Bacterial Adhesion to Grade 4 and Grade 5 Turned and Mildly Acid-Etched Titanium Implant Surfaces: An In Vitro and Ex Vivo Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Products and Reagents
2.2. Specimen Preparation
2.3. Surface Characterization
2.3.1. 3D Surface Profiling
2.3.2. SEM-EDS Analysis
2.4. In Vitro Bacterial Adhesion
2.4.1. Bacterial Culture
2.4.2. ESEM Analysis
2.4.3. Determination of Bacterial Adhesion
2.5. Ex Vivo Bacterial Adhesion
2.6. Statistical Analysis
3. Results
3.1. Surface Characterization of Ti Samples
3.1.1. SEM/EDS Analysis
3.1.2. 3D Surface Profiling
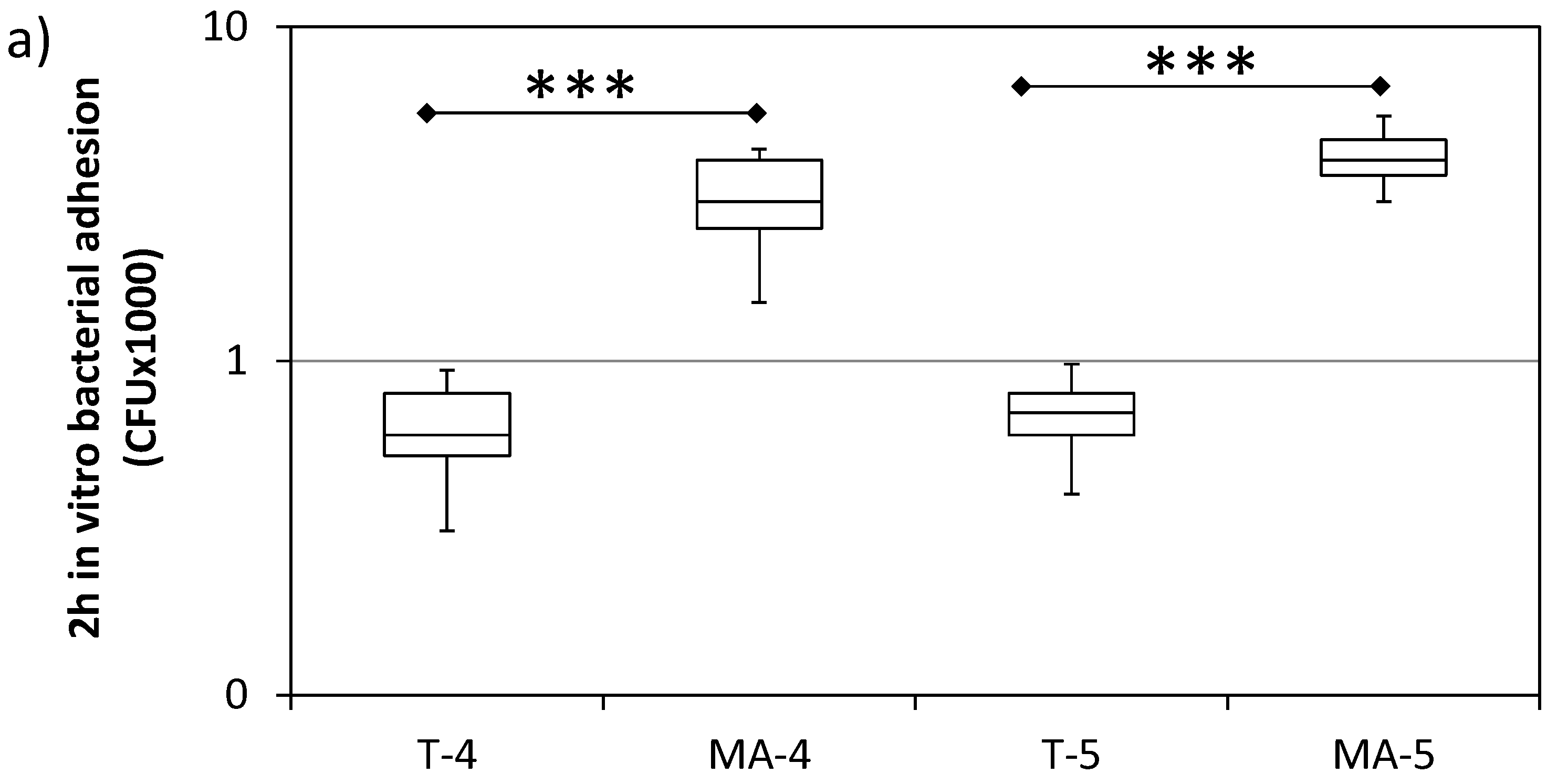
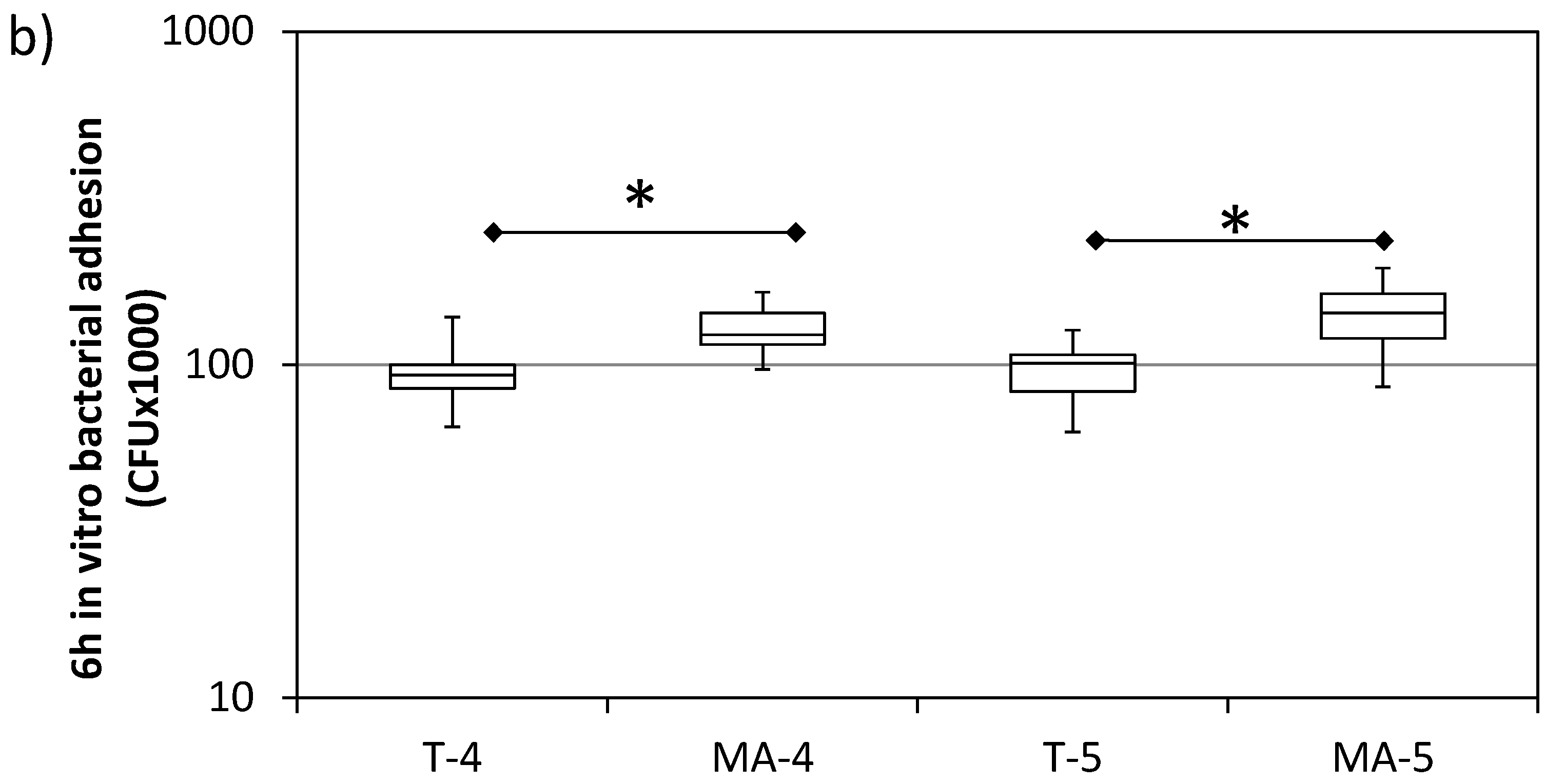
3.2. Bacterial Adhesion In Vitro
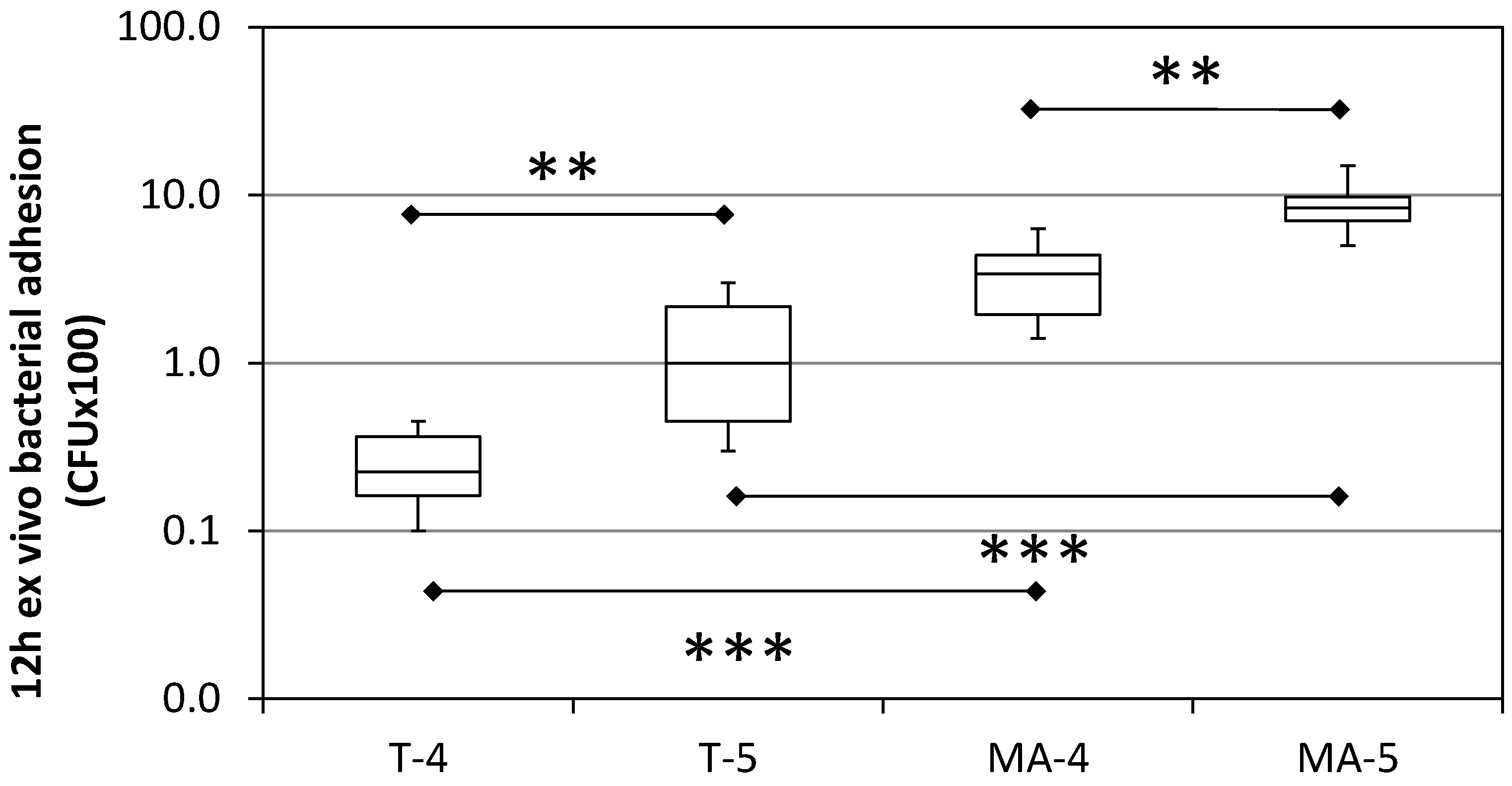
3.3. Ex Vivo Bacterial Adhesion
4. Discussion
5. Conclusions
- Bacterial adhesion, particularly when tested ex vivo, was shown to be sensitive to both Ti surface topography and composition. MA surfaces were shown to be more attractive for bacteria than T surfaces, although having similar roughness values. Even slight differences in the surface topography of tested surfaces were found to have a significant effect on the bacterial adhesion.
