Doxorubicin Assisted by Microsecond Electroporation Promotes Irreparable Morphological Alternations in Sensitive and Resistant Human Breast Adenocarcinoma Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture
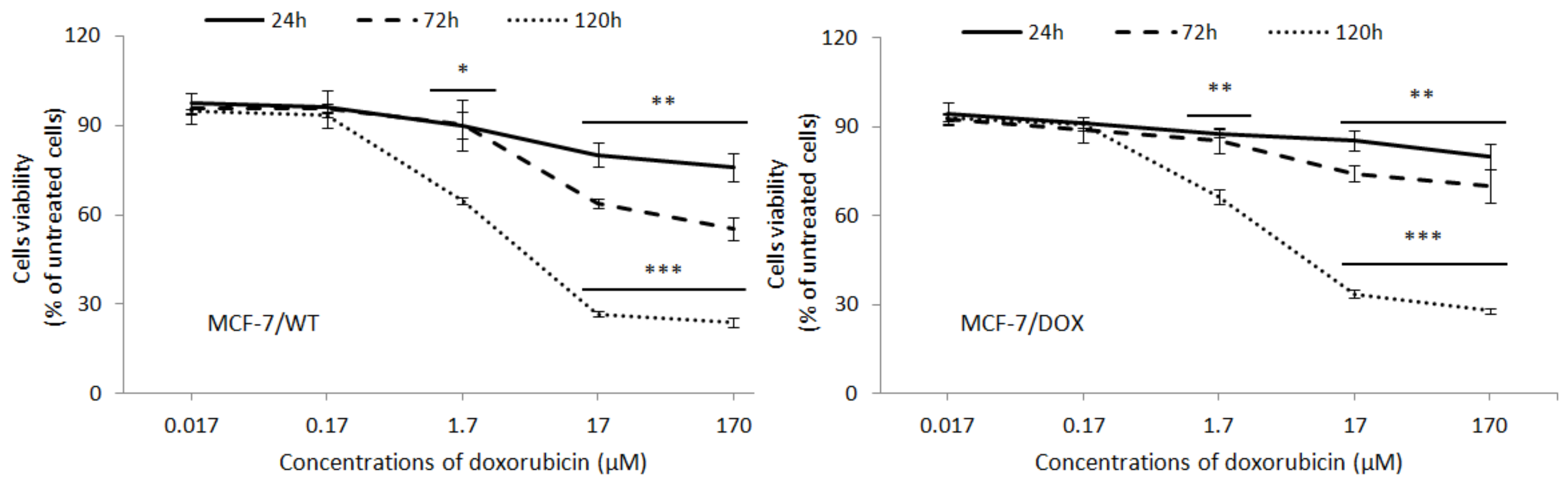
2.2. Chemosensitivity Tests
2.3. Electropermeabilization Protocol
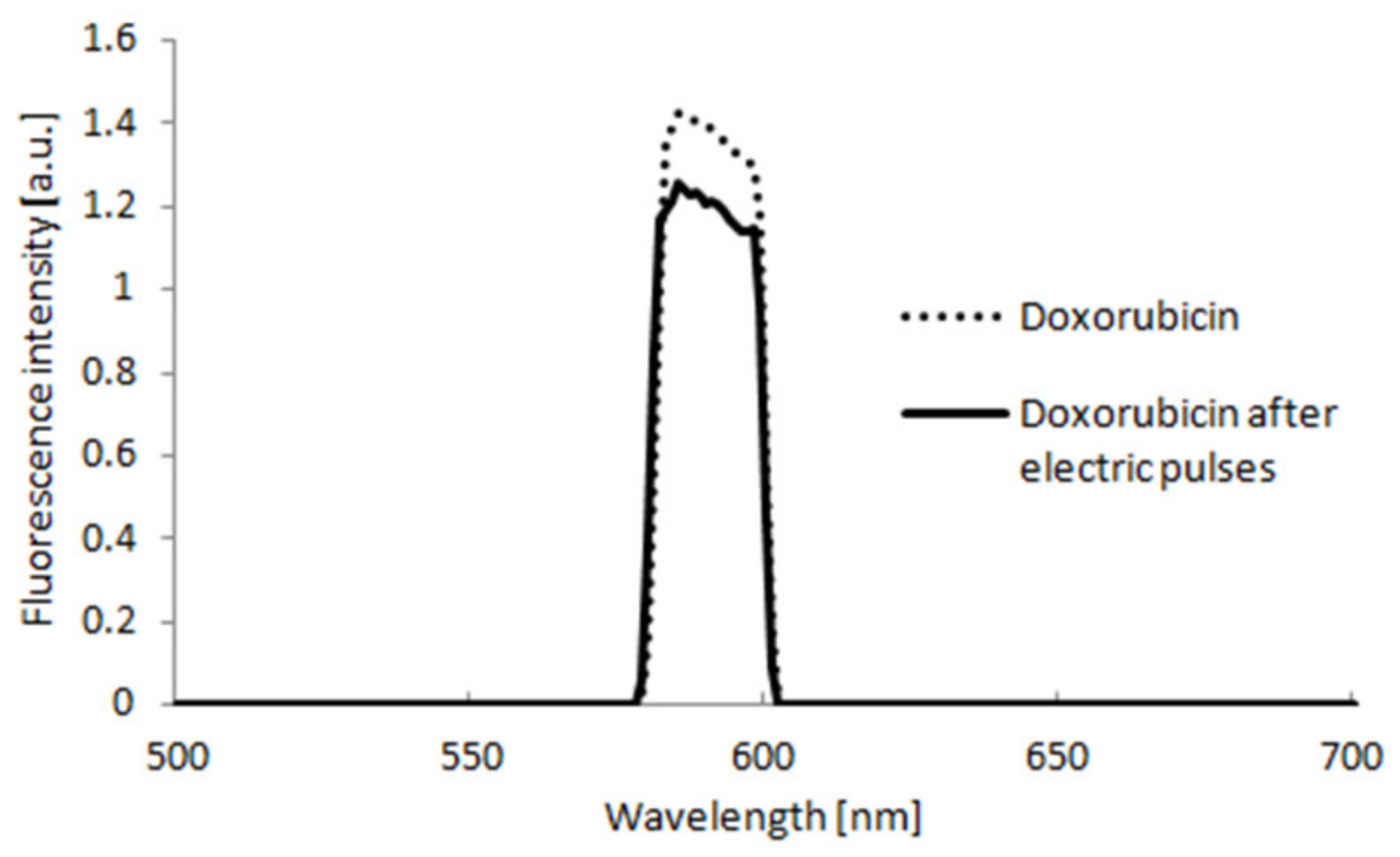
2.4. Doxorubicin Fluorescence Spectra
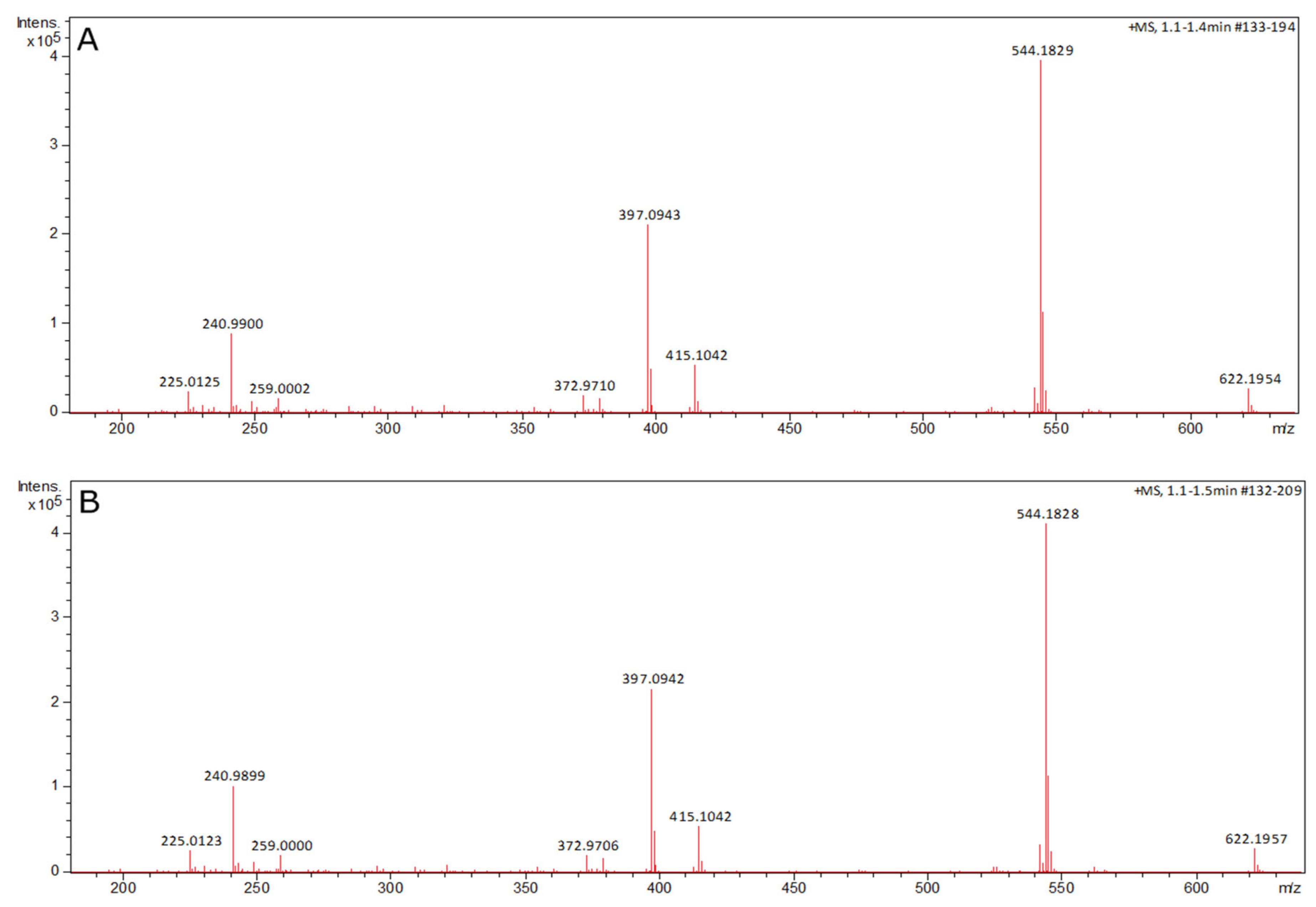
2.5. Doxorubicin Mass Spectroscopy Analysis
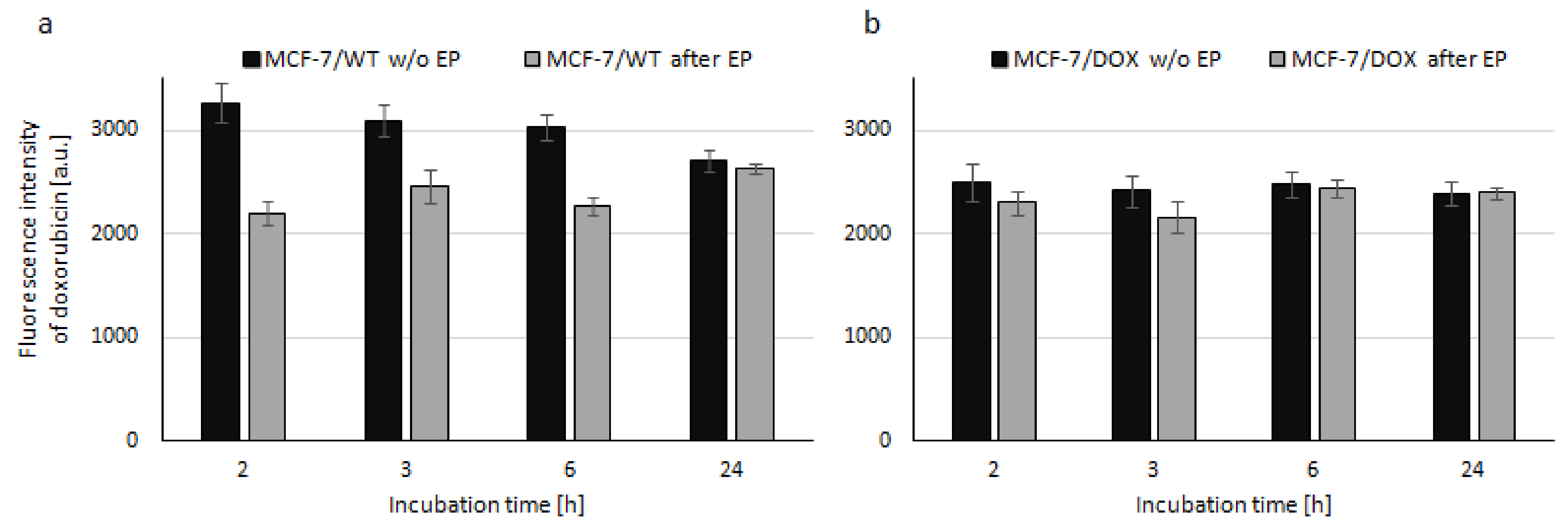
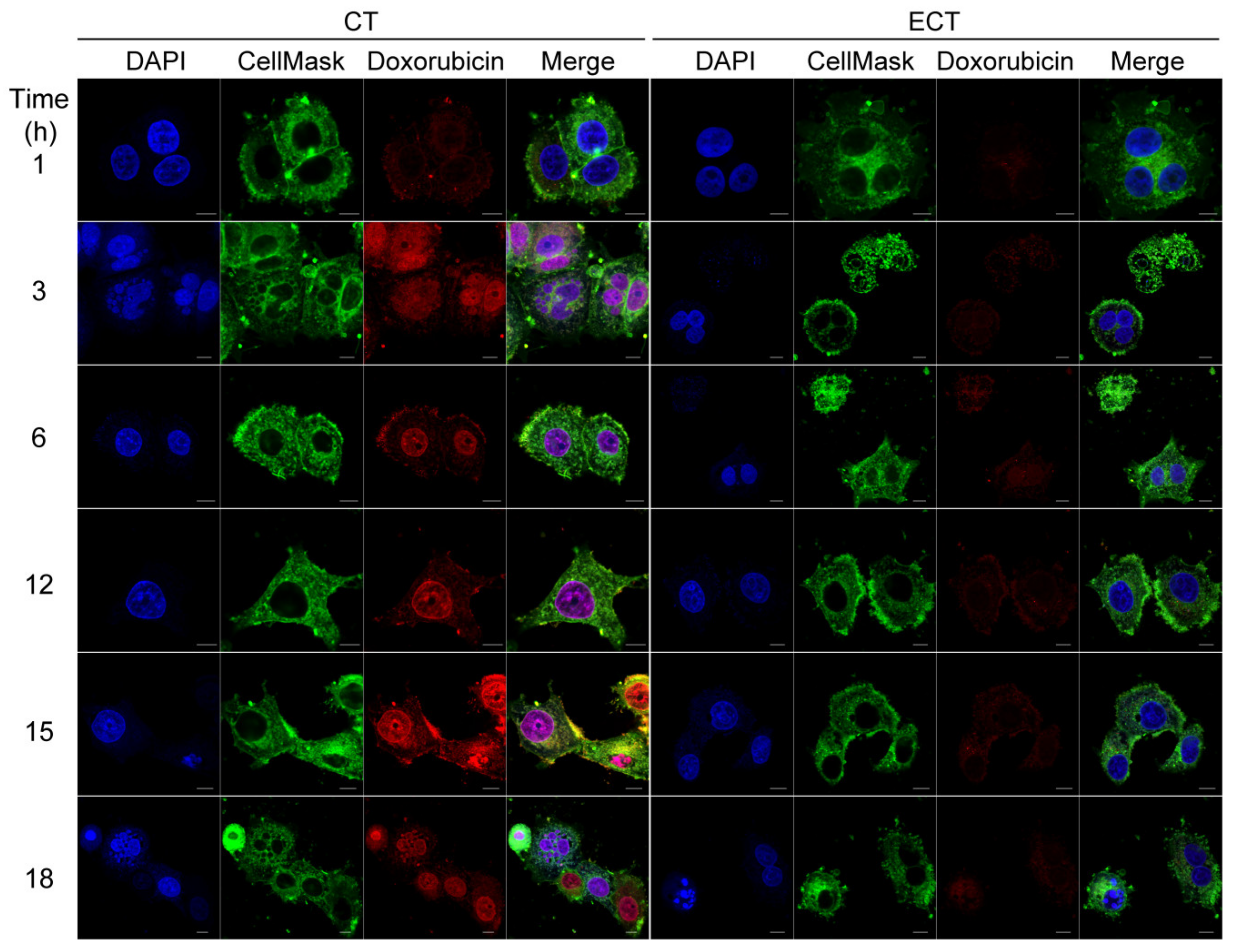
2.6. Intracellular Localization of Doxorubicin
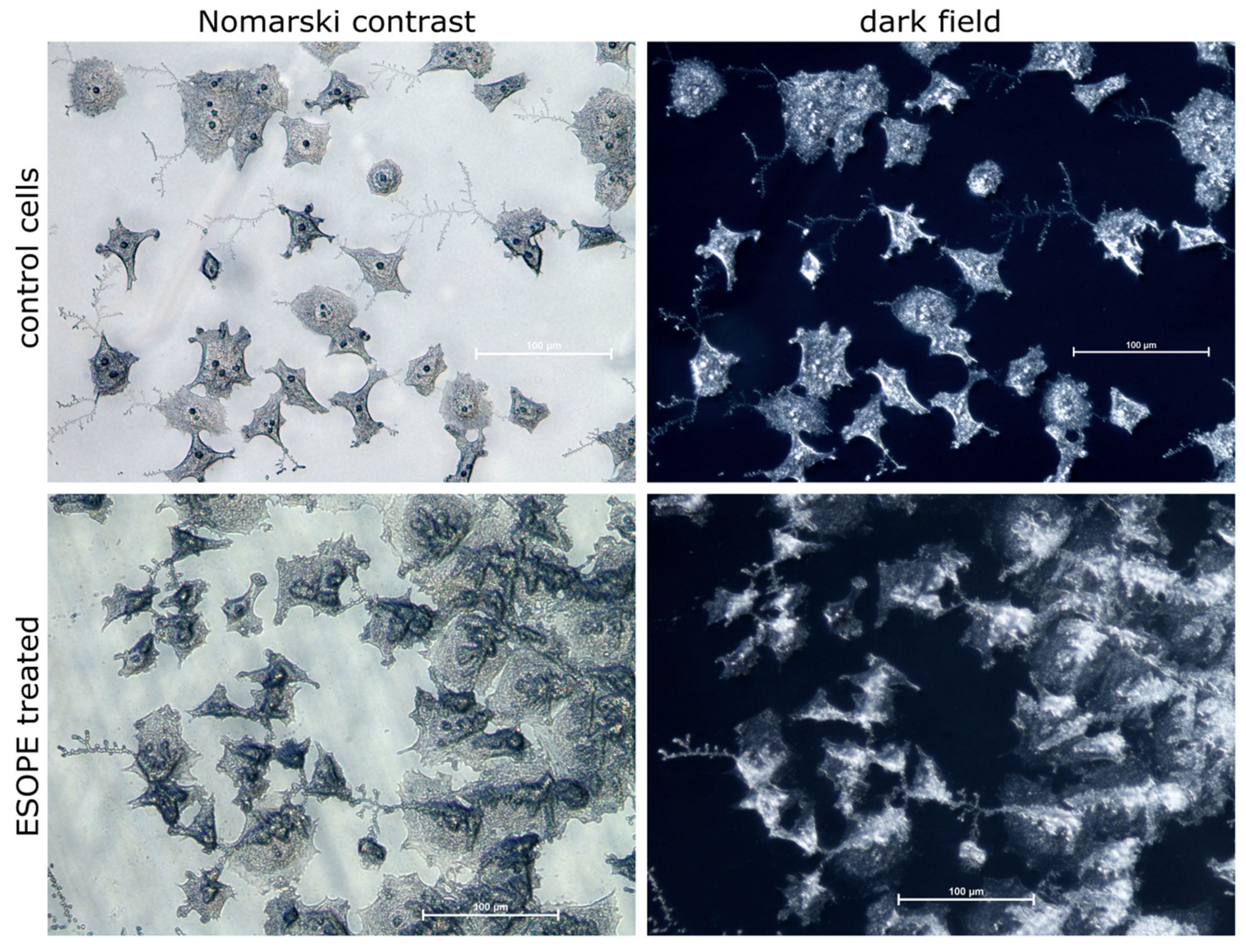
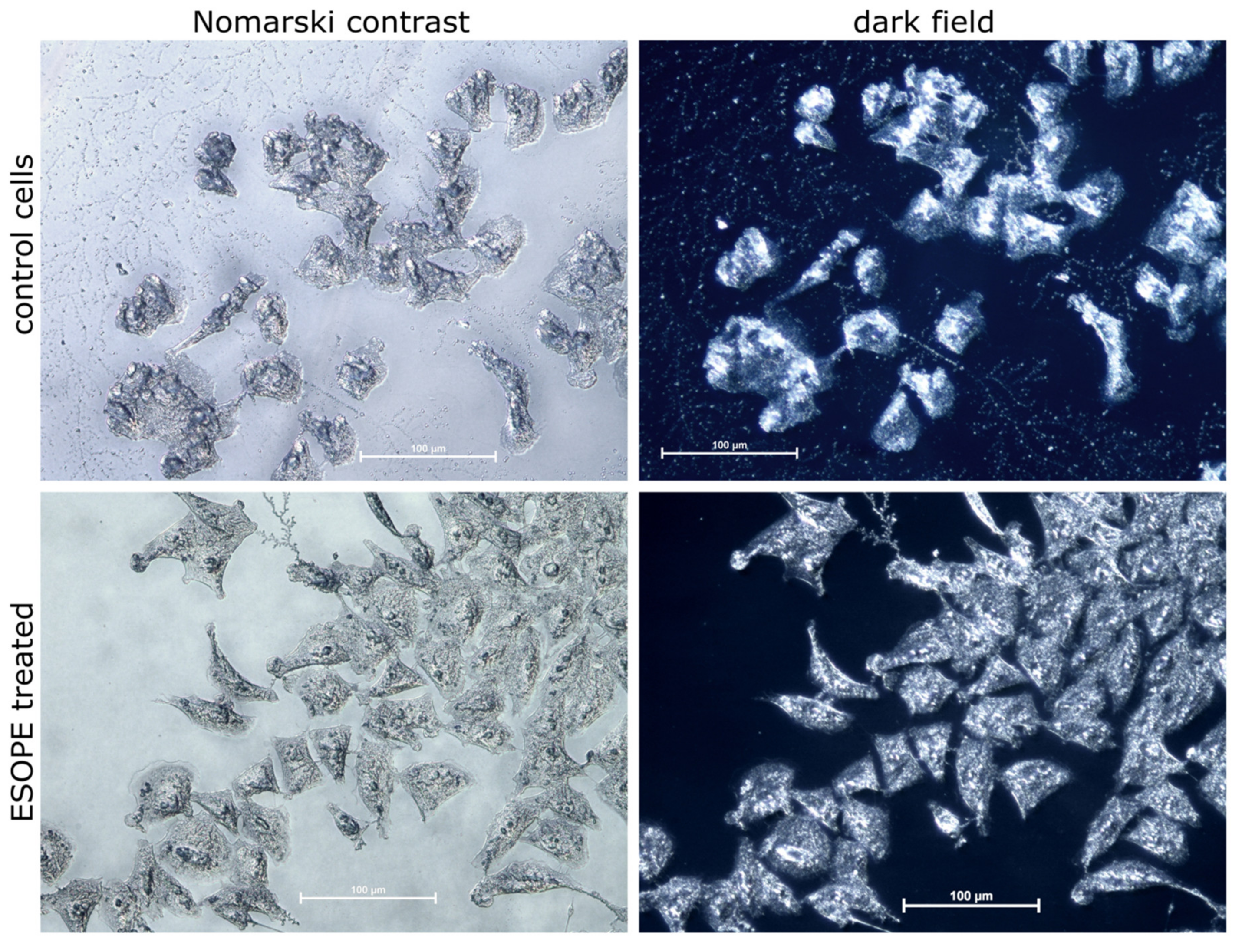
2.7. Metallurgical Microscope
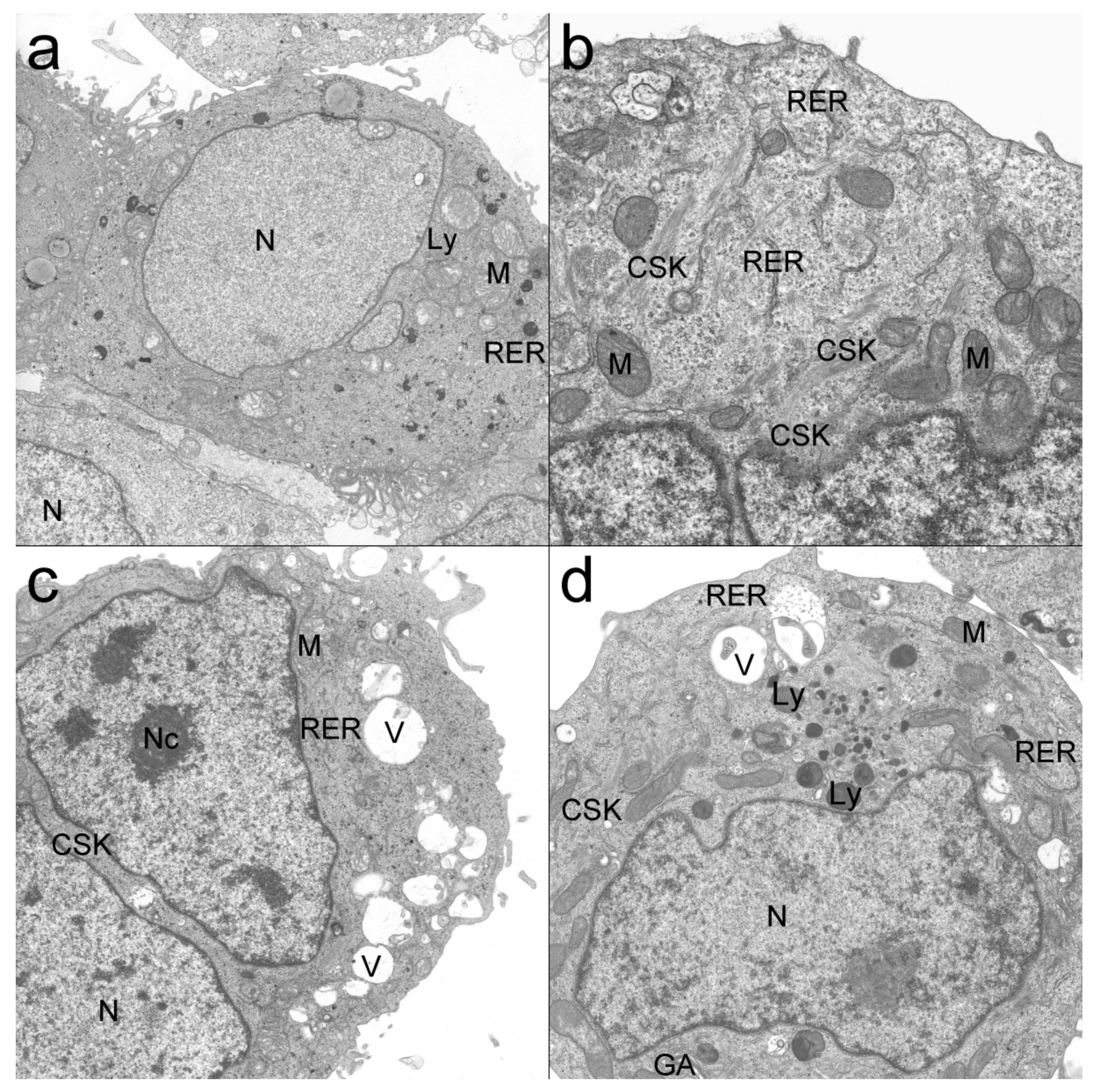
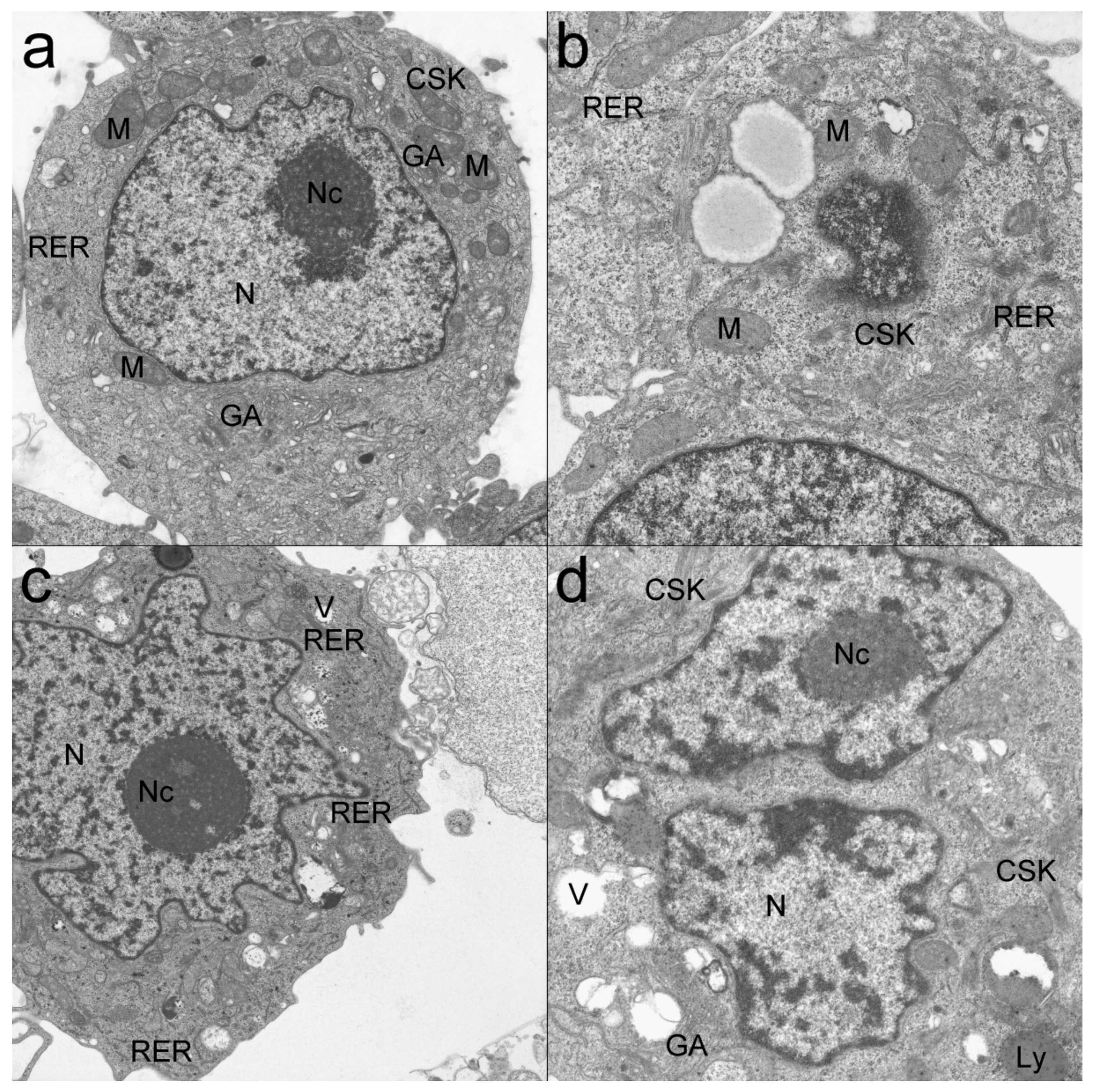
2.8. Transmission Electron Microscope (TEM)
2.9. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gothelf, A.; Mir, L.M.; Gehl, J. Electrochemotherapy: Results of cancer treatment using enhanced delivery of bleomycin by electroporation. Cancer Treat. Rev. 2003, 5, 371–387. [Google Scholar] [CrossRef]
- Loi, S.; Sirtaine, N.; Piette, F.; Salgado, R.; Viale, G.; Van Eenoo, F.; Rouas, G.; Francis, P.; Crown, J.P.A.; Hitre, E.; et al. Prognostic and predictive value of tumor-infiltrating lymphocytes in a phase III randomized adjuvant breast cancer trial in node-positive breast cancer comparing the addition of docetaxel to doxorubicin with doxorubicin-based chemotherapy: BIG 02-98. J. Clin. Oncol. 2013. [Google Scholar] [CrossRef] [PubMed]
- Adams, S.; Gray, R.J.; Demaria, S.; Goldstein, L.; Perez, E.A.; Shulman, L.N.; Martino, S.; Wang, M.; Jones, V.E.; Saphner, T.J.; et al. Prognostic value of tumor-infiltrating lymphocytes in triple-negative breast cancers from two phase III randomized adjuvant breast cancer trials: ECOG 2197 and ECOG 1199. J. Clin. Oncol. 2014, 32, 2959–2966. [Google Scholar] [CrossRef]
- Mir, L.M.; Orlowski, S. Mechanisms of electrochemotherapy. Adv. Drug Deliv. Rev. 1999, 35, 107–118. [Google Scholar] [CrossRef]
