High Genetic Diversity and Low Population Differentiation in Wild Hop (Humulus lupulus L.) from Croatia
Abstract
:1. Introduction
2. Materials and Methods
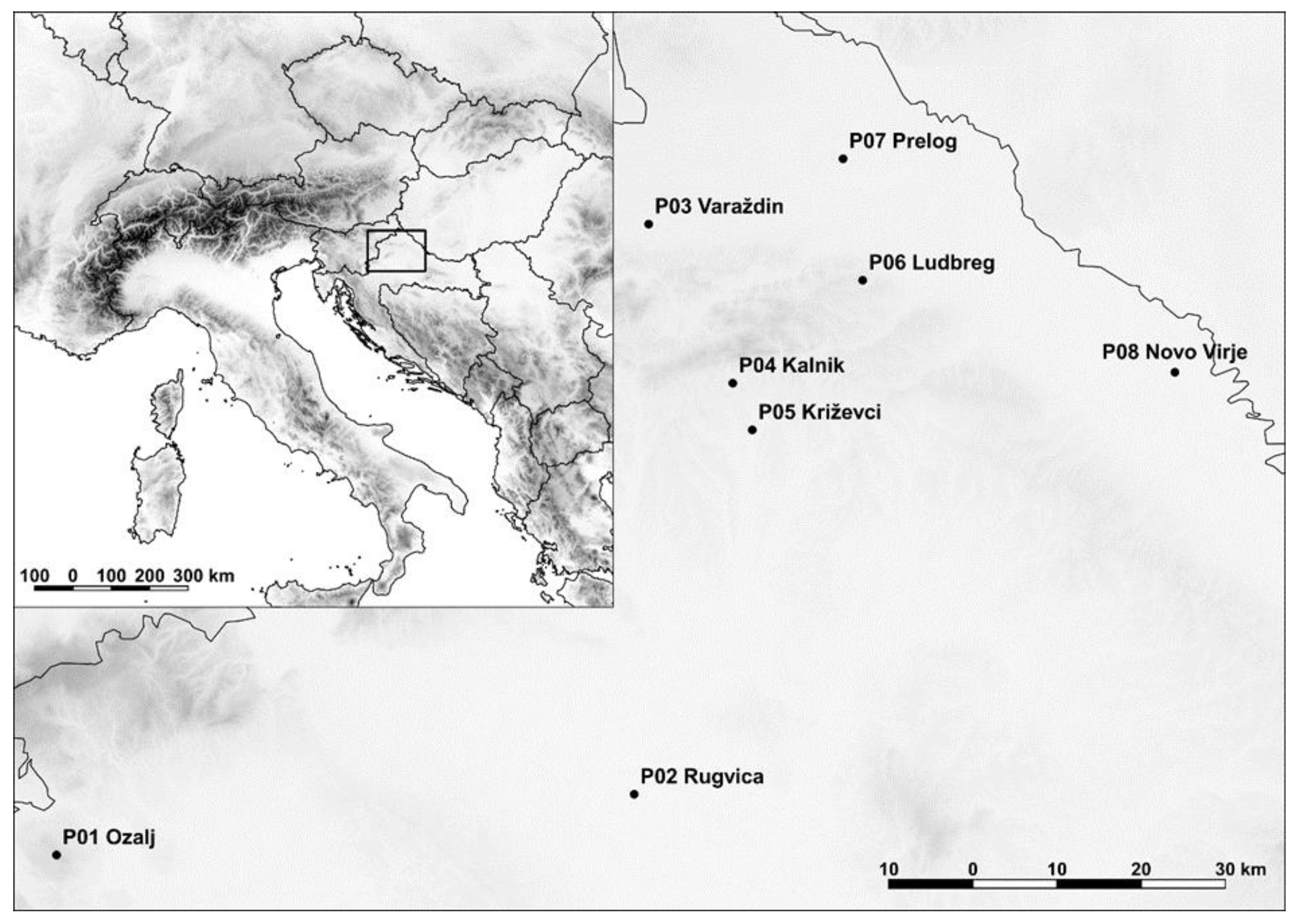
2.1. Sampling and Plant Material
2.2. DNA Extraction and SSR Genotyping
2.3. Data Analysis
2.3.1. Microsatellite Diversity
2.3.2. Within-Population Diversity
2.3.3. Population Differentiation and Structure
2.3.4. Isolation by Distance
3. Results and Discussion
3.1. Microsatellite Diversity
3.2. Within-Population Diversity
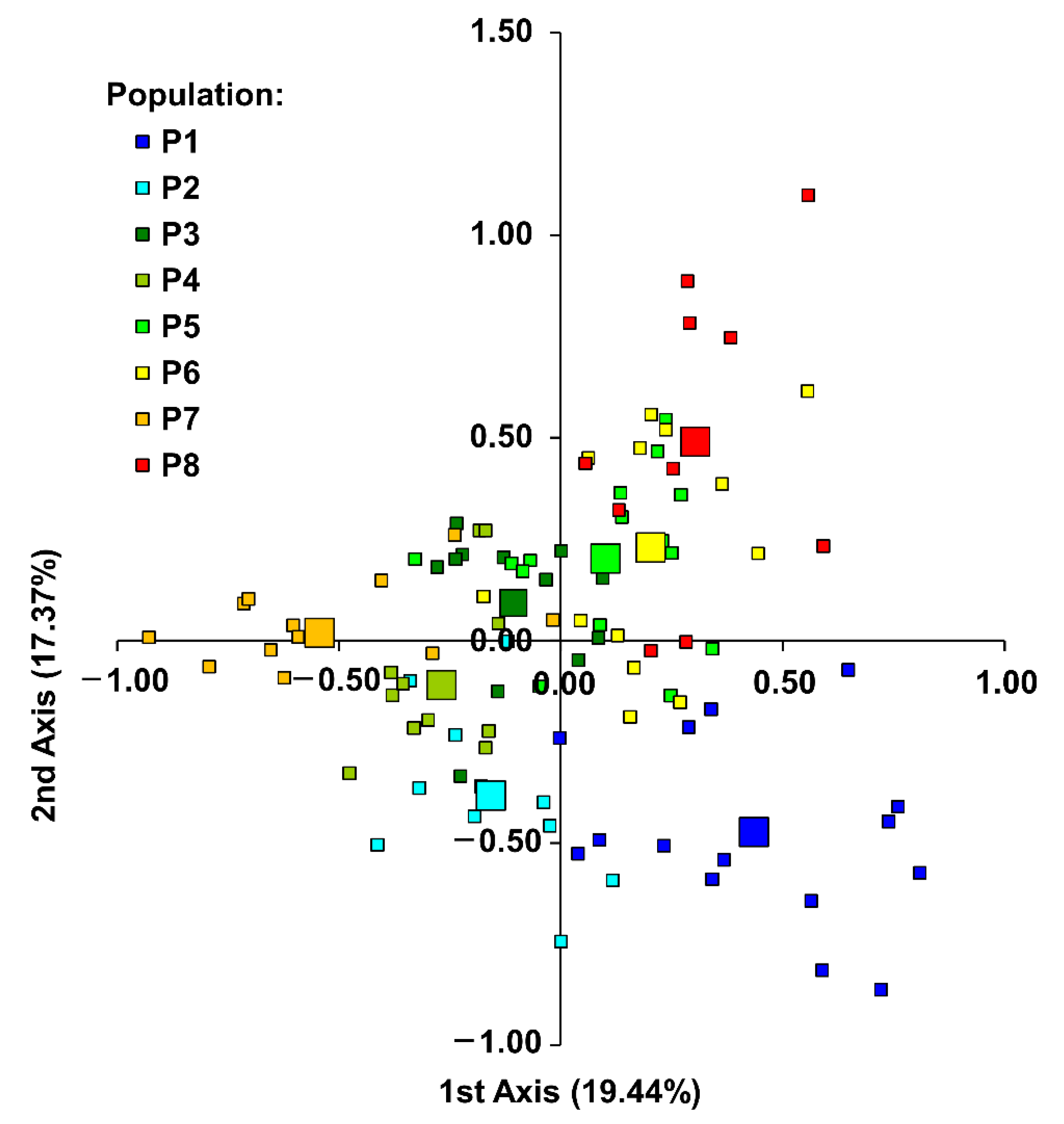
3.3. Population Differentiation and Structure
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wilson, D.G. Plant Remains From the Graveney Boat and the Early History of Humulus Lupulus L. in W. Europe. New Phytol. 1975, 75, 627–648. [Google Scholar] [CrossRef]
- Behre, K.-E. The history of beer additives in Europe—A review. Veg. Hist. Archaeobot. 1999, 8, 35–48. [Google Scholar] [CrossRef]
- Small, E. A Numerical and Nomenclatural Analysis of Morpho-Geographic Taxa of Humulus. Syst. Bot. 1978, 3, 37–76. [Google Scholar] [CrossRef]
- Burgess, A.H. Hops: Botany, Cultivation and Utilization; Leonard Hill Ltd.: London, UK, 1964. [Google Scholar]
- Delyser, D.Y.; Kasper, W.J. Hopped Beer: The Case For Cultivation. Econ. Bot. 1994, 48, 166–170. [Google Scholar] [CrossRef]
- Chadwick, L.R.; Pauli, G.F.; Farnsworth, N.R. The pharmacognosy of Humulus lupulus L. (hops) with an emphasis on estrogenic properties. Phytomedicine 2006, 13, 119–131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Neve, R.A. Hops; Chapman and Hall: London, UK, 1991; ISBN 978-0-412-30330-2. [Google Scholar]
- Patzak, J.; Nesvadba, V.; Henychová, A.; Krofta, K. Assessment of the genetic diversity of wild hops (Humulus lupulus L.) in Europe using chemical and molecular analyses. Biochem. Syst. Ecol. 2010, 38, 136–145. [Google Scholar] [CrossRef]
- Štajner, N.; Šatović, Z.; Čerenak, A.; Javornik, B. Genetic structure and differentiation in hop (Humulus lupulus L.) as inferred from microsatellites. Euphytica 2008, 161, 301–311. [Google Scholar] [CrossRef]
- Shephard, H.L.; Parker, J.S.; Darby, P.; Ainsworth, C.C. Sexual development and sex chromosomes in hop. New Phytol. 2000, 148, 397–411. [Google Scholar] [CrossRef] [PubMed]
- Čerenak, A.; Pavlovič, M.; Luskar, M.; Košir, I. Characterization of slovenian hop (Humulus lupulus L.) varieties by analysis of essential oil. Hop Bull. 2011, 18, 27–32. [Google Scholar]
- Negri, G.; di Santi, D.; Tabach, R. Bitter acids from hydroethanolic extracts of Humulus lupulus L., Cannabaceae, used as anxiolytic. Rev. Bras. Farmacogn. 2010, 20, 850–859. [Google Scholar] [CrossRef] [Green Version]
- Van Cleemput, M.; Cattoor, K.; De Bosscher, K.; Haegeman, G.; De Keukeleire, D.; Heyerick, A. Hop (Humulus lupulus)-derived bitter acids as multipotent bioactive compounds. J. Nat. Prod. 2009, 72, 1220–1230. [Google Scholar] [CrossRef] [PubMed]
