Effect of Fly Ash as Cement Replacement on Chloride Diffusion, Chloride Binding Capacity, and Micro-Properties of Concrete in a Water Soaking Environment
Abstract
Featured Application
Abstract
1. Introduction
2. Experimental Investigation
2.1. Materials
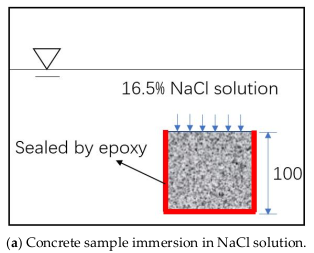
2.2. Immersion Test Methodology
2.3. Measurement of Chloride Content
2.4. Microstructural Characterization
3. Results and Discussion
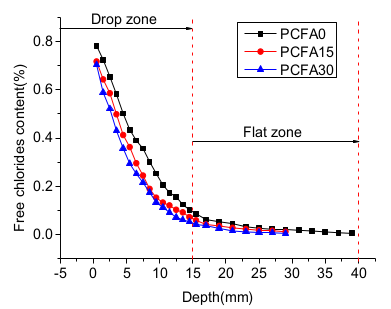
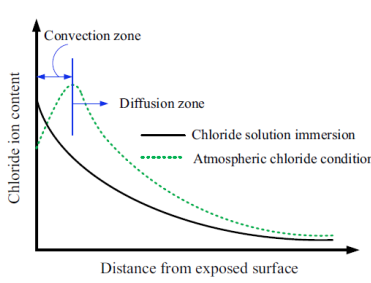
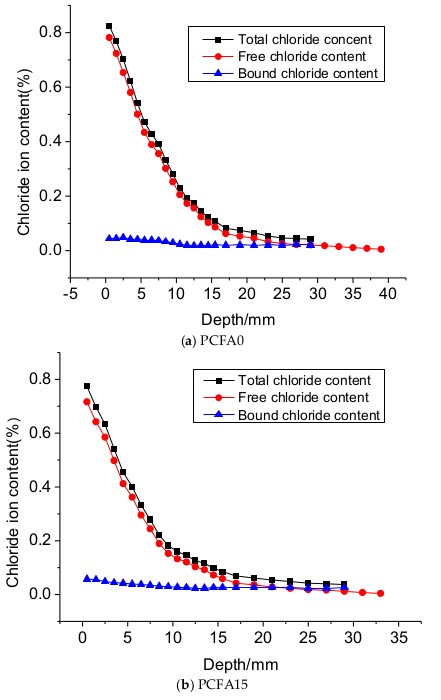
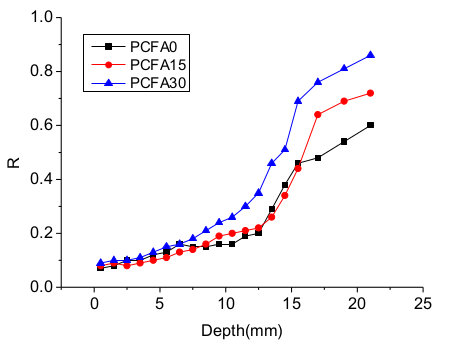
3.1. Effect of Fly Ash on Chloride Transportation
