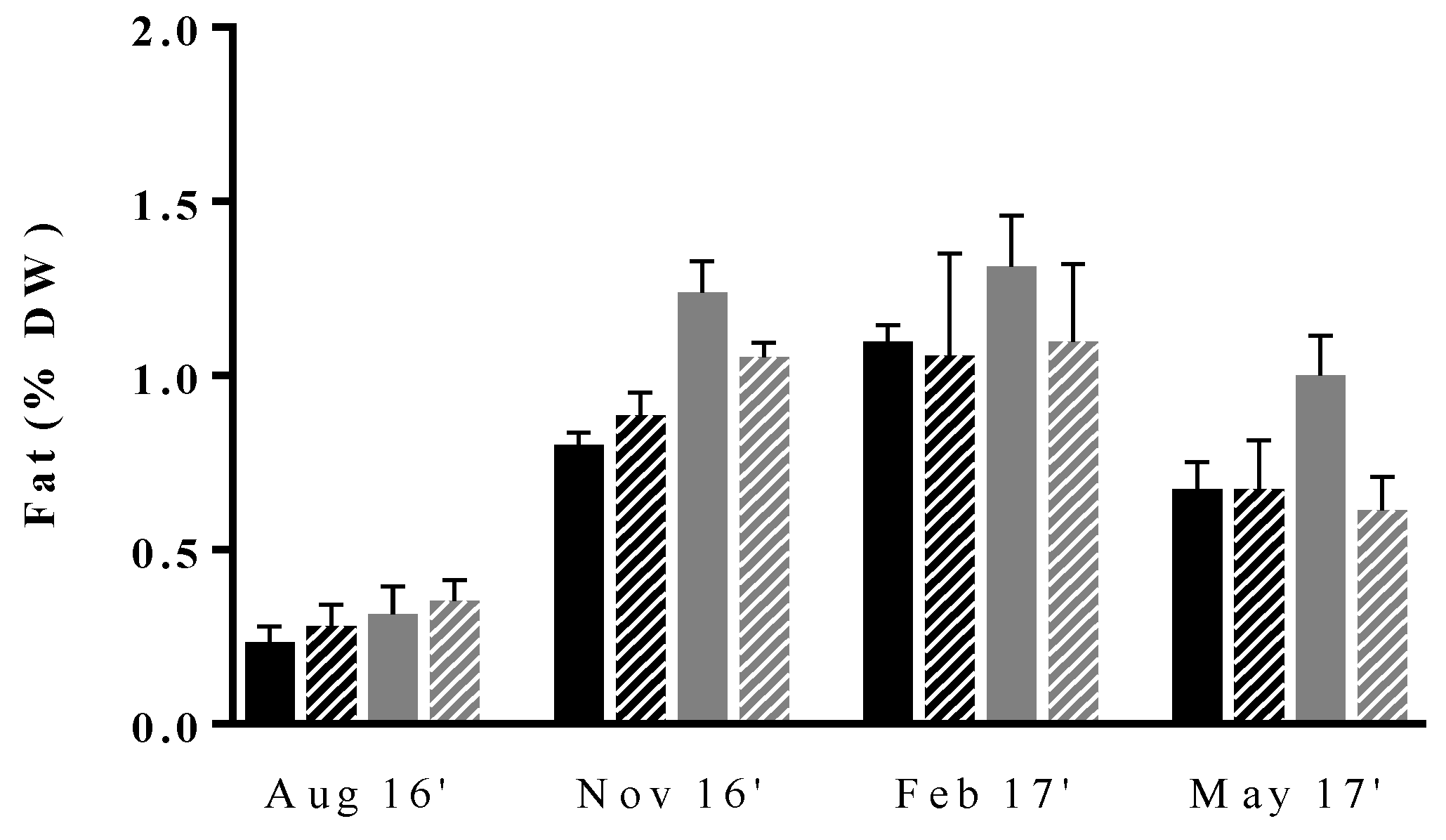Abstract
Ulva macroalgae species are recognized to be an underexploited source of key nutrients, including proteins, fibers and minerals. The present work evaluated the nutritional value protein, ash, mineral composition, fat, fatty acid profile and dietary fiber of the green macroalgae Ulva rigida produced in an open land-based integrated multi-trophic aquaculture (IMTA) system, over four seasons. Overall, protein and fat content of the farmed algae ranged between 7.6 and 25.8% DW and between 0.2 and 1.3% DW, respectively, reaching the highest levels during autumn/winter seasons. In turn, total dietary fiber and ashes showed a contrary seasonal tendency, reaching maximum levels in spring (40.9–58.3% DW and 25.5–38.8% DW, respectively). Notably, the latter were particularly characterized by their richness in potassium, magnesium and iron, and a sodium/potassium ratio bellow 1.7. Variable cultivation conditions of stocking density (Sd) and flow rate (Fr) were also tested, allowing to confirm that manipulation of cultivation conditions in an IMTA system may help to improve the nutritional value of this macroalga and to boost its market value through its use as a functional food ingredient.
1. Introduction
According to the United Nations States, the world population will reach 9.8 billion in 2050 and, by that time, fish and seafood will be one of the most-sought food resources [1]. Indeed, global fish production reached approximately 179 million tons in 2018 and is expected to grow about 15% in the following decade, mainly fueled by aquaculture, which is projected to represent more than half of the global fish production in 2030 [2].
Nevertheless, aquaculture has been raising controversy due to increasing concerns regarding the quality of fish and/or seafood, as well as the impacts of its practices on the environment. Common worries related to the quality of aquaculture products arise from the presence of trace elements and drugs, whereas ecosystems-related issues are mostly associated with discharges that might change levels of oxygen and increase the amounts of suspended organic matter and inorganic nutrients in the waters [3,4,5,6,7]. Hence, the development of more responsible, innovative, sustainable and profitable technologies and practices is needed in order to reduce the environmental impact of aquaculture and simultaneously give it an opportunity to grow. One of the most recognized ecological approach is integrated multi-trophic aquaculture (IMTA), which basically combines the cultivation of species from different trophic or nutritional levels in the same system [8]. This strategy allows the exploitation of synergetic interactions between different species, associating the production of fed species (e.g., finfish) with other extractive organisms, such as marine invertebrates and/or algae. These, in turn, incorporate organic and inorganic compounds resulting from the metabolism of fed species, as well as from uneaten feed [8,9]. In fact, the cultivation of seaweeds under an IMTA system allows the removal of nutrients excess (e.g., phosphorous and nitrogen) from wastewater [10,11] while enhancing the quality and stability of seaweeds biomass and their biochemical profile [12,13].
Ulva spp., commonly known in the food market as sea lettuce, are the main green macroalgae consumed around the world. Algae from Ulva genus belong to Ulvophyceae class and are recognized by their cosmopolite occurrence, mainly due to their great ability to withstand distinct environmental parameters. Therefore, the wide distribution of Ulva spp. in many climatic and ecological conditions and their opportunistic growth make them suitable and cost effective for cultivation in an IMTA framework [12,14,15].
Ulva species are known to be a rich source of valuable polysaccharides, including ulvans and oligosaccharides, that can comprise almost 40% of their dry weight (DW) [16]. Moreover, similar to other green species, they are a good source of protein (10–25% DW) with an appreciable amount of essential amino acids (48%) [16,17] and macro and trace elements, particularly iron and manganese [18]. Moreover, despite the fact that lipids may only represent from residual to 3% of the whole algal DM, they display an important nutritional value, being rich in n-3 polyunsaturated fatty acids (PUFAs), such as α-linolenic acid, eicosapentaenoic acid and docosahexaenoic acid [17,19]. The specific chemical composition of Ulva spp. is, however, dependent on several factors, including the specific species [17,20] and the growing conditions (e.g., location [18,21], seasonality [20,22,23] and cultivation conditions [12,24,25,26]). Most of the studies evaluating the chemical composition of Ulva have been performed on wild samples, therefore those focusing on farmed samples are very limited [27,28]. Hence, this work was planned to further understand the nutritional profile of U. rigida cultivated in an IMTA system over four seasons. Moreover, algae were grown under different conditions of water flow rate and stocking conditions, in order to evaluate possible impacts of such cultivation factors on the chemical profile of this green macroalga.