- Zanoli, P.; Zavatti, M.; Rivasi, M.; Brusiani, F.; Losi, G.; Puia, G.; Avallone, R.; Baraldi, M. Evidence that the β-acids fraction of hops reduces central GABAergic neurotransmission. J. Ethnopharmacol. 2007, 109, 87–92. [Google Scholar] [CrossRef]
- Zanoli, P.; Zavatti, M. Pharmacognostic and pharmacological profile of Humulus lupulus L. J. Ethnopharmacol. 2008, 116, 383–396. [Google Scholar] [CrossRef]
- Kac, J.; Plazar, J.; Mlinarič, A.; Žegura, B.; Lah, T.T.; Filipič, M. Antimutagenicity of hops (Humulus lupulus L.): Bioassay-directed fractionation and isolation of xanthohumol. Phytomedicine 2008, 15, 216–220. [Google Scholar] [CrossRef]
- Gerhäuser, C. Beer constituents as potential cancer chemopreventive agents. Eur. J. Cancer 2005, 41, 1941–1954. [Google Scholar] [CrossRef]
- Jirovetz, L.; Bail, S.; Buchbauer, G.; Denkova, Z.; Slavchev, A.; Stoyanova, A.; Schmidt, E.; Geissler, M. Antimicrobial testings, gas chromatographic analysis and olfactory evaluation of an essential oil of hop cones (Humulus lupulus L.) from Bavaria and some of its main compounds. Sci. Pharm. 2006, 74, 189–201. [Google Scholar] [CrossRef] [Green Version]
- Tofană, M.; Socaci, S.A.; Socaciu, C.; Mihăiescu, D.E.; Semeniuc, C.; Truţa, D. Optimization of HS/GC-MS Method for the Determination of Volatile Compounds from some Indigenous Hop Varieties. Bull. Univ. Agric. Sci. Vet. Med. Cluj-Napoca Agric. 2009, 66, 1843–5386. [Google Scholar] [CrossRef]
- Liu, M.; Zeng, Z.; Xiong, B. Preparation of novel solid-phase microextraction fibers by sol-gel technology for headspace solid-phase microextraction-gas chromatographic analysis of aroma compounds in beer. J. Chromatogr. A 2005, 1065, 287–299. [Google Scholar] [CrossRef] [PubMed]
- Pinho, O.; Ferreira, I.M.P.L.V.O.; Santos, L.H.M.L.M. Method optimization by solid-phase microextraction in combination with gas chromatography with mass spectrometry for analysis of beer volatile fraction. J. Chromatogr. A 2006, 1121, 145–153. [Google Scholar] [CrossRef]
- Srečec, S.; Zechner-Krpan, V.; Petravić-Tominac, V.; Čerenak, A.; Liber, Z.; Šatović, Z. Phenotypic and alpha-acid content diversity of wild hop populations in Croatia. Plant Soil Environ. 2010, 56, 37–42. [Google Scholar] [CrossRef] [Green Version]
- Nance, M.R.; Setzer, W.N. Volatile components of aroma hops (Humulus lupulus L.) commonly used in beer brewing. J. Brew. Distill. 2011, 2, 16–22. [Google Scholar]
- Mongelli, A.; Rodolfi, M.; Ganino, T.; Marieschi, M.; Dall’Asta, C.; Bruni, R. Italian hop germplasm: Characterization of wild Humulus lupulus L. genotypes from Northern Italy by means of phytochemical, morphological traits and multivariate data analysis. Ind. Crops Prod. 2015, 70, 16–27. [Google Scholar] [CrossRef]
