Fluoride and Pineal Gland
Abstract
1. Introduction
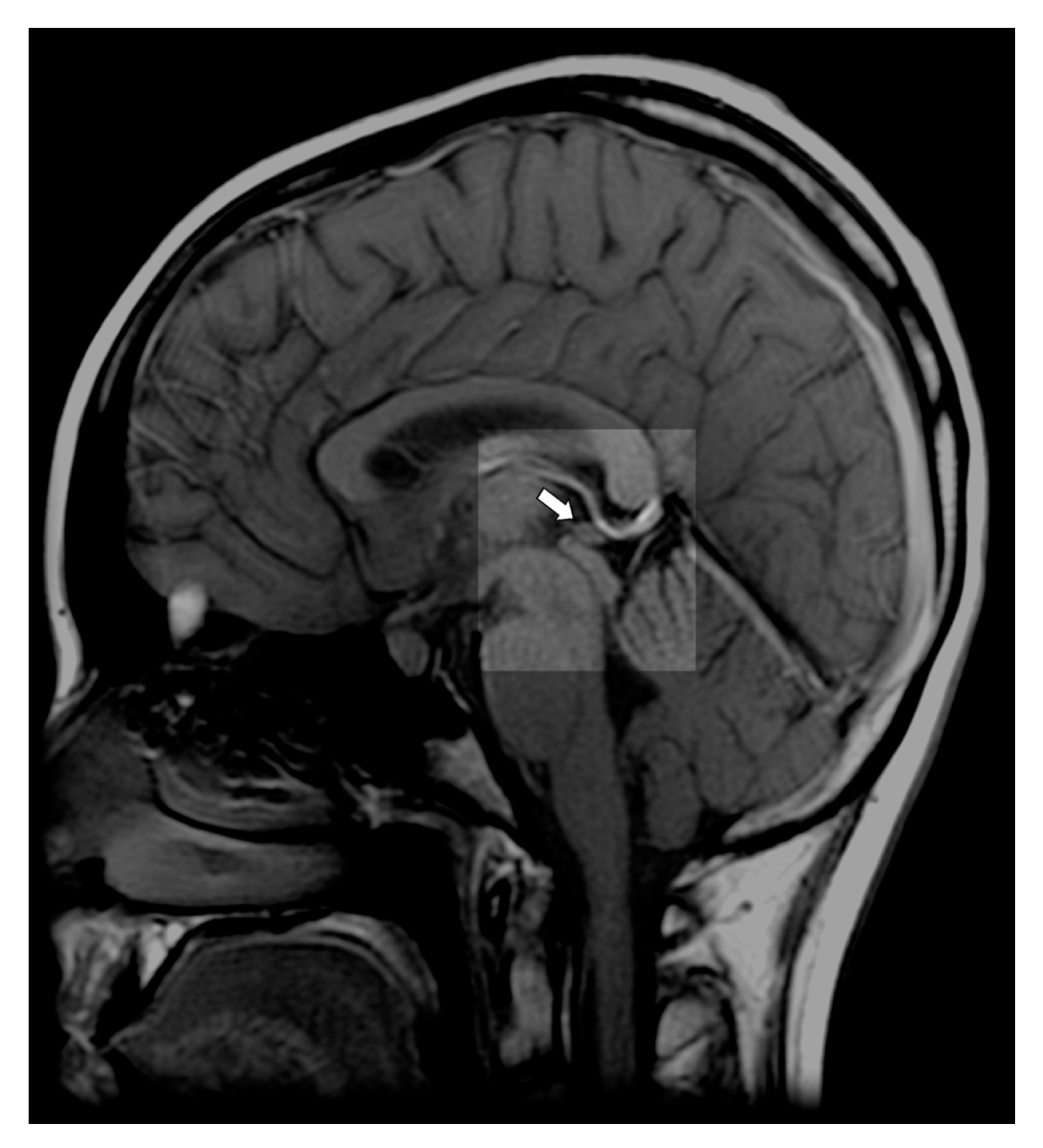
2. Pineal Gland—Anatomy and Physiology
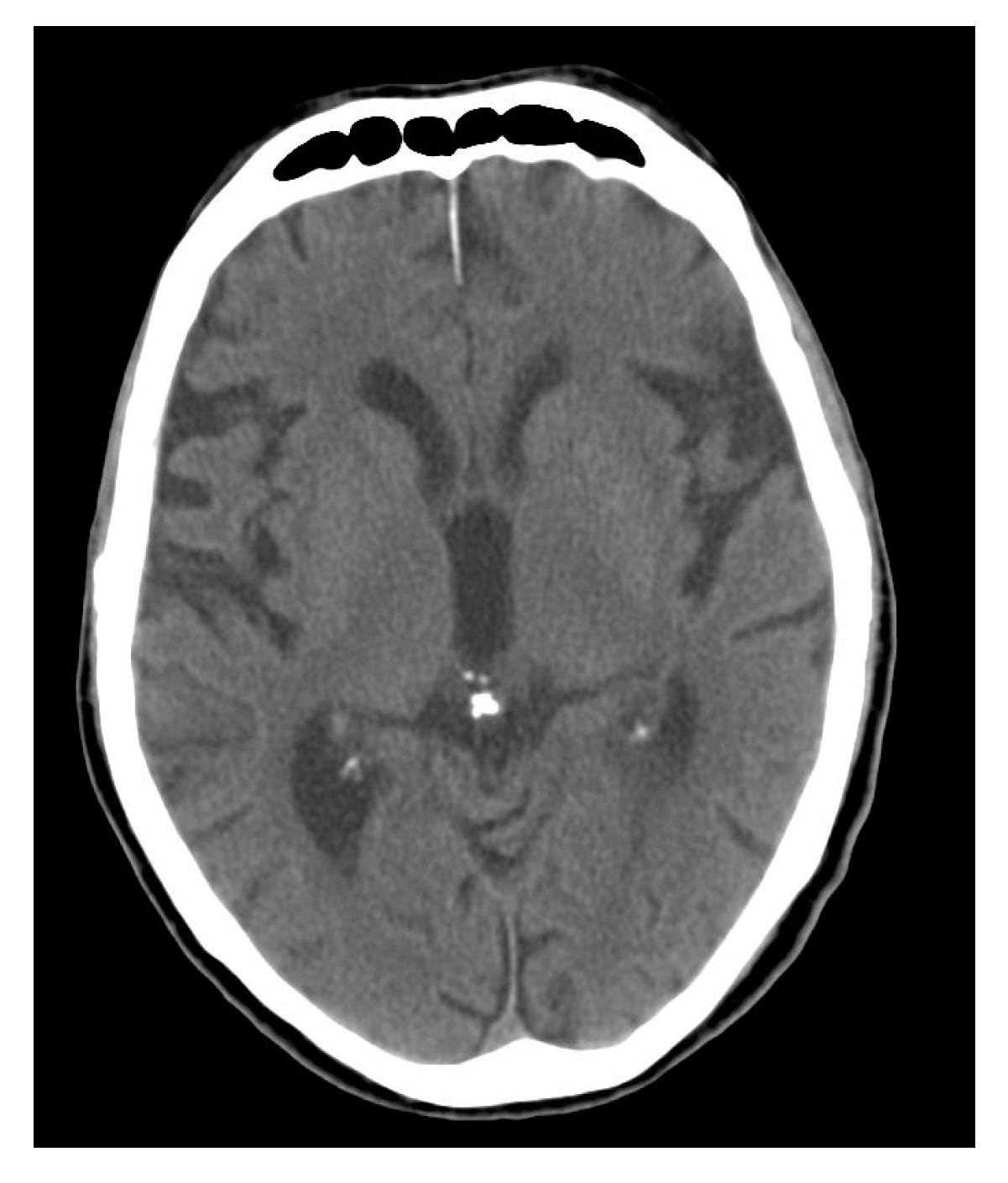
3. Calcium Accumulation in the Pineal Gland
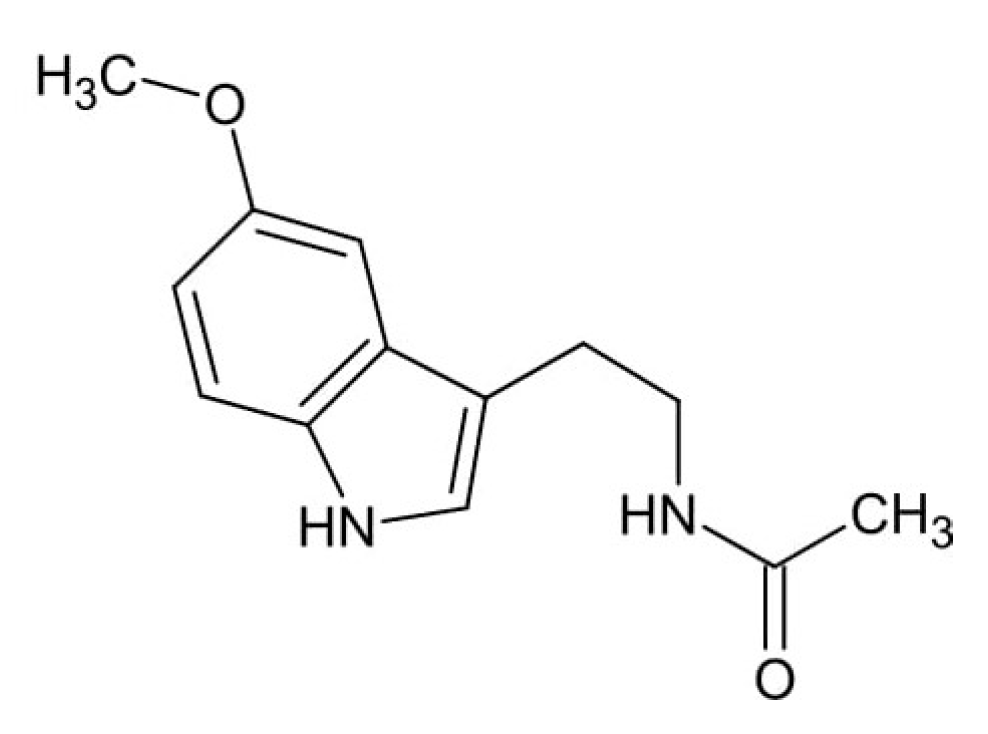
4. Fluoride Accumulation in the Pineal Gland and Its Consequences
Author Contributions
Funding
Conflicts of Interest
References
- Kalisińska, E.; Baranowska-Bosiacka, I.; Łanocha, N.; Kosik-Bogacka, D.; Królaczyk, K.; Wilk, A.; Kavetska, K.; Budis, H.; Gutowska, I.; Chlubek, D. Fluoride concentrations in the pineal gland, brain and bone of goosander (Mergus merganser) and its prey in Odra River estuary in Poland . Environ. Geochem. Health 2014, 36, 1063–1077. [Google Scholar] [CrossRef] [PubMed]
- Kanduti, D.; Sterbenk, P.; Artnik, B. Fluoride: A review of use and effects on health. Mater. Sociomed. 2016, 28, 133–137. [Google Scholar] [CrossRef]
- Kupnicka, P.; Kojder, K.; Metryka, E.; Kapczuk, P.; Jeżewski, D.; Gutowska, I.; Goschorska, M.; Chlubek, D.; Baranowska-Bosiacka, I. Morphine-element interactions—The influence of selected chemical elements on neural pathways associated with addiction. J. Trace Elem. Med. Biol. 2020, 60, 126495. [Google Scholar] [CrossRef]
- Choi, A.L.; Sun, G.; Zhang, Y.; Grandjean, P. Developmental fluoride neurotoxicity: A systematic review and meta-analysis. Environ. Health Perspect. 2012, 10, 1362–1368. [Google Scholar] [CrossRef]
- Dec, K.; Łukomska, A.; Maciejewska, D.; Jakubczyk, K.; Baranowska-Bosiacka, I.; Chlubek, D.; Wąsik, A.; Gutowska, I. The influence of fluorine on the disturbances of homeostasis in the central nervous system. Biol. Trace Elem. Res. 2017, 177, 224–234. [Google Scholar] [CrossRef]
- Duan, Q.; Jiao, J.; Chen, X.; Wang, X. Association between water fluoride and the level of children’s intelligence: A dose-response meta-analysis. Public Health 2018, 154, 87–97. [Google Scholar] [CrossRef]
- Ghosh, A.; Mukherjee, K.; Ghosh, S.K.; Saha, B. Sources and toxicity of fluoride in the environment. Res. Chem. Intermediat. 2013, 39, 2881–2915. [Google Scholar] [CrossRef]
- Palczewska-Komsa, M.; Barczak, K.; Kotwas, A.; Sikora, M.; Chlubek, D.; Buczkowska-Radlińska, J. Fluoride concentration in dentin of human permanent teeth. Fluoride 2019, 52, 489–496. [Google Scholar]
- Waszkiel, D.; Opalko, K.; Łagocka, R.; Chlubek, D. Fluoride and magnesium content in superficial enamel layers of teeth with erosions. Fluoride 2004, 37, 285–291. [Google Scholar]
- Buzalaf, M.A.; Whitford, G.M. Fluoride metabolism. Monogr. Oral Sci. 2011, 22, 20–36. [Google Scholar] [PubMed]
- Malin, A.J.; Bose, S.; Busgang, S.A.; Gennings, C.; Thorpy, M.; Wright, R.O.; Wright, R.J.; Arora, M. Fluoride exposure and sleep patterns among older adolescents in the United States: A cross-sectional study of NHANES 2015–2016. Environ. Health 2019, 18, 106. [Google Scholar] [CrossRef] [PubMed]
- ten Cate, J.M.; Featherstone, J.D. Mechanistic aspects of the interactions between fluoride and dental enamel. Crit. Rev. Oral Biol. Med. 1991, 2, 283–296. [Google Scholar] [CrossRef] [PubMed]