2. Materials and Methods
2.1. Chemicals
Nitric acid (HNO3) 69% (w/w) and hydrogen peroxide (H2O2) 30% (w/w) were purchased from Merck. Light petroleum, n-hexane and pyridine were purchased from Fisher Scientific. Tetracosane and trimethylchlorosilane (TMSCI, 99% purity) were purchased from Sigma (St. Louis, MO, USA). N,O-bis(trimethylsilyl)trifluoroacetamide (BSTFA, 99% purity), cholesterol and palmitic acid were purchased from Acros (Hampton, NH, USA).
2.2. Biomass
The green macroalgae U. rigida was cultivated by ALGAplus at Ria de Aveiro coastal lagoon. ALGAplus cultivation production is based on an integrated multi-trophic aquaculture (IMTA) system, composed of a semi-intensive organic aquaculture of seabream (1/3) and seabass (2/3) on earthen ponds, with seaweed production on a land-based outdoor tank system. Seaweeds are cultivated exclusively by using nutrient-rich effluent water that flows from the fish production ponds to the seaweed tanks and is subsequently discharged into a settling basin connected to the coastal lagoon, with a significantly lower load of nutrients. The general cultivation conditions used by ALGAplus are available at the website of the company, while some specific conditions used in this study are part of internal protocols of the industry, which are subjected to confidentiality issues. In this context, the exact values of water flow rate applied in the present study to the cultivation tanks cannot be publicized here and are simply denoted as high flow rate (HFr) and low flow rate (LFr), being the last correspondent to half of the HFr value. Likewise, the high stocking density (HSd) values correspond to 2-fold that applied in the low stocking density (LSd) conditions, while the exact values cannot be disclosed. The two variables were combined, setting 4 distinct growing conditions. Overall, the green macroalgae was grown under 4 different conditions, which resulted in 12 independent tanks (3 independent tanks for each condition), sustaining a high flow rate (HFr) or low flow rate (LFr, corresponding to half of HFr) and a high density (HSd) or low density (LSd, corresponding to half of HSd) of cultivation. The grown was performed by clonal propagation methods and the cultivation period took 2 weeks in the tanks, occurring between August 2016 and June 2017. In that period, average water temperature (°C) and salinity (PSU) were 21.9 ± 0.8 and 40.5 ± 0.3 in the summer, 15.1 ± 2.3 and 36.5 ± 0.9 in autumn, 12.5 ± 0.2 and 30.1 ± 0.4 in the winter, and 17.5 ± 1.5 and 31.1 ± 1.8 in spring.
The macroalgae were harvested and processed according to ALGAplus standard practices, which include hand-harvesting and rinsing with freshwater to remove attached mud and epiphytes, centrifugation to remove excess of water, and drying at 25 °C in a convective dryer. After arriving at the lab, macroalgae were milled to a particle size of <0.25 mm. Residual moisture was measured before each analysis by oven-drying the samples at 105 °C, overnight, until constant weight.
2.3. Nitrogen Content and Crude Protein Estimation
Levels of nitrogen were assessed by elemental analysis (LECO TruSpec-Micro CHNS 630-200-200 elemental analyzer) at combustion furnace temperature 1075 °C and afterburner temperature 850 °C. The protein content was estimated with the application of the factor 6.25. Note that the conversion factor 5 is claimed to be more suitable for the estimation of protein in seaweeds [29]. Effectively, the factor 6.25 slightly overestimates the protein content of seaweeds; however, it was herein applied for direct comparison with the literature data, which use it for the most part. When using 6.25 factor, the term “crude protein” is preferred.
2.4. Ash Content and Mineral Composition
Oven-dried samples (105 °C, overnight) in the crucibles were previously pre-incinerated for 20 min, using a heating plate, and then placed in a muffle furnace at 550 °C for 6 h. Ashes were quantified gravimetrically.
Mineral analysis was performed by mineralization of the samples by a microwave-assisted acid-digestion methodology [30] and quantification by inductively coupled plasma–mass spectrometry (ICP–MS), on a Thermo ICP–MS XSeries equipped with a Burgener nebulizer. Quantification limits of the method were for Na, K, Ca and Mg: 0.0125 g/kg; Fe: 0.0125 g/kg; Mn, Zn and Cu: 0.5 mg/kg.
2.5. Fat Content and Fatty Acid Profile
The crude lipid content of each sample (ca. 10 g) was determined gravimetrically after Soxhlet extraction with light petroleum for 8 h. Then the extraction solvent was filtered through a 0.2 µm nylon filter and dried in a rotary evaporator, at a temperature below 30 °C. Further, the residue was dried in an oven, at 105 °C, overnight, and weighted. Subsequently, the residue was re-suspended in hexane, sprayed with nitrogen gas and kept in the freezer until further analysis by GC–MS to determine fatty acid composition.
The fatty acid profile was analyzed by GC–MS, under the previous described conditions [27].
2.6. Dietary Fiber
The contents of total dietary fiber, along with soluble and insoluble fibers were estimated by the enzymatic gravimetric method AOAC 991.43, using the Total Dietary Fiber Assay kit from Megazyme (Bray, UK).
2.7. Statistics
With exception of fatty acids, the remaining results are expressed as mean ± standard deviation. Statistical analysis was performed by using a trial version of GraphPad Prism 6.01 software (OriginLab Corporation, Northampton, MA, USA), using two-way ANOVA and Tukey–HSD multiple comparisons test (p < 0.05).
3. Results and Discussion
3.1. Crude Protein
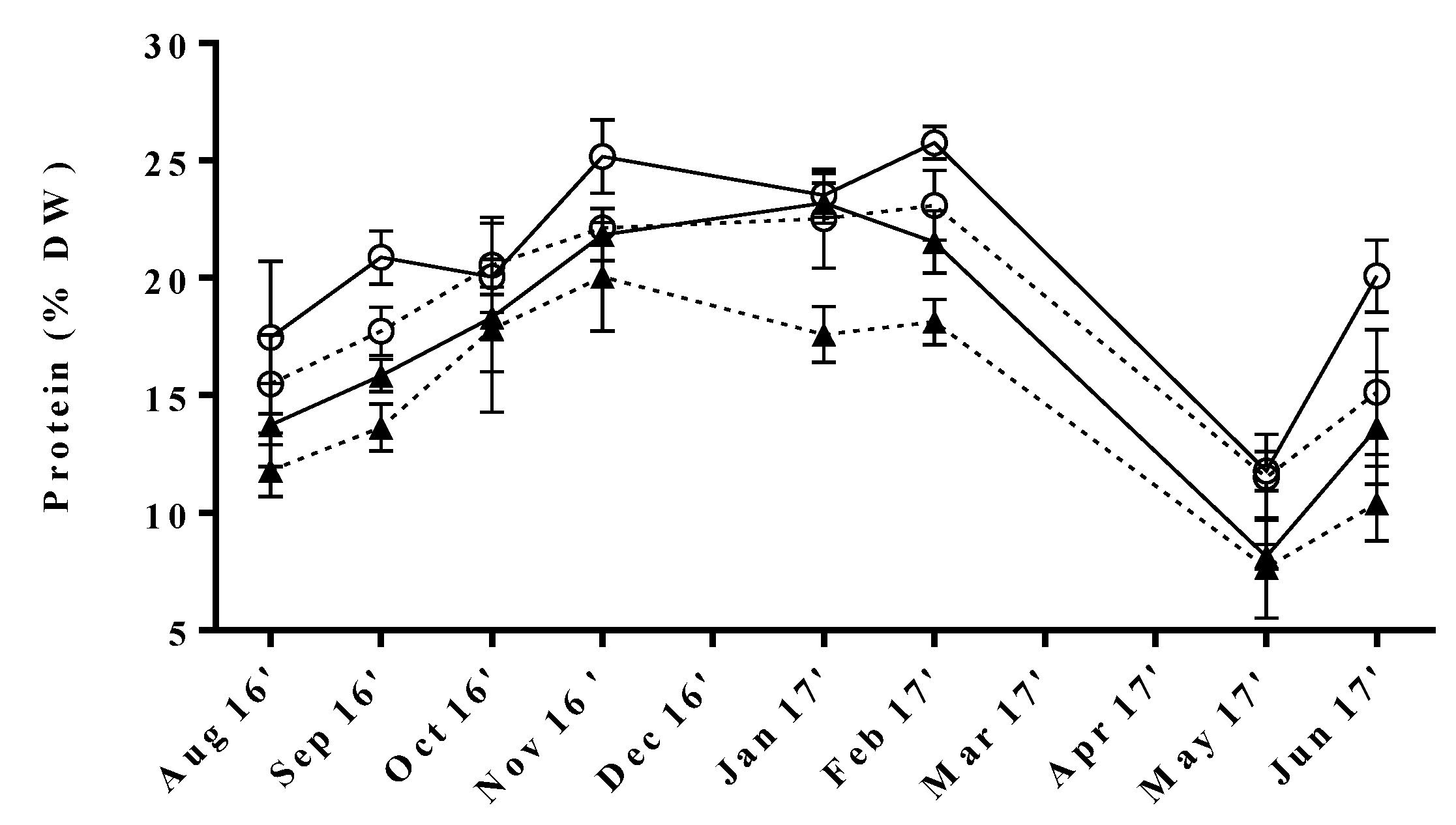
The collected data reflected significant seasonal variations of protein content common to all tested samples (Figure 1). The maximum levels were obtained during autumn/winter (17.6 ± 1.2 to 25.8 ± 0.7% DW), whereas the lowest were observed in spring, particularly in May (7.6−11.8% DW). Similar trends were found by Gadberry et al. [31], who reported slightly higher protein levels during fall/winter seasons corresponding to 30% DW for Ulva spp. cultivated in a land-based intensive culture system in Pacific Northwest, albeit without statistical differences [31]. Furthermore, some studies have reported higher protein levels in farmed macroalgae in comparison to their wild counterparts, as aquaculture effluent is richer in nutrients, especially dissolved nitrogen, than seawater [32,33]. Indeed, this fact could in part justify that the maximum value found in our study was slightly higher than those reported for several specimens of wild Ulva from Northwest Europe (25.8 vs. 23.6% DW) [34,35,36].
