Clinical Evaluation of Stretchable and Wearable Inkjet-Printed Strain Gauge Sensor for Respiratory Rate Monitoring at Different Body Postures
Abstract
1. Introduction
2. Methods
2.1. Physiological Measurements
2.1.1. Subjects
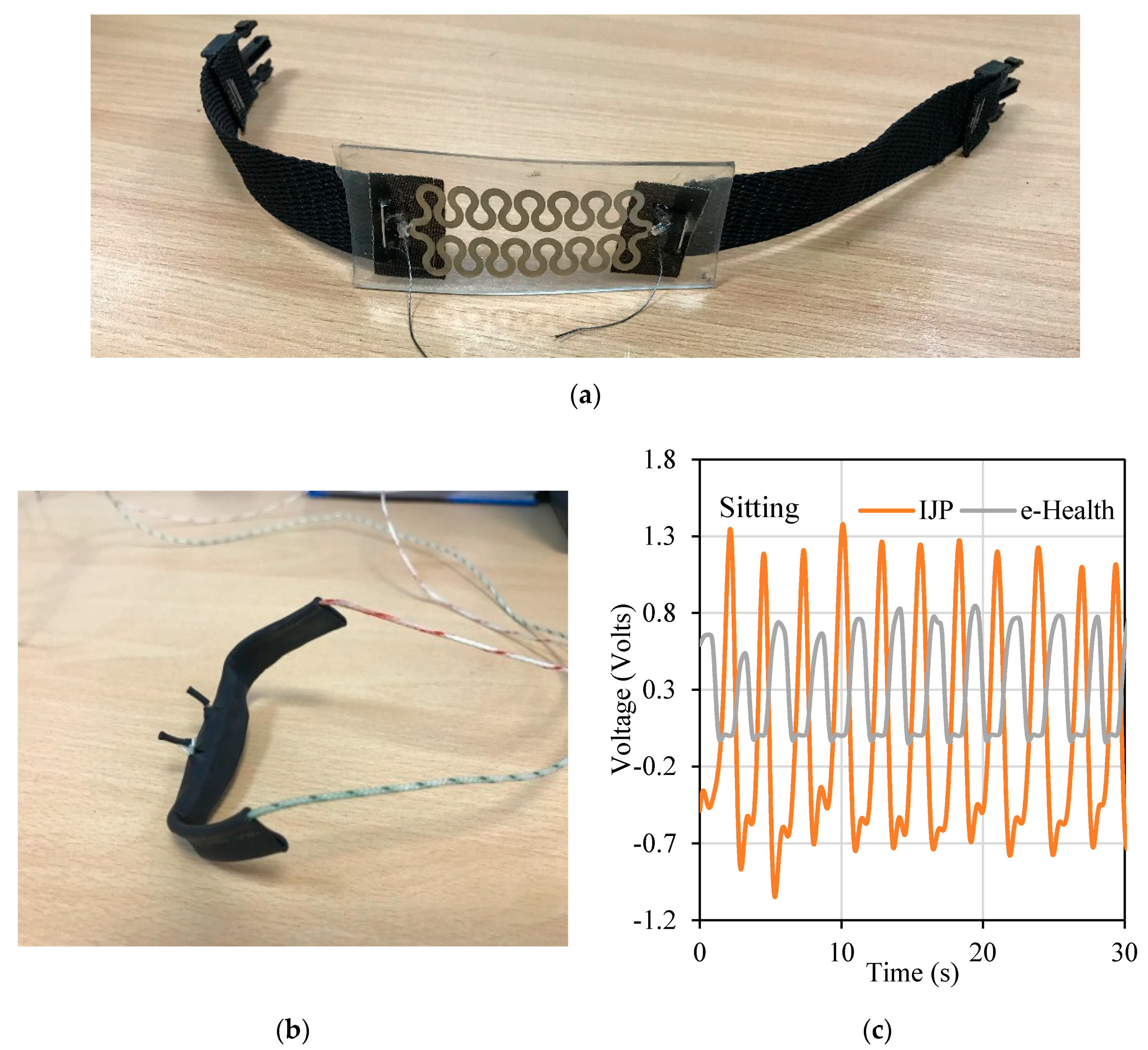
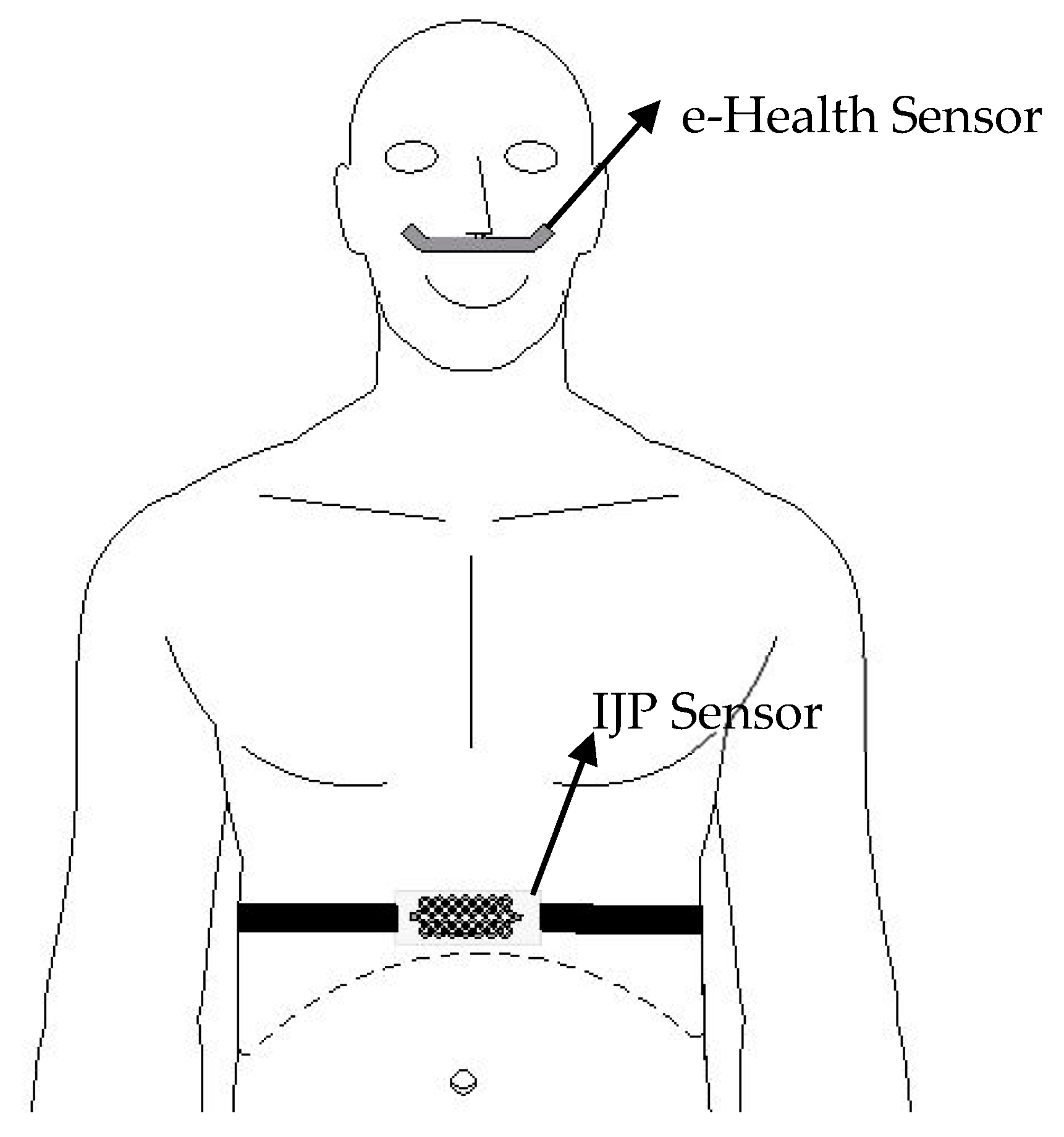
2.1.2. Sensors
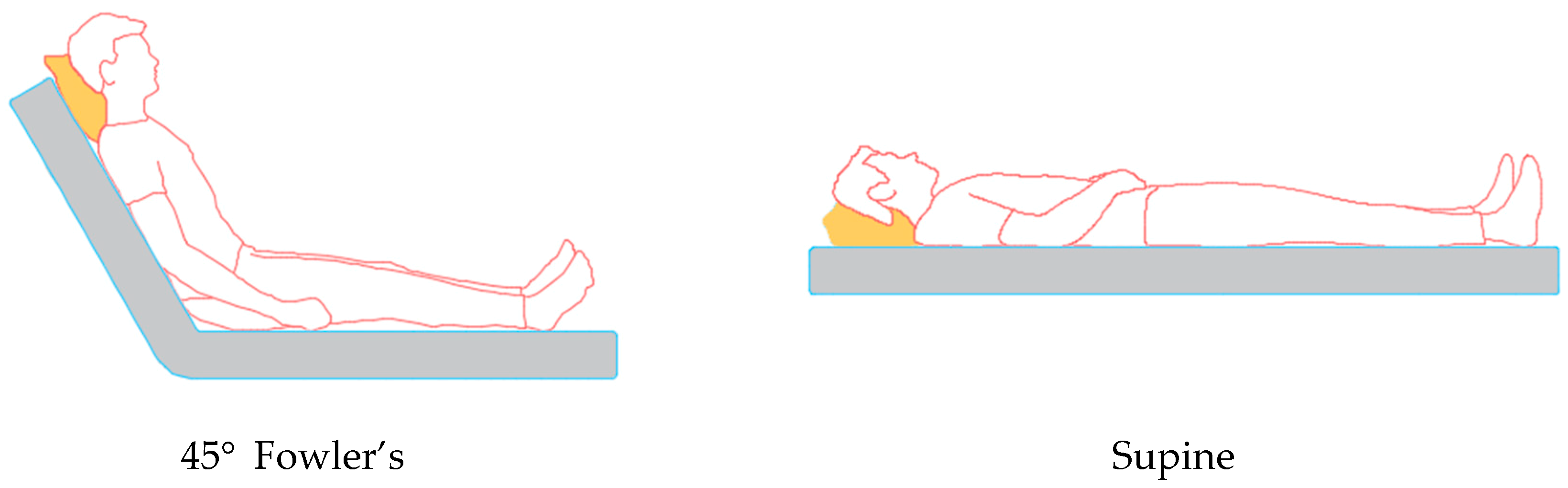
2.1.3. Measurement Procedure
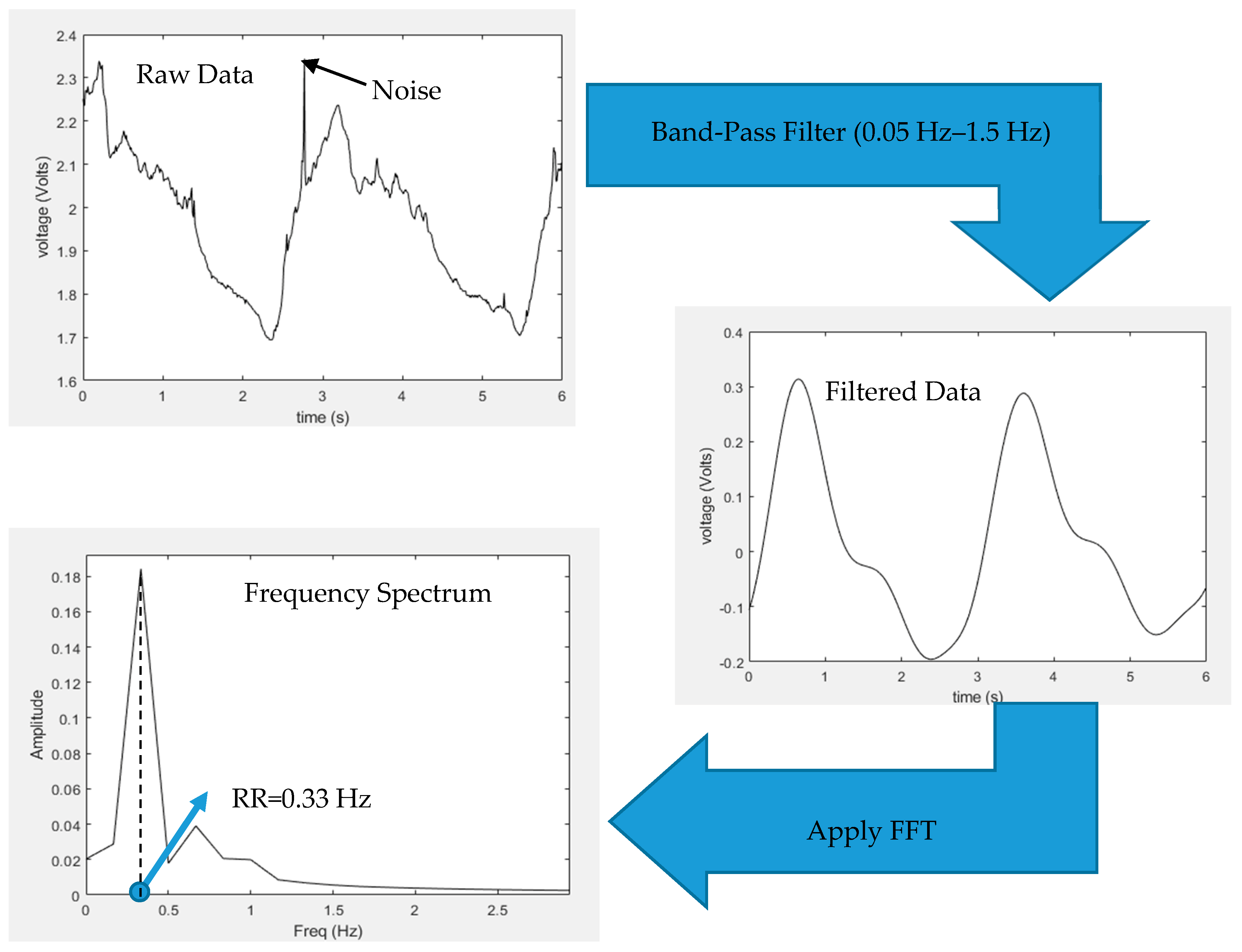
2.2. Respiratory Rate Derivation
2.3. Statistical Analysis
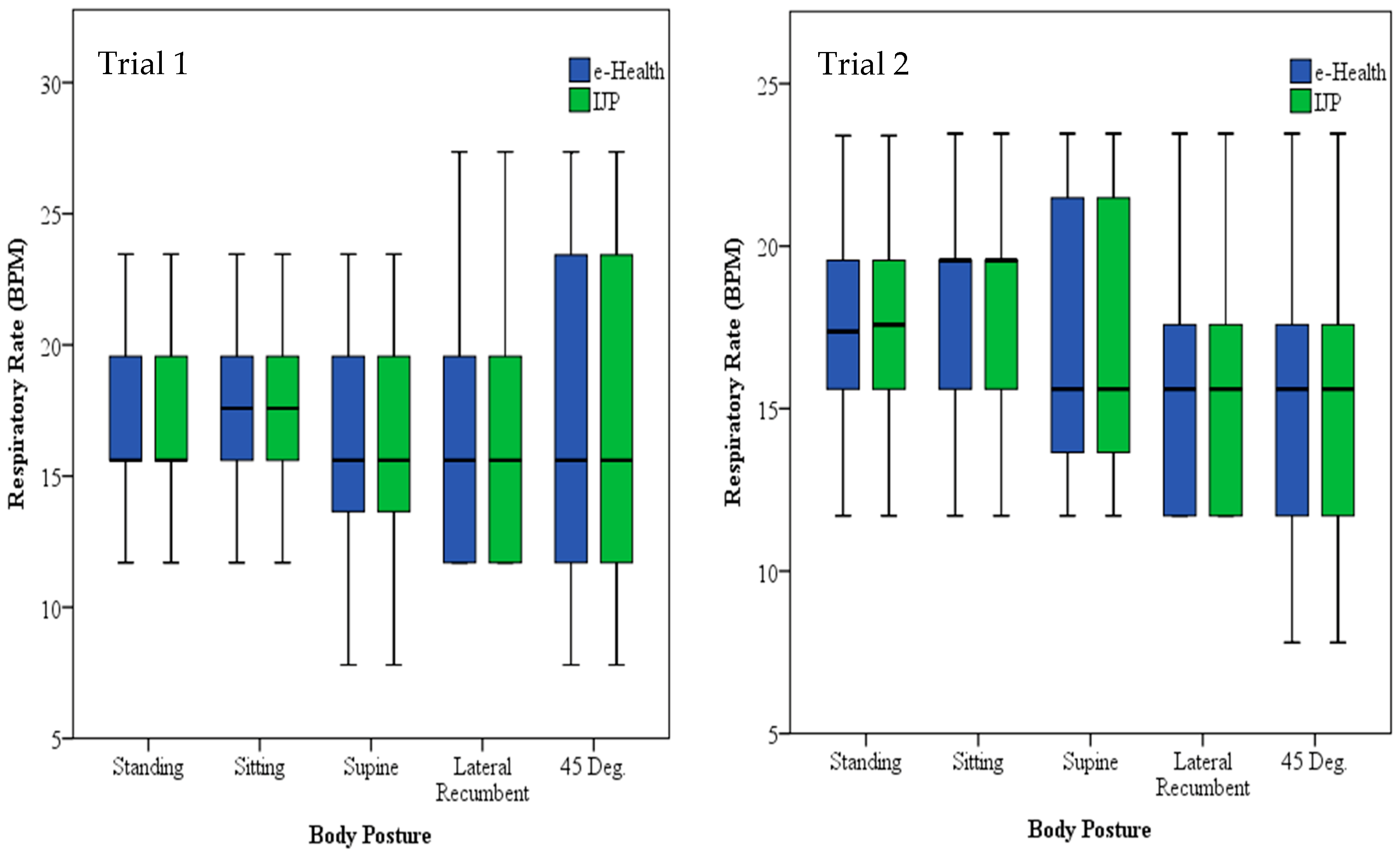
3. Results
4. Discussion
4.1. IJP Sensor Accuracy: Comparison with Other Sensors
4.2. RR Measurements at Different Body Postures
4.3. Application of the IJP Sensor
4.4. Limitations and Future Work
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Gender | Age | Standing | Sitting | 45 Deg. | Supine | Lateral | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Our | Ref | Our | Ref | Our | Ref | Our | Ref | Our | Ref | ||
| M | 24 | 11.7 | 11.7 | 13.65 | 13.65 | 11.7 | 11.7 | 15.6 | 15.6 | 11.7 | 11.7 |
| M | 23 | 21.51 | 21.51 | 21.51 | 21.51 | 17.58 | 17.58 | 19.56 | 19.56 | 23.46 | 23.46 |
| M | 25 | 21.48 | 21.48 | 21.51 | 21.51 | 23.46 | 23.46 | 21.51 | 21.51 | 19.56 | 19.56 |
| M | 25 | 19.56 | 19.56 | 19.56 | 19.56 | 17.58 | 17.58 | 15.63 | 15.63 | 15.6 | 15.6 |
| M | 23 | 17.58 | 17.58 | 21.51 | 21.51 | 13.65 | 13.65 | 21.51 | 21.51 | 17.58 | 17.58 |
| M | 24 | 15.6 | 15.6 | 13.65 | 13.65 | 11.7 | 11.7 | 13.65 | 13.65 | 11.7 | 11.7 |
| M | 24 | 13.65 | 13.65 | 13.65 | 13.65 | 9.75 | 9.75 | 9.75 | 9.75 | 11.7 | 11.7 |
