Bioactive Compounds and Nanodelivery Perspectives for Treatment of Cardiovascular Diseases
Abstract
:1. Introduction
2. Plant Bioactive Compounds
3. Types of Bioactive Compounds
3.1. Flavonoids
3.2. Anthocyanins
3.3. Tannins
3.4. Betalains
3.5. Carotenoids
3.6. Plant Sterols
3.7. Glucosinolates
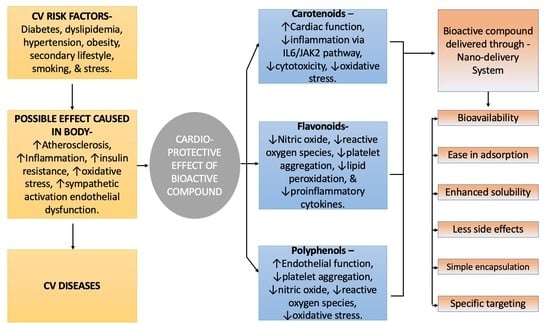
4. Therapeutic Effect of Bioactive Compounds on Cardiovascular System
4.1. Carotenoids
4.2. Polyphenols (Anthocyanins)
4.3. Lycopene
4.4. Flavonoids
Effect on CV System
5. Nano-Delivery Systems of Bioactive Compounds
5.1. Clinical Trials
5.2. Nanoparticles for Theranostic
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Martínez-Augustin, O.; Aguilera, C.M.; Gil-Campos, M.; Sánchez de Medina, F.; Gil, A. Bioactive anti-obesity food components. Int. J. Vitam. Nutr. Res. 2012, 82, 148–156. [Google Scholar] [CrossRef] [PubMed]
- NIH National Cancer Institute. What Is Cancer? Available online: https://www.cancer.gov/about-cancer/understanding/what-is-cancer (accessed on 5 May 2021).
- Felman, A.; Kohli, P. What to know about cardiovascular disease? Med. News Today 2019. Available online: https://www.medicalnewstoday.com/articles/257484 (accessed on 26 July 2019).
- Hemler, E.C.; Hu, F.B. Plant-Based Diets for Cardiovascular Disease Prevention: All Plant Foods Are Not Created Equal. Curr. Atheroscler. Rep. 2019, 21, 18. [Google Scholar] [CrossRef] [PubMed]
- Alwan, A.; World Health Organization. Global Status Report on Noncommunicable Diseases 2010; World Health Organization: Geneva, Switzerland, 2011; Volume 9, p. 162. Available online: https://www.who.int/nmh/publications/ncd_report_full_en.pdf (accessed on 5 May 2021).
- Gensini, G.F.; Comeglio, M.; Colella, A. Classical risk factors and emerging elements in the risk profile for coronary artery disease. Eur. Heart J. 1998, 19 (Suppl. A), A53–A61. [Google Scholar]
- Mahmood, S.S.; Levy, D.; Vasan, R.S.; Wang, T.J. The Framingham Heart Study and the epidemiology of cardiovascular disease: A historical perspective. Lancet 2014, 383, 999–1008. [Google Scholar] [CrossRef] [Green Version]
- Kris-Etherton, P.M.; Lefevre, M.; Beecher, G.R.; Gross, M.D.; Keen, C.L.; Etherton, T.D. Bioactive compounds in nutrition and health-research methodologies for establishing biological function: The antioxidant and anti-inflammatory effects of flavonoids on atherosclerosis. Annu. Rev. Nutr. 2004, 24, 511–538. [Google Scholar] [CrossRef]
- GBD 2013 Mortality and Causes of Death Collaborators. Global, regional, and national age–sex specific all-cause and cause-specific mortality for 240 causes of death, 1990–2013: A systematic analysis for the Global Burden of Disease Study 2013. Lancet 2015, 385, 117–171. [Google Scholar] [CrossRef]
- Bowen, K.J.; Sullivan, V.K.; Kris-Etherton, P.M.; Petersen, K.S. Nutrition and Cardiovascular Disease-an Update. Curr. Atheroscler. Rep. 2018, 20, 8. [Google Scholar] [CrossRef] [PubMed]
- Satija, A.; Hu, F.B. Plant-based diets and cardiovascular health. Trends Cardiovasc. Med. 2018, 28, 437–441. [Google Scholar] [CrossRef] [PubMed]
- Guaadaoui, A.; Benaicha, S.; Elmajdoub, N.; Bellaoui, M.; Hamal, A. What is a Bioactive Compound? A Combined Definition for a Preliminary Consensus. Int. J. Nutr. Food Sci. 2014, 3, 174–179. [Google Scholar] [CrossRef]
- González, S. Dietary Bioactive Compounds and Human Health and Disease. Nutrients 2020, 12, 348. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- NCI Dictionary of Cancer Terms. Bioactive Compounds. Available online: https://www.cancer.gov/publications/dictionaries/cancer-terms/def/bioactive-compound (accessed on 24 February 2021).
- Middleton, E., Jr.; Kandaswami, C.; Theoharides, T.C. The effects of plant flavonoids on mammalian cells: Implications for inflammation, heart disease, and cancer. Pharmacol. Rev. 2000, 52, 673–751. [Google Scholar] [PubMed]
- Conti, M.V.; Guzzetti, L.; Panzeri, D.; De Giuseppe, R.; Coccetti, P.; Labra, M.; Cena, H. Bioactive compounds in legumes: Implications for sustainable nutrition and health in the elderly population. Trends Food Sci. Technol. 2021. [Google Scholar] [CrossRef]
- Bazzano, L.A.; He, J.; Ogden, L.G.; Loria, C.M.; Vupputuri, S.; Myers, L.; Whelton, P.K. Fruit and vegetable intake and risk of cardiovascular disease in US adults: The first National Health and Nutrition Examination Survey Epidemiologic Follow-up Study. Am. J. Clin. Nutr. 2002, 76, 93–99. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, F.B. Dietary pattern analysis: A new direction in nutritional epidemiology. Curr. Opin. Lipidol. 2002, 13, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Trumbo, P.; Schlicker, S.; Yates, A.A.; Poos, M. Food and Nutrition Board of the Institute of Medicine, The National Academies. Dietary reference intakes for energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein and amino acids. J. Am. Diet. Assoc. 2002, 102, 1621–1630. [Google Scholar] [CrossRef]
- Jimeno, J.; Faircloth, G.; Sousa-Faro, J.M.F.; Scheuer, P.; Rinehart, K. New Marine Derived Anticancer Therapeutics—A Journey from the Sea to Clinical Trials. Mar. Drugs. 2004, 2, 14–29. [Google Scholar] [CrossRef] [Green Version]
- Vignesh, S.; Raja, A.; James, A.R. Marine drugs: Implication and Future Studies. Int. J. Pharmacol. 2011, 7, 22–30. [Google Scholar] [CrossRef]
- Food and Drug Administration. FDA Basics—Dietary Supplements; Office of Dietary Supplements Programs, HFS-810 (FDA). 2019, USA. Available online: https://www.fda.gov/food/dietary-supplements (accessed on 5 May 2021).
- Scottish Intercollegiate Guidelines Network (SIGN). Risk Estimation and the Prevention of Cardiovascular Disease: A National Clinical Guideline. Edinburgh (Scotland): Scottish Intercollegiate Guidelines Network; 2017. (SIGN publication no. 149); 1–118. Available online: http://www.sign.ac.uk (accessed on 5 May 2021).
- Rahman, K.; Lowe, G.M. Garlic and cardiovascular disease: A critical review. J. Nutr. 2006, 136 (Suppl. 3), 736S–740S. [Google Scholar] [CrossRef]
- Zhang, J.; Wen, C.; Zhang, H.; Duan, Y.; Ma, H. Recent advances in the extraction of bioactive compounds with subcritical water: A review. Trends Food Sci. Technol. 2020, 95, 183–195. [Google Scholar] [CrossRef]
- Gonçalves, R.F.S.; Martins, J.T.; Duarte, C.M.M.; Vicente, A.A.; Pinheiro, A.C. Advances in nutraceutical delivery systems: From formulation design for bioavailability enhancement to efficacy and safety evaluation. Trends Food Sci. Technol. 2018, 78, 270–291. [Google Scholar] [CrossRef] [Green Version]
- Sharma, M.; Koul, A.; Sharma, D.; Kaul, S.; Swamy, M.; Dhar, M.K. Metabolic engineering strategies for enhancing the production of bio-active compounds from medicinal plants. In Natural Bio-Active Compounds; Akhtar, M., Swamy, M., Eds.; Springer: Singapore, 2019. [Google Scholar] [CrossRef]
- Depuydt, S.; Van Praet, S.; Nelissen, H.; Vanholme, B.; Vereecke, D. How plant hormones and their interactions affect cell growth. In Molecular Cell Biology of the Growth and Differentiation of Plant Cells; Rose, R.J., Ed.; CRC Press: Boca Raton, FL, USA, 2016; pp. 174–195. [Google Scholar]
- Chaves Lobón, N.; Ferrer de la Cruz, I.; Alías Gallego, J.C. Autotoxicity of Diterpenes Present in Leaves of Cistus ladanifer L. Plants 2019, 8, 27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Azmir, J.; Zaidul, I.S.M.; Rahman, M.M.; Sharif, K.M.; Mohamed, A.; Sahena, F.; Jahurul, M.H.A.; Ghafoor, K.; Norulaini, N.A.N.; Omar, A.K.M. Techniques for extraction of bioactive compounds from plant materials: A review. J. Food Eng. 2013, 117, 426–436. [Google Scholar] [CrossRef]
- Ahmed, B.; Mohamed, S.; Adel, A.-R. Antioxidant Activities and Potential Impacts to Reduce Aflatoxins Utilizing Jojoba and Jatropha Oils and Extracts. Int. J. Pharmacol. 2017, 13, 1103–1114. [Google Scholar]
- Loi, M.; Paciolla, C.; Logrieco, A.F.; Mulè, G. Plant Bioactive Compounds in Pre- and Postharvest Management for Aflatoxins Reduction. Front. Microbiol. 2020, 11, 243. [Google Scholar] [CrossRef] [PubMed]
- Walden, R.; Tomlinson, B. Cardiovascular Disease. In Herbal Medicine: Biomolecular and Clinical Aspects, 2nd ed.; Benzie, I.F.F., Wachtel-Galor, S., Eds.; CRC Press/Taylor & Francis: Boca Raton, FL, USA, 2011; Chapter 16. Available online: www.ncbi.nlm.nih.gov/books/NBK92767/ (accessed on 5 May 2021).
