGP) | 4 | 9 | 5 | 3 | 0 |
| Supercritical Extract of Green Propolis (ScGP) | 0 | 2 | 8 | 0 | 0 |
| Ethanolic Extract of Red Propolis (EtRP) | 8 | 11 | 3 | 4 | 3 |
| Supercritical Extract of Red Propolis (ScRP) | 0 | 9 | 5 | 1 | 0 |
| Ethanolic Extract of Brown Propolis (EtBP) | 0 | 2 | 9 | 1 | 0 |
| Supercritical Extract of Brown Propolis (ScBP) | 0 | 0 | 9 | 0 | 0 |
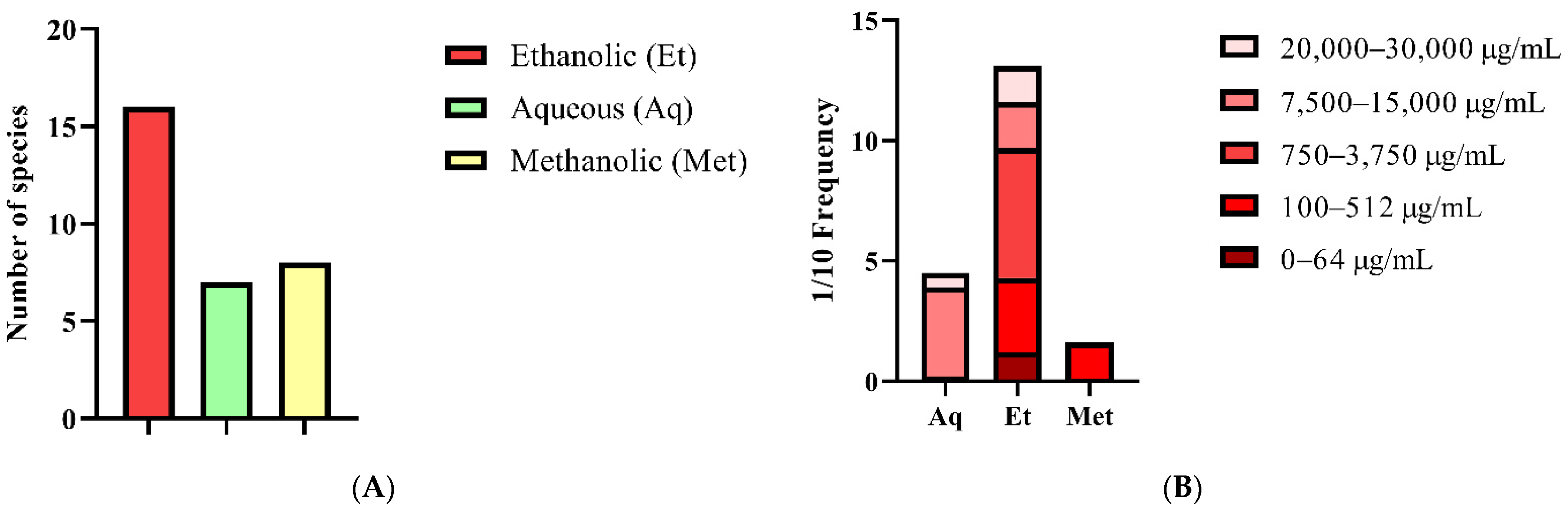
| Distribution of Mexican Propolis Minimal Inhibitory Concentrations | |||||
| 0–64 | 100–512 | 750–3750 | 7500–15,000 | 20,000–30,000 | |
| Aqueous Extract (Aq) | 0 | 0 | 20 | 37 | 60 |
| Ethanolic Extract (Et) | 12 | 31 | 54 | 19 | 15 |
| Methanolic Extract (Met) | 10 | 15 | 0 | 0 | 0 |
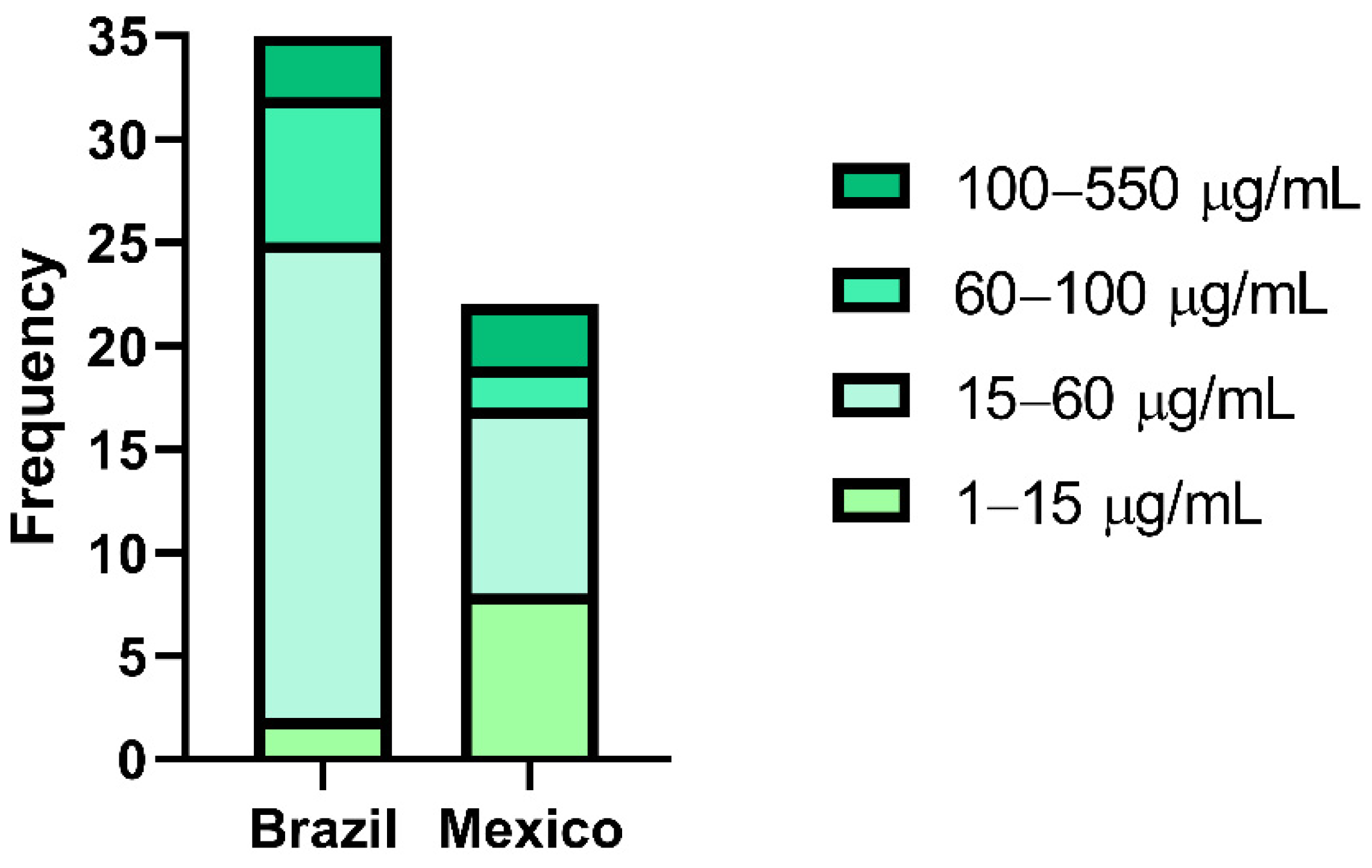
| Distribution of Brazilian and Mexican Propolis Inhibitory Concentration (IC50) | |||||
| 1–15 | 15–60 | 60–100 | 100–550 | ||
| Brazilian Propolis | 2 | 23 | 7 | 3 | |
| Mexican Propolis | 8 | 9 | 2 | 3 | |
| Microorganism | Color | Region of the Country | Extraction | MIC (μg/mL) | Reference |
|---|---|---|---|---|---|
| Fungi Mexico | |||||
| Candida albicans | ND | South | Ethanolic | 32 | [106] |
| ND | Southeast | Ethanolic | 1.6–2.30 | [107] | |
| Brown | Southeast | Ethanolic | 750 | ||
| Center | Ethanolic | 750–2400 | [85] | ||
| Cryptococcus neoformans | ND | South | Ethanolic | 32 | [106] |
| Candida albicans (ATCC 14065) Aspergillus flavus | Green, Brow, Yellow | Southeast | Ethanolic | 1.6–2.30 | [108] |
| Aspergillus fumigatus | ND | Center | Ethanolic | 32 | [106] |
| Fungi Brazil | |||||
