Properties of Foamed Lightweight High-Performance Phosphogypsum-Based Ternary System Binder
Abstract
Featured Application
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Properties of High-Performance Ternary System Binder and Mortar
3.1.1. Appearance of Ternary System Binder and Mortar
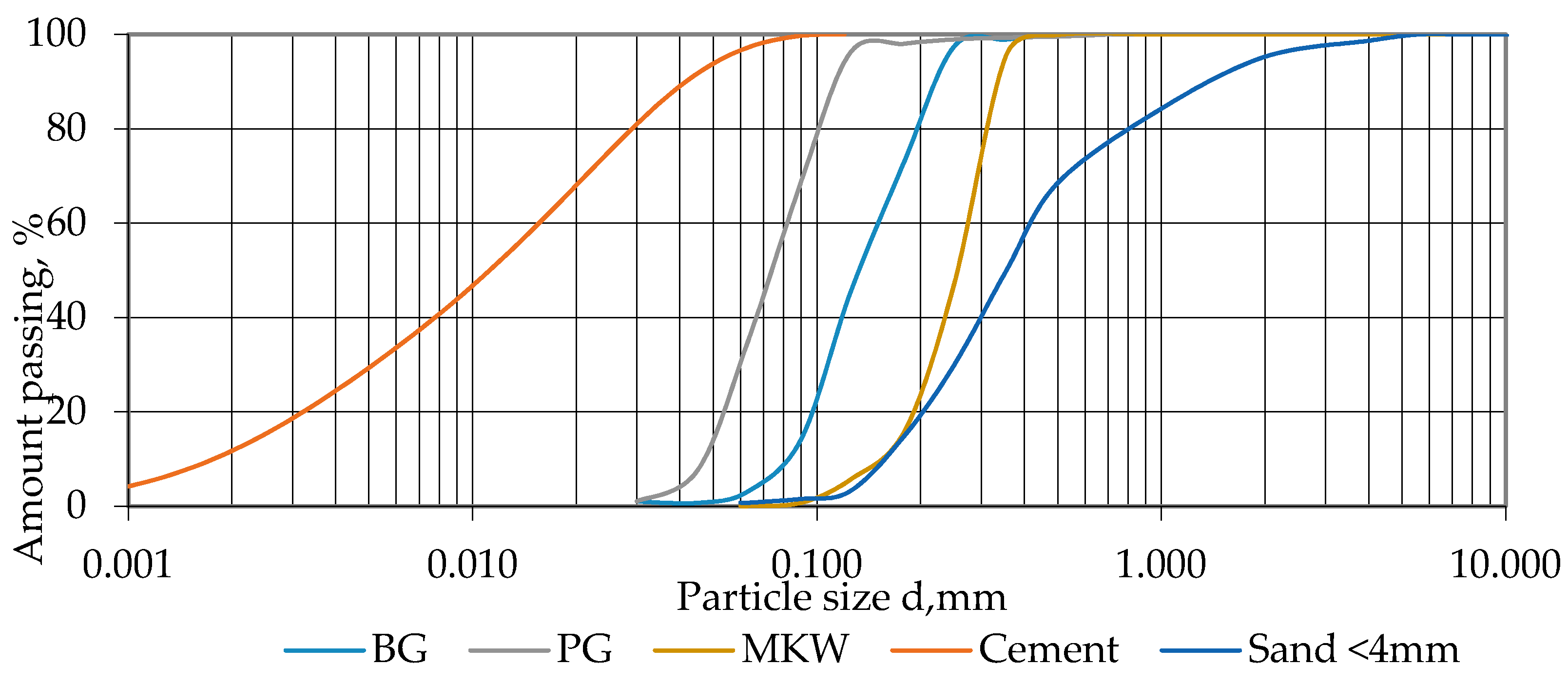
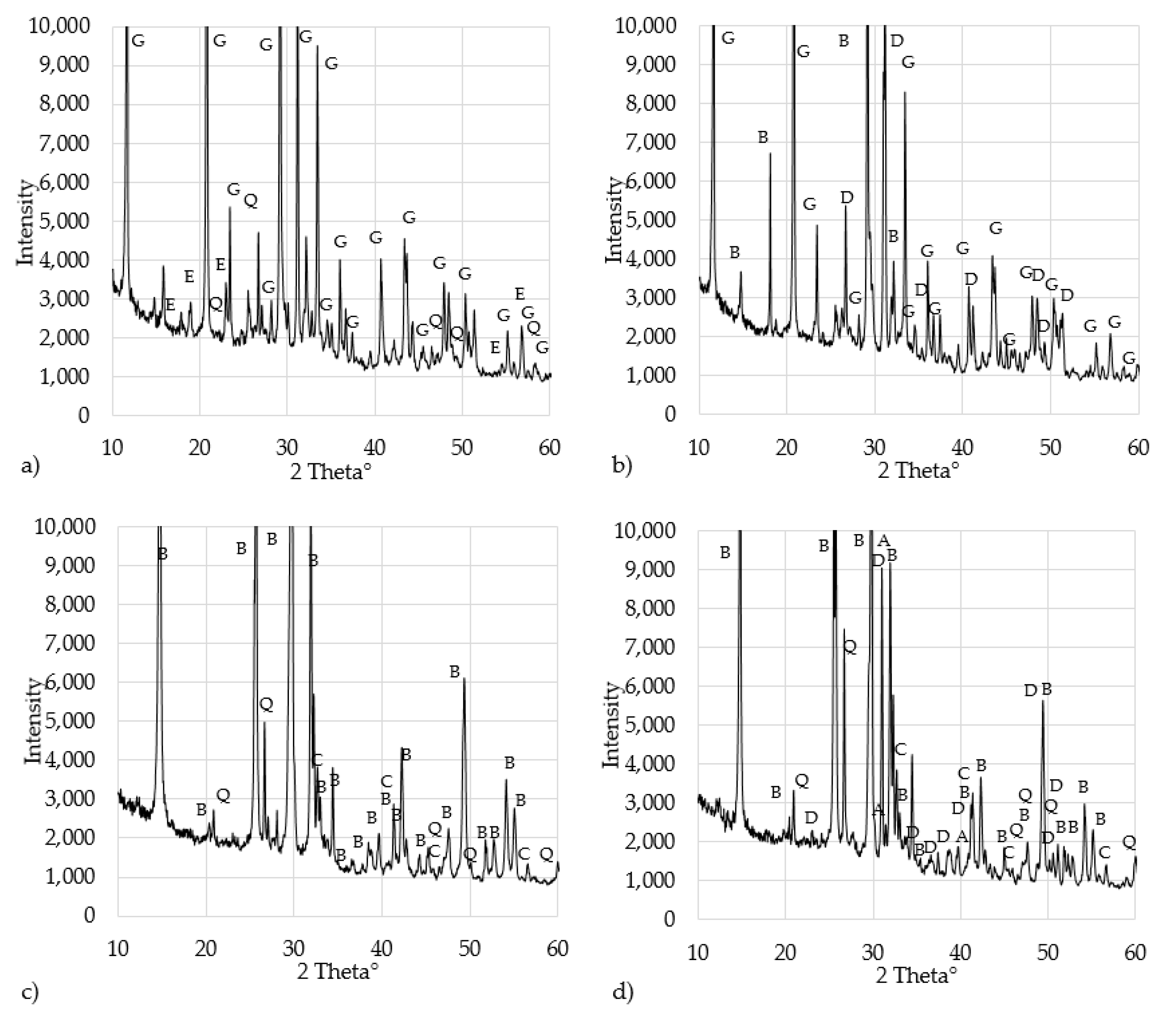
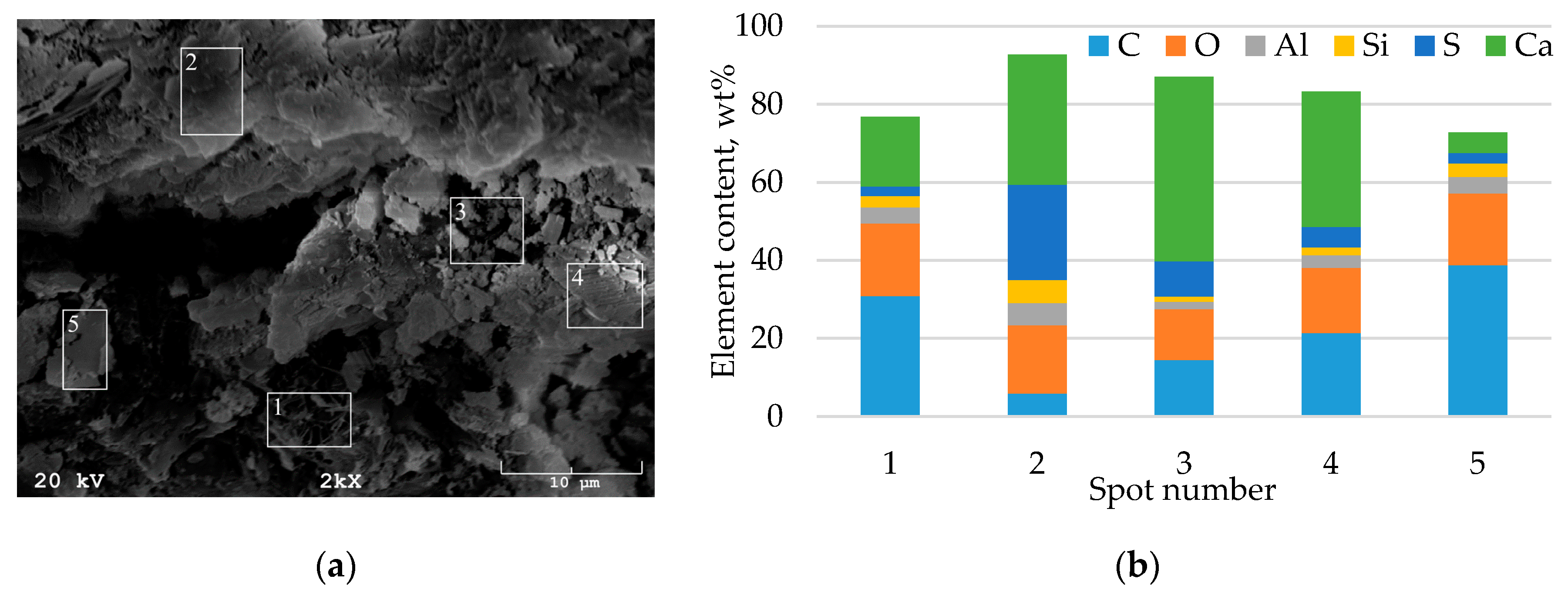
3.1.2. Chemical and Mineralogical Characterization
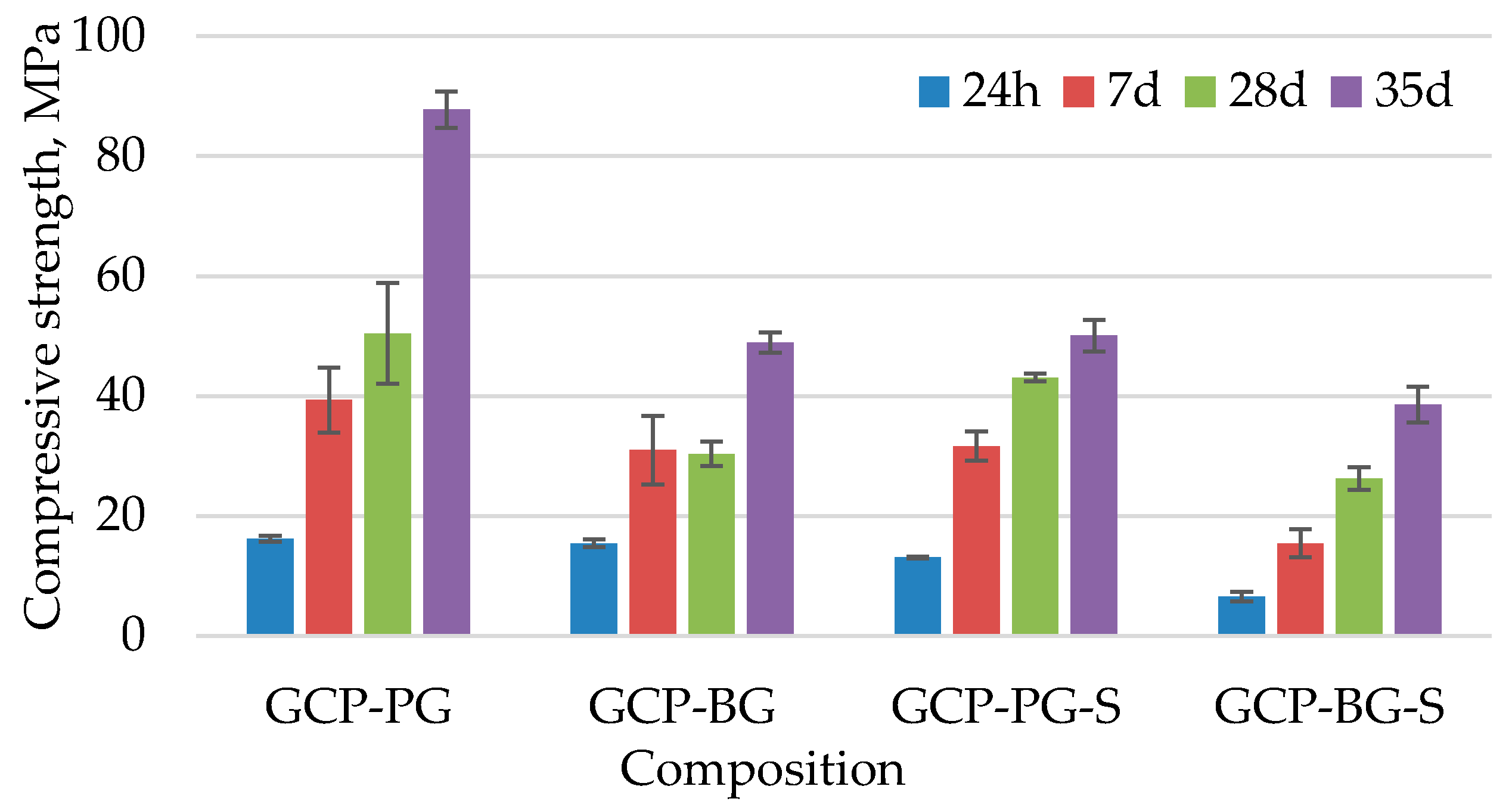
3.1.3. Physical and Mechanical Properties
3.2. Properties of Foamed Ternary System Binder
3.2.1. Appearance of Foamed Ternary System Binder
3.2.2. Physical and Mechanical Properties
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bicer, A.; Kar, F. Thermal and mechanical properties of gypsum plaster mixed with expanded polystyrene and tragacanth. J. Therm. Sci. Eng. Prog. 2017, 1, 59–65. [Google Scholar] [CrossRef]
- Lushnikova, N.; Dvorkin, L. Sustainability of gypsum products as a construction material. J. Sustain. Constr. Mater. 2016, 643–681. [Google Scholar] [CrossRef]
- British Standards Institution. BS EN 12859:2011, Gypsum Blocks—Definitions, Requirements and Test Methods; BSI: London, UK, 30 April 2011; p. 38. ISBN 9780580710360. [Google Scholar]
