Diversity of Synthetic Dyes from Textile Industries, Discharge Impacts and Treatment Methods
Abstract
1. Introduction
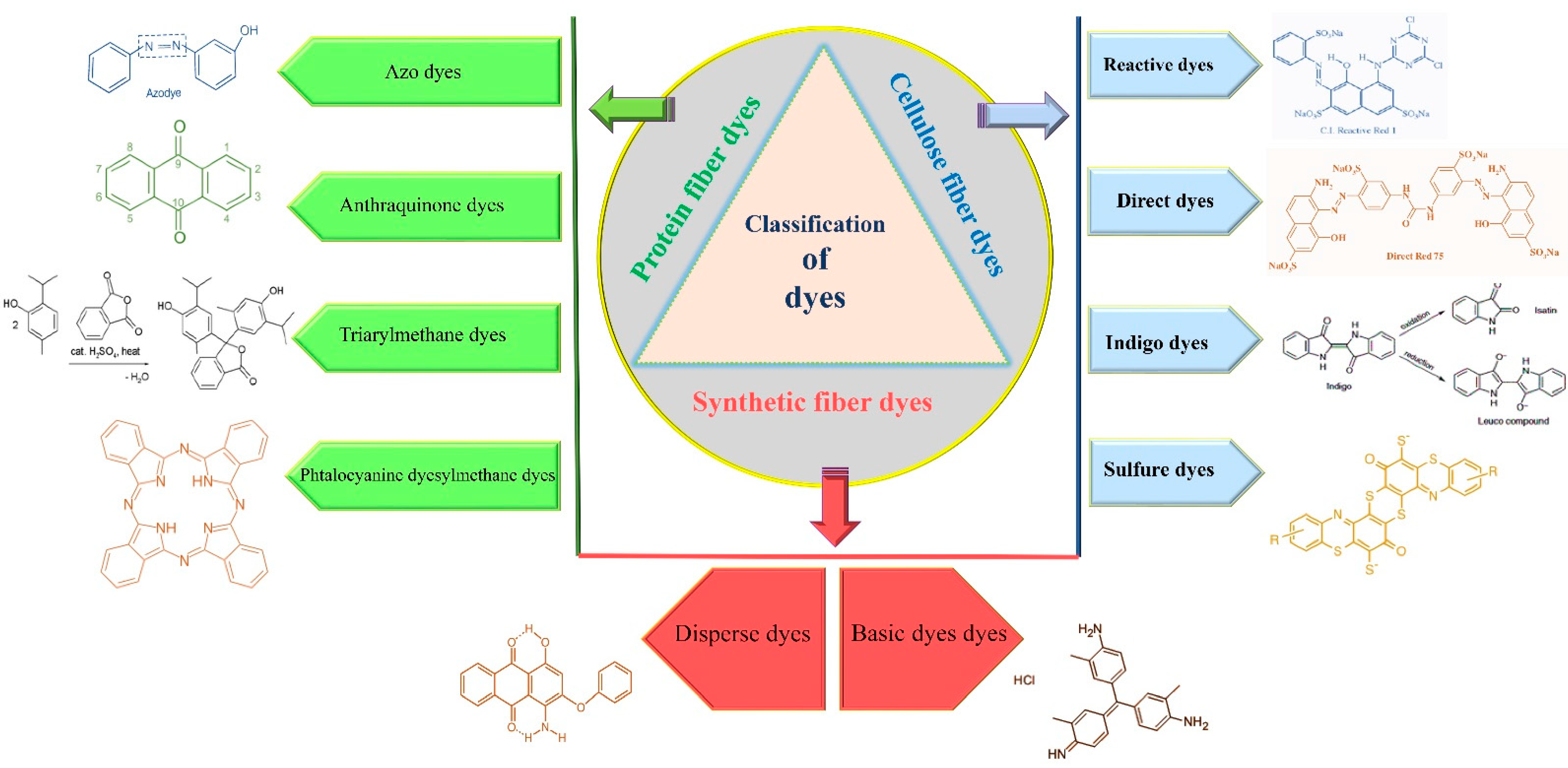
2. Classification of Dyes
2.1. Cellulose Fiber Dyes
2.1.1. Reactive Dyes
2.1.2. Direct Dyes
2.1.3. Indigo Dyes
2.1.4. Sulfur Dyes
2.2. Protein Fiber Dyes
2.2.1. Azo Dyes
2.2.2. Anthraquinone Dyes
2.2.3. Triarylmethane Dyes
2.2.4. Phtalocyanine Dyes
2.3. Synthetic Fiber Dyes
2.3.1. Disperse Dyes
2.3.2. Basic Dyes
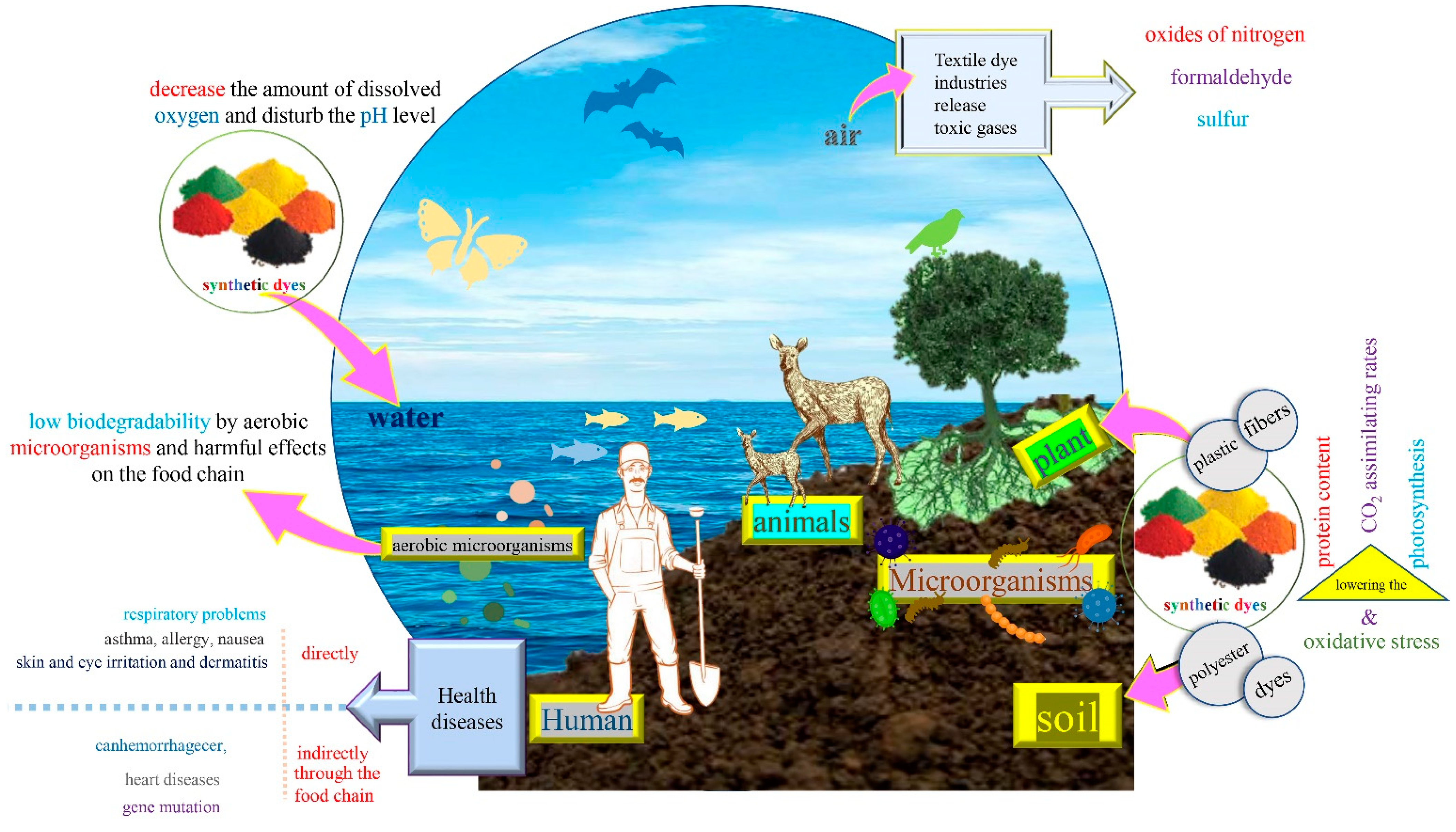
3. Characteristics and Impacts of Synthetic Dyes
3.1. Harmful Impacts on Soil and Plants
3.2. Harmful Impacts on Air
3.3. Harmful Impacts on Water
3.4. Harmful Impacts on Humans
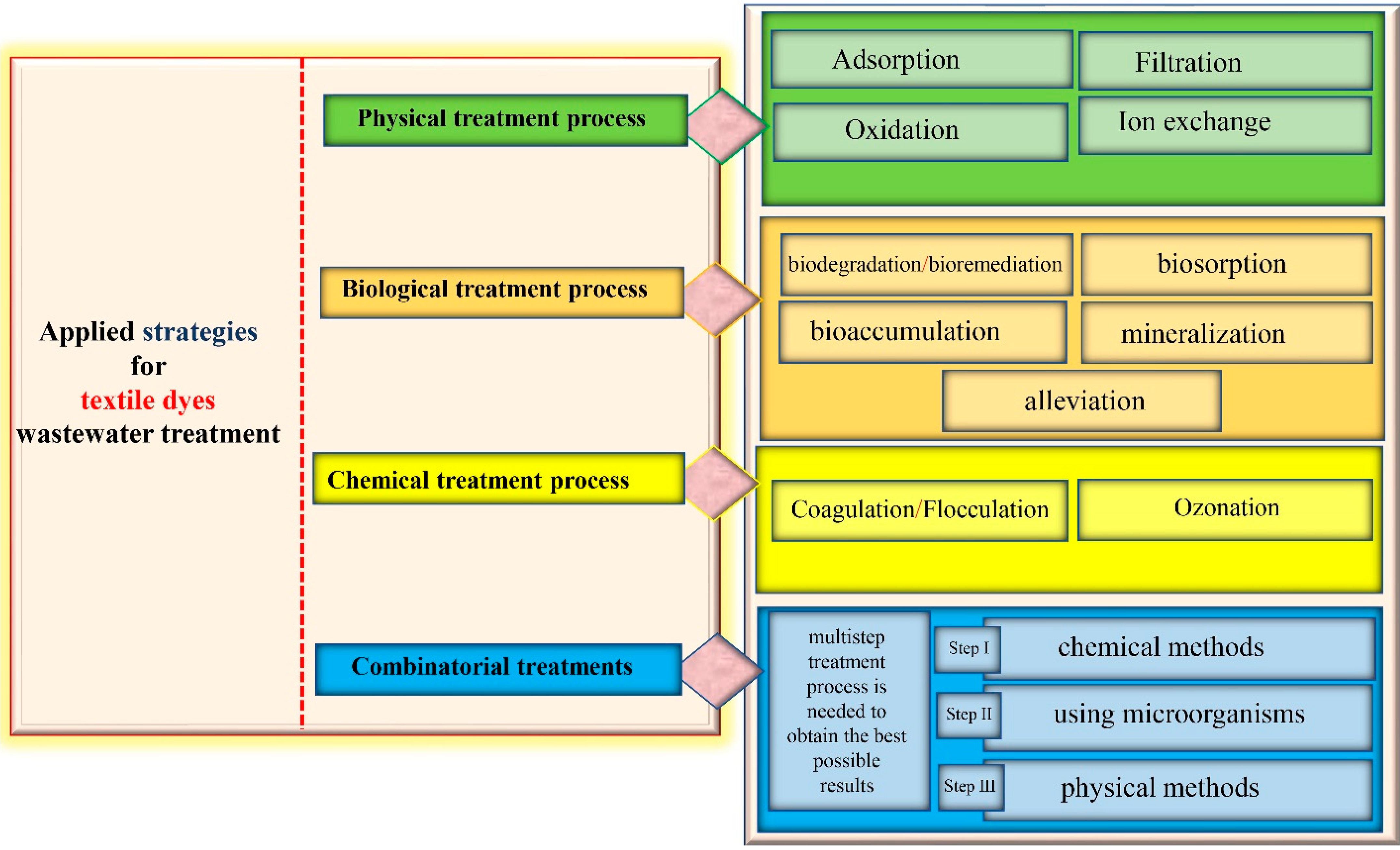
4. Applied Strategies for Textile Dye Wastewater Treatment
4.1. Physical Treatment Process
4.1.1. Adsorption
4.1.2. Nanoparticle Utilization
4.1.3. Filtration
4.1.4. Ion-Exchange
4.1.5. Oxidation
4.2. Chemical Treatment Process
4.2.1. Coagulation/Flocculation
4.2.2. Ozonation
4.3. Biological Treatment Process
4.4. Combinatorial Treatments
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bharagava, R.N.; Saxena, G.; Mulla, S.I. Introduction to industrial wastes containing organic and inorganic pollutants and bioremediation approaches for environmental management. In Bioremediation of Industrial Waste for Environmental Safety; Springer: New York, NY, USA, 2020; pp. 1–18. [Google Scholar]
- Khan, S.; Malik, A. Toxicity Evaluation of Textile Effluents and Role of Native Soil Bacterium in Biodegradation of a Textile Dye. Environ. Sci. Pollut. Res. 2018, 25, 4446–4458. [Google Scholar] [CrossRef] [PubMed]
- Celia, M.P.; Suruthi, S. Textile Dye Degradation Using Bacterial Strains Isolated from Textile Mill Effluent. Int. J. Appl. Res. 2016, 2, 337–341. [Google Scholar]
- Shindhal, T.; Rakholiya, P.; Varjani, S.; Pandey, A.; Ngo, H.H.; Guo, W.; Ng, H.Y.; Taherzadeh, M.J. A Critical Review on Advances in the Practices and Perspectives for the Treatment of Dye Industry Wastewater. Bioengineered 2021, 12, 70–87. [Google Scholar] [CrossRef] [PubMed]
- Druding, S.C. (Ed.) Dye History from 2600 BC to the 20th Century. 1982. Available online: http://www.straw.com/sig/dyehist.html (accessed on 1 June 2021).