| M | 23 | 23.4 | 23.4 | 23.4 | 23.4 | 23.4 | 23.4 | 23.4 | 23.4 | 23.4 | 23.4 |
| M | 26 | 15.6 | 15.6 | 17.58 | 17.58 | 15.6 | 15.6 | 15.6 | 15.6 | 17.58 | 17.58 |
| M | 21 | 19.56 | 19.35 | 19.56 | 19.56 | 21.51 | 21.51 | 19.56 | 19.56 | 19.56 | 19.56 |
| M | 25 | 17.58 | 17.58 | 19.56 | 19.56 | 19.53 | 19.53 | 17.58 | 17.58 | 15.6 | 15.6 |
| M | 24 | 23.46 | 23.46 | 29.31 | 29.31 | 23.46 | 23.46 | 23.46 | 23.46 | 19.53 | 19.53 |
| M | 24 | 7.8 | 7.8 | 9.75 | 9.75 | 9.75 | 9.75 | 11.7 | 11.7 | 11.7 | 11.7 |
| M | 22 | 15.6 | 15.6 | 15.6 | 15.6 | 11.7 | 11.7 | 9.75 | 9.75 | 11.7 | 11.7 |
| M | 24 | 17.58 | 17.58 | 17.58 | 17.58 | 15.6 | 15.6 | 15.6 | 15.6 | 13.65 | 13.65 |
References
- AL-Khalidi, F.Q.; Saatchi, R.; Burke, D.; Elphick, H.; Tan, S. Respiration rate monitoring methods: A review. Pediatr. Pulmonol. 2011, 46, 523–529. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.J. Clinical evaluation of a novel respiratory rate monitor. J. Clin. Monit. Comput. 2016, 30, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.; Xu, W.; Liu, J.; Samy, L.; Vajid, A.; Alshurafa, N.; Sarrafzadeh, M. Inconspicuous on-bed respiratory rate monitoring. In Proceedings of the 6th International Conference on PErvasive Technologies Related to Assistive Environments, Rhodes, Greece, 29–31 May 2013; ACM Press: New York, NY, USA, 2013; pp. 1–8. [Google Scholar]
- Nam, Y.; Kong, Y.; Reyes, B.; Reljin, N.; Chon, K.H. Monitoring of Heart and Breathing Rates Using Dual Cameras on a Smartphone. PLoS ONE 2016, 11, e0151013. [Google Scholar] [CrossRef]