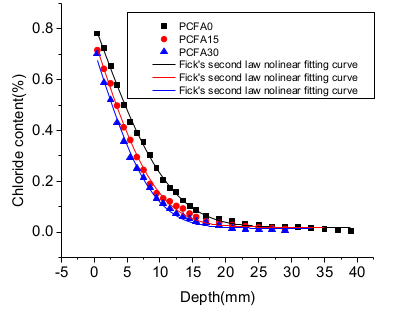
3.2. Influence of Diffusion Coefficient
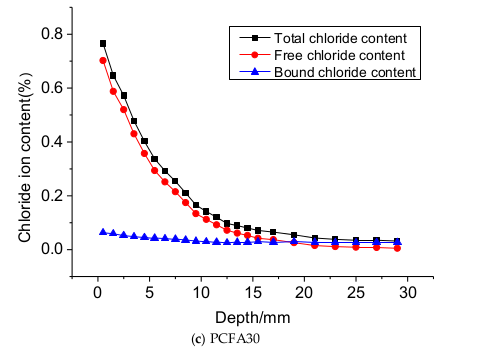
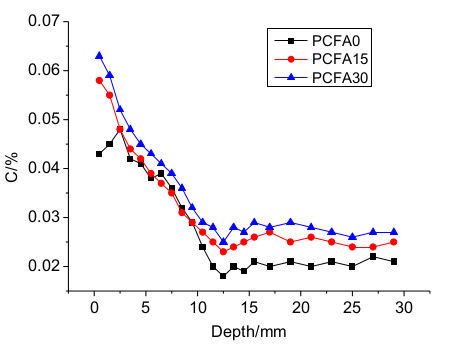
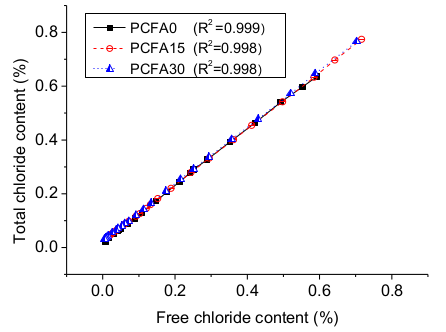
3.3. Influence of Free and Binding Chlorides
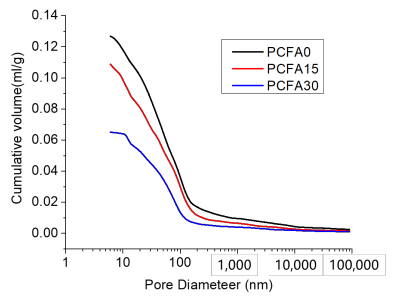
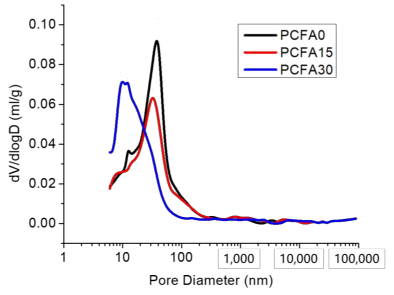
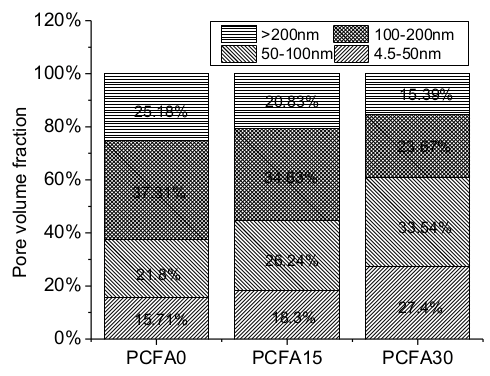
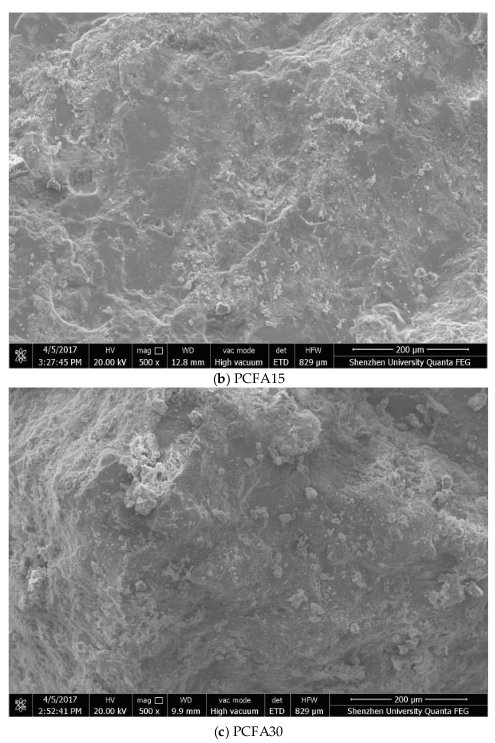
3.4. Effect of Fly Ash on Porosity and Microstructure
4. Conclusions
- (1)
- The addition of fly ash reduced free chloride diffusion, increased the chloride binding capacity, and improved the concrete compactness, which could further improve the durability of concrete.
