Uncaria tomentosa (Willd. ex Schult.) DC.: A Review on Chemical Constituents and Biological Activities
Abstract
1. Introduction
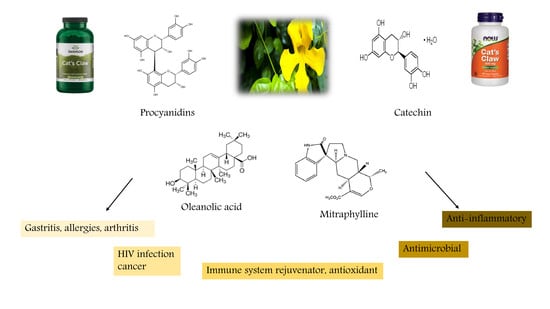
2. Chemical Constituents of U. tomentosa
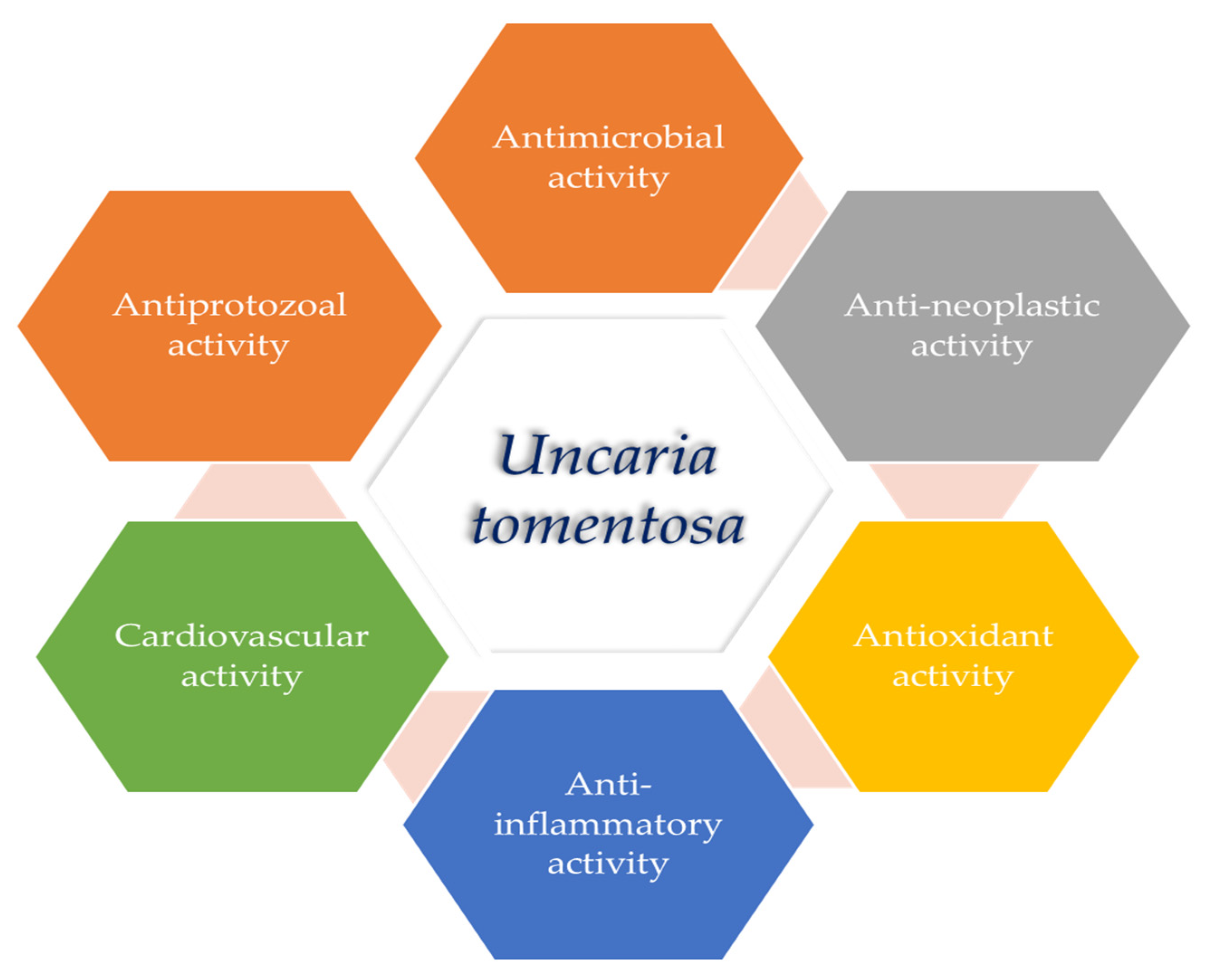
3. Biological Activities of U. tomentosa Extracts and Compounds
3.1. Traditional Uses
3.2. Antioxidant Activity
3.3. Anti-Neoplastic Activity
3.4. Anti-Inflammatory Activity
3.5. Antimicrobial, Antiprotozoal and Antiviral Activities
3.6. Immunomodulatory Activity
3.7. Cardiovascular Activity
3.8. Anti-Alzheimer’s Disease (AD) Activity
4. Reported Side Effects
5. Precautions
5.1. Drug-Drug Interactions
5.1.1. Immunosuppressant Drugs
5.1.2. Anticoagulants
5.1.3. Diuretics
5.1.4. Antihypertensive Drugs
5.1.5. Cytochrome P450 Substrates
5.2. Drug Safety
6. Recommended Doses
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| HIV | human immunodeficiency viruses |
| TOA | tetracyclic oxindole alkaloid |
| POA | pentacyclic oxindole alkaloid |
| IUPAC | International Union of Pure and Applied Chemistry |
| NSAIDs | nonsteroidal anti-inflammatory drugs |
| TBARs | thiobarbituric acid-reactive substances |
| iNOS | inducible nitric oxide synthase |
| IL-2 | interleukin-2 |
| AC11 of U. tomentosa extract | standardized aqueous extract of U. tomentosa |
| CSFs | colony-stimulating growth factors |
| RNA | ribonucleic acid |
| HSV | herpes simplex virus |
| MAPK | mitogen-activated protein kinases |
| AD | Alzheimer’s disease |
| Aβ | beta-amyloid |
| AHPA | The American Herbal Products Association |
References
- Batiha, G.E.S.; Beshbishy, A.M.; Tayebwa, D.S.; Adeyemi, O.S.; Shaheen, H.; Yokoyama, N.; Igarashi, I. The effects of trans-chalcone and chalcone 4 hydrate on the growth of Babesia and Theileria. PLoS Negl. Trop. Dis. 2019, 13, e0007030. [Google Scholar] [CrossRef] [PubMed]
- Beshbishy, A.M.; Batiha, G.E.; Yokoyama, N.; Igarashi, I. Ellagic acid microspheres restrict the growth of Babesia and Theileria in vitro and Babesia microti in vivo. Parasit. Vectors 2019, 12, 269. [Google Scholar] [CrossRef] [PubMed]
- Atanasov, A.G.; Waltenberger, B.; Pferschy-Wenzig, E.M.; Linder, T.; Wawrosch, C.; Uhrin, P.; Temml, V.; Wang, L.; Schwaiger, S.; Heiss, E.H.; et al. Discovery and resupply of pharmacologically active plant-derived natural products: A review. Biotech. Adv. 2015, 33, 1582–1614. [Google Scholar] [CrossRef] [PubMed]
- Adeyemi, O.S.; Atolani, O.; Awakan, O.J.; Olaolu, T.D.; Nwonuma, C.O.; Alejolowo, O.; Otohinoyi, D.A.; Rotimi, D.; Owolabi, A.; Batiha, G.E. Focus: Organelles: In vitro screening to identify anti-Toxoplasma compounds and in silico modeling for bioactivities and toxicity. Yale J. Biol. Med. 2019, 92, 369. [Google Scholar]