- Ciumărnean, L.; Milaciu, M.V.; Runcan, O.; Vesa, Ș.C.; Răchișan, A.L.; Negrean, V.; Perné, M.G.; Donca, V.I.; Alexescu, T.G.; Para, I.; et al. The Effects of Flavonoids in Cardiovascular Diseases. Molecules 2020, 25, 4320. [Google Scholar] [CrossRef]
- McCullough, M.L.; Peterson, J.J.; Patel, R.; Jacques, P.F.; Shah, R.; Dwyer, J.T. Flavonoid intake and cardiovascular disease mortality in a prospective cohort of US adults. Am. J. Clin. Nutr. 2012, 95, 454–464. [Google Scholar] [CrossRef]
- Durazzo, A.; Lucarini, M.; Novellino, E.; Souto, E.B.; Daliu, P.; Santini, A. Abelmoschus esculentus L.: Bioactive Components’ Beneficial Properties-Focused on Antidiabetic Role-For Sustainable Health Applications. Molecules 2018, 24, 38. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saida, M.; Lamjed, B.; Amina, B.; Rym, E.; Soumaya, H.; Salem, E. Biological activities, and phytocompounds of northwest Algeria Ajuga iva (L) extracts: Partial identification of the antibacterial fraction. Microb. Pathog. 2018, 121, 173–178. [Google Scholar]
- Wang, S.; Zhao, Y.; Song, J.; Wang, R.; Gao, L.; Zhang, L.; Fang, L.; Lu, Y.; Du, G. Total flavonoids from Anchusa italica Retz. Improve cardiac function and attenuate cardiac remodeling post myocardial infarction in mice. J. Ethnopharmacol. 2020, 257, 112887. [Google Scholar] [CrossRef]
- Bondonno, N.P.; Bondonno, C.P.; Blekkenhorst, L.C.; Considine, M.J.; Maghzal, G.; Stocker, R.; Woodman, R.J.; Ward, N.C.; Hodgson, J.M.; Croft, K.D. Flavonoid-Rich Apple Improves Endothelial Function in Individuals at Risk for Cardiovascular Disease: A Randomized Controlled Clinical Trial. Mol. Nutr. Food Res. 2018, 62, 1700674. [Google Scholar] [CrossRef]
- Panthi, M.; Subba, R.K.; Raut, B.; Khanal, D.P.; Koirala, N. Bioactivity evaluations of leaf extract fractions from young barley grass and correlation with their phytochemical profiles. BMC Complement. Med. Ther. 2020, 20, 64. [Google Scholar] [CrossRef] [Green Version]
- Pereyra, K.V.; Andrade, D.C.; Toledo, C.; Schwarz, K.G.; Uribe-Ojeda, A.; RÃ-os-Gallardo, A.P.; Quintanilla, R.A.; Contreras, S.A.; Del Rio, R. Dietary supplementation of a sulforaphane-enriched broccoli extract protects the heart from acute cardiac stress. J. Funct. Foods 2020, 75, 104267. [Google Scholar] [CrossRef]
- Zaini, R.G.; Brandt, K.; Clench, M.R.; Le Maitre, C.L. Effects of bioactive compounds from carrots (Daucus carota L.), polyacetylenes, beta-carotene and lutein on human lymphoid leukaemia cells. Anticancer Agents Med. Chem. 2012, 12, 640–652. [Google Scholar] [CrossRef] [PubMed]
- Bartłomiej, S.; Justyna, R.K.; Ewa, N. Bioactive compounds in cereal grains—Occurrence, structure, technological significance and nutritional benefits—A review. Food Sci. Technol. Int. 2012, 18, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Martin, M.Á.; Ramos, S. Impact of cocoa flavanols on human health. Food Chem. Toxicol. 2021, 151, 112121. [Google Scholar] [CrossRef]
- Neetu, K.; Choudhary, S.B.; Sharma, H.K.; Singh, B.K.; Kumar, A.A. Health-promoting properties of Corchorus leaves: A review. J. Herb. Med. 2019, 15, 100240. [Google Scholar]
- Haidar, B.; Ferdous, M.; Fatema, B.; Ferdous, A.S.; Islam, M.R.; Khan, H. Population diversity of bacterial endophytes from jute (Corchorus olitorius) and evaluation of their potential role as bioinoculants. Microbiol. Res. 2018, 208, 43–53. [Google Scholar] [CrossRef]
- Polley, K.R.; Oswell, N.J.; Pegg, R.B.; Paton, C.M.; Cooper, J.A. A 5-day high-fat diet rich in cottonseed oil improves cholesterol profiles and triglycerides compared to olive oil in healthy men. Nutr. Res. 2018, 60, 43–53. [Google Scholar] [CrossRef]
- Simha, P.; Mathew, M.; Ganesapillai, M. Empirical modeling of drying kinetics and microwave assisted extraction of bioactive compounds from Adathoda vasica and Cymbopogon citratus. Alex. Eng. J. 2016, 55, 141–150. [Google Scholar] [CrossRef] [Green Version]
- Orgah, J.O.; He, S.; Wang, Y.; Jiang, M.; Wang, Y.; Orgah, E.A.; Duan, Y.; Zhao, B.; Zhang, B.; Han, J.; et al. Pharmacological potential of the combination of Salvia miltiorrhiza (Danshen) and Carthamus tinctorius (Honghua) for diabetes mellitus and its cardiovascular complications. Pharmacol. Res. 2020, 153, 104654. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, S.; Liu, Q.; Zhang, H.; Wang, S.; Liu, Q.; Zheng, H.; Liu, X.; Wang, X.; Shen, T.; et al. Dracomolphin AE, new lignans from Dracocephalum moldavica. Fitoterapia 2021, 150, 104841. [Google Scholar] [CrossRef]
- Bayang, J.P.; Laya, A.; Kolla, M.C.; Koubala, B.B. Variation of physical properties, nutritional value and bioactive nutrients in dry and fresh wild edible fruits of twenty-three species from Far North region of Cameroon. J. Agric. Food Res. 2021, 4, 100146. [Google Scholar] [CrossRef]
- Miao, S.M.; Zhang, Q.; Bi, X.B.; Cui, J.L.; Wang, M.L. A review of the phytochemistry and pharmacological activities of Ephedra herb. Chin. J. Nat. Med. 2020, 18, 321–344. [Google Scholar] [CrossRef]
- Graefe, E.U.; Veit, M. Urinary metabolites of flavonoids and hydroxycinnamic acids in humans after application of a crude extract from Equisetum arvense. Phytomedicine 1999, 6, 239–246. [Google Scholar] [CrossRef]
- Shim, Y.Y.; Gui, B.; Arnison, P.G.; Shim, Y.Y.; Gui, B.; Arnison, P.G.; Wang, Y.; Reaney, M.J.T.; Jiang, M.; Wang, Y.; et al. Flaxseed (Linum usitatissimum L.) bioactive compounds and peptide nomenclature: A review. Trends Food Sci. Technol. 2014, 38, 5–20. [Google Scholar] [CrossRef] [Green Version]
- Al-Snafi, A. Phytochemical constituents and medicinal properties of digitalis lanata and digitalis purpurea—A Review. IAJPS 2017, 4, 225–234. [Google Scholar]
- Martins, N.; Petropoulos, S.; Ferreira, I.C. Chemical composition and bioactive compounds of garlic (Allium sativum L.) as affected by pre- and post-harvest conditions: A review. Food Chem. 2016, 211, 41–50. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hassan, I.; Ibrahim, W.; Yusuf, F.M.; Ahmad, S.A.; Ahmad, S. Biochemical Constituent of Ginkgo biloba (Seed) 80% Methanol Extract Inhibits Cholinesterase Enzymes in Javanese Medaka (Oryzias javanicus) Model. J. Toxicol. 2020, 2020, 8815313. [Google Scholar] [CrossRef]
- Yu, H.; Zhao, J.; You, J.; Li, J.; Ma, H.; Chen, X. Factors influencing cultivated ginseng (Panax ginseng CA Meyer) bioactive compounds. PLoS ONE 2019, 14, e0223763. [Google Scholar] [CrossRef]
- Rojas, R.; Castro-lópez, C.; Sánchez-Alejo, E.J.; Niño-Medina, G.; Martínez-Ávila, C.G. Phenolic compound recovery from grape fruit and by- products: An overview of extraction methods. Grape Wine Biotechnol. 2016. [Google Scholar] [CrossRef]
- Rababa’h, A.M.; Al Yacoub, O.N.; El-Elimat, T.; Rabab’ah, M.; Altarabsheh, S.; Deo, S.; Al-Azayzih, A.; Zayed, A.; Alazzam, S.; Alzoubi, K.H. The effect of hawthorn flower and leaf extract (Crataegus Spp.) on cardiac hemostasis and oxidative parameters in Sprague Dawley rats. Heliyon 2020, 6, e04617. [Google Scholar] [CrossRef] [PubMed]
- Souza, J.S.N.; Machado, L.L.; Pessoa, O.D.; Braz-Filho, R.; Overk, C.R.; Yao, P.; Lemos, T.L. Pyrrolizidine alkaloids from heliotropium indicum. J. Braz. Chem. Soc. 2005, 16, 1410–1414. [Google Scholar] [CrossRef] [Green Version]