- Mokrzyński, S.; Machoy, Z. Fluoride incorporation into fetal bone. Fluoride 1994, 27, 151–154. [Google Scholar]
- Mokrzyński, S.; Chlubek, D.; Mikulski, T.; Machoy, Z. The use of microdensitometric examinations for evaluating the influence of fluorine on bone mineralization in fetus. Pol. Przegl. Radiol. 1994, 58, 62–64. [Google Scholar]
- Mokrzyński, S.; Chlubek, D.; Machoy, Z.; Samujło, D. Fluoride in the organism of mother and fetus. II. Fluoride cumulation in the organism of fetus. Ginekol. Pol. 1994, 65, 678–681. [Google Scholar] [PubMed]
- Sikora, M.; Kwiatkowska, B.; Chlubek, D. Fluoride content in superficial enamel layers of human teeth from archeological excavations. Fluoride 2014, 47, 341–348. [Google Scholar]
- Chlubek, D.; Noceń, I.; Dąbkowska, E.; Żyluk, B.; Machoy, Z.; Kwiatkowska, B. Fluoride accumulation in human skulls in relation to chronological age. Fluoride 1996, 29, 131–134. [Google Scholar]
- Chlubek, D.; Sikora, M.; Kwiatkowska, B.; Gronkiewicz, S. Determinations of mineral composition in superficial enamel layers of human teeth from archeological excavations by means of enamel biopsy. Biul. Magnezol. 2001, 6, 110–117. [Google Scholar]
- Patil, M.M.; Lakhkar, B.B.; Patil, S.S. Course of fluorosis. Indian J. Pediatr. 2018, 85, 375–383. [Google Scholar] [CrossRef]
- Shruthi, M.N.; Anil, N.S. A comparative study of dental fluorosis and non-skeletal manifestations of fluorosis in areas with different water fluoride concentrations in rural Kolar. J. Family Med. Prim. Care. 2018, 7, 1222–1228. [Google Scholar]
- Rajapakse, P.S.; Jayawardhane, W.M.; Lokubandara, A.; Gamage, R.; Dasanayake, A.P.; Goonaratna, C. High prevalence of dental fluorosis among schoolchildren in three villages in Vavuniya District: An observational study. Ceylon Med. J. 2017, 62, 218–221. [Google Scholar] [CrossRef] [PubMed]
- Hewavithana, P.B.; Jayawardhane, W.M.; Gamage, R.; Goonaratna, C. Skeletal fluorosis in Vavuniya District: An observational study. Ceylon Med. J. 2018, 63, 139–142. [Google Scholar] [CrossRef] [PubMed]
- Sellami, M.; Riahi, H.; Maatallah, K.; Ferjani, H.; Bouaziz, M.C.; Ladeb, M.F. Skeletal fluorosis: Don’t miss the diagnosis! Skeletal Radiol. 2020, 49, 345–357. [Google Scholar] [CrossRef] [PubMed]
- Aoba, T.; Fejerskov, O. Dental fluorosis: Chemistry and biology. Crit. Rev. Oral Biol. Med. 2002, 13, 155–170. [Google Scholar] [CrossRef]
- Waldbott, G.L. Fluoride and calcium levels in the aorta. Exeprientia 1966, 22, 835–837. [Google Scholar] [CrossRef] [PubMed]
- Zipkin, I.; Zucas, S.M.; Lavender, D.R.; Fullmer, H.M.; Schiffmann, E.; Corcoran, B.A. Fluoride and calcification of rat aorta. Calcif. Tissue Res. 1970, 6, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Susheela, A.K.; Kharb, P. Aortic calcification in chronic fluoride poisoning: Biochemical and electronmicroscopic evidence. Exp. Mol. Pathol. 1990, 53, 72–80. [Google Scholar] [CrossRef]