- Rols, M.P. Electropermeabilization, a physical method for the delivery of therapeutic molecules into cells. Biochim. Biophys. Acta Biomembr. 2006, 1758, 423–428. [Google Scholar] [CrossRef] [PubMed]
- Yarmush, M.L.; Golberg, A.; Serša, G.; Kotnik, T.; Miklavčič, D. Electroporation-based technologies for medicine: Principles, Applications, and challenges. Annu. Rev. Biomed. Eng. 2014, 16, 295–320. [Google Scholar] [CrossRef]
- Jaroszeski, M.J.; Heller, R.; Gilbert, R. Electrochemotherapy, Electrogenetherapy, and Transdermal Drug Delivery: Electrically Mediated Delivery of Molecules to Cells; University of South Florida Press: Tampa, FL, USA, 2000; ISBN 0896036065. [Google Scholar]
- Gehl, J. Electroporation for drug and gene delivery in the clinic: Doctors go electric. In Electroporation Protocols; Humana Press: Totowa, NJ, USA, 2008; Volume 423, pp. 351–359. [Google Scholar]
- Campana, L.G.; Valpione, S.; Falci, C.; Mocellin, S.; Basso, M.; Corti, L.; Balestrieri, N.; Marchet, A.; Rossi, C.R. The activity and safety of electrochemotherapy in persistent chest wall recurrence from breast cancer after mastectomy: A phase-II study. Breast Cancer Res. Treat. 2012, 134, 1169–1178. [Google Scholar] [CrossRef]
- Benevento, R.; Santoriello, A.; Perna, G.; Canonico, S. Electrochemotherapy of cutaneous metastastes from breast cancer in elderly patients: A preliminary report. BMC Surg. 2012. [Google Scholar] [CrossRef]
- Davalos, R.V.; Mir, L.M.; Rubinsky, B. Tissue ablation with irreversible electroporation. Ann. Biomed. Eng. 2005. [Google Scholar] [CrossRef]
- Rubinsky, B. Irreversible electroporation in medicine. Technol. Cancer Res. Treat. 2007. [Google Scholar] [CrossRef]
- Dev, S.B.; Rabussay, D.P.; Widera, G.; Hofmann, G.A. Medical applications of electroporation. IEEE Trans. Plasma Sci. 2000, 28, 206–223. [Google Scholar] [CrossRef]
