In Situ Generation of Hydrogen Peroxide Using Polymetallic-Doped g-C3N4 for Pollutant Removal
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Catalysts Preparation
2.3. Characterization Methods
2.4. Performances Tests
3. Results and Discussion
3.1. Morphology and Structure
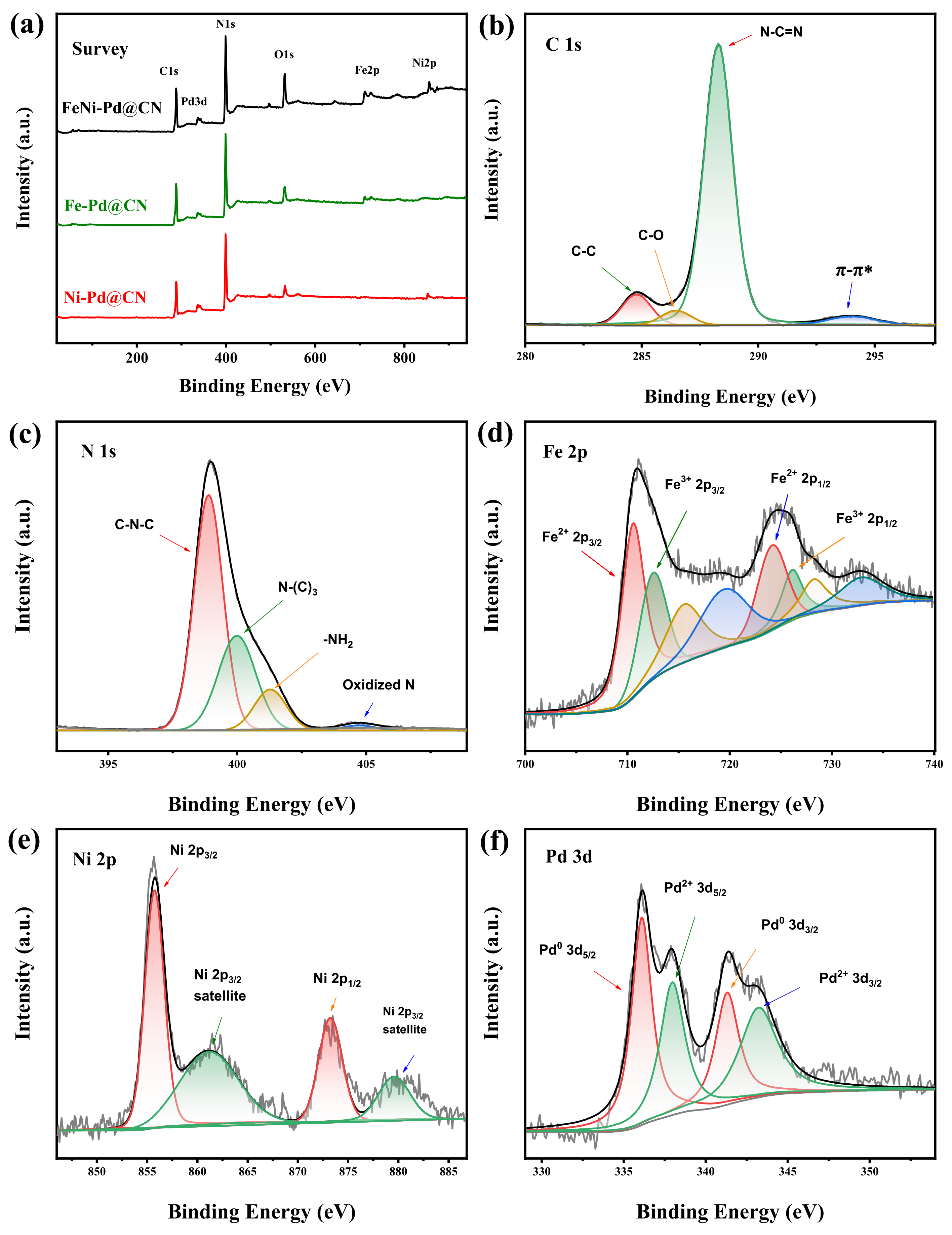
3.2. Chemical Composition of the Catalyst
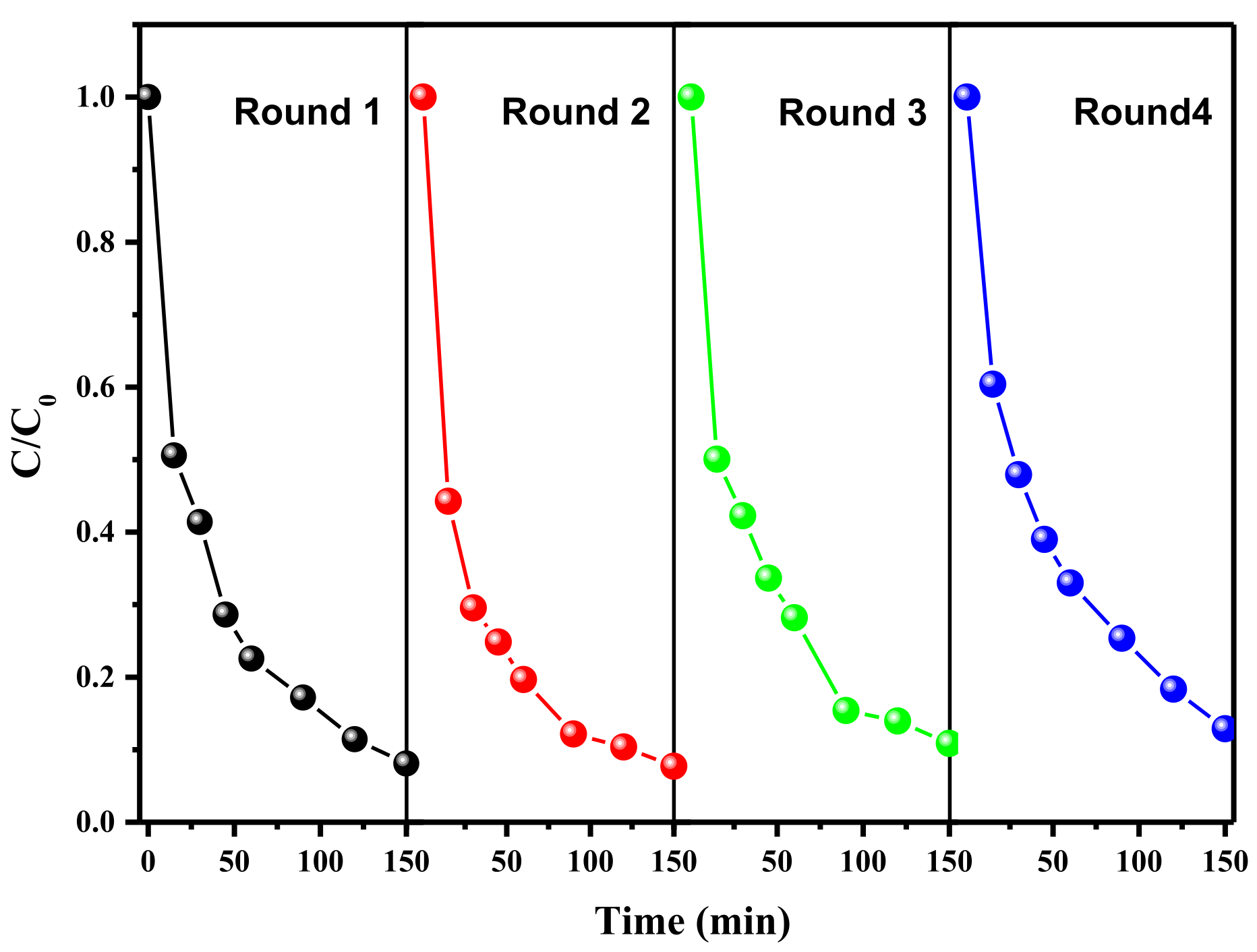
3.3. The Catalytic Performances of Catalysts
3.4. Enhanced H2O2 Production
3.5. Reaction Mechanism
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gągol, M.; Przyjazny, A.; Boczkaj, G. Wastewater treatment by means of advanced oxidation processes based on cavitation—A review. Chem. Eng. J. 2018, 338, 599–627. [Google Scholar] [CrossRef]
- Collivignarelli, M.C.; Pedrazzani, R.; Sorlini, S.; Abbà, A.; Bertanza, G. H2O2 Based Oxidation Processes for the Treatment of Real High Strength Aqueous Wastes. Sustainability 2017, 9, 244. [Google Scholar] [CrossRef] [Green Version]
- Pouran, S.R.; Aziz, A.A.; Daud, W.M.A.W. Review on the main advances in photo-Fenton oxidation system for recalcitrant wastewaters. J. Ind. Eng. Chem. 2015, 21, 53–69. [Google Scholar] [CrossRef]
- Wang, N.; Zheng, T.; Zhang, G.; Wang, P. A review on Fenton-like processes for organic wastewater treatment. J. Environ. Chem. Eng. 2016, 4, 762–787. [Google Scholar] [CrossRef] [Green Version]
- Liu, Q.; Yang, Y.; Lv, X.; Ding, Y.; Zhang, Y.; Jing, J.; Xu, C. One-step synthesis of uniform nanoparticles of porphyrin functionalized ceria with promising peroxidase mimetics for H2O2 and glucose colorimetric detection. Sens. Actuators B Chem. 2017, 240, 726–734. [Google Scholar] [CrossRef]
- Zárate-Guzmán, A.I.; González-Gutiérrez, L.V.; Godínez, L.A.; Medel-Reyes, A.; Carrasco-Marín, F.; Romero-Cano, L.A. Towards understanding of heterogeneous Fenton reaction using carbon-Fe catalysts coupled to in-situ H2O2 electro-generation as clean technology for wastewater treatment. Chemosphere 2019, 224, 698–706. [Google Scholar] [CrossRef]
- William, R.S. Cleaner industrial processes using hydrogen peroxide. Pure Appl. Chem. 2000, 72, 1289–1304. [Google Scholar]