- Sharma, H.; Sharma, K.K. ECG-derived respiration using Hermite expansion. Biomed. Signal Process. Control 2018, 39, 312–326. [Google Scholar] [CrossRef]
- Di Fiore, J.M. Neonatal cardiorespiratory monitoring techniques. Semin. Neonatol. 2004, 9, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Koch, E.; Dietzel, A. Stretchable sensor array for respiratory monitoring. In Proceedings of the 2017 19th International Conference on Solid-State Sensors, Actuators and Microsystems (TRANSDUCERS), Kaohsiung, Taiwan, 18–22 June 2017; pp. 2227–2230. [Google Scholar]
- Jeong, J.W.; Jang, Y.W.; Lee, I.; Shin, S.; Kim, S. Wearable Respiratory Rate Monitoring using Piezo-resistive Fabric Sensor. In World Congress on Medical Physics and Biomedical Engineering, 7–12 September 2009, Munich, Germany; Dössel, O., Schlegel, W.C., Eds.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 282–284. ISBN 978-3-642-03904-1. [Google Scholar]
- Ciocchetti, M.; Massaroni, C.; Saccomandi, P.; Caponero, M.; Polimadei, A.; Formica, D.; Schena, E. Smart Textile Based on Fiber Bragg Grating Sensors for Respiratory Monitoring: Design and Preliminary Trials. Biosensors 2015, 5, 602–615. [Google Scholar] [CrossRef] [PubMed]
- Chu, M.; Nguyen, T.; Pandey, V.; Zhou, Y.; Pham, H.N.; Bar-Yoseph, R.; Radom-Aizik, S.; Jain, R.; Cooper, D.M.; Khine, M. Respiration rate and volume measurements using wearable strain sensors. NPJ Digit. Med. 2019, 2, 8. [Google Scholar] [CrossRef]
- Liu, H.; Allen, J.; Zheng, D.; Chen, F. Recent development of respiratory rate measurement technologies. Physiol. Meas. 2019, 40, 07TR01. [Google Scholar] [CrossRef]