- Fiz, F.; Morbelli, S.; Bauckneht, M.; Piccardo, A.; Ferrarazzo, G.; Nieri, A.; Artom, N.; Cabria, M.; Marini, C.; Canepa, M.; et al. Correlation between thoracic aorta 18F-natrium fluoride uptake and cardiovascular risk. World J. Radiol. 2016, 8, 82–89. [Google Scholar] [CrossRef]
- Ericsson, Y.; Hammarström, L. Autoradiographic localization of fluoride and calcium deposition in the atherosclerotic aorta of cholesterol-fed rabbits. Gerontology 1964, 9, 150–156. [Google Scholar] [CrossRef]
- Li, Y.; Berenji, G.R.; Shaba, W.F.; Tafti, B.; Yevdayev, E.; Dadparvar, S. Association of vascular fluoride uptake with vascular calcification and coronary artery disease. Nucl. Med. Commun. 2012, 33, 14–20. [Google Scholar] [CrossRef]
- Chen, W.; Dilsizian, V. Targeted PET/CT imaging of vulnerable atherosclerotic plaques: Microcalcification with sodium fluoride and inflammation with fluorodeoxyglucose. Curr. Cardiol. Rep. 2013, 15, 364. [Google Scholar] [CrossRef]
- Chlubek, D.; Poręba, R.; Machaliński, B. Fluoride and calcium distribution in human placenta. Fluoride 1998, 31, 131–136. [Google Scholar]
- Chlubek, D.; Rzeuski, R. Toxic effects of fluorine compounds on the fetus and their effect on the course of pregnancy. Ginekol. Pol. 1996, 67, 141–418. [Google Scholar]
- Shen, Y.W.; Taves, D.R. Fluoride concentrations in human placenta and maternal and cord blood. Am. J. Obstet. Gynecol. 1974, 119, 205–207. [Google Scholar] [CrossRef]
- Gurumurthy, S.M.; Mohanty, S.; Vyakaranam, S.; Bhongir, A.V.; Rao, P. Transplacental transport of fluoride, calcium and magnesium. Natl. J. Integr. Res. Med. 2011, 2, 51–55. [Google Scholar]
- Gurumurthy, S.M.; Mohanty, S.; Rao, P. Role of placenta to combat fluorosis (in fetus) in endemic fluorosis area. Natl. J. Integr. Res. Med. 2010, 1, 16–19. [Google Scholar]
- Chlubek, D.; Machoy, Z.; Samujło, D. Fluoride concentration in human placenta in the region of fluorine industrial emissions. Bromat. Chem. Toksykol. 1997, 30, 299–302. [Google Scholar]
- Feltman, R.; Kosel, G. Prenatal ingestion of fluorides and their transfer to the fetus. Science 1955, 122, 560–561. [Google Scholar] [CrossRef]
- Chlubek, D.; Machoy, Z. Role of placenta in fluoride metabolism. Ginekol. Pol. 1991, 42, 568–572. [Google Scholar]
- Chlubek, D. Some aspects of prenatal fluoride metabolism in humans. Studies performed during the perinatal period. Ann. Acad. Med. Stetin. 1996, 42 (Suppl. S31), 1–99. [Google Scholar]
- Chlubek, D.; Zawierta, J.; Kaźmierczyk, A.; Kramek, J.; Olszewska, M.; Stachowska, E. Effect of different fluoride ion concentrations on malondialdehyde (MDA) formation in the mitochondrial fraction of human placental cells. Bromat. Chem. Toksykol. 1999, 32, 119–122. [Google Scholar]
- Kot, K.; Ciosek, Ż.; Łanocha-Arendarczyk, N.; Kosik-Bogacka, D.; Ziętek, P.; Karaczun, G.; Baranowska-Bosiacka, I.; Gutowska, I.; Kalisińska, E.; Chlubek, D. Fluoride concentrations in cartilage, spongy bone, anterior cruciate ligament, meniscus, and infrapatellar fat pad of patients undergoing primary knee joint arthroplasty. Fluoride 2017, 50, 175–181. [Google Scholar]