- De Angelis, D.; Pasqualone, A.; Costantini, M.; Ricciardi, L.; Lotti, C.; Pavan, S.; Summo, C. Data on the proximate composition, bioactive compounds, physicochemical and functional properties of a collection of faba beans (Vicia faba L.) and lentils (Lens culinaris Medik.). Data Brief 2020, 34, 106660. [Google Scholar] [CrossRef]
- Lu, J.; He, R.; Sun, P.; Zhang, F.; Linhardt, R.J.; Zhang, A. Molecular mechanisms of bioactive polysaccharides from Ganoderma lucidum (Lingzhi), a review. Int. J. Biol. Macromol. 2020, 150, 765–774. [Google Scholar] [CrossRef]
- Sudheer, S.; Alzorqi, I.; Manickam, S. Bioactive Compounds of the Wonder Medicinal Mushroom “Ganoderma lucidum”. In Bioactive Molecules in Food. Reference Series in Phytochemistry; Mérillon, J.M., Ramawat, K., Eds.; Springer: Cham, Switzerland, 2019. [Google Scholar]
- Leone, A.; Spada, A.; Battezzati, A.; Schiraldi, A.; Aristil, J.; Bertoli, S. Moringa oleifera Seeds and Oil: Characteristics and Uses for Human Health. Int. J. Mol. Sci. 2016, 17, 2141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tian, Y.; Deng, F. Phytochemistry and biological activity of mustard (Brassica juncea): A review. CyTA-J. Food 2020, 18, 704–718. [Google Scholar] [CrossRef]
- Wu, J.R.; Leu, H.B.; Yin, W.H. The benefit of secondary prevention with oat fiber in reducing future cardiovascular event among CAD patients after coronary intervention. Sci. Rep. 2019, 9, 3091. [Google Scholar] [CrossRef] [PubMed]
- Boskou, D. Table Olives as Sources of Bioactive Compounds. In Olive and Olive Oil Bioactive Constituents. J. Agric. Food Chem. 2015, 217–259. [Google Scholar] [CrossRef]
- Marrelli, M.; Amodeo, V.; Statti, G.; Conforti, F. Biological Properties and Bioactive Components of Allium cepa L.: Focus on Potential Benefits in the Treatment of Obesity and Related Comorbidities. Molecules 2018, 24, 119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alper, C.M.; Mattes, R.D. Peanut consumption improves indices of cardiovascular disease risk in healthy adults. J. Am. Coll. Nutr. 2003, 22, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, M.; Boughalleb, F.; Bouhemda, T.; Abdellaoui, R.; Nasri, N. Unexploited polygonum equisetiforme seeds: Potential source of useful natural bioactive products. Ind. Crop. Prod. 2018, 122, 349–357. [Google Scholar] [CrossRef]
- Shah, A.R.; Sharma, P.; Gour, V.S.; Kothari, S.L.; Dar, K.B.; Ganie, S.A.; Shah, Y.R. Antioxidant, Nutritional, Structural, Thermal and Physico-Chemical Properties of Psyllium (Plantago Ovata) Seeds. Curr. Res. Nutr. Food Sci. 2020, 8. [Google Scholar] [CrossRef]
- Ferraz da Costa, D.C.; Pereira Rangel, L.; Quarti, J.; Santos, R.A.; Silva, J.L.; Fialho, E. Bioactive compounds and metabolites from grapes and red wine in breast cancer chemoprevention and therapy. Molecules 2020, 25, 3531. [Google Scholar] [CrossRef] [PubMed]
- Salehi, E.; Sardarodiyan, M. Bioactive phytochemicals in rice bran: Processing and functional properties. Biochem. Ind. J. 2016, 10, 101. [Google Scholar]
- Zampelas, A. The Effects of Soy and its Components on Risk Factors and End Points of Cardiovascular Diseases. Nutrients 2019, 11, 2621. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahmed, H.M.; Al-Zubaidy, A.M.A. Exploring natural essential oil components and antibacterial activity of solvent extracts from twelve Perilla frutescens L. Genotypes. Arab. J. Chem. 2020, 13, 7390–7402. [Google Scholar] [CrossRef]
- Cheng, H.M.; Koutsidis, G.; Lodge, J.K.; Ashor, A.; Siervo, M.; Lara, J. Tomato and lycopene supplementation and cardiovascular risk factors: A systematic review and meta-analysis. Atherosclerosis 2017, 257, 100–108. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, S.; Xu, T.; Akoh, C. Effect of roasting on the volatile constituents of trichosanthes kirilowii seeds. J. Food Drug Anal. 2014, 22, 310–317. [Google Scholar] [CrossRef]
- Melini, V.; Acquistucci, R. Health-promoting compounds in pigmented thai and wild rice. Foods 2017, 6, 9. [Google Scholar] [CrossRef] [Green Version]
- Dai, W.; Zhang, Z.; Zhao, S. Baseline levels of serum high sensitivity C reactive protein and lipids in predicting the residual risk of cardiovascular events in Chinese population with stable coronary artery disease: A prospective cohort study. Lipids Health Dis. 2018, 17, 273. [Google Scholar] [CrossRef] [Green Version]
- Ververidis, F.; Trantas, E.; Douglas, C.; Vollmer, G.; Kretzschmar, G.; Panopoulos, N. Biotechnology of flavonoids and other phenylpropanoid-derived natural products. Part I: Chemical diversity, impacts on plant biology and human health. Biotechnol. J. 2007, 2, 1214–1234. [Google Scholar] [CrossRef] [PubMed]
- Lotito, S.B.; Frei, B. Consumption of flavonoid-rich foods and increased plasma antioxidant capacity in humans: Cause, consequence, or epiphenomenon? Free Radic. Biol. Med. 2006, 41, 1727–1746. [Google Scholar] [CrossRef] [PubMed]
- Izzi, V.; Masuelli, L.; Tresoldi, I.; Sacchetti, P.; Modesti, A.; Galvano, F.; Bei, R. The effects of dietary flavonoids on the regulation of redox inflammatory networks. Front. Biosci. 2012, 17, 2396–2418. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, C.F.; Cho, S.; Wang, J. Epicatechin protects hemorrhagic brain via synergistic Nrf2 pathways. Ann. Clin. Transl. Neurol. 2014, 1, 258–271. [Google Scholar] [CrossRef] [PubMed]
- Nishiumi, S.; Miyamoto, S.; Kawabata, K.; Ohnishi, K.; Mukai, R.; Murakami, A.; Ashida, H.; Terao, J. Dietary flavonoids as cancer-preventive and therapeutic biofactors. Front. Biosci. 2011, 3, 1332–1362. [Google Scholar] [CrossRef]
- McKay, D.L.; Chen, C.Y.; Saltzman, E.; Blumberg, J.B. Hibiscus sabdariffa L. tea (tisane) lowers blood pressure in prehypertensive and mildly hypertensive adults. J. Nutr. 2010, 140, 298–303. [Google Scholar] [CrossRef] [PubMed]
- Clark, J.L.; Zahradka, P.; Taylor, C.G. Efficacy of flavonoids in the management of high blood pressure. Nutr. Rev. 2015, 73, 799–822. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, A.M.; Hernández Bautista, R.J.; Sandhu, M.A.; Hussein, O.E. Beneficial Effects of Citrus Flavonoids on Cardiovascular and Metabolic Health. Oxid. Med. Cell. Longev. 2019, 2019, 5484138. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Su, J.; Xu, H.-T.; Yu, J.-J.; Gao, J.; Lei, J.; Yin, Q.S.; Li, B.; Pang, M.X.; Su, M.X.; Mi, W.J.; et al. Luteolin ameliorates hypertensive vascular remodeling through inhibiting the proliferation and migration of vascular smooth muscle cells. Evid. Based Complement. Altern. Med. 2015, 2015, 364876. [Google Scholar] [CrossRef] [Green Version]
- Thamcharoen, N.; Susantitaphong, P.; Wongrakpanich, S.; Chongsathidkiet, P.; Tantrachoti, P.; Pitukweerakul, S.; Avihingsanon, Y.; Praditpornsilpa, K.; Jaber, B.L.; Eiam-Ong, S. Effect of N- and T-type calcium channel blocker on proteinuria, blood pressure and kidney function in hypertensive patients: A meta-analysis. Hypertens. Res. 2015, 38, 902. [Google Scholar] [CrossRef] [Green Version]