- (2)
- Two individual zones of chloride deposition, namely the descending zone (0–15 mm) and the gentle zone (15–40 mm), were identified through the measurement of chloride content. The chloride concentration of samples at the 0–7.5 mm zone reduced by 54.5%, 65.9%, and 69.4%, respectively, for PCFA0, PCFA15 and PCFA30. Fly ash addition indeed decreased the free chloride concentration.
- (3)
- In natural immersion, the chloride ion diffusion coefficients of concrete with 15% and 30% fly ash replacements for cement were reduced by 26.1% and 37.9%, respectively, compared to concrete without any fly ash addition, which indicated that the more fly ash was added in the concrete, the lower the chloride diffusion coefficient.
- (4)
- Both chemical ion exchange and physical adsorption processes contributed to the reduction of free chloride ion concentrations. The free chloride ion content had a linear relation with the total amount of chloride ions. An equation was proposed to predict the binding capacity of chloride. Concretes with 0%, 15%, and 30% replacement for cement had binding capacity indexes of 0.1426, 0.1882, and 0.2134, respectively, which indicated that the chloride binding capacity was improved by fly ash. In this case, fly ash reduced the free chloride ion concentration, and if this concentration was less than the critical value, the rebar corrosion could be prevented, thus resulting in a longer service life of reinforced concrete. Moreover, a future study is expected to differentiate between chemical and physical adsorption process.
- (5)
- Fly ash particles and hydration products (filling and pozzolanic effect) led to the densification of microstructures. SEM results showed that the volume of pores less than 100 nm obviously increased for concrete with 15% fly ash replacement for cement, the volume of pores sized from 4.5 to 50 nm, and 50 to 100 nm pronouncedly increased, while the volume of pores larger than 100 nm decreased significantly.
Author Contributions
Funding
Conflicts of Interest
References
- Hasanbeigi, A.; Menke, C.; Price, L. The CO2 abatement cost curve for the Thailand cement industry. J. Clean. Prod. 2010, 18, 1509–1518. [Google Scholar] [CrossRef]
- Chen, C.; Habert, G.; Bouzidi, Y.; Jullien, A. Environmental impact of cement production: Detail of the different processes and cement plant variability evaluation. J. Clean. Prod. 2010, 18, 478–485. [Google Scholar] [CrossRef]
- Madhavi, T.C.; Swamy Raju, L.; Mathur, D. Durabilty and Strength Properties of High Volume Fly Ash Concrete. J. Civil. Eng. Res. 2014, 4, 7–11. [Google Scholar]
- Abdullah, M.M.A.; Tahir, M.F.M.; Hussin, K.; Binhussain, M.; Sandu, I.G.; Yahya, Z.; Sandu, A.V. Fly ash based lightweight geopolymer concrete using foaming agent technology. Rev. Chim. 2015, 66, 1001–1003. [Google Scholar]
- Mishra, S.R.; Kumar, S.; Park, A.; Rho, J.; Losby, J.; Hoffmeister, B.K. Ultrasonic characterization of the curing process of PCC fly ash–cement composites. Mater. Charact. 2003, 50, 317–323. [Google Scholar] [CrossRef]
- Hemalatha, T.; Ramaswamy, A. A review on fly ash characteristics-Towards promoting high volume utilization in developing sustainable concrete. J. Clean. Prod. 2017, 147, 546–559. [Google Scholar] [CrossRef]
- Mukherjee, A.B.; Zevenhoven, R.; Bhattacharya, P.; Sajwan, K.S.; Kikuchi, R. Mercury flow via coal and coal utilization by-products: A global perspective. Resour. Conserv. Recycl. 2008, 52, 571–591. [Google Scholar] [CrossRef]