- Giachini, M.; Pierleoni, F. Fluoride toxicity. Minerva Stomatol. 2004, 53, 171–177. [Google Scholar] [PubMed]
- Fordyce, F.M.; Vrana, K.; Zhovinsky, E.; Povoroznuk, V.; Toth, G.; Hope, B.C.; Iljinsky, U.; Baker, J. A health risk assessment for fluoride in Central Europe. Environ. Geochem. Health 2007, 29, 83–102. [Google Scholar] [CrossRef]
- Kosik-Bogacka, D.; Łanocha-Arendarczyk, N.; Kot, K.; Ziętek, P.; Karaczun, M.; Gutowska, I.; Baranowska-Bosiacka, I.; Grzeszczak, K.; Sikora, M.; Chlubek, D. Fluoride concentration in synovial fluid, bone marrow, and cartilage in patients with osteoarthritis. Fluoride 2018, 51, 164–170. [Google Scholar]
- Dołęgowska, B.; Machoy, Z.; Chlubek, D. Changes in the content of zinc and fluoride during growth of the femur in chicken. Biol. Trace Elem. Res. 2003, 91, 67–76. [Google Scholar] [CrossRef]
- Jamal, A.; Moshfeghi, M.; Moshfeghi, S.; Mohammadi, N.; Zarean, E.; Jahangiri, N. Is preterm placental calcification related to adverse maternal and foetal outcome? J. Obstet. Gynecol. 2017, 37, 605–609. [Google Scholar] [CrossRef]
- Moran, M.; Higgins, M.; Zombori, G.; Ryan, J.; McAuliffe, F.M. Computerized assessment of placental calcification post-ultrasound: A novel software tool. Ultrasound Obstet. Gynecol. 2013, 41, 545–549. [Google Scholar] [CrossRef]
- Wang, W.; Kong, L.; Zhao, H.; Dong, R.; Li, J.; Jia, Z.; Ji, N.; Deng, S.; Sun, Z.; Zhou, J. Thoracic ossification of ligamentum flavum caused by skeletal fluorosis. Eur. Spine J. 2007, 16, 1119–1128. [Google Scholar] [CrossRef]
- Chlubek, D.; Mokrzyński, S.; Machoy, Z.; Samujło, D.; Węgrzynowski, J. Fluoride concentration in mother and fetus. I. Placental transport of fluorides. Ginekol. Pol. 1994, 65, 611–615. [Google Scholar]
- Chlubek, D.; Mokrzyński, S.; Machoy, Z.; Olszewska, M. Fluorides in the body of mother and in the fetus. III. Fluorides in amniotic fluid. Ginekol Pol. 1995, 66, 614–617. [Google Scholar]
- Luke, J. Fluoride deposition in the aged human pineal gland. Caries Res. 2001, 35, 125–128. [Google Scholar] [CrossRef]
- Tharnpanich, T.; Johns, J.; Subongkot, S.; Johns, N.P.; Kitkhuandee, A.; Toomsan, Y.; Luengpailin, S. Association between high pineal fluoride content and pineal calcification in a low fluoride area. Fluoride 2016, 49, 472–484. [Google Scholar]
- Luke, J. The Effect of Fluoride on the Physiology of the Pineal Gland. Ph.D. Thesis, University of Surrey, Guildford, UK, 1997. [Google Scholar]
- National Research Council. Fluoride in Drinking Water: A Scientific Review of EPAs Standards; Advisors of the Nation on Science Engineering and Medicine; Committee on Fluoride in Drinking Water; Board on Environmental Studies and Toxicology; The National Academies: Washington, DC, USA, 2006; pp. 262–263. [Google Scholar]
- Del Rio-Hortega, P. Cytology and cellular pathology of the nervous system. In Pineal Gland; Penfield, W., Ed.; Hoeber: New York, NY, USA, 1932; pp. 637–703. [Google Scholar]