- Jiang, R.W.; Lau, K.M.; Lam, H.M.; Yam, W.S.; Leung, L.K.; Choi, K.L.; Waye, M.M.; Mak, T.C.; Woo, K.S.; Fung, K.P. A comparative study on aqueous root extracts of Pueraria thomsonii and Pueraria lobata by antioxidant assay and HPLC fingerprint analysis. J. Ethnopharmacol. 2005, 96, 133–138. [Google Scholar] [CrossRef]
- Tsuda, T. Dietary anthocyanin-rich plants: Biochemical basis and recent progress in health benefits studies. Mol. Nutr. Food Res. 2012, 56, 159–170. [Google Scholar] [CrossRef] [PubMed]
- Srilakshami, B. Food Science, 7th ed.; New Age International Publishers: New Delhi, India, 2018. [Google Scholar]
- Celestino, S.B.; Augustin, S. Proanthocyanidins and tannin-like compounds—Nature, occurrence, dietary intake and effects on nutrition and health. J. Sci. Food Agric. 2000, 80, 1094–1117. [Google Scholar]
- Salisbury, F.; Ross, C. Plant Physiology, 4th ed.; Wadsworth: Belmont, CA, USA, 1991; pp. 325–326. [Google Scholar]
- Rahimi, P.; Abedimanesh, S.; Mesbah-Namin, S.A.; Ostadrahimi, A. Betalains, the nature-inspired pigments, in health and diseases. Crit. Rev. Food Sci. Nutr. 2019, 59, 2949–2978. [Google Scholar] [CrossRef]
- Antonescu, A.-I.; Miere, F.; Fritea, L.; Ganea, M.; Zdrinca, M.; Dobjanschi, L.; Antonescu, A.; Vicas, S.I.; Bodog, F.; Sindhu, R.K.; et al. Perspectives on the Combined Effects of Ocimum basilicum and Trifolium pratense Extracts in Terms of Phytochemical Profile and Pharmacological Effects. Plants 2021, 10, 1390. [Google Scholar] [CrossRef] [PubMed]
- Leoncini, E.; Nedovic, D.; Panic, N.; Pastorino, R.; Edefonti, V.; Boccia, S. Carotenoid Intake from Natural Sources and Head and Neck Cancer: A Systematic Review and Meta-analysis of Epidemiological Studies. Cancer Epidemiol. Biomark. Prev. 2015, 24, 1003–1011. [Google Scholar] [CrossRef] [Green Version]
- Voutilainen, S.; Nurmi, T.; Mursu, J.; Rissanen, T.H. Carotenoids and cardiovascular health. Am. J. Clin. Nutr. 2006, 83, 1265–1271. [Google Scholar] [CrossRef] [PubMed]
- Granado-Lorencio, F.; Lagarda, M.J.; Garcia-López, F.J.; Sánchez-Siles, L.M.; Blanco-Navarro, I.; Alegría, A.; Pérez-Sacristán, B.; Garcia-Llatas, G.; Donoso-Navarro, E.; Silvestre-Mardomingo, R.A.; et al. Effect of β-cryptoxanthin plus phytosterols on cardiovascular risk and bone turnover markers in post-menopausal women: A randomized crossover trial. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 1090–1096. [Google Scholar] [CrossRef] [PubMed]
- Osganian, S.K.; Stampfer, M.J.; Rimm, E.; Spiegelman, D.; Manson, J.E.; Willett, W.C. Dietary carotenoids and risk of coronary artery disease in women. Am. J. Clin. Nutr. 2003, 77, 1390–1399. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ross, A.C.; Zolfaghari, R.; Weisz, J. Vitamin A: Recent advances in the biotransformation, transport, and metabolism of retinoids. Curr. Opin. Gastroenterol. 2001, 17, 184–192. [Google Scholar] [CrossRef] [PubMed]
- Holden, J.M.; Eldridge, A.L.; Beecher, G.R.; Buzzard, I.M.; Bhagwat, S.; Davis, C.S.; Douglass, L.W.; Gebhardt, S.; Haytowiz, D.; Schakel, S. Carotenoid content of US foods: An update of the database. J. Food Comp. Anal. 1999, 12, 169–196. [Google Scholar] [CrossRef] [Green Version]
- Stahl, W.; Sies, H. Lycopene: A biologically important carotenoid for humans? Arch. Biochem. Biophys. 1996, 336, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Sommerburg, O.; Keunen, J.E.; Bird, A.; Van Kuijk, F. J G M. Fruits and vegetables that are sources for lutein and zeaxanthin: The macular pigment in human eyes. Br. J. Ophthalmol. 1998, 82, 907–910. [Google Scholar] [CrossRef] [PubMed]
- Jialal, I.; Norkus, E.P.; Cristol, L.; Grundy, S.M. beta-Carotene inhibits the oxidative modification of low-density lipoprotein. Biochim. Biophys. Acta 1991, 1086, 134–138. [Google Scholar] [CrossRef]
- Ciccone, M.M.; Cortese, F.; Gesualdo, M.; Carbonara, S.; Zito, A.; Ricci, G.; De Pascalis, F.; Scicchitano, P.; Riccioni, G. Dietary intake of carotenoids and their antioxidant and anti-inflammatory effects in cardiovascular care. Mediat. Inflamm. 2013, 2013, 782137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Trautwein, E.A.; Vermeer, M.A.; Hiemstra, H.; Ras, R.T. LDL-Cholesterol Lowering of Plant Sterols and Stanols-Which Factors Influence Their Efficacy? Nutrients 2018, 10, 1262. [Google Scholar] [CrossRef] [Green Version]
- Radojčić Redovniković, I.; Glivetić, T.; Delonga, K.; Vorkapic-Furac, J. Glucosinolates and their potential role in plant. Period. Biol. 2008, 110, 297–309. [Google Scholar]
- Tse, G.; Eslick, G.D. Cruciferous vegetables and risk of colorectal neoplasms: A systematic review and meta-analysis. Nutr. Cancer 2014, 66, 128–139. [Google Scholar] [CrossRef] [PubMed]
- Soundararajan, P.; Kim, J.S. Anti-Carcinogenic Glucosinolates in Cruciferous Vegetables and Their Antagonistic Effects on Prevention of Cancers. Molecules 2018, 23, 2983. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Loef, M.; Walach, H. Fruit, vegetables and prevention of cognitive decline or dementia: A systematic review of cohort studies. J. Nutr. Health Aging 2012, 16, 626–630. [Google Scholar] [CrossRef]
- Walia, A.; Gupta, A.K.; Sharma, V. Role of Bioactive Compounds in Human Health. Acta Sci. Med. Sci. 2019, 3, 25–33. [Google Scholar]
- Greenberg, E.R.; Baron, J.A.; Karagas, M.R.; Greenberg, E.R.; Baron, J.A.; Karagas, M.R.; Stukel, T.A.; Nierenberg, D.W.; Stevens, M.M.; Mandel, J.S.; et al. Mortality associated with low plasma concentration of beta carotene and the effect of oral supplementation. JAMA 1996, 275, 699–703. [Google Scholar] [CrossRef] [PubMed]
- Heart Protection Study Collaborative Group. MRC/BHF Heart Protection Study of antioxidant vitamin supplementation in 20,536 high-risk individuals: A randomised placebo-controlled trial. Lancet 2002, 360, 23–33. [Google Scholar] [CrossRef]
- Liu, S.; Lee, I.M.; Ajani, U.; Cole, S.R.; Buring, J.E.; Manson, J.E. Physicians’ Health Study. Intake of vegetables rich in carotenoids and risk of coronary heart disease in men: The Physicians’ Health Study. Int. J. Epidemiol. 2001, 30, 130–135. [Google Scholar] [CrossRef] [Green Version]
- Hennekens, C.H.; Buring, J.E.; Manson, J.E.; Stampfer, M.; Rosner, B.; Cook, N.R.; Belanger, C.; LaMotte, F.; Gaziano, J.M.; Ridker, P.M.; et al. Lack of effect of long-term supplementation with beta carotene on the incidence of malignant neoplasms and cardiovascular disease. N. Engl. J. Med. 1996, 334, 1145–1149. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Basu, A.; Rhone, M.; Lyons, T.J. Berries: Emerging impact on cardiovascular health. Nutr. Rev. 2010, 68, 168–177. [Google Scholar] [CrossRef] [Green Version]
- Basu, A.; Du, M.; Leyva, M.J.; Sanchez, K.; Betts, N.M.; Wu, M.; Aston, C.E.; Lyons, T.J. Blueberries decrease cardiovascular risk factors in obese men and women with metabolic syndrome. J. Nutr. 2010, 140, 1582–1587. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goszcz, K.; Duthie, G.G.; Stewart, D.; Leslie, S.J.; Megson, I.L. Bioactive polyphenols and cardiovascular disease: Chemical antagonists, pharmacological agents or xenobiotics that drive an adaptive response? Br. J. Pharmacol. 2017, 174, 1209–1225. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Asgary, S.; Sahebkar, A.; Afshani, M.R.; Keshvari, M.; Haghjooyjavanmard, S.; Rafieian-Kopaei, M. Clinical evaluation of blood pressure lowering, endothelial function improving, hypolipidemic and anti-inflammatory effects of pomegranate juice in hypertensive subjects. Phytother. Res. 2014, 28, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Dohadwala, M.M.; Holbrook, M.; Hamburg, N.M.; Shenouda, S.M.; Chung, W.B.; Titas, M.; Kluge, M.A.; Wang, N.; Palmisano, J.; Milbury, P.E.; et al. Effects of cranberry juice consumption on vascular function in patients with coronary artery disease. Am. J. Clin. Nutr. 2011, 93, 934–940. [Google Scholar] [CrossRef]