- Čeh, B.; Kač, M.; Košir, I.J.; Abram, V. Relationships between Xanthohumol and Polyphenol Content in Hop Leaves and Hop Cones with Regard to Water Supply and Cultivar. Int. J. Mol. Sci. 2007, 8, 989. [Google Scholar] [CrossRef] [Green Version]
- Okada, Y.; Ito, K. Cloning and analysis of valerophenone synthase gene expressed specifically in lupulin gland of hop (Humulus lupulus L.). Biosci. Biotechnol. Biochem. 2001, 65, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Jakše, J.; Bandelj, D.; Javornik, B. Eleven new microsatellites for hop (Humulus lupulus L.). Mol. Ecol. Notes 2002, 2, 544–546. [Google Scholar] [CrossRef]
- McAdam, E.L.; Vaillancourt, R.E.; Koutoulis, A.; Whittock, S.P. Quantitative genetic parameters for yield, plant growth and cone chemical traits in hop (Humulus lupulus L.). BMC Genet. 2014, 15, 1–18. [Google Scholar] [CrossRef]
- Shephard, H.; Parker, J.; Darby, P.; Charles, C.A. Sex expression in hop (Humulus lupulus L. and H.japonicus Sieb. et Zucc.) floral morphology and sex chromosomes. In Sex Determination in Plants; Ainsworth, C.C., Ed.; BIOS Scientific Publishers Ltd.: Oxford, UK, 1999; pp. 139–150. [Google Scholar]
- McKey, D.; Elias, M.; Pujol, M.E.; Duputié, A. The evolutionary ecology of clonally propagated domesticated plants. New Phytol. 2010, 186, 318–332. [Google Scholar] [CrossRef]
- Šuštar-Vozlič, J.; Javornik, B. Genetic relationships in cultivars of hop, Humulus lupulus L., determined by RAPD analysis. Plant Breed. 1999, 118, 175–181. [Google Scholar] [CrossRef]
- Murakami, A. Hop variety classification using the genetic distance based on RAPD. J. Inst. Brew. 2000, 106, 157–162. [Google Scholar] [CrossRef]
- Henning, J.A.; Steiner, J.J.; Hummer, K.E. Genetic diversity among world hop accessions grown in the USA. Crop Sci. 2004, 44, 411–417. [Google Scholar] [CrossRef]
- Seefelder, S.; Ehrmaier, H.; Schweizer, G.; Seigner, E. Genetic diversity and phylogenetic relationships among accessions of hop, Humulus lupulus, as determined by amplified fragment length polymorphism fingerprinting compared with pedigree data. Plant Breed. 2000, 119, 257–263. [Google Scholar] [CrossRef]
- Murakami, A.; Darby, P.; Javornik, B.; Pais, M.S.S.; Seigner, E.; Lutz, A.; Svoboda, P. Microsatellite DNA analysis of wild hops, Humulus lupulus L. Genet. Resour. Crop Evol. 2006, 53, 1553–1562. [Google Scholar] [CrossRef]
- Patzak, J.; Nesvadba, V.; Krofta, K.; Henychova, A.; Marzoev, A.I.; Richards, K. Evaluation of genetic variability of wild hops (Humulus lupulus L.) in Canada and the Caucasus region by chemical and molecular methods. Genome 2010, 53, 545–557. [Google Scholar] [CrossRef]
