The Role of Water in the Effect of Weak Combined Magnetic Fields on Production of Reactive Oxygen Species (ROS) by Neutrophils
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
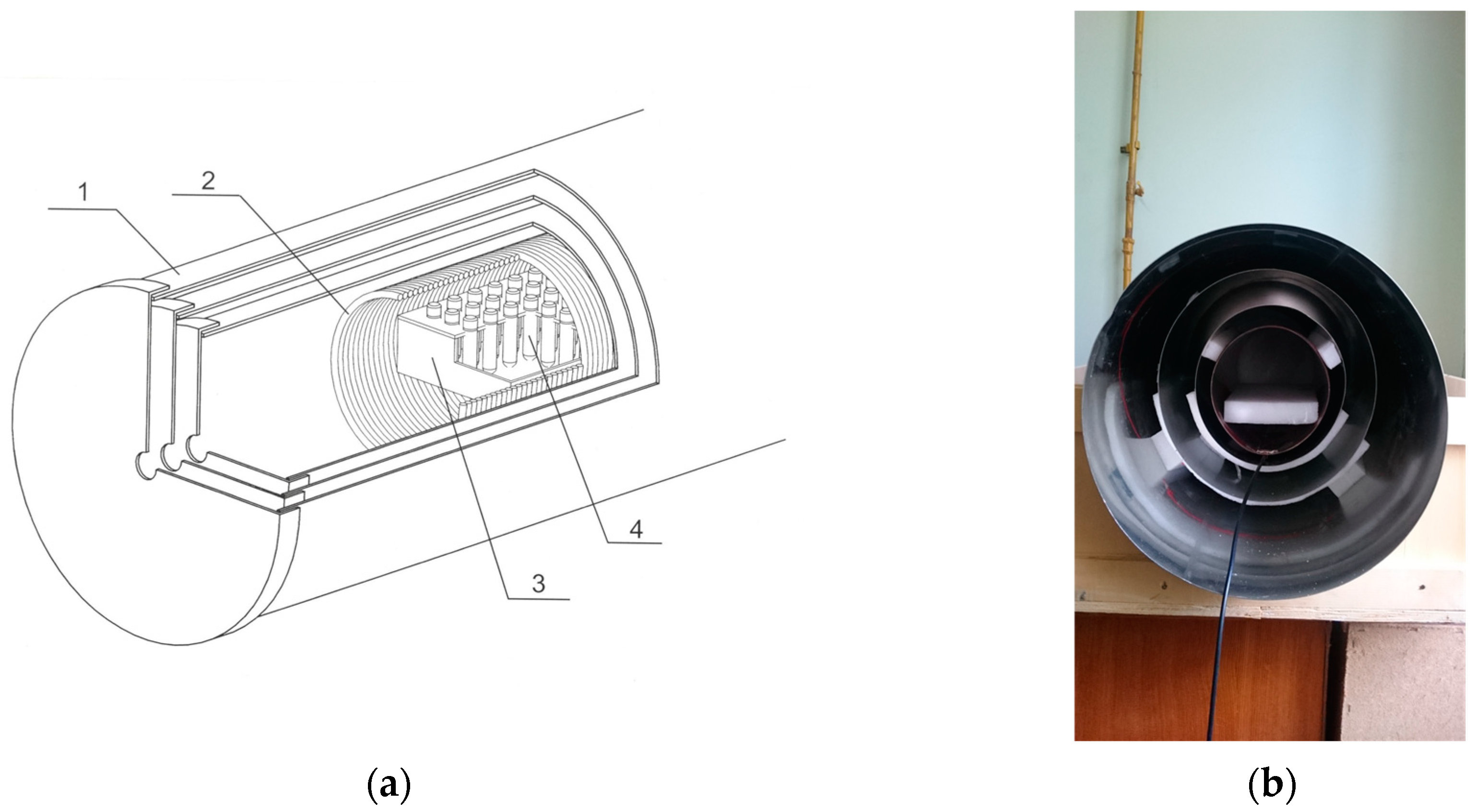
2.2. CMF Generation
2.2.1. Generation of the CMF with a Single Frequency Regime
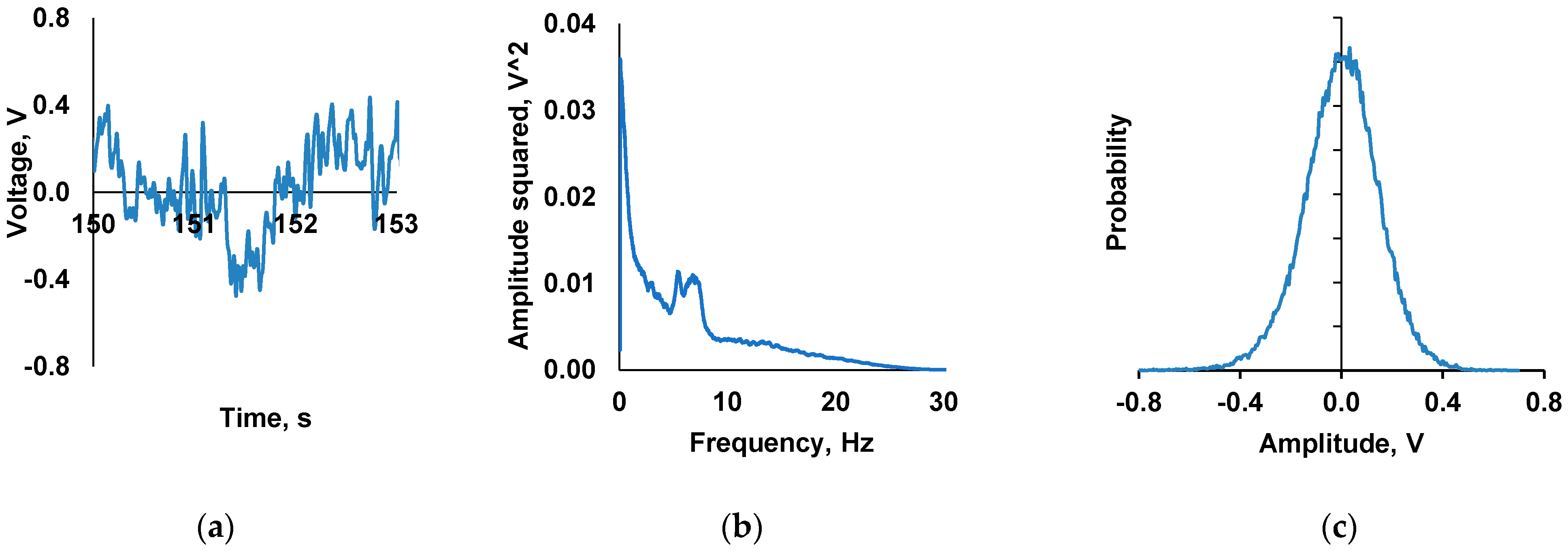
2.2.2. Generation of the CMF with a Spectral Range Regime
2.3. Obtaining Neutrophil Suspension
2.4. Neutrophil Suspension Exposure to the Combined Magnetic Field
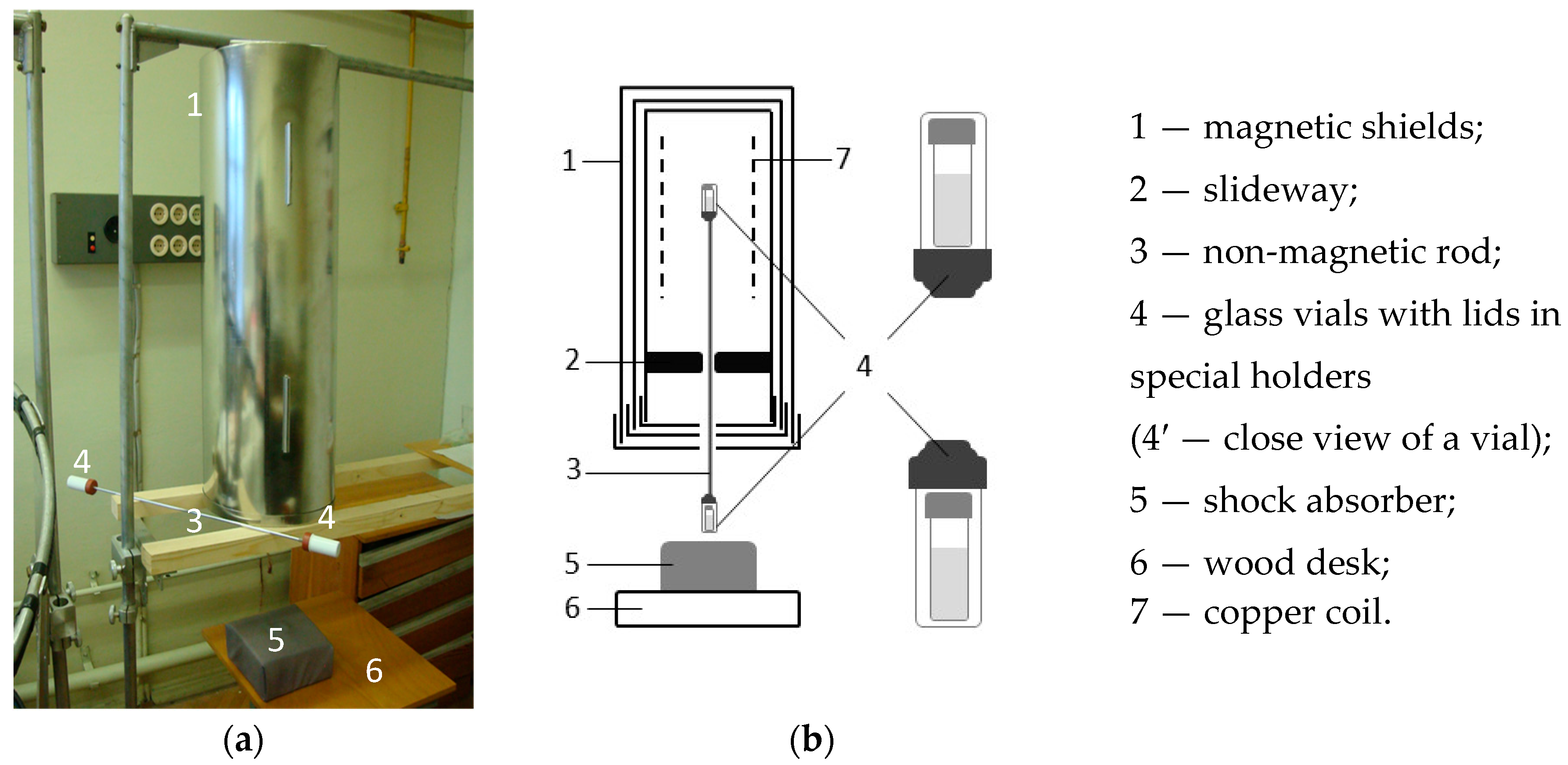
2.5. CMF Water Treatment with a Series of Dilutions and Vigorous Shaking
2.5.1. Sample Preparation for Investigation of Water-Mediated Activation of Neutrophils. The Role of Dilutions, Magnetic Vacuum, and Methods of Mechanical Treatment