- Singh, L.; Singh, V.P. Textile dyes degradation: A microbial approach for biodegradation of pollutants. In Microbial Degradation of Synthetic Dyes in Wastewaters; Springer: New York, NY, USA, 2015; pp. 187–204. [Google Scholar]
- Bhatia, D.; Sharma, N.R.; Singh, J.; Kanwar, R.S. Biological Methods for Textile Dye Removal from Wastewater: A Review. Crit. Rev. Environ. Sci. Technol. 2017, 47, 1836–1876. [Google Scholar] [CrossRef]
- Jamee, R.; Siddique, R. Biodegradation of Synthetic Dyes of Textile Effluent by Microorganisms: An Environmentally and Economically Sustainable Approach. Eur. J. Microbiol. Immunol. 2019, 9, 114–118. [Google Scholar] [CrossRef]
- Madhav, S.; Ahamad, A.; Singh, P.; Mishra, P.K. A Review of Textile Industry: Wet Processing, Environmental Impacts, and Effluent Treatment Methods. Environ. Qual. Manag. 2018, 27, 31–41. [Google Scholar] [CrossRef]
- Thakur, S.; Chauhan, M.S. Treatment of Dye Wastewater from Textile Industry by Electrocoagulation and Fenton Oxidation: A Review. In Proceedings of the Water Quality Management; Singh, V.P., Yadav, S., Yadava, R.N., Eds.; Springer: Singapore, 2018; pp. 117–129. [Google Scholar]
- Desore, A.; Narula, S.A. An Overview on Corporate Response towards Sustainability Issues in Textile Industry. Environ. Dev. Sustain. 2018, 20, 1439–1459. [Google Scholar] [CrossRef]
- Lellis, B.; Fávaro-Polonio, C.Z.; Pamphile, J.A.; Polonio, J.C. Effects of Textile Dyes on Health and the Environment and Bioremediation Potential of Living Organisms. Biotechnol. Res. Innov. 2019, 3, 275–290. [Google Scholar] [CrossRef]
- Hossen, M.Z.; Hussain, M.E.; Hakim, A.; Islam, K.; Uddin, M.N.; Azad, A.K. Biodegradation of Reactive Textile Dye Novacron Super Black G by Free Cells of Newly Isolated Alcaligenes Faecalis AZ26 and Bacillus Spp Obtained from Textile Effluents. Heliyon 2019, 5, e02068. [Google Scholar] [CrossRef] [PubMed]
- Barathi, S.; Karthik, C.; Nadanasabapathi, S.; Padikasan, I.A. Biodegradation of Textile Dye Reactive Blue 160 by Bacillus Firmus (Bacillaceae: Bacillales) and Non-Target Toxicity Screening of Their Degraded Products. Toxicol. Rep. 2020, 7, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Yaseen, D.A.; Scholz, M. Textile Dye Wastewater Characteristics and Constituents of Synthetic Effluents: A Critical Review. Int. J. Environ. Sci. Technol. 2019, 16, 1193–1226. [Google Scholar] [CrossRef]
- Gita, S.; Hussan, A.; Choudhury, T.G. Impact of Textile Dyes Waste on Aquatic Environments and Its Treatment. Environ. Ecol. 2017, 35, 2349–2353. [Google Scholar]
- Ghaly, A.E.; Ananthashankar, R.; Alhattab, M.; Ramakrishnan, V.V. Production, Characterization and Treatment of Textile Effluents: A Critical Review. J. Chem. Eng. Process Technol. 2014, 5, 1–18. [Google Scholar]
- Burkinshaw, S.M.; Salihu, G. The Role of Auxiliaries in the Immersion Dyeing of Textile Fibres: Part 5 Practical Aspects of the Role of Inorganic Electrolytes in Dyeing Cellulosic Fibres with Direct Dyes. Dyes Pigment. 2019, 161, 581–594. [Google Scholar] [CrossRef]
- Paz, A.; Carballo, J.; Pérez, M.J.; Domínguez, J.M. Biological Treatment of Model Dyes and Textile Wastewaters. Chemosphere 2017, 181, 168–177. [Google Scholar] [CrossRef]
- Chowdhury, M.F.; Khandaker, S.; Sarker, F.; Islam, A.; Rahman, M.T.; Awual, M.R. Current Treatment Technologies and Mechanisms for Removal of Indigo Carmine Dyes from Wastewater: A Review. J. Mol. Liq. 2020, 318, 114061. [Google Scholar] [CrossRef]
- Nguyen, T.A.; Juang, R.S. Treatment of Waters and Wastewaters Containing Sulfur Dyes: A Review. Chem. Eng. J. 2013, 219, 109–117. [Google Scholar] [CrossRef]
- Khattab, T.A.; Abdelrahman, M.S.; Rehan, M. Textile Dyeing Industry: Environmental Impacts and Remediation. Environ. Sci. Pollut. Res. 2020, 27, 3803–3818. [Google Scholar] [CrossRef] [PubMed]
- dos Santos Pisoni, D.; de Abreu, M.P.; Petzhold, C.L.; Rodembusch, F.S.; Campo, L.F. Synthesis, Photophysical Study and BSA Association of Water-Insoluble Squaraine Dyes. J. Photochem. Photobiol. A Chem. 2013, 252, 77–83. [Google Scholar] [CrossRef]
- Wan, Z.; Li, D.; Jiao, Y.; Ouyang, X.; Chang, L.; Wang, X. Bifunctional MoS2 Coated Melamine-Formaldehyde Sponges for Efficient Oil–Water Separation and Water-Soluble Dye Removal. Appl. Mater. Today 2017, 9, 551–559. [Google Scholar] [CrossRef]
- Salem, M.Z.; Ibrahim, I.H.; Ali, H.M.; Helmy, H.M. Assessment of the Use of Natural Extracted Dyes and Pancreatin Enzyme for Dyeing of Four Natural Textiles: HPLC Analysis of Phytochemicals. Processes 2020, 8, 59. [Google Scholar] [CrossRef]
- Li, W.; Mu, B.; Yang, Y. Feasibility of Industrial-Scale Treatment of Dye Wastewater via Bio-Adsorption Technology. Bioresour. Technol. 2019, 277, 157–170. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Shao, Q.; Xu, C. Enhanced Azo Dye Removal from Wastewater by Coupling Sulfidated Zero-Valent Iron with a Chelator. J. Clean. Prod. 2019, 213, 753–761. [Google Scholar] [CrossRef]
- Singh, R.L.; Singh, P.K.; Singh, R.P. Enzymatic Decolorization and Degradation of Azo Dyes—A Review. Int. Biodeterior. Biodegrad. 2015, 104, 21–31. [Google Scholar] [CrossRef]
- Louati, I.; Elloumi-Mseddi, J.; Cheikhrouhou, W.; Hadrich, B.; Nasri, M.; Aifa, S.; Woodward, S.; Mechichi, T. Simultaneous Cleanup of Reactive Black 5 and Cadmium by a Desert Soil Bacterium. Ecotoxicol. Environ. Saf. 2020, 190, 110103. [Google Scholar] [CrossRef] [PubMed]
- Al-Tohamy, R.; Sun, J.; Fareed, M.F.; Kenawy, E.R.; Ali, S.S. Ecofriendly Biodegradation of Reactive Black 5 by Newly Isolated Sterigmatomyces Halophilus SSA1575, Valued for Textile Azo Dye Wastewater Processing and Detoxification. Sci. Rep. 2020, 10, 12370. [Google Scholar] [CrossRef]
- Liu, S.H.; Tsai, S.L.; Guo, P.Y.; Lin, C.W. Inducing Laccase Activity in White Rot Fungi Using Copper Ions and Improving the Efficiency of Azo Dye Treatment with Electricity Generation Using Microbial Fuel Cells. Chemosphere 2020, 243, 125304. [Google Scholar] [CrossRef]
- Shahid, M.; Wertz, J.; Degano, I.; Aceto, M.; Khan, M.I.; Quye, A. Analytical Methods for Determination of Anthraquinone Dyes in Historical Textiles: A Review. Anal. Chim. Acta 2019, 1083, 58–87. [Google Scholar] [CrossRef]
- Novotnỳ, Č.; Dias, N.; Kapanen, A.; Malachová, K.; Vándrovcová, M.; Itävaara, M.; Lima, N. Comparative Use of Bacterial, Algal and Protozoan Tests to Study Toxicity of Azo-and Anthraquinone Dyes. Chemosphere 2006, 63, 1436–1442. [Google Scholar] [CrossRef]
- Gürses, A.; Açıkyıldız, M.; Güneş, K.; Gürses, M.S. Classification of Dye and Pigments. In Dyes and Pigments; Gürses, A., Açıkyıldız, M., Güneş, K., Gürses, M.S., Eds.; Springer Briefs in Molecular Science; Springer International Publishing: Cham, Switzerland, 2016; pp. 31–45. ISBN 978-3-319-33892-7. [Google Scholar]
- Cao, D.J.; Wang, J.J.; Zhang, Q.; Wen, Y.Z.; Dong, B.; Liu, R.J.; Yang, X.; Geng, G. Biodegradation of Triphenylmethane Dye Crystal Violet by Cedecea Davisae. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2019, 210, 9–13. [Google Scholar] [CrossRef]
- Silva, M.C.; Corrêa, A.D.; Amorim, M.T.; Parpot, P.; Torres, J.A.; Chagas, P.M. Decolorization of the Phthalocyanine Dye Reactive Blue 21 by Turnip Peroxidase and Assessment of Its Oxidation Products. J. Mol. Catal. B Enzym. 2012, 77, 9–14. [Google Scholar] [CrossRef]
- Dindaş, G.B.; Şahin, Z.; Cengiz Yatmaz, H.; İşci, Ü. Cobalt Phthalocyanine-TiO2 Nanocomposites for Photocatalytic Remediation of Textile Dyes under Visible Light Irradiation. J. Porphyr. Phthalocyanines 2019, 23, 561–568. [Google Scholar] [CrossRef]
- Cesa, F.S.; Turra, A.; Baruque-Ramos, J. Synthetic Fibers as Microplastics in the Marine Environment: A Review from Textile Perspective with a Focus on Domestic Washings. Sci. Total Environ. 2017, 598, 1116–1129. [Google Scholar] [CrossRef]
- Almroth, B.M.; Åström, L.; Roslund, S.; Petersson, H.; Johansson, M.; Persson, N.K. Quantifying Shedding of Synthetic Fibers from Textiles; a Source of Microplastics Released into the Environment. Environ. Sci. Pollut. Res. 2018, 25, 1191–1199. [Google Scholar] [CrossRef]
- Meireles, G.; Daam, M.A.; Sanches, A.L.; Zanoni, M.V.; Soares, A.M.; Gravato, C.; de Oliveira, D.P. Red Disperse Dyes (DR 60, DR 73 and DR 78) at Environmentally Realistic Concentrations Impact Biochemical Profile of Early Life Stages of Zebrafish (Danio Rerio). Chem.-Biol. Interact. 2018, 292, 94–100. [Google Scholar] [CrossRef]