- Batiha, G.E.S.; Beshbishy, A.A.; Tayebwa, D.S.; Shaheen, M.H.; Yokoyama, N.; Igarashi, I. Inhibitory effects of Syzygium aromaticum and Camellia sinensis methanolic extracts on the growth of Babesia and Theileria parasites. Ticks Tick. Borne Dis. 2019, 10, 949–958. [Google Scholar] [CrossRef]
- Beshbishy, A.M.; Batiha, G.E.S.; Adeyemi, O.S.; Yokoyama, N.; Igarashi, I. Inhibitory effects of methanolic Olea europaea and acetonic Acacia laeta on the growth of Babesia and Theileria. Asian Pac. J. Trop. Med. 2019, 12, 425–434. [Google Scholar]
- Batiha, G.E.S.; Beshbishy, A.A.; Tayebwa, D.S.; Adeyemi, O.S.; Yokoyama, N.; Igarashi, I. Anti-piroplasmic potential of the methanolic Peganum harmala seeds and ethanolic Artemisia absinthium leaf extracts. J. Protoz. Res. 2019, 29, 8–27. [Google Scholar]
- Batiha, G.E.S.; Beshbishy, A.M.; Tayebwa, D.S.; Adeyemi, O.S.; Shaheen, H.; Yokoyama, N.; Igarashi, I. Evaluation of the inhibitory effect of ivermectin on the growth of Babesia and Theileria parasites in vitro and in vivo. Trop. Med. Health. 2019, 47, 42. [Google Scholar] [CrossRef]
- World Health Organization. WHO Monographs on Selected Medicinal Plants; World Health Organization: Geneva, Switzerland, 1999; Volume 2. [Google Scholar]
- Heitzman, M.E.; Neto, C.C.; Winiarz, E.; Vaisberg, A.J.; Hammond, G.B. Ethnobotany, phytochemistry and pharmacology of Uncaria (Rubiaceae). Phytochemistry 2005, 66, 5–29. [Google Scholar] [CrossRef]
- Santos, K.F.; Gutierres, J.M.; Pillat, M.M.; Rissi, V.B.; dos Santos Araújo, M.D.C.; Bertol, G.; Gonçalves, P.B.D.; Schetinger, M.R.C.; Morsch, V.M. Uncaria tomentosa extract alters the catabolism of adenine nucleotides and expression of ecto 5′-nucleotidase/CD73 and P2X7 and A1 receptors in the MDA-MB-231 cell line. J. Ethnopharmacol. 2016, 194, 108–116. [Google Scholar] [CrossRef]
- Navarro Hoyos, M.; Sánchez-Patán, F.; Murillo Masis, R.; Martín-Álvarez, P.J.; Zamora Ramirez, W.; Monagas, M.J.; Bartolomé, B. Phenolic assessment of Uncaria tomentosa L. (cat’s claw): Leaves, stem, bark and wood extracts. Molecules 2015, 20, 22703–22717. [Google Scholar] [CrossRef] [PubMed]
- Laus, G.; Brössner, D.; Kiplinger, K. Alkaloids of Peruvian Uncaria tomentosa. Phytochemistry 1997, 45, 855–860. [Google Scholar] [CrossRef]
- Peñaloza, E.M.C.; Kaiser, S.; Resende, P.E.d.; Pittol, V.; Carvalho, Â.R.; Ortega, G.G. Chemical composition variability in the Uncaria tomentosa (cat’s claw) wild population. Química Nova 2015, 38, 378–386. [Google Scholar]
- Gable, R.S. Risk assessment of ritual use of oral dimethyltryptamine (DMT) and harmala alkaloids. Addiction 2007, 102, 24–34. [Google Scholar] [CrossRef]