- Riccioni, C.; Belfiori, B.; Sileoni, V.; Marconi, O.; Perretti, G.; Bellucci, M.; Rubini, A. High genetic and chemical diversity of wild hop populations from Central Italy with signals of a genetic structure influenced by both sexual and asexual reproduction. Plant Sci. 2021, 304, 110794. [Google Scholar] [CrossRef]
- Mafakheri, M.; Kordrostami, M.; Rahimi, M.; Matthews, P.D. Evaluating genetic diversity and structure of a wild hop (Humulus lupulus L.) germplasm using morphological and molecular characteristics. Euphytica 2020, 216, 1–19. [Google Scholar] [CrossRef]
- Jakše, J.; Šatović, Z.; Javornik, B. Microsatellite variability among wild and cultivated hops (Humulus lupulus L.). Genome 2004, 47, 889–899. [Google Scholar] [CrossRef]
- Peredo, E.L.; Revilla, M.Á.; Reed, B.M.; Javornik, B.; Cires, E.; Prieto, J.A.F.; Arroyo-García, R. The influence of European and American wild germplasm in hop (Humulus lupulus L.) cultivars. Genet. Resour. Crop Evol. 2010, 57, 575–586. [Google Scholar] [CrossRef]
- Rodolfi, M.; Silvanini, A.; Chiancone, B.; Marieschi, M.; Fabbri, A.; Bruni, R.; Ganino, T. Identification and genetic structure of wild Italian Humulus lupulus L. and comparison with European and American hop cultivars using nuclear microsatellite markers. Genet. Resour. Crop Evol. 2018, 65, 1405–1422. [Google Scholar] [CrossRef]
- Dabbous-Wach, A.; Rodolfi, M.; Paolini, J.; Costa, J.; Ganino, T. Characterization of wild corsican hops and assessment of the performances of german hops in corsican environmental conditions through a multidisciplinary approach. Appl. Sci. 2021, 11, 3756. [Google Scholar] [CrossRef]
- Jakše, J.; Kindlhofer, K.; Javornik, B. Assessment of genetic variation and differentiation of hop genotypes by microsatellite and AFLP markers. Genome 2001, 44, 773–782. [Google Scholar] [CrossRef] [PubMed]
- Srečec, S.; Kvaternjak, I.; Kaučić, D.; Špoljar, A.; Erhatić, R. Influence of Climatic Conditions on Accumulation of α-acids in Hop Cones. Agric. Conspec. Sci. 2008, 73, 161–166. [Google Scholar]
- Pavlovic, M.; Pavlovic, V.; Rozman, C.; Udovc, A.; Stajnko, D.; Wang, D.; Gavric, M.; Srecec, S. Market value assessment of hops by modeling of weather attributes. Plant Soil Environ. 2013, 59, 267–272. [Google Scholar] [CrossRef] [Green Version]
- Rossini, F.; Loreti, P.; Provenzano, M.E.; De Santis, D.; Ruggeri, R. Agronomic performance and beer quality assessment of twenty hop cultivars grown in central Italy. Ital. J. Agron. 2016, 11, 180–187. [Google Scholar] [CrossRef] [Green Version]
- Mackinnon, D.; Pavlovič, V.; Čeh, B.; Naglič, B.; Pavlovič, M. The impact of weather conditions on alpha-acid content in hop (Humulus lupulus L.) cv. aurora. Plant Soil Environ. 2020, 66, 519–525. [Google Scholar] [CrossRef]
- Kimura, M. The Neutral Theory of Molecular Evolution; Cambridge University Press: Cambridge, MA, USA; New York, NY, USA, 1983. [Google Scholar]