- Arab, L.; Steck, S. Lycopene and cardiovascular disease. Am. J. Clin. Nutr. 2000, 71 (Suppl. 6), 1691S–1697S. [Google Scholar] [CrossRef] [Green Version]
- Rees, A.; Dodd, G.F.; Spencer, J.P.E. The Effects of Flavonoids on Cardiovascular Health: A Review of Human Intervention Trials and Implications for Cerebrovascular Function. Nutrients 2018, 10, 1852. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, X.; Ouyang, Y.Y.; Liu, J.; Zhao, G. Flavonoid intake and risk of CVD: A systematic review and meta-analysis of prospective cohort studies. Br. J. Nutr. 2014, 111, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, P.; Mahato, D.K.; Kamle, M.; Mohanta, T.K.; Kang, S.G. Aflatoxins: A global concern for food safety, human health and their management. Front. Microbial. 2017, 7, 2170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Williamson, G.; Kay, C.D.; Crozier, A. The bioavailability, transport, and bioactivity of dietary flavonoids: A review from a historical perspective. Compr. Rev. Food Sci. Food Saf. 2018, 17, 1054–1112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thilakarathna, S.H.; Rupasinghe, H.P. Flavonoid bioavailability and attempts for bioavailability enhancement. Nutrients 2013, 5, 3367–3387. [Google Scholar] [CrossRef]
- Kawabata, K.; Yoshioka, Y.; Terao, J. Role of Intestinal Microbiota in the Bioavailability and Physiological Functions of Dietary Polyphenols. Molecules 2019, 24, 370. [Google Scholar] [CrossRef] [Green Version]
- Pei, R.; Liu, X.; Bolling, B. Flavonoids and gut health. Curr. Opin. Biotechnol. 2020, 61, 153–159. [Google Scholar] [CrossRef]
- Cinta-Pinzaru, S.; Cavalu, S.; Leopold, N.; Petry, R.; Kiefer, W. Raman and surface-enhanced Raman spectroscopy of tempyo spin labelled ovalbumin. J. Mol. Struct. 2001, 565, 225–229. [Google Scholar] [CrossRef]
- Arriagada, F.; Günther, G.; Morales, J. Nanoantioxidant-Based Silica Particles as Flavonoid Carrier for Drug Delivery Applications. Pharmaceutics 2020, 12, 302. [Google Scholar] [CrossRef] [Green Version]
- Zuo, L.; Ao, X.; Guo, Y. Study on the synthesis of dual-chain ionic liquids and their application in the extraction of flavonoids. J. Chromatogr. A 2020, 1628, 461446. [Google Scholar] [CrossRef]
- De Whalley, C.V.; Rankin, S.M.; Hoult, J.R.; Jessup, W.; Leake, D.S. Flavonoids inhibit the oxidative modification of low-density lipoproteins by macrophages. Biochem. Pharmacol. 1990, 39, 1743–1750. [Google Scholar] [CrossRef]
- Fuhrman, B.; Lavy, A.; Aviram, M. Consumption of red wine with meals reduces the susceptibility of human plasma and low-density lipoprotein to lipid peroxidation. Am. J. Clin. Nutr. 1995, 61, 549–554. [Google Scholar] [CrossRef]
- Arai, Y.; Watanabe, S.; Kimira, M.; Shimoi, K.; Mochizuki, R.; Kinae, N. Dietary intakes of flavonols, flavones and isoflavones by Japanese women and the inverse correlation between quercetin intake and plasma LDL cholesterol concentration. J. Nutr. 2000, 130, 2243–2250. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lou, F.Q.; Zhang, M.F.; Zhang, X.G.; Liu, J.M.; Yuan, W.L. A study on tea-pigment in prevention of atherosclerosis. Chin. Med. J. 1989, 102, 579–583. [Google Scholar] [PubMed]
- Osman, H.E.; Maalej, N.; Shanmuganayagam, D.; Folts, J.D. Grape juice but not orange or grapefruit juice inhibits platelet activity in dogs and monkeys. J. Nutr. 1998, 128, 2307–2312. [Google Scholar] [CrossRef] [Green Version]
- Gryglewski, R.J.; Korbut, R.; Robak, J.; Swies, J. On the mechanism of antithrombotic action of flavonoids. Biochem. Pharmacol. 1987, 36, 317–322. [Google Scholar] [CrossRef]
- Kandaswami, C.; Middleton, E., Jr. Free radical scavenging and antioxidant activity of plant flavonoids. Adv. Exp. Med. Biol. 1994, 366, 351–376. [Google Scholar] [PubMed]
- Rodríguez-García, C.; Sánchez-Quesada, C.; Gaforio, J. Dietary Flavonoids as Cancer Chemopreventive Agents: An Updated Review of Human Studies. Antioxidants 2019, 8, 137. [Google Scholar] [CrossRef] [Green Version]
- Bast, A.; Kaiserová, H.; den Hartog, G.J.; Haenen, G.R.; Van der Vijgh, W.J. Protectors against doxorubicin-induced cardiotoxicity: Flavonoids. Cell Biol. Toxicol. 2007, 23, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Fuhrman, B.; Aviram, M. Flavonoids protect LDL from oxidation and attenuate atherosclerosis. Curr. Opin. Lipidol. 2001, 12, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Bernátová, I.; Pechánová, O.; Babál, P.; Kyselá, S.; Stvrtina, S.; Andriantsitohaina, R. Wine polyphenols improve cardiovascular remodeling and vascular function in NO-deficient hypertension. Am. J. Physiol. Heart Circ. Physiol. 2002, 282, H942–H948. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jayakody, R.L.; Senaratne, M.P.; Thomson, A.B.; Kappagoda, C.T. Cholesterol feeding impairs endothelium-dependent relaxation of rabbit aorta. Can. J. Physiol. Pharmacol. 1985, 63, 1206–1209. [Google Scholar] [CrossRef] [PubMed]
- Hertog, M.G.; Feskens, E.J.; Hollman, P.C.; Katan, M.B.; Kromhout, D. Dietary antioxidant flavonoids and risk of coronary heart disease: The Zutphen Elderly Study. Lancet 1993, 342, 1007–1011. [Google Scholar] [CrossRef]
- Khoo, H.E.; Azlan, A.; Tang, S.T.; Lim, S.M. Anthocyanidins and anthocyanins: Colored pigments as food, pharmaceutical ingredients, and the potential health benefits. Food Nutr. Res. 2013, 61, 1361779. [Google Scholar] [CrossRef] [Green Version]
- Andriambeloson, E.; Kleschyov, A.L.; Muller, B.; Beretz, A.; Stoclet, J.C.; Andriantsitohaina, R. Nitric oxide production and endothelium-dependent vasorelaxation induced by wine polyphenols in rat aorta. Br. J. Pharmacol. 1997, 120, 1053–1058. [Google Scholar] [CrossRef] [Green Version]
- Burns, J.; Gardner, P.T.; O’Neil, J.; Crawford, S.; Morecroft, I.; McPhail, D.B.; Lister, C.; Matthews, D.; MacLean, M.R.; Lean, M.E.; et al. Relationship among antioxidant activity, vasodilation capacity, and phenolic content of red wines. J. Agric. Food Chem. 2000, 48, 220–230. [Google Scholar] [CrossRef]
- Trombino, S.; Cassano, R.; Muzzalupo, R.; Pingitore, A.; Cione, E.; Picci, N. Stearyl ferulate-based solid lipid nanoparticles for the encapsulation and stabilization of beta-carotene and alpha-tocopherol. Colloids Surf. B Biointerfaces 2009, 72, 181–187. [Google Scholar] [CrossRef]
- Vicas, S.I.; Cavalu, S.; Laslo, V.; Tocai, M.; Costea, T.O.; Moldovan, L. Growth, Photosynthetic Pigments, Phenolic, Glucosinolates Content and Antioxidant Capacity of Broccoli Sprouts in Response to Nanoselenium Particles Supply. Not. Bot. Horti Agrobot. Cluj Napoca 2019, 47, 821–828. [Google Scholar] [CrossRef]