- Such findings could have possible implications on peri-implant tissue health maintenance. Turned surfaces, especially grade 4 Ti ones, could be more advantageously used for transmucosal implant parts and components, whereas mildly acid-etched surfaces, especially grade 5 Ti ones, should be kept far from the oral environment, remaining confined at the endosseous level, being more prone to bacterial colonization.
- Further studies are needed to investigate the effect of chemical and topographic characteristics of implant surfaces on bacterial adhesion and peri-implant tissue health. In order to obtain a more accurate topographic surface characterization, the use of high resolution equipment, 3D methods, multiple parameters and filtering procedures is recommended, as well as the use of ex vivo experimental models to increase the clinical relevance of the obtained results.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Howe, M.-S.; Keys, W.; Richards, D. Long-term (10-year) dental implant survival: A systematic review and sensitivity meta-analysis. J. Dent. 2019, 84, 9–21. [Google Scholar] [CrossRef]
- Albrektsson, T.; Brånemark, P.-I.; Hansson, H.-A.; Lindström, J. Osseointegrated Titanium Implants:Requirements for Ensuring a Long-Lasting, Direct Bone-to-Implant Anchorage in Man. Acta Orthop. Scand. 1981, 52, 155–170. [Google Scholar] [CrossRef] [Green Version]
- Quirynen, M.; De Soete, M.; Van Steenberghe, D. Infectious risks for oral implants: A review of the literature. Clin. Oral Implant Res. 2002, 13, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Wennerberg, A.; Albrektsson, T. Effects of titanium surface topography on bone integration: A systematic review. Clin. Oral Implant Res. 2009, 20, 172–184. [Google Scholar] [CrossRef]
- Annunziata, M.; Guida, L. The Effect of Titanium Surface Modifications on Dental Implant Osseointegration. Biomater. Oral Craniomaxillofacial Appl. 2015, 17, 62–77. [Google Scholar]
- Teughels, W.; Van Assche, N.; Sliepen, I.; Quirynen, M. Effect of material characteristics and/or surface topography on biofilm development. Clin. Oral Implant Res. 2006, 17, 68–81. [Google Scholar] [CrossRef] [PubMed]
- Subramani, K.; Jung, R.E.; Molenberg, A.; Hämmerle, C.H.F. Biofilm on dental implants: A review of the literature. Int. J. Oral Maxillofac. Implant 2009, 24, 616–626. [Google Scholar] [CrossRef]
- Renvert, S.; Quirynen, M. Risk indicators for peri-implantitis. A narrative review. Clin. Oral Implant Res. 2015, 26, 15–44. [Google Scholar] [CrossRef]
- Barone, A.; Orlando, B.; Cingano, L.; Marconcini, S.; Derchi, G.; Covani, U. A randomized clinical trial to evaluate and compare implants placed in augmented versus non-augmented extraction sockets: 3-year results. J. Periodontol. 2012, 83, 836–846. [Google Scholar] [CrossRef]