- Hague, M.T.J.; Routman, E.J. Does population size affect genetic diversity? A test with sympatric lizard species. Heredity 2015, 116, 92–98. [Google Scholar] [CrossRef] [Green Version]
- Wagner, T. Autochthonous Hop in Yugoslavia and Its Usability for Breeding New Varieties in Comparison with the Grown in Present; Institute of Hop Research: Žalec, Slovenia, 1974. [Google Scholar]
- Kišgeci, J.; Mijavec, A.; Aćimović, M.; Spevak, P.; Vučić, N. Hmeljarstvo; Faculty of Agriculture–Institute of Field and Vegetable Crops: Novi Sad, Serbia, 1984. [Google Scholar]
- Rybáček, V. Hop Production; SZN: Prague, Czech Republic, 1991. [Google Scholar]
- Srečec, S. Hmeljarstvo; Križevci College of Agriculture: Križevci, Croatia, 2004. [Google Scholar]
- Hijmans, R.J.; Cameron, S.E.; Parra, J.L.; Jones, P.G.; Jarvis, A. Very high resolution interpolated climate surfaces for global land areas. Int. J. Climatol. 2005, 25, 1965–1978. [Google Scholar] [CrossRef]
- Jakše, J.; Luthar, Z.; Javornik, B. New polymorphic dinucleotide and trinucleotide microsatellite loci for hop Humulus lupulus L. Mol. Ecol. Resour. 2008, 8, 769–772. [Google Scholar] [CrossRef] [PubMed]
- Kalinowski, S.T.; Taper, M.L.; Marshall, T.C. Revising how the computer program CERVUS accommodates genotyping error increases success in paternity assignment. Mol. Ecol. 2007, 16, 1099–1106. [Google Scholar] [CrossRef]
- Arnaud-Haond, S.; Belkhir, K. GENCLONE: A computer program to analyse genotypic data, test for clonality and describe spatial clonal organization. Mol. Ecol. Notes 2007, 7, 15–17. [Google Scholar] [CrossRef]
- Bowcock, A.M.; Ruiz-Linares, A.; Tomfohrde, J.; Minch, E.; Kidd, J.R.; Cavalli-Sforza, L.L. High resolution of human evolutionary trees with polymorphic microsatellites. Nature 1994, 368, 455–457. [Google Scholar] [CrossRef] [PubMed]
- Minch, E.; Ruiz-Linares, A.; Goldstein, D.; Feldman, M.; Cavalli-Sforza, L.L. MICROSAT: A Computer Program for Calculating Various Statistics on Microsatellite Allele Data; Version 1.5; Stanford University: Stanford, CA, USA, 1997. [Google Scholar]
- Van Oosterhout, C.; Hutchinson, W.F.; Wills, D.P.M.; Shipley, P. MICRO-CHECKER: Software for identifying and correcting genotyping errors in microsatellite data. Mol. Ecol. Notes 2004, 4, 535–538. [Google Scholar] [CrossRef]
- Dempster, A.P.; Laird, N.M.; Rubin, D.B. Maximum Likelihood from Incomplete Data Via the EM Algorithm. J. R. Stat. Soc. Ser. B 1977, 39, 1–22. [Google Scholar] [CrossRef]
- Chapuis, M.P.; Estoup, A. Microsatellite null alleles and estimation of population differentiation. Mol. Biol. Evol. 2007, 24, 621–631. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kalinowski, S.T. HP-RARE 1.0: A computer program for performing rarefaction on measures of allelic richness. Mol. Ecol. Notes 2005, 5, 187–189. [Google Scholar] [CrossRef]