- Sheng, Y.; Akesson, C.; Holmgren, K.; Bryngelsson, C.; Giamapa, V.; Pero, R.W. An active ingredient of cat’s claw water extracts identification and efficacy of quinic acid. J. Ethnopharmacol. 2005, 96, 577–584. [Google Scholar] [CrossRef]
- Lee, J.S.; Kim, J.; Kim, B.Y.; Lee, H.S.; Ahn, J.S.; Chang, Y.S. Inhibition of phospholipase cgamma1 and cancer cell proliferation by triterpene esters from Uncaria rhynchophylla. J. Nat. Prod. 2000, 63, 753–756. [Google Scholar] [CrossRef]
- Sheng, Y.; Bryngelsson, C.; Pero, R.W. Enhanced DNA repair, immune function and reduced toxicity of C-MED-100, a novel aqueous extract from Uncaria tomentosa. J. Ethnopharmacol. 2000, 69, 115–126. [Google Scholar] [CrossRef]
- Dreifuss, A.A.; Bastos-Pereira, A.L.; Fabossi, I.A.; Lívero, F.A.; Stolf, A.M.; Alves de Souza, C.E.; Gomes Lde, O.; Constantin, R.P.; Furman, A.E.; Strapasson, R.L.; et al. Uncaria tomentosa exerts extensive anti-neoplastic effects against the Walker-256 tumor by modulating oxidative stress and not by alkaloid activity. PLoS ONE 2013, 8, e54618. [Google Scholar] [CrossRef]
- Laus, G. Advances in chemistry and bioactivity of the genus Uncaria. Phytother. Res. 2004, 18, 259–274. [Google Scholar] [CrossRef]
- Azevedo, B.C.; Morel, L.J.F.; Carmona, F.; Cunha, T.M.; Contini, S.H.T.; Delprete, P.G.; Ramalho, F.S.; Crevelin, E.; Bertoni, B.W.; França, S.C.; et al. Aqueous extracts from Uncaria tomentosa (Willd. ex Schult.) DC. reduce bronchial hyper responsiveness and inflammation in a murine model of asthma. J. Ethnopharmacol. 2018, 218, 76–89. [Google Scholar] [CrossRef]
- Sandoval, M.; Charbonnet, R.M.; Okuhama, N.N.; Roberts, J.; Krenova, Z.; Trentacosti, A.M.; Miller, M.J. Cat’s claw inhibits TNF-α production and scavenges free radicals: Role in cytoprotection. Free Radic. Biol. Med. 2000, 29, 71–78. [Google Scholar] [CrossRef]
- Piscoya, J.; Rodriguez, Z.; Bustamante, S.A.; Okuhama, N.N.; Miller, M.J.; Sandoval, M. Efficacy and safety of freeze-dried cat’s claw in osteoarthritis of the knee: Mechanisms of action of the species Uncaria guianensis. Inflamm. Res. 2001, 50, 442–448. [Google Scholar] [CrossRef] [PubMed]
- Sandoval, M.; Okuhama, N.N.; Zhang, X.J.; Condezo, L.A.; Lao, J.; Angeles, F.M.; Musah, R.A.; Bobrowski, P.; Miller, M.J. Anti-inflammatory and antioxidant activities of cat’s claw (Uncaria tomentosa and Uncaria guianensis) are independent of their alkaloid content. Phytomedicine 2002, 9, 325–337. [Google Scholar] [CrossRef] [PubMed]
- Bors, M.; Bukowska, B.; Pilarski, R.; Gulewicz, K.; Oszmiański, J.; Michałowicz, J.; Koter-Michalak, M. Protective activity of the Uncaria tomentosa extracts on human erythrocytes in oxidative stress induced by 2,4-dichlorophenol (2,4-DCP) and catechol. Food Chem. Toxicol. 2011, 49, 2202–2211. [Google Scholar] [CrossRef]