- Weżgowiec, J.; Kulbacka, J.; Saczko, J.; Rossowska, J.; Chodaczek, G.; Kotulska, M. Biological effects in photodynamic treatment combined with electropermeabilization in wild and drug resistant breast cancer cells. Bioelectrochemistry 2018, 123, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Batista Napotnik, T.; Miklavčič, D. In vitro electroporation detection methods—An overview. Bioelectrochemistry 2018, 120, 166–182. [Google Scholar] [CrossRef] [PubMed]
- Waks, A.G.; Winer, E.P. Breast Cancer Treatment: A Review. JAMA J. Am. Med. Assoc. 2019, 321, 288–300. [Google Scholar] [CrossRef]
- Sun, Y.S.; Zhao, Z.; Yang, Z.N.; Xu, F.; Lu, H.J.; Zhu, Z.Y.; Shi, W.; Jiang, J.; Yao, P.P.; Zhu, H.P. Risk factors and preventions of breast cancer. Int. J. Biol. Sci. 2017, 13, 1387–1397. [Google Scholar] [CrossRef]
- Cai, F.; Luis, M.; Lin, X.; Wang, M.; Cai, L.; Cen, C.; Biskup, E. Anthracycline-induced cardiotoxicity in the chemotherapy treatment of breast cancer: Preventive strategies and treatment (Review). Mol. Clin. Oncol. 2019, 11, 15–23. [Google Scholar] [CrossRef]
- Barretf-Lee, P.J. Growth factor signalling in clinical breast cancer and its impact on response to conventional therapies: A review of chemotherapy. Endocr.-Relat. Cancer 2005, 12, 125–133. [Google Scholar] [CrossRef]
- Canela, A.; Maman, Y.; Jung, S.; Wong, N.; Callen, E.; Day, A.; Kieffer-Kwon, K.R.; Pekowska, A.; Zhang, H.; Rao, S.S.P.; et al. Genome organization drives chromosome fragility. Cell 2017, 170, 507–521. [Google Scholar] [CrossRef]
- Henriksen, P.A. Anthracycline cardiotoxicity: An update on mechanisms, monitoring and prevention. Heart 2018, 104, 971–977. [Google Scholar] [CrossRef]
- Avril, T.; Vauléon, E.; Chevet, E. Endoplasmic reticulum stress signaling and chemotherapy resistance in solid cancers. Oncogenesis 2017, 6, 373. [Google Scholar] [CrossRef]
- Zhao, N.; Martin, C.; Woodle, M.; Mixson, A.J. Advances in delivery systems for doxorubicin. J. Nanomed. Nanotechnol. 2018, 9, 519. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, P.G.; Miranda-Silva, D.; Costa, S.M.; Barros, C.; Hamdani, N.; Moura, C.; Mendes, M.J.; Sousa-Mendes, C.; Trindade, F.; Fontoura, D.; et al. Early myocardial changes induced by doxorubicin in the nonfailing dilated ventricle. Am. J. Physiol.-Heart Circ. Physiol. 2019, 316. [Google Scholar] [CrossRef] [PubMed]
