The Application of Seabed Silt in the Preparation of Artificial Algal Reefs
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Specimen Preparation
2.3. Experimental Method
2.3.1. Compressive Strength Test
2.3.2. Dissolution Test
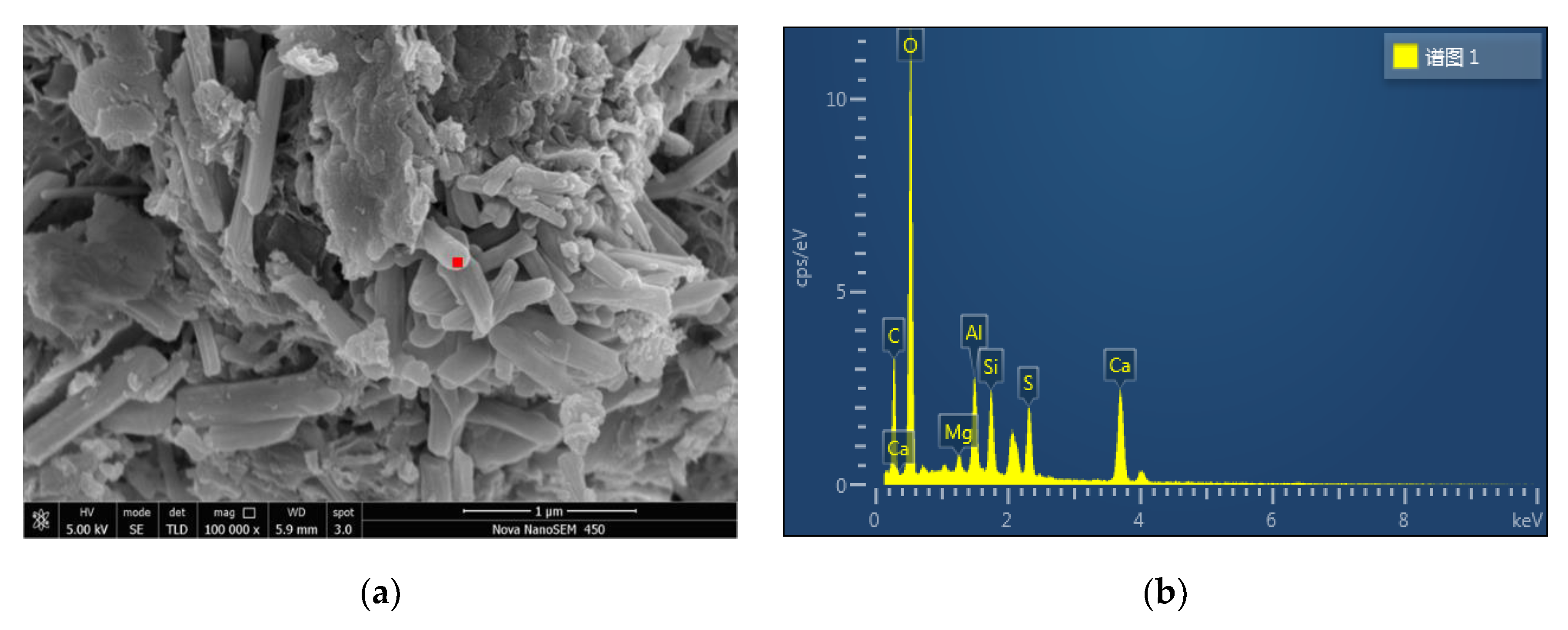
2.3.3. Microstructure Tests