- Dragomir, A.M.; Lisnic, R.; Prisecaru, T.; Prisecaru, M.M.; Vîjan, C.A.; Nastac, D.C. Study on synthetic gypsum obtained from wet flue gas desulphurisation in thermal power plants. J. Rev. Rom. Mater. Rom. Mater. 2017, 47, 551–556. [Google Scholar]
- Tayibi, H.; Choura, M.; López, F.A.; Alguacil, F.J.; López-Delgado, A. Environmental impact and management of phosphogypsum. J. Environ. Manag. 2009, 90, 2377–2386. [Google Scholar] [CrossRef] [PubMed]
- Bumanis, G.; Zorica, J.; Bajare, D.; Korjakins, A. Technological properties of phosphogypsum binder obtained from fertilizer production waste. J. Energy Procedia 2018, 147, 301–308. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, A.; Li, K.; Wang, Q.; Han, Y.; Yao, B.; Gao, X.; Feng, L. Research on the pretreatment and mechanical performance of undisturbed phosphogypsum. J. Case Stud. Constr. Mater. 2020, 13, e00400. [Google Scholar] [CrossRef]
- Kumar, S. Fly ash-lime-phosphogympsum hollow blocks for walls and partitions. J. Build. Env. 2003, 38, 291–295. [Google Scholar] [CrossRef]
- Morsy, M.S.; Rashad, A.M.; Shoukry, H.; Mokhtar, M.M.; El-Khodary, S.A. Development of lime-pozzolan green binder: The influence of anhydrous gypsum and high ambient temperature curing. J. Build. Eng. 2020, 28, 101026. [Google Scholar] [CrossRef]