- Golan, J.; Torres, K.; Staśkiewicz, G.J.; Opielak, G.; Maciejewski, R. Morphometric parameters of the human pineal gland in relation to age, body weight and height. Folia Morphol. 2002, 61, 111–113. [Google Scholar]
- Macchi, M.M.; Bruce, J.N. Human pineal physiology and functional significance of melatonin. Front. Neuroendocrinol. 2004, 25, 177–195. [Google Scholar] [CrossRef]
- Arendt, J. Melatonin and the Mammalian Pineal Gland, 1st ed.; Chapman & Hall: London, UK, 1995; p. 17. [Google Scholar]
- Tan, D.-X.; Manchester, L.C.; Fuentes-Broto, L.; Paredes, S.D.; Reiter, R.J. Significance and application of melatonin in the regulation of brown adipose tissue metabolism. Relation to human obesity. Obes. Rev. 2011, 12, 167–188. [Google Scholar] [CrossRef]
- Kunz, D.; Schmitz, S.; Mahlberg, R.; Mohr, A.; Stöter, C.; Wolf, K.J.; Herrmann, W.M. A new concept for melatonin deficit: On pineal calcification and melatonin excretion. Neuropsychopharmacology 1999, 21, 765–772. [Google Scholar] [CrossRef]
- Mahlberg, R.; Kienast, T.; Hödel, S.; Heidenreich, J.O.; Schmitz, S.; Kunz, D. Degree of pineal calcification (DOC) is associated with polysomnographic sleep measures in primary insomnia patients. Sleep Med. 2009, 10, 439–445. [Google Scholar] [CrossRef]
- Patel, S.; Rahmani, B.; Gandhi, J.; Seyam, O.; Joshi, G.; Reid, I.; Smith, N.L.; Waltzer, W.C.; Khan, S.A. Revisiting the pineal gland: A review of calcification, masses, precocious puberty, and melatonin functions. Int. J. Neurosci. 2020, 25, 1–12. [Google Scholar] [CrossRef]
- Tan, D.-X.; Xu, B.; Zhou, X.; Reiter, R.J. Calcification, melatonin production, aging, associated health consequences and rejuvenation of the pineal gland. Molecules 2018, 23, 301. [Google Scholar] [CrossRef]
- Kiroğlu, Y.; Çalli, C.; Karabulut, N.; Oncel, C. Intracranial calcifications on CT. Diagn. Interv. Radiol. 2010, 16, 263–269. [Google Scholar]
- McKinney, A.M. Atlas of Normal Imaging Variations of the Brain, Skull, and Craniocervical Vasculature; Springer International Publishing: Cham, Switzerland, 2017. [Google Scholar]
- Demmel, U.; Höck, A.; Kasperek, K.; Feinendegen, L.E. Trace element concentration in the human pineal body. Activation analysis of cobalt, iron, rubidium, selenium, zinc, antimony and cesium. Sci. Total Environ. 1982, 24, 135–146. [Google Scholar] [CrossRef]
- Reiter, R.J.; Mayo, J.C.; Tan, D.-X.; Sainz, R.M.; Alatorre-Jimenez, M.; Qin, I. Melatonin as an antioxidant: Under promises but over delivers. J. Pineal Res. 2016, 61, 253–278. [Google Scholar] [CrossRef]
- Reiter, R.J. Oxidative damage in the central nervous system: Protection by melatonin. Prog. Neurobiol. 1998, 56, 359–384. [Google Scholar] [CrossRef]
- Pandi-Perumal, S.R.; BaHammam, A.S.; Brown, G.M.; Spence, D.W.; Bharti, W.K.; Kaur, C.; Hardeland, R.; Cardinali, D.P. Melatonin antioxidative defense: Therapeutical implications for aging and neurodegenerative processes. Neurotox. Res. 2013, 23, 267–300. [Google Scholar] [CrossRef]