2.4. Statistical Method
3. Results and Discussion
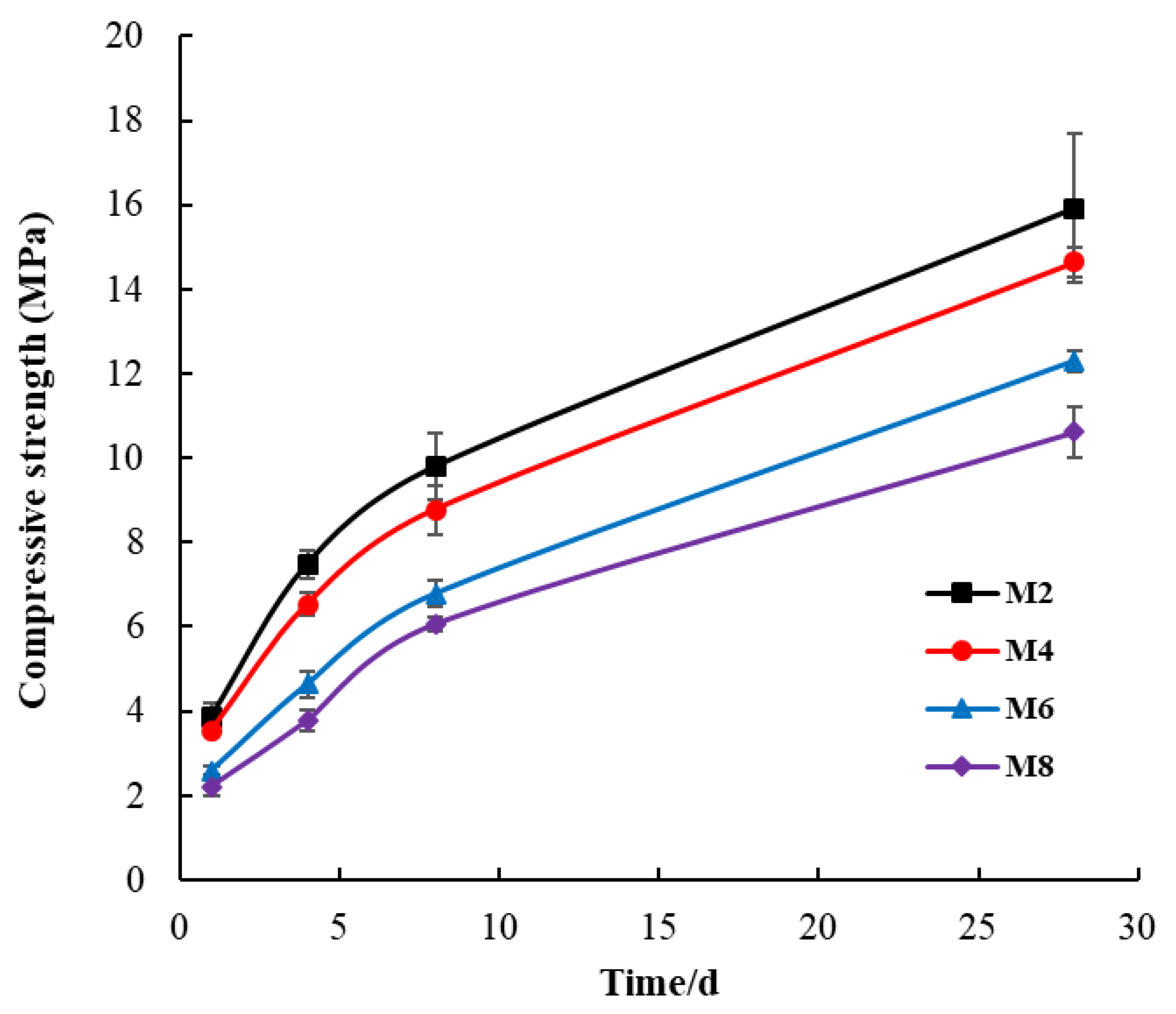
3.1. Compressive Strength
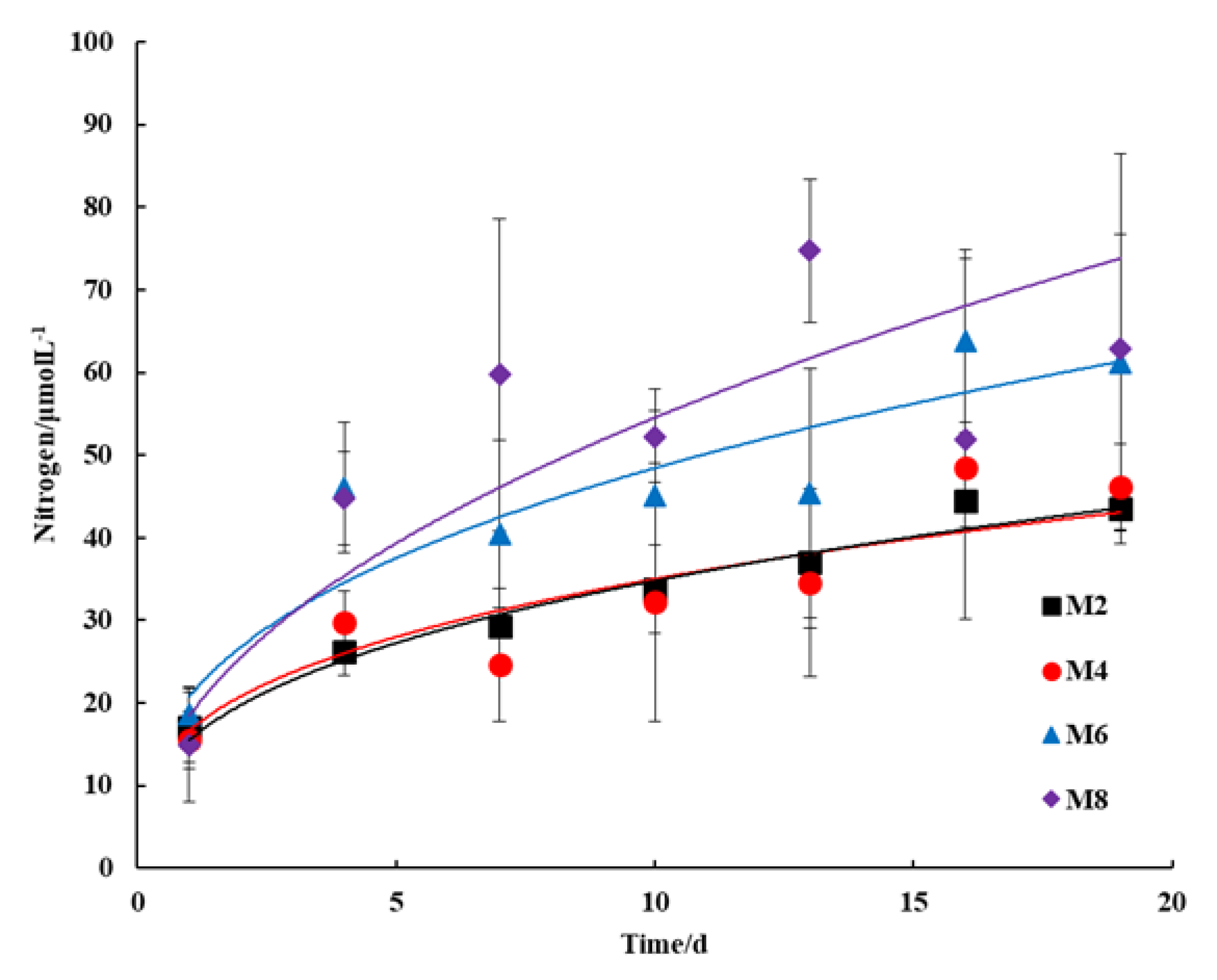
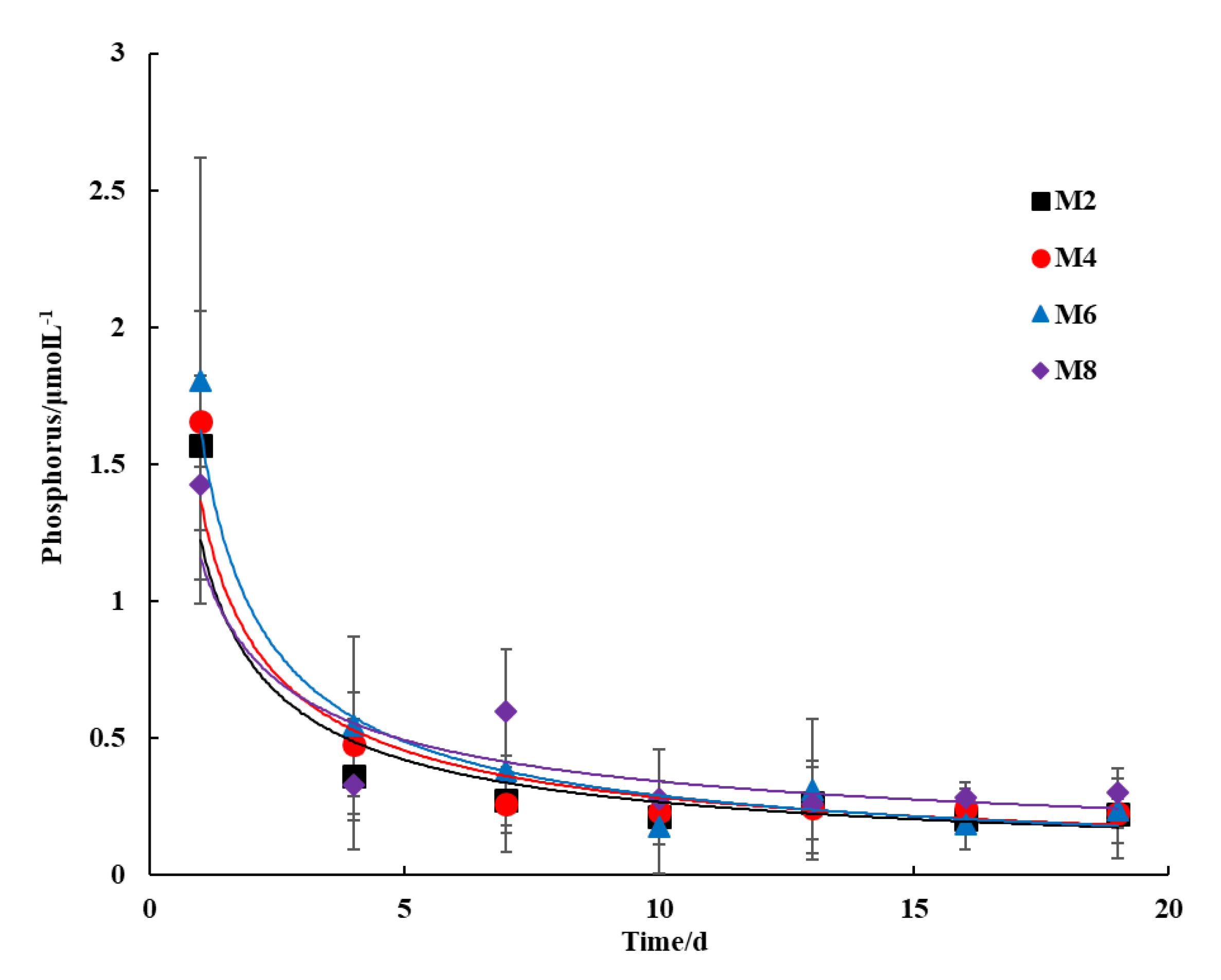
3.2. Nutrient Dissolution
3.2.1. Nitrogen Content
3.2.2. Phosphorus Content
3.2.3. Ratio of Nitrogen to Phosphorus