- Al-Etaibi, A.M.; El-Apasery, M.A. Dyeing Performance of Disperse Dyes on Polyester Fabrics Using Eco-Friendly Carrier and Their Antioxidant and Anticancer Activities. Int. J. Environ. Res. Public Health 2019, 16, 4603. [Google Scholar] [CrossRef]
- Bayramoglu, G.; Kunduzcu, G.; Arica, M.Y. Preparation and Characterization of Strong Cation Exchange Terpolymer Resin as Effective Adsorbent for Removal of Disperse Dyes. Polym. Eng. Sci. 2020, 60, 192–201. [Google Scholar] [CrossRef]
- Berradi, M.; Hsissou, R.; Khudhair, M.; Assouag, M.; Cherkaoui, O.; El Bachiri, A.; El Harfi, A. Textile Finishing Dyes and Their Impact on Aquatic Environs. Heliyon 2019, 5, e02711. [Google Scholar] [CrossRef] [PubMed]
- Ito, T.; Adachi, Y.; Yamanashi, Y.; Shimada, Y. Long–Term Natural Remediation Process in Textile Dye–Polluted River Sediment Driven by Bacterial Community Changes. Water Res. 2016, 100, 458–465. [Google Scholar] [CrossRef] [PubMed]
- Singh, L. Biodegradation of Synthetic Dyes: A Mycoremediation Approach for Degradation/Decolourization of Textile Dyes and Effluents. J. Appl. Biotechnol. Bioeng. 2017, 3, 430–435. [Google Scholar] [CrossRef]
- Yang, C.; Li, L.; Shi, J.; Long, C.; Li, A. Advanced Treatment of Textile Dyeing Secondary Effluent Using Magnetic Anion Exchange Resin and Its Effect on Organic Fouling in Subsequent RO Membrane. J. Hazard. Mater. 2015, 284, 50–57. [Google Scholar] [CrossRef]
- Yaseen, D.A.; Scholz, M. Shallow Pond Systems Planted with Lemna Minor Treating Azo Dyes. Ecol. Eng. 2016, 94, 295–305. [Google Scholar] [CrossRef]
- Gürses, A.; Açıkyıldız, M.; Güneş, K.; Gürses, M.S. Colorants in health and environmental aspects. In Dyes and Pigments; Springer: New York, NY, USA, 2016; pp. 69–83. [Google Scholar]
- Donkadokula, N.Y.; Kola, A.K.; Naz, I.; Saroj, D. A Review on Advanced Physico-Chemical and Biological Textile Dye Wastewater Treatment Techniques. Rev. Environ. Sci. Biotechnol. 2020, 19, 543–560. [Google Scholar] [CrossRef]
- Khandare, R.V.; Govindwar, S.P. Phytoremediation of Textile Dyes and Effluents: Current Scenario and Future Prospects. Biotechnol. Adv. 2015, 33, 1697–1714. [Google Scholar] [CrossRef] [PubMed]
- Tabish, T.A.; Memon, F.A.; Gomez, D.E.; Horsell, D.W.; Zhang, S. A Facile Synthesis of Porous Graphene for Efficient Water and Wastewater Treatment. Sci. Rep. 2018, 8, 1817. [Google Scholar] [CrossRef] [PubMed]
- Aldalbahi, A.; El-Naggar, M.E.; El-Newehy, M.H.; Rahaman, M.; Hatshan, M.R.; Khattab, T.A. Effects of Technical Textiles and Synthetic Nanofibers on Environmental Pollution. Polymers 2021, 13, 155. [Google Scholar] [CrossRef]
- Vikrant, K.; Giri, B.S.; Raza, N.; Roy, K.; Kim, K.H.; Rai, B.N.; Singh, R.S. Recent Advancements in Bioremediation of Dye: Current Status and Challenges. Bioresour. Technol. 2018, 253, 355–367. [Google Scholar] [CrossRef] [PubMed]
- Park, M.; Lee, K.S.; Shim, J.; Liu, Y.; Lee, C.; Cho, H.; Kim, M.J.; Park, S.J.; Yun, Y.J.; Kim, H.Y.; et al. Environment Friendly, Transparent Nanofiber Textiles Consolidated with High Efficiency PLEDs for Wearable Electronics. Org. Electron. 2016, 36, 89–96. [Google Scholar] [CrossRef]
- Muthu, S.S. Introduction. In Sustainability in the Textile Industry; Muthu, S.S., Ed.; Textile Science and Clothing Technology; Springer: Singapore, 2017; pp. 1–8. ISBN 978-981-10-2639-3. [Google Scholar]
- Eslami, H.; Shariatifar, A.; Rafiee, E.; Shiranian, M.; Salehi, F.; Hosseini, S.S.; Eslami, G.; Ghanbari, R.; Ebrahimi, A.A. Decolorization and Biodegradation of Reactive Red 198 Azo Dye by a New Enterococcus Faecalis–Klebsiella Variicola Bacterial Consortium Isolated from Textile Wastewater Sludge. World J. Microbiol. Biotechnol. 2019, 35, 38. [Google Scholar] [CrossRef]
- Imran, M.; Crowley, D.E.; Khalid, A.; Hussain, S.; Mumtaz, M.W.; Arshad, M. Microbial Biotechnology for Decolorization of Textile Wastewaters. Rev. Environ. Sci. Biotechnol. 2015, 14, 73–92. [Google Scholar] [CrossRef]
- Sakib, A.A.; Masum, S.M.; Hoinkis, J.; Islam, R.; Molla, M.; Islam, A. Synthesis of CuO/ZnO Nanocomposites and Their Application in Photodegradation of Toxic Textile Dye. J. Compos. Sci. 2019, 3, 91. [Google Scholar] [CrossRef]
- Kant, R. Textile Dyeing Industry an Environmental Hazard. Nat. Sci. 2011, 4, 17027. [Google Scholar] [CrossRef]
- Rovira, J.; Domingo, J.L. Human Health Risks Due to Exposure to Inorganic and Organic Chemicals from Textiles: A Review. Environ. Res. 2019, 168, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Yadav, A.K.; Jain, C.K.; Malik, D.S. Toxic Characterization of Textile Dyes and Effluents in Relation to Human Health Hazards. J. Sustain. Environ. Res. 2014, 3, 95–102. [Google Scholar]
- Ahmad, M.; Yousaf, M.; Nasir, A.; Bhatti, I.A.; Mahmood, A.; Fang, X.; Jian, X.; Kalantar-Zadeh, K.; Mahmood, N. Porous Eleocharis@MnPE Layered Hybrid for Synergistic Adsorption and Catalytic Biodegradation of Toxic Azo Dyes from Industrial Wastewater. Environ. Sci. Technol. 2019, 53, 2161–2170. [Google Scholar] [CrossRef] [PubMed]
- Dai, Q.; Zhang, S.; Liu, H.; Huang, J.; Li, L. Sulfide-Mediated Azo Dye Degradation and Microbial Community Analysis in a Single-Chamber Air Cathode Microbial Fuel Cell. Bioelectrochemistry 2020, 131, 107349. [Google Scholar] [CrossRef]
- George, G.; Saravanakumar, M.P. Facile Synthesis of Carbon-Coated Layered Double Hydroxide and Its Comparative Characterisation with Zn–Al LDH: Application on Crystal Violet and Malachite Green Dye Adsorption—Isotherm, Kinetics and Box-Behnken Design. Environ. Sci. Pollut. Res. 2018, 25, 30236–30254. [Google Scholar] [CrossRef]
- Arora, C.; Kumar, P.; Soni, S.; Mittal, J.; Mittal, A.; Singh, B. Efficient Removal of Malachite Green Dye from Aqueous Solution Using Curcuma Caesia Based Activated Carbon. Desalination Water Treat. 2020, 195, 341–352. [Google Scholar] [CrossRef]
- Ghasemi, M.; Mashhadi, S.; Asif, M.; Tyagi, I.; Agarwal, S.; Gupta, V.K. Microwave-Assisted Synthesis of Tetraethylenepentamine Functionalized Activated Carbon with High Adsorption Capacity for Malachite Green Dye. J. Mol. Liq. 2016, 213, 317–325. [Google Scholar] [CrossRef]
- Shedbalkar, U.; Jadhav, J.P. Detoxification of Malachite Green and Textile Industrial Effluent by Penicillium Ochrochloron. Biotechnol. Bioprocess Eng. 2011, 16, 196. [Google Scholar] [CrossRef]
- Chen, C.Y.; Kuo, J.T.; Cheng, C.Y.; Huang, Y.T.; Ho, I.H.; Chung, Y.C. Biological Decolorization of Dye Solution Containing Malachite Green by Pandoraea Pulmonicola YC32 Using a Batch and Continuous System. J. Hazard. Mater. 2009, 172, 1439–1445. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, T.; Das, M. Degradation of Malachite Green by Enterobacter Asburiae Strain XJUHX-4TM. CLEAN–Soil Air Water 2014, 42, 849–856. [Google Scholar] [CrossRef]
- Liu, Y.; Jin, J.H.; Zhou, Y.G.; Liu, H.C.; Liu, Z.P. Flavobacterium Caeni Sp. Nov., Isolated from a Sequencing Batch Reactor for the Treatment of Malachite Green Effluents. Int. J. Syst. Evol. Microbiol. 2010, 60, 417–421. [Google Scholar] [CrossRef]
- Muthukumaran, C.; Sivakumar, V.M.; Thirumarimurugan, M. Adsorption Isotherms and Kinetic Studies of Crystal Violet Dye Removal from Aqueous Solution Using Surfactant Modified Magnetic Nanoadsorbent. J. Taiwan Inst. Chem. Eng. 2016, 63, 354–362. [Google Scholar] [CrossRef]
- Fabryanty, R.; Valencia, C.; Soetaredjo, F.E.; Putro, J.N.; Santoso, S.P.; Kurniawan, A.; Ju, Y.H.; Ismadji, S. Removal of Crystal Violet Dye by Adsorption Using Bentonite–Alginate Composite. J. Environ. Chem. Eng. 2017, 5, 5677–5687. [Google Scholar] [CrossRef]
- Kumari, H.J.; Krishnamoorthy, P.; Arumugam, T.K.; Radhakrishnan, S.; Vasudevan, D. An Efficient Removal of Crystal Violet Dye from Waste Water by Adsorption onto TLAC/Chitosan Composite: A Novel Low Cost Adsorbent. Int. J. Biol. Macromol. 2017, 96, 324–333. [Google Scholar] [CrossRef]
- Wu, J.; Gao, H.; Yao, S.; Chen, L.; Gao, Y.; Zhang, H. Degradation of Crystal Violet by Catalytic Ozonation Using Fe/Activated Carbon Catalyst. Sep. Purif. Technol. 2015, 147, 179–185. [Google Scholar] [CrossRef]
- Mbacké, M.K.; Kane, C.; Diallo, N.O.; Diop, C.M.; Chauvet, F.; Comtat, M.; Tzedakis, T. Electrocoagulation Process Applied on Pollutants Treatment-Experimental Optimization and Fundamental Investigation of the Crystal Violet Dye Removal. J. Environ. Chem. Eng. 2016, 4, 4001–4011. [Google Scholar] [CrossRef]