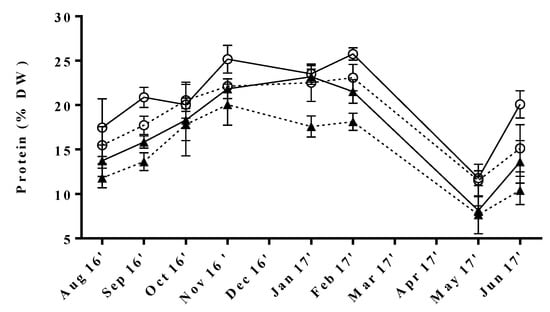
Figure 1.
Seasonal variation of protein content in U. rigida produced by IMTA under different conditions of flow rate, high (○) and low (▲); and of stocking density, high (full lines) and low (dotted lines). Standard deviations are represented as bars (n = 3, in triplicate).
The annual protein content fluctuations observed in our samples were comparable to those assessed by Rouxel et al. [34] for wild U. rigida collected at the Brittany coast in France. Interestingly, Shuuluka et al. [26] reported the opposite trend for protein variation of wild U. rigida collected at the west coast of the Cape Peninsula in South Africa, with maximum protein contents being registered during March and April. Still, although these results might seem discrepant, in both cases, the minimum levels of protein were found in seasons characterized by intense sunlight and high temperatures. In fact, the exposure to light saturating conditions is known to reduce nitrogen uptake in macroalgae tissue, resulting in decreased protein levels [37]. In contrast, the lower durability of the days and temperatures during the autumn/winter months seem to promote the uptake of nitrogen and the consequent synthesis of protein. However, one must not forget that other factors might affect the levels of protein in seaweeds. In this respect, Marinho-Soriano et al. [38] registered a positive correlation with nitrogen content and a negative correlation with water temperature and salinity, in Gracilaria cervicornis and Sargassum vulgare from Brazil [39]. In fact, the water temperature and salinity of tanks from different conditions during our study showed a decreasing tendency of these parameters from November to January and February, respectively (Supplementary Materials Figure S1). This might be partly associated with the high protein levels obtained in this period. However, further data would be needed in order to assess the influence of such abiotic factors.
Our results also indicate that protein content in the green macroalgae grown under an IMTA regime could be changed by setting flow rate (Fr) and stocking density (Sd), or both parameters. Between these two, flow rate was the most relevant factor and macroalgae submitted to high flow rate (HFr) showed in general higher protein content than those cultivated under low flow rate (LFr) conditions. In more detail, the combination of HFr and high stocking density (HSd) yielded the best results, whereas the opposite was achieved for LFr with low stocking density (LSd) conditions. Still, significant differences were found among samples cultivated under fixed Fr and variable Sd. These results are in line with previous studies that reported increased levels of nitrogen accumulation in both green [12,40] and red [41,42] seaweeds when cultivated in the presence of higher nutrient loadings, as there is more nitrogen available for protein synthesis. Furthermore, a positive correlation between protein content and stocking density has been previously reported for U. lactuca cultivated in IMTA grown in Gulf of Aqaba [43,44]. In fact, as the number of specimens increases in the cultivation tanks, macroalgae are naturally less exposed to light, and, as already mentioned, in these conditions, nitrogen-uptake is favored [37], which results in increased nitrogen available for protein synthesis, consequently leading to higher levels of protein in the seaweed.
3.2. Ash and Mineral Profile
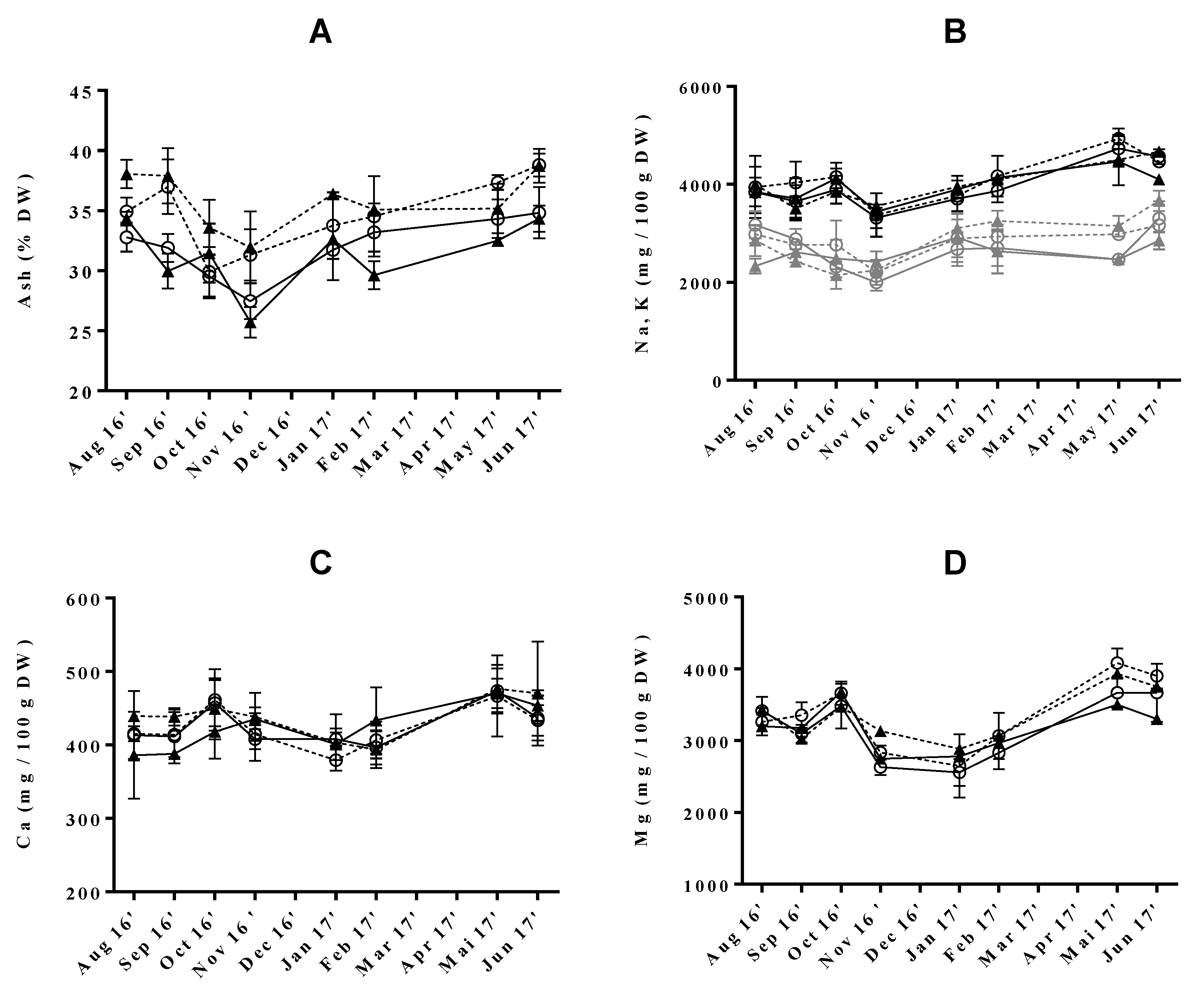
Throughout the year, ash content varied between 25.7 ± 1.3% DW and 38.8 ± 1.0% DW, reaching the maximum levels in June and the minimum in November (Figure 2A). These results were not directly comparable to those of the literature, as, to our knowledge, there is no reported information regarding seasonal variation of ash content specifically in U. rigida, even in wild samples. Nevertheless, recently, Gadberry and coworkers [31] revealed slightly higher ash levels in summer (38% DW) comparatively to fall/winter (31% DW) seasons for Ulva spp. cultivated in a land-based intensive cultivation system (p < 0.05). Still, the yearly average (33.5 ± 3.5% DW) falls within the range of previously reported values for wild specimens of the same species collected in several countries (14−31% DW) [24,35,45].