- Homonymous “Experimental” sample measured without any additional exposure.
- “GMF, dilution factor 1099”—the sample was subjected to 50 serial centesimal dilutions with vigorous shaking between dilution steps. Both dilution and shaking procedures were performed under the GMF conditions (outside the magnetic field induction device).
- “Magnetic vacuum”, dilution factor 1099”—the sample was subjected to 50 serial centesimal dilutions with vigorous shaking between dilution steps. However, dilutions were made under the GMF conditions and each shaking was performed inside the magnetic field induction device with disconnected power supply and with residual magnetic field intensity ~10 nT. Noteworthy, “GMF, dilution factor 1099” and “Magnetic vacuum” samples were subjected to shaking identically due to the fact that they were interconnected by a rigid nonmagnetic bar at the time of shaking (Figure 3).
- “Dilution without shaking, dilution factor 1099”—the sample was subjected to 50 serial centesimal dilutions, but without vigorous shaking under the GMF conditions.
2.5.2. Preparation of Samples to Investigate Water-Mediated Activation of Neutrophils. The Role of Single-Frequency Combined Magnetic Field at Dilution
- Homonymous “Experimental” sample measured without any additional exposure.
- “in GMF, dilution factor 1099”—the sample was subjected to 50 serial centesimal dilutions with vigorous shaking between dilution steps. Both dilution and shaking procedures were performed under the GMF conditions (outside the magnetic field induction device).