- Huang, Y.; Lin, Z.S. Investigation on phosphogypsum-steel slag-granulated blast-furnace slag-limestone cement. J. Constr. Build. Mater. 2010, 24, 1296–1301. [Google Scholar] [CrossRef]
- Zhang, G.; Li, G.; Li, Y. Effects of superplasticizers and retarders on the fluidity and strength of sulphoaluminate cement. J. Constr. Build. Mater. 2016, 126, 44–54. [Google Scholar] [CrossRef]
- Akhlaghi, O.; Aytas, T.; Tatli, B.; Sezer, D.; Hodaei, A.; Favier, A.; Scrivener, K.; Menceloglu, Y.Z.; Akbulut, O. Modified poly(carboxylate ether)-based superplasticizer for enhanced flowability of calcined clay-limestone-gypsum blended Portland cement. J. Cem. Concr. Res. 2017, 101, 114–122. [Google Scholar] [CrossRef]
- Namsone, E.; Šahmenko, G.; Korjakins, A. Durability Properties of High Performance Foamed Concrete. J. Procedia Eng. 2017, 172, 760–767. [Google Scholar] [CrossRef]
- Skujans, J.; Iljins, U.; Ziemelis, I.; Gross, U.; Ositis, N.; Brencis, R.; Veinbergs, A.; Kukuts, O. Experimental research of foam gypsum acoustic absorption and heat flow. J. Chem. Eng. Trans. 2010, 19, 79–84. [Google Scholar] [CrossRef]