- Liu, Z.; Gan, L.; Xu, Y.; Luo, D.; Ren, Q.; Wu, S.; Sun, C. Melatonin alleviates inflammasome-induced pyroptosis through inhibiting NF-κB/GSDMD signal in mice adipose tissue. J. Pineal Res. 2017, 63, e12414. [Google Scholar] [CrossRef]
- Hardeland, R.; Cardinali, D.P.; Brown, G.M.; Pandi-Perumal, S.R. Melatonin and brain inflammaging. Prog. Neurobiol. 2015, 127–128, 46–63. [Google Scholar] [CrossRef]
- Slominski, A.T.; Zmijewski, M.A.; Semak, I.; Kim, T.K.; Janjetovic, Z.; Slominski, R.M.; Zmijewski, J.W. Melatonin, mitochondria, and the skin. Cell. Mol. Life Sci. 2017, 74, 3913–3925. [Google Scholar] [CrossRef]
- Reiter, R.J.; Rosales-Corral, S.; Boga, J.A.; Tan, D.-X.; Davis, J.M.; Konturek, P.C.; Konturek, S.J.; Brzozowski, T. The photoperiod, circadian regulation and chronodisruption: The requisite interplay between the suprachiasmatic nuclei and the pineal and gut melatonin. J. Physiol. Pharmacol. 2011, 62, 269–274. [Google Scholar]
- Conti, A.; Conconi, S.; Hertens, E.; Skwarlo-Sonta, K.; Markowska, M.; Maestroni, J.M. Evidence for melatonin synthesis in mouse and human bone marrow cells. J. Pineal Res. 2000, 28, 193–202. [Google Scholar] [CrossRef]
- Tijmes, M.; Pedraza, R.; Valladares, I. Melatonin in the rat testis: Evidence for local synthesis. Steroids 1996, 61, 65–68. [Google Scholar] [CrossRef]
- Itoh, M.T.; Ishizuka, B.; Kudo, Y.; Fusama, S.; Amemiya, A.; Sumi, Y. Detection of melatonin and serotonin N-acetyltransferase and hydroxyindole-O-methyltransferase activities in rat ovary. Mol. Cell. Endocrinol. 1997, 136, 7–13. [Google Scholar] [CrossRef]
- Soliman, A.; Lacasse, A.-A.; Lanoix, D.; Sagrillo-Fagundes, L.; Boulard, V.; Vaillancourt, C. Placental melatonin system is present throughout pregnancy and regulates villous trophoblast differentiation. J. Pineal Res. 2015, 59, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Mrvelj, A.; Womble, M.D. Fluoride-free diet stimulates pineal growth in aged male rats. Biol. Trace Elem. Res. 2020. (In press) [CrossRef]
- Ibañez Rodriguez, M.P.; Noctor, S.C.; Muñoz, E.M. Cellular basis of pineal gland development: Emerging role of microglia as phenotype regulator. PLoS ONE 2016, 11, e0167063. [Google Scholar] [CrossRef]
- Murcia Garcia, J.; Muñoz Hovos, A.; Molina Carballo, A.; Fernández Garcia, J.M.; Narbona López, E.; Uberos Fernández, J. Puberty and melatonin. An. Esp. Pediatr. 2002, 57, 121–126. [Google Scholar] [CrossRef]
- Lewy, A.J.; Cutler, N.L.; Sack, R.L. The endogenous melatonin profile as a marker for circadian phase position. J. Biol. Rhythms 1999, 14, 227–236. [Google Scholar] [CrossRef]
- Selmaoui, B.; Touitou, Y. Reproducibility of the circadian rhythms of serum cortisol and melatonin in healthy subjects: A study of three different 24-h cycles over six weeks. Life Sci. 2003, 73, 3339–3349. [Google Scholar] [CrossRef]
- Van Someren, E.; Nagtegaal, E. Improving melatonin circadian phase estimates. Sleep Med. 2007, 8, 590–601. [Google Scholar] [CrossRef]