- Kalinowski, S.T. Counting Alleles with Rarefaction: Private Alleles and Hierarchical Sampling Designs. Conserv. Genet. 2004, 5, 539–543. [Google Scholar] [CrossRef]
- Raymond, M.; Rousset, F. GENEPOP (Version 1.2): Population Genetics Software for Exact Tests and Ecumenicism. J. Hered. 1995, 86, 248–249. [Google Scholar] [CrossRef]
- Zachariah Peery, M.; Kirby, R.; Reid, B.N.; Stoelting, R.; Doucet-Bëer, E.; Robinson, S.; Vásquez-Carrillo, C.; Pauli, J.N.; Palsboll, P.J. Reliability of genetic bottleneck tests for detecting recent population declines. Mol. Ecol. 2012, 21, 3403–3418. [Google Scholar] [CrossRef]
- Luikart, G.; Allendorf, F.W.; Cornuet, J.M.; Sherwin, W.B. Distortion of allele frequency distributions provides a test for recent population bottlenecks. J. Hered. 1998, 89, 238–247. [Google Scholar] [CrossRef]
- Cornuet, J.M.; Luikart, G. Description and power analysis of two tests for detecting recent population bottlenecks from allele frequency data. Genetics 1996, 144, 2001–2014. [Google Scholar] [CrossRef]
- Goudet, J. FSTAT (Version 1.2): A Computer Program to Calculate F-statistics. J. Hered. 1995, 86, 485–486. [Google Scholar] [CrossRef]
- Excoffier, L.; Lischer, H.E.L. Arlequin suite ver 3.5: A new series of programs to perform population genetics analyses under Linux and Windows. Mol. Ecol. Resour. 2010, 10, 564–567. [Google Scholar] [CrossRef]
- Excoffier, L.; Smouse, P.E.; Quattro, J.M. Analysis of molecular variance inferred from metric distances among DNA haplotypes: Application to human mitochondrial DNA restriction data. Genetics 1992, 131, 479–491. [Google Scholar] [CrossRef]
- Belkhir, K.; Borsa, P.; Chikhi, L.; Raufaste, N.; Bonhomme, F. GENETIX 4.05, Logiciel Sous Windows TM pour la Génétique des Populations. Laboratoire Génome, Populations, Interactions, CNRS UMR 5000; Université de Montpellier II: Montpellier, France, 2004. [Google Scholar]
- Pritchard, J.K.; Stephens, M.; Donnelly, P. Inference of population structure using multilocus genotype data. Genetics 2000, 155, 945–959. [Google Scholar] [CrossRef]
- Hubisz, M.J.; Falush, D.; Stephens, M.; Pritchard, J.K. Inferring weak population structure with the assistance of sample group information. Mol. Ecol. Resour. 2009, 9, 1322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Evanno, G.; Regnaut, S.; Goudet, J. Detecting the number of clusters of individuals using the software STRUCTURE: A simulation study. Mol. Ecol. 2005, 14, 2611–2620. [Google Scholar] [CrossRef] [Green Version]
- Earl, D.A.; vonHoldt, B.M. STRUCTURE HARVESTER: A website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv. Genet. Resour. 2012, 4, 359–361. [Google Scholar] [CrossRef]
- Kopelman, N.M.; Mayzel, J.; Jakobsson, M.; Rosenberg, N.A.; Mayrose, I. Clumpak: A program for identifying clustering modes and packaging population structure inferences across K. Mol. Ecol. Resour. 2015, 15, 1179–1191. [Google Scholar] [CrossRef] [Green Version]