- Vega-Villa, K.R.; Takemoto, J.K.; Yáñez, J.A.; Remsberg, C.M.; Forrest, M.L.; Davies, N.M. Clinical toxicities of nanocarrier systems. Adv. Drug Deliv. Rev. 2008, 60, 929–938. [Google Scholar] [CrossRef]
- Pala, R.; Anju, V.T.; Dyavaiah, M.; Busi, S.; Nauli, S.M. Nanoparticle-Mediated Drug Delivery for the Treatment of Cardiovascular Diseases. Int. J. Nanomed. 2020, 15, 3741–3769. [Google Scholar] [CrossRef]
- Borel, T.; Sabliov, C.M. Nanodelivery of bioactive components for food applications: Types of delivery systems, properties, and their effect on ADME profiles and toxicity of nanoparticles. Annu. Rev. Food Sci. Technol. 2014, 5, 197–213. [Google Scholar] [CrossRef] [PubMed]
- Vaiserman, A.; Koliada, A.; Zayachkivska, A.; Lushchak, O. Nanodelivery of Natural Antioxidants: An Anti-aging Perspective. Front. Bioeng. Biotechnol. 2020, 7, 447. [Google Scholar] [CrossRef] [Green Version]
- Naahidi, S.; Jafari, M.; Edalat, F.; Raymond, K.; Khademhosseini, A.; Chen, P. Biocompatibility of engineered nanoparticles for drug delivery. J. Control. Release 2013, 166, 182–194. [Google Scholar] [CrossRef]
- Bertrand, N.; Leroux, J.C. The journey of a drug-carrier in the body: An anatomo-physiological perspective. J. Control. Release 2012, 161, 152–163. [Google Scholar] [CrossRef]
- Tena, N.; Martín, J.; Asuero, A.G. State of the Art of Anthocyanins: Antioxidant Activity, Sources, Bioavailability, and Therapeutic Effect in Human Health. Antioxidants 2020, 9, 451. [Google Scholar] [CrossRef]
- Dube, A.; Ng, K.; Nicolazzo, J.; Larson, I.C. Effective use of reducing agents and nanoparticle encapsulation in stabilizing catechins in alkaline solution. Food Chem. 2010, 122, 662–667. [Google Scholar] [CrossRef]
- Oliveira, G.; Volino, M.; Conte-Junior, C.A.; Alvares, T.S. Food-derived polyphenol compounds and cardiovascular health: A nano-technological perspective. Food Biosci. 2021, 41, 101033. [Google Scholar] [CrossRef]
- Cavalu, S.; Bisboaca, S.; Mates, I.M.; Pasca, P.; Vasile, L.; Costea, T.; Luminita, F.; Vicas, S.L. Novel Formulation Based on Chitosan-Arabic Gum Nanoparticles Entrapping Propolis Extract Production, physico-chemical and structural characterization. Rev. Chim. 2018, 69, 3756–3760. [Google Scholar] [CrossRef]
- Alotaibi, B.; Tousson, E.; El-Masry, T.A.; Altwaijry, N.; Saleh, A. Ehrlich ascites carcinoma as model for studying the cardiac protective effects of curcumin nanoparticles against cardiac damage in female mice. Environ. Toxicol. 2021, 36, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhou, Y.; Zhang, W.; Bao, C.; Xie, Z. Relief of oxidative stress and cardiomyocyte apoptosis by using curcumin nanoparticles. Colloids Surf. B Biointerfaces 2017, 153, 174–182. [Google Scholar] [CrossRef]
- Rachmawati, H.; Soraya, I.S.; Kurniati, N.F.; Rahma, A. In Vitro Study on Antihypertensive and Anti-hypercholesterolemic Effects of a Curcumin Nano emulsion. Sci. Pharm. 2016, 84, 131–140. [Google Scholar] [CrossRef] [Green Version]
- Carlson, L.J.; Cote, B.; Alani, A.W.; Rao, D.A. Polymeric micellar co-delivery of resveratrol and curcumin to mitigate in vitro doxorubicin-induced cardiotoxicity. J. Pharm. Sci. 2014, 103, 2315–2322. [Google Scholar] [CrossRef]
- Zhang, L.; Zhu, K.; Zeng, H.; Zhang, J.; Pu, Y.; Wang, Z.; Zhang, T.; Wang, B. Resveratrol solid lipid nanoparticles to trigger credible inhibition of doxorubicin cardiotoxicity. Int. J. Nanomed. 2019, 14, 6061–6071. [Google Scholar] [CrossRef] [Green Version]
- Xu, H.; Hua, Y.; Zhong, J.; Li, X.; Xu, W.; Cai, Y.; Mao, Y.; Lu, X. Resveratrol Delivery by Albumin Nanoparticles Improved Neurological Function and Neuronal Damage in Transient Middle Cerebral Artery Occlusion Rats. Front. Pharmacol. 2018, 9, 1403. [Google Scholar] [CrossRef]
- Lozano, O.; Lázaro-Alfaro, A.; Silva-Platas, C.; Oropeza-Almazán, Y.; Torres-Quintanilla, A.; Bernal-Ramírez, J.; Alves-Figueiredo, H.; García-Rivas, G. Nanoencapsulated Quercetin Improves Cardioprotection during Hypoxia-Reoxygenation Injury through Preservation of Mitochondrial Function. Oxid. Med. Cell. Longev. 2019, 2019, 7683051. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, C.J.; Yao, L.; Hu, Y.M.; Zhao, B.T. Effect of Quercetin-Loaded Mesoporous Silica Nanoparticles on Myocardial Ischemia-Reperfusion Injury in Rats and Its Mechanism. Int. J. Nanomed. 2021, 16, 741–752. [Google Scholar] [CrossRef] [PubMed]
- Quagliariello, V.; Vecchione, R.; De Capua, A.; Lagreca, E.; Iaffaioli, R.V.; Botti, G.; Netti, P.A.; Maurea, N. Nano-Encapsulation of Coenzyme Q10 in Secondary and Tertiary Nano-Emulsions for Enhanced Cardioprotection and Hepatoprotection in Human Cardiomyocytes and Hepatocytes During Exposure to Anthracyclines and Trastuzumab. Int. J. Nanomed. 2020, 15, 4859–4876. [Google Scholar] [CrossRef] [PubMed]
- Imaizumi, A. Highly bioavailable curcumin (Theracurmin): Its development and clinical application. PharmaNutrition 2015, 3, 123–130. [Google Scholar] [CrossRef]
- Kanai, M.; Imaizumi, A.; Otsuka, Y.; Sasaki, H.; Hashiguchi, M.; Tsujiko, K.; Matsumoto, S.; Ishiguro, H.; Chiba, T. Dose-escalation and pharmacokinetic study of nanoparticle curcumin, a potential anticancer agent with improved bioavailability, in healthy human volunteers. Cancer Chemother. Pharmacol. 2012, 69, 65–70. [Google Scholar] [CrossRef] [Green Version]
- Sasaki, H.; Sunagawa, Y.; Takahashi, K.; Imaizumi, A.; Fukuda, H.; Hashimoto, T.; Wada, H.; Katanasaka, Y.; Kakeya, H.; Fujita, M.; et al. Innovative preparation of curcumin for improved oral bioavailability. Biol. Pharm. Bull. 2011, 34, 660–665. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muthu, M.S.; Mei, L.; Feng, S.S. Nanotheranostics: Advanced nanomedicine for the integration of diagnosis and therapy. Nanomedicine 2014, 9, 1277–1280. [Google Scholar] [CrossRef] [PubMed]
- Pala, R.; Pattnaik, S.; Busi, S.; Nauli, S.M. Nanomaterials as Novel Cardiovascular Theranostics. Pharmaceutics 2021, 13, 348. [Google Scholar] [CrossRef]
- Cavalu, S.; Fritea, L.; Brocks, M.; Barbaro, K.; Murvai, G.; Costea, T.O.; Antoniac, I.; Verona, C.; Romani, M.; Latini, A.; et al. Novel Hybrid Composites Based on PVA/SeTiO2Nanoparticles and Natural Hydroxyapatite for Orthopedic Applications: Correlations between Structural, Morphological and Biocompatibility Properties. Materials 2020, 13, 2077. [Google Scholar] [CrossRef] [PubMed]
- Fritea, L.; Banica, F.; Costea, T.O.; Moldovan, L.; Iovan, C.; Cavalu, S. A gold-nanoparticles—Graphene based electrochemical sensor for sensitive determination of nitrazepam. J. Electroanal. Chem. 2018, 830, 63–71. [Google Scholar] [CrossRef]
- Suzuki, M.; Bachelet-Violette, L.; Rouzet, F.; Beilvert, A.; Autret, G.; Maire, M.; Menager, C.; Louedec, L.; Choqueux, C.; Saboural, P.; et al. Ultrasmall superparamagnetic iron oxide nanoparticles coated with fucoidan for molecular MRI of intraluminal thrombus. Nanomedicine 2015, 10, 73–87. [Google Scholar] [CrossRef] [PubMed]
- Danila, D.; Johnson, E.; Kee, P. CT imaging of myocardial scars with collagen-targeting gold nanoparticles. Nanomedicine 2013, 9, 1067–1076. [Google Scholar] [CrossRef]