| Candida albicans | Red | Northeast | Ethanolic/Sc-CO2 | ≥1000 | [26] |
| Red | Northeast | Ethanolic/Sc-CO2 | 1.56 | [76] | |
| Brown | Northeast | Ethanolic/Sc-CO2 | ≥1000 | [105] | |
| Malassezia pachydermatis | Red | Northeast | Ethanolic/Sc-CO2 | 4000–8000 | [105] |
| Green | Southeast | Ethanolic | 4000–8000 | ||
| Browns | South | Ethanolic | ≥16,000 | ||
| Veterinary Diseases | Microorganisms | Product with Propolis Extract | Author |
|---|---|---|---|
| Effect canine dermatophytosis effect in dogs | Microsporum gypseum Microsporum canis | Soap | [147] |
| Treatment of Dermatomycosis in Horses | Trichophyton mentagrophytes Candida albicans | Shampoo | [148,149] |
| Post-surgical Treatment of Caseous Lymphadenitis in Sheep | Corynebacterium pseudotuberculosis | Ointment | [79] |
| Mexican Propolis | |||||
|---|---|---|---|---|---|
| Number of Samples | Geographical Origin | Elements Analyzed | Toxic Elements Found | Analytical Methods | References |
| N/E | South | Dichlorvos, Diazinon, Methyl parathion, Malathion, and Coumaphos | No found | GC/MS-SIM | [164] |
| 4 | South | Dichlorvos, Diazinon, Methyl parathion, Malathion, and Coumaphos | Organophosphate pesticides (≤025 μg/mL) | GC-MS | [160] |
| Brazilian Propolis | |||||
| 19 | Northeast (Ba, Se, Al); South (SC, Pr). Southeast (MG) and Centersouth (MT) | AMPA Atrazine Glyphosate Picloram | AMPA (10.2 ± 1.39–11.3 ± 2.65) Atrazine (9.7 ± 0.11–17.4 ± 2.60) | HPLC | [15] |
| 50 | South (SP) | Organochlorines, Organophosphates, Pyrethroids, Carbamates, Herbicides, Fungicides, and Acaricides any pesticide | ND | (GC/ECD), (GC/NPD) | [165] |