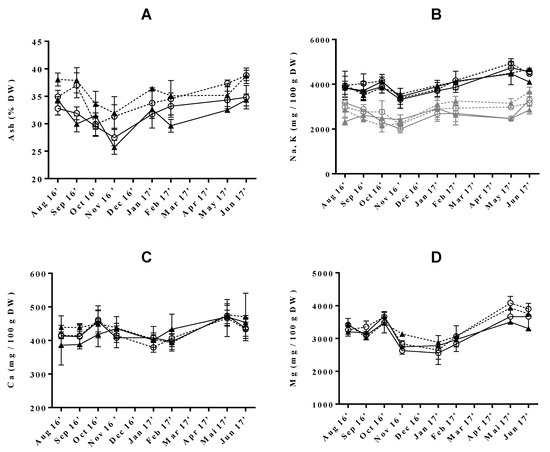
Figure 2.
Seasonal variation of (A) ash (% DW), (B) sodium and potassium, (C) calcium and (D) magnesium (mg/100 g DW) in Ulva rigida produced by IMTA under different conditions of flow rate, high (○) and low (▲); and of stocking density, high (full lines) and low (dotted lines). Standard deviations are represented as bars (n = 3, in triplicate).
Regarding the effect of cultivation conditions, stocking density was found to have a significant influence on the modulation of this nutrient, leading to increased ash levels when set to low. This tendency was also observed in a small study comparing the nutrient composition of Ulva ohnoi cultivated under three different stocking densities [46]. Apart from this study, little information is available regarding the influence of this growing condition on the ash content of macroalgae. In opposition, variation of flow rate in IMTA conditions had no significant impact on the ash content of the analysed samples, although values tended to increase when this parameter was set at low. These observations agree with those of previous authors who have studied the effect of nitrogen availability in the composition of farmed U. rigida [47] and U. lactuca [12]. In their study, researchers hypothesized ash content to be controlled by nitrogen levels, in the presence of low concentrations of this nutrient [48]. This could also explain why ash levels varied inversely to those of protein throughout the present study. Indeed, the previous protein content reached its minimum and maximum in the summer and winter months, respectively, whereas, for ash levels, an opposite tendency was observed.
Among macrominerals, sodium (Na) was predominant (3604 ± 384 to 4700 ± 290 mg/100 g DW), followed by magnesium (Mg, 2695 ± 241 to 3992 ± 196 mg/100 g DW), potassium (K, 2157 ± 204 to 3460 ± 336 mg/100 g DW) and calcium (Ca, 387 ± 37 to 498 ± 71 mg/100 g DW) (Figure 2B−D). Na and K levels were substantially higher than those established for wild U. rigida (400–576 mg/100g DW and 817–1100 mg/100 g DW) [17,49]. As previously described, there are seasonal, environmental and physiological factors causing variations in the mineral content of seaweeds, even among the same species [18,50,51,52]. However, the disparity in sodium content could be related to the methodology used in sample preparation, as our samples were washed with freshwater from “Lagoon of Aveiro” (naturally salted), instead of distilled water. As a result, Na/K ratio varied between 1.2 ± 0.1 and 1.7 ± 0.3), which is higher than that determined by other authors for the same species (0.4–0.7) [17,49]. Nevertheless, the obtained values were comparable to those reported in the literature for U. lactuca (< 2.4) [53]. The benefits of consuming foods with a low Na/K ratio in the regulation of blood pressure are well documented [54]. In this sense, the introduction of macroalgae in Western diets could be an asset to improve or maintain cardiovascular health.
The analyzed green macroalgae also proved to be an important source of Mg and Ca, with values fitting within the range previously described for this species (1775–8686 mg/100 g DW and 325–1266 mg/100 g DW, respectively) and even exceeded those of some edible higher plants and other common foods which are known to be rich in these nutrients [17,27,35,49,55]. Interestingly, the consumption of 8 g of dry U. rigida covered between 62 and 91% of the recommended daily intake (RDI) of magnesium (Supplementary Materials Table S1). Seasonality did not affect the levels of Ca but those of Mg followed a close pattern to that registered for ash, with minimum values being registered upon October and maximum in spring. In addition, in this season, Mg was predominately accumulated in macroalgae grown under LSd conditions. Please note that, to our knowledge, macromineral content variation in Ulva genus as a function of seasonality has only been described for wild U. lactuca from Alexandria [56], in Egypt, while the effect of Fr and Sd is herein focused on for the first time.
The trace elements iron (Fe), manganese (Mn), copper (Cu), and zinc (Zn) were also assessed in each season (Table 1). Samples were rich in iron (35.7–170 mg/100 g DW), followed by manganese (2.6–11.5 mg/100 g DW), copper (1.1–3.4 mg/100 g DW) and zinc (0.8–1.9 mg/100 g DW). In fact, eating a portion of 8 g of dry U. rigida covered up to 196% of the RDI of iron, while Mn, Cu and Zn did not cover RDI above 15%. For this specific level of Fe, results of the recommended daily allowance (RDA) (Supplementary Materials Table S1) showed that the use of no more than 30 g/day of U. rigida would not compromise health due to the iron content of this seaweed.

Table 1.
Seasonal variation of trace minerals (mg/100 g dry weight) in Ulva rigida produced by IMTA under different conditions of flow rate, high (HFr) and low (LFr); and of stocking density, high (HSd) and low (LSd).
These results were comparable to those reported for wild samples collected at Galician Coast (Spain) and Gabes Gulf area (Tunisia) [35,49,55]. Another study focusing on U. rigida from different sites of Italy documented similar concentrations of the mentioned elements, but the relative abundance differed slightly, as copper was present in higher concentrations than zinc [22]. The same study also reported seasonal variability of these elements, which mainly peaked in autumn and spring, as a result of the proliferation of the macroalgae. In opposition, in the present study, no seasonal patterns were detected, mainly due to the high variability of results among replicates under the same experimental conditions. As for the influence of growing conditions, no significant differences were found as well. Nevertheless, this was the first study evaluating the effect of cultivation conditions on trace elements in cultivated Ulva.
3.3. Fat and Fatty Acid Profile
The fat content and fatty acid profile of the green macroalgae cultivated in IMTA under distinct conditions were evaluated throughout the four seasons, specifically in samples collected in August, November, February and May (Figure 3). The fat content varied between 0.23 ± 0.05% DW and 1.31 ± 0.15% DW throughout the entire experiment. These values are close to those of the range reported in the literature for wild U. rigida collected in different sites of the world (0.37−1.63% DW) [17,19,45]; however, they are inferior either to those reported for IMTA grown specimens also cultivated by ALGAplus (1.7 to 3.4% DW) [28] or Ulva spp. cultivated in a land-based system (2.6 to 3.5% DW) [31]. Most probably, differences are due to the distinct extraction methodologies and/or solvents applied in the two studies.