- “in CMF, dilution factor 1099”—the sample was subjected to 50 serial centesimal dilutions with vigorous shaking between dilution steps. The dilutions were made under the GMF conditions and shaking—inside the magnetic field induction device (CMF) with static component of 60 µT and alternating component of 100 nT, which was formed according to the sinusoidal regime with 12.6 Hz. Noteworthy, “GMF, dilution factor 1099” and “Magnetic vacuum” samples were subjected to shaking identically due to the fact that they were interconnected by a rigid nonmagnetic bar at the time of shaking (Figure 3).
2.5.3. Preparation of Samples to Investigate Water-Mediated Activation of Neutrophils. The Role of Mechanical Treatment Under Geomagnetic Field Conditions
- Homonymous “Control” sample measured without any additional exposure.
- “in GMF” (without shaking), dilution factor 1099”—the sample was subjected to 50 serial centesimal dilutions under the GMF conditions, but without vigorous shaking between dilution steps.
- “in GMF, dilution factor 1099”—the sample was subject to 50 serial centesimal dilutions with vigorous shaking between dilution steps. Both dilution and shaking were performed under the GMF conditions (outside the magnetic field induction device).
2.5.4. Preparation of Samples to Investigate Water-Mediated Activation of Neutrophils. The Role of the CMF Formed with Spectral Signal and Dilution Method
- Homonymous “Experimental” sample measured without any additional exposure.
- “in GMF, dilution factor 1099”—the sample was subjected to 50 serial centesimal dilutions with vigorous shaking between dilution steps. Both dilutions and shaking were performed under CMF conditions with static component of 60 µT and alternating component of 100 nT, which was formed by a random signal (described in the subsection “Generation of the CMF with a spectral range regime”).
2.6. Measurement of Luminol-Enhanced Cell Chemiluminescence
2.7. Statistical Analysis
3. Results
3.1. Effect of Artificial and Geomagnetic Field on Neutrophil Activity
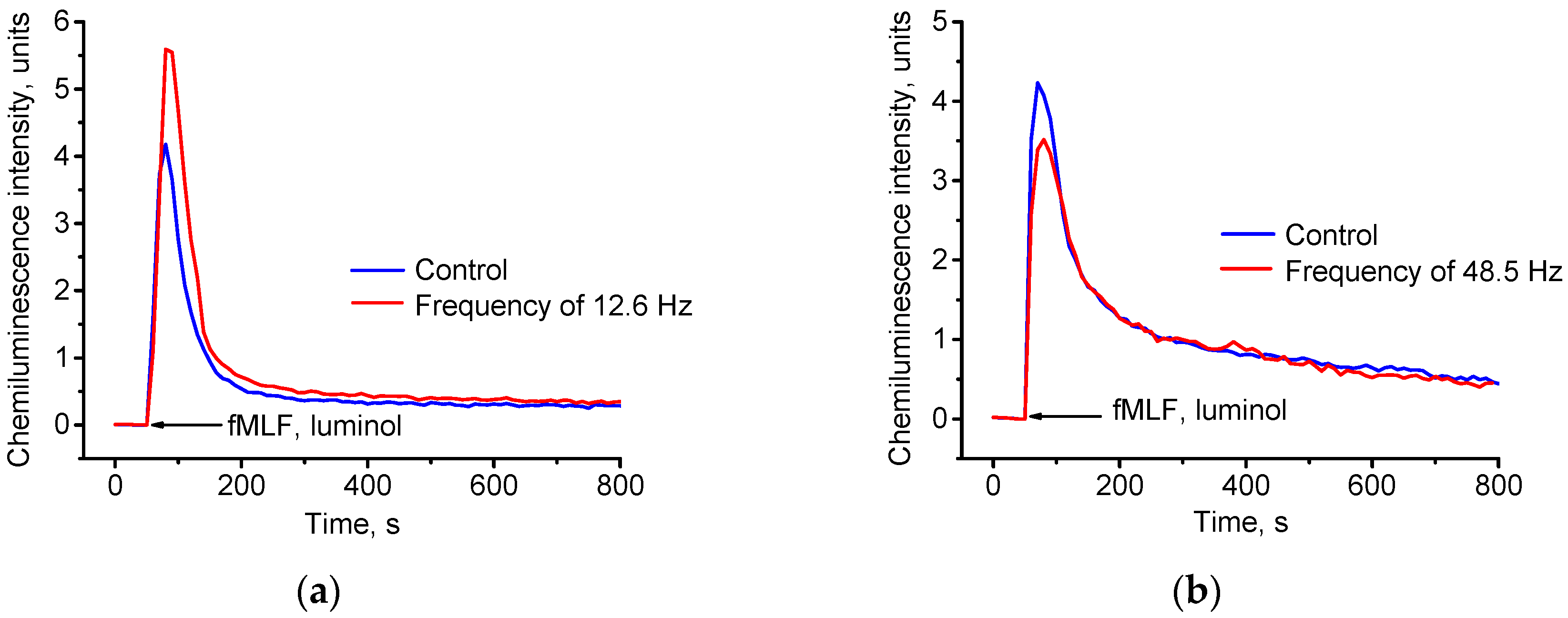
3.2. Effect of Combined Magnetic Fields on ROS Generation by Neutrophils
3.2.1. Direct Neutrophil Exposure to the Magnetic Field
3.2.2. Water-Mediated Effect of Magnetic Fields on Neutrophils
3.2.3. Water-Mediated Activation of Neutrophils with Samples Exposed to Magnetic Fields, Dilution, and Intensive Mechanical Treatment
- “GMF at 1099 dilution”—dilution and shaking were performed under the GMF (outside the magnetic field induction device).
- “Magnetic vacuum” at 1099 dilution”—dilutions were made under the GMF (outside the magnetic field induction device), while shaking was made inside the device under the so-called “magnetic vacuum” (~10 nT).
- “Experimental sample dilution to 1099 without shaking”—dilutions were made under the GMF (outside the magnetic field induction device), but without shaking between dilution steps.
3.2.4. Water-Mediated Activation of Neutrophils Exposed to the Spectral-Formed CMF with Intensive Mechanical Exposure and Dilution