- Teissié, J.; Escoffre, J.M.; Paganin, A.; Chabot, S.; Bellard, E.; Wasungu, L.; Rols, M.P.; Golzio, M. Drug delivery by electropulsation: Recent developments in oncology. Int. J. Pharm. 2012, 423, 3–6. [Google Scholar] [CrossRef] [PubMed]
- Pehlivanova, V.N.; Tsoneva, I.H.; Tzoneva, R.D. Multiple effects of electroporation on the adhesive behaviour of breast cancer cells and fibroblasts. Cancer Cell Int. 2012, 12, 9. [Google Scholar] [CrossRef] [PubMed]
- Gibot, L.; Wasungu, L.; Teissié, J.; Rols, M.P. Antitumor drug delivery in multicellular spheroids by electropermeabilization. J. Control. Release 2013, 167, 138–147. [Google Scholar] [CrossRef]
- Elmore, S. Apoptosis: A review of programmed cell death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef]
- Choromanska, A.; Lubinska, S.; Szewczyk, A.; Saczko, J.; Kulbacka, J. Mechanisms of antimelanoma effect of oat β-glucan supported by electroporation. Bioelectrochemistry 2018, 123, 255–259. [Google Scholar] [CrossRef]
- Kulbacka, J.; Pucek, A.; Wilk, K.A.; Dubińska-Magiera, M.; Rossowska, J.; Kulbacki, M.; Kotulska, M. The effect of millisecond pulsed electric fields (msPEF) on intracellular drug transport with negatively charged large nanocarriers made of solid lipid nanoparticles (SLN): In vitro study. J. Membr. Biol. 2016, 249, 645–661. [Google Scholar] [CrossRef]
- Kulbacka, J.; Kotulska, M.; Rembiałkowska, N.; Choromańska, A.; Kamińska, I.; Garbiec, A.; Rossowska, J.; Daczewska, M.; Jachimska, B.; Saczko, J. Cellular stress induced by photodynamic reaction with CoTPPS and MnTMPyPCl5 in combination with electroporation in human colon adenocarcinoma cell lines (LoVo and LoVoDX). Cell Stress Chaperones 2013, 18, 719–731. [Google Scholar] [CrossRef]