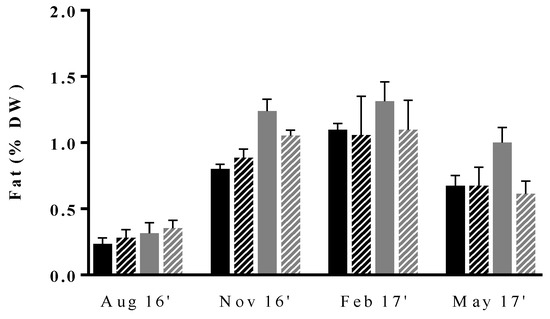
Figure 3.
Seasonal variation of fat content (% DW) in Ulva rigida produced by IMTA under different conditions of flow rate, high (dark bars) and low (gray bars); and of stocking density, high (full bars) and low (lined bars). Standard deviations are represented (n = 3, in triplicate).
Lipid contents were shown to vary seasonally, reaching the maximum and minimum values in winter and summer, respectively. This trend is in accordance with that previously reported by Moreira and co-workers for IMTA, as well as by other authors when monitoring fat content in wild macroalgae of Ulva genus [28,56,57,58]. In fact, despite the taxonomic and geographical differences, the observed trends point to higher and lower lipid values during low and high temperature seasons, respectively. These observations are also in accordance with those of Floreto et al. [23], who concluded that temperature was one of the most important abiotic factors modulating lipids in Ulva pertusa and that lipid synthesis was favored at low temperatures, as evaluated in a randomized laboratory experiment using an artificial seawater medium.
In addition to seasonality, our results also suggest that cultivation conditions influence fat content. Indeed, except for summer when lipid synthesis is at minimum values, samples cultivated at LFr/HSd had superior fat ratios than those of the remaining experimental conditions. To our knowledge, there are no published studies assessing the influence of growing conditions on the fat content of U. rigida. Nevertheless, a previous work on U. pertusa have reported the increase in the fat content of the macroalgae when exposed to nitrogen deficiency conditions [59]. This fact could partially be associated with the superior levels of fat in LFr/HSd samples. The authors hypothesized lipid production to be increased in these conditions, as a result of a storage mechanism or due to a continuous synthesis of lipid compounds, as this process does not require nitrogen. In the first case, excess carbon would be stored as fat, instead of being converted into protein as a result of a high C:N ratio.
The fatty acid (FA) profile of U. rigida was mainly composed of palmitic acid, C16:0; oleic acid, C18:1 (n-9); and α-linolenic acid, C18:3 (n-3) (Table 2). Such results are consistent with those of previous studies on wild U. rigida [17,19,45,49]. Curiously, this trend was not totally coincident with that previously reported for U. rigida grown in IMTA, since the authors also demonstrated the existence of high proportions of other polyunsaturated fatty acids, particularly of C16:4 and C18:4 [28]. Differences are probably due to the different extraction conditions used and/or to the superior capacity of DB-FFAP column (used by Moreira et al. [28]) to analyze polyunsaturated fatty acids, compared to DB-5 ms. According to this, we must note that is possible that polyunsaturated fatty acids (n = 4) were underestimated in our study.

Table 2.
Seasonal variation of fatty acid composition (mg/kg dry weight) in Ulva rigida produced by IMTA under different conditions of flow rate, high (HFr) and low (LFr); and of stocking density, high (HSd) and low (LSd).
Albeit that, ratios Ω6/Ω3 (0.2–0.4) reflected higher levels of omega-3 FA than omega-6 FA, as previously reported for this species. The intake of foods with a low ratio Ω6/Ω3 (below 5:1) have been proven to contribute to the prevention of certain diseases promoted by the excessive consumption of omega-6 FA relative to omega-3 FA [60]. Notably, the Atherogenic Index (AI) and Thrombogenic Index (TI), which are related to the protection against coronary artery diseases, varied between 1.5 and 3.4 and between 1 and 2.1, respectively, being then more favorable than those reported for U. fasciata (2.85 and 2.78–5.48, respectively) [61]. Moreover, more beneficial fatty acid hypocholesterolemic/hypercholesterolemic (h/H) ratios were herein found for U. rigida in comparison to those found in U. fasciata [61].
In addition, seasonal variations were detected in terms of FA concentration, concomitant with the temporal changes observed for total fat content. Thus, higher concentrations of FA were found in autumn and winter, whereas the lowest were observed in summer and spring. As for the relative proportions of FA, no coherent significant differences were found due to the high variability of results among replicates (samples from three distinct cultivation tanks). One should highlight that it is possible that part of such variability may arise due to slight variations of environmental factors (e.g., light exposure) that were not considered in this study. In fact, previous studies performed with Ulva specimens have attributed FA seasonal variations to environmental conditions, such as temperature, irradiation or even to vegetative and reproductive development [21,56,58], and particularly, Floreto et al. [23] have found both light and temperature to significantly influence FA profile in Ulva pertusa.
As for differences between samples cultivated under different water flows and stocking densities, it was also not possible to establish a cause–effect relation between the quantitative composition of FA and the cultivation parameters. Despite the high variability of results, one cannot ignore the possibility that the increased fat content in samples grown under LFr/HSd conditions (Figure 2) might not be directly associated with FA but instead with other lipid components such as pigments. Hence, further studies are necessary to understand the impact of Fr and Sd on the fat constituents of Ulva species. To our knowledge, this topic has only been previously addressed by Floreto et al. [59] and Pinchetti et al. [47], who respectively evaluated changes in the FA composition as a function of nitrogen and phosphorus availability in U. pertusa by using artificial seawater at a laboratorial scale and of nitrogen in tank-cultivated U. rigida by using seawater. Curiously, these authors reported opposing trends: whereas nitrogen-starvation caused an increment of certain PUFAs and a decrement of SFA and MUFAs in U. pertusa, the tank-cultivated U. rigida showed the reverse pattern.
3.4. Dietary Fiber
Dietary fibers are one of the most abundant nutrients in U. rigida [16]. In this study, the contents of insoluble (IDF), soluble (SDF) and total (TDF) dietary fibers were estimated for the samples collected in August, November, February and May. During these four seasons, TDF levels ranged between 30.9 ± 1.4% DW and 57.2 ± 1.5% DW (Table 3), which are at the top limit, or above those previously reported for wild specimens of U. rigida (38–41% DW) from different geographical origins [16,24,62], while fitting well on the range reported for wild U. lactuca (38.1–60.5% DW) [63,64,65]. Interestingly, TDF levels were shown to be higher than those found in certain vegetables, cereals and legumes [66].

Table 3.
Seasonal variation of dietary fiber composition (g/100 g dry weight) in Ulva produced by IMTA under different conditions of flow rate, high (HFr) and low (LFr), and of stocking density, high (HSd) and low (LSd).
All samples showed to contain approximate amounts of IDF (19.0 ± 2.1 to 29.2 ± 1.6% DW) and SDF (20.8 ± 0.6 to 30.6 ± 1.3% DW), respectively [17,24,62]. Moreover, the ratio soluble/insoluble fiber varied between 0.9 and 1.2. It is to be noted that only one sample presented a value below 1, reflecting the prevalence of SDF over IDF. This trend seems to be the opposite to that of the few studies available on this topic, in which IDF was shown to be the most abundant fraction, with soluble/insoluble fiber ratios ranging between 0.8 and 0.9 [17,24,62]. Factors such as methodology, geographical origin and time of harvest could explain the observed differences.