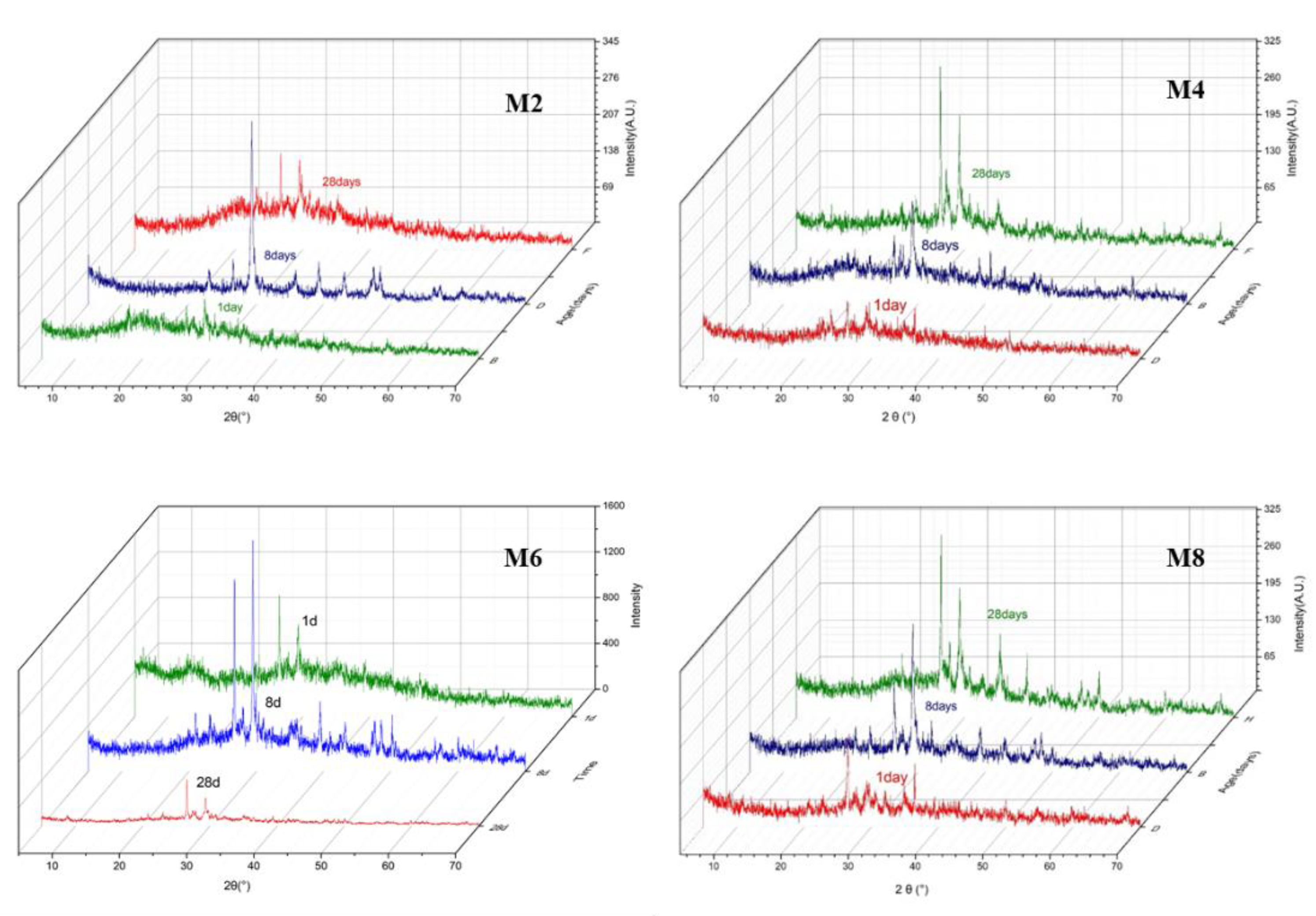
3.3. XRD Analysis
3.4. SEM Analysis
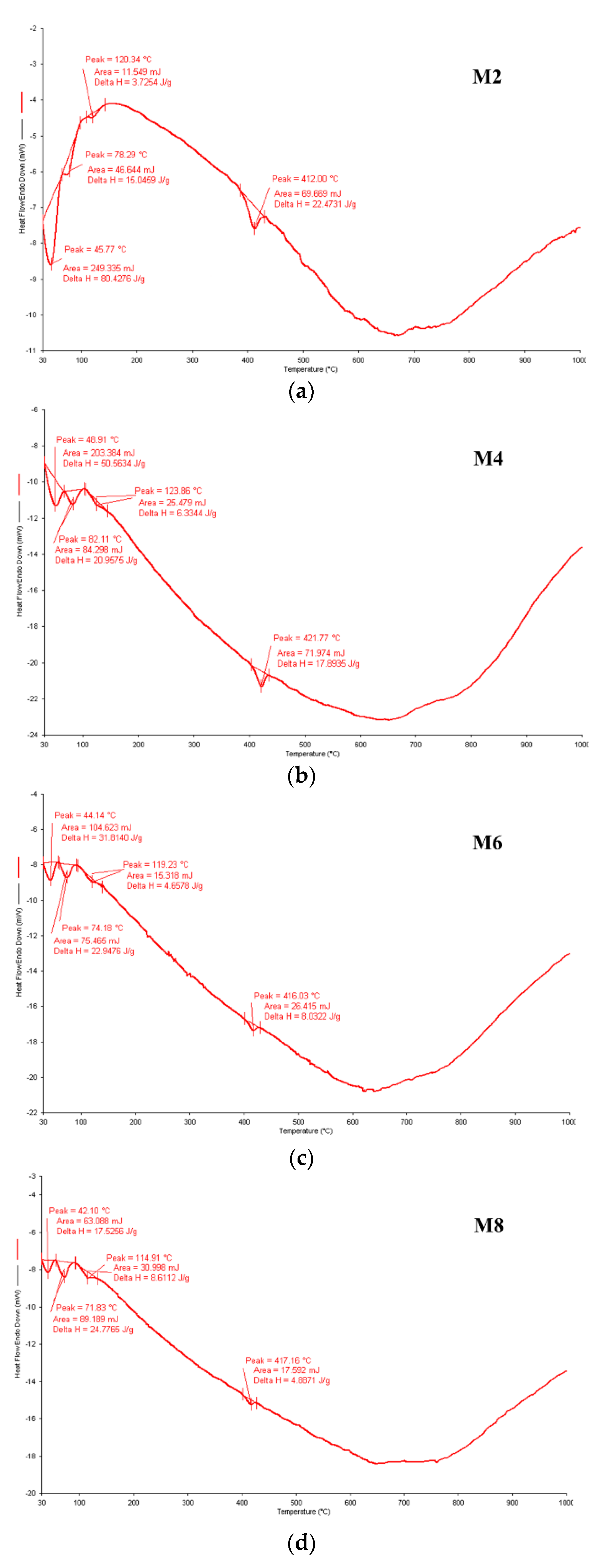
3.5. DSC
4. Conclusion
- In this study, seabed silt was tested as a potential raw material for artificial reefs. The SAR specimens were composed purely of seabed silt and cement in four proportions. The compressive strength development and nutrient dissolution were examined in the SAR specimens. The hydration products of the SAR paste were investigated through XRD, SEM, and DSC techniques. The compressive strength of the SAR specimen was between that of C15 and C20, which was inversely proportional to their seabed silt content. The SAR specimens were able to continuously dissolve nitrogen-based nutrients, which is conducive to the improvement of primary productivity in the surrounding water. However, the effect on the N/P ratio in the surrounding water depends on the conditions of the seabed silt itself.