- Schwarz, F.; Derks, J.; Monje, A. Peri-implantitis. J. Periodontol. 2018, 89, S267–S290. [Google Scholar] [CrossRef]
- McCracken, M. Dental Implant Materials: Commercially Pure Titanium and Titanium Alloys. J. Prosthodont. 1999, 8, 40–43. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Chen, S.; Tsoi, J.K.; Matinlinna, J.P. Binary titanium alloys as dental implant materials—A review. Regen. Biomater. 2017, 4, 315–323. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elias, C.N.; Fernandes, D.; de Souza, F.M.; Monteiro, E.D.S.; de Biasi, R.S. Mechanical and clinical properties of titanium and titanium-based alloys (Ti G2, Ti G4 cold worked nanostructured and Ti G5) for biomedical applications. J. Mater. Res. Technol. 2019, 8, 1060–1069. [Google Scholar] [CrossRef]
- Barão, V.A.R.; Yoon, C.J.; Mathew, M.T. Attachment of Porphyromonas gingivalis to corroded commercially pure titanium and titanium-aluminum-vanadium alloy. J. Periodontol. 2014, 85, 1275–1282. [Google Scholar] [CrossRef]
- Di Giulio, M.; Traini, T.; Sinjari, B. Porphyromonas gingivalis biofilm formation in different titanium surfaces, an in vitro study. Clin. Oral. Implants Res. 2016, 27, 918–925. [Google Scholar] [CrossRef]
- Wennerberg, A.; Albrektsson, T. Suggested guidelines for the topographic evaluation of implant surfaces. Int. J. Oral Maxillofac. Implant 2000, 15, 331–344. [Google Scholar]
- De Bruyn, H.; Christiaens, V.; Doornewaard, R. Implant surface roughness and patient factors on long-term peri-implant bone loss. Periodontol. 2000 2017, 73, 218–227. [Google Scholar] [CrossRef]
- Bürgers, R.; Gerlach, T.; Hahnel, S. In vivo and in vitro biofilm formation on two different titanium implant surfaces. Clin. Oral Implants Res. 2010, 21, 156–164. [Google Scholar] [CrossRef]
- Abranches, J.; Zeng, L.; Kajfasz, J.K.; Palmer, S.R.; Chakraborty, B.; Wen, Z.T.; Richards, V.; Brady, L.J.; Lemos, J. Biology of Oral Streptococci. Microbiol. Spectr. 2018, 6. [Google Scholar] [CrossRef]
- Bjerkan, G.; Witsø, E.; Bergh, K. Sonication is superior to scraping for retrieval of bacteria in biofilm on titanium and steel surfaces in vitro. Acta Orthop. 2009, 80, 245–250. [Google Scholar] [CrossRef] [Green Version]
- Größner-Schreiber, B.; Griepentrog, M.; Haustein, I.; Müller, W.-D.; Briedigkeit, H.; Göbel, U.B.; Lange, K.-P. Plaque formation on surface modified dental implants. Clin. Oral Implant Res. 2001, 12, 543–551. [Google Scholar] [CrossRef] [PubMed]