- Guan, H.; Yang, X.; Sun, W.; Ren, A.; Fan, D.; Zhao, N.; Guan, L.; Haider, D.; Abbasi, Q. Posture-Specific Breathing Detection. Sensors 2018, 18, 4443. [Google Scholar] [CrossRef]
- Furtak, N.T.; Skrzetuska, E. Development of Screen-Printed Breathing Rate Sensors. FIBRES Text. East. Eur. 2013, 6, 84–88. [Google Scholar]
- Zhu, Y.; Maniyeri, J.; Fook, V.F.S.; Zhang, H. Estimating respiratory rate from FBG optical sensors by using signal quality measurement. In Proceedings of the 2015 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Milan, Italy, 25–29 August 2015; pp. 853–856. [Google Scholar]
- Wu, D.; Wang, L.; Zhang, Y.; Huang, B.; Wang, B.; Lin, S.; Xu, X. A wearable respiration monitoring system based on digital respiratory inductive plethysmography. In Proceedings of the 2009 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Minneapolis, MN, USA, 3–6 September 2009; pp. 4844–4847. [Google Scholar]
- Hesse, M.; Christ, P.; Hormann, T.; Ruckert, U. A respiration sensor for a chest-strap based wireless body sensor. In Proceedings of the IEEE SENSORS 2014 Proceedings, Valencia, Spain, 2–5 November 2014; pp. 490–493. [Google Scholar]
- Sun, X.; Qiu, L.I.; Wu, Y.; Tang, Y.; Cao, G.; State, T.P. SleepMonitor: Monitoring Respiratory Rate and Body Position During Sleep Using Smartwatch. PACM Interact. Mob. Wearable Ubiquitous Technol. 2017, 13, 104. [Google Scholar] [CrossRef]
- Al-Halhouli, A.; Qitouqa, H.; Alashqar, A.; Abu-Khalaf, J. Inkjet printing for the fabrication of flexible/stretchable wearable electronic devices and sensors. Sens. Rev. 2018, 38, 438–452. [Google Scholar] [CrossRef]