- Selic, E.; Herbell, J.D. Utilization of fly ash from coal-fired power plants in China. J. Zhejiang Univ.-Sci. A 2008, 9, 681–687. [Google Scholar]
- Liu, J.; Gu, Z.; Xiang, J.; Pan, D. Permeation properties and pore structure of surface layer of fly ash concrete. Materials 2014, 7, 4282–4296. [Google Scholar] [CrossRef]
- Liu, J.; Xing, F.; Dong, B.; Ma, H.; Pan, D. Study on water sorptivity of the surface layer of concrete. Mater. Struct. 2014, 47, 1941–1951. [Google Scholar] [CrossRef]
- Argiz, C.; Moragues, A.; Menéndez, E. Use of ground coal bottom ash as cement constituent in concretes exposed to chloride environments. J. Clean. Prod. 2017, 170, 25–33. [Google Scholar] [CrossRef]
- Zou, D.J.; Liu, T.J.; Qiao, G.F. Experimental investigation on the dynamic properties of RC structures affected by the reinforcement corrosion. Adv. Struct. Eng. 2014, 17, 851–860. [Google Scholar] [CrossRef]
- Mehta, P.K. High-performance, high-volume fly ash concrete for sustainable development. In Proceedings of the International Workshop on Sustainable Development and Concrete Technology, Beijing, China, 20–21 May 2004. [Google Scholar]
- Moghaddam, F.; Sirivivatnanon, V.; Vessalas, K. The effect of fly ash fineness on heat of hydration, microstructure, flow and compressive strength of blended cement pastes. Case Stud. Constr. Mater. 2019, 10, e00218. [Google Scholar] [CrossRef]
- Liu, J.; Qiu, Q.; Chen, X.; Wang, X.; Xing, F.; Han, N.; He, Y. Degradation of fly ash concrete under the coupled effect of carbonation and chloride aerosol ingress. Corros. Sci. 2016, 112, 364–372. [Google Scholar] [CrossRef]
- Thomas, M.D.A.; Matthews, J.D. Carbonation of fly ash concrete. Mag. Concr. Res. 1992, 44, 217–228. [Google Scholar] [CrossRef]
- Liu, T.; Zou, D.; Teng, J.; Yan, G. The influence of sulfate attack on the dynamic properties of concrete column. Constr. Build. Mater. 2012, 28, 201–207. [Google Scholar] [CrossRef]
- Naik, T.R.; Singh, S.; Ramme, B. Mechanical properties and durability of concrete made with blended fly ash. ACI Mater. J. 1998, 95, 454–462. [Google Scholar]
- Malhotra, V.M. Role of supplementary cementing materials in reducing greenhouse gas emissions CANMET. In Concrete Technology for a Sustainable Development in the 21st Century; Gjorv, O.E., Sakai, K., Eds.; Spon Press: London, UK, 1999. [Google Scholar]
- Liu, J.; Qiu, Q.; Chen, X.; Xing, F.; Han, N.; Ma, Y. Understanding the interacted mechanism between carbonation and chloride aerosol attack in ordinary Portland cement concrete. Cem. Concr. Res. 2017, 95, 217–225. [Google Scholar] [CrossRef]
- Liu, J.; Wang, X.; Qiu, Q.; Ou, G.; Xing, F. Understanding the effect of curing age on the chloride resistance of fly ash blended concrete by rapid chloride migration test. Mater. Chem. Phys. 2017, 196, 315–323. [Google Scholar] [CrossRef]
- Tripathi, S.R.; Ogura, H.; Inoue, H.; Hasegawa, T.; Takeya, K.; Kawase, K. Measurement of chloride ion concentration in concrete structures using terahertz time domain spectroscopy (THz-TDS). Corros. Sci. 2012, 62, 5–10. [Google Scholar] [CrossRef]
- Al-Mattarneh, H. Determination of chloride content in concrete using near- and far-field microwave non-destructive methods. Corros. Sci. 2016, 105, 133–140. [Google Scholar] [CrossRef]
- Liu, J.; Ou, G.; Qiu, Q.; Chen, X.; Hong, J.; Xing, F. Chloride transport and microstructure of concrete with/without fly ash under atmospheric chloride condition. Constr. Build. Mater. 2017, 146, 493–501. [Google Scholar] [CrossRef]