- Navarro, M.; Arnaez, E.; Moreira, I.; Hurtado, A.; Monge, D.; Monagas, M. Polyphenolic composition and antioxidant activity of Uncaria tomentosa commercial bark products. Antioxidants 2019, 8, 339. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Hoyos, M.; Lebrón-Aguilar, R.; Quintanilla-López, J.E.; Cueva, C.; Hevia, D.; Quesada, S.; Azofeifa, G.; Moreno-Arribas, M.V.; Monagas, M.; Bartolomé, B. Proanthocyanidin characterization and bioactivity of extracts from different parts of Uncaria tomentosa L. (Cat’s Claw). Antioxidants 2017, 6, 12. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhao, J.J.; Xu, J.; Feng, F.; Qu, W. Medicinal uses, phytochemistry and pharmacology of the genus Uncaria. J. Ethnopharmacol. 2015, 173, 48–80. [Google Scholar] [CrossRef]
- Akesson, C.; Lindgren, H.; Pero, R.W.; Leanderson, T.; Ivars, F. An extract of Uncaria tomentosa inhibiting cell division and NF-kappa B activity without inducing cell death. Int. Immunopharmacol. 2003, 3, 1889–1900. [Google Scholar] [CrossRef]
- Cheng, A.C.; Jian, C.B.; Huang, Y.T.; Lai, C.S.; Hsu, P.C.; Pan, M.H. Induction of apoptosis by Uncaria tomentosa through reactive oxygen species production, cytochrome c release, and caspases activation in human leukemia cells. Food Chem. Toxicol. 2007, 45, 2206–2218. [Google Scholar] [CrossRef]
- Erowele, G.I.; Kalejaiye, A.O. Pharmacology and therapeutic uses of cat’s claw. Am. J. Health Syst. Pharm. 2009, 66, 992–995. [Google Scholar] [CrossRef]
- Rinner, B.; Li, Z.X.; Haas, H.; Siegl, V.; Sturm, S.; Stuppner, H.; Pfragner, R. Antiproliferative and pro-apoptotic effects of Uncaria tomentosa in human medullary thyroid carcinoma cells. Anticancer Res. 2009, 29, 4519–4528. [Google Scholar] [PubMed]
- Xiao, D.; Gu, Z.L.; Qian, Z.N. Effects of quercetin on platelet and reperfusion-induced arrhythmias in rats. Zhongguo Yao Li Xue Bao 1993, 14, 505–508. [Google Scholar] [PubMed]
- Xiang, L.; Li, Y.; Deng, X.; Kosanovic, D.; Schermuly, R.T.; Li, X. Natural plant products in treatment of pulmonary arterial hypertension. Pulm. Circ. 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- Allen-Hall, L.; Arnason, J.T.; Cano, P.; Lafrenie, R.M. Uncaria tomentosa acts as a potent TNF-alpha inhibitor through NF-kappaB. J. Ethnopharmacol. 2010, 127, 685–693. [Google Scholar] [CrossRef] [PubMed]
- Paniagua-Pérez, R.; Madrigal-Bujaidar, E.; Molina-Jasso, D.; Reyes-Cadena, S.; Alvarez-González, I.; Sánchez-Chapul, L.; Pérez-Gallaga, J. Antigenotoxic, antioxidant and lymphocyte induction effects produced by pteropodine. Basic Clin. Pharmacol. Toxicol. 2009, 104, 222–227. [Google Scholar] [CrossRef]
- Bacher, N.; Tiefenthaler, M.; Sturm, S.; Stuppner, H.; Ausserlechner, M.J.; Kofler, R.; Konwalinka, G. Oxindole alkaloids from Uncaria tomentosa induce apoptosis in proliferating, G0/G1-arrested and bcl-2-expressing acute lymphoblastic leukemia cells. Br. J. Haematol. 2006, 132, 615–622. [Google Scholar] [CrossRef]