- Wezgowiec, J.; Derylo, M.B.; Teissie, J.; Orio, J.; Rols, M.P.; Kulbacka, J.; Saczko, J.; Kotulska, M. Electric field-assisted delivery of photofrin to human breast carcinoma cells. J. Membr. Biol. 2013, 246, 725–735. [Google Scholar] [CrossRef]
- Marty, M.; Sersa, G.; Garbay, J.R.; Gehl, J.; Collins, C.G.; Snoj, M.; Billard, V.; Geertsen, P.F.; Larkin, J.O.; Miklavcic, D.; et al. Electrochemotherapy—An easy, highly effective and safe treatment of cutaneous and subcutaneous metastases: Results of ESOPE (European Standard Operating Procedures of Electrochemotherapy) study. Eur. J. Cancer Suppl. 2006, 4. [Google Scholar] [CrossRef]
- Romeo, S.; Sannino, A.; Scarfì, M.R.; Vernier, P.T.; Cadossi, R.; Gehl, J.; Zeni, O. ESOPE-Equivalent pulsing protocols for calcium electroporation: An in vitro optimization study on 2 cancer cell models. Technol. Cancer Res. Treat. 2018, 17, 1533033818788072. [Google Scholar] [CrossRef]
- Ma, W.; Wang, J.; Guo, Q.; Tu, P. Simultaneous determination of doxorubicin and curcumin in rat plasma by LC–MS/MS and its application to pharmacokinetic study. J. Pharm. Biomed. Anal. 2015, 111, 215–221. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, E.S. The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J. Cell Biol. 1963, 17, 208–212. [Google Scholar] [CrossRef] [PubMed]
- Cemazar, M.; Parkins, C.S.; Holder, A.L.; Chaplin, D.J.; Tozer, G.M.; Sersa, G. Electroporation of human microvascular endothelial cells: Evidence for an anti-vascular mechanism of electrochemotherapy. Br. J. Cancer 2001, 84, 565–570. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, B.; Teissié, J. Direct observation in the millisecond time range of fluorescent molecule asymmetrical interaction with the electropermeabilized cell membrane. Biophys. J. 1997, 75, 2630–2637. [Google Scholar] [CrossRef]
- Serša, G.; čemažar, M.; Miklavčič, D. Antitumor effectiveness of electrochemotherapy with cis–Diamminedichloroplatinum(II) in mice. Cancer Res. 1995, 55, 3450–3455. [Google Scholar]