Seasonality was observed to influence dietary fiber levels, which reached a maximum in spring and minimum values in autumn–winter. To our knowledge, there are no data available in the literature directly assessing the influence of seasonality on the levels of fiber content in U. rigida. Nevertheless, it is feasible that these variations may reflect in part those of proteins whose levels in general had the opposite trend (Figure 1). Moreover, it is possible to hypothesize that the high fiber levels in May might also be partly due to a longer and more intense light exposition, as this factor has been positively correlated with increasing percentages of carbon in the seaweed, concomitant with an increasing C:N ratio [67]. As for the influence of growing conditions, no significant differences were found among samples cultivated under distinct water flows and stocking densities. Although no previous studies have assessed the effect of Fr and Sd on fiber content of U. rigida, it has been observed that they are closely related to nutrient limitation in the medium. For instance, Lahaye et al. [24] investigated changes in dietary fiber content in U. rigida as a function of nitrogen variations in the cultivation medium. These authors observed an increase in TDF, IDF and SDF concomitant with an increase in polysaccharides in conditions of nitrogen limitation. In this case, compounds containing nitrogen would be consumed or lost, leading to an increase in carbon percentage. Once again, these results are in accordance with our study, in which the nitrogen content of macroalgae was shown to be low in spring/summer months, whereas dietary fiber was shown to be increased.
4. Conclusions
Our work highlights the richness of cultivated Ulva rigida in valuable nutrients, albeit their variations caused by seasonal aspects. Among them, when cultivated in IMTA system, this species maintains high levels of proteins and fibers that may reach about 26 and 58% of their DW, respectively. The knowledge of seasonal biochemical composition of this green macroalgae produced in a sustainable IMTA system hopefully will inspire future studies on its valorization, also by considering other cultivation conditions and nutrient limits. These could even manipulate environmental parameters to induce different responses/adaptations in this macroalga. In addition, our study may also help promote the consumption of Ulva produced under controlled conditions, as its modulated biochemical profile may represent a good source of macro- and micronutrients, which are essential in a balanced diet.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/app11136137/s1: Figure S1: Seasonal variation: (A) water temperature and (B) water salinity of Ulva rigida tanks produced by IMTA under different conditions of flow rate, high (○) and low (π); and of stocking density, high (full lines) and low (dotted lines). Standard deviations are represented as bars (n = 3, in triplicate). Table S1: Recommended Daily Intakes (%) and Recommended Daily Allowances (g/day) for macro- and microminerals of dry Ulva rigida produced by IMTA under different conditions of flow rate, high (HFr) and low (LFr), and of stocking density, high (HSd) and low (LSd).
Author Contributions
A.S.Q., A.R.C. and M.V. performed the experimental work; A.S.Q. and A.R.C. wrote the manuscript, performed the analysis, and the interpretation of data; M.H.A. was responsible for setting IMTA farming and providing of Ulva; E.P. and A.M.S.S. co-wrote the manuscript; S.M.C. coordinated the work and co-wrote the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
R&D Co-promotion project POCI-01-0247-FEDER-003419, SHARP–Seaweed for Healthier Traditional Products, co-financed by the European Regional Development Fund through the Competitive and Internationalization Thematic Operational Program under the Portugal 2020 Program financed the experimental work, the fellowships of A.S.Q. and A.R.C. and the research contract of S.M.C.
Institutional Review Board Statement
Not Applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data is contained within the article and supplementary material.
Acknowledgments
Science and Technology Foundation/Ministry of Education and Science (FCT/MEC) funded the Organic Chemistry, Natural Products and Food Stuffs Research Unit (QOPNA) (FCT UID/QUI/00062/2019) and the Associated Laboratory for Green Chemistry (LAQV) of the Network of Chemistry and Technology (REQUIMTE) (UIDB/50006/2020), through national funds and, where applicable, co-financed by the European Regional Development Fund (FEDER), within the Portugal 2020 PT2020 Partnership Agreement, and to the Portuguese NMR Network.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Diana, J.S.; Egna, H.S.; Chopin, T.; Peterson, M.S.; Cao, L.; Pomeroy, R.; Verdegem, M.; Slack, W.T.; Bondad-Reantaso, M.G.; Cabello, F. Responsible aquaculture in 2050: Valuing local conditions and human innovations will be key to success. BioScience 2013, 63, 255–262. [Google Scholar] [CrossRef] [Green Version]
- FAO. The State of World Fisheries and Aquaculture 2020; Sustainability in Action: Rome, Italy, 2020. [Google Scholar]
- Granada, L.; Sousa, N.; Lopes, S.; Lemos, M.F.L. Is Integrated Multitrophic Aquaculture the solution to the sectors’ major challenges? A review. Rev. Aquacult. 2016, 8, 283–300. [Google Scholar] [CrossRef]
- Olesen, I.; Myhr, A.I.; Rosendal, G.K. Sustainable aquaculture: Are We getting there? Ethical perspectives on salmon farming. J. Agric. Environ. Ethics 2011, 24, 381–408. [Google Scholar] [CrossRef]
- Jansen, H.; Broch, O.; Bannister, R.; Cranford, P.; Handå, A.; Husa, V.; Jiang, Z.; Strohmeier, T.; Strand, Ø. Spatio-temporal dynamics in the dissolved nutrient waste plume from Norwegian salmon cage aquaculture. Aquacult. Environ. Interac. 2018, 10, 385–399. [Google Scholar] [CrossRef] [Green Version]
- Pal, D.; Maiti, S.K. Seasonal variation of heavy metals in water, sediment, and highly consumed cultured fish (Labeo rohita and Labeo bata) and potential health risk assessment in aquaculture pond of the coal city, Dhanbad (India). Environ. Sci. Pollut. Res. Int. 2018, 25, 12464–12480. [Google Scholar] [CrossRef] [PubMed]
- De Casabianca, M.; Laugier, T.; Marinho-Soriano, E. Seasonal changes of nutrients in water and sediment in a Mediterranean lagoon with shellfish farming activity (Thau Lagoon, France). ICES J. Mar. Sci. 1997, 54, 905–916. [Google Scholar] [CrossRef] [Green Version]
- Chopin, T. Aquaculture, Integrated Multi-Trophic (IMTA). In Sustainable Food Production; Christou, P., Savin, R., Costa-Pierce, B.A., Misztal, I., Whitelaw, C.B.A., Eds.; Springer: New York, NY, USA, 2013; pp. 184–205. [Google Scholar]
- Neori, A.; Chopin, T.; Troell, M.; Buschmann, A.H.; Kraemer, G.P.; Halling, C.; Shpigel, M.; Yarish, C. Integrated aquaculture: Rationale, evolution and state of the art emphasizing seaweed biofiltration in modern mariculture. Aquaculture 2004, 231, 361–391. [Google Scholar] [CrossRef]
- Copertino, M.d.S.; Tormena, T.; Seeliger, U. Biofiltering efficiency, uptake and assimilation rates of Ulva clathrata (Roth) J. Agardh (Clorophyceae) cultivated in shrimp aquaculture waste water. J. Appl. Phycol. 2009, 21, 31–45. [Google Scholar] [CrossRef]