- Ma, B.; Jin, Z.; Su, Y.; Lu, W.; Qi, H.; Hu, P. Utilization of hemihydrate phosphogypsum for the preparation of porous sound absorbing material. J Constr. Build. Mater. 2020, 234, 117346. [Google Scholar] [CrossRef]
- Nizevičienė, D.; Vaičiukynienė, D.; Michalik, B.; Bonczyk, M.; Vaitkevičius, V.; Jusas, V. The treatment of phosphogypsum with zeolite to use it in binding material. J Constr. Build. Mater. 2018, 180, 134–142. [Google Scholar] [CrossRef]
- Bumanis, G.; Vitola, L.; Stipniece, L.; Locs, J.; Korjakins, A.; Bajare, D. Evaluation of Industrial by-products as pozzolans: A road map for use in concrete production. J Case Stud. Constr. Mater. 2020, 13, e00424. [Google Scholar] [CrossRef]
- Nguyen, V.H.; Leklou, N.; Aubert, J.E.; Mounanga, P. The effect of natural pozzolan on delayed ettringite formation of the heat-cured mortars. J Constr. Build. Mater. 2013, 48, 479–484. [Google Scholar] [CrossRef]
- Bajare, D.; Bumanis, G.; Shakhmenko, G.; Justs, J. High performance and conventional concrete properties affected by ashes obtained from different type of grasses. J Am. Concr. Inst. 2012, 289, 317–330. [Google Scholar]
- Žvironaite, J.; Kligys, M.; Pundiene, I.; Pranckevičiene, J. Effect of limestone particles on rheological properties and hardening process of plasticized cement pastes. J Medzg. 2015, 21, 143–148. [Google Scholar] [CrossRef]