- Li, X.F.; Liu, W.P.; Ma, J.Q.; Wen, Y.Z.; Wu, Z.C. High catalytic activity of magnetic FeOx/NiOy/SBA-15: The role of Ni in the bimetallic oxides at the nanometer level. Appl. Catal. B Environ. 2015, 179, 239–248. [Google Scholar] [CrossRef]
- Shaegh, S.A.M.; Nguyen, N.-T.; Ehteshami, S.M.M.; Chan, S.H. A membraneless hydrogen peroxide fuel cell using Prussian Blue as cathode material. Energy Environ. Sci. 2012, 5, 8225–8228. [Google Scholar] [CrossRef] [Green Version]
- Samanta, C. Direct synthesis of hydrogen peroxide from hydrogen and oxygen: An overview of recent developments in the process. Appl. Catal. A Gen. 2008, 350, 133–149. [Google Scholar] [CrossRef]
- Demirci, U.B.; Miele, P. Chemical hydrogen storage: ‘material’ gravimetric capacity versus ‘system’ gravimetric capacity. Energy Environ. Sci. 2011, 4, 3334–3341. [Google Scholar] [CrossRef]
- Kopinke, F.-D.; Mackenzie, K.; Koehler, R.; Georgi, A. Alternative sources of hydrogen for hydrodechlorination of chlorinated organic compounds in water on Pd catalysts. Appl. Catal. A Gen. 2004, 271, 119–128. [Google Scholar] [CrossRef]
- Yalfani, M.S.; Contreras, S.; Medina, F.; Sueiras, J. Direct generation of hydrogen peroxide from formic acid and O2 using heterogeneous Pd/γ-Al2O3 catalysts. Chem. Commun. 2008, 3885–3887. [Google Scholar] [CrossRef]
- Tan, N.; Yang, Z.; Gong, X.; Wang, Z.-R.; Fu, T.; Liu, Y. In situ generation of H2O2 using MWCNT-Al/O2 system and possible application for glyphosate degradation. Sci. Total Environ. 2018, 650, 2567–2576. [Google Scholar] [CrossRef]
- Thomas, A.; Fischer, A.; Goettmann, F.; Antonietti, M.; Müller, J.-O.; Schlögl, R.; Carlsson, J.M. Graphitic carbon nitride materials: Variation of structure and morphology and their use as metal-free catalysts. J. Mater. Chem. 2008, 18, 4893–4908. [Google Scholar] [CrossRef] [Green Version]
- Ge, L.; Peng, Z.; Wang, W.; Tan, F.; Wang, X.; Su, B.; Qiao, X.; Wong, P.K. g-C3N4/MgO nanosheets: Light-independent, metal-poisoning-free catalysts for the activation of hydrogen peroxide to degrade organics. J. Mater. Chem. A 2018, 6, 16421–16429. [Google Scholar] [CrossRef]
- Sun, B.-W.; Li, H.-J.; Yu, H.-Y.; Qian, D.-J.; Chen, M. In situ synthesis of polymetallic Co-doped g-C3N4 photocatalyst with increased defect sites and superior charge carrier properties. Carbon 2017, 117, 1–11. [Google Scholar] [CrossRef]