- Parshetti, G.K.; Parshetti, S.G.; Telke, A.A.; Kalyani, D.C.; Doong, R.A.; Govindwar, S.P. Biodegradation of Crystal Violet by Agrobacterium Radiobacter. J. Environ. Sci. 2011, 23, 1384–1393. [Google Scholar] [CrossRef]
- Grassi, P.; Reis, C.; Drumm, F.C.; Georgin, J.; Tonato, D.; Escudero, L.B.; Kuhn, R.; Jahn, S.L.; Dotto, G.L. Biosorption of Crystal Violet Dye Using Inactive Biomass of the Fungus Diaporthe Schini. Water Sci. Technol. 2019, 79, 709–717. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, J.; Wattoo, F.H.; Wattoo, M.H.; Malik, R.; Tirmizi, S.A.; Imran, M.; Ghangro, A.B. Adsorption of Acid Yellow Dye on Flakes of Chitosan Prepared from Fishery Wastes. Arab. J. Chem. 2011, 4, 389–395. [Google Scholar] [CrossRef]
- Khan, J.; Sayed, M.; Ali, F.; Khan, H.M. Removal of Acid Yellow 17 Dye by Fenton Oxidation Process. Z. Phys. Chem. 2018, 232, 507–525. [Google Scholar] [CrossRef]
- Kashefialasl, M.; Khosravi, M.; Marandi, R.; Seyyedi, K. Treatment of Dye Solution Containing Colored Index Acid Yellow 36 by Electrocoagulation Using Iron Electrodes. Int. J. Environ. Sci. Technol. 2006, 2, 365. [Google Scholar]
- Jedynak, K.; Wideł, D.; Rędzia, N. Removal of Rhodamine b (a Basic Dye) and Acid Yellow 17 (an Acidic Dye) from Aqueous Solutions by Ordered Mesoporous Carbon and Commercial Activated Carbon. Colloids Interfaces 2019, 3, 30. [Google Scholar] [CrossRef]
- Lops, C.; Ancona, A.; Di Cesare, K.; Dumontel, B.; Garino, N.; Canavese, G.; Hérnandez, S.; Cauda, V. Sonophotocatalytic Degradation Mechanisms of Rhodamine B Dye via Radicals Generation by Micro-and Nano-Particles of ZnO. Appl. Catal. B Environ. 2019, 243, 629–640. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.; Zou, B.; Gao, W.; Liu, Q.; Wang, Z.; Guo, Y.; Wang, X.; Liu, Y. Adsorption of Rhodamine-B from Aqueous Solution Using Treated Rice Husk-Based Activated Carbon. Colloids Surf. A Physicochem. Eng. Asp. 2014, 446, 1–7. [Google Scholar] [CrossRef]
- Machado, Ê.L.; de Sales Dambros, V.; Kist, L.T.; Lobo, E.A.; Tedesco, S.B.; Moro, C.C. Use of Ozonization for the Treatment of Dye Wastewaters Containing Rhodamine B in the Agate Industry. Water Air Soil Pollut. 2012, 223, 1753–1764. [Google Scholar] [CrossRef]
- Khan, S.S.; Arunarani, A.; Chandran, P. Biodegradation of Basic Violet 3 and Acid Blue 93 by Pseudomonas Putida. CLEAN–Soil Air Water 2015, 43, 67–72. [Google Scholar] [CrossRef]
- Deivasigamani, C.; Das, N. Biodegradation of Basic Violet 3 by Candida Krusei Isolated from Textile Wastewater. Biodegradation 2011, 22, 1169–1180. [Google Scholar] [CrossRef]
- Abubakar, A.; Gaya, U. Box-Behnken-Optimized Cu (I) Photo-Fenton-like Degradation of Basic Violet 3. J. Mater. Environ. Sci. 2019, 10, 15–21. [Google Scholar]
- Sorbiun, M.; Mehr, E.S.; Ramazani, A.; Fardood, S.T. Biosynthesis of Ag, ZnO and Bimetallic Ag/ZnO Alloy Nanoparticles by Aqueous Extract of Oak Fruit Hull (Jaft) and Investigation of Photocatalytic Activity of ZnO and Bimetallic Ag/ZnO for Degradation of Basic Violet 3 Dye. J. Mater. Sci. Mater. Electron. 2018, 29, 2806–2814. [Google Scholar] [CrossRef]
- Satapanajaru, T.; Chompuchan, C.; Suntornchot, P.; Pengthamkeerati, P. Enhancing Decolorization of Reactive Black 5 and Reactive Red 198 during Nano Zerovalent Iron Treatment. Desalination 2011, 266, 218–230. [Google Scholar] [CrossRef]
- Nazari, P.; Setayesh, S.R. Effective Degradation of Reactive Red 195 via Heterogeneous Electro-Fenton Treatment: Theoretical Study and Optimization. Int. J. Environ. Sci. Technol. 2019, 16, 6329–6346. [Google Scholar] [CrossRef]
- Sonai, G.G.; de Souza, S.M.; de Oliveira, D.; de Souza, A.A. The Application of Textile Sludge Adsorbents for the Removal of Reactive Red 2 Dye. J. Environ. Manag. 2016, 168, 149–156. [Google Scholar] [CrossRef]
- Kalyani, D.C.; Telke, A.A.; Dhanve, R.S.; Jadhav, J.P. Ecofriendly Biodegradation and Detoxification of Reactive Red 2 Textile Dye by Newly Isolated Pseudomonas Sp. SUK1. J. Hazard. Mater. 2009, 163, 735–742. [Google Scholar] [CrossRef]
- Wang, H.; Su, J.Q.; Zheng, X.W.; Tian, Y.; Xiong, X.J.; Zheng, T.L. Bacterial Decolorization and Degradation of the Reactive Dye Reactive Red 180 by Citrobacter Sp. CK3. Int. Biodeterior. Biodegrad. 2009, 63, 395–399. [Google Scholar] [CrossRef]
- Moussavi, G.; Mahmoudi, M. Degradation and Biodegradability Improvement of the Reactive Red 198 Azo Dye Using Catalytic Ozonation with MgO Nanocrystals. Chem. Eng. J. 2009, 152, 1–7. [Google Scholar] [CrossRef]
- Zhang, F.; Yediler, A.; Liang, X.; Kettrup, A. Effects of Dye Additives on the Ozonation Process and Oxidation By-Products: A Comparative Study Using Hydrolyzed CI Reactive Red 120. Dyes Pigments 2004, 60, 1–7. [Google Scholar] [CrossRef]
- Saratale, R.G.; Saratale, G.D.; Chang, J.S.; Govindwar, S.P. Ecofriendly Degradation of Sulfonated Diazo Dye CI Reactive Green 19A Using Micrococcus Glutamicus NCIM-2168. Bioresour. Technol. 2009, 100, 3897–3905. [Google Scholar] [CrossRef]
- Sari, A.A.; Tachibana, S.; Muryanto; Hadibarata, T. Development of Bioreactor Systems for Decolorization of Reactive Green 19 Using White Rot Fungus. Desalination Water Treat. 2016, 57, 7029–7039. [Google Scholar] [CrossRef]
- Zuorro, A.; Lavecchia, R. Evaluation of UV/H2O2 Advanced Oxidation Process (AOP) for the Degradation of Diazo Dye Reactive Green 19 in Aqueous Solution. Desalination Water Treat. 2014, 52, 1571–1577. [Google Scholar] [CrossRef]
- Palma-Goyes, R.E.; Silva-Agredo, J.; Vazquez-Arenas, J.; Romero-Ibarra, I.; Torres-Palma, R.A. The Effect of Different Operational Parameters on the Electrooxidation of Indigo Carmine on Ti/IrO2-SnO2-Sb2O3. J. Environ. Chem. Eng. 2018, 6, 3010–3017. [Google Scholar] [CrossRef]
- Labiadh, L.; Barbucci, A.; Carpanese, M.P.; Gadri, A.; Ammar, S.; Panizza, M. Direct and Indirect Electrochemical Oxidation of Indigo Carmine Using PbO2 and TiRuSnO2. J. Solid State Electrochem. 2017, 21, 2167–2175. [Google Scholar] [CrossRef]
- Ramesh, T.N.; Kirana, D.V.; Ashwini, A.; Manasa, T.R. Calcium Hydroxide as Low Cost Adsorbent for the Effective Removal of Indigo Carmine Dye in Water. J. Saudi Chem. Soc. 2017, 21, 165–171. [Google Scholar] [CrossRef]
- Zapata-Castillo, P.; Villalonga-Santana, L.; Islas-Flores, I.; Rivera-Muñoz, G.; Ancona-Escalante, W.; Solís-Pereira, S. Synergistic Action of Laccases from Trametes Hirsuta Bm2 Improves Decolourization of Indigo Carmine. Lett. Appl. Microbiol. 2015, 61, 252–258. [Google Scholar] [CrossRef]
- Li, H.; Zhang, R.; Tang, L.; Zhang, J.; Mao, Z. Manganese Peroxidase Production from Cassava Residue by Phanerochaete Chrysosporium in Solid State Fermentation and Its Decolorization of Indigo Carmine. Chin. J. Chem. Eng. 2015, 23, 227–233. [Google Scholar] [CrossRef]
- Cho, E.A.; Seo, J.; Lee, D.W.; Pan, J.G. Decolorization of Indigo Carmine by Laccase Displayed on Bacillus Subtilis Spores. Enzym. Microb. Technol. 2011, 49, 100–104. [Google Scholar] [CrossRef]
- Lu, R.; Ma, L.; He, F.; Yu, D.; Fan, R.; Zhang, Y.; Long, Z.; Zhang, X.; Yang, Y. White-Rot Fungus Ganoderma Sp. En3 Had a Strong Ability to Decolorize and Tolerate the Anthraquinone, Indigo and Triphenylmethane Dye with High Concentrations. Bioprocess Biosyst. Eng. 2016, 39, 381–390. [Google Scholar] [CrossRef]
- Adnan, L.A.; Hadibarata, T.; Sathishkumar, P.; Mohd Yusoff, A.R. Biodegradation Pathway of Acid Red 27 by White-Rot Fungus Armillaria Sp. F022 and Phytotoxicity Evaluation. CLEAN–Soil Air Water 2016, 44, 239–246. [Google Scholar] [CrossRef]
- Jocic, D.; Vílchez, S.; Topalovic, T.; Molina, R.; Navarro, A.; Jovancic, P.; Julià, M.R.; Erra, P. Effect of Low-Temperature Plasma and Chitosan Treatment on Wool Dyeing with Acid Red 27. J. Appl. Polym. Sci. 2005, 97, 2204–2214. [Google Scholar] [CrossRef]
- Khandegar, V.; Saroha, A.K. Electrochemical Treatment of Textile Effluent Containing Acid Red 131 Dye. J. Hazard. Toxic Radioact. Waste 2014, 18, 38–44. [Google Scholar] [CrossRef]
- Jorfi, S.; Barzegar, G.; Ahmadi, M.; Soltani, R.D.; Takdastan, A.; Saeedi, R.; Abtahi, M. Enhanced Coagulation-Photocatalytic Treatment of Acid Red 73 Dye and Real Textile Wastewater Using UVA/Synthesized MgO Nanoparticles. J. Environ. Manag. 2016, 177, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Bhole, B.D.; Ganguly, B.; Madhuram, A.; Deshpande, D.; Joshi, J. Biosorption of Methyl Violet, Basic Fuchsin and Their Mixture Using Dead Fungal Biomass. Curr. Sci. 2004, 86, 1641–1645. [Google Scholar]