- Neori, A. Essential role of seaweed cultivation in integrated multi-trophic aquaculture farms for global expansion of mariculture: An analysis. In Nineteenth International Seaweed Symposium. Developments in Applied Phycology; Borowitzka, M.A., Critchley, A.T., Kraan, S., Peters, A., Sjøtun, K., Notoya, M., Eds.; Springer: Dordrecht, The Netherlands, 2008; Volume 2, pp. 567–570. [Google Scholar]
- Msuya, F.E.; Neori, A. Effect of Water aeration and nutrient load level on biomass yield, N uptake and protein content of the seaweed Ulva lactuca cultured in seawater tanks. J. Appl. Phycol. 2008, 20, 1021–1031. [Google Scholar] [CrossRef]
- Abreu, M.H.; Pereira, R.; Sassi, J.-F. Marine algae and the global food industry. In Marine Algae: Biodiversity, Taxonomy, Environmental Assessment, and Biotechnology; Pereira, L., Neto, J., Eds.; CRC Press Taylor & Francis Group: Boca Raton, FL, USA, 2014; pp. 300–319. [Google Scholar]
- Marinho, G.; Nunes, C.; Sousa-Pinto, I.; Pereira, R.; Rema, P.; Valente, L.M.P. The IMTA-cultivated chlorophyta Ulva spp. as a sustainable ingredient in Nile tilapia (Oreochromis Niloticus) diets. J. Appl. Phycol. 2013, 25, 1359–1367. [Google Scholar] [CrossRef]
- Shpigel, M.; Guttman, L.; Shauli, L.; Odintsov, V.; Ben-Ezra, D.; Harpaz, S. Ulva lactuca from an Integrated Multi-Trophic Aquaculture (IMTA) biofilter system as a protein supplement in gilthead seabream (Sparus Aurata) diet. Aquaculture 2017, 481, 112–118. [Google Scholar] [CrossRef]
- Pereira, L. A review of the nutrient composition of selected edible seaweeds. In Seaweed: Ecology, Nutrient Composition and Medicinal Uses; Pomin, V.H., Ed.; Nova Science Publishers, Inc.: Coimbra, Portugal, 2011; pp. 15–49. [Google Scholar]
- Paiva, L.; Lima, E.; Neto, A.I.; Marcone, M.; Baptista, J. Health-promoting ingredients from four selected Azorean macroalgae. Food Res. Int. 2016, 89, 432–438. [Google Scholar] [CrossRef]
- Circuncisão, A.R.; Catarino, M.D.; Cardoso, S.M.; Silva, A.M.S. Minerals from macroalgae origin: Health benefits and risks for consumers. Mar. Drugs 2018, 16, 400. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumari, P.; Bijo, A.J.; Mantri, V.A.; Reddy, C.R.K.; Jha, B. Fatty acid profiling of tropical marine macroalgae: An analysis from chemotaxonomic and nutritional perspectives. Phytochemistry 2013, 86, 44–56. [Google Scholar] [CrossRef]
- Robic, A.; Sassi, J.-F.; Dion, P.; Lerat, Y.; Lahaye, M. Seasonal variability of physicochemical and rheological properties of ulvan in two Ulva species (chlorophyta) from the Brittany coast. J. Phycol. 2009, 45, 962–973. [Google Scholar] [CrossRef] [PubMed]
- Serviere-Zaragoza, E.; Hurtado, M.A.; Manzano-Sarabia, M.; Mazariegos-Villarreal, A.; Reza, M.; Arjona, O.; Palacios, E. Seasonal and interannual variation of fatty acids in macrophytes from the Pacific coast of Baja California Peninsula (Mexico). J. Appl. Phycol. 2015, 27, 1297–1306. [Google Scholar] [CrossRef]
- Favero, N.; Cattalini, F.; Bertaggia, D.; Albergoni, V. Metal accumulation in a biological indicator (Ulva rigida) from the Lagoon of Venice (Italy). Arch. Environ. Contam. Toxicol. 1996, 31, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Floreto, E.A.T.; Hirata, H.; Ando, S.; Yamasaki, S. Effects of temperature, light intensity, salinity and source of nitrogen on the growth, total lipid and fatty acid composition of Ulva pertusa kjellman (chlorophyta). Bot. Mar. 1993, 36, 149–158. [Google Scholar] [CrossRef]
- Lahaye, M.; Gomez-Pinchetti, J.-L.; Del Rio, M.J.; Garcia-Reina, G. Natural decoloration, composition and increase in dietary fibre content of an edible marine algae, Ulva rigida (chlorophyta), grown under different nitrogen conditions. J. Sci. Food Agric. 1995, 68, 99–104. [Google Scholar] [CrossRef]
- Korzen, L.; Abelson, A.; Israel, A. Growth, Protein and carbohydrate contents in Ulva rigida and Gracilaria bursa-pastoris integrated with an offshore fish farm. J. Appl. Phycol. 2016, 28, 1835–1845. [Google Scholar] [CrossRef]
- Shuuluka, D.; Bolton, J.J.; Anderson, R.J. Protein content, amino acid composition and nitrogen-to-protein conversion factors of Ulva rigida and Ulva capensis from natural populations and Ulva lactuca from an aquaculture system, in South Africa. J. Appl. Phycol. 2013, 25, 677–685. [Google Scholar] [CrossRef]
- Neto, R.; Marçal, C.; Queirós, A.; Abreu, H.; Silva, A.; Cardoso, S. Screening of Ulva rigida, Gracilaria Sp., Fucus vesiculosus and Saccharina latissima as functional ingredients. Int. J. Mol. Sci. 2018, 19, 2987. [Google Scholar] [CrossRef] [Green Version]
- Moreira, A.S.P.; da Costa, E.; Melo, T.; Sulpice, R.; Cardoso, S.M.; Pitarma, B.; Pereira, R.; Abreu, M.H.; Domingues, P.; Calado, R.; et al. Seasonal plasticity of the polar lipidome of Ulva rigida cultivated in a sustainable integrated multi-trophic aquaculture. Algal Res. 2020, 49, 101958. [Google Scholar] [CrossRef]
- Lourenço, S.O.; Barbarino, E.; De-Paula, J.C.; Marquez, U.M.L. Amino acid composition, protein content and calculation of nitrogen-to-protein conversion factors for 19 tropical seaweeds. Phycol. Res. 2002, 50, 233–241. [Google Scholar] [CrossRef]
- Pinheiro, V.F.; Marçal, C.; Abreu, H.; Lopes da Silva, J.A.; Silva, A.M.S.; Cardoso, S.M. Physicochemical Changes of Air-Dried and Salt Processed Ulva rigida over Storage Time. Molecules 2020, 24, 2955. [Google Scholar] [CrossRef] [Green Version]
- Gadberry, B.A.; Colt, J.; Maynard, D.; Boratyn, D.C.; Webb, K.; Johnson, R.B.; Saunders, G.W.; Boyer, R.H. Intensive land-based production of red and green macroalgae for human consumption in the Pacific Northwest: An evaluation of seasonal growth, yield, nutritional composition, and contaminant levels. ALGAE 2018, 33, 109–125. [Google Scholar] [CrossRef] [Green Version]
- Robertson-Andersson, D.V.; Maneveldt, G.W.; Naidoo, K. Effects of wild and farm-grown macroalgae on the growth of juvenile South African abalone Haliotis midae linnaeus. Afr. J. Aquat. Sci. 2011, 36, 331–337. [Google Scholar] [CrossRef] [Green Version]
- Robertson-Andersson, D.V.; Potgieter, M.; Hansen, J.; Bolton, J.J.; Troell, M.; Anderson, R.J.; Halling, C.; Probyn, T. Integrated seaweed cultivation on an abalone farm in South Africa. J. Appl. Phycol. 2008, 20, 579–595. [Google Scholar] [CrossRef]
- Rouxel, C.; Bonnabeze, E.; Daniel, A.; Jerôme, M.; Etienne, M.; Fleurence, J. Identification by SDS PAGE of green seaweeds (Ulva and Enteromorpha) used in the food industry. J. Appl. Phycol. 2001, 13, 215–219. [Google Scholar] [CrossRef]
- Taboada, C.; Millán, R.; MÃguez, I. Composition, nutritional aspects and effect on serum parameters of marine algae Ulva rigida. J. Sci. Food Agric. 2009, 90, 445–449. [Google Scholar] [CrossRef]
- Fleurence, J.; Le Coeur, C.; Mabeau, S.; Maurice, M.; Landrein, A. Comparison of different extractive procedures for proteins from the edible seaweeds Ulva rigida and Ulva rotundata. J. Appl. Phycol. 1995, 7, 577–582. [Google Scholar] [CrossRef]
- Lapointe, B.E.; Duke, C.S. Biochemical strategies for growth of Gracilaria tikvahiae (Rhodophyta) in relation to light intensity and nitrogen availability. J. Phycol. 1984, 20, 488–495. [Google Scholar] [CrossRef]
