Molding Wetting by Laser-Induced Nanostructures
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Van Driel, H.M.; Sipe, J.E.; Young, J.F. Laser-Induced Periodic Surface Structure on Solids: A Universal Phenomenon. Phys. Rev. Lett. 1982, 49, 1955–1958. [Google Scholar] [CrossRef]
- Sipe, J.E.; Young, J.F.; Preston, J.S.; van Driel, H.M. Laser-induced periodic surface structure. I. Theory. Phys. Rev. B 1983, 27, 1141–1154. [Google Scholar] [CrossRef]
- Young, J.F.; Sipe, J.E.; van Driel, H.M. Laser-induced periodic surface structure. III. Fluence regimes, the role of feedback, and details of the induced topography in germanium. Phys. Rev. B 1984, 30, 2001–2015. [Google Scholar] [CrossRef]
- Ursu, I.; Mihăilescu, I.N.; Prokhorov, A.M.; Konov, V.I.; Tokarev, V.N. On the role of the periodical structures induced by powerful laser irradiation of metallic surfaces in the energy coupling process. Phys. B + C 1985, 132, 395–402. [Google Scholar] [CrossRef]
- Wang, J.; Guo, C. Ultrafast dynamics of femtosecond laser-induced periodic surface pattern formation on metals. Appl. Phys. Lett. 2005, 87, 251914. [Google Scholar] [CrossRef]
- Bonse, J.; Krüger, J.; Höhm, S.; Rosenfeld, A. Femtosecond laser-induced periodic surface structures. J. Laser Appl. 2012, 24, 042006. [Google Scholar] [CrossRef]
- Sokolowski-Tinten, K.; Bialkowski, J.; Boing, M.; Cavalleri, A.; von der Linde, D. Thermal and nonthermal melting of gallium arsenide after femtosecond laser excitation. Phys. Rev. B 1998, 58, R11805–R11808. [Google Scholar] [CrossRef]
- Bonse, J.; Rosenfeld, A.; Krüger, J. Pulse number dependence of laser-induced periodic surface structures for femtosecond laser irradiation of silicon. J. Appl. Phys. 2009, 106, 104910. [Google Scholar] [CrossRef]
- Vorobyev, A.Y.; Guo, C. Femtosecond laser-induced periodic surface structure formation on tungsten. J. Appl. Phys. 2008, 104, 063523. [Google Scholar] [CrossRef]
- Tsibidis, G.D.; Fotakis, C.; Stratakis, E. From ripples to spikes: A hydrodynamical mechanism to interpret femtosecond laser-induced self-assembled structures. Phys. Rev. B 2015, 92, 041405. [Google Scholar] [CrossRef]
- Kovačević, A.; Petrović, S.; Bokić, B.; Gaković, B.; Bokorov, M.; Vasić, B.; Gajić, R.; Trtica, M.; Jelenković, B. Surface nanopatterning of Al/Ti multilayer thin films and Al single layer by a low-fluence UV femtosecond laser beam. Appl. Surf. Sci. 2015, 326, 91–98. [Google Scholar] [CrossRef]