- Zhao, W.; Su, Q.; Wang, W.; Niu, L.; Liu, T. Experimental study on the effect of water on the properties of cast in situ foamed concrete. J Adv. Mater. Sci. Eng. 2018. [Google Scholar] [CrossRef]




| Element | Commercial Gypsum BG | Phosphogypsum PG | Metakaolin MKW | Cement CEM I 42.5N |
|---|---|---|---|---|
| SiO2 | 3.73 | 1.07 | 51.80 | 22.64 |
| Al2O3 | 1.68 | 0.70 | 34.20 | 5.93 |
| Fe2O3 | 0.46 | 0.22 | 0.50 | 3.26 |
| CaO | 35.64 | 37.16 | 0.10 | 57.04 |
| MgO | 3.92 | 0.21 | 0.10 | 4.26 |
| SO3 | 30.90 | 37.38 | - | 3.30 |
| Na2O | 0.31 | 0.48 | 0.60 | 0.10 |
| K2O | - | - | - | 2.40 |
| TiO2 | 0.05 | 0.11 | 0.60 | 0.38 |
| Cl | - | - | - | 0.14 |
| P2O5 | - | 0.57 | - | 0.46 |
| LOI | 22.43 | 19.24 | 11.5 | - |
| Total | 99.42 | 99.74 | 99.60 | 99.90 |
| Mixture | PG | BG | CEM | MKW | G-2 | R | Sand 0/4 mm | W/B |
|---|---|---|---|---|---|---|---|---|
| GCP-PG | 1 | 0.4 | 0.4 | 1.5% | 0.2% | - | 0.34 | |
| GCP-BG | 1 | 0.4 | 0.4 | 1.5% | - | - | 0.34 | |
| GCP-PG-S | 1 | 0.4 | 0.4 | 1.5% | 0.2 | 1.75 | 0.36 | |
| GCP-BG-S | 1 | 0.4 | 0.4 | 1.5% | - | 1.75 | 0.36 |