- Bocchi, G.V.G. Physical, chemical, and mineralogical characterization of carbonatehydroxyapatite concretions of the human pineal gland. J. Inorg. Biochem. 1993, 49, 209–220. [Google Scholar] [CrossRef]
- Mabie, C.P.; Wallace, M.M. Optical, physical and chemical properties of pineal gland calcifications. Calcif. Tissue Res. 1974, 16, 59–71. [Google Scholar] [CrossRef] [PubMed]
- Baconnier, S.; Lang, S.B.; Polomska, M.; Hilczer, B.; Berkovic, G.; Meshulam, G. Calcite microcrystals in the pineal gland of the human brain: First physical and chemical studies. Bioelectromagnetics 2002, 23, 488–495. [Google Scholar] [CrossRef] [PubMed]
- Doyle, A.J.; Anderson, G.D. Physiologic calcification of the pineal gland in children on computed tomography: Prevalence, observer reliability and association with choroid plexus calcification. Acad. Radiol. 2006, 13, 822–826. [Google Scholar] [CrossRef] [PubMed]
- Winkler, P.; Helmke, K. Age-related incidence of pineal gland calcification in children: A roentgenological study of 1044 skull films and a review of the literature. J. Pineal Res. 1987, 4, 247–252. [Google Scholar] [CrossRef]
- Maślińska, D.; Laure-Kamionowska, M.; Deręgowski, K.; Maśliński, S. Association of mast cells with calcification in the human pineal gland. Folia Neuropathol. 2010, 48, 276–282. [Google Scholar]
- Kay, S.R.; Sandyk, R. Experimental models of schizophrenia. Int. J. Neurosci. 1991, 58, 69–82. [Google Scholar] [CrossRef]
- Sandyk, R.; Kay, S.R. Abnormal EEG and calcification of the pineal gland in schizophrenia. Int. J. Neurosci. 1992, 62, 107–111. [Google Scholar] [CrossRef]
- Sandyk, R.; Pardeshi, R. The relationship between ECT nonresponsiveness and calcification of the pineal gland in bipolar patients. Int. J. Neurosci. 1990, 54, 301–306. [Google Scholar] [CrossRef]
- Friedland, R.P.; Luxenberg, J.S.; Koss, E.A. A quantitative study of intracranial calcification in dementia of the Alzheimer type. Int. Psychogeriatr. 1990, 2, 36–43. [Google Scholar] [CrossRef]
- Mahlberg, L.; Walther, S.; Kalus, P.; Bohner, G.; Haedel, S.; Reischies, F.M.; Kuhl, K.P.; Hellweg, R.; Kunz, D. Pineal calcification in Alzheimer’s disease: An in vivo study using computed tomography. Neurobiol. Aging 2008, 29, 203–209. [Google Scholar] [CrossRef]
- Tuntapakul, S.; Kitkhuandee, A.; Kanpittaya, J.; Johns, J.; Johns, N.P. Pineal calcification is associated with pediatric primary brain tumor. Asia Pac. J. Clin. Oncol. 2016, 12, e405–e410. [Google Scholar] [CrossRef] [PubMed]
- Kitkhuandee, A.; Sawanyawisuth, K.; Johns, N.P.; Kanpittaya, J.; Johns, J. Pineal calcification is associated with symptomatic cerebral infarction. J. Stroke Cerebrovasc. Dis. 2014, 23, 249–253. [Google Scholar] [CrossRef] [PubMed]
- Ozlece, H.K.; Akyuz, O.; Huseyinoglu, N.; Aydin, S.; Can, S.; Serim, V.A. Is there a correlation between the pineal gland calcification and migraine? Eur. Rev. Med. Pharmacol. Sci. 2015, 19, 3861–3864. [Google Scholar] [PubMed]
- Kunz, D.; Bes, F.; Schlattmann, P.; Herrmann, W.M. On pineal calcification and its relation to subjective sleep perception: A hypothesis-driven pilot study. Psychiatry Res. 1998, 82, 187–191. [Google Scholar] [CrossRef]