- Dinchuk, J.E.; Kelley, K.A.; Callahan, G.N. Flow cytometric analysis of transport activity in lymphocytes electroporated with a fluorescent organic anion dye. J. Immunol. Methods 1992, 155, 257–265. [Google Scholar] [CrossRef]
- Saczko, J.; Nowak, M.; Skolucka, N.; Kulbacka, J.; Kotulska, M. The effects of the electro-photodynamic in vitro treatment on human lung adenocarcinoma cells. Bioelectrochemistry 2010, 79, 90–94. [Google Scholar] [CrossRef]
- Skolucka, N.; Daczewska, M.; Saczko, J.; Chwilkowska, A.; Choromanska, A.; Kotulska, M.; Kaminska, I.; Kulbacka, J. ETM study of electroporation influence on cell morphology in human malignant melanoma and human primary gingival fibroblast cells. Asian Pac. J. Trop. Biomed. 2011, 1, 94–98. [Google Scholar] [CrossRef]
- Maillet, A.; Tan, K.; Chai, X.; Sadananda, S.N.; Mehta, A.; Ooi, J.; Hayden, M.R.; Pouladi, M.A.; Ghosh, S.; Shim, W.; et al. Modeling doxorubicin–induced cardiotoxicity in human pluripotent stem cell derived-cardiomyocytes. Sci. Rep. 2016. [Google Scholar] [CrossRef] [PubMed]
- Novickij, V.; Zinkevičienė, A.; Perminaitė, E.; Čėsna, R.; Lastauskienė, E.; Paškevičius, A.; Švedienė, J.; Markovskaja, S.; Novickij, J.; Girkontaitė, I. Non–invasive nanosecond electroporation for biocontrol of surface infections: An in vivo study. Sci. Rep. 2018. [Google Scholar] [CrossRef] [PubMed]
- Whelan, M.C.; Larkin, J.O.; Collins, C.G.; Cashman, J.; Breathnach, O.; Soden, D.M.; O’Sullivan, G.C. Effective treatment of an extensive recurrent breast cancer which was refractory to multimodal therapy by multiple applications of electrochemotherapy. Eur. J. Cancer Suppl. 2006, 4, 32–34. [Google Scholar] [CrossRef]
- Xiao, F.; Salameh, T.; Camarillo, I.; Dykstra, C.; Lenarduzzi, N.; Reece, L.; Leary, J.F.; Natarajan, A.; Sundararajan, R. Electrically-enhanced chemodrug delivery to human breast cancer cells. In Proceedings of the Electrostatic Society of America Annual Meeting, Boston, MA, USA, 16–18 June 2009. [Google Scholar]