4. Discussion
5. Conclusions
- (1)
- The results of our experiments demonstrate that water exposure to a physical effect in the form of CMF with alternating component, which changes by sinusoidal signal at 12.6 Hz, modifies its properties (66% increase in intensity of neutrophil suspension chemiluminescence relative to control samples).
- (2)
- These properties are retained during serial dilution and vigorous shaking of the exposed water (44% increase in chemiluminescence of neutrophil suspension relative to control samples).
- (3)
- Similar but less pronounced indirect effect of the CMF pre-exposed water is observed when using the CMF formed by a random signal proportional to electroencephalogram recording (which also includes 12.6 Hz band). Chemiluminescence intensity increased both after adding to the cells the CMF pre-exposed water (56% increase) and after adding the same CMF pre-exposed water but mechanically treated between serial dilutions (33% increase).
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Novikov, V.V.; Yablokova, E.V.; Fesenko, E.E. Priming of the respiratory burst in neutrophils exposed to a combination of weak constant and alternating low-frequency magnetic fields in vitro. Biophysics 2016, 61, 429–434. [Google Scholar] [CrossRef]
- Dahlgren, C.; Karlsson, A. Respiratory burst in human neutrophils. J. Immunol. Methods 1999, 232, 3–14. [Google Scholar] [CrossRef]
- Novikov, G.V.; Novikov, V.V.; Fesenko, E.E. Effect of weak combined static and low-frequency alternating magnetic fields on the Ehrlich ascites carcinoma in mice. Biophysics 2009, 54, 741–747. [Google Scholar] [CrossRef]
- Novikov, V.V.; Novikov, G.V.; Fesenko, E.E. Effect of weak combined static and extremely low-frequency alternating magnetic fields on tumor growth in mice inoculated with the Ehrlich ascites carcinoma. Bioelectromagnetics 2009, 30, 343–351. [Google Scholar] [CrossRef]
- Novikov, V.V.; Ponomarev, V.O.; Novikov, G.V.; Kuvichkin, V.V.; Yablokova, E.V.; Fesenko, E.E. Effects and molecular mechanisms of the biological action of weak and extremely weak magnetic fields. Biophysics 2010, 55, 565–572. [Google Scholar] [CrossRef]
- Novikov, V.V.; Yablokova, E.V.; Novikov, G.V.; Fesenko, E.E. The role of lipid peroxidation and myeloperoxidase in priming a respiratory burst in neutrophils under the action of combined constant and alternating magnetic fields. Biophysics 2017, 62, 759–763. [Google Scholar] [CrossRef]
- Novikov, V.V.; Yablokova, E.V.; Fesenko, E.E. The role of hydroxyl radicals and calcium ions in the priming of a respiratory burst in neutrophils and the increase in luminol-dependent blood chemiluminescence on exposure to combined magnetic fields with a very weak low-frequency alternating component. Biophysics 2017, 62, 440–443. [Google Scholar] [CrossRef]
- Novikov, V.V.; Yablokova, E.V.; Fesenko, E.E. The Role of Oxygen in the Priming of Neutrophils on Exposure to a Weak Magnetic Field. Biophysics 2018, 63, 193–196. [Google Scholar] [CrossRef]