- Rousset, F. Genetic differentiation and estimation of gene flow from F-statistics under isolation by distance. Genetics 1997, 145, 1219–1228. [Google Scholar] [CrossRef]
- Rohlf, F.J. NTSYS-pc. Version 2.2. Numerical Taxonomy and Multivariate Analysis System; Applied Biostatistics Inc.: New York, NY, USA, 2000. [Google Scholar]
- Carlsson, J. Effects of microsatellite null alleles on assignment testing. J. Hered. 2008, 99, 616–623. [Google Scholar] [CrossRef] [Green Version]
- Čerenak, A.; Jakše, J.; Javornik, B. Identification and Differentiation of Hop Varieties Using Simple Sequence Repeat Markers. J. Am. Soc. Brew. Chem. 2004, 62, 1–7. [Google Scholar] [CrossRef]
- Park, Y.-J.; Dixit, A.; Ma, K.-H.; Lee, J.-K.; Lee, M.-H.; Chung, C.-S.; Nitta, M.; Okuno, K.; Kim, T.-S.; Cho, E.-G.; et al. Evaluation of genetic diversity and relationships within an on-farm collection of Perilla frutescens (L.) Britt. using microsatellite markers. Genet. Resour. Crop Evol. 2008, 55, 523–535. [Google Scholar] [CrossRef]
- Hamrick, J.L.; Godt, M.J.W. Allozyme diversity in plant species. In Plant Population Genetics, Breeding, and Genetic Resources; Brown, A.H.D., Clegg, M.T., Kahler, A.L., Weir, B.S., Eds.; Sinauer Associates Inc.: Sunderland, MA, USA, 1990; pp. 43–63. ISBN 0878931171. [Google Scholar]
- Nybom, H. Comparison of different nuclear DNA markers for estimating intraspecific genetic diversity in plants. Mol. Ecol. 2004, 13, 1143–1155. [Google Scholar] [CrossRef] [PubMed]
- Millar, C.I.; Libby, W.J. Strategies for Conserving Clinal, Ecotypic, and Disjunct Population Diversiv in Widespread Species. In Genetics and Conservation of Rare Plants; Falk, D.A., Holsinger, K.E., Eds.; Oxford University Press: Oxford, UK, 1991. [Google Scholar]
- Bittencourt, J.V.M.; Sebbenn, A.M. Patterns of pollen and seed dispersal in a small, fragmented population of the wind-pollinated tree Araucaria angustifolia in southern Brazil. Heredity (Edinb.) 2007, 99, 580–591. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hamrick, J.L.; Godt, M.J.W. Effects of Life History Traits on Genetic Diversity in Plant Species. Philos. Trans. Biol. Sci. 1996, 351, 1291–1298. [Google Scholar]
- Nybom, H.; Bartish, I.V. Effects of life history traits and sampling strategies on genetic diversity estimates obtained with RAPD markers in plants. Perspect. Plant Ecol. Evol. Syst. 2000, 3, 93–114. [Google Scholar] [CrossRef]
| No. | Locus | Fluorophore Label | Repeat Motif | Size Range | Na | PIC | PI |
|---|---|---|---|---|---|---|---|
| M01 | GA4-K16-8 | PET | (GA)16 | 190–206 | 5 | 0.604 | 0.176 |
| M02 | GA4-P11-9 | VIC | (GA)18 | 217–229 | 7 | 0.791 | 0.060 |
| M03 | GA5-G3-10 | NED | (CT)22 | 123–179 | 25 | 0.874 | 0.021 |
| M04 | GA6-N13-14 | VIC | (TC)21 | 209–271 | 24 | 0.823 | 0.042 |