- Ghosh, D.; Periyasamy, G.; Pandey, B.; Pati, S.K. Computational studies on magnetism and the optical properties of transition metal embedded graphitic carbon nitride sheets. J. Mater. Chem. C 2014, 2, 7943–7951. [Google Scholar] [CrossRef]
- Jose, D.; Jagirdar, B.R. Synthesis and characterization of Pd(0), PdS, and Pd@PdO core–shell nanoparticles by solventless thermolysis of a Pd–thiolate cluster. J. Solid State Chem. 2010, 183, 2059–2067. [Google Scholar] [CrossRef]
- Feng, Y.; Liao, C.; Kong, L.; Wu, D.; Liu, Y.; Lee, P.-H.; Shih, K. Facile synthesis of highly reactive and stable Fe-doped g-C3N4 composites for peroxymonosulfate activation: A novel nonradical oxidation process. J. Hazard. Mater. 2018, 354, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Niu, P.; Zhang, L.; Liu, G.; Cheng, H.-M. Graphene-Like Carbon Nitride Nanosheets for Improved Photocatalytic Activities. Adv. Funct. Mater. 2012, 22, 4763–4770. [Google Scholar] [CrossRef]
- Ma, T.; Tang, Y.; Dai, S.; Qiao, S.Z. Proton-Functionalized Two-Dimensional Graphitic Carbon Nitride Nanosheet: An Excellent Metal-/Label-Free Biosensing Platform. Small 2014, 10, 2382–2389. [Google Scholar] [CrossRef] [PubMed]
- Laokul, P.; Amornkitbamrung, V.; Seraphin, S.; Maensiri, S. Characterization and magnetic properties of nanocrystalline CuFe2O4, NiFe2O4, ZnFe2O4 powders prepared by the Aloe vera extract solution. Curr. Appl. Phys. 2011, 11, 101–108. [Google Scholar] [CrossRef]
- Bertolini, J.C.; Delichere, P.; Khanra, B.C.; Massardier, J.; Noupa, C.; Tardy, B. Electronic properties of supported Pd aggregates in relation with their reactivity for 1,3-butadiene hydrogenation. Catal. Lett. 1990, 6, 215–223. [Google Scholar] [CrossRef]
- Lei, W.; Portehault, D.; Dimova, R.; Antonietti, M. Boron Carbon Nitride Nanostructures from Salt Melts: Tunable Water-Soluble Phosphors. J. Am. Chem. Soc. 2011, 133, 7121–7127. [Google Scholar] [CrossRef]
- Yu, H.; Shi, R.; Zhao, Y.; Bian, T.; Zhao, Y.; Zhou, C.; Waterhouse, G.I.N.; Wu, L.Z.; Tung, C.H.; Zhang, T. Alkali-Assisted Synthesis of Nitrogen Deficient Graphitic Carbon Nitride with Tunable Band Structures for Efficient Visible-Light-Driven Hydrogen Evolution. Adv. Mater. 2017, 29, 1605148. [Google Scholar] [CrossRef]