- John, G.; Becker, J.; Schwarz, F. Modified Implant Surface with Slower and Less Initial Biofilm Formation. Clin. Implant Dent. Relat. Res. 2013, 17, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Bollen, C.M.L.; Papaioanno, W.; Van Eldere, J.; Schepers, E.; Quirynen, M.; Van Steenberghe, D. The influence of abutment surface roughness on plaque accumulation and peri-implant mucositis. Clin. Oral Implant Res. 1996, 7, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Bollenl, C.M.; Lambrechts, P.; Quirynen, M. Comparison of surface roughness of oral hard materials to the threshold surface roughness for bacterial plaque retention: A review of the literature. Dent. Mater. 1997, 13, 258–269. [Google Scholar] [CrossRef]
- Annunziata, M.; Rizzo, A.; Leone, C.; Mangano, C.; Mazzola, N.; Nastri, L.; Papale, F.; Rullo, F.; Guida, L. Bacterial adhesion to direct laser metal formed and mildly acid etched implant surfaces. Surf. Coat. Technol. 2017, 328, 390–397. [Google Scholar] [CrossRef]
- Canullo, L.; Annunziata, M.; Pesce, P.; Tommasato, G.; Nastri, L.; Guida, L. Influence of abutment material and modifications on peri-implant soft-tissue attachment: A systematic review and meta-analysis of histological animal studies. J. Prosthet. Dent. 2021, 125, 426–436. [Google Scholar] [CrossRef]
- Schwarz, F.; Mihatovic, I.; Golubovic, V.; Eick, S.; Iglhaut, T.; Becker, J. Experimental peri-implant mucositis at different implant surfaces. J. Clin. Periodontol. 2014, 41, 513–520. [Google Scholar] [CrossRef]
- Quirynen, M.; Van Assche, N. RCT comparing minimally with moderately rough implants. Part 2: Microbial observations. Clin. Oral Implant Res. 2011, 23, 625–634. [Google Scholar] [CrossRef] [PubMed]
- Renvert, S.; Lindahl, C.; Persson, G.R. The incidence of peri-implantitis for two different implant systems over a period of thirteen years. J. Clin. Periodontol. 2012, 39, 1191–1197. [Google Scholar] [CrossRef] [PubMed]
- Doornewaard, R.; Christiaens, V.; De Bruyn, H. Long-Term Effect of Surface Roughness and Patients’ Factors on Crestal Bone Loss at Dental Implants. A Systematic Review and Meta-Analysis. Clin. Implant Dent. Relat. Res. 2017, 19, 372–399. [Google Scholar] [CrossRef]
- Lin, C.-Y.; Chen, Z.; Pan, W.-L.; Wang, H.-L. Is History of Periodontal Disease Still a Negative Risk Indicator for Peri-implant Health under Supportive Post-Implant Treatment Coverage? A Systematic Review and Meta-analysis. Int. J. Oral Maxillofac. Implant 2020, 35, 52–62. [Google Scholar] [CrossRef]
- Figuero, E.; Graziani, F.; Sanz, I.; Herrera, D.; Sanz, M. Management of peri-implant mucositis and peri-implantitis. Periodontol. 2000 2014, 66, 255–273. [Google Scholar] [CrossRef]