- Čemažar, M.; Jarm, T.; Miklavčič, D.; Lebar, A.M.; Ihan, A.; Kopitar, N.A.; Serša, G. Effect of electric-field intensity on electropermeabilization and electrosensitivity of various tumor-cell lines in vitro. Electromagn. Biol. Med. 1998, 263–272. [Google Scholar] [CrossRef]
- Shil, P.; Kumar, A.; Vidyasagar, P.B.; Mishra, K.P. Electroporation enhances radiation and doxorubicin–induced toxicity in solid tumor in vivo. J. Environ. Pathol. Toxicol. Oncol. 2006, 25, 625–632. [Google Scholar] [CrossRef]
- Beyer, U.; Rothen-Rutishauser, B.; Unger, C.; Wunderli-Allenspach, H.; Kratz, F. Differences in the intracellular distribution of acid-sensitive doxorubicin-protein conjugates in comparison to free and liposomal formulated doxorubicin as shown by confocal microscopy. Pharm. Res. 2001, 18, 29–38. [Google Scholar] [CrossRef]
- Wu, J.; Lu, Y.; Lee, A.; Pan, X.; Yang, X.; Zhao, X.; Lee, R.J. Reversal of multidrug resistance by transferrin-conjugated liposomes co-encapsulating doxorubicin and verapamil. J. Pharm. Pharm. Sci. 2007, 10, 350–357. [Google Scholar]
- Xu, D.H.; Gao, J.Q.; Liang, W.Q. Liposome-based intracellular kinetics of doxorubicin in K562/DOX cells. Pharmazie 2008, 63, 646–649. [Google Scholar]
- Ho, J.A.A.; Fan, N.C.; Fang-Ju Jou, A.; Wu, L.C.; Sun, T.P. Monitoring the subcellular localization of doxorubicin in CHO-K1 using MEKC-LIF: Liposomal carrier for enhanced drug delivery. Talanta 2012, 99, 683–688. [Google Scholar] [CrossRef]
- Zeng, X.; Morgenstern, R.; Nyström, A.M. Nanoparticle-directed sub-cellular localization of doxorubicin and the sensitization breast cancer cells by circumventing GST-mediated drug resistance. Biomaterials 2014, 35, 1227–1239. [Google Scholar] [CrossRef]
- Lou, P.J.; Lai, P.S.; Shieh, M.J.; MacRobert, A.J.; Berg, K.; Bown, S.G. Reversal of doxorubicin resistance in breast cancer cells by photochemical internalization. Int. J. Cancer 2006, 119, 2692–2698. [Google Scholar] [CrossRef] [PubMed]