- Durmus, Z.; Kurt, B.Z.; Durmus, A. Synthesis and Characterization of Graphene Oxide/Zinc Oxide (GO/ZnO) Nanocomposite and Its Utilization for Photocatalytic Degradation of Basic Fuchsin Dye. ChemistrySelect 2019, 4, 271–278. [Google Scholar] [CrossRef]
- Gupta, V.K.; Mittal, A.; Gajbe, V.; Mittal, J. Adsorption of Basic Fuchsin Using Waste Materials—Bottom Ash and Deoiled Soya—As Adsorbents. J. Colloid Interface Sci. 2008, 319, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.H.; Geng, J.T.; Cai, J.C.; Cai, Y.F.; Cao, C.Y. Adsorption Performance of Basic Fuchsin on Alkali-Activated Diatomite. Adsorpt. Sci. Technol. 2020, 38, 151–167. [Google Scholar] [CrossRef]
- El Haddad, M. Removal of Basic Fuchsin Dye from Water Using Mussel Shell Biomass Waste as an Adsorbent: Equilibrium, Kinetics, and Thermodynamics. J. Taibah Univ. Sci. 2016, 10, 664–674. [Google Scholar] [CrossRef]
- Wang, L.; Wang, J.; Pan, H.; Zhao, M.; Chen, J. Kinetics and Removal Pathwayof Basic Fuchsin by Electrochemical Oxidization. J. Electroanal. Chem. 2021, 880, 114792. [Google Scholar] [CrossRef]
- Senthilkumar, S.; Perumalsamy, M.; Prabhu, H.J. Decolourization Potential of White-Rot Fungus Phanerochaete Chrysosporium on Synthetic Dye Bath Effluent Containing Amido Black 10B. J. Saudi Chem. Soc. 2014, 18, 845–853. [Google Scholar] [CrossRef]
- Angelova, R.; Baldikova, E.; Pospiskova, K.; Safarikova, M.; Safarik, I. Magnetically Modified Sheaths of Leptothrix Sp. as an Adsorbent for Amido Black 10B Removal. J. Magn. Magn. Mater. 2017, 427, 314–319. [Google Scholar] [CrossRef]
- Sun, J.H.; Sun, S.P.; Wang, G.L.; Qiao, L.P. Degradation of Azo Dye Amido Black 10B in Aqueous Solution by Fenton Oxidation Process. Dyes Pigments 2007, 74, 647–652. [Google Scholar] [CrossRef]
- Garg, A.; Mainrai, M.; Bulasara, D.V.; Barman, S. Experimental Investigation on Adsorption of Amido Black 10b Dye Onto Zeolite Synthesized from Fly Ash. Chem. Eng. Commun. 2015, 202, 123–130. [Google Scholar] [CrossRef]
- Ahmad, R.; Kumar, R. Conducting Polyaniline/Iron Oxide Composite: A Novel Adsorbent for the Removal of Amido Black 10B. J. Chem. Eng. Data 2010, 55, 3489–3493. [Google Scholar] [CrossRef]
- Tanzifi, M.; Yaraki, M.T.; Kiadehi, A.D.; Hosseini, S.H.; Olazar, M.; Bharti, A.K.; Agarwal, S.; Gupta, V.K.; Kazemi, A. Adsorption of Amido Black 10B from Aqueous Solution Using Polyaniline/SiO2 Nanocomposite: Experimental Investigation and Artificial Neural Network Modeling. J. Colloid Interface Sci. 2018, 510, 246–261. [Google Scholar] [CrossRef]
- Aragaw, T.A. Utilizations of Electro-Coagulated Sludge from Wastewater Treatment Plant Data as an Adsorbent for Direct Red 28 Dye Removal. Data Brief 2020, 28, 104848. [Google Scholar] [CrossRef]
- Ay, F.; Catalkaya, E.C.; Kargi, F. A Statistical Experiment Design Approach for Advanced Oxidation of Direct Red Azo-Dye by Photo-Fenton Treatment. J. Hazard. Mater. 2009, 162, 230–236. [Google Scholar] [CrossRef]
- Mokhtari-Shourijeh, Z.; Langari, S.; Montazerghaem, L.; Mahmoodi, N.M. Synthesis of Porous Aminated PAN/PVDF Composite Nanofibers by Electrospinning: Characterization and Direct Red 23 Removal. J. Environ. Chem. Eng. 2020, 8, 103876. [Google Scholar] [CrossRef]
- Safa, Y.; Bhatti, H.N. Biosorption of Direct Red-31 and Direct Orange-26 Dyes by Rice Husk: Application of Factorial Design Analysis. Chem. Eng. Res. Des. 2011, 89, 2566–2574. [Google Scholar] [CrossRef]
- Guendouz, S.; Khellaf, N.; Zerdaoui, M.; Ouchefoun, M. Biosorption of Synthetic Dyes (Direct Red 89 and Reactive Green 12) as an Ecological Refining Step in Textile Effluent Treatment. Environ. Sci. Pollut. Res. 2013, 20, 3822–3829. [Google Scholar] [CrossRef] [PubMed]
- Bayramoğlu, G.; Yakup Arıca, M. Biosorption of Benzidine Based Textile Dyes “Direct Blue 1 and Direct Red 128” Using Native and Heat-Treated Biomass of Trametes Versicolor. J. Hazard. Mater. 2007, 143, 135–143. [Google Scholar] [CrossRef]
- Silambarasan, S.; Vangnai, A.S. Biodegradation of 4-Nitroaniline by Plant-Growth Promoting Acinetobacter Sp. AVLB2 and Toxicological Analysis of Its Biodegradation Metabolites. J. Hazard. Mater. 2016, 302, 426–436. [Google Scholar] [CrossRef] [PubMed]
- Silambarasan, S.; Vangnai, A.S. Plant-Growth Promoting Candida Sp. AVGB4 with Capability of 4-Nitroaniline Biodegradation under Drought Stress. Ecotoxicol. Environ. Saf. 2017, 139, 472–480. [Google Scholar] [CrossRef] [PubMed]
- Das, P.; Ghosh, S.; Sen, M.B. Heterogeneous Catalytic Reduction of 4-Nitroaniline by RGO-Ni Nanocomposite for Water Resource Management. J. Mater. Sci. Mater. Electron. 2019, 30, 19731–19737. [Google Scholar] [CrossRef]
- Szyguła, A.; Guibal, E.; Palacín, M.A.; Ruiz, M.; Sastre, A.M. Removal of an Anionic Dye (Acid Blue 92) by Coagulation–Flocculation Using Chitosan. J. Environ. Manag. 2009, 90, 2979–2986. [Google Scholar] [CrossRef] [PubMed]
- Parsa, J.B.; Negahdar, S.H. Treatment of Wastewater Containing Acid Blue 92 Dye by Advanced Ozone-Based Oxidation Methods. Sep. Purif. Technol. 2012, 98, 315–320. [Google Scholar] [CrossRef]
- Saravanan, M.; Sambhamurthy, N.P.; Sivarajan, M. Treatment of Acid Blue 113 Dye Solution Using Iron Electrocoagulation. CLEAN–Soil Air Water 2010, 38, 565–571. [Google Scholar] [CrossRef]
- Hairom, N.H.; Mohammad, A.W.; Kadhum, A.A. Effect of Various Zinc Oxide Nanoparticles in Membrane Photocatalytic Reactor for Congo Red Dye Treatment. Sep. Purif. Technol. 2014, 137, 74–81. [Google Scholar] [CrossRef]
- Vimonses, V.; Lei, S.; Jin, B.; Chow, C.W.; Saint, C. Kinetic Study and Equilibrium Isotherm Analysis of Congo Red Adsorption by Clay Materials. Chem. Eng. J. 2009, 148, 354–364. [Google Scholar] [CrossRef]
- Gharbani, P.; Tabatabaii, S.M.; Mehrizad, A. Removal of Congo Red from Textile Wastewater by Ozonation. Int. J. Environ. Sci. Technol. 2008, 5, 495–500. [Google Scholar] [CrossRef]
- Fu, Y.; Viraraghavan, T. Removal of Congo Red from an Aqueous Solution by Fungus Aspergillus Niger. Adv. Environ. Res. 2002, 7, 239–247. [Google Scholar] [CrossRef]
- Kishor, R.; Purchase, D.; Saratale, G.D.; Ferreira, L.F.; Bilal, M.; Iqbal, H.M.; Bharagava, R.N. Environment Friendly Degradation and Detoxification of Congo Red Dye and Textile Industry Wastewater by a Newly Isolated Bacillus Cohnni (RKS9). Environ. Technol. Innov. 2021, 22, 101425. [Google Scholar] [CrossRef]
- Sarayu, K.; Sandhya, S. Aerobic Biodegradation Pathway for Remazol Orange by Pseudomonas Aeruginosa. Appl. Biochem. Biotechnol. 2010, 160, 1241–1253. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.M.; Asiri, A.M.; Youssef, T.E.; Marwani, H.M. Photocatalytic Degradation of Remazol Brilliant Orange 3R Using Wet-Chemically Prepared CdO-ZnO Nanofibers for Environmental Remediation. Mater. Express 2016, 6, 137–148. [Google Scholar] [CrossRef]
- Lin, J.; Zhang, X.; Li, Z.; Lei, L. Biodegradation of Reactive Blue 13 in a Two-Stage Anaerobic/Aerobic Fluidized Beds System with a Pseudomonas Sp. Isolate. Bioresour. Technol. 2010, 101, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Tizaoui, C.; Grima, N. Kinetics of the Ozone Oxidation of Reactive Orange 16 Azo-Dye in Aqueous Solution. Chem. Eng. J. 2011, 173, 463–473. [Google Scholar] [CrossRef]
- KARAMAN, C.; Zümriye, A. Modelling of Remazol Black-B Adsorption on Chemically Modified Waste Orange Peel: PH Shifting Effect of Acidic Treatment. Sak. Üniversitesi Fen Bilimleri Enstitüsü Derg. 2020, 24, 1127–1142. [Google Scholar] [CrossRef]
- Jadhav, S.U.; Jadhav, M.U.; Kagalkar, A.N.; Govindwar, S.P. Decolorization of Brilliant Blue G Dye Mediated by Degradation of the Microbial Consortium of Galactomyces Geotrichum and Bacillus Sp. J. Chin. Inst. Chem. Eng. 2008, 39, 563–570. [Google Scholar] [CrossRef]
- Mafra, M.R.; Igarashi-Mafra, L.; Zuim, D.R.; Vasques, E.C.; Ferreira, M.A. Adsorption of Remazol Brilliant Blue on an Orange Peel Adsorbent. Braz. J. Chem. Eng. 2013, 30, 657–665. [Google Scholar] [CrossRef]
- Lazim, Z.M.; Mazuin, E.; Hadibarata, T.; Yusop, Z. The Removal of Methylene Blue and Remazol Brilliant Blue R Dyes by Using Orange Peel and Spent Tea Leaves. J. Teknol. 2015, 74. [Google Scholar] [CrossRef]
- Hamzeh, Y.; Ashori, A.; Azadeh, E.; Abdulkhani, A. Removal of Acid Orange 7 and Remazol Black 5 Reactive Dyes from Aqueous Solutions Using a Novel Biosorbent. Mater. Sci. and Eng. C 2012, 32, 1394–1400. [Google Scholar] [CrossRef]