- Ungureanu, A.; Dragoi, B.; Chirieac, A.; Ciotonea, C.; Royer, S.; Duprez, D.; Mamede, A.S.; Dumitriu, E. Composition-Dependent Morphostructural Properties of Ni-Cu Oxide Nanoparticles Confined within the Channels of Ordered Mesoporous SBA-15 Silica. ACS Appl. Mater. Interfaces 2013, 5, 3010–3025. [Google Scholar] [CrossRef]
- Berríos, C.; Cardenas-Jiron, G.; Marco, J.; Gutiérrez, C.; Ureta-Zañartu, M.S. Theoretical and Spectroscopic Study of Nickel(II) Porphyrin Derivatives. J. Phys. Chem. A 2007, 111, 2706–2714. [Google Scholar] [CrossRef]
- Mansour, A.N.; Melendres, C.A. Characterization of Slightly Hydrated Ni(OH)2 by XPS. Surf. Sci. Spectra 1994, 3, 247–254. [Google Scholar] [CrossRef]
- Yao, Y.; Chen, H.; Lian, C.; Wei, F.; Zhang, D.; Wu, G.; Chen, B.; Wang, S. Fe, Co, Ni nanocrystals encapsulated in nitrogen-doped carbon nanotubes as Fenton-like catalysts for organic pollutant removal. J. Hazard. Mater. 2016, 314, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.-C.; Feng, J.-J.; Lin, X.-X.; Zhang, L.; Yuan, J.; Zhang, Q.-L.; Wang, A.-J. One-step hydrothermal synthesis of three-dimensional nitrogen-doped reduced graphene oxide hydrogels anchored PtPd alloyed nanoparticles for ethylene glycol oxidation and hydrogen evolution reactions. Electrochim. Acta 2018, 293, 504–513. [Google Scholar] [CrossRef]
- Hong, W.; Shang, C.; Wang, J.; Wang, E. Bimetallic PdPt nanowire networks with enhanced electrocatalytic activity for ethylene glycol and glycerol oxidation. Energy Environ. Sci. 2015, 8, 2910–2915. [Google Scholar] [CrossRef]
- Qin, X.; Li, H.; Xie, S.; Li, K.; Jiang, T.-W.; Ma, X.-Y.; Jiang, K.; Zhang, Q.; Terasaki, O.; Wu, Z.; et al. Mechanistic Analysis-Guided Pd-Based Catalysts for Efficient Hydrogen Production from Formic Acid Dehydrogenation. ACS Catal. 2020, 10, 3921–3932. [Google Scholar] [CrossRef]
- Jiang, B.; Zhang, X.-G.; Jiang, K.; Wu, D.-Y.; Cai, W.-B. Boosting Formate Production in Electrocatalytic CO2 Reduction over Wide Potential Window on Pd Surfaces. J. Am. Chem. Soc. 2018, 140, 2880–2889. [Google Scholar] [CrossRef]