- Meschini, S.; Condello, M.; Lista, P.; Vincenzi, B.; Baldi, A.; Citro, G.; Arancia, G.; Spugnini, E.P. Electroporation adopting trains of biphasic pulses enhances in vitro and in vivo the cytotoxic effect of doxorubicin on multidrug resistant colon adenocarcinoma cells (LoVo). Eur. J. Cancer 2012, 48, 2236–2243. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Xiong, Z.; Liu, Y.; Yao, C.; Li, C. Low voltage irreversible electroporation induced apoptosis in HeLa cells. J. Cancer Res. Ther. 2012, 8, 80–85. [Google Scholar] [CrossRef] [PubMed]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rembiałkowska, N.; Dubińska-Magiera, M.; Sikora, A.; Szlasa, W.; Szewczyk, A.; Czapor-Irzabek, H.; Daczewska, M.; Saczko, J.; Kulbacka, J. Doxorubicin Assisted by Microsecond Electroporation Promotes Irreparable Morphological Alternations in Sensitive and Resistant Human Breast Adenocarcinoma Cells. Appl. Sci. 2020, 10, 2765. https://doi.org/10.3390/app10082765
Rembiałkowska N, Dubińska-Magiera M, Sikora A, Szlasa W, Szewczyk A, Czapor-Irzabek H, Daczewska M, Saczko J, Kulbacka J. Doxorubicin Assisted by Microsecond Electroporation Promotes Irreparable Morphological Alternations in Sensitive and Resistant Human Breast Adenocarcinoma Cells. Applied Sciences. 2020; 10(8):2765. https://doi.org/10.3390/app10082765
Chicago/Turabian StyleRembiałkowska, Nina, Magda Dubińska-Magiera, Andrzej Sikora, Wojciech Szlasa, Anna Szewczyk, Hanna Czapor-Irzabek, Małgorzata Daczewska, Jolanta Saczko, and Julita Kulbacka. 2020. "Doxorubicin Assisted by Microsecond Electroporation Promotes Irreparable Morphological Alternations in Sensitive and Resistant Human Breast Adenocarcinoma Cells" Applied Sciences 10, no. 8: 2765. https://doi.org/10.3390/app10082765
APA StyleRembiałkowska, N., Dubińska-Magiera, M., Sikora, A., Szlasa, W., Szewczyk, A., Czapor-Irzabek, H., Daczewska, M., Saczko, J., & Kulbacka, J. (2020). Doxorubicin Assisted by Microsecond Electroporation Promotes Irreparable Morphological Alternations in Sensitive and Resistant Human Breast Adenocarcinoma Cells. Applied Sciences, 10(8), 2765. https://doi.org/10.3390/app10082765