- Abu-Khalaf, J.; Al-Ghussain, L.; Al-Halhouli, A. Fabrication of Stretchable Circuits on Polydimethylsiloxane (PDMS) Pre-Stretched Substrates by Inkjet Printing Silver Nanoparticles. Materials 2018, 11, 2377. [Google Scholar] [CrossRef] [PubMed]
- Abu-khalaf, J.M.; Saraireh, R.; Eisa, S.M.; Al-halhouli, A. Experimental Characterization of Inkjet-Printed Stretchable Circuits for Wearable Sensor Applications. Sensors 2018, 18, 3476. [Google Scholar] [CrossRef] [PubMed]
- Abu-khalaf, J.M.; Al-Ghussain, L.; Nadi, A.; Saraireh, R.; Rabayah, A.; Altarazi, S.; Al-Halhouli, A. Optimization of Geometry Parameters of Inkjet-Printed Silver Nanoparticle Traces on PDMS Substrates Using Response Surface Methodology. Materials 2019, 12, 3329. [Google Scholar] [CrossRef]
- Al-Halhouli, A.; Al-Ghussain, L.; El Bouri, S.; Liu, H.; Zheng, D. Fabrication and Evaluation of a Novel Non-Invasive Stretchable and Wearable Respiratory Rate Sensor Based on Silver Nanoparticles Using Inkjet Printing Technology. Polymers 2019, 11, 1518. [Google Scholar] [CrossRef]
- Bayo-Monton, J.L.; Martinez-Millana, A.; Han, W.; Fernandez-Llatas, C.; Sun, Y.; Traver, V. Wearable Sensors Integrated with Internet of Things for Advancing eHealth Care. Sensors 2018, 18, 1851. [Google Scholar] [CrossRef]
- Rákay, R.; Višňovský, M.; Galajdová, A.; Šimšík, D. Testing Properties of E-health System Based on Arduino. J. Autom. Control. 2015, 3, 122–126. [Google Scholar]
- Mezidi, M.; Guérin, C. Effects of patient positioning on respiratory mechanics in mechanically ventilated ICU patients. Ann. Transl. Med. 2018, 6, 384. [Google Scholar] [CrossRef]