- Garcia Prado, E.; Garcia Gimenez, M.D.; De la Puerta Vazquez, R.; Espartero Sánchez, J.L.; Sáenz Rodríguez, M.T. Antiproliferative effects of mitraphylline, a pentacyclic oxindole alkaloid of Uncaria tomentosa on human glioma and neuroblastoma cell lines. Phytomedicine 2007, 14, 280–284. [Google Scholar] [CrossRef]
- Lopes, C.M.; Dourado, A.; Oliveira, R. Phytotherapy and nutritional supplements on breast cancer. Biomed. Res. Int. 2017, 2017, 42. [Google Scholar] [CrossRef]
- Sheng, Y.; Pero, R.W.; Wagner, H. Treatment of chemotherapy-induced leukopenia in a rat model with aqueous extract from Uncaria tomentosa. Phytomedicine 2000, 7, 137–143. [Google Scholar] [CrossRef]
- Farias, I.L.G.; Araújo, M.C.S.; Farias, J.G.; Rossato, L.V.; Elsenbach, L.I.; Dalmora, S.L.; Flores, N.M.; Durigon, M.; Cruz, I.B.; Morsch, V.M.; et al. Uncaria tomentosa for reducing side effects caused by chemotherapy in CRC Patients: Clinical trial. Evid. Based Complement. Alternat. Med. 2012, 2012, 892182. [Google Scholar] [CrossRef]
- Cisneros, F.J.; Jayo, M.; Niedziela, L. An Uncaria tomentosa (cat’s claw) extract protects mice against ozone-induced lung inflammation. J. Ethnopharmacol. 2005, 96, 355–364. [Google Scholar] [CrossRef]
- Ccahuana-Vasquez, R.A.; Santos, S.S.; Koga-Ito, C.Y.; Jorge, A.O. Antimicrobial activity of Uncaria tomentosa against oral human pathogens. Braz. Oral Res. 2007, 21, 46–50. [Google Scholar] [CrossRef]
- Moraes, R.C.; Dalla Lana, A.J.; Kaiser, S.; Carvalho, A.R.; de Oliveira, L.F.S.; Fuentefria, A.M.; Ortega, G.G. Antifungal activity of Uncaria tomentosa (Willd.) DC against resistant non-albicans Candida isolates. Ind. Crops Prod. 2015, 69, 7–14. [Google Scholar] [CrossRef]
- Batiha, G.E.S.; Beshbishy, A.A.; Tayebwa, D.S.; Shaheen, M.H.; Yokoyama, N.; Igarashi, I. Inhibitory effects of Uncaria tomentosa bark, Myrtus communis roots, Origanum vulgare leaves and Cuminum cyminum seeds extracts against the growth of Babesia and Theileria in vitro. Jpn. J. Vet. Parasitol. 2018, 17, 1–13. [Google Scholar]
- Caon, T.; Kaiser, S.; Feltrin, C.; de Carvalho, A.; Sincero, T.C.; Ortega, G.G.; Simões, C.M. Antimutagenic and antiherpetic activities of different preparations from Uncaria tomentosa (cat’s claw). Food Chem. Toxicol. 2014, 66, 30–35. [Google Scholar] [CrossRef]
- Smith, V.J.; Brown, J.H.; Hauton, C. Immunostimulation in crustaceans: Does it really protect against infection? Fish Shellfish Immunol. 2003, 15, 71–90. [Google Scholar] [CrossRef]