- Kousha, M.; Daneshvar, E.; Sohrabi, M.S.; Jokar, M.; Bhatnagar, A. Adsorption of Acid Orange II Dye by Raw and Chemically Modified Brown Macroalga Stoechospermum Marginatum. Chem. Eng. J. 2012, 192, 67–76. [Google Scholar] [CrossRef]
- Daneshvar, E.; Sohrabi, M.S.; Kousha, M.; Bhatnagar, A.; Aliakbarian, B.; Converti, A.; Norrström, A.C. Shrimp Shell as an Efficient Bioadsorbent for Acid Blue 25 Dye Removal from Aqueous Solution. J. Taiwan Inst. Chem. Eng. 2014, 45, 2926–2934. [Google Scholar] [CrossRef]
- Umar, A.; Khan, M.S.; Alam, S.; Zekker, I.; Burlakovs, J.; Bhowmick, G.D.; Kallistova, A.; Pimenov, N.; Zahoor, M. Synthesis and Characterization of Pd-Ni Bimetallic Nanoparticles as Efficient Adsorbent for the Removal of Acid Orange 8 Present in Wastewater. Water 2021, 13, 1095. [Google Scholar] [CrossRef]
- Alam, S.; Khan, M.S.; Umar, A.; Khattak, R.; Zekker, I.; Burlakovs, J.; Ghangrekar, M.M.; Bhowmick, G.D.; Kallistova, A.; Pimenov, N. Preparation of Pd–Ni Nanoparticles Supported on Activated Carbon for Efficient Removal of Basic Blue 3 from Water. Water 2021, 13, 1211. [Google Scholar] [CrossRef]
- Jabeen, S.; Sufaid Khan, M.; Khattak, R.; Zekker, I.; Burlakovs, J.; Ghangrekar, M.M.; Kallistova, A.; Pimenov, N.; Zahoor, M.; Khan, G.S. Palladium-Supported Zirconia-Based Catalytic Degradation of Rhodamine-B Dye from Wastewater. Water 2021, 13, 1522. [Google Scholar] [CrossRef]
- Alam, S.; Khan, M.S.; Bibi, W.; Zekker, I.; Burlakovs, J.; Ghangrekar, M.M.; Bhowmick, G.D.; Kallistova, A.; Pimenov, N.; Zahoor, M. Preparation of Activated Carbon from the Wood of Paulownia Tomentosa as an Efficient Adsorbent for the Removal of Acid Red 4 and Methylene Blue Present in Wastewater. Water 2021, 13, 1453. [Google Scholar] [CrossRef]
- Galán, J.; Rodríguez, A.; Gómez, J.M.; Allen, S.J.; Walker, G.M. Reactive Dye Adsorption onto a Novel Mesoporous Carbon. Chem. Eng. J. 2013, 219, 62–68. [Google Scholar] [CrossRef]
- Holkar, C.R.; Jadhav, A.J.; Pinjari, D.V.; Mahamuni, N.M.; Pandit, A.B. A Critical Review on Textile Wastewater Treatments: Possible Approaches. J. Environ. Manag. 2016, 182, 351–366. [Google Scholar] [CrossRef]
- Rápó, E.; Aradi, L.E.; Szabó, Á.; Posta, K.; Szép, R.; Tonk, S. Adsorption of Remazol Brilliant Violet-5R Textile Dye from Aqueous Solutions by Using Eggshell Waste Biosorbent. Sci. Rep. 2020, 10, 8385. [Google Scholar] [CrossRef]
- El Malah, T.; Nour, H.F.; Radwan, E.K.; Abdel Mageid, R.E.; Khattab, T.A.; Olson, M.A. A Bipyridinium-Based Polyhydrazone Adsorbent That Exhibits Ultrahigh Adsorption Capacity for the Anionic Azo Dye, Direct Blue 71. Chem. Eng. J. 2021, 409, 128195. [Google Scholar] [CrossRef]
- Ruan, W.; Hu, J.; Qi, J.; Hou, Y.; Zhou, C.; Wei, X. Removal Of Dyes From Wastewater By Nanomaterials: A Review. Adv. Mater. Lett. 2019, 10, 9–20. [Google Scholar] [CrossRef]
- Yogalakshmi, K.N.; Das, A.; Rani, G.; Jaswal, V.; Randhawa, J.S. Nano-bioremediation: A new age technology for the treatment of dyes in textile effluents. In Bioremediation of Industrial Waste for Environmental Safety; Springer: New York, NY, USA, 2020; pp. 313–347. [Google Scholar]
- Dhand, C.; Dwivedi, N.; Loh, X.J.; Ying, A.N.; Verma, N.K.; Beuerman, R.W.; Lakshminarayanan, R.; Ramakrishna, S. Methods and Strategies for the Synthesis of Diverse Nanoparticles and Their Applications: A Comprehensive Overview. Rsc Adv. 2015, 5, 105003–105037. [Google Scholar] [CrossRef]
- Krishnan, S.K.; Subbiah, K.; Kandasamy, S.; Subramaniam, K. Application of Metal Nanoparticles for Textile Dye Remediation. In Proceedings of the Sustainable Development in Energy and Environment; Sivasubramanian, V., Pugazhendhi, A., Moorthy, I.G., Eds.; Springer: Singapore, 2020; pp. 217–223. [Google Scholar]
- Tara, N.; Siddiqui, S.I.; Rathi, G.; Chaudhry, S.A.; Asiri, A.M. Nano-Engineered Adsorbent for the Removal of Dyes from Water: A Review. Curr. Anal. Chem. 2020, 16, 14–40. [Google Scholar] [CrossRef]
- Ali, Z.; Ahmad, R. Nanotechnology for Water Treatment. In Environmental Nanotechnology Volume 3; Dasgupta, N., Ranjan, S., Lichtfouse, E., Eds.; Environmental Chemistry for a Sustainable World; Springer International Publishing: Cham, Switzerland, 2020; pp. 143–163. ISBN 978-3-030-26672-1. [Google Scholar]
- Sadegh, H.; Ali, G.A.; Gupta, V.K.; Makhlouf, A.S.; Shahryari-Ghoshekandi, R.; Nadagouda, M.N.; Sillanpää, M.; Megiel, E. The Role of Nanomaterials as Effective Adsorbents and Their Applications in Wastewater Treatment. J. Nanostruct. Chem. 2017, 7, 1–14. [Google Scholar] [CrossRef]
- Wang, T.; Lin, J.; Chen, Z.; Megharaj, M.; Naidu, R. Green Synthesized Iron Nanoparticles by Green Tea and Eucalyptus Leaves Extracts Used for Removal of Nitrate in Aqueous Solution. J. Clean. Prod. 2014, 83, 413–419. [Google Scholar] [CrossRef]
- Nagajyothi, P.C.; Vattikuti, S.V.; Devarayapalli, K.C.; Yoo, K.; Shim, J.; Sreekanth, T.V. Green Synthesis: Photocatalytic Degradation of Textile Dyes Using Metal and Metal Oxide Nanoparticles-Latest Trends and Advancements. Crit. Rev. Environ. Sci. Technol. 2020, 50, 2617–2723. [Google Scholar] [CrossRef]
- Pal, U.; Sandoval, A.; Madrid, S.I.; Corro, G.; Sharma, V.; Mohanty, P. Mixed Titanium, Silicon, and Aluminum Oxide Nanostructures as Novel Adsorbent for Removal of Rhodamine 6G and Methylene Blue as Cationic Dyes from Aqueous Solution. Chemosphere 2016, 163, 142–152. [Google Scholar] [CrossRef]
- Kalia, A.; Singh, S. Myco-Decontamination of Azo Dyes: Nano-Augmentation Technologies. 3 Biotech 2020, 10, 384. [Google Scholar] [CrossRef]
- Sahoo, T.; Sahu, J.R.; Panda, J.; Hembram, M.; Sahoo, S.K.; Sahu, R. Nanotechnology: An Efficient Technique of Contaminated Water Treatment. In Contaminants in Drinking and Wastewater Sources: Challenges and Reigning Technologies; Kumar, M., Snow, D.D., Honda, R., Mukherjee, S., Eds.; Springer Transactions in Civil and Environmental Engineering; Springer: Singapore, 2021; pp. 251–270. ISBN 9789811545993. [Google Scholar]
- Gupta, M.K.; Tandon, P.K.; Shukla, N. Nanotechnology: Environmentally Sustainable Solutions for Water Treatment. In Nanostructured Materials for Treating Aquatic Pollution; Springer: New York, NY, USA, 2019; pp. 225–242. [Google Scholar]
- Chu, T.P.; Nguyen, N.T.; Vu, T.L.; Dao, T.H.; Dinh, L.C.; Nguyen, H.L.; Hoang, T.H.; Le, T.S.; Pham, T.D. Synthesis, Characterization, and Modification of Alumina Nanoparticles for Cationic Dye Removal. Materials 2019, 12, 450. [Google Scholar] [CrossRef]
- Sahinkaya, E.; Sahin, A.; Yurtsever, A.; Kitis, M. Concentrate Minimization and Water Recovery Enhancement Using Pellet Precipitator in a Reverse Osmosis Process Treating Textile Wastewater. J. Environ. Manag. 2018, 222, 420–427. [Google Scholar] [CrossRef]
- Long, Q.; Zhang, Z.; Qi, G.; Wang, Z.; Chen, Y.; Liu, Z.Q. Fabrication of Chitosan Nanofiltration Membranes by the Film Casting Strategy for Effective Removal of Dyes/Salts in Textile Wastewater. ACS Sustain. Chem. Eng. 2020, 8, 2512–2522. [Google Scholar] [CrossRef]
- Jin, P.; Zhu, J.; Yuan, S.; Zhang, G.; Volodine, A.; Tian, M.; Wang, J.; Luis, P.; Van der Bruggen, B. Erythritol-Based Polyester Loose Nanofiltration Membrane with Fast Water Transport for Efficient Dye/Salt Separation. Chem. Eng. J. 2021, 406, 126796. [Google Scholar] [CrossRef]
- Yin, H.; Qiu, P.; Qian, Y.; Kong, Z.; Zheng, X.; Tang, Z.; Guo, H. Textile Wastewater Treatment for Water Reuse: A Case Study. Processes 2019, 7, 34. [Google Scholar] [CrossRef]
- Ji, J.; Kulshreshtha, S.; Kakade, A.; Majeed, S.; Li, X.; Liu, P. Bioaugmentation of Membrane Bioreactor with Aeromonas Hydrophila LZ-MG14 for Enhanced Malachite Green and Hexavalent Chromium Removal in Textile Wastewater. Int. Biodeterior. Biodegrad. 2020, 150, 104939. [Google Scholar] [CrossRef]
- Rashidi, H.R.; Sulaiman, N.M.; Hashim, N.A.; Hassan, C.R.; Ramli, M.R. Synthetic Reactive Dye Wastewater Treatment by Using Nano-Membrane Filtration. Desalination Water Treat. 2015, 55, 86–95. [Google Scholar] [CrossRef]
- Liang, P.; Rivallin, M.; Cerneaux, S.; Lacour, S.; Petit, E.; Cretin, M. Coupling Cathodic Electro-Fenton Reaction to Membrane Filtration for AO7 Dye Degradation: A Successful Feasibility Study. J. Membr. Sci. 2016, 510, 182–190. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, J.; Lu, M.; Liang, L.; Zhang, H.; Wei, J. Biosynthesis Based Membrane Filtration Coupled with Iron Nanoparticles Reduction Process in Removal of Dyes. Chem. Eng. J. 2020, 387, 124202. [Google Scholar] [CrossRef]