| Composition | Components, Weights Parts | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Water, Foams | Water, Binder | PG | BG | PBLUX | CEMII | MKW | R | G-2 | SDS | |
| GCP-PG-1 | 200 | 410 | 750 | - | 5 | 312 | 312 | 2.5 | 21 | 2.5 |
| GCP-PG-2 | 200 | 560 | 1050 | - | 5 | 437 | 437 | 3.5 | 29.5 | 3.5 |
| GCP-BG-1 | 200 | 370 | - | 750 | 5 | 312 | 312 | 2.5 | 21 | 2.5 |
| GCP-BG-1 | 200 | 520 | - | 1050 | 5 | 437 | 437 | 3.5 | 29.5 | 3.5 |
| Mixture | Consistency by Suttard Viscosimeter, mm | Setting Time, min | Dry Density, kg/m3 | Total Porosity, vol.% | |
|---|---|---|---|---|---|
| Initial | Final | ||||
| GCP-PG | 370 | 165 | 280 | 1766 ± 9 | 23 ± 2.2 |
| GCP-PG-S | 230 | 130 | 170 | 2027 ± 14 | 11 ± 1.2 |
| GCP-BG | 295 | 70 | 130 | 1609 ± 24 | 29 ± 2.4 |
| GCP-BG-S | 210 | 75 | 85 | 1886 ± 38 | 17 ± 1.9 |
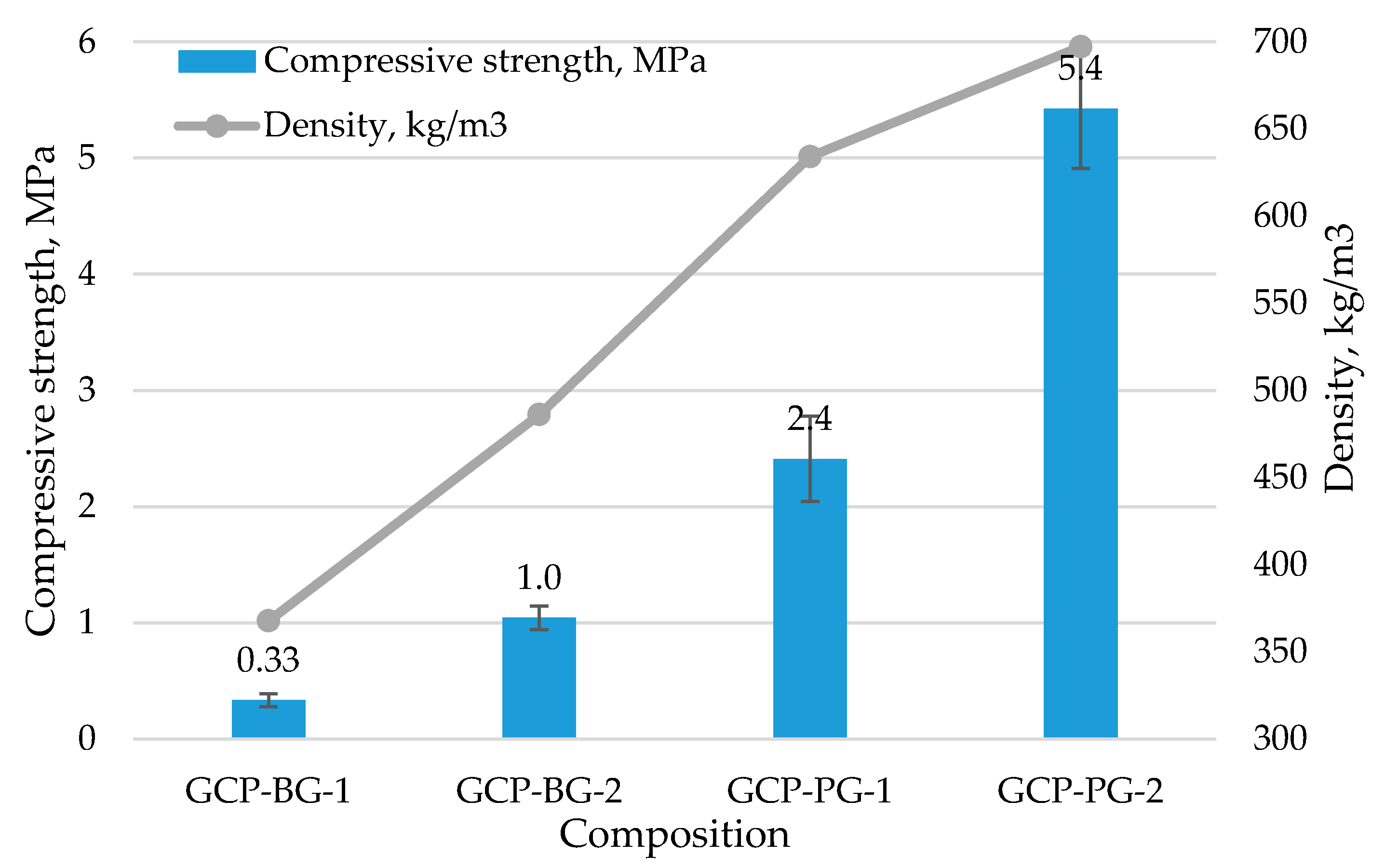
| Composition | ρo, kg/m3 | ρowet, kg/m3 | Water Absorption, wt.% | Open Porosity, vol.% | Closed Porosity, vol.% | Total Porosity, vol.% | Thermal Conductivity, λ, W/m·K |
|---|---|---|---|---|---|---|---|
| GCP-BG-1 | 368 | 758 | 105 | 39 | 45 | 84 | 0.086 |
| GCP-BG-2 | 486 | 901 | 91 | 43 | 36 | 79 | 0.123 |
| GCP-PG-1 | 634 | 876 | 46 | 28 | 45 | 72 | 0.111 |
| GCP-PG-2 | 697 | 901 | 39 | 25 | 44 | 70 | 0.153 |