- Schwarz, F.; Schmucker, A.; Becker, J.C. Efficacy of alternative or adjunctive measures to conventional treatment of peri-implant mucositis and peri-implantitis: A systematic review and meta-analysis. Int. J. Implant Dent. 2015, 1, 1–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barrak, I.; Baráth, Z.; Tián, T.; Venkei, A.; Gajdács, M.; Urbán, E.; Stájer, A. Effects of different decontaminating solutions used for the treatment of peri-implantitis on the growth of Porphyromonas gingivalis-an in vitro study. Acta Microbiol. Immunol. Hung. 2021, 68, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Stájer, A.; Kajári, S.; Gajdács, M.; Musah-Eroje, A.; Baráth, Z. Utility of Photodynamic Therapy in Dentistry: Current Concepts. Dent. J. 2020, 8, 43. [Google Scholar] [CrossRef] [PubMed]
- Carcuac, O.; Derks, J.; Charalampakis, G. Adjunctive Systemic and Local Antimicrobial Therapy in the Surgical Treatment of Peri-implantitis: A Randomized Controlled Clinical Trial. J. Dent. Res. 2016, 95, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Carcuac, O.; Derks, J.; Abrahamsson, I.; Wennström, J.L.; Berglundh, T. Risk for recurrence of disease following surgical therapy of peri-implantitis—A prospective longitudinal study. Clin. Oral Implant Res. 2020, 31, 1072–1077. [Google Scholar] [CrossRef]

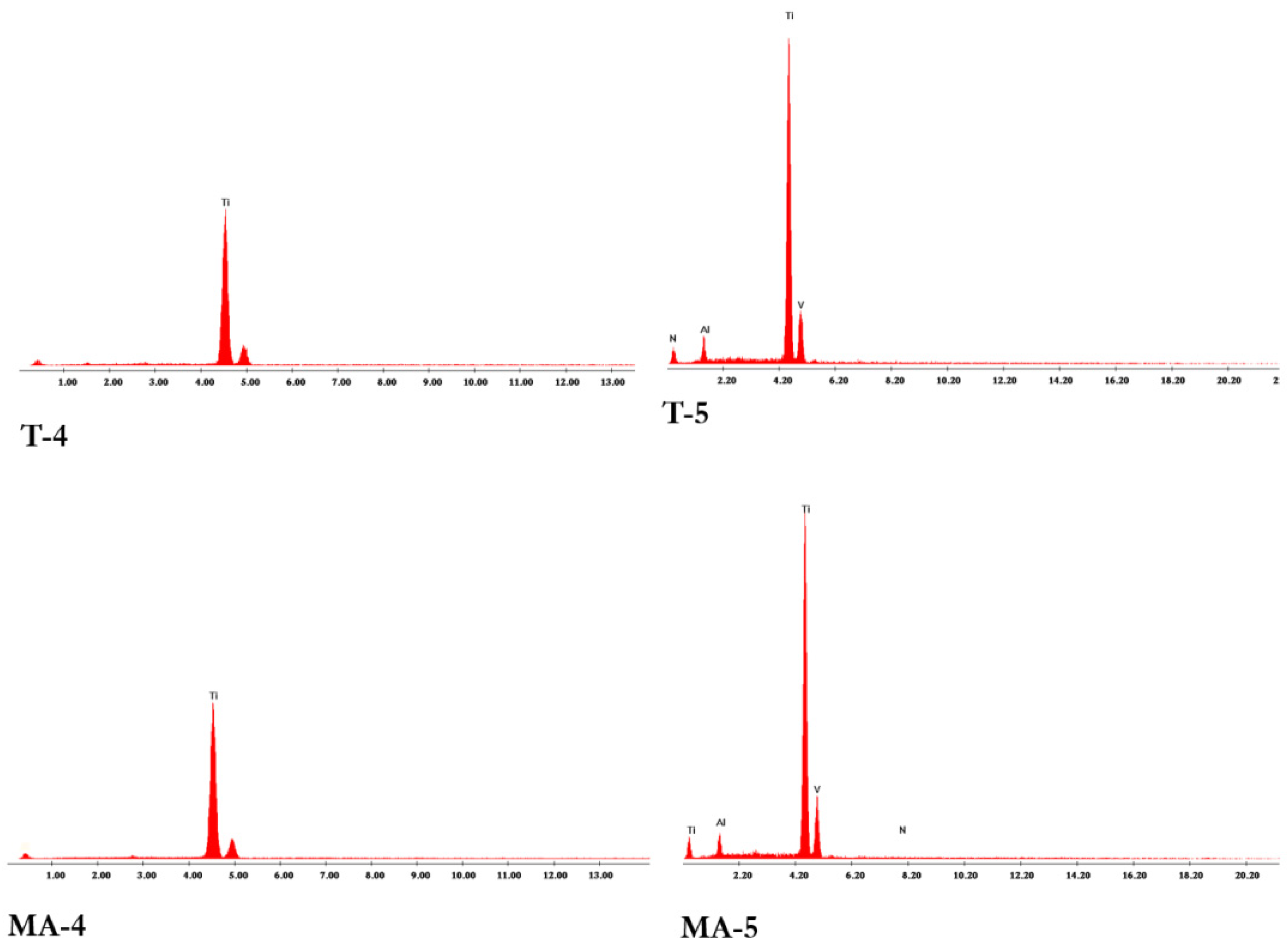

| Sample Code | Sa (µm) | Sds (µm−2) | Sdr (%) |
|---|---|---|---|
| T-4 | 0.126 ± 0.04 | 0.243 ± 0.01 | 10.47 ± 7.50 |
| T-5 | 0.134 ± 0.02 | 0.249 ± 0.01 | 11.16 ± 7.12 |
| MA-4 | 0.225 ± 0.02 * | 0.218 ± 0.01 | 12.43 ± 4.51 |
| MA-5 | 0.212 ± 0.02¤ | 0.205 ± 0.05 | 8.31 ± 7.65 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sbricoli, L.; Cecoro, G.; Leone, C.; Nastri, L.; Guazzo, R.; Guida, L.; Annunziata, M. Bacterial Adhesion to Grade 4 and Grade 5 Turned and Mildly Acid-Etched Titanium Implant Surfaces: An In Vitro and Ex Vivo Study. Appl. Sci. 2021, 11, 7185. https://doi.org/10.3390/app11167185
Sbricoli L, Cecoro G, Leone C, Nastri L, Guazzo R, Guida L, Annunziata M. Bacterial Adhesion to Grade 4 and Grade 5 Turned and Mildly Acid-Etched Titanium Implant Surfaces: An In Vitro and Ex Vivo Study. Applied Sciences. 2021; 11(16):7185. https://doi.org/10.3390/app11167185
Chicago/Turabian StyleSbricoli, Luca, Gennaro Cecoro, Claudio Leone, Livia Nastri, Riccardo Guazzo, Luigi Guida, and Marco Annunziata. 2021. "Bacterial Adhesion to Grade 4 and Grade 5 Turned and Mildly Acid-Etched Titanium Implant Surfaces: An In Vitro and Ex Vivo Study" Applied Sciences 11, no. 16: 7185. https://doi.org/10.3390/app11167185
APA StyleSbricoli, L., Cecoro, G., Leone, C., Nastri, L., Guazzo, R., Guida, L., & Annunziata, M. (2021). Bacterial Adhesion to Grade 4 and Grade 5 Turned and Mildly Acid-Etched Titanium Implant Surfaces: An In Vitro and Ex Vivo Study. Applied Sciences, 11(16), 7185. https://doi.org/10.3390/app11167185