- Hassan, M.M.; Carr, C.M. A Critical Review on Recent Advancements of the Removal of Reactive Dyes from Dyehouse Effluent by Ion-Exchange Adsorbents. Chemosphere 2018, 209, 201–219. [Google Scholar] [CrossRef] [PubMed]
- Yurtsever, A.; Basaran, E.; Uçar, D. Process Optimization and Filtration Performance of an Anaerobic Dynamic Membrane Bioreactor Treating Textile Wastewaters. J. Environ. Manag. 2020, 273, 111114. [Google Scholar] [CrossRef] [PubMed]
- Arslan, S.; Eyvaz, M.; Gürbulak, E.; Yüksel, E. A Review of State-of-the-Art Technologies in Dye-Containing Wastewater Treatment—The Textile Industry Case. Text. Wastewater Treat. 2016. [Google Scholar] [CrossRef]
- Marin, N.M.; Pascu, L.F.; Demba, A.; Nita-Lazar, M.; Badea, I.A.; Aboul-Enein, H.Y. Removal of the Acid Orange 10 by Ion Exchange and Microbiological Methods. Int. J. Environ. Sci. Technol. 2019, 16, 6357–6366. [Google Scholar] [CrossRef]
- Joseph, J.; Radhakrishnan, R.C.; Johnson, J.K.; Joy, S.P.; Thomas, J. Ion-Exchange Mediated Removal of Cationic Dye-Stuffs from Water Using Ammonium Phosphomolybdate. Mater. Chem. Phys. 2020, 242, 122488. [Google Scholar] [CrossRef]
- Raghu, S.; Basha, C.A. Chemical or Electrochemical Techniques, Followed by Ion Exchange, for Recycle of Textile Dye Wastewater. J. Hazard. Mater. 2007, 149, 324–330. [Google Scholar] [CrossRef] [PubMed]
- Cseri, L.; Topuz, F.; Abdulhamid, M.A.; Alammar, A.; Budd, P.M.; Szekely, G. Electrospun Adsorptive Nanofibrous Membranes from Ion Exchange Polymers to Snare Textile Dyes from Wastewater. Adv. Mater. Technol. 2021, 2000955. [Google Scholar] [CrossRef]
- Babu, D.S.; Srivastava, V.; Nidheesh, P.V.; Kumar, M.S. Detoxification of Water and Wastewater by Advanced Oxidation Processes. Sci. Total Environ. 2019, 696, 133961. [Google Scholar] [CrossRef]
- Al-Sakkaf, B.M.; Nasreen, S.; Ejaz, N. Degradation Pattern of Textile Effluent by Using Bio and Sono Chemical Reactor. Available online: https://www.hindawi.com/journals/jchem/2020/8965627/ (accessed on 10 February 2021).
- Nidheesh, P.V.; Rajan, R. Removal of Rhodamine B from a Water Medium Using Hydroxyl and Sulphate Radicals Generated by Iron Loaded Activated Carbon. RSC Adv. 2016, 6, 5330–5340. [Google Scholar] [CrossRef]
- Javaid, R.; Qazi, U.Y. Catalytic Oxidation Process for the Degradation of Synthetic Dyes: An Overview. Int. J. Environ. Res. Public Health 2019, 16, 2066. [Google Scholar] [CrossRef]
- Zhang, L.P.; Liu, Z.; Faraj, Y.; Zhao, Y.; Zhuang, R.; Xie, R.; Ju, X.J.; Wang, W.; Chu, L.Y. High-Flux Efficient Catalytic Membranes Incorporated with Iron-Based Fenton-like Catalysts for Degradation of Organic Pollutants. J. Membr. Sci. 2019, 573, 493–503. [Google Scholar] [CrossRef]
- Lyu, L.; Han, M.; Cao, W.; Gao, Y.; Zeng, Q.; Yu, G.; Huang, X.; Hu, C. Efficient Fenton-like Process for Organic Pollutant Degradation on Cu-Doped Mesoporous Polyimide Nanocomposites. Environ. Sci. Nano 2019, 6, 798–808. [Google Scholar] [CrossRef]
- Ameta, R.; Chohadia, A.K.; Jain, A.; Punjabi, P.B. Chapter 3—Fenton and Photo-Fenton Processes. In Advanced Oxidation Processes for Waste Water Treatment; Ameta, S.C., Ameta, R., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 49–87. ISBN 978-0-12-810499-6. [Google Scholar]
- Tarkwa, J.B.; Oturan, N.; Acayanka, E.; Laminsi, S.; Oturan, M.A. Photo-Fenton Oxidation of Orange G Azo Dye: Process Optimization and Mineralization Mechanism. Environ. Chem. Lett. 2019, 17, 473–479. [Google Scholar] [CrossRef]
- Dotto, J.; Fagundes-Klen, M.R.; Veit, M.T.; Palácio, S.M.; Bergamasco, R. Performance of Different Coagulants in the Coagulation/Flocculation Process of Textile Wastewater. J. Clean. Prod. 2019, 208, 656–665. [Google Scholar] [CrossRef]
- GilPavas, E.; Dobrosz-Gómez, I.; Gómez-García, M.Á. Coagulation-Flocculation Sequential with Fenton or Photo-Fenton Processes as an Alternative for the Industrial Textile Wastewater Treatment. J. Environ. Manag. 2017, 191, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Chen, H.; Yuan, R.; Wang, F.; Ma, F.; Zhou, B. Intensified Degradation of Textile Wastewater Using a Novel Treatment of Hydrodynamic Cavitation with the Combination of Ozone. J. Environ. Chem. Eng. 2020, 8, 103959. [Google Scholar] [CrossRef]
- Siddique, K.; Rizwan, M.; Shahid, M.J.; Ali, S.; Ahmad, R.; Rizvi, H. Textile Wastewater Treatment Options: A Critical Review. Enhancing Cleanup Environ. Pollut. 2017, 183–207. [Google Scholar]
- Su, Z.; Liu, T.; Yu, W.; Li, X.; Graham, N.J. Coagulation of Surface Water: Observations on the Significance of Biopolymers. Water Res. 2017, 126, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Miralles-Cuevas, S.; Oller, I.; Agüera, A.; Llorca, M.; Pérez, J.S.; Malato, S. Combination of Nanofiltration and Ozonation for the Remediation of Real Municipal Wastewater Effluents: Acute and Chronic Toxicity Assessment. J. Hazard. Mater. 2017, 323, 442–451. [Google Scholar] [CrossRef]
- Yang, Y.C.; Zeng, S.S.; Ouyang, Y.; Sang, L.; Yang, S.Y.; Zhang, X.Q.; Huang, Y.Y.; Ye, J.; Xiao, M.T.; Zhang, N. An Intensified Ozonation System in a Tank Reactor with Foam Block Stirrer: Synthetic Textile Wastewater Treatment and Mass Transfer Modeling. Sep. Purif. Technol. 2021, 257, 117909. [Google Scholar] [CrossRef]
- Powar, A.; Perwuelz, A.; Behary, N.; Hoang, L.V.; Aussenac, T.; Loghin, C.; Maier, S.S.; Guan, J.; Chen, G. Environmental Profile Study of Ozone Decolorization of Reactive Dyed Cotton Textiles by Utilizing Life Cycle Assessment. Sustainability 2021, 13, 1225. [Google Scholar] [CrossRef]
- Kurade, M.B.; Waghmode, T.R.; Patil, S.M.; Jeon, B.H.; Govindwar, S.P. Monitoring the Gradual Biodegradation of Dyes in a Simulated Textile Effluent and Development of a Novel Triple Layered Fixed Bed Reactor Using a Bacterium-Yeast Consortium. Chem. Eng. J. 2017, 307, 1026–1036. [Google Scholar] [CrossRef]
- Song, L.; Shao, Y.; Ning, S.; Tan, L. Performance of a Newly Isolated Salt-Tolerant Yeast Strain Pichia Occidentalis G1 for Degrading and Detoxifying Azo Dyes. Bioresour. Technol. 2017, 233, 21–29. [Google Scholar] [CrossRef]
- Brahmbhatt, N.H.; Jasrai, R.T. The Role of Algae in Bioremediation of Textile Effluent. Methods 2016, 21, 28. [Google Scholar]
- Das, S.; Dash, H.R. Handbook of Metal-Microbe Interactions and Bioremediation; CRC Press: Boca Raton, FL, USA, 2017. [Google Scholar]
- AI-Jawhari, I.F. Decolorization of Methylene Blue and Crystal Violet by Some Filamentous Fungi. Int. J. Environ. Bioremediat. Biodegrad. 2015, 3, 62–65. [Google Scholar]
- Kaushik, P.; Malik, A. Mycoremediation of Synthetic Dyes: An Insight into the Mechanism, Process Optimization and Reactor Design. In Microbial Degradation of Synthetic Dyes in Wastewaters; Singh, S.N., Ed.; Environmental Science and Engineering; Springer International Publishing: Cham, Switzerland, 2015; pp. 1–25. ISBN 978-3-319-10942-8. [Google Scholar]
- Varjani, S.; Rakholiya, P.; Ng, H.Y.; You, S.; Teixeira, J.A. Microbial Degradation of Dyes: An Overview. Bioresour. Technol. 2020, 314, 123728. [Google Scholar] [CrossRef] [PubMed]
- Rekik, H.; Zaraî Jaouadi, N.; Bouacem, K.; Zenati, B.; Kourdali, S.; Badis, A.; Annane, R.; Bouanane-Darenfed, A.; Bejar, S.; Jaouadi, B. Physical and Enzymatic Properties of a New Manganese Peroxidase from the White-Rot Fungus Trametes Pubescens Strain I8 for Lignin Biodegradation and Textile-Dyes Biodecolorization. Int. J. Biol. Macromol. 2019, 125, 514–525. [Google Scholar] [CrossRef]
- Srinivasan, S.; Sadasivam, S.K.; Gunalan, S.; Shanmugam, G.; Kothandan, G. Application of Docking and Active Site Analysis for Enzyme Linked Biodegradation of Textile Dyes. Environ. Pollut. 2019, 248, 599–608. [Google Scholar] [CrossRef] [PubMed]
- Giovanella, P.; Vieira, G.A.; Ramos Otero, I.V.; Pais Pellizzer, E.; de Jesus Fontes, B.; Sette, L.D. Metal and Organic Pollutants Bioremediation by Extremophile Microorganisms. J. Hazard. Mater. 2020, 382, 121024. [Google Scholar] [CrossRef] [PubMed]
- Ji, D.; Xiao, C.; Zhao, J.; Chen, K.; Zhou, F.; Gao, Y.; Zhang, T.; Ling, H. Green Preparation of Polyvinylidene Fluoride Loose Nanofiltration Hollow Fiber Membranes with Multilayer Structure for Treating Textile Wastewater. Sci. Total Environ. 2021, 754, 141848. [Google Scholar] [CrossRef]
- Khan, R.; Bhawana, P.; Fulekar, M.H. Microbial Decolorization and Degradation of Synthetic Dyes: A Review. Rev. Environ. Sci. Biotechnol. 2013, 12, 75–97. [Google Scholar] [CrossRef]