- The results of compressive strength and microstructure tests showed that the strength of the specimens increased continuously during the hydration process of the mixture of seabed silt and cement. The ettringite became interlaced with C–S–H gel to form a compact structure, but the main hydration products of the SAR paste were SiO2 and CaCO3. Ca(OH)2 was not detected in the SAR specimens, which may help to improve its adhesion and biological effects. This suggests that seabed silt can be partially mixed into AARs as a raw material.
- The potential bioadhesion effect of seabed silt and cement mixture needs further experimental verification, especially the influence of interaction between microorganisms and concrete materials. In addition, seabed silt may also contain heavy metals and other organic materials. Whether these substances will be dissolved out or influence the attachment and growth of algae also needs further study.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Seaman, W.; Jensen, A.C. Purposes and practices of artificial reef evaluation. In Artificial Reef Evaluation with Application to Natural Marine Habitats; Seaman, W., Ed.; CRC Press: Boca Raton, FL, USA, 2000; pp. 95–120. [Google Scholar]
- Guilbeau, B.P.; Harry, F.P.; Gambrell, R.P.; Knopf, F.C.; Dooley, K.M. Algae attachment on carbonated cements in fresh and brackish waters—Preliminary results. Ecol. Eng. 2003, 20, 309–319. [Google Scholar] [CrossRef]
- Jiang, Z.Y.; Liang, Z.L.; Liu, Y. Application of Sludge in the preparation of artificial algal reef. Trans. Chin. Soc. Agric. Eng. 2014, 31, 242–245, (In Chinese with English abstract). [Google Scholar]
- Hanner, S.E.; McIntosh, T.L.; Blair, S.M. Development of benthic and fish assemblages on artificial reef materials compared to adjacent natural reef assemblages in Miami-Dade County, Florida. Bull. Mar. Sci. 2006, 78, 57–70. [Google Scholar]
- Yu, Y.G.; Gao, W.; Castel, A.; Liu, A.; Chen, X.J.; Liu, M.Y. Assessing external sulfate attack on thin-shell artificial reef structures under uncertainty. Ocean. Eng. 2020, 207, 107397. [Google Scholar] [CrossRef]
- Perkol-Finkel, S.; Zilman, G.; Sella, I.; Miloh, T.; Benayahu, Y. Floating and fixed artificial habitats: Spatial and temporal patterns of benthic communities in a coral reef environment. Estuar. Coast. Shelf Sci. 2008, 77, 491–500. [Google Scholar] [CrossRef]
- Relini, M.; Torchia, G.; Relini, G. Seasonal-variation of fish assemblages in the loano artificial reef (ligurian sea Northwestern Mediterranean). Bull. Mar. Sci. 1994, 55, 401–417. [Google Scholar]
- Patranella, A.; Kilfoyle, K.; Pioch, S.; Spieler, RE. Artificial reefs as juvenile fish habitat in a marina. J. Coast. Res. 2017, 33, 1341–1351. [Google Scholar] [CrossRef]
- Lemoine, H.R.; Paxton, A.B.; Anisfeld, S.C.; Rosemond, R.C. Selecting the optimal artificial reefs to achieve fish habitat enhancement goals. Biol. Conserv. 2019, 238, 108200. [Google Scholar] [CrossRef]
- Frease, R.A.; Windsor, J.G. Behavior of selected polycyclic aromatic-hydrocarbons associated with stabilized oil and coal ash artificial reef. Mar. Pollut. Bull. 1991, 22, 15–19. [Google Scholar] [CrossRef]
- Kress, N.; Tom, M.; Spanier, E. The use of coal fly ash in concrete for marine artificial reefs in the southeastern Mediterranean: Compressive strength, sessile biota, and chemical composition. ICES J. Mar. Sci. 2002, 59, S231–S237. [Google Scholar] [CrossRef][Green Version]