- Lestini, B.J.; Sagnella, S.M.; Xu, Z.; Shive, M.S.; Richter, N.J.; Jayaseharan, J.; Case, A.J.; Kottke-Marchant, K.; Anderson, J.M.; Marchant, R.E. Surface modification of liposomes for selective cell targeting in cardiovascular drug delivery. J. Control. Release 2002, 78, 235–247. [Google Scholar] [CrossRef]
- Bilia, A.R.; Piazzini, V.; Guccione, C.; Risaliti, L.; Asprea, M.; Capecchi, G.; Bergonzi, M.C. Improving on Nature: The Role of Nanomedicine in the Development of Clinical Natural Drugs. Planta Med. 2017, 83, 366–381. [Google Scholar] [CrossRef] [Green Version]
- Piazzini, V.; Lemmi, B.; D’Ambrosio, M.; Luceri, C.; Cinci, L.; Ghelardini, C.; Bilia, A.R.; Di Cesare Mannelli, L.; Bergonzi, M.C. Nanostructured Lipid Carriers as Promising Delivery Systems for Plant Extracts: The Case of Silymarin. Appl. Sci. 2018, 8, 1163. [Google Scholar] [CrossRef] [Green Version]


| No. | Source | Bioactive Compounds | Therapeutic Potential | Ref. |
|---|---|---|---|---|
| 1. | Abelmoschus esculentus | Quercetin | Anti-inflammatory, antioxidant, hypolipidemic. | [36] |
| 2. | Ajuga iva | Naringein, apigenin-7-O-neohesperidoside. | Antioxidant, anti-inflammatory, antihypercholesterolemia. | [37] |
| 3. | Anchusa italica Retz. | Rutin, hesperidin, quercetin, kaempferol, naringenin. | Anti-inflammatory, antioxidant, anticoagulant. | [38] |
| 4. | Apples | Lutein, carotenoids, antioxidant: phlorizin, quercetin, catechin, procyanidin, epicatechin. | Antioxidant, antifungal, anti-proliferative. | [39] |
| 5. | Barley leaves | 2β-O-Glucosylisovitexin. | Antioxidant, membrane stabilizer, antitumor. | [40] |
| 6. | Broccoli | Lutein, zeaxanthin, β-carotene, flavonoids. | Antioxidant, anti-inflammatory, anticarcinogenic. | [41] |
| 7. | Carrots | Lignin, carotene. | Treat leukemia. | [42] |
| 8. | Cereal crops | Orizanol, isovitexin, cyanidine-3- O-β-D-glycopyranoside, pinoresinol. | Prevent cardiovascular diseases and cancer diseases. | [43] |
| 9. | Cocoa | Phytochemicals: methylxanthine, proantho-cyanidin, theobromin. | Antioxidant, anti-inflammatory, hypoglycemic, antiplatelet, antihypertensive. | [44] |
| 10. | Corchorus olitorius Leaf and Corchorus capsularis | Luteolin | Antioxidant, hypotensive, diuretic. | [45,46] |
| 11. | Cotton seed oil | Quercetin, rutin, kaempferol, gossypeti, heracetin, dihydroquercetin, quercetrin, isoquercetrin. | Total cholesterol, low-density lipoprotein (LDL) cholesterol, and triglycerides (TG) lowered. | [47] |
| 12. | Cymbopogon citratus (DC) Sapf. | Tannins, luteolin, apigenin. | Vasorelaxation, antioxidant, anti-inflammatory. | [48] |
| 13. | Danshen (Salvia miltiorrhiza) | Tanshinones, salvianolic acids. | Angina, ischemic stroke, hyperlipidemia, antithrombotic. | [49] |
| 14. | Dracocephalum moldavica L. | Tallianine, luteolin, apigenin, diosmetin. | Anticancer, anti-inflammatory, antioxidant. | [50] |
| 15. | Edible wild fruit | Polyphenols: procyanidin, quercetin, phenolic acid, anthocyanin, carotenoids. | Anti-inflammatory, anti-obesity, antidiabetic. | [51] |
| 16. | Ephedra herb | Epiafzelechin (flavanol), quercetin, gallocatechin, apigenin, luteolin. | Diuretic, anti-inflammatory, hypotensive, antioxidant. | [52] |
| 17. | Equisetum arvense L. (Horsetail) | Resveratrol, apigenin, quercetin. | Antioxidant, anti-inflammatory. | [53] |
| 18. | Flaxseed oil | Phytoestrogens lignans, coumestran, enterolactone, enterodial, coumestrol. | Reduce the growth rate of mammary cancer, lowered plasma LDL cholesterol. | [54] |
| 19. | Foxglove (Digitalis species) | Glycoside digoxi. | Treat heart failure, atrial fibrillation. | [55] |
| 20. | Garlic (Allium sativum) | Allicin. | Treat hypertension, hyperlipidemia, antithrombotic. | [56] |
| 21. | Ginkgo (Ginkgo biloba) | Flavonol glycosides–quercetin and catechin and terpenoids–ginkgolides and bilobalides. | Cerebral insufficiency, peripheral vascular disease, antithrombotic. | [57] |
| 22. | Ginseng (Panax species) | Triterpene saponins– ginsenosides. | Angina, hypertension, antidiabetic. | [58] |
| 23. | Grapes | Polyphenols: resveratrol, carotenoids, flavonoids. | Antioxidant, anti-inflammatory, antihypertensive, antidiabetic. | [59] |
| 24. | Hawthorn (Crataegus species) | Phenolic acids, quercetin, pyrocatechin, phlorodizin, terpenoids, lignans, steroids, organic acids, and sugars. | Heart failure, angina, hyperlipidemia. | [60] |
| 25. | Heliotropium taltalense (Phil.) | Naringenin, pinocembrin, quercetin. | Anti-inflammatory, antioxidant, vasorelaxation. | [61] |
| 26. | Lentil (Lens culinaris Medik.) | Quercetin, kaempferol. | Anticoagulant, antiplatelet. | [62] |
| 27. | Lingzhi (Ganoderma lucidum) | Polysaccharides, triterpenes, polyphenols, proteins, amino acids, and organic germanium. | Hyperlipidemia, hypertension, antidiabetic. | [63,64] |
| 28. | Moringa oleifera Lam. | Catechin, epicatechin, kaempferol, quercetin. | Antioxidant, anti-inflammatory. | [65] |
| 29. | Mustard seed | Glucosinolates, sinigrin, phenolic acids, sinapic acid, methyl ester. | Anti-inflammatory, Anticancer, antioxidant, antihyperglycemia. | [66] |
| 30. | Oats | β-Glucan, pectin, psyllium, esters of caffeic and ferulic acids. | Reduce both total cholesterol and LDL cholesterol. | [67] |
| 31. | Olives | Phenolic compounds, hydroxy-tyrosol, oleuropein, polyphenols, flavonoids, theanine, quercetin. | Antioxidant, anti-inflammatory, antihypertensive, anti-obesity. | [68] |
| 32. | Onions | Quercetin, myricetin. | Treat obesity, coagulation, inflammation, atherosclerosis, hyperlipidemia. | [69] |
| 33. | Peanuts | Taxifolin, resveratrol. | Triacylglycerol (TAG) reduced. | [70] |
| 34. | Polygonum minus (Persicaria minor) | Myricetin, quercetin, methyl-flavonol. | Antioxidant, anti-inflammatory. | [71] |
| 35. | Psyllium seed | β-glucan, pectin, psyllium, soluble dietary fibers. | Antioxidant, laxative. | [72] |
| 36. | Red wine | Resveratrol. | Anticancer activity. | [73] |
| 37. | Rice bran oil | Plant sterols, sitostanol, stigmasterol, campesterol. | Hypocholesterolemic effect, total cholesterol and LDL cholesterol reduced. | [74] |
| 38. | Soybeans | Genistein, daidzein, isoflavones. | Reduced LDL-cholesterol level. | [75] |
| 39. | Thai Perilla frutescens | Cyanidins, luteolin, phenolic acids. | Anti-inflammatory, antioxidant. | [76] |
| 40. | Tomato | Phenols: phenolic acid, flavonoids, carotenoids. | Antioxidant, anti-inflammatory, antiplatelet, antihypertensive. | [77] |
| 41. | Trichosanthes kirilowii | Luteolin. | Hypolipidemic, antioxidant, antiatherosclerotic. | [78] |
| 42. | Wild rice | Phytic acid, luteolin glycoside, p- hydroxy acetophenone glycoside, 3,4,5-trimethoxycinnamin acid. | Health-promoting. | [79] |
| Flavonoids with Main Features | Natural Sources | Bountiful Health Benefits |
|---|---|---|
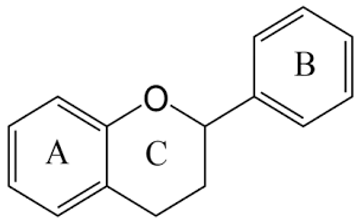
 | Berries, spices, leeks, ginger, herbs, grapefruit, carrot, grapes, apple, onion, broccoli, cabbage, kale, tomato, lemon, parsley, buckwheat, legumes, coffee, tea. |
|
| No. | Type of Flavonoid | Major Food Flavonoids | Functional Unit of B-Ring | Unsaturation in B-Ring | Common Plant Source | Therapeutic Effect | Ref. |
|---|---|---|---|---|---|---|---|
| 1. | Anthocyanins | Cyanidin, delphinidin, malvidin, peonidin. | 3-Hydroxy. | 1-2, 3-4 Double bond. | Strawberries, red wine, and blueberries. |