- Turnbull, H.; Kasereka, M.C.; Amirav, I.; Sahika, S.E.; Solomon, I.; Aldar, Y.; Hawkes, M.T. Development of a novel device for objective respiratory rate measurement in low-resource settings. BMJ Innov. 2018, 4, 185–191. [Google Scholar] [CrossRef]
- Jarchi, D.; Salvi, D.; Tarassenko, L.; Clifton, D. Validation of Instantaneous Respiratory Rate Using Reflectance PPG from Different Body Positions. Sensors 2018, 18, 3705. [Google Scholar] [CrossRef] [PubMed]
- Van Loon, K.; Breteler, M.J.M.; van Wolfwinkel, L.; Rheineck Leyssius, A.T.; Kossen, S.; Kalkman, C.J.; van Zaane, B.; Peelen, L.M. Wireless non-invasive continuous respiratory monitoring with FMCW radar: A clinical validation study. J. Clin. Monit. Comput. 2016, 30, 797–805. [Google Scholar] [CrossRef] [PubMed]
- Guechi, Y.; Pichot, A.; Frasca, D.; Rayeh-Pelardy, F.; Lardeur, J.-Y.; Mimoz, O. Assessment of noninvasive acoustic respiration rate monitoring in patients admitted to an Emergency Department for drug or alcoholic poisoning. J. Clin. Monit. Comput. 2015, 29, 721–726. [Google Scholar] [CrossRef] [PubMed]
- Shen, C.-L.; Huang, T.-H.; Hsu, P.-C.; Ko, Y.-C.; Chen, F.-L.; Wang, W.-C.; Kao, T.; Chan, C.-T. Respiratory Rate Estimation by Using ECG, Impedance, and Motion Sensing in Smart Clothing. J. Med. Biol. Eng. 2017, 37, 826–842. [Google Scholar] [CrossRef] [PubMed]
- Moreno, F.; Lyons, H.A. Effect of body posture on lung volumes. J. Appl. Physiol. 2017, 16, 27–29. [Google Scholar] [CrossRef] [PubMed]