- Karelina, E.A.; Ganina, K.K.; Kosmachev, V.N.; Tarasov, S.A. Results of the blind placebo-controlled trial of the novel anti-stress drug Anoten efficacy against neurotic disoders in dogs. Ross. Vet. (Russ. Vet. J.) 2018, 2, 39–42. [Google Scholar]
- Penkov, N. Peculiarities of the Perturbation of Water Structure by Ions with Various Hydration in Concentrated Solutions of CaCl 2, CsCl, KBr, and KI. Phys. Wave Phenom. 2019, 27, 128–134. [Google Scholar] [CrossRef]
- Epstein, O. The spatial homeostasis hypothesis. Symmetry 2018, 10, 103. [Google Scholar] [CrossRef]
- D’Emilia, E.; Ledda, M.; Foletti, A.; Lisi, A.; Giuliani, L.; Grimaldi, S.; Liboff, A.R. Weak-field H3O(+) ion cyclotron resonance alters water refractive index. Electromagn. Biol. Med. 2017, 36, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Noachtar, S.; Binnie, C.; Ebersole, J.; Mauguiere, F.; Sakamoto, A.; Westmoreland, B. A glossary of terms most commonly used by clinical electroencephalographers and proposal for the report form for the EEG findings. The International Federation of Clinical Neurophysiology. Electroencephalogr. Clin. Neurophysiol. Suppl. 1999, 52, 21–41. [Google Scholar] [PubMed]
- Fesenko, E.E.; Gluvstein, A. Changes in the state of water, induced by radiofrequency electromagnetic fields. FEBS Lett. 1995, 367, 53–55. [Google Scholar] [CrossRef]
- Fesenko, E.E.; Geletyuk, V.I.; Kazachenko, V.N.; Chemeris, N.K. Preliminary microwave irradiation of water solutions changes their channel-modifying activity. FEBS Lett. 1995, 366, 49–52. [Google Scholar] [CrossRef]
- Liboff, A.R. Geomagnetic cyclotron resonance in living cells. J. Biol. Phys. 1985, 13, 99–102. [Google Scholar] [CrossRef]
- Liboff, A.R. ION cyclotron resonance: Geomagnetic strategy for living systems? Electromagn. Biol. Med. 2019, 38, 143–148. [Google Scholar] [CrossRef]
- D’Emilia, E.; Giuliani, L.; Lisi, A.; Ledda, M.; Grimaldi, S.; Montagnier, L.; Liboff, A. Lorentz force in water: Evidence that hydronium cyclotron resonance enhances polymorphism. Electromagn. Biol. Med. 2015, 34, 370–375. [Google Scholar] [CrossRef]
- Novikov, V.V.; Zhadin, M.N. Combined action of weak constant and variable low-frequency magnetic fields on ionic currents in aqueous solutions of amino acid. Biophysics 1994, 39, 41–45. [Google Scholar]
- Novikov, V.V. Initiate action of weak magnetic fields on intermolecular bond formation in aqueous solution of amino acids. Biophysics 1994, 39, 851–856. [Google Scholar]
- Novikov, V.V. Cooperative effect of the resonance amplification of ionic current in aqueous solutions of amino acids under the action of weak electromagnetic fields. Approaches to experimental and theoretical analysis. Biophysics 1996, 41, 983–988. [Google Scholar]
- Zhadin, M.N.; Novikov, V.V.; Barnes, F.S.; Pergola, N.F. Combined action of static and alternating magnetic fields on ionic current in aqueous glutamic acid solution. Bioelectromagnetics 1998, 19, 41–45. [Google Scholar] [CrossRef]
- Novikov, V.V.; Yablokova, E.V.; Fesenko, E.E. Respiratory burst reduction in neutrophils after exposure to certain modes of weak combined magnetic fields. Biophysics 2020, 65, 82–87. [Google Scholar] [CrossRef]