- Li, X.; Yuan, C.; Yang, H.; Li, J.; Huang, W.; Tan, D.; Hu, Q. Morphology and composition on Al surface irradiated by femtosecond laser pulses. Appl. Surf. Sci. 2010, 256, 4344–4349. [Google Scholar] [CrossRef]
- Vorobyev, A.T.; Guo, C. Direct femtosecond laser surface nano/microstructuring and its applications. Laser Photonics Rev. 2013, 7, 385–407. [Google Scholar] [CrossRef]
- Zorba, V.; Persano, L.; Pisignano, D.; Athanassiou, A.; Stratakis, E.; Cingolani, R.; Tzanetakis, P.; Fotakis, C. Making silicon hydrophobic: Wettability control by two-lengthscale simultaneous patterning with femtosecond laser irradiation. Nanotechnology 2006, 17, 3234–3238. [Google Scholar] [CrossRef]
- Baldacchini, T.; Carey, J.E.; Zhou, M.; Mazur, E. Superhydrophobic Surfaces Prepared by Microstructuring of Silicon Using a Femtosecond Laser. Langmuir 2006, 22, 4917–4919. [Google Scholar] [CrossRef]
- Kietzig, A.-M.; Hatzikiriakos, S.G.; Englezos, P. Patterned superhydrophobic metallic surfaces. Langmuir 2009, 25, 4821–4827. [Google Scholar] [CrossRef]
- Kietzig, A.; Mirvakili, M.N.; Kamal, S.; Englezos, P.; Hatzikiriakos, S.G. Laser-Patterned Super-Hydrophobic Pure Metallic Substrates: Cassie to Wenzel Wetting Transitions. J. Adhesion Sci. Technol. 2011, 25, 2789–2809. [Google Scholar] [CrossRef]
- De Gennes, P.G. Wetting: Statics and dynamics. Rev. Modern Phys. 1985, 57, 827–863. [Google Scholar] [CrossRef]
- Chan, H.B.; Aksyuk, V.A.; Kleiman, R.N.; Bishop, D.J.; Capa, F. Quantum mechanical actuation of microelectromechanical systems by the Casimir force. Science 2001, 291, 1941–1944. [Google Scholar] [CrossRef]
- Alhambra, Á.M.; Kempf, A.; Eduardo Martín-Martínez, E. Casimir forces on atoms in optical cavities. Phys. Rev. A 2014, 89, 033835. [Google Scholar] [CrossRef]
- Dellieu, L.; Deparis, O.; Muller, J.; Kolaric, B.; Sarrazin, M.M. Quantum vacuum photon modes and repulsive Lifshitz–van der Waals interactions. Phys. Rev. B 2015, 92, 235418. [Google Scholar] [CrossRef]
- Dellieu, L.; Deparis, O.; Muller, J.; Sarrazin, M. Quantum vacuum photon modes and superhydrophobicity. Phys. Rev. Lett. 2015, 114, 024501. [Google Scholar] [CrossRef] [PubMed]