- Leipälä, J.A.; Bhat, R.Y.; Rafferty, G.F.; Hannam, S.; Greenough, A. Effect of posture on respiratory function and drive in preterm infants prior to discharge. Pediatr. Pulmonol. 2003, 36, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Pal, A.K. Effect of Recumbent Body Positions on Dynamic Lung Function Parameters in Healthy Young Subjects. J. Clin. Diagn. Res. 2017, 11, CC08. [Google Scholar] [CrossRef]
- Sun, J.; Jiang, J.; Bao, B.; Wang, S.; He, M.; Zhang, X.; Song, Y. Fabrication of Bendable Circuits on a Polydimethylsiloxane (PDMS) Surface by Inkjet Printing Semi-Wrapped Structures. Materials 2016, 9, 253. [Google Scholar] [CrossRef]
- Nag, A.; Afasrimanesh, N.; Feng, S.; Mukhopadhyay, S.C. Strain induced graphite/PDMS sensors for biomedical applications. Sensors Actuators A Phys. 2018, 271, 257–269. [Google Scholar] [CrossRef]
- Amjadi, M.; Kyung, K.-U.; Park, I.; Sitti, M. Stretchable, Skin-Mountable, and Wearable Strain Sensors and Their Potential Applications: A Review. Adv. Funct. Mater. 2016, 26, 1678–1698. [Google Scholar] [CrossRef]
- Al-Rousan, T.; Schwabkey, Z.; Jirmanus, L.; Nelson, B.D. Health needs and priorities of syrian refugees in camps and urban settings in jordan: Perspectives of refugees and health care providers | Besoins et priorités sanitaires des réfugiés syriens dans les camps et en milieu urbain en jordanie: Perspectives de. East. Mediterr. Health J. 2018, 24, 243–253. [Google Scholar] [CrossRef] [PubMed]
- Kelly, C. Respiratory rate 1: Why measurement and recording are crucial. Nurs. Times 2018, 114, 23–24. [Google Scholar]
- Kwak, Y.H.; Kim, J.; Kim, K. Sleep monitoring sensor using flexible metal strain gauge. Jpn. J. Appl. Phys. 2018, 57, 05GD03. [Google Scholar] [CrossRef]