- Katherine, K.Y.L. Coral recruitment onto an experimental pulverised fuel ash–concrete artificial reef. Mar. Pollut. Bull. 2003, 46, 642–653. [Google Scholar]
- Bigdeli, Y.; Barbato, M.; Lofton, C.D.; Gutierrez-Wing, M.T.; Rusch, K.A. Mechanical properties and performance under laboratory and field conditions of a lightweight fluorogypsum-based blend for economic artificial-reef construction. J. Mater. Civ. Eng. 2020, 32, 04020172. [Google Scholar] [CrossRef]
- Huang, X.Y.; Wang, Z.J.; Liu, Y.; Hu, W.T.; Ni, W. On the use of blast furnace slag and steel slag in the preparation of green artificial reef concrete. Constr. Build. Mater. 2016, 112, 241–246. [Google Scholar] [CrossRef]
- Pan, S.Y.; Adhikari, R.; Chen, Y.H.; Li, P.; Chiang, P.C. Integrated and innovative steel slag utilization for iron reclamation, green material production and CO2 fixation via accelerated carbonation. J. Clean Prod. 2016, 137, 617–631. [Google Scholar] [CrossRef]
- Onoue, K.; Shimono, S. Iron supply capacity of porous concrete using steelmaking slag aggregate for seaweed beds or fish reef blocks. J. Sustain. Met. 2018, 4, 333–342. [Google Scholar] [CrossRef]
- Okumura, S.; Kayano, Y.; Kusaka, K.; Tsumura, S.; Maruyama, K. Experimental release of juvenile red spotted grouper Epinephelus akaara into the artificial reefs made of scallop Patinopecten yessoensis shells. Nippon. Suisan Gakkaishi 2003, 69, 917–925. [Google Scholar] [CrossRef][Green Version]
- Xu, Q.Z.; Zhang, L.B.; Zhang, T.; Zhou, Y.; Xia, S.D.; Liu, H.; Yang, H.S. Effects of an artificial oyster shell reef on macrobenthic communities in Rongcheng Bay, East China. Chin. J. Oceanol. Limnol. 2014, 32, 99–110. [Google Scholar] [CrossRef]
- Liu, G.S.; Li, W.T.; Zhang, X.M. Assessment of the benthic macrofauna in an artificial shell reef zone in Shuangdao Bay, Yellow Sea. Mar. Pollut. Bull. 2017, 114, 778–785. [Google Scholar] [CrossRef]
- Callaway, R. Interstitial space and trapped sediment drive benthic communities in artificial shell and rock reefs. Front. Mar. Sci. 2018, 5, 1–10. [Google Scholar] [CrossRef]
- Souche, J.C.; Saout, G.L.; Salgues, M.; Pioch, S. Effect of concrete with bio-active admixture on marine colonisation in mediterranean environment. Matériaux Tech. 2016, 104, 504–512. [Google Scholar] [CrossRef]
- Hayek, M.; Salgues, M.; Habouzit, F.; Bayle, S.; Souche, J.C.; De Weerdt, K.; Pioch, S. The influence of carbonation on the biocolonization of cementitious materials in the marine environment. Matériaux Tech. 2020, 108, 202. [Google Scholar] [CrossRef]
- Lu, X.X.; Ran, L.S.; Liu, S.; Jiang, T.; Zhang, S.R.; Wang, J.J. Sediment loads response to climate change: A preliminary study of eight large Chinese rivers. Int. J. Sediment. Res. 2013, 28, 1–14. [Google Scholar] [CrossRef]
- Liu, C.; Sui, J.Y.; He, Y.; Hirshfield, F. Changes in runoff and sediment load from major Chinese rivers to the Pacific Ocean over the period 1955–2010. Int. J. Sediment. Res. 2013, 28, 486–492. [Google Scholar] [CrossRef]
- Liu, G.S.; Cai, X.Y.; Tong, F.; Wang, L.; Zhang, X.M. Investigation of massive death of sea cucumber in artificial reef zone of Shuangdao Bay, Weihai. Fish. Inf. Strategy 2014, 29, 122–129, (In Chinese with English abstract). [Google Scholar]
- Ministry of Construction; The State Administration of Quality Supervision; Inspection and Quarantine People’s Republic of China. GB/T50081-2002 Standard for Test Method of Mechanical Properties on Ordinary Concrete; Ministry of Construction: Bejing, China, 2002.