| Composition | Dry Compression Strength, MPa | Saturated Compression Strength, MPa | Softening Coefficient |
|---|---|---|---|
| GCP-BG-1 | 0.3 ± 0.06 | 0.2 ± 0.02 | 0.56 |
| GCP-BG-2 | 1.0 ± 0.10 | 0.6 ± 0.06 | 0.59 |
| GCP-PG-1 | 2.4 ± 0.37 | 1.2 ± 0.06 | 0.50 |
| GCP-PG-2 | 5.4 ± 0.52 | 3.5 ± 0.17 | 0.64 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bumanis, G.; Zorica, J.; Bajare, D. Properties of Foamed Lightweight High-Performance Phosphogypsum-Based Ternary System Binder. Appl. Sci. 2020, 10, 6222. https://doi.org/10.3390/app10186222
Bumanis G, Zorica J, Bajare D. Properties of Foamed Lightweight High-Performance Phosphogypsum-Based Ternary System Binder. Applied Sciences. 2020; 10(18):6222. https://doi.org/10.3390/app10186222
Chicago/Turabian StyleBumanis, Girts, Jelizaveta Zorica, and Diana Bajare. 2020. "Properties of Foamed Lightweight High-Performance Phosphogypsum-Based Ternary System Binder" Applied Sciences 10, no. 18: 6222. https://doi.org/10.3390/app10186222
APA StyleBumanis, G., Zorica, J., & Bajare, D. (2020). Properties of Foamed Lightweight High-Performance Phosphogypsum-Based Ternary System Binder. Applied Sciences, 10(18), 6222. https://doi.org/10.3390/app10186222