- Mathew, J.; Semenova, Y.; Farrell, G. A miniature optical breathing sensor. Biomed. Opt. Express 2012, 3, 3325. [Google Scholar] [CrossRef] [PubMed]
- Hamdani, S.; Fernando, A. The Application of a Piezo-Resistive Cardiorespiratory Sensor System in an Automobile Safety Belt. Sensors 2015, 15, 7742–7753. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Halhouli, A.; Al-Ghussain, L.; El Bouri, S.; Habash, F.; Liu, H.; Zheng, D. Clinical Evaluation of Stretchable and Wearable Inkjet-Printed Strain Gauge Sensor for Respiratory Rate Monitoring at Different Body Postures. Appl. Sci. 2020, 10, 480. https://doi.org/10.3390/app10020480
Al-Halhouli A, Al-Ghussain L, El Bouri S, Habash F, Liu H, Zheng D. Clinical Evaluation of Stretchable and Wearable Inkjet-Printed Strain Gauge Sensor for Respiratory Rate Monitoring at Different Body Postures. Applied Sciences. 2020; 10(2):480. https://doi.org/10.3390/app10020480
Chicago/Turabian StyleAl-Halhouli, Ala’aldeen, Loiy Al-Ghussain, Saleem El Bouri, Fuad Habash, Haipeng Liu, and Dingchang Zheng. 2020. "Clinical Evaluation of Stretchable and Wearable Inkjet-Printed Strain Gauge Sensor for Respiratory Rate Monitoring at Different Body Postures" Applied Sciences 10, no. 2: 480. https://doi.org/10.3390/app10020480
APA StyleAl-Halhouli, A., Al-Ghussain, L., El Bouri, S., Habash, F., Liu, H., & Zheng, D. (2020). Clinical Evaluation of Stretchable and Wearable Inkjet-Printed Strain Gauge Sensor for Respiratory Rate Monitoring at Different Body Postures. Applied Sciences, 10(2), 480. https://doi.org/10.3390/app10020480