| [85] |
| 2. | Flavan-3-ols [flavanols] | Cathechin, gallocatechin, epicatechin, epiallocatechin-3-gallate. | 3-Hydroxy, 3-O-gallate. | None. | Red wine, blueberries, apples, chocolate, pears, cocoa, tea, and grape. |
| [86] |
| 3. | Flavanones | Eriodictyol, naringenin, hesperetin. | 4-Oxo. | None. | Herbal tea, juice, fruit peels, and citrus fruit. |
| [87] |
| 4. | Flavones | Apigenin, luteolin, tangeretin, baicalein. | 4-Oxo. | 2-3 Double bond. | Herbal tea, garlic, celery, chamomile tea, and green peppers. |
| [88] |
| 5. | Flavonols | Myricetin, quercetin, isorhamnetin, kaempferol. | 3-Hydroxy, 4-Oxo. | 2-3 Double bond. | Red wine, kale, broccoli, cherry, tomato, garlic, onions, tea, strawberries, beans, Spinach. |
| [89] |
| 6. | Isoflavones | Daidzein, genistein, glycitein, biochaninA, glycitein. | 4-Oxo. | 2-3 Double bond. | Peanuts, legumes, and soy products. |
| [90] |
| Chemical Structure and Main Features | Sources | Bountiful Health Benefits |
|---|---|---|
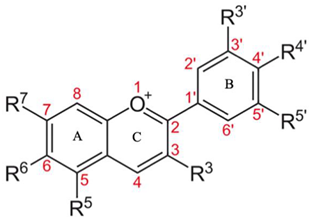 | Acai, blackcurrant, blueberry, bilberry cherry, red grape, and purple corn. |
|
| Chemical Structure and Main Features | Natural Sources | Bountiful Health Benefits |
|---|---|---|

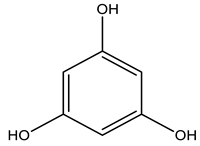 | Teas, coffee, pomegranates, persimmons, cranberries, strawberries, blueberries, grapes, red wine, cinnamon, vanilla, cloves, thyme, and oak gallnuts (tannin content of 50–70%). |
|
| Chemical Structure and Main Features | Sources | Bountiful Health Benefits |
|---|---|---|
 |
|
|
| Chemical Structure and Main Features | Sources | Bountiful Health Benefits |
|---|---|---|
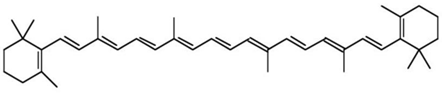
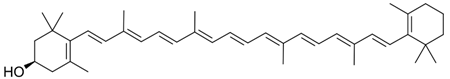 | Apricots, carrots, cilantro, collard, cantaloupes, mango, plum, thyme, turnip, squash, kale. |
|
| No | Types of Carotenoids | Composition | Source | Properties | Health Benefits | Ref. |
|---|---|---|---|---|---|---|
| 1 | α-Carotene | C40H56, carotene with β-ionone ring and α-ionone ring. | Carrots, pumpkin, broccoli, spinach, avocado, sweet potato, squash. | This is second most common carotenoid. |
| [100] |
| 2 | β-Carotene | C40H56, group consisting of isoprene units. | Carrots, apricots, mango, red pepper, greens (kale, spinach, broccoli). | This is the major and natural carotenoid present. |
| [101,102] |
| 3 | β-Cryptoxanthin | C40H56O, isoprene unit with a hydroxyl unit. | Orange, peaches, tangerines, papaya, egg yolk, butter, apples. | Natural carotenoid pigments. |
| [103] |
| 4 | Lutein and Zeaxanthin | C40H56O2, they have similar isomers but differ in double bond location. Lutein shows the presence of three chiral canters, while zeaxanthin has two. | Kale, spinach, broccoli, sprouts, lettuce, egg yolk, yellow corns, and parsley. | These are dietary oxygenated carotenoids. |
| [104,105] |
| 5 | Lycopene | C40H56, in this a tetraterpene, is arranged with eight isoprene units of carbon and hydrogen. | Tomato, watermelon, grape, papaya, guava, rose. | These are hydrophilic, acyclic carotenoids with eleven conjugated double bonds. |
| [106] |
| Chemical Structure and Main Features | Sources | Bountiful Health Benefits |
|---|---|---|
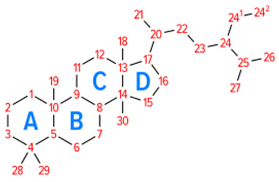
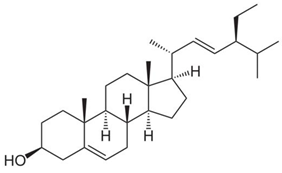 | Vegetable oils, nuts, legumes, grains, cereals, wood pulp, margarine, milk, yogurt, soybean oil, and leaves. | Reduce the level of the LDL cholesterol in blood. |
| Chemical Structure and Main Features | Sources | Bountiful Health Benefits |
|---|---|---|
 | Cruciferous vegetables: wasabia japonica, cabbage, garden cress, kale, broccoli, and watercress. |
|
| Bioactive Compound | Nanoformulation | In Vitro/In Vivo Experimental Model | Main Results | Ref. |
|---|---|---|---|---|
| Curcumin | Chitosan NPs | In vitro/Ehrlich ascites carcinoma and in vivo animal model (albino mice) | Protection against myocardial injury and cardiac function, ameliorates EAC-induced cardiotoxicity. | [162] |
| PEG-PDLLA (polyethylene glycol methyl ether-block-poly (D, L lactide) NPs | In vitro/cardiomyocytes exposed to palmitate | Inhibition of cell apoptosis and NADPH-mediated oxidative stress; protective effect possibly mediated by endoplasmic reticulum stress-related signaling pathway. | [163] | |
| Nano emulsion/glyceryl monooleate oil phase | In vitro/HMG-CoA reductase assay | Increased not only the HMGR inhibition (showing antihypercholesterolemic effect) but also angiotensin-converting enzyme (antihypertensive effect). | [164] | |
| Curcumin + resveratrol | Pluronic® F127 micelles | In vitro/embryonic rat cardiomyocytes | Cardioprotection, reduction in apoptosis and ROS of cardiomyocytes treated with doxorubicin. | [165] |
| Resveratrol NPs | Solid–lipid NPs/glycerol monostearate oil phase | In vivo pharmacodynamic study/male mice | Protecting the myocardium, improving the cardiomyocyte calcium cycling, inhibiting of doxorubicin cardiotoxicity, inhibiting the production of reactive oxygen species. | [166] |
| Albumin NPs | In vivo/male Sprague–Dawley rats | Improved neurological score and decreased infarct volume at 24 h after administration in a dose-dependent manner; significantly attenuated oxidative stress due to prolonged circulation in blood and sustained release pattern. | [167] | |
| Quercetin | PLGA NPs | In vitro/H9c2 cells, a surrogate model of cardiac cells | Improves cardioprotection during hypoxia–reoxygenation injury through the preservation of mitochondrial function; superior protection capability of PLGA–quercetin NP with respect to free quercetin. | [168] |
| Mesoporous silica nanoparticles | In vivo rat model of myocardial ischemia reperfusion | Improves the apoptosis degree and oxidative stress level of myocardial cells by regulating the JAK2/STAT3 signaling pathway, promoting the recovery of cardiac blood flow. | [169] | |
| Coenzyme Q10 | Nano emulsion/soybean oil and egg lecithin surfactant | In vitro/cardiomyocytes and Fibroblasts model | Providing multiple molecular mechanisms of cardioprotection during doxorubicin and trastuzumab treatments; anti-inflammatory activities modulating the heart microenvironment. | [170] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sindhu, R.K.; Goyal, A.; Algın Yapar, E.; Cavalu, S. Bioactive Compounds and Nanodelivery Perspectives for Treatment of Cardiovascular Diseases. Appl. Sci. 2021, 11, 11031. https://doi.org/10.3390/app112211031
Sindhu RK, Goyal A, Algın Yapar E, Cavalu S. Bioactive Compounds and Nanodelivery Perspectives for Treatment of Cardiovascular Diseases. Applied Sciences. 2021; 11(22):11031. https://doi.org/10.3390/app112211031
Chicago/Turabian StyleSindhu, Rakesh K., Annima Goyal, Evren Algın Yapar, and Simona Cavalu. 2021. "Bioactive Compounds and Nanodelivery Perspectives for Treatment of Cardiovascular Diseases" Applied Sciences 11, no. 22: 11031. https://doi.org/10.3390/app112211031
APA StyleSindhu, R. K., Goyal, A., Algın Yapar, E., & Cavalu, S. (2021). Bioactive Compounds and Nanodelivery Perspectives for Treatment of Cardiovascular Diseases. Applied Sciences, 11(22), 11031. https://doi.org/10.3390/app112211031