- Angst, U.; Elsener, B.; Larsen, C.K.; Vennesland, Ø. Critical chloride content in reinforced concrete: A review. Cem. Concr. Res. 2009, 39, 1122–1138. [Google Scholar] [CrossRef]
- Sun, G.; Zhang, Y.; Sun, W.; Liu, Z.; Wang, C. Multi-scale prediction of the effective chloride diffusion coefficient of concrete. Constr. Build. Mater. 2011, 25, 3820–3831. [Google Scholar] [CrossRef]
- Wu, J.; Li, H.; Wang, Z.; Liu, J. Transport model of chloride ions in concrete under loads and drying-wetting cycles. Constr. Build. Mater. 2016, 112, 733–738. [Google Scholar] [CrossRef]
- Mehta, P.K.; Monteriropj, M. Concrete: Microstructure, Properties and Materials; McGraw-Hill: New York, NY, USA, 2006; pp. 203–300. [Google Scholar]
- Muigai, R.; Alexander, M. Durability design of reinforced concrete structures: A comparison of the use of durability indexes in the deemed-to-satisfy approach and the full-probabilistic approach. Mater. Struct. 2012, 45, 1233–1244. [Google Scholar] [CrossRef]
- Kwon, S.J.; Na, U.J.; Park, S.S.; Jung, S.H. Service life prediction of concrete wharves with early-aged crack: Probabilistic approach for chloride diffusion. Struct. Saf. 2009, 31, 75–83. [Google Scholar] [CrossRef]
- Huang, Z.; Padmaja, K.; Li, S.; Liew, J.R. Mechanical properties and microstructure of ultra-lightweight cement composites with fly ash cenospheres after exposure to high temperatures. Constr. Build. Mater. 2018, 164, 760–774. [Google Scholar] [CrossRef]
- Huang, Z.Y.; Liew, J.Y.R.; Li, W. Evaluation of compressive behavior of ultra-lightweight cement composite after elevated temperature exposure. Constr. Build. Mater. 2017, 148, 579–589. [Google Scholar] [CrossRef]
- Berry, E.E.; Hemmings, R.T.; Cornelius, B.J. Mechanisms of hydration reactions in high volume fly ash pastes and mortars. Cem. Concr. Compos. 1990, 12, 253–261. [Google Scholar] [CrossRef]
- Kouloumbi, N.; Batis, G.; Malami, C. The anticorrosive effect of fly ash, slag and a Greek pozzolan in reinforced concrete. Cem. Concr. Compos. 1994, 16, 253–260. [Google Scholar] [CrossRef]
- Kouloumbi, N.; Batis, G. Chloride corrosion of steel rebars in mortars with fly ash admixtures. Cem. Concr. Compos. 1992, 14, 199–207. [Google Scholar] [CrossRef]
- Cheewaket, T.; Jaturapitakkul, C.; Chalee, W. Long term performance of chloride binding capacity in fly ash concrete in a marine environment. Constr. Build. Mater. 2010, 24, 1352–1357. [Google Scholar] [CrossRef]
- NT (NORDTEST) Build 443. Concrete Hardened: Accelerated Chloride Penetration, Nordtest, Tekniikantie 12, FIN-02150 Espoo, Finland. 1995. Available online: http://www.nordtest.info/index.php/methods/building/item/concrete-hardened-accelerated-chloride-penetration-nt-build-443.html (accessed on 8 September 2020).
- AASHO T260. Standard Method of Test for Sampling and Testing for Chloride Ion in Concrete and Concrete Raw Materials; American Association of State Highway and Transportation Officials: Washington, DC, USA, 2009. [Google Scholar]
- Ma, H.Y.; Li, Z.J. Realistic Pore Structure of Portland Cement Paste: Experimental Study and Numerical Simulation. Comput. Concr. 2013, 11, 317–336. [Google Scholar] [CrossRef]
- Huang, Z.Y.; Huang, Y.S.; Han, N.X.; Zhou, Y.W.; Xing, F.; Sui, T.B.; Wang, B.; Ma, H.Y. Development of limestone calcined clay cement (LC3) concrete in South China and its bond behavior with steel bar. J. Zhejiang Univ.-Sci. A 2020, 21. [Google Scholar] [CrossRef]