- Kośmider, A.; Czepielewska, E.; Kuraś, M.; Gulewicz, K.; Pietrzak, W.; Nowak, R.; Nowicka, G. Uncaria tomentosa Leaves Decoction Modulates Differently ROS Production in Cancer and Normal Cells, and Effects Cisplatin Cytotoxicity. Molecules 2017, 22, 620. [Google Scholar] [CrossRef]
- Eberlin, S.; dos Santos, L.M.; Queiroz, M.L. Uncaria tomentosa extract increases the number of myeloid progenitor cells in the bone marrow of mice infected with Listeria monocytogenes. Int. Immunopharmacol. 2005, 5, 1235–1246. [Google Scholar] [CrossRef] [PubMed]
- Allen-Hall, L.; Cano, P.; Arnason, J.T.; Rojas, R.; Lock, O.; Lafrenie, R.M. Treatment of THP-1 cells with Uncaria tomentosa extracts differentially regulates the expression if IL-1beta and TNF-alpha. J. Ethnopharmacol. 2007, 109, 312–317. [Google Scholar] [CrossRef]
- Zhu, K.; Yang, S.N.; Ma, F.F.; Gu, X.F.; Zhu, Y.C.; Zhu, Y.Z. The novel analogue of hirsutine as an anti-hypertension and vasodilatary agent both in vitro and in vivo. PLoS ONE 2015, 10, e0119477. [Google Scholar] [CrossRef]
- Leenen, F.H.; Ruzicka, M.; Huang, B.S. Central sympathoinhibitory effects of calcium channel blockers. Curr. Hypertens. Rep. 2001, 3, 314–321. [Google Scholar] [CrossRef] [PubMed]
- Snow, A.D.; Castillo, G.M.; Nguyen, B.P.; Choi, P.Y.; Cummings, J.A.; Cam, J.; Hu, Q.; Lake, T.; Pan, W.; Kastin, A.J.; et al. The Amazon rain forest plant Uncaria tomentosa (cat’s claw) and its specific proanthocyanidin constituents are potent inhibitors and reducers of both brain plaques and tangles. Sci. Rep. 2019, 9, 561. [Google Scholar] [CrossRef] [PubMed]
- Rojas-Duran, R.; González-Aspajo, G.; Ruiz-Martel, C.; Bourdy, G.; Doroteo-Ortega, V.H.; Alban-Castillo, J.; Robert, G.; Auberger, P.; Deharo, E. Anti-inflammatory activity of Mitraphylline isolated from Uncaria tomentosa bark. J. Ethnopharmacol. 2012, 143, 801–804. [Google Scholar] [CrossRef] [PubMed]
- Azevedo, B.C.; Roxo, M.; Borges, M.C.; Peixoto, H.; Crevelin, E.J.; Bertoni, B.W.; Contini, S.H.T.; Lopes, A.A.; França, S.C.; Pereira, A.M.S.; et al. Antioxidant activity of an aqueous leaf extract from Uncaria tomentosa and its major alkaloids mitraphylline and isomitraphylline in Caenorhabditis elegans. Molecules 2019, 24, 3299. [Google Scholar] [CrossRef]
- Kim, T.J.; Lee, J.H.; Lee, J.J.; Yu, J.Y.; Hwang, B.Y.; Ye, S.K.; Shujuan, L.; Gao, L.; Pyo, M.Y.; Yun, Y.P. Corynoxeine isolated from the hook of Uncaria rhynchophylla inhibits rat aortic vascular smooth muscle cell proliferation through the blocking of extracellular signal regulated kinase 1/2 phosphorylation. Biol. Pharm. Bull. 2008, 31, 2073–2078. [Google Scholar] [CrossRef]
- Chen, C.X.; Jin, R.M.; Li, Y.K.; Zhong, J.; Yue, L.; Chen, S.C.; Zhou, J.Y. Inhibitory effect of rhynchophylline on platelet aggregation and thrombosis. Zhongguo Yao Li Xue Bao 1999, 13, 126–130. [Google Scholar]