| Number of Samples | Toxic Elements Found | Analytical Methods | References |
|---|---|---|---|
| 19 | Pb, Cd, As, Cu | GFAAS and FAAS | [14] |
| 106 | Hg, Cd, Pb, Sn | FAAS | [177] |
| 6 | Pb, Cd, Cr | FAAS | [178] |
| 42 | Cd, Cr, Pb | FAAS | [179] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silva-Beltrán, N.P.; Umsza-Guez, M.A.; Ramos Rodrigues, D.M.; Gálvez-Ruiz, J.C.; de Paula Castro, T.L.; Balderrama-Carmona, A.P. Comparison of the Biological Potential and Chemical Composition of Brazilian and Mexican Propolis. Appl. Sci. 2021, 11, 11417. https://doi.org/10.3390/app112311417
Silva-Beltrán NP, Umsza-Guez MA, Ramos Rodrigues DM, Gálvez-Ruiz JC, de Paula Castro TL, Balderrama-Carmona AP. Comparison of the Biological Potential and Chemical Composition of Brazilian and Mexican Propolis. Applied Sciences. 2021; 11(23):11417. https://doi.org/10.3390/app112311417
Chicago/Turabian StyleSilva-Beltrán, Norma Patricia, Marcelo Andrés Umsza-Guez, Daniela Méria Ramos Rodrigues, Juan Carlos Gálvez-Ruiz, Thiago Luiz de Paula Castro, and Ana Paola Balderrama-Carmona. 2021. "Comparison of the Biological Potential and Chemical Composition of Brazilian and Mexican Propolis" Applied Sciences 11, no. 23: 11417. https://doi.org/10.3390/app112311417
APA StyleSilva-Beltrán, N. P., Umsza-Guez, M. A., Ramos Rodrigues, D. M., Gálvez-Ruiz, J. C., de Paula Castro, T. L., & Balderrama-Carmona, A. P. (2021). Comparison of the Biological Potential and Chemical Composition of Brazilian and Mexican Propolis. Applied Sciences, 11(23), 11417. https://doi.org/10.3390/app112311417