- Fesenko, E.E.; Novikov, V.V.; Kuvichkin, V.V.; Iablokova, E.V. Effect of treated with weak magnetic field aqueous salt solutions on the intrinsic fluorescence of bovine serum albumin. Isolation from solutions and partial characterization of the biologically active fluorescing fraction. Biophysics 2000, 45, 232–239. [Google Scholar]
- Fesenko, E.E.; Popov, V.I.; Novikov, V.V.; Khutsian, S.S. Water structure formation by weak magnetic fields and xenon. Electron microscopic analysis. Biophysics 2002, 47, 389–394. [Google Scholar]
- Novikov, V.V.; Kuvichkin, V.V.; Fesenko, E.E. Effect of weak combined low frequency static and low-frequency alternative magnetic fields on the intrinsic fluorescence of some proteins in aqueous solutions. Biophysics 1999, 44, 224–230. [Google Scholar]
- Novikov, V.V.; Fesenko, E.E. Hydrolysis of various peptides and proteins in weak permanent and low frequency fluctuating magnetic fields. Biofizika 2001, 46, 235–241. [Google Scholar]
- Novikov, V.V.; Sheiman, I.M.; Lisitsyn, A.S.; Kliubin, A.V.; Fesenko, E.E. Dependence of effects of weak combined low-frequency variable and constant magnetic fields on the intensity of asexual reproduction of planarians Dugesia tigrina on the magnitude of the variable field. Biofizika 2002, 47, 564–567. [Google Scholar]
- Fukushima, M.; Mohri, K.; Kataoka, T.; Matsumoto, M. Milli Gauss pursed Magnetic Field Applied Phosphate Buffeted Saline Solution Elevates Intracellular Ca2+ Level and Stimulates Phagocytic Activity of Human Neutrophils. Trans. Magn. Soc. Jpn. 2002, 2, 15–18. [Google Scholar] [CrossRef]
- Ayrapetyan, S.N.; Grigorian, K.V.; Avanesian, A.S.; Stamboltsian, K.V. Magnetic fields alter electrical properties of solutions and their physiological effects. Bioelectromagnetics 1994, 15, 133–142. [Google Scholar] [CrossRef]
- Ayrapetyan, S.; Hunanyan, A.S.; Hakobyan, S. 4 Hz EMF treated physiological solution depresses Ach-induced neuromembrane current. Bioelectromagnetics 2004, 25, 397–399. [Google Scholar] [CrossRef] [PubMed]
- Foletti, A.; Ledda, M.; Lolli, M.G.; Grimaldi, S.; Lisi, A. Electromagnetic information transfer through aqueous system. Electromagn. Biol. Med. 2017, 36, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Yinnon, T.; Kalia, K.; Kikar, D. Very Dilute Aqueous Solutions—Structural and Electromagnetic Phenomena. Water 2017, 9, 28–66. [Google Scholar]
- Konovalov, A.; Ryzhkina, I.; Maltzeva, E.; Murtazina, L.; Kiseleva, Y.; Kasparov, V.; Palmina, N. Nanoassociate formation in highly diluted water solutions of potassium phenosan with and without permalloy shielding. Electromagn. Biol. Med. 2015, 34, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Góra, U.; Podeszwa, R.; Cencek, W.; Szalewicz, K. Interaction energies of large clusters from many-body expansion. J. Chem. Phys. 2011, 135, 224102. [Google Scholar] [CrossRef] [PubMed]

| Experimental Neutrophil Group | Chemiluminescence Intensity (%) | Chemiluminescence Intensity (V) |