- Washburn, E.W. Note on a Method of Determining the Distribution of Pore Sizes in a Porous Material. Proc. Natl. Acad. Sci. USA 1921, 7, 115. [Google Scholar] [CrossRef] [PubMed]
- Collepardi, M.; Marcialis, A.; Turriziani, R.C. Penetration of chloride ions into cement paste and concrete. Am. Ceram. Soc. 1972, 55, 534–535. [Google Scholar] [CrossRef]
- Funahashi, M. Predicting corrosion free service life of a concrete structure in a chloride environment. ACI Mater. J. 1998, 87, 581–587. [Google Scholar]
- Wang, C.; Yang, C.; Qian, J.; Zhong, M.; Zhao, S. Behavior and Mechanism of Pozzolanic Reaction Heat of Fly Ash and Ground Granulated Blast Furnace Slag at Early Age. J. Chin. Ceram. Soc. 2012, 40, 1050–1058. [Google Scholar]
- Zibara, H.; Hooton, R.D.; Thomas, M.D.A.; Stanish, K. Influence of the C/S and C/A ratios of hydration products on the chloride ion binding capacity of lime-SF and lime-MK mixtures. Cem. Concr. Res. 2008, 38, 422–426. [Google Scholar] [CrossRef]
- Yuan, Q.; Shi, C.; De Schutter, G.; Audenaert, K.; Deng, D. Chloride binding of cement-based materials subjected to external chloride environment—A review. Constr. Build. Mater. 2009, 23, 1–13. [Google Scholar] [CrossRef]
- Elakneswaran, Y.; Nawa, T.; Kurumisawa, K. Electrokinetic potential of hydrated cement in relation to adsorption of chlorides. Cem. Concr. Res. 2009, 39, 340–344. [Google Scholar] [CrossRef]
- Ben-Yair, M. The effect of chlorides on concrete in hot and arid regions. Cem. Concr. Res. 1974, 4, 405–416. [Google Scholar] [CrossRef]
- Tang, L.P.; Nilsson, L.O. Resistance of Concrete to Chloride Ingress: Testing and Modelling; Spon Press: London, UK, 2012. [Google Scholar]



| NO | OPC | Sand | CA | FA | W | W/C |
|---|---|---|---|---|---|---|
| PCFA0 | 409.0 | 720.0 | 1079.0 | 0.0 | 192 | 0.47 |
| PCFA15 | 347.7 | 697.3 | 1054.0 | 61.3 | 192 | 0.47 |
| PCFA30 | 286.3 | 689.0 | 1041.0 | 122.7 | 192 | 0.47 |
| Material | CaO | SiO2 | Al2O3 | Fe2O3 | MgO | SO3 | K2O |
|---|---|---|---|---|---|---|---|
| Cement | 64.67 | 18.59 | 4.62 | 4.17 | 2.35 | 3.32 | 0.92 |
| Fly ash | 4.74 | 62.32 | 23.95 | 1.33 | 2.04 | 1.25 | 0.76 |
| ID | D/(×10−12 m2/s) | Cs/% | R2 | σ2 |
|---|---|---|---|---|
| PCFA0 | 12.2617 | 0.8210 | 0.9988 | 0.0006 |
| PCFA15 | 9.0633 | 0.7614 | 0.9970 | 0.0014 |
| PCFA30 | 7.6176 | 0.7187 | 0.9966 | 0.0014 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, J.; Liu, J.; Huang, Z.; Zhu, J.; Liu, W.; Zhang, W. Effect of Fly Ash as Cement Replacement on Chloride Diffusion, Chloride Binding Capacity, and Micro-Properties of Concrete in a Water Soaking Environment. Appl. Sci. 2020, 10, 6271. https://doi.org/10.3390/app10186271
Liu J, Liu J, Huang Z, Zhu J, Liu W, Zhang W. Effect of Fly Ash as Cement Replacement on Chloride Diffusion, Chloride Binding Capacity, and Micro-Properties of Concrete in a Water Soaking Environment. Applied Sciences. 2020; 10(18):6271. https://doi.org/10.3390/app10186271
Chicago/Turabian StyleLiu, Jun, Jiaying Liu, Zhenyu Huang, Jihua Zhu, Wei Liu, and Wei Zhang. 2020. "Effect of Fly Ash as Cement Replacement on Chloride Diffusion, Chloride Binding Capacity, and Micro-Properties of Concrete in a Water Soaking Environment" Applied Sciences 10, no. 18: 6271. https://doi.org/10.3390/app10186271
APA StyleLiu, J., Liu, J., Huang, Z., Zhu, J., Liu, W., & Zhang, W. (2020). Effect of Fly Ash as Cement Replacement on Chloride Diffusion, Chloride Binding Capacity, and Micro-Properties of Concrete in a Water Soaking Environment. Applied Sciences, 10(18), 6271. https://doi.org/10.3390/app10186271