- Marinho-Soriano, E.; Fonseca, P.C.; Carneiro, M.A.A.; Moreira, W.S.C. Seasonal variation in the chemical composition of two tropical seaweeds. Bioresour. Technol. 2006, 97, 2402–2406. [Google Scholar] [CrossRef]
- Stengel, D.B.; Connan, S.; Popper, Z.A. Algal chemodiversity and bioactivity: Sources of natural variability and implications for commercial application. Biotechnol. Adv. 2011, 29, 483–501. [Google Scholar] [CrossRef] [PubMed]
- Del Río, M.J.; Ramazanov, Z.; García-Reina, G. Ulva rigida (Ulvales, Chlorophyta) tank culture as biofilters for dissolved inorganic nitrogen from fishpond effluents. Hydrobiologia 1996, 326, 61–66. [Google Scholar] [CrossRef]
- Abreu, M.H.; Pereira, R.; Yarish, C.; Buschmann, A.H.; Sousa-Pinto, I. IMTA with Gracilaria vermiculophylla: Productivity and nutrient removal performance of the seaweed in a land-based pilot scale system. Aquaculture 2011, 312, 77–87. [Google Scholar] [CrossRef]
- Matos, J.; Costa, S.; Rodrigues, A.; Pereira, R.; Sousa Pinto, I. Experimental integrated aquaculture of fish and red seaweeds in Northern Portugal. Aquaculture 2006, 252, 31–42. [Google Scholar] [CrossRef]
- Cohen, I.; Neori, A. Ulva lactuca biofilters for marine fishpond effluents I. Ammonia uptake kinetics and nitrogen content. Bot. Mar. 1991, 34, 475–482. [Google Scholar] [CrossRef]
- Neori, A.; Cohen, I.; Gordin, H. Ulva lactuca biofilters for marine fishpond effluents II. Growth rate, yield and C:N ratio. Bot. Mar. 1991, 34, 483–489. [Google Scholar] [CrossRef]
- Trigui, M.; Gasmi, L.; Zouari, I.; Tounsi, S. Seasonal variation in phenolic composition, antibacterial and antioxidant activities of Ulva rigida (Chlorophyta) and Assessment of Antiacetylcholinesterase potential. J. Appl. Phycol. 2013, 25, 319–328. [Google Scholar] [CrossRef]
- Masaló, I.; Oca, J.; Ferrer, J.; Cremades, J.; Pintado, J.; Jiménez, P. Influence of Growing Conditions on Ulva Ohnoi Composition Cultivated in an IMTA-RAS System. Aquac Eur. 2016, 16, 6–8. [Google Scholar]
- Pinchetti, J.L.G.; Fernández, E.d.C.; Díez, P.M.; Reina, G.G. Nitrogen availability influences the biochemical composition and photosynthesis of tank-cultivated Ulva rigida (Chlorophyta). J. Appl. Phycol. 1998, 10, 383–389. [Google Scholar] [CrossRef]
- Lapointe, B.E.; Ryther, J.H. The effects of nitrogen and seawater flow rate on the growth and biochemical composition of Gracilaria foliifera Var. Angustissima in mass outdoor cultures. Bot. Mar. 1979, 22, 529–537. [Google Scholar] [CrossRef]
- Frikha, F.; Kammoun, M.; Hammami, N.; Mchirgui, R.; Belbahri, L.; Gargouri, Y.; Miled, N.; Ben-Rebah, F. Chemical composition and some biological activities of marine algae collected in Tunisia. Cienc. Mar. 2011, 37, 113–124. [Google Scholar] [CrossRef] [Green Version]
- Rupérez, P. Mineral content of edible marine seaweeds. Food Chem. 2002, 79, 23–26. [Google Scholar] [CrossRef]
- Mabeau, S.; Fleurence, J. Seaweed in food products: Biochemical and nutritional aspects. Trends Food Sci. Technol. 1993, 4, 103–107. [Google Scholar] [CrossRef]
- Astorga-España, M.S.; Rodríguez Galdón, B.; Rodríguez Rodríguez, E.M.; Díaz Romero, C. Mineral and trace element concentrations in seaweeds from the Sub-Antarctic ecoregion of Magallanes (Chile). J. Food Compost. Anal. 2015, 39, 69–76. [Google Scholar] [CrossRef]
- Khairy, H.M.; El-Sheikh, M.A. Antioxidant activity and mineral composition of three Mediterranean common seaweeds from Abu-Qir Bay, Egypt. Saudi J. Biol. Sci. 2015, 22, 623–630. [Google Scholar] [CrossRef] [Green Version]
- Koliaki, C.; Katsilambros, N. Dietary sodium, potassium, and alcohol: Key players in the pathophysiology, prevention, and treatment of human hypertension. Nutr. Rev. 2013, 71, 402–411. [Google Scholar] [CrossRef] [PubMed]
- Moreda-Piñeiro, J.; Alonso-Rodríguez, E.; López-Mahía, P.; Muniategui-Lorenzo, S.; Prada-Rodríguez, D.; Moreda-Piñeiro, A.; Bermejo-Barrera, P. Development of a new sample pre-treatment procedure based on pressurized liquid extraction for the determination of metals in edible seaweed. Anal. Chim. Acta 2007, 598, 95–102. [Google Scholar] [CrossRef]
- Khairy, H.M.; El-Shafay, S.M. Seasonal variations in the biochemical composition of some common seaweed species from the Coast of Abu Qir Bay, Alexandria, Egypt. Oceanologia 2013, 55, 435–452. [Google Scholar] [CrossRef] [Green Version]
- El Maghraby, D.M.; Fakhry, E.M. Lipid content and fatty acid composition of Mediterranean macro-algae as dynamic factors for biodiesel production. Oceanologia 2015, 57, 86–92. [Google Scholar] [CrossRef] [Green Version]
- Nelson, M.M.; Phleger, C.F.; Nichols, P.D. Seasonal lipid composition in macroalgae of the Northeastern Pacific Ocean. Bot. Mar. 2002, 45, 58–65. [Google Scholar] [CrossRef]
- Floreto, E.A.T.; Teshima, S.; Ishikawa, M. Effects of nitrogen and phosphorus on the growth and fatty acid composition of Ulva pertusa Kjellman (Chlorophyta). Bot. Mar. 1996, 39, 69–74. [Google Scholar] [CrossRef]
- Simopoulos, A.P. The importance of the omega-6/omega-3 fatty acid ratio in cardiovascular disease and other chronic diseases. Exp. Biol. Med. 2008, 233, 674–688. [Google Scholar] [CrossRef]
- Magdugo, R.P.; Terme, N.; Lang, M.; Pliego-Cortés, H.; Marty, C.; Hurtado, A.Q.; Bedoux, G.; Bourgougnon, N. An analysis of the nutritional and health values of Caulerpa racemosa (Forsskål) and Ulva fasciata (Delile)—Two chlorophyta collected from the Philippines. Molecules 2020, 25, 2901. [Google Scholar] [CrossRef]
- Godard, M.; Décordé, K.; Ventura, E.; Soteras, G.; Baccou, J.-C.; Cristol, J.-P.; Rouanet, J.-M. Polysaccharides from the green alga Ulva rigida improve the antioxidant status and prevent fatty streak lesions in the high cholesterol fed hamster, an animal model of nutritionally-induced atherosclerosis. Food Chem. 2009, 115, 176–180. [Google Scholar] [CrossRef]
- Ortiz, J.; Romero, N.; Robert, P.; Araya, J.; Lopez-Hernández, J.; Bozzo, C.; Navarrete, E.; Osorio, A.; Rios, A. Dietary fiber, amino acid, fatty acid and tocopherol contents of the edible seaweeds Ulva lactuca and Durvillaea antarctica. Food Chem. 2006, 99, 98–104. [Google Scholar] [CrossRef]
- Jard, G.; Marfaing, H.; Carrère, H.; Delgenes, J.P.; Steyer, J.P.; Dumas, C. French Brittany macroalgae screening: Composition and methane potential for potential alternative sources of energy and products. Bioresour. Technol. 2013, 144, 492–498. [Google Scholar] [CrossRef]
- Lahaye, M. Marine Algae as sources of fibres: Determination of soluble and insoluble dietary fibre contents in some ‘sea vegetables’. J. Sci. Food Agric. 1991, 54, 587–594. [Google Scholar] [CrossRef]
- Jiménez-Escrig, A.; Sánchez-Muniz, F.J. Dietary fibre from edible seaweeds: Chemical structure, physicochemical properties and effects on cholesterol metabolism. Nutr. Res. 2000, 20, 585–598. [Google Scholar] [CrossRef]
- Lapointe, B.E.; Tenore, K.R. Experimental outdoor studies with Ulva fasciata Delile. I. Interaction of light and nitrogen on nutrient uptake, growth, and biochemical composition. J. Exp. Mar. Biol. Ecol. 1981, 53, 135–152. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).