- Mori, R.; Kodaka, T.; Sano, T. Preliminary report on the correlation among pineal concretions, prostatic calculi and age in human adult males. Anat. Sci. Int. 2003, 78, 181–184. [Google Scholar] [CrossRef]
- Liebrich, L.S.; Schredl, M.; Findeisen, P.; Groden, C.; Bumb, J.M.; Nölte, I.S. Morphology and function: MR pineal volume and melatonin level in human saliva are correlated. J. Magn. Reson. Imaging 2014, 40, 966–971. [Google Scholar] [CrossRef]
- Tan, D.-X.; Manchester, L.C.; Reiter, R.J. CSF generation by pineal gland results in a robust melatonin circadian rhythm in the third ventricle as a unique light/dark signal. Med. Hypotheses 2016, 86, 3–9. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.-X.; Kim, S.J.; Cruz, M.H.C. Delivery of pineal melatonin to the brain and SCN: Role of canaliculi, cerebrospinal fluid, tanycytes and Virchow-Robin perivascular spaces. Brain Struct. Funct. 2014, 219, 1873–1887. [Google Scholar] [CrossRef]
- Zhou, J.-N.; Liu, R.-Y.; Kamphorst, W.; Hofman, M.A.; Swaab, D.F. Early neuropathological Alzheimer’s changes in aged individuals are accompanied by decreased cerebrospinal fluid melatonin levels. J. Pineal Res. 2003, 35, 125–130. [Google Scholar] [CrossRef]
- Michotte, Y.; Lowenthal, A.; Knaepen, L.; Colland, M.; Massart, D.L. A morphological and chemical study of calcification of the pineal gland. J. Neurol. 1977, 215, 209–219. [Google Scholar] [CrossRef]
- Bharti, V.K.; Srivastava, R.S. Effect of pineal proteins at different dose level on fluoride-induced changes in plasma biochemicals and blood antioxidants enzymes in rats. Biol. Trace Elem. Res. 2011, 141, 275–282. [Google Scholar] [CrossRef]
- Bharti, V.K.; Srivastava, R.S. Fluoride-induced oxidative stress in rat’s brain and its amelioration by buffalo (Bubalus bubalis) pineal proteins and melatonin. Biol. Trace Elem. Res. 2009, 130, 131–140. [Google Scholar] [CrossRef]
- Chlubek, D. Fluoride and oxidative stress. Fluoride 2003, 36, 217–228. [Google Scholar]
- Rzeuski, R.; Chlubek, D.; Machoy, Z. Interactions between fluoride and biological free radical reactions. Fluoride 1998, 31, 43–45. [Google Scholar]
- Chlubek, D.; Machoy, Z. Significance of the effect of fluorine dose on enzymes activity in in vivo and in vitro studies. Bromat. Chem. Toksykol. 1989, 22, 235–245. [Google Scholar]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chlubek, D.; Sikora, M. Fluoride and Pineal Gland. Appl. Sci. 2020, 10, 2885. https://doi.org/10.3390/app10082885
Chlubek D, Sikora M. Fluoride and Pineal Gland. Applied Sciences. 2020; 10(8):2885. https://doi.org/10.3390/app10082885
Chicago/Turabian StyleChlubek, Dariusz, and Maciej Sikora. 2020. "Fluoride and Pineal Gland" Applied Sciences 10, no. 8: 2885. https://doi.org/10.3390/app10082885
APA StyleChlubek, D., & Sikora, M. (2020). Fluoride and Pineal Gland. Applied Sciences, 10(8), 2885. https://doi.org/10.3390/app10082885