- The State Administration of Quality Supervision; Inspection and Quarantine People’s Republic of China. GB/T 12763.4-2007 Specifications for Oceanographic Survey–Part 4: Survey of Chemical Parameters in Sea Water; The State Administration of Quality Supervision: Beijing, China, 2007.
- Ministry of Construction; The State Administration of Quality Supervision; Inspection and Quarantine People’s Republic of China. GB 50010-2010 Code for Design of Concrete Structures; Ministry of Construction: Bejing, China, 2010.
- Xu, Q.; Ji, T.; Yang, Z.; Ye, Y. Preliminary investigation of artificial reef concrete with sulphoaluminate cement, marine sand and sea water. Constr. Build. Mater. 2019, 211, 837–846. [Google Scholar] [CrossRef]
- Lourenco, S.O.; Barbarino, E.; Nascimento, A.; Freitas, J.N.P.; Diniz, G.S. Tissue nitrogen and phosphorus in seaweeds in a tropical eutrophic environment: What a long-term study tells us. J. Appl. Phycol. 2006, 18, 389–398. [Google Scholar] [CrossRef]
- Huo, S.; Liu, J.; Zhu, F.; Basheer, S.; Necas, D.; Zhang, R.; Li, K.; Chen, D.; Cheng, P.; Cobb, K.; et al. Post treatment of swine anaerobic effluent by weak electric field following intermittent vacuum assisted adjustment of N:P ratio for oil-rich filamentous microalgae production. Bioresour. Technol. 2020, 314, 123718. [Google Scholar] [CrossRef]
- Ghorbel, H.; Samet, B. Effect of iron on pozzolanic activity of kaolin. Constr. Build. Mater. 2013, 44, 185–191. [Google Scholar] [CrossRef]



| Components | SiO2 | Al2O3 | Fe2O3 | CaO | MgO | TiO2 | Na2O | K2O |
|---|---|---|---|---|---|---|---|---|
| Content (%) | 53.47 | 15.76 | 8.25 | 2.09 | 2.49 | 0.91 | 3.27 | 1.93 |
| Specimen | M2 | M4 | M6 | M8 |
|---|---|---|---|---|
| N/P | 193.88 | 203.62 | 298.52 | 210.01 |
| Element | wt% | Atomic Percent |
|---|---|---|
| C | 16.96 | 26.70 |
| O | 43.26 | 51.14 |
| Mg | 0.90 | 0.70 |
| Al | 5.19 | 3.64 |
| Si | 5.24 | 3.53 |
| S | 7.34 | 4.33 |
| Ca | 21.11 | 9.96 |
| Total quantity | 100.00 | 100.00 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, Z.; Zhang, J.; Nie, Z.; Guo, Z.; Zhu, L.; Cong, W.; Chen, Y.; Liang, Z. The Application of Seabed Silt in the Preparation of Artificial Algal Reefs. Appl. Sci. 2020, 10, 7279. https://doi.org/10.3390/app10207279
Jiang Z, Zhang J, Nie Z, Guo Z, Zhu L, Cong W, Chen Y, Liang Z. The Application of Seabed Silt in the Preparation of Artificial Algal Reefs. Applied Sciences. 2020; 10(20):7279. https://doi.org/10.3390/app10207279
Chicago/Turabian StyleJiang, Zhaoyang, Jiating Zhang, Zhaoyi Nie, Zhansheng Guo, Lixin Zhu, Wei Cong, Yuan Chen, and Zhenlin Liang. 2020. "The Application of Seabed Silt in the Preparation of Artificial Algal Reefs" Applied Sciences 10, no. 20: 7279. https://doi.org/10.3390/app10207279
APA StyleJiang, Z., Zhang, J., Nie, Z., Guo, Z., Zhu, L., Cong, W., Chen, Y., & Liang, Z. (2020). The Application of Seabed Silt in the Preparation of Artificial Algal Reefs. Applied Sciences, 10(20), 7279. https://doi.org/10.3390/app10207279