|---|---|---|
| GMF (Control) | 100.0 ± 9.7 | 3.38 ± 0.33 |
| SMF (60 µT) “sham control” | 103.6 ± 12.1 | 3.50 ± 0.42 |
| AMF Parameters | Chemiluminescence Intensity | |||
|---|---|---|---|---|
| Control | Experimental | |||
| % | V | % | V | |
| 12.6 Hz, 100 nT | 100 ± 3.0 | 4.23 ± 0.13 | 136.6 ± 8.6 | 5.78 ± 0.50 p = 0.002165 |
| 48.5 Hz, 100 nT | 100 ± 6.8 | 4.33 ± 0.29 | 81.5 ± 8.4 | 3.53 ± 0.30 p = 0.002165 |
| AMF Parameters | Water Samples | |||||
|---|---|---|---|---|---|---|
| Effect of Water Pre-Exposed to CMF | Effect of Water Pre-Exposed to CMF at Intense Mechanical Exposure and Dilution | |||||
| Control | Experimental | GMF | “Magnetic Vacuum” | CMF (without Shaking) | ||
| Dilution Factor 1099 | ||||||
| 12.6 Hz, 100 nT | V | 3.88 ± 0.37 | 6.46 ± 0.69 | 5.60 ± 0.79 | 3.72 ± 0.41 | 3.65 ± 0.50 |
| % | 100 ± 9.6 | 166.0 ± 18.0 | 144.3 ± 20.0 | 95.9 ± 10.2 | 94.1 ± 13.6 | |
| 48.5 Hz, 100 nT | V | 4.79 ± 0.37 | 5.02 ± 0.84 | 5.22 ± 0.79 | 4.64 ± 0.63 | 4.73 ± 0.64 |
| % | 100 ± 7.7 | 104.8 ± 16.7 | 109.0 ± 15.1 | 96.9 ± 9.1 | 99.0 ± 13.4 | |
| AMF Parameters | Water Samples | ||||
|---|---|---|---|---|---|
| Control | Experimental | GMF | CMF | ||
| Dilution Factor 1099 | |||||
| 12.6 Hz, 100 nT | V | 3.72 ± 0.27 | 5.68 ± 0.36 | 5.33 ± 0.35 | 5.08 ± 0.31 |
| % | 100 ± 7.2 | 152.7 ± 6.4 | 143.3 ± 6.6 | 136.6 ± 6.1 | |
| 48.5 Hz, 100 nT | V | 3.30 ± 0.42 | 3.44 ± 0.28 | 3.32 ± 0.36 | 3.69 ± 0.49 |
| % | 100 ± 12.7 | 104.2 ± 8.1 | 100.6 ± 11.0 | 111.8 ± 13.4 | |
| Field Parameters | Control | GMF (Without Shaking) | GMF | |
|---|---|---|---|---|
| Dilution Factor 1099 | ||||
| GMF | V | 2.37 ± 0.25 | 2.26 ± 0.20 | 2.20 ± 0.31 |
| % | 100 ± 10.5 | 95.4 ± 9.1 | 92.8 ± 14.1 | |
| CMF Parameters. | Control | Experimental | In CMF | |
|---|---|---|---|---|
| Dilution Factor 1099 | ||||
| Static 60 µT; Alternating ~100 nT; Modulation with a random signal | V | 2.37 ± 0.25 | 3.69 ± 0.97 | 3.15 ± 0.76 |
| % | 100 ± 11 | 156 ± 41 | 133 ± 32 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Novikov, V.V.; Yablokova, E.V.; Fesenko, E.E. The Role of Water in the Effect of Weak Combined Magnetic Fields on Production of Reactive Oxygen Species (ROS) by Neutrophils. Appl. Sci. 2020, 10, 3326. https://doi.org/10.3390/app10093326
Novikov VV, Yablokova EV, Fesenko EE. The Role of Water in the Effect of Weak Combined Magnetic Fields on Production of Reactive Oxygen Species (ROS) by Neutrophils. Applied Sciences. 2020; 10(9):3326. https://doi.org/10.3390/app10093326
Chicago/Turabian StyleNovikov, Vadim V., Elena V. Yablokova, and Evgeny E. Fesenko. 2020. "The Role of Water in the Effect of Weak Combined Magnetic Fields on Production of Reactive Oxygen Species (ROS) by Neutrophils" Applied Sciences 10, no. 9: 3326. https://doi.org/10.3390/app10093326
APA StyleNovikov, V. V., Yablokova, E. V., & Fesenko, E. E. (2020). The Role of Water in the Effect of Weak Combined Magnetic Fields on Production of Reactive Oxygen Species (ROS) by Neutrophils. Applied Sciences, 10(9), 3326. https://doi.org/10.3390/app10093326