- Maharani, V.; Vijayalakshmi, S.; Balasubramanian, T. Degradation and Detoxification of Reactive Azo Dyes by Native Bacterial Communities. Afr. J. Microbiol. Res. 2013, 7, 2274–2282. [Google Scholar]
- Saratale, R.G.; Gandhi, S.S.; Purankar, M.V.; Kurade, M.B.; Govindwar, S.P.; Oh, S.E.; Saratale, G.D. Decolorization and Detoxification of Sulfonated Azo Dye CI Remazol Red and Textile Effluent by Isolated Lysinibacillus Sp. RGS. J. Biosci. Bioeng. 2013, 115, 658–667. [Google Scholar] [CrossRef]
- Abdulrazzaq, N.N.; Al-Sabbagh, B.H.; Shanshool, H.A. Coupling of Electrocoagulation and Microflotation for the Removal of Textile Dyes from Aqueous Solutions. J. Water Process Eng. 2021, 40, 101906. [Google Scholar] [CrossRef]
- Logroño, W.; Pérez, M.; Urquizo, G.; Kadier, A.; Echeverría, M.; Recalde, C.; Rákhely, G. Single Chamber Microbial Fuel Cell (SCMFC) with a Cathodic Microalgal Biofilm: A Preliminary Assessment of the Generation of Bioelectricity and Biodegradation of Real Dye Textile Wastewater. Chemosphere 2017, 176, 378–388. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, A.; Rai, B.N.; Singh, R.S.; Jaiswal, R.P. A Comprehensive Review on the Integration of Advanced Oxidation Processes with Biodegradation for the Treatment of Textile Wastewater Containing Azo Dyes. Rev. Chem. Eng. 2021, 1. [Google Scholar] [CrossRef]
| Textile Dyes | Treatment Methods | Type of the Treatment Methods | References |
|---|---|---|---|
| Malachite green | Adsorption with curcuma-based activated carbon | Physical | [65] |
| Adsorption with tetraethylenepentamine functionalized activated carbon | [66] | ||
| Penicillim ochrochloron | Biological | [67] | |
| Pandoraea pulmonicola YC32 | [68] | ||
| Enterobacter asburiae XJUHX-4TM | [69] | ||
| Flavobacterium caeni sp. | [70] | ||
| Crystal violet and malachite green | Adsorption with carbon-coated layered double hydroxide | Physical | [64] |
| Crystal violet | Surfactant modified magnetic nanoadsorbent | Physical | [71] |
| Adsorption with bentonite-alginate composite | [72] | ||
| Adsorption onto TLAC/Chitosan composite | [73] | ||
| Ozonation | Chemical | [74] | |
| Electrociagulation | [75] | ||
| Agrobacterium radiobacter | Biological | [76] | |
| Diaporthe schini | [77] | ||
| Acid yellow | Adsorption on flakes of chitosan | Physical | [78] |
| Fenton oxidation | [79] | ||
| Electrocoagulation using iron electrolites | Chemical | [80] | |
| Rhodamine B and Acid yellow | Adsorption by ordered mesoporous carbon and commercial activated carbon | Physical | [81] |
| Rhodamine B | Adsorption by micro and nano-particles of ZnO | Physical | [82] |
| Adsorption with treated rice husk-based activated carbon | [83] | ||
| Ozonation | Chemical | [84] | |
| Basic violet and Acid blue 93 | Pseudomonas putida | Biological | [85] |
| Basic violet 3 | Candida krusei | Biological | [86] |
| Fenton oxidation | Physical | [87] | |
| Ag, ZnO and bimetallic Ag/ZnO alloy nanoparticles | [88] | ||
| Reactive Black 5 and Reactive red | Nano zerovalent iron treatment | Physical | [89] |
| Reactive red 195 | Electro-fenton | Physical | [90] |
| Reactive red 2 | Adsorption sludge | Physical | [91] |
| Pseudomonas sp. SUK1 | Biological | [92] | |
| Reactive red 180 | Citrobacter sp. CK3 | Biological | [93] |
| Reactive red 198 | Catalytic ozonation | Chemical | [94] |
| Reactive red 120 | Ozonation | Chemical | [95] |
| Reactive green | Micrococcus glutamicus NCIM-2168 | Biological | [96] |
| White rot fungus | [97] | ||
| UV/H2O2 advanced oxidation process (AOP) | Physical | [98] | |
| Indigo carmine | Electrooxidation on Ti/IrO2-SnO2-Sb2O3 | Physical | [99] |
| Electrochemical oxidation | [100] | ||
| Adsorption with calcium hydroxide | [101] | ||
| Trametes hirsuta laccase production | Biological | [102] | |
| Phanerochaete chrysosporium manganese peroxidase production | [103] | ||
| Bacillus amyloliquefaciens laccase production | [93,104] | ||
| Anthraquinone, indigo and triphenylmethane | Ganoderma sp. En3 | Biological | [105] |
| Acid red 27 | Armillaria sp. F022 | Biological | [106] |
| Chitosan adsorption | Physical | [107] | |
| Acid red 131 | Eectrochemical coagulation | Chemical | [108] |
| Acid red 73 | Coagulation | [109] | |
| Methyl violet, basic fuchsin and their mixture | Biosorption using fungal biomass | Biological | [110] |
| Basic fuchsin | Adsorption by graphene oxide/zinc oxide (GO/ZnO) nanocomposite | Physical | [111] |
| Adsorption by bottom ash and deoiled soya | [112] | ||
| Adsorption on alkali-activated diatomite | [113] | ||
| Adsorption with mussel shell biomass waste | [114] | ||
| Electrochemical oxidation | Chemical | [115] | |
| Amido black 10B | Phanerochaete chrysosporium | Biological | [116] |
| Leptothrix sp. | [117] | ||
| Fenton oxidation | Physical | [118] | |
| Adsorption with zeolite | [119] | ||
| Adsorption with polyaniline/iron oxide composite | [120] | ||
| Adsorption usingpolyaniline/SiO2 nanocomposite | [121] | ||
| Direct red 28 | Electrocoagulated sludge | Chemical | [122] |
| Direct red | Oxidation with photo-Fenton | Physical | [123] |
| Direct red 23 | Adsorption with PAN/PVDF composite ananofibers | [124] | |
| Direct red 31 and Direct orange 26 | Biosorption by rice husk | Physical | [125] |
| Direct red 89 and Reactive green 12 | Biosorption | Physical | [126] |
| Direct Blue 1 and Direct Red 128 | Biosorption using Trametes versicolor | Biological | [127] |
| 4-nitroaniline | Acinetobacter sp. AVLB2 | Biological | [128] |
| Candida sp. AVGB4 | [129] | ||
| RGO-Ni nanocomposite | Physical | [130] | |
| Acid Blue 92 | Coagulation/Flocculation | Chemical | [131] |
| Ozone based oxidation | Physical | [132] | |
| Acid Blue 113 | Electrocoagulation | Chemical | [133] |
| Congo red | Nanofiltration | Physical | [134] |
| Adsorption by clay materials | [135] | ||
| Ozonation | Chemical | [136] | |
| Aspergillus niger | Biological | [137] | |
| Congo red | Bacillus cohnii | Biological | [138] |
| Remazol orange | Pseudomonas aeruginosa | Biological | [139] |
| Remazol brilliant orange 3R | CdO–ZnO nanofibers | Physical | [140] |
| Reactive blue 13 | Pseudomonas sp. | Biological | [141] |
| Reactive orange 16 | Ozone oxidation | Physical | [142] |
| Remazol Black-B | Adsorption on waste orange peel | Physical | [143] |
| Brilliant blue G | Galactomyces geotrichum and Bacillus sp. | Biological | [144] |
| Remazol brilliant blue and orange | Peel adsorption | Physical | [145] |
| Brilliant blue R | Adsorption with orange peel and spent tea leaves | Physical | [146] |
| Acid orange 7 and Remazol black 5 | Biosorption | Physical | [147] |
| Orange 2 | Adsorption by row and chemically modified brown macroalga | Physical | [148] |
| Acid blue 25 | Biosorption with shrimp shells | Physical | [149] |
| Acid orange 8 | Pd-Ni bimetallic nanoparticles | Physical | [150] |
| Basic blue 3 | Pd-Ni nanoparticles supported on activated carbon | Physical | [151] |
| Rhodamine B | palladium-supported zirconia-based catalytic degradation | - | [152] |
| Acid red 4 | Adsorption with activated carbon | Physical | [153] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Slama, H.B.; Chenari Bouket, A.; Pourhassan, Z.; Alenezi, F.N.; Silini, A.; Cherif-Silini, H.; Oszako, T.; Luptakova, L.; Golińska, P.; Belbahri, L. Diversity of Synthetic Dyes from Textile Industries, Discharge Impacts and Treatment Methods. Appl. Sci. 2021, 11, 6255. https://doi.org/10.3390/app11146255
Slama HB, Chenari Bouket A, Pourhassan Z, Alenezi FN, Silini A, Cherif-Silini H, Oszako T, Luptakova L, Golińska P, Belbahri L. Diversity of Synthetic Dyes from Textile Industries, Discharge Impacts and Treatment Methods. Applied Sciences. 2021; 11(14):6255. https://doi.org/10.3390/app11146255
Chicago/Turabian StyleSlama, Houda Ben, Ali Chenari Bouket, Zeinab Pourhassan, Faizah N. Alenezi, Allaoua Silini, Hafsa Cherif-Silini, Tomasz Oszako, Lenka Luptakova, Patrycja Golińska, and Lassaad Belbahri. 2021. "Diversity of Synthetic Dyes from Textile Industries, Discharge Impacts and Treatment Methods" Applied Sciences 11, no. 14: 6255. https://doi.org/10.3390/app11146255
APA StyleSlama, H. B., Chenari Bouket, A., Pourhassan, Z., Alenezi, F. N., Silini, A., Cherif-Silini, H., Oszako, T., Luptakova, L., Golińska, P., & Belbahri, L. (2021). Diversity of Synthetic Dyes from Textile Industries, Discharge Impacts and Treatment Methods. Applied Sciences, 11(14), 6255. https://doi.org/10.3390/app11146255