- Tian, P.; Ouyang, L.; Xu, X.; Xu, J.; Han, Y.-F. Density functional theory study of direct synthesis of H2O2 from H2 and O2 on Pd(111), Pd(100), and Pd(110) surfaces. Chin. J. Catal. 2013, 34, 1002–1012. [Google Scholar] [CrossRef]
- Bai, N.B. Evaluation of the energy of activation of chemisorption for H2 and O2 on transition d-metals. J. Chem. Ind. Eng. 1981, 3, 217–223. [Google Scholar]
- Xu, Y.; Ge, F.; Xie, M.; Huang, S.; Qian, J.; Wang, H.; He, M.; Xu, H.; Li, H. Fabrication of magnetic BaFe12O19/Ag3PO4 composites with an in situ photo-Fenton-like reaction for enhancing reactive oxygen species under visible light irradiation. Catal. Sci. Technol. 2019, 9, 2563–2570. [Google Scholar] [CrossRef]
- Li, C.; Wu, J.; Peng, W.; Fang, Z.; Liu, J. Peroxymonosulfate activation for efficient sulfamethoxazole degradation by Fe3O4/β-FeOOH nanocomposites: Coexistence of radical and non-radical reactions. Chem. Eng. J. 2019, 356, 904–914. [Google Scholar] [CrossRef]
- de Souza, W.F.; Guimarães, I.R.; Oliveira, L.C.; Giroto, A.S.; Guerreiro, M.C.; Silva, C.L. Effect of Ni incorporation into goethite in the catalytic activity for the oxidation of nitrogen compounds in petroleum. Appl. Catal. A Gen. 2010, 381, 36–41. [Google Scholar] [CrossRef]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, L.; Ma, J.; Guo, Q.; Liu, L.; Shou, J.; Sun, A.; Zhao, L. In Situ Generation of Hydrogen Peroxide Using Polymetallic-Doped g-C3N4 for Pollutant Removal. Appl. Sci. 2021, 11, 10797. https://doi.org/10.3390/app112210797
Wang L, Ma J, Guo Q, Liu L, Shou J, Sun A, Zhao L. In Situ Generation of Hydrogen Peroxide Using Polymetallic-Doped g-C3N4 for Pollutant Removal. Applied Sciences. 2021; 11(22):10797. https://doi.org/10.3390/app112210797
Chicago/Turabian StyleWang, Liyan, Jianqing Ma, Qianhui Guo, Liang Liu, Jiangnan Shou, Aojie Sun, and Liaoyuan Zhao. 2021. "In Situ Generation of Hydrogen Peroxide Using Polymetallic-Doped g-C3N4 for Pollutant Removal" Applied Sciences 11, no. 22: 10797. https://doi.org/10.3390/app112210797
APA StyleWang, L., Ma, J., Guo, Q., Liu, L., Shou, J., Sun, A., & Zhao, L. (2021). In Situ Generation of Hydrogen Peroxide Using Polymetallic-Doped g-C3N4 for Pollutant Removal. Applied Sciences, 11(22), 10797. https://doi.org/10.3390/app112210797