- Intravaia, F.; Koev, S.; Jung, I.W.; Talin, A.A.; Davids, P.S.; Decca, R.S.; Aksyuk, V.A.; Dalvit, D.A.R.; López, D. Strong Casimir force reduction through metallic surface nanostructuring. Nat. Commun. 2013, 4, 2515. [Google Scholar] [CrossRef] [PubMed]
- Gwyddion. Available online: www.gwyddion.net (accessed on 29 November 2019).
- Israelachvili, J. Intermolecular and Surface Forces; Academic Press—Elsevier: San Diego, CA, USA, 2011. [Google Scholar]
- Bordag, M.; Klimchitskaya, G.L.; Mohideen, U.; Mostepanenko, V.M. Advances in the Casimir Effect; Oxford University Press: New York, NY, USA, 2009. [Google Scholar]
- Gnilitskyi, I.; Derrien, T.J.-Y.; Levy, Y.; Bulgakova, N.M.; Mocek, T.; Orazi, L. High-speed manufacturing of highly regular femtosecond laser-induced periodic surface structures: Physical origin of regularity. Sci. Rep.-UK 2017, 7, 8485. [Google Scholar] [CrossRef]
- Petrović, S.; Peruško, D.; Skoulas, E.; Kovač, J.; Mitrić, M.; Potočnik, J.; Rakočević, Z.; Stratakis, E. Laser-Assisted Surface Texturing of Ti/Zr Multilayers for Mesenchymal Stem Cell Respose. Coatings 2019, 9, 854. [Google Scholar] [CrossRef]
- Navrotsky, A. Energetics of nanoparticle oxides: Interplay between surface energy and polymorphism. Geochem. T. 2003, 4, 34–37. [Google Scholar] [CrossRef]
- Noginov, M.A.; Barnakov, Y.A.; Liberman, V.; Prayakarao, S.; Bonner, C.E.; Narimanov, E.E. Long-range wetting transparency on top of layered metal-dielectric substrates. Sci. Rep.-UK 2016, 6, 27834. [Google Scholar] [CrossRef]
- Lu, G.; Bernasek, S.L.; Schwartz, J. Oxidation of a polycrystalline titanium surface by oxygen and water. Surf. Sci. 2000, 458, 80–90. [Google Scholar] [CrossRef]
- Rudakova, A.V.; Maevskaya, M.V.; Emeline, A.V.; Bahnemann, D.W. Light-Controlled ZrO2 Surface Hydrophilicity. Sci. Rep. 2016, 6, 34285. [Google Scholar] [CrossRef]
- González-Martín, M.L.; Labajos-Broncano, L.; Jańczuk, B.; Bruque, B.M. Wettability and surface free energy of zirconia ceramics and their constituents. J. Mater. Sci. 1999, 34, 5923–5926. [Google Scholar] [CrossRef]
- Stevens, N.; Priest, C.I.; Sedev, R.; Ralston, J. Wettability of Photoresponsive Titanium Dioxide Surfaces. Langmuir 2003, 19, 3272–3275. [Google Scholar] [CrossRef]
- Petrović, S.; Gaković, B.; Kovač, J.; Panjan, P.; Stratakis, E.; Trtica, M.; Fotakis, C.; Jelenković, B. Synthesis of ultra-thin oxide layer in laser-treated 3x (Al/Fe)/Si multilayer structure. J. Mater. Sci. 2014, 49, 7900–7907. [Google Scholar] [CrossRef]