| M05 | GA6-N21-14 | NED | (CT)17 | 203–219 | 7 | 0.599 | 0.170 |
| M06 | GA6-P20-14 | PET | (TC)16 | 198–206 | 5 | 0.547 | 0.216 |
| M07 | GA7-07-16 | VIC | (AG)28 | 183–227 | 20 | 0.881 | 0.021 |
| M08 | GA7-16-16 | NED | (GA)16 | 218–252 | 14 | 0.782 | 0.062 |
| M09 | GA7-A6-14 | 6-FAM | (TC)10 | 179–191 | 5 | 0.588 | 0.183 |
| M10 | GA8-K15-4 | 6-FAM | (CT)19 | 238–268 | 12 | 0.794 | 0.057 |
| M11 | GT1-K1-4 | 6-FAM | (TG)10C(GT)9 | 168–220 | 20 | 0.866 | 0.026 |
| M12 | GT2-010-8 | PET | (TA)6(TG)16 | 188–202 | 8 | 0.626 | 0.155 |
| Average | 12.67 | 0.731 | 0.099 |
| No. | Locality | M | F | n | Na | Nar | Npr | Npar | HO | HE | FIS | PBottleneck | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| P1 | Ozalj | 9 | 6 | 15 | 6.917 | 5.955 | 5 | 0.369 | 0.689 | 0.725 | 0.050 | ns | 0.575 |
| P2 | Rugvica | 5 | 5 | 10 | 6.500 | 6.500 | 1 | 0.168 | 0.767 | 0.769 | 0.004 | ns | 0.235 |
| P3 | Varaždin | 5 | 7 | 12 | 6.333 | 5.992 | 3 | 0.305 | 0.785 | 0.760 | −0.033 | ns | 0.117 |
| P4 | Kalnik | 4 | 8 | 12 | 6.833 | 6.362 | 5 | 0.474 | 0.736 | 0.771 | 0.045 | ns | 0.765 |
| P5 | Križevci | 5 | 10 | 15 | 7.167 | 6.226 | 4 | 0.272 | 0.750 | 0.753 | 0.004 | ns | 0.575 |
| P6 | Ludbreg | 3 | 10 | 13 | 7.250 | 6.619 | 2 | 0.256 | 0.840 | 0.789 | −0.065 | ns | 0.396 |
| P7 | Prelog | 4 | 10 | 14 | 7.000 | 6.240 | 10 | 0.714 | 0.792 | 0.759 | −0.043 | ns | 0.311 |
| P8 | Novo Virje | 3 | 7 | 10 | 6.500 | 6.500 | 8 | 0.713 | 0.800 | 0.750 | −0.067 | ns | 0.715 |
| Average | 4.750 | 7.875 | 12.630 | 6.813 | 6.299 | 4.75 | 0.409 | 0.770 | 0.759 | - | - | ||
| Total | 38 | 63 | 101 | 12.667 | - | 38 | - | 0.767 | 0.768 | 0.001 | 0.924 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grdiša, M.; Šatović, Z.; Liber, Z.; Jakše, J.; Varga, F.; Erhatić, R.; Srečec, S. High Genetic Diversity and Low Population Differentiation in Wild Hop (Humulus lupulus L.) from Croatia. Appl. Sci. 2021, 11, 6484. https://doi.org/10.3390/app11146484
Grdiša M, Šatović Z, Liber Z, Jakše J, Varga F, Erhatić R, Srečec S. High Genetic Diversity and Low Population Differentiation in Wild Hop (Humulus lupulus L.) from Croatia. Applied Sciences. 2021; 11(14):6484. https://doi.org/10.3390/app11146484
Chicago/Turabian StyleGrdiša, Martina, Zlatko Šatović, Zlatko Liber, Jernej Jakše, Filip Varga, Renata Erhatić, and Siniša Srečec. 2021. "High Genetic Diversity and Low Population Differentiation in Wild Hop (Humulus lupulus L.) from Croatia" Applied Sciences 11, no. 14: 6484. https://doi.org/10.3390/app11146484
APA StyleGrdiša, M., Šatović, Z., Liber, Z., Jakše, J., Varga, F., Erhatić, R., & Srečec, S. (2021). High Genetic Diversity and Low Population Differentiation in Wild Hop (Humulus lupulus L.) from Croatia. Applied Sciences, 11(14), 6484. https://doi.org/10.3390/app11146484