- Cosentino, C.; Torres, L. Reversible worsening of Parkinson’s disease motor symptoms after oral intake of Uncaria tomentosa (cat’s claw). Clin. Neuropharmacol. 2008, 31, 293–294. [Google Scholar] [CrossRef]
- Navarro, V.J.; Barnhart, H.; Bonkovsky, H.L.; Davern, T.; Fontana, R.J.; Grant, L.; Reddy, K.R.; Seeff, L.B.; Serrano, J.; Sherker, A.H.; et al. Liver injury from herbals and dietary supplements in the U.S. Drug-Induced Liver Injury Network. Hepatology 2014, 60, 1399–1408. [Google Scholar] [CrossRef] [PubMed]
- Ernst, E. Cardiovascular adverse effects of herbal medicines: A systematic review of the recent literature. Can. J. Cardiol. 2003, 19, 818–827. [Google Scholar]
- Vogel, J.H.; Bolling, S.F.; Costello, R.B.; Guarneri, E.M.; Krucoff, M.W.; Longhurst, J.C.; Olshansky, B.; Pelletier, K.R.; Tracy, C.M.; Vogel, R.A.; et al. Integrating complementary medicine into cardiovascular medicine: A report of the American college of cardiology foundation task force on clinical expert consensus documents (writing committee to develop an expert consensus document on complementary and integrative medicine). J. Am. Coll. Cardiol. 2005, 46, 184–221. [Google Scholar]
- Standard, N. Natural Standard Herb & Supplement Guide-E-Book: An Evidence-Based Reference; Elsevier Health Sciences: Amsterdam, The Netherlands, 2016. [Google Scholar]
- Gabardi, S.; Munz, K.; Ulbricht, C. A review of dietary supplement–induced renal dysfunction. Clin. J. Amer. Soc. Nephr. 2007, 2, 757–765. [Google Scholar] [CrossRef] [PubMed]
- Kemper, K.J. Shark cartilage, cat’s claw, and other complementary cancer therapies. Contemp. Ped. 1999, 16, 101. [Google Scholar]
- Yano, S.; Horiuchi, H.; Horie, S.; Aimi, N.; Sakai, S.; Watanabe, K. Ca2+, channel-blocking effects of hirsutine, an indole alkaloid from Uncaria genus, in the isolated rat aorta. Planta Medica. 1991, 57, 403–405. [Google Scholar] [CrossRef] [PubMed]
- Budzinski, J.W.; Foster, B.C.; Vandenhoek, S.; Arnason, J.T. An in vitro evaluation of human cytochrome P450 3A4 inhibition by selected commercial herbal extracts and tinctures. Phytomedicine 2000, 7, 273–282. [Google Scholar] [CrossRef]
- Scott, G.N.; Elmer, G.W. Update on natural product—Drug interactions. Am. J. Health Syst. Pharm. 2002, 59, 339–347. [Google Scholar] [CrossRef] [PubMed]
- Sheng, Y.; Li, L.; Holmgren, K.; Pero, R.W. DNA repair enhancement of aqueous extracts of Uncaria tomentosa in a human volunteer study. Phytomedicine 2010, 8, 275–282. [Google Scholar] [CrossRef]
- Lehmann-Willenbrock, E.; Riedel, H.H. Clinical and endocrinologic studies of the treatment of ovarian insufficiency manifestations following hysterectomy with intact adnexa. Zentralbl. Gynakol. 1988, 110, 611–618. [Google Scholar]