- Lee, Y.-E.; Lee, D.-K.; Cho, Y.H. Fabrication of anisotropic wetting surface with asymmetric structures using geometrical similarity and capillary force. Micro Nano Syst. Lett. 2019, 7, 16. [Google Scholar] [CrossRef]
- Sarrazin, M.; Septembre, I.; Hendrickx, A.; Reckinger, N.; Dellieu, L.; Fleury, G.; Seassal, C.; Mazurczyk, R.; Faniel, S.; Devouge, S.; et al. Towards an experimental proof of superhydrophobicity enhanced by quantum fluctuations freezing on a broadband-absorber metamaterial. arXiv 2007, arXiv:2007.11909. [Google Scholar]
- Öktem, B.; Pavlov, I.; Ilday, S.; Kalaycıoğlu, H.; Rybak, A.; Yavaş, S.; Erdoğan, M.; Ilday, F.Ö. Nonlinear laser lithography for indefinitely large area nanostructuring with femtosecond pulses. Nat. Photonics 2013, 7, 897. [Google Scholar] [CrossRef]
- Léopoldès, J.; Damman, P. From a two-dimensional chemical pattern to a three-dimensional topology through selective inversion of a liquid–liquid bilayer. Nat. Mater. 2006, 5, 957–961. [Google Scholar] [CrossRef]


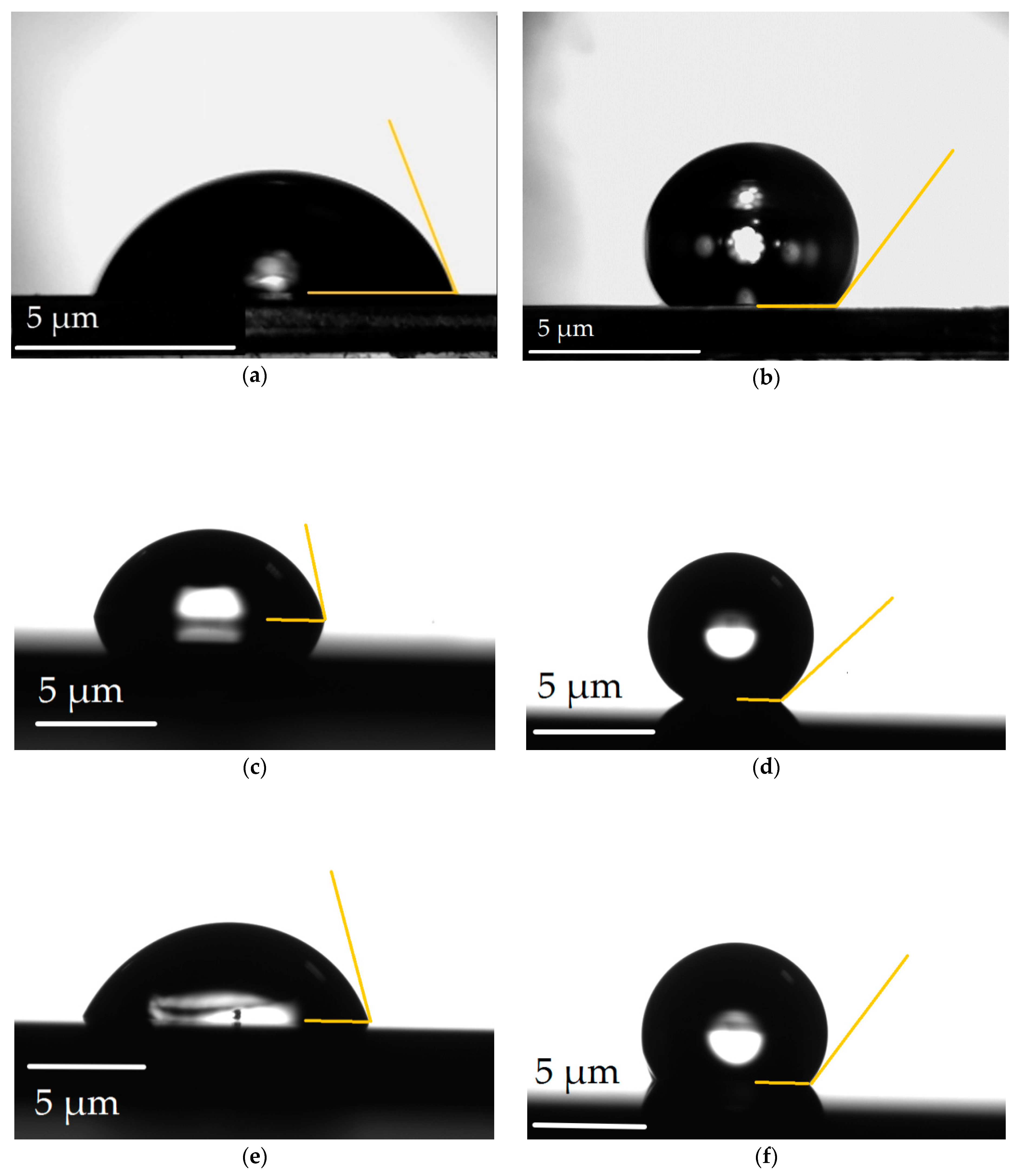
| Material | LIPSS Period (nm) | CA (°) | ∆CA (°) | CA Increase (%) | |
|---|---|---|---|---|---|
| untreated surface | corrugated surface | ||||
| 15×(Ti/Zr) | 740 | 72.11 ± 3.32 | 137.15 ± 11.63 | 65.04 | 90 |
| 15×(Zr/Ti) | 740 | 77.29 ± 1.71 | 144.49 ± 14.97 | 67.20 | 87 |
| 8×(Zr/Cr/Ti) | 790 | 68.10 ± 6.41 | 123.45 ± 25.26 | 55.35 | 81 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kovačević, A.G.; Petrović, S.; Mimidis, A.; Stratakis, E.; Pantelić, D.; Kolaric, B. Molding Wetting by Laser-Induced Nanostructures. Appl. Sci. 2020, 10, 6008. https://doi.org/10.3390/app10176008
Kovačević AG, Petrović S, Mimidis A, Stratakis E, Pantelić D, Kolaric B. Molding Wetting by Laser-Induced Nanostructures. Applied Sciences. 2020; 10(17):6008. https://doi.org/10.3390/app10176008
Chicago/Turabian StyleKovačević, Aleksander G., Suzana Petrović, Alexandros Mimidis, Emmanuel Stratakis, Dejan Pantelić, and Branko Kolaric. 2020. "Molding Wetting by Laser-Induced Nanostructures" Applied Sciences 10, no. 17: 6008. https://doi.org/10.3390/app10176008
APA StyleKovačević, A. G., Petrović, S., Mimidis, A., Stratakis, E., Pantelić, D., & Kolaric, B. (2020). Molding Wetting by Laser-Induced Nanostructures. Applied Sciences, 10(17), 6008. https://doi.org/10.3390/app10176008