- Valerio, L.G., Jr.; Gonzales, G.F. Toxicological aspects of the South American herbs cat’s claw (Uncaria tomentosa) and Maca (Lepidium meyenii): A critical synopsis. Toxicol. Rev. 2005, 24, 11–35. [Google Scholar] [CrossRef]
- Williams, J.E. Review of antiviral and immunomodulating properties of plants of the Peruvian rainforest with a particular emphasis on Una de Gato and Sangre de Grado. Altern. Med. Rev. 2001, 6, 567–579. [Google Scholar]
| Compounds | Molecular Formula | Structure | Biological Activity | References |
|---|---|---|---|---|
| Pentacyclic oxindole alkaloids (POA) | ||||
| Uncarine F | C21H24N2O4 |  | Anticancer activity | [36] |
| Speciophylline | C21H24N2O4 |  | Anti-neoplastic activity | [19] |
| Mitraphylline | C21H24N2O4 |  | Anti-inflammatory, antioxidant activities | [54,55] |
| Isomitraphylline | C21H24N2O4 | 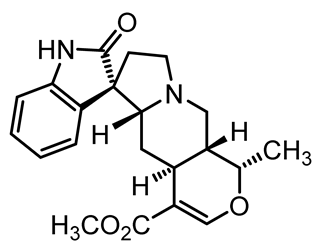 | Antioxidant activity | [55] |
| Pteropodine | C21H24N2O4 | 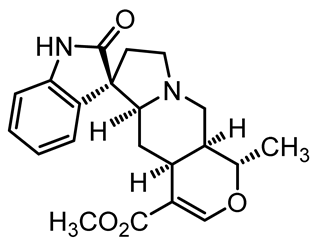 | Immunomodulating properties | [48] |
| Isopteropodine | C21H24N2O4 | 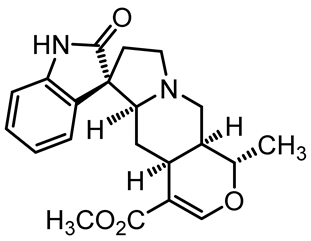 | Antimicrobial activity | [37] |
| Tetracyclic oxindole alkaloid (TOA) | ||||
| Corynoxeine | C22H26N2O4 |  | Antiproliferative activity | [56] |
| Rhynchophylline | C22H28N2O4 | 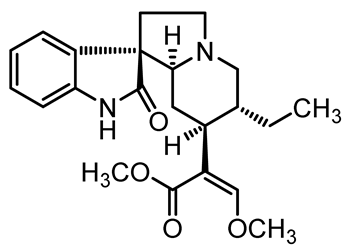 | Inhibit the platelet aggregation and thrombosis | [57] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Batiha, G.E.-S.; Magdy Beshbishy, A.; Wasef, L.; Elewa, Y.H.A.; Abd El-Hack, M.E.; Taha, A.E.; Al-Sagheer, A.A.; Devkota, H.P.; Tufarelli, V. Uncaria tomentosa (Willd. ex Schult.) DC.: A Review on Chemical Constituents and Biological Activities. Appl. Sci. 2020, 10, 2668. https://doi.org/10.3390/app10082668
Batiha GE-S, Magdy Beshbishy A, Wasef L, Elewa YHA, Abd El-Hack ME, Taha AE, Al-Sagheer AA, Devkota HP, Tufarelli V. Uncaria tomentosa (Willd. ex Schult.) DC.: A Review on Chemical Constituents and Biological Activities. Applied Sciences. 2020; 10(8):2668. https://doi.org/10.3390/app10082668
Chicago/Turabian StyleBatiha, Gaber El-Saber, Amany Magdy Beshbishy, Lamiaa Wasef, Yaser H. A. Elewa, Mohamed E. Abd El-Hack, Ayman E. Taha, Adham Abdullah Al-Sagheer, Hari Prasad Devkota, and Vincenzo Tufarelli. 2020. "Uncaria tomentosa (Willd. ex Schult.) DC.: A Review on Chemical Constituents and Biological Activities" Applied Sciences 10, no. 8: 2668. https://doi.org/10.3390/app10082668
APA StyleBatiha, G. E.-S., Magdy Beshbishy, A., Wasef, L., Elewa, Y. H. A., Abd El-Hack, M. E., Taha, A. E., Al-Sagheer, A. A., Devkota, H. P., & Tufarelli, V. (2020). Uncaria tomentosa (Willd. ex Schult.) DC.: A Review on Chemical Constituents and Biological Activities. Applied Sciences, 10(8), 2668. https://doi.org/10.3390/app10082668